Polyphenols and Human Health: The Role of Bioavailability
Abstract
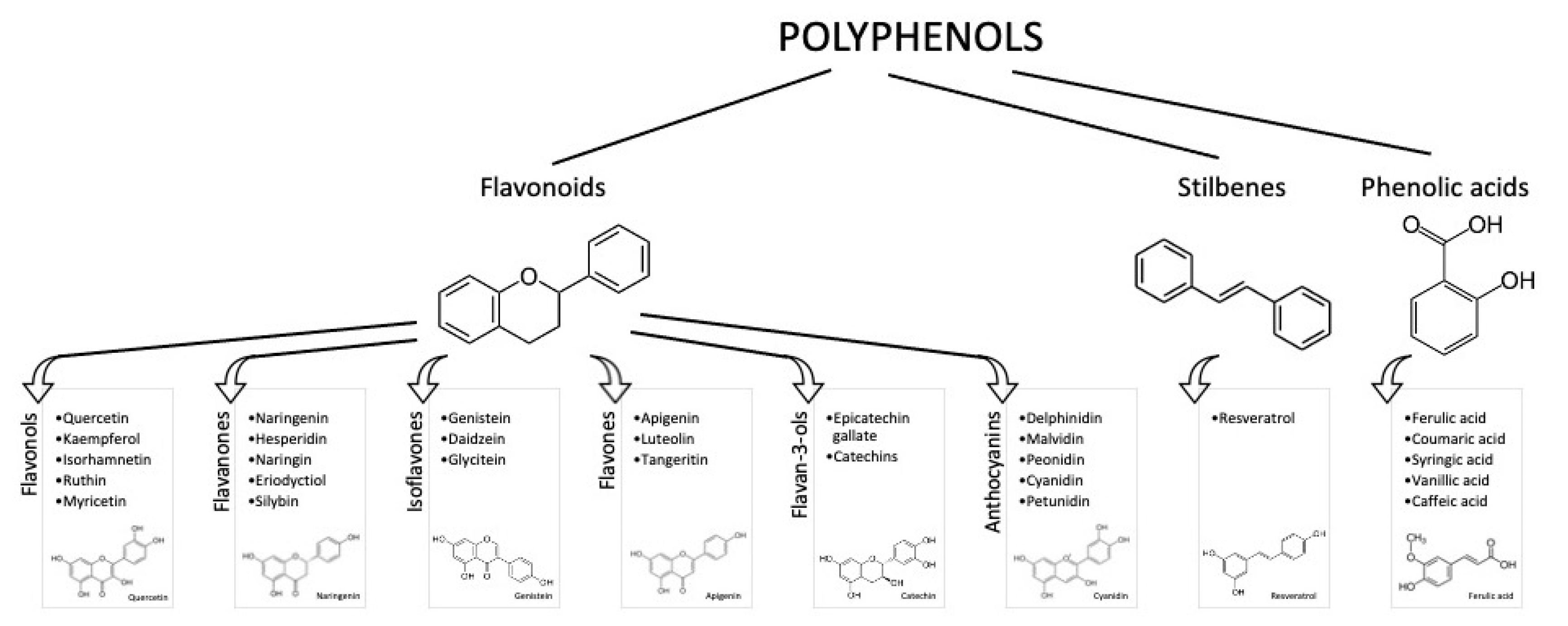
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Anthocyanins
3.2. Flavanols
3.3. Flavonols
3.4. Phenolic Acids
3.5. Stilbenes, Isoflavones, and Flavanones
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tufarelli, V.; Casalino, E.; D’Alessandro, A.G.; Laudadio, V. Dietary Phenolic Compounds: Biochemistry, Metabolism and Significance in Animal and Human Health. Curr. Drug Metab. 2017, 18, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Stockley, C.; Teissedre, P.L.; Boban, M.; Di Lorenzo, C.; Restani, P. Bioavailability of wine-derived phenolic compounds in humans: A review. Food Funct. 2012, 3, 995–1007. [Google Scholar] [CrossRef] [PubMed]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef] [PubMed]
- Teng, H.; Chen, L. Polyphenols and bioavailability: An update. Crit. Rev. Food Sci. Nutr. 2019, 59, 2040–2051. [Google Scholar] [CrossRef]
- Tresserra-Rimbau, A.; Lamuela-Raventos, R.M.; Moreno, J.J. Polyphenols, food and pharma. Current knowledge and directions for future research. Biochem. Pharmacol. 2018, 156, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Ros, E.; Martínez-González, M.A.; Estruch, R.; Salas-Salvadó, J.; Fitó, M.; Martínez, J.A.; Corella, D. Mediterranean diet and cardiovascular health: Teachings of the PREDIMED Study. Adv. Nutr. 2014, 5, 330S–336S. [Google Scholar] [CrossRef]
- Del Bo, C.; Bernardi, S.; Marino, M.; Porrini, M.; Tucci, M.; Guglielmetti, S.; Cherubini, A.; Carrieri, B.; Kirkup, B.; Kroon, P.; et al. Systematic Review on Polyphenol Intake and Health Outcomes: Is there Sufficient Evidence to Define a Health-Promoting Polyphenol-Rich Dietary Pattern? Nutrients 2019, 11, 1355. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Systematic analysis of the content of 502 Polyphenols in 452 foods and beverages: An application of the phenol-explorer database. J. Agric. Food Chem. 2010, 58, 4959–4969. [Google Scholar] [CrossRef]
- Pounis, G.; Tabolacci, C.; Costanzo, S.; Cordella, M.; Bonaccio, M.; Rago, L.; D’Arcangelo, D.; Filippo Di Castelnuovo, A.; de Gaetano, G.; Donati, M.B.; et al. Reduction by coffee consumption of prostate cancer risk: Evidence from the Moli-sani cohort and cellular models. Int. J. Cancer 2017, 141, 72–82. [Google Scholar] [CrossRef]
- Miranda, A.M.; Steluti, J.; Fisberg, R.M.; Marchioni, D.M. Dietary intake and food contributors of polyphenols in adults and elderly adults of Sao Paulo: A population-based study. Br. J. Nutr. 2016, 115, 1061–1070. [Google Scholar] [CrossRef]
- Kent, K.; Charlton, K.E.; Lee, S.; Mond, J.; Russell, J.; Mitchell, P.; Flood, V.M. Dietary flavonoid intake in older adults: How many days of dietary assessment are required and what is the impact of seasonality? Nutr. J. 2018, 17, 7. [Google Scholar] [CrossRef] [PubMed]
- Tena, N.; Martín, J.; Asuero, A.G. State of the art of anthocyanins: Antioxidant activity, sources, bioavailability, and therapeutic effect in human health. Antioxidants 2020, 9, 451. [Google Scholar] [CrossRef]
- Xie, L.; Vance, T.; Kim, B.; Lee, S.G.; Caceres, C.; Wang, Y.; Hubert, P.A.; Lee, J.Y.; Chun, O.K.; Bolling, B.W. Aronia berry polyphenol consumption reduces plasma total and low-density lipoprotein cholesterol in former smokers without lowering biomarkers of inflammation and oxidative stress: A randomized controlled trial. Nutr. Res. 2017, 37, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Broncel, M.; Koziróg, M.; Duchnowicz, P.; Koter-Michalak, M.; Sikora, J.; Chojnowska-Jezierska, J. Aronia melanocarpa extract reduces blood pressure, serum endothelin, lipid, and oxidative stress marker levels in patients with metabolic syndrome. Med. Sci. Monit. 2010, 16, CR28–CR34. [Google Scholar] [PubMed]
- Skoczyńska, A.; Jedrychowska, I.; Poreba, R.; Affelska-Jercha, A.; Turczyn, B.; Wojakowska, A.; Andrzejak, R. Influence of chokeberry juice on arterial blood pressure and lipid parameters in men with mild hypercholesterolemia. Pharmacol. Rep. 2007, 59, 177–182. [Google Scholar]
- Jin, Y.; Alimbetov, D.; George, T.; Gordon, M.H.; Lovegrove, J.A. A randomised trial to investigate the effects of acute consumption of a blackcurrant juice drink on markers of vascular reactivity and bioavailability of anthocyanins in human subjects. Eur. J. Clin. Nutr. 2011, 65, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Prior, R.L. Anthocyanins are detected in human plasma after oral administration of an elderberry extract. Clin. Chem. 1999, 45, 574–576. [Google Scholar] [CrossRef]
- Murkovic, M.; Toplak, H.; Adam, U.; Pfannhauser, W. Analysis of anthocyanins in plasma for determination of their bioavailability. J. Food Compos. Anal. 2000, 13, 291–296. [Google Scholar] [CrossRef]
- Sangiovanni, E.; Di Lorenzo, C.; Colombo, E.; Colombo, F.; Fumagalli, M.; Frigerio, G.; Restani, P.; Dell’Agli, M. The effect of in vitro gastrointestinal digestion on the anti-inflammatory activity of Vitis vinifera L. leaves. Food Funct. 2015, 6, 2453–2463. [Google Scholar] [CrossRef]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230–242. [Google Scholar] [CrossRef]
- Hollands, W.; Brett, G.M.; Radreau, P.; Saha, S.; Teucher, B.; Bennett, R.N.; Kroon, P.A. Processing blackcurrants dramatically reduces the content and does not enhance the urinary yield of anthocyanins in human subjects. Food Chem. 2008, 108, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Wedick, N.M.; Pan, A.; Cassidy, A.; Rimm, E.B.; Sampson, L.; Rosner, B.; Willett, W.; Hu, F.B.; Sun, Q.; Van Dam, R.M. Dietary flavonoid intakes and risk of type 2 diabetes in US men and women. Am. J. Clin. Nutr. 2012, 95, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, A.; Bertoia, M.; Chiuve, S.; Flint, A.; Forman, J.; Rimm, E.B. Habitual intake of anthocyanins and flavanones and risk of cardiovascular disease in men. Am. J. Clin. Nutr. 2016, 104, 587–594. [Google Scholar] [CrossRef]
- Milbury, P.E.; Vita, J.A.; Blumberg, J.B. Anthocyanins are bioavailable in humans following an acute dose of cranberry juice. J. Nutr. 2010, 140, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Duthie, S.J.; Jenkinson, A.M.E.; Crozier, A.; Mullen, W.; Pirie, L.; Kyle, J.; Yap, L.S.; Christen, P.; Duthie, G.G. The effects of cranberry juice consumption on antioxidant status and biomarkers relating to heart disease and cancer in healthy human volunteers. Eur. J. Nutr. 2006, 45, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Giordano, L.; Coletta, W.; Tamburrelli, C.; D’Imperio, M.; Crescente, M.; Silvestri, C.; Rapisarda, P.; Reforgiato Recupero, G.; De Curtis, A.; Iacoviello, L.; et al. Four-week ingestion of blood orange juice results in measurable anthocyanin urinary levels but does not affect cellular markers related to cardiovascular risk: A randomized cross-over study in healthy volunteers. Eur. J. Nutr. 2012, 51, 541–548. [Google Scholar] [CrossRef]
- Riso, P.; Visioli, F.; Gardana, C.; Grande, S.; Brusamolino, A.; Galvano, F.; Galvano, G.; Porrini, M. Effects of blood orange juice intake on antioxidant bioavailability and on different markers related to oxidative stress. J. Agric. Food Chem. 2005, 53, 941–947. [Google Scholar] [CrossRef]
- Vogiatzoglou, A.; Mulligan, A.A.; Luben, R.N.; Lentjes, M.A.H.; Heiss, C.; Kelm, M.; Merx, M.W.; Spencer, J.P.E.; Schroeter, H.; Kuhnle, G.G.C. Assessment of the dietary intake of total flavan-3-ols, monomeric flavan-3-ols, proanthocyanidins and theaflavins in the European Union. Br. J. Nutr. 2014, 111, 1463–1473. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Kroon, P.A.; Philo, M.; Mensink, M.; Kromhout, D.; Hollman, P.C.H. Does epicatechin contribute to the acute vascular function effects of dark chocolate? A randomized, crossover study. Mol. Nutr. Food Res. 2016, 60, 2379–2386. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the modification of the authorisation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilation pursuant to Article 13(5) of Regulation (EC) No 1924/2006 following a request in accordan. EFSA J. 2014, 12, 3654. [Google Scholar] [CrossRef]
- Engler, M.B.; Engler, M.M.; Chen, C.Y.; Malloy, M.J.; Browne, A.; Chiu, E.Y.; Kwak, H.-K.; Milbury, P.; Paul, S.M.; Blumberg, J.; et al. Flavonoid-rich dark chocolate improves endothelial function and increases plasma epicatechin concentrations in healthy adults. J. Am. Coll. Nutr. 2004, 23, 197–204. [Google Scholar] [CrossRef]
- Taubert, D.; Roesen, R.; Lehmann, C.; Jung, N.; Schömig, E. Effects of low habitual cocoa intake on blood pressure and bioactive nitric oxide: A randomized controlled trial. JAMA 2007, 298, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Ostertag, L.M.; Kroon, P.A.; Wood, S.; Horgan, G.W.; Cienfuegos-Jovellanos, E.; Saha, S.; Duthie, G.G.; De Roos, B. Flavan-3-ol-enriched dark chocolate and white chocolate improve acute measures of platelet function in a gender-specific way-a randomized-controlled human intervention trial. Mol. Nutr. Food Res. 2013, 57, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Mena, P.; Bresciani, L.; Brindani, N.; Ludwig, I.A.; Pereira-Caro, G.; Angelino, D.; Llorach, R.; Calani, L.; Brighenti, F.; Clifford, M.N.; et al. Phenyl-γ-valerolactones and phenylvaleric acids, the main colonic metabolites of flavan-3-ols: Synthesis, analysis, bioavailability, and bioactivity. Nat. Prod. Rep. 2019, 36, 714–752. [Google Scholar] [CrossRef] [PubMed]
- Montagnana, M.; Danese, E.; Angelino, D.; Mena, P.; Rosi, A.; Benati, M.; Gelati, M.; Salvagno, G.L.; Favaloro, E.J.; Del Rio, D.; et al. Dark chocolate modulates platelet function with a mechanism mediated by flavan-3-ol metabolites. Medicine 2018, 97, e13432. [Google Scholar] [CrossRef]
- Aherne, S.A.; O’Brien, N.M. Dietary flavonols: Chemistry, food content, and metabolism chemistry and structure of the flavonoids. Nutrition 2002, 18, 75–81. [Google Scholar] [CrossRef]
- Bondonno, N.P.; Bondonno, C.P.; Ward, N.C.; Woodman, R.J.; Hodgson, J.M.; Croft, K.D. Enzymatically modified isoquercitrin improves endothelial function in volunteers at risk of cardiovascular disease. Br. J. Nutr. 2020, 123, 182–189. [Google Scholar] [CrossRef]
- Suomela, J.P.; Ahotupa, M.; Yang, B.; Vasankari, T.; Kallio, H. Absorption of flavonols derived from sea buckthorn (Hippophaë rhamnoides L.) and their effect on emerging risk factors for cardiovascular disease in humans. J. Agric. Food Chem. 2006, 54, 7364–7369. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Mah, E.; Davis, C.G.; Jalili, T.; Ferruzzi, M.G.; Chun, O.K.; Bruno, R.S. Dietary fat increases quercetin bioavailability in overweight adults. Mol. Nutr. Food Res. 2013, 57, 896–905. [Google Scholar] [CrossRef]
- Hubbard, G.P.; Wolffram, S.; de Vos, R.; Bovy, A.; Gibbins, J.M.; Lovegrove, J.A. Ingestion of onion soup high in quercetin inhibits platelet aggregation and essential components of the collagen-stimulated platelet activation pathway in man: A pilot study. Br. J. Nutr. 2006, 96, 482–488. [Google Scholar] [CrossRef]
- Beatty, E.R.; O’Reilly, J.D.; England, T.G.; McAnlis, G.T.; Young, I.S.; Halliwell, B.; Geissler, C.A.; Sanders, T.A.B.; Wiseman, H. Effect of dietary quercetin on oxidative DNA damage in healthy human subjects. Br. J. Nutr. 2000, 84, 919–925. [Google Scholar] [CrossRef]
- O’Reilly, J.D.; Mallet, A.I.; McAnlis, G.T.; Young, I.S.; Halliwell, B.; Sanders, T.A.B.; Wiseman, H. Consumption of flavonoids in onions and black tea: Lack of effect on F2-isoprostanes and autoantibodies to oxidized LDL in healthy humans. Am. J. Clin. Nutr. 2001, 73, 1040–1044. [Google Scholar] [CrossRef] [PubMed]
- Larmo, P.S.; Yang, B.; Hurme, S.A.M.; Alin, J.A.; Kallio, H.P.; Salminen, E.K.; Tahvonen, R.L. Effect of a low dose of sea buckthorn berries on circulating concentrations of cholesterol, triacylglycerols, and flavonols in healthy adults. Eur. J. Nutr. 2009, 48, 277–282. [Google Scholar] [CrossRef]
- Arts, I.C.W.; Hollman, P.C.H. Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr. 2005, 81, 317S–325S. [Google Scholar] [CrossRef]
- State Pharmacopoeia Commission of the PRC. Pharmacopoeia of the People’s Republic of China (The First Part); People’s Medical Publishing House: Beijing, China, 2000; Volume I, ISBN 9787117069823. (In English)
- Wang, J.; Zhang, M.; Xu, Z.; Zhang, T.; Cheng, Y. Clinical observation on effects of sea buckthorn total flavones on ischemic heart diseases. Shanxi Med. Res. 1985, 2, 60–67. [Google Scholar]
- Bondonno, N.P.; Bondonno, C.P.; Rich, L.; Mas, E.; Shinde, S.; Ward, N.C.; Hodgson, J.M.; Croft, K.D. Acute effects of quercetin-3-O-glucoside on endothelial function and blood pressure: A randomized dose-response study. Am. J. Clin. Nutr. 2016, 104, 97–103. [Google Scholar] [CrossRef]
- Hubbard, G.P.; Wolffram, S.; Lovegrove, J.A.; Gibbins, J.M. Ingestion of quercetin inhibits platelet aggregation and essential components of the collagen-stimulated platelet activation pathway in humans. J. Thromb. Haemost. 2004, 2, 2138–2145. [Google Scholar] [CrossRef] [PubMed]
- Loke, W.M.; Hodgson, J.M.; Proudfoot, J.M.; McKinley, A.J.; Puddey, I.B.; Croft, K.D. Pure dietary flavonoids quercetin and (-)-epicatechin augment nitric oxide products and reduce endothelin-1 acutely in healthy men. Am. J. Clin. Nutr. 2008, 88, 1018–1025. [Google Scholar] [CrossRef]
- Perez, A.; Gonzalez-Manzano, S.; Jimenez, R.; Perez-Abud, R.; Haro, J.M.; Osuna, A.; Santos-Buelga, C.; Duarte, J.; Perez-Vizcaino, F. The flavonoid quercetin induces acute vasodilator effects in healthy volunteers: Correlation with beta-glucuronidase activity. Pharmacol. Res. 2014, 89, 11–18. [Google Scholar] [CrossRef]
- Bento-Silva, A.; Koistinen, V.M.; Mena, P.; Bronze, M.R.; Hanhineva, K.; Sahlstrøm, S.; Kitrytė, V.; Moco, S.; Aura, A.M. Factors affecting intake, metabolism and health benefits of phenolic acids: Do we understand individual variability? Eur. J. Nutr. 2020, 59, 1275–1293. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, K.; Nagel, C.W. Occurrence and content of hydroxycinnamic and hydroxybenzoic acid compounds in foods. Crit. Rev. Food Sci. Nutr. 1989, 28, 315–347. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, I.A.; Mena, P.; Calani, L.; Cid, C.; Del Rio, D.; Lean, M.E.J.; Crozier, A. Variations in caffeine and chlorogenic acid contents of coffees: What are we drinking? Food Funct. 2014, 5, 1718–1726. [Google Scholar] [CrossRef] [PubMed]
- El-Seedi, H.R.; El-Said, A.M.A.; Khalifa, S.A.M.; Göransson, U.; Bohlin, L.; Borg-Karlson, A.K.; Verpoorte, R. Biosynthesis, natural sources, dietary intake, pharmacokinetic properties, and biological activities of hydroxycinnamic acids. J. Agric. Food Chem. 2012, 60, 10877–10895. [Google Scholar] [CrossRef] [PubMed]
- Mills, C.E.; Flury, A.; Marmet, C.; Poquet, L.; Rimoldi, S.F.; Sartori, C.; Rexhaj, E.; Brenner, R.; Allemann, Y.; Zimmermann, D.; et al. Mediation of coffee-induced improvements in human vascular function by chlorogenic acids and its metabolites: Two randomized, controlled, crossover intervention trials. Clin. Nutr. 2017, 36, 1520–1529. [Google Scholar] [CrossRef]
- Mubarak, A.; Bondonno, C.P.; Liu, A.H.; Considine, M.J.; Rich, L.; Mas, E.; Croft, K.D.; Hodgson, J.M. Acute effects of chlorogenic acid on nitric oxide status, endothelial function, and blood pressure in healthy volunteers: A randomized trial. J. Agric. Food Chem. 2012, 60, 9130–9136. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Nam, Y.; Kim, J.; Choi, H.; Won, C. Coffee consumption and stroke risk: A meta-analysis of epidemiologic studies. Korean J. Fam. Med. 2012, 33, 356–365. [Google Scholar] [CrossRef]
- Lafay, S.; Morand, C.; Manach, C.; Besson, C.; Scalbert, A. Absorption and metabolism of caffeic acid and chlorogenic acid in the small intestine of rats. Br. J. Nutr. 2006, 96, 39–46. [Google Scholar] [CrossRef]
- Agudelo-Ochoa, G.M.; Pulgarín-Zapata, I.C.; Velásquez-Rodriguez, C.M.; Duque-Ramírez, M.; Naranjo-Cano, M.; Quintero-Ortiz, M.M.; Lara-Guzmán, O.J.; Muñoz-Durango, K. Coffee consumption increases the antioxidant capacity of plasma and has no effect on the lipid profile or vascular function in healthy adults in a randomized controlled trial. J. Nutr. 2016, 146, 524–531. [Google Scholar] [CrossRef]
- Boon, E.A.J.; Croft, K.D.; Shinde, S.; Hodgson, J.M.; Ward, N.C. The acute effect of coffee on endothelial function and glucose metabolism following a glucose load in healthy human volunteers. Food Funct. 2017, 8, 3366–3373. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Rendeiro, C.; Bergillos-Meca, T.; Tabatabaee, S.; George, T.W.; Heiss, C.; Spencer, J.P.E. Intake and time dependence of blueberry flavonoid-induced improvements in vascular function: A randomized, controlled, double-blind, crossover intervention study with mechanistic insights into biological activity. Am. J. Clin. Nutr. 2013, 98, 1179–1191. [Google Scholar] [CrossRef]
- Vitaglione, P.; Mennella, I.; Ferracane, R.; Rivellese, A.A.; Giacco, R.; Ercolini, D.; Gibbons, S.M.; La Storia, A.; Gilbert, J.A.; Jonnalagadda, S.; et al. Whole-grain wheat consumption reduces inflammation in a randomized controlled trial on overweight and obese subjects with unhealthy dietary and lifestyle behaviors: Role of polyphenols bound to cereal dietary fiber. Am. J. Clin. Nutr. 2015, 101, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Ward, N.C.; Hodgson, J.M.; Woodman, R.J.; Zimmermann, D.; Poquet, L.; Leveques, A.; Actis-Goretta, L.; Puddey, I.B.; Croft, K.D. Acute effects of chlorogenic acids on endothelial function and blood pressure in healthy men and women. Food Funct. 2016, 7, 2197–2203. [Google Scholar] [CrossRef] [PubMed]
- Kempf, K.; Herder, C.; Erlund, I.; Kolb, H.; Martin, S.; Carstensen, M.; Koenig, W.; Sundvall, J.; Bidel, S.; Kuha, S.; et al. Effects of coffee consumption on subclinical inflammation and other risk factors for type 2 diabetes: A clinical trial. Am. J. Clin. Nutr. 2010, 91, 950–957. [Google Scholar] [CrossRef]
- Lara-Guzmán, O.J.; Álvarez-Quintero, R.; Osorio, E.; Naranjo-Cano, M.; Muñoz-Durango, K. GC/MS method to quantify bioavailable phenolic compounds and antioxidant capacity determination of plasma after acute coffee consumption in human volunteers. Food Res. Int. 2016, 89, 219–226. [Google Scholar] [CrossRef]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the healthy gut microbiota composition? A changing ecosystem across age, environment, diet, and diseases. Microorganisms 2019, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Renouf, M.; Guy, P.A.; Marmet, C.; Fraering, A.L.; Longet, K.; Moulin, J.; Enslen, M.; Barron, D.; Dionisi, F.; Cavin, C.; et al. Measurement of caffeic and ferulic acid equivalents in plasma after coffee consumption: Small intestine and colon are key sites for coffee metabolism. Mol. Nutr. Food Res. 2010, 54, 760–766. [Google Scholar] [CrossRef]
- Wightman, E.L.; Haskell-Ramsay, C.F.; Reay, J.L.; Williamson, G.; Dew, T.; Zhang, W.; Kennedy, D.O. The effects of chronic trans-resveratrol supplementation on aspects of cognitive function, mood, sleep, health and cerebral blood flow in healthy, young humans. Br. J. Nutr. 2015, 114, 1427–1437. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Wightman, E.L.; Reay, J.L.; Lietz, G.; Okello, E.J.; Wilde, A.; Haskell, C.F. Effects of resveratrol on cerebral blood flow variables and cognitive performance in humans: A double-blind, placebo-controlled, crossover investigation. Am. J. Clin. Nutr. 2010, 91, 1590–1597. [Google Scholar] [CrossRef] [PubMed]
- Pastor, R.F.; Restani, P.; Di Lorenzo, C.; Orgiu, F.; Teissedre, P.L.; Stockley, C.; Ruf, J.C.; Quini, C.I.; Garcìa Tejedor, N.; Gargantini, R.; et al. Resveratrol, human health and winemaking perspectives. Crit. Rev. Food Sci. Nutr. 2019, 59, 1237–1255. [Google Scholar] [CrossRef] [PubMed]
- Hanwell, H.E.C.; Kay, C.D.; Lampe, J.W.; Holub, B.J.; Duncan, A.M. Acute fish oil and soy isoflavone supplementation increase postprandial serum (n-3) polyunsaturated fatty acids and isoflavones but do not affect triacylglycerols or biomarkers of oxidative stress in overweight and obese hypertriglyceridemic men. J. Nutr. 2009, 139, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.N.T.; Thybo, C.B.; Lykkeboe, S.; Rasmussen, L.M.; Frette, X.; Christensen, L.P.; Jeppesen, P.B. Combined bioavailable isoflavones and probiotics improve bone status and estrogen metabolism in postmenopausal osteopenic women: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 106, 909–920. [Google Scholar] [CrossRef] [PubMed]
- Brink, E.; Coxam, V.; Robins, S.; Wahala, K.; Cassidy, A.; Branca, F. Long-term consumption of isoflavone-enriched foods does not affect bone mineral density, bone metabolism, or hormonal status in early postmenopausal women: A randomized, double-blind, placebo controlled study. Am. J. Clin. Nutr. 2008, 87, 761–770. [Google Scholar] [CrossRef]
- Schar, M.Y.; Curtis, P.J.; Hazim, S.; Ostertag, L.M.; Kay, C.D.; Potter, J.F.; Cassidy, A. Orange juice-derived flavanone and phenolic metabolites do not acutely affect cardiovascular risk biomarkers: A randomized, placebo-controlled, crossover trial in men at moderate risk of cardiovascular disease1-5. Am. J. Clin. Nutr. 2015, 101, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Morand, C.; Dubray, C.; Milenkovic, D.; Lioger, D.; Martin, J.F.; Scalbert, A.; Mazur, A. Hesperidin contributes to the vascular protective effects of orange juice: A randomized crossover study in healthy volunteers. Am. J. Clin. Nutr. 2011, 93, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Rendeiro, C.; Dong, H.; Saunders, C.; Harkness, L.; Blaze, M.; Hou, Y.; Belanger, R.L.; Corona, G.; Lovegrove, J.A.; Spencer, J.P.E. Flavanone-rich citrus beverages counteract the transient decline in postprandial endothelial function in humans: A randomised, controlled, double-masked, cross-over intervention study. Br. J. Nutr. 2016, 116, 1999–2010. [Google Scholar] [CrossRef] [PubMed]
| Cohort and Study Details | Anthocyanin Intake | Aim | Bioavailability Data | Outcome | Reference |
|---|---|---|---|---|---|
| 49 healthy adult former smokers (25 M; 24 F) (mean age 35 ± 2.8 years) Duration: 12 weeks Randomized controlled trial |
| Modulation plasma lipids (change in LDL cholesterol was the primary outcome), blood pressure, biomarkers of inflammation and oxidative stress, lipid transport genes of peripheral blood mononuclear cells. | Overnight urinary anthocyanins were significantly higher in the treatment group vs. placebo group (0.332 ± 0.136 vs. 0.051 ± 0.022 mg mg−1 creatinine). Urinary peonidin-3-galactoside was 0.0062 ± 0.0026 mg mg−1 creatinine in the treatment group vs. 0.0008 ± 0.0005 mg mg−1 creatinine in placebo group (p < 0.05). The excretion of the other polyphenols was not significantly affected. | After 12 weeks, Aronia consumption compared with the placebo group showed: ↓ 8% fasting plasma total cholesterol (p = 0.0140), ↓ 11% LDL cholesterol (p = 0.02), ↓ LDL receptor protein in peripheral blood mononuclear cells (p = 0.0036). | [13] |
| 20 Healthy subjects (UK) (9 M; 11 F) mean age (44.55 ± 13.34 years) Duration: acute consumption Randomized, double-blind, placebo-controlled crossover study | 250 mL of either a blackcurrant juice drink (20% of anthocyanins) or the control drink. Sample collection after consumption:
| To measure vascular reactivity at 120 min after juice consumption. | The urinary percentage anthocyanins excreted after 120 min was 0.021 ± 0.003% and 0.009 ± 0.002% of the dietary intake of delphinidin glycosides and cyanidin glycosides, respectively. | No significant effects on vascular reactivity were found. An increase in plasmatic vitamin C was observed (p = 0.006). | [16] |
| 15 subjects with coronary artery disease (13 M; 2 F) (mean age: 62 ± 8 years) Duration: acute consumption Pilot study | 480 mL cranberry juice (54% juice; 835 mg total polyphenols; 94.47 mg anthocyanins). Sample collection after consumption:
| To evaluate plasma redox capacity. | Plasma concentrations ranged between 0.56 and 4.64 nmol L−1. Total urinary anthocyanins were 0.79 ± 0.90% of the amount taken. Cyanidin-glucoside and peonidin-glucoside were the most available (0.007 ± 0.004% and 0.029 ± 0.059% of the dose delivered). | Anthocyanin plasmatic concentrations were not able to reduce oxidative stress. | [24] |
| 20 healthy females (mean age: 27.8 ± 7 years) Randomized controlled trial Duration: 2 weeks |
| Plasma antioxidant activity and biomarkers of oxidative stress (total phenol concentrations, reduced glutathione levels (GSH) and plasma free radical trapping capacity (FRAP)). Activity of blood’s antioxidant enzymes (SOD, catalase and GSH-Px). Urinary excretion of MDA and 8-oxo-deoxyguanosine (as marker of DNA damage). | Neither anthocyanins nor catechins were detectable in plasma samples isolated from both groups, only vitamin C increased significantly (p < 0.01) in the cranberry juice group. Further, catechins were not detectable in the urine samples. | Hcy, TC, TG, HDL, and LDL were unchanged. The antioxidant potential of the plasma, GSH-Px, catalase and SOD activities, MDA and 8-oxo-deoxyguanosine levels were not significantly different in both groups. | [25] |
| 18 healthy subjects (10 M; 8 F) (25–47 years) Duration: 4 weeks Randomized crossover study | 1 L per day of either blood orange juice (OJ) (from Moro, Tarocco, and Sanguinello varieties) or blond OJ that contains no anthocyanins (from Valencia, navel, and Belladonna varieties). Blood orange juice contained: 53.09 ± 5.31 mg L−1 total anthocyanins, 3.96 ± 0.20 mg L−1 delphinidin-3-glucoside, 25.79 ± 1.17 mg L−1 cyandin-3-glucoside, 17.88 ± 0.95 mg L−1 cyanidin-3-(6-malonylglucoside). | Potential effects on cell markers of platelet and leukocyte activation (P-selectin, PAC-1, leukocyte activation markers CD11b) due to anthocyanins absorption after daily supplementation with blood OJ for 1 month. | Mean levels of anthocyanins (11.47 ± 5.63 nmol L−1) significantly differed from baseline in 24-h urinary excretion. Anthocyanins levels remained substantially unchanged until the end of treatment (p = 0.1). | The anthocyanin plasma levels reached were insufficient to significantly modify cell markers of platelet and leukocyte activation and interaction. Urinary excretions of anthocyanins considered showed a significant increase after blood OJ consumption (p < 0.05). | [26] |
| 16 healthy females (20–27 years) Duration: 3 weeks Randomized crossover study |
- Wash out period; - Standardized diet with 600 mL/day of blood orange juice.
- Wash-out period; - Standardized diet without orange juice. Each period was of 21 days. 100 mL of orange juice contained: 75.2 mg vitamin C, 67 μg of β-cryptoxanthin, 20 μg of lutein, 18 μg of zeaxanthin, 17 μg of lycopene, 10 μg of β-carotene, and 8 μg of α-carotene, 3.5 mg cyanidin-3-glucoside, 1.2 mg cyanidin-3-glucoside-6″-malonyl. | To evaluate the effect on plasma antioxidant concentrations and on markers of lipid peroxidation malondialdehyde (MDA) and 11-dehydro-tromboxane 2 (TXB2) | Cyanidin 3-glucoside mean plasma concentration was, after the washout period, about 0.6 nmol L−1 and increased from 0.56 nmol L−1 to ∼8 nmol L−1 after 3 weeks of blood orange juice intake (p < 0.05). Both the aglycone and the cyanidin-3-glucoside-6″-malonyl were not detected in plasma. | Plasma antioxidant capacity did not increase after the 3 weeks of juice intake. The daily intake of orange juice did not affect the biomarkers of lipid oxidation malondialdehyde (MDA), and 11-dehydro-TXB2. | [27] |
| Cohort and Study Details | Flavanol Intake | Aim | Bioavailability Data | Outcome | Reference |
|---|---|---|---|---|---|
| 20 healthy men (40–80 years) Duration: acute consumption randomized, placebo controlled, crossover study | Subjects consumed the following:
| To study the effect of epicatechin from different matrices (cocoa and supplement) on vascular function (FMD and AIx). | DC intake determined an Area Under the Curve of 16.2 μM (per 0.100 g of EC) that was not significantly different from Area Under the Curve calculated for pure EC (p = 0.14). | Pure EC didn’t ameliorate Flow Mediated Dilatation (+0.75%; p = 0.10) or Augmentation Index (2.2%; p = 0.23) respect to placebo. DC significantly ameliorated Flow Mediated Dilatation (+0.96%; p = 0.04) and Augmentation Index (4.6%; p = 0.02). Amelioration of FMD (+0.21%; p = 0.65) or Aix (–2.4%; p = 0.20) was not different after pure EC and DC intake. EC bioavailability was not different after pure EC and DC (p = 0.14). | [29] |
| 21 healthy subjects (11 M; 11 F) (mean age: 32.2 ± 3.1 years) Duration: 2 weeks Randomized, double-blind, controlled with placebo study | The following treatments were assigned:
| To evaluate the impact of DC enriched with flavonoids on FMD, blood pressure, markers of oxidative stress (LDL-ox, total antioxidant power, 8-isoprostane levels and plasmatic lipid concentrations). | Plasmatic EC levels were significantly higher at two weeks after high-flavonoid DC consumption (204.4 ± 18.5 nmol L−1, p < or =0.001) than after low-flavonoid DC intake (17.5 ± 9 nmol L−1, p = 0.99). | The intake of chocolate containing high levels of flavonoids ameliorated FMD (average change = 1.3 ± 0.7%) respect to the groups consuming chocolate with low levels of flavonoids (mean change = −0.96 ± 0.5%) (p = 0.024). LDL-ox, antioxidant power, 8-isoprostanes, blood pressure, lipid biomarkers were not significantly in the two groups. | [31] |
| 44 adults (20 M; 24 F, 20) (Age: 63.6 ± 4.8 years) with not-treated hypertension (at stage 1) without other diseases Duration: 18 weeks Randomized study | Treated subjects (n = 22): 6.3 g dark chocolate per day providing 30 mg of polyphenols per day (1.4 mg gallic acid, 1.7 mg catechin, 5.1 mg epicatechin, 0.3 mg epicatechin-gallate, 6.8 mg procyanidin dimer, 1.8 procyanidin dimer-gallate, 5.3 mg procyanidin trimer, 1.0 mg procyanidin trimer-gallate, 3.7 mg procyanidin tetramer, 2.6 mg procyanidin pentamer, <0.05 mg flavonols, 3.1 mg caffeine, 26.4 mg theobromine) Control group (n = 22) polyphenol-free white chocolate. | To evaluate the change in blood pressure. Secondary objectives were the assessment of nitric oxide and oxidative stress plasmatic biomarkers (S-nitrosoglutathione and 8-isoprostane, respectively). | Only catechin, epicatechin and the dimers procyanidin B2 and procyanidin B2 gallate were measured in plasma and didn’t change after 18 weeks. Pharmacokinetic data were: Epicatechin: AUC from 761 (±210) ng mL−1 × min on day 1 to 774 (±253) ng mL−1 × min (p = 0.82) at 18 weeks; Cmax from 3.63 (±1.02) ng/mL on day 1 to 3.58 (±0.92) ng mL−1 (p = 0.78) at 18 weeks; Tmax from 77 (±4) min on day 1 to 81 (±6) min at 18 weeks (p = 0.70); T1/2 from 54 (±3) min on day 1 to 56 (±5) min (p = 0.79) at 18 weeks. Catechin: AUC from 234 (±61) (day 1) to 228 (±56) ng mL−1 × min (p = 0.77); Cmax from 1.12 (±0.31) ng mL−1 on day 1 to 1.01 (±0.26) ng mL−1 (p = 0.58) at 18 weeks; Tmax from 78 (±9) min on day 1 to 82 (±6) min at 18 weeks (p = 0.69); T1/2 from 54 (±6) min on day 1 to 58 (±7) min (p = 0.68) at 18 weeks. Procyanidin B2: AUC from 99 (±30) ng mL−1 × min on day 1 to 102 (±32) ng mL−1 × min (p = 0.90); Cmax from 0.45 (±0.15) ng mL−1 on day 1 to 0.43 (±0.14) ng mL−1 (p = 0.94) at 18 weeks; Tmax from 81 (±8) min on day 1 to 86 (±9) min at 18 weeks (p = 0.62); T1/2 from 56 (±6) min on day 1 to 57 (±5) min (p = 0.91) at 18 weeks; procyanidin B2 gallate: AUC from 33 (±14) ng mL−1 × min on day 1 to 33 (±13) ng mL−1 × min (p = 0.91); Cmax from 0.14 (±0.06) ng mL−1- day 1 to 0.14 (±0.06) ng mL−1 (p = 0.98) at 18 weeks; Tmax from 89 (±10) min on day 1 to 85 (±8) min at 18 weeks (p = 0.72); T1/2 from 62 (±7) min on day 1 to 59 (±6) min (p = 0.76) at 18 weeks. | At the end of the study, the treatment with dark chocolate educed mean systolic blood pressure by 2.9 (±1.6) mm Hg (p < 0.001) and diastolic blood pressure by 1.9 (±1) mm Hg (p < 0.001) but 8-isoprostane levels didn’t change. Hypertension prevalence was reduced from 86% to 68%. S-nitrosoglutathione levels (increased by 0.23 nmol L−1 (±0.12) (p < 0.001). | [32] |
| 42 healthy subjects (26 M; 16 F) (mean age 41 ± 2.0 years) Duration: acute consumption Observer-blinded randomized-controlled crossover acute intervention trial | Subjects received 60 g of dark chocolate enriched with flavan-3-ols (with 400 mL water), 60 g of a “standard” dark chocolate or 60 g of white chocolate (14 days wash out period was used between treatments). Sixty grams of chocolate enriched with flavan-3-ols contained: 0.257 (±1.06) g epicatechin, 53.6 (±0.27) mg catechin, 0.198 (±1.22) g dimer B2, 0.168 (±1.42) g trimers, 0.105 ± (12.75) g tetramers, 0.125 ± (6.04) g pentamers (total flavonoids = 907.4 ± 22.75 mg). Sixty grams of “standard” dark chocolate contained: 84.1 (±0.67) mg epicatechin, 25.8 (±1.02) mg catechin, 74.4 (±0.76) mg dimer B2, 47 (±2.23) mg trimers, 32.1 (±4.4) mg tetramers, 0.119 (±39.5) g pentamers (total flavonoids: 382.3 ± 45.2 mg). White chocolate = ND. | To evaluate if flavan-3-ol-enriched dark chocolate could influence markers of platelet status (adenosine diphosphate-induced platelet aggregation, expression of P-selectin, thrombin receptor-activating peptide-induced platelet aggregation and thrombin receptor-activating peptide-induced fibrinogen binding, collagen/epinephrine-induced bleeding time). | Plasmatic levels of total C/EC significantly increased 120 min after flavan-3-oil enriched or “standard “dark chocolate in comparison with white chocolate (p < 0.001) (Cmax = 1.20 μmol L−1 120 min after consumption of dark chocolate enriched with flavan3-ols) and decreased after 6 h. Urinary levels of total catechins increased 120 and 360 min after consumption of enriched dark or “standard” dark chocolate in comparison with white chocolate (p < 0.001), reaching a peak concentration of 13.4 mmol mol−1 creatinine. Comparable results were observed for procyanidin dimer B 2 (p < 0.001), reaching a peak concentration at 57 μmol mol−1 creatinine at 360 min. The peak of flavan-3-ol concentrations was higher in biological fluids of women (p = 0.047). | Enriched dark and white chocolate ameliorated different biomarkers of platelet status (adenosine diphosphate-induced platelet aggregation, P-selectin expression, thrombin receptor-activating peptide-induced platelet aggregation and thrombin receptor-activating peptide-induced fibrinogen binding) following a gender-specific fashion. | [33] |
| 18 healthy male subjects (mean age: 36 ± 10 years) Duration: acute consumption Observational study | Subjects consumed 50 g of 90% cocoa chocolate (containing 7.5 g total polyphenols expressed as GA equivalents) within 5 min. | To investigate the impact of dark chocolate on platelet status and the correlation with flavanols metabolites. | After 4 h from the ingestion, epicatechin metabolites concentration in plasma significantly increased (epicatechin glucuronide, sulfate, methyl-epicatechin-sulfate) (p < 0.05). | An inhibitory effect of cocoa was observed on platelet aggregation and adhesion caused by ADP/collagen 4 h after cocoa intake (98.5 ± 13.0 respect to 114.5 ± 22 s, p < 0.001) was noticed, while the closure time of collagen/epinephrine didn’t change (128.5 ± 27.0 vs. 122.5 ± 34. s, p = 0.33), probably associated with EC metabolites. | [35] |
| Cohort and Study Details | Flavonol Intake | Aim | Bioavailability Data | Outcome | Reference |
|---|---|---|---|---|---|
| 25 participants (12 M; 13 F) (mean age: 64.1 ± 6.3 years) with at least one CVD risk factor (sBP 120–160 mmHg, FPG 5.6–6.5 mM, total cholesterol 5–8 mM or a waist circumference > 94 cm for men or >80 cm for women) Duration: acute consumption Randomized, controlled crossover trial | Treatment group: - 4.89 mg/kg bw/day of EMIQ® (Enzymatically Modified IsoQuercitrin), - half teaspoon of maltodextrin, - one and half tablespoons of Cottee’s Raspberry flavored cordial. Placebo group: - half teaspoon of maltodextrin, - one and half tablespoons of Cottee’s Raspberry flavored cordial, The treatments were given in 250 mL of water. | To evaluate if FMD, BP, and cognitive function improve whether an acute intake of EMIQ® was administered. | After 3 h from the consumption of EMIQ®, quercetin metabolites concentration was significantly higher in plasma respect placebo group (quercetin aglycone 144.9 ± 12.3 nM vs. 12.6 ± 12.3 nM; and isorhamnetin 245.5 ± 16.5 nM vs. 41.7 ± 16.5 nM) (p < 0.001). | EMIQ® significantly affected FMD compared with the placebo (p = 0.025). | [37] |
| 14 healthy males (46.6 ± 5.6 years) with a slightly elevated total cholesterol level (5.3–7.2 mmol/L) Duration: 4 weeks Double-blind, placebo-controlled, crossover study |
0.4 g of extract, added to the porridge, contained: 78 mg of total flavonol aglycones, of which 70% isorhamnetin, 26% quercetin, and 4% kaempferol. | To evaluate the effects on CRP, conjugated dienes and oxidized LDL, homocysteine, and paraoxonase activity (potential risk factors of CVD) of a flavonols extract of sea buckthorn. | Flavonols were mainly present as glucuronide and sulfate metabolites in plasma fluid. When was intake porridge added with flavonols extract, AUC was significantly higher for kaempferol and isorhamnetin (p < 0.05). | The flavonols ingested did not significantly affect the following: - Oxidized LDL; - CRP; - Homocysteine levels; - Plasma antioxidant potential; - Paraoxonase activity. | [38] |
| 9 overweight/obese men (n = 4) and post-menopausal women (n = 5) (mean age = 55.9 ± 2.1 years) Duration: acute consumption Randomized, crossover study | Subjects ingested quercetin aglycone (1095 mg) with 3 types of standardized breakfast: - Fat-free (<0.5 g); - Low-fat (4.0 g); - High-fat (15.4 g). | To verify whether dietary fat improve quercetin and its metabolites bioavailability in adults with high CVD risk. | During the high-fat breakfast, compared to the fat-free trial: - Plasma quercetin: ↑ 45% Cmax; ↑ 32% AUC (0–24 h); - plasma isorhamnetin: ↑ 40% Cmax; ↑ 19% AUC (0–24 h); - Plasma O-methyl-isorhamnetin: ↑ 46% Cmax; ↑ 43% AUC (0–24 h). | Dietary fat improved quercetin bioavailability by increasing its absorption leading to a possible dietary approach for reducing CVD risk. | [39] |
| 6 heathy subjects (4 M; 2 F) (mean age 34 ± 7 years) Duration: 1 day Randomized, double-blind, crossover study | Participants ingested either a high- or a low-quercetin soup (600 mL), made using 500 g of onions for portion. 1 L of low quercetin onion soup (LQS) contained 0.1 mg L−1 quercetin aglycone, 3.8 mg L−1 quercetin-4′-glucoside, 4.3 mg L−1 quercetin 3,4′-glucoside. 1 L of high quercetin onion soup (HQS) contained: 1.1 mg L−1 quercetin aglycone, 53.2 mg L−1 quercetin-4′-glucoside, 60.5 mg L−1 quercetin 3,4′-glucoside. | To investigate the possible inhibitory effects of quercetin ingestion from a dietary source on platelet function (collagen-stimulated platelet aggregation and collagen-stimulated tyrosine phosphorylation). | After HQS treatment, plasma levels of quercetin did the following: - Peaked at 2.59 ± 0.42 mmol L−1 (p = 0.0001); - AUC 911.61 ± 85·17 mmol L−1 per min (p = 0.001). Plasma levels of isorhamnetin peaked after 2 h at 0.119 ± 0.02 mmol L−1 (HQS) and 0.0133 ± 0.04 mmol L−1 (LQS) (p = 0.0001). Plasma levels of tamarixetin peaked after 2.5 h at 0.172 ± 0.035) mmol L−1 (HQS) and 0.0049 ± 0.001 mmol L−1 (LQS) (p = 0.0001). | HQS treatment inhibited the following: - Collagen-stimulated platelet aggregation (time-dependent); - Collagen-stimulated tyrosine phosphorylation (p = 0.001). The inhibition of tyrosine phosphorylation was correlated with AUC of quercetin after HQS intake. | [40] |
| 36 healthy human subjects (16 M; 20 F) (mean age: 31.4 ± 7.7 years) Duration: 4 weeks Randomized crossover study | Treatment period: high flavonol (HF) diet based on daily consumption of 150 g onion cake (89.7 mg quercetin) + 300 mL black tea (1.4 mg quercetin). Control period: low flavonol (LF) period based on exclusion of flavonol and flavone foods and tea. | To determine the effect of dietary intake of quercetin from onions and black tea on oxidative damage to leukocytes DNA bases. | Plasma quercetin was <LOD (66.2 nmol L−1) after the HF period and increased at 228.5 ± 34.7 nmol L−1 after HF period. | The concentrations of the products of oxidative damage to DNA bases did not differ significantly between the two dietary treatment periods for any of the products measured. | [41] |
| 32 healthy subjects (mean age 30.4 ± 7.3 years) Duration: 4 weeks Randomized crossover study | Treatment period: high flavonol (HF) diet based on daily consumption of 150 g onion cake (89.7 mg quercetin) + 300 mL black tea (1.4 mg quercetin). Control period: low flavonol (LF) period based on exclusion of flavonol and flavone foods and tea. | To investigate the effects of a high-flavonoid (HF) diet on markers of oxidative stress (F2 -isoprostanes and malondialdehyde (MDA)-modified LDL) compared with a low-flavonoid (LF) diet. | After the HF treatment, plasma quercetin concentrations were significantly higher (221.6 ± 37.4 nmol L−1) than after the LF treatment (compared with less than the LOD of 66.2 nmol L−1). | There were no significant differences in plasma F2-isoprostane concentrations, and MDA–LDL between the HF and LF dietary treatments. | [42] |
| 229 healthy subjects (mean age 31.05 ± 8.9 years) Duration: 3 months Randomized double-blind, placebo controlled study | Participants consumed 16.7 mg/day of sea buckthorn extract or placebo, added to 28 g of puree. The daily dose of sea buckthorn extract contained: - 5.8 ± 0.7 mg isorhamnetin 3-O-glucoside-7-O-rhamnoside; - 1.5 ± 0.9 mg quercetin 3-Orutinoside; - 1.6 ± 0.4 mg quercetin 3-O-glucoside; - 5.1 ± 0.8 mg isorhamnetin 3-O-rutinoside; - 2.4 ± 0.4 mg isorhamnetin 3-O-glucoside; - 0.3 ± 0.4 mg kaempferol 3-O-rutinoside. | To study the effect of flavonoid-rich sea buckthorn berry on circulating lipid markers associated with CVD risk (total, HDL and LDL cholesterol, triacylglycerols) and CRP. | The consumption of sea buckthorn extract significantly modified the plasma concentration in treated group: ↑ quercetin (3.0 ng mL−1, p = 0.03); ↑ isorhamnetin (3.9 ng mL−1, p < 0.01). | Sea buckthorn extract did not affect serum concentration of any CVD risk factors considered. | [43] |
| 15 healthy volunteers (6 M; 9 F) (mean age 60.8 ± 9.3 years) Duration: 1 week Randomized, controlled, crossover study | Each subject received 5 doses of quercetin-3-O-glucoside: - 0 mg; - 50 mg; - 100 mg; - 200 mg; - 400 mg. Each compound (control or treatment) was provided once in the morning in a cup of coffee. | To determine whether endothelial function, BP and NO were affected in a dose-dependent mode of administration of quercetin-3-O-glucoside. | After the intake of increasing doses of quercetin-3-O-glucoside, was observed: ↑ quercetin dose-dependent plasma concentrations (R2 = 0.52, p < 0.001), ↑ isorhamnetin dose-dependent plasma concentrations (R2 = 0.12, p = 0.005). Baseline: - free quercetin 1.90 ± 1.1 mM; - isorhamnetin 0.99 ± 0.06 mM. | After any intervention, no improvements were observed in: - endothelial function, - BP; - NO production. | [47] |
| 6 healthy subjects (4 M; 2 F) (mean age 34 ± 7 years) Duration: 1 day Randomized placebo-controlled crossover study | Participants were randomly treated with the following: - 150 mg Q-4-G in 5% ethanol; - 300 mg Q-4-G in 5% ethanol; - 5% (v/v) ethanol control drink. | To investigate the effect of the dietary ingestion of quercetin on platelet function (platelet aggregation and platelet collagen-stimulated tyrosine phosphorylation). | Plasma concentrations peaked 30 min after ingestion. Group 150 mg: - Quercetin 4.66 ± 0.77 μM; - Isorhamnetin 0.16 ± 0.05 μM; - Tamarixetin 0.24 ± 0.07 μM; - Total flavonoid 5.07 ± 0.90 μM. Group 300 mg: - Quercetin 9.72 ± 1.38 μM; - Isorhamnetin 0.44 ± 0.07 μM; - Tamarixetin 0.54 ± 0.09 μM (after 45 min); - Total flavonoid 10.66 ± 1.55 μM. These results indicating dose-dependent bioavailability of flavonoid. | After 30 and 120 min since intake of both doses of Q-4-G were inhibited: - platelet aggregation (p = 0.001); - collagen-stimulated tyrosine phosphorylation of TPP (p = 0.001). | [48] |
| 12 healthy men (mean age of 43.2 ± 4.3 years) Duration: acute consumption Randomized, placebo-controlled, crossover trial | Each participant received, in random order, 4 treatments: - 300 mL water (control); - 0.67 mg/mL quercetin; - 0.67 mg/mL epicatechin; - 0.67 mg/mL EGCG. | To evaluate the effects of quercetin and epicatechin on the endothelial function (measuring endothelin-1 and NO production) and oxidative stress (measuring urinary F2-isoprostanes). | Acute treatment with quercetin and epicatechin significantly increased (p < 0.001) the total circulating concentration of each flavonoid (from 0.84 ± 0.39 μmol L−1 to 3.54 ± 1.57 μmol L−1 for quercetin and from and 0.70 ± 0.34 μmol L−1 to 3.57 ± 1.21 μmol L−1 for epicatechin). In urine, concentrations of total quercetin increased from 0.61 ± 0.15 to 2.51 ± 0.65 μmol mmol−1 creatinine and total epicatechin from 0.50 ± 0.28 to 2.62 ± 0.98 μmol mmol−1 creatinine (p < 0.001). Plasma concentrations of EGCG increased from 0.06 ± 0.01 to 0.10 ± 0.01 μmol L−1 (p < 0.05). EGCG was not detected in urine. | EGCG did not affect NO production. Quercetin and epicatechin significantly reduced plasma endothelin-1 concentration (p < 0.05), but only quercetin significantly decreased the urinary endothelin-1 concentration. None of the 3 treatments significantly decreased plasma or urinary F2-isoprostane concentrations. | [49] |
| 15 healthy subjects (9 M; 6 F) (mean age 25.8 ± 5.2 years) Duration: 3 weeks Double blind, randomized, placebo-controlled trial. | Subjects received a capsule containing the following: - Placebo; - 200 mg of quercetin; - 400 mg of quercetin. | To evaluate whether the deconjugation of quercetin-3-O-glucuronide (Q3GA) may improve vasodilator effects of quercetin. | At 2 h post ingestion, plasma levels were as follows: - 200 mg quercetin group: 0.35 μM Q3GA, 0.043 μM quercetin aglycone, 0.008 μM isorhamnetin aglycone, - 400 mg quercetin group: 0.95 μM Q3GA, 0.031 μM quercetin aglycone, 0.035 μM isorhamnetin aglycone. Glucuronides of isorhamnetin were not detected. | After ingestion (2 or 5 h) of both doses, were not changes in systolic and diastolic blood pressure. A time-dependent increase in brachial artery diameter was detected after 400 mg quercetin intake, correlated with the levels of Q3GA mediated by glucuronidase activity. | [50] |
| Cohort and Study Details | Phenolic Acids Intake | Aim | Bioavailability Data | Outcome | Reference |
|---|---|---|---|---|---|
| 1° study: 15 healthy male subjects (mean age: 26.3 ± 1.6 years) 2° study: 24 healthy male subjects (mean age: 23.8 ± 1.4 years) Duration: acute consumption Randomized controlled crossover studies | 1° study: Subjects consumed 50 mL coffee containing high (310 mg) or low (89 mg) chlorogenic acid levels. Control intervention contained 0 mg chlorogenic acid. 2° study: Subjects consumed (a) 0.45 g purified 5-caffeoylquinic acid (5-CQA) + 1 g maltodextrin (MDX); (b) 0.90 g purified 5-CQA + 1 g MDX; 1 g MDX (negative control) and 0.20 g purified epicatechin + 1 g MDX (positive control). Each preparation was solubilized in 200 mL of hot water. | To evaluate the vascular function (% flow-mediated dilation, FMD) after the consumption of coffee rich in chlorogenic acid. | 1° study: After 1 h of treatment was observed, a significant positive correlation between total plasma chlorogenic acids metabolites (CgAM) and % FMD: 3-caffeoylquinic acid (3CQA) 4-caffeoylquinic acid (4CQA), 5-feruloylquinic acid (5FQA) and caffeic-3′-O-sulfate (CA3S) (p < 0.005). After 5 h, where a second peak of % FMD was observed, the correlation was significant for ferulic-4′-O-sulfate, isoferulic-3′-O-glucuronide, caffeic-4′-O-sulfate, m-coumaric acid-3′-O-sulfate and 4-methoxycinnamic acid (p < 0.005). 2° study: Total plasma CgAM post intervention of 0.45 g of chlorogenic acids were lower than following intake of the 0.90 g dose, but both significantly ↑ (p = 0.032 and p = 0.006, respectively). | 1° study: Both low and high chlorogenic enriched coffee improved vascular function 2° study: Only after the 0.45 g dose of purified 5-CQA, %FMD ↑ in comparison to the water control (6.02 ± 0.28 to 6.77 ± 0.42, p = 0.06), whereas intake of the control (5.90 ± 0.33% to 5.86 ± 0.30%) or the higher dose of 5-CQA (0.900 g) had no effect on %FMD (6.17 ± 0.31% to 6.46 ± 0.35%). | [55] |
| 23 healthy subjects (4 M; 19 F) (mean age: 52.3 ± 10.6 years) Duration: acute consumption Randomized, double-blind, placebo-controlled, crossover trial | The treatments included: - water (used as control); - 0.40 g of chlorogenic acid (3-O-caffeoylquinic acid) solubilized in 0.20 L of water (corresponding to 2 cups of coffee). | To investigate the acute effects of chlorogenic acid on different parameters: - Nitric oxide level; - Endothelial function; - Blood pressure. | Chlorogenic acid concentration in plasma was significantly higher 150 min after the consumption of 0.40 g of CGA as compared to control group (p < 0.001). No significant differences were observed in term of chlorogenic acid metabolites (isoferulic acid, ferulic acid, phloretic acid, caffeic acid, and hydrocaffeic acid) between the two treatments. | The mean systolic blood pressure (−2.41 mmHg, p = 0.05) and diastolic blood pressure (−1.53 mmHg, p = 0.04) were significantly ↓ in the chlorogenic acid group compared with control group. NO levels (p > 0.10) and endothelial function (p = 0.60) were not significantly affected. | [56] |
| 75 healthy subjects (38 M; 37 F) (mean age: 38.5 ± 9 years) Duration: 8 weeks Randomized, placebo-controlled trial | Group A: consumption 400 mL coffee/day containing high levels chlorogenic acids (HCCGA) (780 mg/400 mL); group B: (400 mL coffee/day containing medium levels chlorogenic acid (MCCGA) (420 mg/400 mL); group C: placebo. Chlorogenic acids were composed by: 5-O-caffeoylquinic; 3-O-caffeoylquinic; 4-O-caffeoylquinic; caffeic; ferulic; 3,4-di-O-caffeoyl-quinic; 3,5-di-O-caffeoylquinic; and 4,5-di-O-caffeoylquinic. | To evaluate the effects of chlorogenic acids on the following: - The antioxidant capacity of plasma; - The lipid profile in serum; - The vascular function (flow-mediated dilation-FMD, nitric oxide levels, blood pressure). | The concentration of ferulic and caffeic acid was ↑ in the groups that consumed coffee-drinking and were significantly higher 1 h after the consumption in the MCCGA group than the HCCGA group (caffeic acid: 50.5 ± 6.9 nM vs 20.3 ± 3.3 nM; ferulic acid: 201 ± 18.7 nM vs. 137 ± 6.1) (p < 0.001). In both groups the ferulic and caffeic acids decreased after 8 weeks. | Plasma antioxidant capacity significantly increased only in the group consuming medium levels of chlorogenic acid (6%) and group consuming high levels chlorogenic acids (5%) (p < 0.05). No effects on lipid profile and vascular function were measured. | [59] |
| 16 healthy subjects (10 F, 6 M) (mean age: 59.9 ± 8.2 years) Duration: acute consumption Double-blind, randomized, placebo-controlled crossover study | Subjects received: (1) 0 g purified 5-chlorogenic acid (p5-CgA) (control group); (2) 0.45 g p5-CgA; (3) 0.90 g p5-CgA; and (4) 0.20 g purified (−)-epicatechin (positive control). Each treatment was prepared in 200 mL of warm water and consumed within 10 min. A minimum one-week washouts between visits was required. | To evaluate the acute effect of two doses of (5-CgA) (0.45 and 0.90 g) on vascular function (FMD) and blood pressure. | After 1 h and 4 h from the consumption of 0.90 g 5-CgA, total CgA metabolites, reached 1.5 μM and 1.25 μM, respectively. After 0.45 g, metabolites reached 0.75 μM and 1 μM after 1 and 4 h, respectively. In both cases, the most representative CgA metabolite was 5-caffeoylquinic acid (5-CqA). | None of the doses of 5-CgA used significantly affected FMD response. | [63] |
| 12 healthy subjects (5 F, 7 M) (mean age: 59.4 ± 6.4 years) Duration: acute consumption Randomized, placebo-controlled, crossover trial | Subjects consumed: (1) 18 g of ground caffeinated coffee + 0.3 g CgA in 0.20 L of hot water; (2) 18 g of decaffeinated coffee + 0.287 g CgA in 0.20 L of hot water; (3) 0.20 L of hot water (control). For each group the beverages were consumed twice. The second beverage was consumed after 2 h with a 75 g glucose load. | The aim of the study was to evaluate the effect of coffee on different outcomes: (1) vascular function (FMD), (2) blood pressure, (3) glucose concentration in blood. | The mean values concentration for 5-CgA were: - caffeinated group: 1.89 ± 0.56 nM; - water group: 1.21 ± 0.22 nM; - decaffeinated group: 1.30 ± 0.29 nM. No significant difference in 5-CgA concentration between the three groups were observed. | The FMD response was significantly ↑ in the caffeinated coffee group compared to the other groups (decaffeinated coffee and water) (p < 0.001). No significant difference in the FMD response between decaffeinated coffee and water groups was observed. No differences were observed in term of blood glucose concentrations and blood pressure between the three groups considered. | [60] |
| 68 healthy overweight/obese subjects (23 M; 45 F) (mean age: 38.5 ± 2 years) with sedentary lifestyle and reduced intake of fruit and vegetables Duration: 8 weeks Randomized placebo-controlled, randomized trial | Whole grain (WG) group consumed 70 g wheat/day containing 96.7 mg ferulic acid, 26.5 mg sinapic acid, 9.4 mg coumaric acid, 1.9 mg gallic acid, 1.8 mg syringic acid, 1.6 mg vanillic acid, 0.5 mg salicylic acid, 0.3 mg caffeic acid); control group (CTR) consumed 60 g refined wheat (RW) products/day containing 2.6 mg ferulic acid. | To investigate the role of whole grain (WG) consumption on plasma markers of metabolic disease and inflammation (tumor necrosis factor-α (TNF-α) interleukin-10 (IL)-10), plasminogen activator inhibitor 1. | After 8 weeks, WG consumption was associated with a 4-fold ↑ in serum dihydroferulic acid (DHFA) and a 2-fold ↑ in fecal ferulic acid (FA) compared with RW group. Similarly, after 8 weeks, urinary FA was 2-fold ↑ the baseline concentration only in WG group. | In the WG group was observed a ↓ in TNF-α after 8 weeks and ↑ IL-10 only after 4 weeks, compared with RW group (p = 0.04). No significant differences in plasma metabolic disease markers were observed. | [62] |
| 47 habitual coffee drinkers at risk for type-2 diabetes (11 M; 36 F) (mean age 54.0 ± 9.0 years) Duration: 3 months Crossover clinical trial | The coffee consumption during the trial was set as follow: - First month: subjects avoided to drink coffee; - Second month: subjects consumed 4 cups (150 mL/cup) of filtered coffee/day; - Third month: participants consumed 8 cups of filtered coffee/day. | To evaluate the role of daily coffee consumption on the following: - The modulation of different biomarkers of inflammation (interleukin-18 (IL-18), 8-isoprostane, and adiponectin); - Oxidative stress and glucose; -Lipid metabolism. | Positive correlations between the ↑ in serum concentrations of coffee phenolic acid metabolites (cPAM) and changes in adiponectin concentrations were detected after 8 cups coffee/day consumption vs. baseline (0 cups coffee/day). Significant correlations were observed for isoferulic acid (r = 0.328, p = 0.025) and dihydroisoferulic acid (r = 0.323, p = 0.027). Negative or no correlation was observed for the other cPAM. | Significant differences were observed for serum concentrations of IL-18, 8-isoprostane, only after 8 cups coffee/day consumption compared with baseline (0 cups coffee/day). Serum concentrations of total cholesterol, HDL cholesterol, and apolipoprotein A-I ↑ significantly (+12%, 7%, and 4%, respectively). The ratios of LDL to HDL cholesterol and of apolipoprotein B to apolipoprotein A-I significantly ↓ (−8% and 9%, respectively) after the consumption of 8 cups coffee/day compared with 0 cups coffee/day. No variations were observed for glucose metabolism markers. | [64] |
| 20 healthy subjects (6 M; 14 F) (mean age: 35.7 ± 9.0 years) Duration: acute consumption Randomized placebo-controlled trial | 0.4 L of Arabica coffee containing 0.42 g CgAs (6 g/100 mL: provided 105 ± 4.1 mg of CgAs) were given to the intervention group The sum of the following acids contributed to the total CgAs and phenolic acids content: 5-O-caffeoylquinic; 3-O-caffeoylquinic; 4-O-caffeoylquinic; caffeic; ferulic; 3,4-di-O-caffeoylquinic; 3,5-di-O-caffeoylquinic; and 4,5-di-O-caffeoylquinic. No coffee or placebo were consumed by the control group. | The caffeic acid (CA) and ferulic acid (FA) were quantify and different methods were applied to evaluate the antioxidant capacity of plasma. | The concentrations of FA and CA, measured 1 h after the coffee consumption, were: 202.38 ± 12.87 nM and 49.76 ± 6.44 nM, respectively. Both acids (CA and FA) were not detected at baseline. In the control group, at 1 h, the same trend was observed. | The antioxidant capacity of plasma was measured with ferric reducing antioxidant power assay (FRAP) and oxygen radical absorbance capacity assay (ORAC). Compared to the baseline, the antioxidant capacity, measured with both methods, ↑ significantly (+6.67%; p < 0.001 for FRAP and +7.16%; p < 0.05 for ORAC). A correlation was observed between the ↑ of antioxidant capacity and the plasma concentration of FA and CA. | [65] |
| 21 healthy men (mean age: 27 ± 1.3) Duration: acute consumption Randomized, controlled, double-blind, crossover human- intervention trials | 1° study: participants consumed: - A blueberry drink containing: 0.766, 1.278, and 1.791 g total blueberry polyphenols (corresponding to 0.24, 0.40, and 0.56 kg fresh blueberries, respectively); - Control drink (macronutrient and micronutrient drink). 2° study: participants consumed the following: - A blueberry drink containing 0.319, 0.637, 0.766, 1.278 and 1.791 g total blueberry polyphenols (corresponding to 0.10, 0.20, 0.24, 0.40, and 0.56 kg fresh blueberries, respectively); - Control drink (macronutrient and micronutrient drink). | The endothelial function (FMD) was monitored and the time-dependent (1° study) and intake-dependent (2° study) changes were investigated. | A correlation between the FMD ↑ and the plasma concentration ↑ of different phenolic acid metabolites (ferulic acid, isoferulic acid, vanillic acid, 2-hydroxybenzoic acid, benzoic acid, and caffeic acid—sum of conjugated and nonconjugated compounds) were observed 2 and 6 h after consumption. The phenolic acid metabolites were significantly ↑ at 1–2 h after blueberry polyphenol consumption (p < 0.001). The plasma total concentration of metabolites was about 400 nmol/L (coinciding with the highest FMD value at 1 h). The metabolites of flavanol or anthocyanin were not detected in plasma at any time after the intake of blueberry drink. | The consumption of 0.10–0.24 kg blueberry (corresponding to 0.319, 0.639 and 0.766 g total polyphenols) positively affected vascular function. | [61] |
| Cohort and Study Details | Phenols Intake | Aim | Bioavailability Data | Outcome | Reference |
|---|---|---|---|---|---|
| |||||
| 60 subjects (9 M; 51 F) (mean age: 20.52 years) Duration: 28 days Randomized, double-blind, placebo-controlled, parallel-groups study | 500 mg/day of pure trans-resveratrol (also containing 10 mg of piperine/capsule) or a placebo. | To evaluate the effect of resveratrol on cognitive performance (measured as serial subtractions, rapid visual information processing, 3 Back test), mood, sleep quality and cerebral blood flow (CBF). | Resveratrol 3-O-sulfate was the predominant metabolite in all volunteers, contributing 73–77% of total metabolites, followed by resveratrol 4′ glucuronide and 3′ glucuronide. Total resveratrol metabolites increased in plasma from 3 to 13 μM 110 min after administration. | Although stilbene metabolite levels increase in plasma, supplementing with 500 mg of resveratrol for 28 days did not improve cognitive function. | [68] |
| 22 healthy subjects (4 M; 20 F) (mean age: 20.17 years) Duration: 7 days Randomized, double-blind, placebo-controlled, crossover study | Subjects received the following treatments: - inert placebo; - 250 mg trans-resveratrol; - 500 mg trans-resveratrol; | To evaluate the effects of oral resveratrol on cognitive performance and CBF. In a separate group (n = 9) was investigated plasma levels of resveratrol and its conjugates after the intake of the same treatments. | Resveratrol sulfate and glucuronide were the main metabolites and reached a peak plasma concentration at 90 min after both 250 and 500 mg supplementation. As regards unmetabolized resveratrol, 90 min after both supplementations, reached low concentrations, peaking at 5.65 and 14.4 ng mL−1, respectively. | CBF increased in a dose-dependent fashion of resveratrol intake during task performance. No changes in cognitive function were registered. | [69] |
| |||||
| 10 overweight or obese men (mean age: 56.2 ± 6.18 years) Duration: acute consumption Randomized, double-blind, placebo-controlled, crossover study | Subjects consumed a high-fat, high-fructose breakfast with 4 dietary supplementations: - Placebo: fish oil placebo and isoflavone placebo; - FO: fish oil and isoflavone placebo; - ISO: fish oil placebo and isoflavones; - FO + ISO: fish oil and isoflavones. The soy isoflavone supplements provided 150 mg glycoside isoflavones (eq. to 96 mg aglycone form) in proportions of 1.05/1.0/0.29 for genistein/daidzein/glycitein. Fish oil supplement (1 g of refined fish oil concentrate) providing 0.4 g EPA and 0.2 g DHA. | To evaluate the effect of acute supplementation with fish oil (n-3), PUFA, soy isoflavones, and their combination on postprandial serum triglycerides (TG) and oxidative biomarkers in a proatherogenic high-fat, high-fructose meal. | At 4 h, postprandially serum concentration was as follows: - Genistein ISO: 1.027 ± 0.122 μmol L−1, FO + ISO: 1.185 ± 0.079 μmol L−1; - Daidzein ISO: 0.838 ± 0.096 μmol L−1, FO + ISO: 1.017 ± 0.046 μmol L−1. | The high-fat, high-fructose meal significantly increased serum total FA and TG without affecting oxidative stress biomarkers. Serum TG and oxidative stress biomarkers did not differ between treatments. The FO and ISO were bioavailable but did not reduce the postprandial rise in serum TG. Neither the study meal nor the FO or ISO induced significant changes in oxidative stress. | [71] |
| 78 postmenopausal osteopenic women (mean age: 61.85 ± 1.03 years) Duration: 12 months Double-blind, parallel design, placebo-controlled trial | Participants received supplementation: 1.2 g/day calcium, 0.55 g/day magnesium, 0.025 g/day calcitriol, and a red clover extract (0.06 g/day isoflavone aglycones and probiotics) or a placebo. | To determine the beneficial effects of a bioavailable isoflavone and probiotic treatment in postmenopausal osteopenia. | After 12 month, isoflavone concentration in the treated group was 3.933 μg mL−1 (median), significantly higher from baseline (p = 0.0094) (compared with the control group, where median values were 2.323 μg mL−1). | Treatments with red clover extract: ↓ BMD loss, ↓ plasma concentrations of collagen type 1 crosslinked C-telopeptide (p < 0.05). | [72] |
| 237 women (mean age: 53 ± 3 years) Duration: 12 months Randomized, double-blind, placebo-controlled, parallel, multicenter trial (including the Netherlands, Italy and France) | Subjects, during their habitual diet and lifestyle, consumed 110 mg/day isoflavone aglycones or control. | To evaluate whether bone metabolism and mineral density, and hormonal conditions were affected by chronic consumption of isoflavone-enriched foods. | ↑ isoflavones plasma levels in treated group. Both genistein and daidzein were higher in the Netherlands (1522 ± 1136.2 nmol L−1 and 338.2 ± 261.3 nmol L−1, respectively) than in France (533.4 ± 607.4 nmol L−1 and 92.9 ± 145.8 nmol L−1, respectively) and Italy (541.5 ± 557.6 nmol L−1 and 133.4 ± 188.3 nmol L−1, respectively). | Bone mineral density or biomarkers of bone were not affected by isoflavone-enriched products chronic intake. Hormone concentrations did not differ between the two groups. | [73] |
| |||||
| 16 men at moderate CVD risk (mean age: 60.6 ± 1.4 years) Duration: acute consumption Randomized, placebo-controlled crossover trial | Participants received 767 mL orange juice or a hesperidin supplement (both providing 320 mg hesperidin and 439 mg vitamin C) or control. | To evaluate the effects of orange juice or a hesperidin supplement on plasma concentrations of flavanone metabolites and their effects on cardiovascular risk biomarkers (blood pressure, endothelial function, central arterial stiffness, cardiac autonomic function, platelet activation, and NADPH oxidase gene expression). | After 5 h from the orange juice intake, significantly increased plasma concentrations of 8 flavanones (1.75 ± 0.35 mmol L−1, p < 0.0001), and 15 other phenolic metabolites (13.27 ± 2.22 mmol L−1, p < 0.0001) were significantly increased. In particular, 47% hesperidin-glucuronide, 15% naringenin-7-O-glucuronide, 14% a second hesperidin-glucuronide, 54% hippuric acid, 15% dihydroferulic acid, 8% dihydroferulic acid–glucuronide, 7% 4-hydroxyphenylacetic acid, and 5% vanillic acid increase were detected. | Effects on CVD risk factor were not observed. | [74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Lorenzo, C.; Colombo, F.; Biella, S.; Stockley, C.; Restani, P. Polyphenols and Human Health: The Role of Bioavailability. Nutrients 2021, 13, 273. https://doi.org/10.3390/nu13010273
Di Lorenzo C, Colombo F, Biella S, Stockley C, Restani P. Polyphenols and Human Health: The Role of Bioavailability. Nutrients. 2021; 13(1):273. https://doi.org/10.3390/nu13010273
Chicago/Turabian StyleDi Lorenzo, Chiara, Francesca Colombo, Simone Biella, Creina Stockley, and Patrizia Restani. 2021. "Polyphenols and Human Health: The Role of Bioavailability" Nutrients 13, no. 1: 273. https://doi.org/10.3390/nu13010273
APA StyleDi Lorenzo, C., Colombo, F., Biella, S., Stockley, C., & Restani, P. (2021). Polyphenols and Human Health: The Role of Bioavailability. Nutrients, 13(1), 273. https://doi.org/10.3390/nu13010273






