Association between Breakfast Skipping and Body Weight—A Systematic Review and Meta-Analysis of Observational Longitudinal Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Data Extraction and Quality Assessment of Included Studies
2.3. Statistical Analysis
3. Results
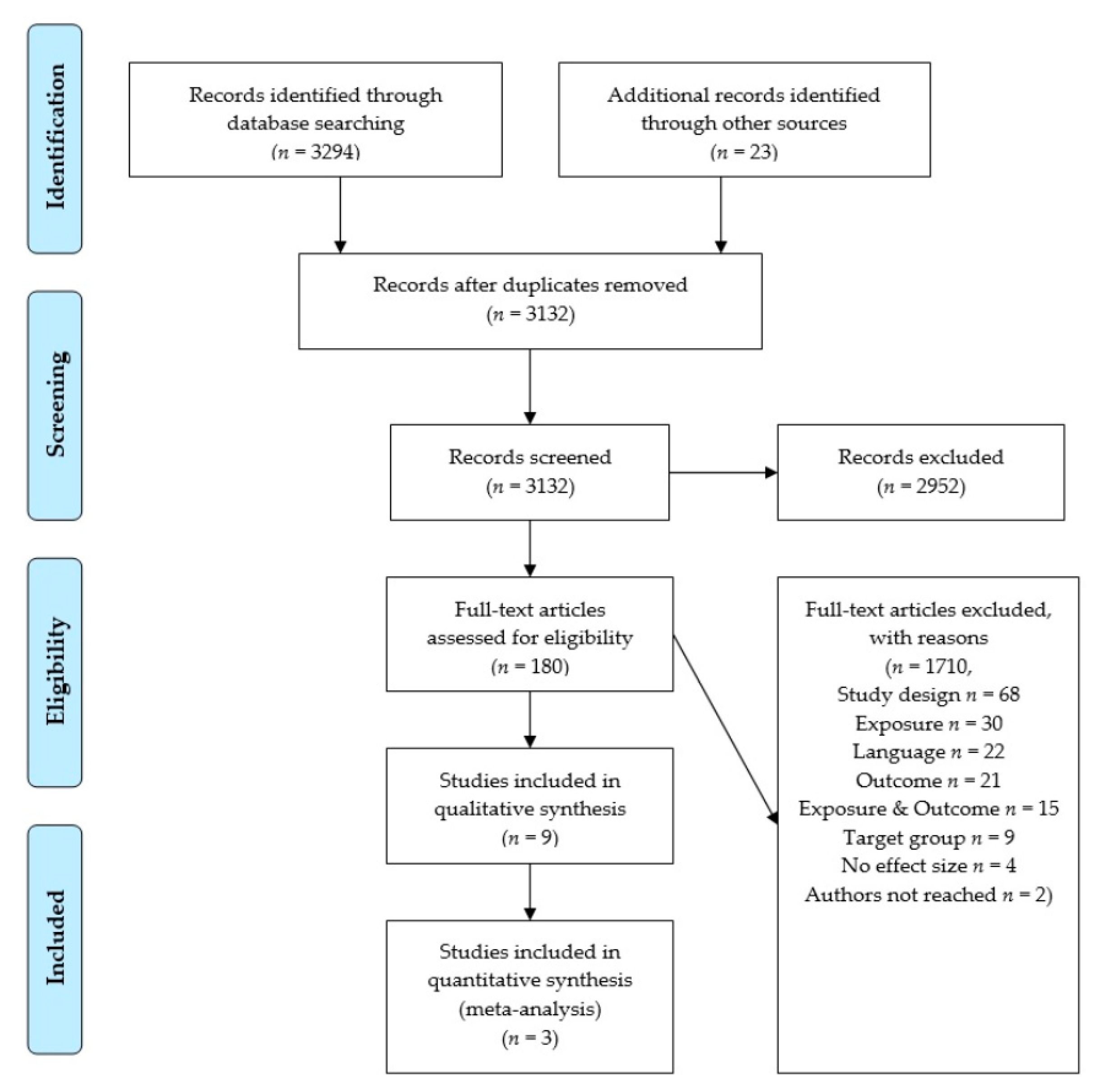
3.1. Identified Literature and Characteristics of Included Studies
3.2. Association between Breakfast Skipping and Body Weight
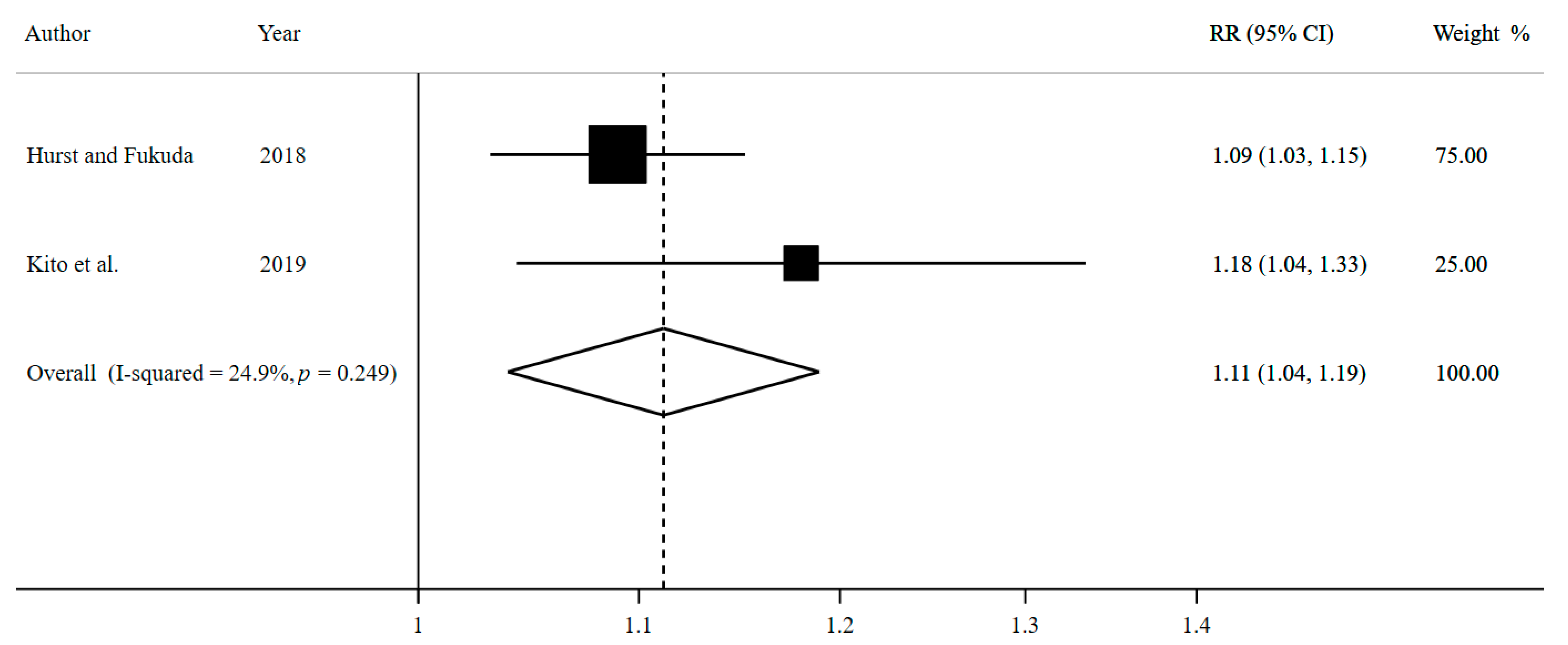
3.2.1. Association between Breakfast Skipping and Overweight/Obesity
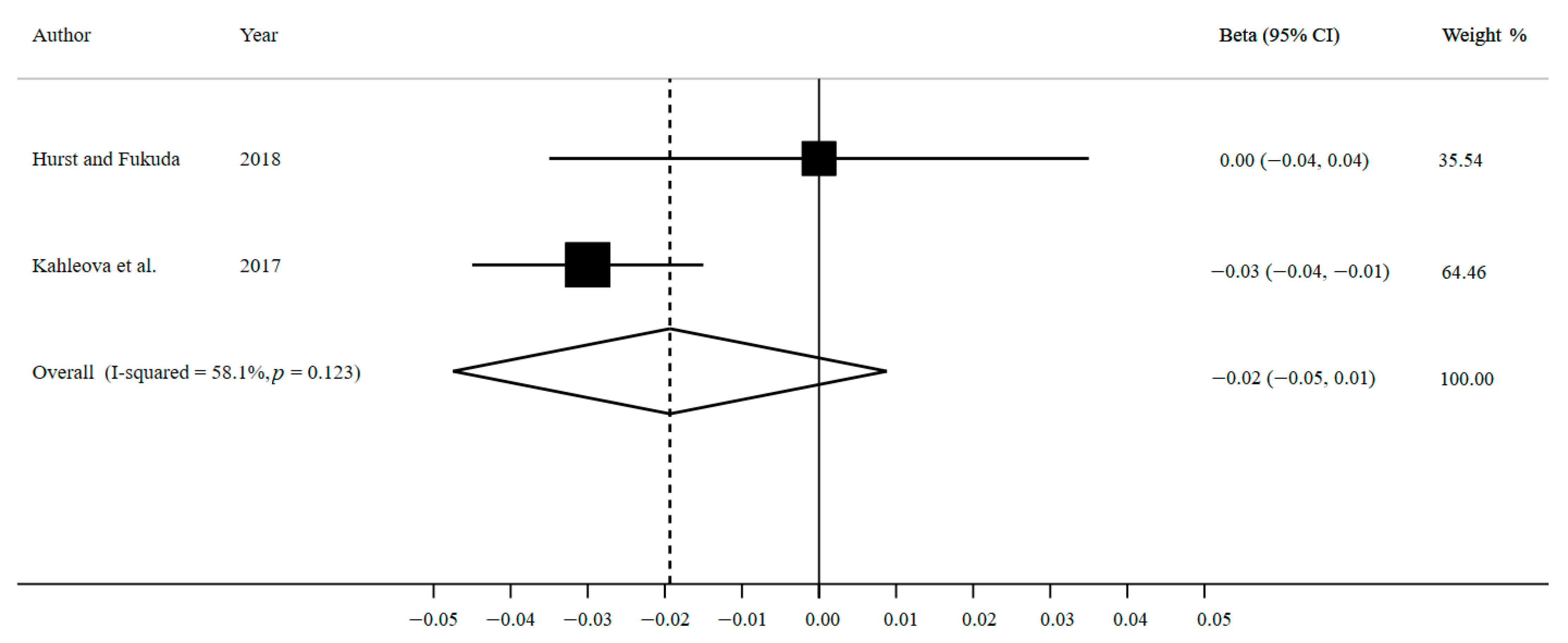
3.2.2. Association between Breakfast Skipping and BMI Change
3.3. Quality of Included Studies
4. Discussion
4.1. Limitations
4.2. Strengths and Further Research Needs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 19 January 2021).
- Gurevich-Panigrahi, T.; Panigrahi, S.; Wiechec, E.; Los, M. Obesity: Pathophysiology and clinical management. Curr. Med. Chem. 2009, 16, 506–521. [Google Scholar] [CrossRef] [PubMed]
- World Obesity Federation. Obesity: Missing the 2025 Global Targets. Trends, Costs and Country Reports. 2020. Available online: https://data.worldobesity.org/publications/WOF-Missing-the-2025-Global-Targets-Report-FINAL-WEB.pdf (accessed on 30 December 2020).
- World Obesity Federation. Women and Men Living with Obesity, Newest Available Data. Available online: https://data.worldobesity.org/maps/ (accessed on 30 December 2020).
- Visscher, T.; Seidell, J.C. The Public Health Impact of Obesity. Annu. Rev. Public Health 2001, 22, 355–375. [Google Scholar] [CrossRef] [PubMed]
- NCD Risk Factor Collaboration. Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19·2 million participants. Lancet 2016, 1377–1396. [Google Scholar] [CrossRef]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- Bray, G.A. Medical consequences of obesity. J. Clin. Endocrinol. Metab. 2004, 89, 2583–2589. [Google Scholar] [CrossRef]
- Tchernof, A.; Després, J.-P. Pathophysiology of human visceral obesity: An update. Physiol. Rev. 2013, 93, 359–404. [Google Scholar] [CrossRef]
- Djalalinia, S.; Qorbani, M.; Peykari, N.; Kelishadi, R. Health impacts of Obesity. Pak. J. Med. Sci. 2015, 31, 239–242. [Google Scholar] [CrossRef]
- Agha, M.; Agha, R. The rising prevalence of obesity: Part A: Impact on public health. Int. J. Surg. Oncol. (N. Y.) 2017, 2, e17. [Google Scholar] [CrossRef]
- Tune, J.D.; Goodwill, A.G.; Sassoon, D.J.; Mather, K.J. Cardiovascular consequences of metabolic syndrome. Transl. Res. 2017, 183, 57–70. [Google Scholar] [CrossRef]
- Freedland, S.J.; Wen, J.; Wuerstle, M.; Shah, A.; Lai, D.; Moalej, B.; Atala, C.; Aronson, W.J. Obesity is a significant risk factor for prostate cancer at the time of biopsy. Urology 2008, 72, 1102–1105. [Google Scholar] [CrossRef]
- van Baal, P.; Polder, J.J.; de Wit, G.A.; Hoogenveen, R.T.; Feenstra, T.L.; Boshulzen, H.C.; Engelfriet, P.M.; Brouwer, W. Lifetime Medical Costs of Obesity: Prevention No Cure for Increasing Health Expenditure. PLoS Med. 2008, 5, e29. [Google Scholar] [CrossRef] [PubMed]
- Tremmel, M.; Gerdtham, U.-G.; Nilsson, P.M.; Saha, S. Economic Burden of Obesity: A Systematic Literature Review. Int. J. Environ. Res. Public Health 2017, 14, 435. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.D.; Basu, A. Estimating the Medical Care Costs of Obesity in the United States: Systematic Review, Meta-Analysis, and Empirical Analysis. Value Health 2016, 19, 602–613. [Google Scholar] [CrossRef] [PubMed]
- Kjellberg, J.; Larsen, A.T.; Ibsen, R.; Højgaard, B. The Socioeconomic Burden of Obesity. Obes. Facts 2017, 10, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Boissonnet, C.; Schargrodsky, H.; Pellegrini, F.; Macchia, A.; Champagne, B.M.; Wilson, E.; Tognoni, G. Educational inequalities in obesity, abdominal obesity, and metabolic syndrome in seven Latin American cities: The CARMELA Study. Eur. J. Cardiovasc. Prev. Rehabil. 2011, 18, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Devaux, M.; Sassi, F. Social inequalities in obesity and overweight in 11 OECD countries. Eur. J. Public Health 2013, 23, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A. Obesity, diets, and social inequalities. Nutr. Rev. 2009, 67 (Suppl. 1), S36–S39. [Google Scholar] [CrossRef]
- World Health Organization. Global Action Plan for the Prevention and Control of Noncommunicable Diseases 2013–2020. 2013. Available online: https://apps.who.int/iris/bitstream/handle/10665/94384/9789241506236_eng.pdf;jsessionid=1D8EA26C28A4F9BCA3A7A10D86569ED1?sequence=1 (accessed on 30 December 2020).
- United Nations. Sustainable Development Goals: Goal 3: Ensure Healthy Lives and Promote Well-Being for All at All Ages. Available online: https://sdgs.un.org/goals/goal3 (accessed on 19 January 2021).
- Jo, J.; Gavrilova, O.; Pack, S.; Jou, W.; Mullen, S.; Sumner, A.E.; Cushman, S.W.; Periwal, V. Hypertrophy and/or Hyperplasia: Dynamics of Adipose Tissue Growth. PLoS Comput. Biol. 2009, 5, e1000324. [Google Scholar] [CrossRef]
- Spiegelman, B.M.; Flier, J.S. Obesity and the Regulation of Energy Balance. Cell 2001, 104, 531–543. [Google Scholar] [CrossRef]
- Jungermann, K.; Barth, C.A. Energy Metabolism and Nutrition. In Comprehensive Human Physiology: From Cellular Mechanisms to Integration; Greger, R., Windhorst, U., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 1425–1457. ISBN 978-3-642-64619-5. [Google Scholar]
- Duren, D.L.; Sherwood, R.J.; Czerwinski, S.A. Body Composition Methods: Comparisons and Interpretation. J. Diabetes Sci. Technol. 2008, 2, 1139–1146. [Google Scholar] [CrossRef]
- Wells, J.C.K.; Fewtrell, M.S. Measuring body composition. Arch. Dis. Child. 2006, 91, 612–617. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Body Mass Index—BMI. Available online: http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 1 August 2019).
- World Health Organization. Waist Circumference and Waist-Hip Ratio. Report of a WHO Expert Consultation; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Ashwell, M.; Gibson, S. Waist-to-height ratio as an indicator of ‘early health risk’: Simpler and more predictive than using a ‘matrix’ based on BMI and waist circumference. BMJ Open 2016, 6, e010159. [Google Scholar] [CrossRef] [PubMed]
- Gibney, M.J.; Barr, S.I.; Bellisle, F.; Drewnowski, A.; Fagt, S.; Livingstone, B.; Masset, G.; Varela Moreiras, G.; Moreno, L.A.; Smith, J.; et al. Breakfast in Human Nutrition: The International Breakfast Research Initiative. Nutrients 2018, 10, 559. [Google Scholar] [CrossRef] [PubMed]
- Gwin, J.A.; Leidy, H.J. A Review of the Evidence Surrounding the Effects of Breakfast Consumption on Mechanisms of Weight Management. Adv. Nutr. 2018, 9, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Timlin, M.T.; Pereira, M.A. Breakfast frequency and quality in the etiology of adult obesity and chronic diseases. Nutr. Rev. 2007, 65, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Keski-Rahkonen, A.; Kaprio, J.; Rissanen, A.; Virkkunen, M.; Rose, R.J. Breakfast skipping and health-compromising behaviors in adolescents and adults. Eur. J. Clin. Nutr. 2003, 57, 842–853. [Google Scholar] [CrossRef]
- Vereecken, C.; Dupuy, M.; Rasmussen, M.; Kelly, C.; Nansel, T.R.; Sabbah, H.; Baldassari, D.; Jordan, M.D.; Maes, L.; Niclasen, B.V.-L.; et al. Breakfast consumption and its socio-demographic and lifestyle correlates in schoolchildren in 41 countries participating in the HBSC study. Int. J. Public Health 2009, 54 (Suppl. 2), 180–190. [Google Scholar] [CrossRef]
- Goto, M.; Kiyohara, K.; Kawamura, T. Lifestyle risk factors for overweight in Japanese male college students. Public Health Nutr. 2010, 13, 1575–1580. [Google Scholar] [CrossRef]
- Hurst, Y.; Fukuda, H. Effects of changes in eating speed on obesity in patients with diabetes: A secondary analysis of longitudinal health check-up data. BMJ Open 2018, 8, e019589. [Google Scholar] [CrossRef]
- Kito, K.; Kuriyama, A.; Takahashi, Y.; Nakayama, T. Impacts of skipping breakfast and late dinner on the incidence of being overweight: A 3-year retrospective cohort study of men aged 20–49 years. J. Hum. Nutr. Diet. 2019. [Google Scholar] [CrossRef]
- Smith, K.J.; Gall, S.L.; McNaughton, S.A.; Cleland, V.J.; Otahal, P.; Dwyer, T.; Venn, A.J. Lifestyle behaviours associated with 5-year weight gain in a prospective cohort of Australian adults aged 26–36 years at baseline. BMC Public Health 2017, 17, 54. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh-Taskar, P.; Nicklas, T.A.; Radcliffe, J.D.; O’Neil, C.E.; Liu, Y. The relationship of breakfast skipping and type of breakfast consumed with overweight/obesity, abdominal obesity, other cardiometabolic risk factors and the metabolic syndrome in young adults. The National Health and Nutrition Examination Survey (NHANES): 1999–2006. Public Health Nutr. 2013, 16, 2073–2082. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.-J.; Lee, Y.; Lee, S.; Choi, K. Breakfast skipping and breakfast type are associated with daily nutrient intakes and metabolic syndrome in Korean adults. Nutr. Res. Pract. 2015, 9, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Parikh, N.I.; Pencina, M.J.; Wang, T.J.; Lanier, K.J.; Fox, C.S.; D’Agostino, R.B.; Vasan, R.S. Increasing trends in incidence of overweight and obesity over 5 decades. Am. J. Med. 2007, 120, 242–250. [Google Scholar] [CrossRef]
- World Health Organization. Global Health Observatory Data Repository: Prevalence of Overweight among Adults. Available online: http://gamapserver.who.int/gho/interactive_charts/ncd/risk_factors/overweight/atlas.html (accessed on 5 September 2019).
- World Health Organization. Prevalence of Obesity among Adults: Interactive Charts at Map Server. Available online: http://gamapserver.who.int/gho/interactive_charts/ncd/risk_factors/obesity/atlas.html (accessed on 5 September 2019).
- U.S. Department of Health and Human Services. National Cholesterol Education Programme Adult Treatment Panel III (NCEP ATP III) Guidelines; U.S. Department of Health and Human Services: Washington, DC, USA, 2001.
- Menche, N. (Ed.) Biologie, Anatomie, Physiologie. Kompaktes Lehrbuch für Pflegeberufe, 7th ed.; Elsevier: München, Germany, 2012; ISBN 978-3-437-26802-1. [Google Scholar]
- Hammond, R.A.; Levine, R. The economic impact of obesity in the United States. Diabetes Metab. Syndr. Obes. 2010, 3, 285–295. [Google Scholar] [CrossRef]
- Seidell, J.C.; Kahn, H.S.; Williamson, D.F.; Lissner, L.; Valdez, R. Report from a Centers for Disease Control and Prevention Workshop on use of adult anthropometry for public health and primary health care. Am. J. Clin. Nutr. 2001, 73, 123–126. [Google Scholar] [CrossRef]
- Albataineh, S.R.; Badran, E.F.; Tayyem, R.F. Dietary factors and their association with childhood obesity in the Middle East: A systematic review. Nutr. Health 2018, 25, 53–60. [Google Scholar] [CrossRef]
- Blondin, S.A.; Anzman-Frasca, S.; Djang, H.C.; Economos, C.D. Breakfast consumption and adiposity among children and adolescents: An updated review of the literature. Pediatr. Obes. 2016, 11, 333–348. [Google Scholar] [CrossRef]
- Gebremariam, M.K.; Lien, N.; Nianogo, R.A.; Arah, O.A. Mediators of socioeconomic differences in adiposity among youth: A systematic review. Obes. Rev. 2017, 18, 880–898. [Google Scholar] [CrossRef]
- Ma, X.; Chen, Q.; Pu, Y.; Guo, M.; Jiang, Z.; Huang, W.; Long, Y.; Xu, Y. Skipping breakfast is associated with overweight and obesity: A systematic review and meta-analysis. Obes. Res. Clin. Pract. 2020, 14, 1–8. [Google Scholar] [CrossRef]
- Ghafari, M.; Doosti-Irani, A.; Amiri, M.; Cheraghi, Z. Prevalence of the Skipping Breakfast among the Iranian Students: A Review Article. Iran. J. Public Health 2017, 46, 882–889. [Google Scholar] [PubMed]
- Hopkins, L.C.; Gunther, C. A Historical Review of Changes in Nutrition Standards of USDA Child Meal Programs Relative to Research Findings on the Nutritional Adequacy of Program Meals and the Diet and Nutritional Health of Participants: Implications for Future Research and the Summer Food Service Program. Nutrients 2015, 7, 10145–10167. [Google Scholar] [CrossRef] [PubMed]
- Minges, K.E.; Whittemore, R.; Grey, M. Overweight and obesity in youth with type 1 diabetes. Ann. Rev. Nurs. Res. 2013, 31, 47–69. [Google Scholar] [CrossRef]
- Monzani, A.; Ricotti, R.; Caputo, M.; Solito, A.; Archero, F.; Bellone, S.; Prodam, F. A Systematic Review of the Association of Skipping Breakfast with Weight and Cardiometabolic Risk Factors in Children and Adolescents. What Should We Better Investigate in the Future? Nutrients 2019, 11, 387. [Google Scholar] [CrossRef]
- Patro, B.; Szajewska, H. Meal patterns and childhood obesity. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 300–304. [Google Scholar] [CrossRef]
- Rampersaud, G.C.; Pereira, M.A.; Girard, B.L.; Adams, J.; Metzl, J.D. Review—Breakfast habits, nutritional status, body weight, and academic performance in children and adolescents. J. Am. Diet. Assoc. 2005, 105, 743–760. [Google Scholar] [CrossRef] [PubMed]
- Rohana, A.J.; Aiba, N. Childhood Obesity in Japan: A Growing Public Health Threat. Int. Med. J. 2012, 19, 146–149. [Google Scholar]
- Szajewska, H.; Ruszczynski, M. Systematic review demonstrating that breakfast consumption influences body weight outcomes in children and adolescents in Europe. Crit. Rev. Food Sci. Nutr. 2010, 50, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.J.; Henley, W.E.; Williams, C.A.; Hurst, A.J.; Logan, S.; Wyatt, K.M. Systematic review and meta-analysis of the association between childhood overweight and obesity and primary school diet and physical activity policies. Phys. Act. 2013, 10, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, J.L.J.; Stanford, J.B. Preventing or improving obesity by addressing specific eating patterns. J. Am. Board Fam. Med. 2008, 21, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, J.P.; Cardel, M.I.; Cellini, J.; Hu, F.B.; Guasch-Ferré, M. Breakfast Skipping, Body Composition, and Cardiometabolic Risk: A Systematic Review and Meta-Analysis of Randomized Trials. Obesity (Silver Spring) 2020, 28, 1098–1109. [Google Scholar] [CrossRef] [PubMed]
- Stroup, D.F.; Berlin, J.A.; Morton, S.C.; Olkin, I.; Williamson, G.D.; Rennie, D.; Moher, D.; Becker, B.J.; Sipe, T.A.; Thacker, S.B.; et al. Meta-analysis of Observational Studies in EpidemiologyA Proposal for Reporting. JAMA 2000, 283, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.; Higgins, J.; Elbers, R.G.; Reeves, B.C.; The Development Group for ROBINS-I. Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I): Detailed Guidance. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef] [PubMed]
- Olatona, F.A.; Onabanjo, O.O.; Ugbaja, R.N.; Nnoaham, K.E.; Adelekan, D.A. Dietary habits and metabolic risk factors for non-communicable diseases in a university undergraduate population. J. Health Popul. Nutr. 2018, 37, 21. [Google Scholar] [CrossRef]
- Batista-Jorge, G.C.; Barcala-Jorge, A.S.; Dias, A.F.O.; Silveira, M.F.; Lelis, D.D.F.; Andrade, J.M.O.; Claro, R.M.; De Paula, A.M.B.; Guimaraes, A.L.S.; Ferreira, A.; et al. Nutritional Status Associated to Skipping Breakfast in Brazilian Health Service Patients. Ann. Nutr. Metab. 2016, 69, 31–40. [Google Scholar] [CrossRef]
- Ball, K.; Mishra, G.D.; Crawford, D. Social factors and obesity: An investigation of the role of health behaviours. Int. J. Obes. 2003, 27, 394–403. [Google Scholar] [CrossRef]
- Bjørnarå, H.B.; Vik, F.N.; Brug, J.; Manios, Y.; De Bourdeaudhuij, I.; Jan, N.; Maes, L.; Moreno, L.A.; Dössegger, A.; Bere, E. The association of breakfast skipping and television viewing at breakfast with weight status among parents of 10–12-year-olds in eight European countries; the ENERGY (EuropeaN Energy balance Research to prevent excessive weight Gain among Youth) cross-sectional study. Public Health Nutr. 2013, 17, 906–914. [Google Scholar] [CrossRef]
- Carels, R.A.; Young, K.M.; Coit, C.; Clayton, A.M.; Spencer, A.; Wagner, M. Skipping meals and alcohol consumption. The regulation of energy intake and expenditure among weight loss participants. Appetite 2008, 51, 538–545. [Google Scholar] [CrossRef]
- Fayet-Moore, F.; McConnell, A.; Cassettari, T.; Petocz, P. Breakfast Choice Is Associated with Nutrient, Food Group and Discretionary Intakes in Australian Adults at Both Breakfast and the Rest of the Day. Nutrients 2019, 11, 175. [Google Scholar] [CrossRef]
- Fransen, H.P.; Boer, J.M.; Beulens, J.W.; De Wit, G.A.; Bueno-De-Mesquita, H.B.; Hoekstra, J.; May, A.M.; Peeters, P.H. Associations between lifestyle factors and an unhealthy diet. Eur. J. Public Health 2016, 27, ckw190. [Google Scholar] [CrossRef]
- Heinrich, K.M.; Maddock, J. Multiple health behaviors in an ethnically diverse sample of adults with risk factors for cardiovascular disease. Perm. J. 2011, 15, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.-J.; Hu, H.-T.; Fan, Y.-C.; Liao, Y.-M.; Tsai, P.-S. Associations of breakfast skipping with obesity and health-related quality of life: Evidence from a national survey in Taiwan. Int. J. Obes. 2010, 34, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K.; Andon, M.B.; Angelopoulos, T.J.; Rippe, J.M. Association of breakfast energy density with diet quality and body mass index in American adults: National Health and Nutrition Examination Surveys, 1999–2004. Am. J. Clin. Nutr. 2008, 88, 1396–1404. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, M.; Hasani-Ranjbar, S.; Yaghubi, H.; Rahmani, J.; Tabrizi, Y.M.; Keshtkar, A.; Varmaghani, M.; Sharifi, F.; Sadeghi, O. Breakfast consumption pattern and its association with overweight and obesity among university students: A population-based study. Eat. Weight Disord. 2018, 25, 379–387. [Google Scholar] [CrossRef]
- Nishiyama, M.; Muto, T.; Minakawa, T.; Shibata, T. The Combined Unhealthy Behaviors of Breakfast Skipping and Smoking Are Associated with the Prevalence of Diabetes Mellitus. Tohoku J. Exp. Med. 2009, 218, 259–264. [Google Scholar] [CrossRef]
- Park, S.H.; Jang, S.Y.; Kim, H.; Lee, S.W. An association rule mining-based framework for understanding lifestyle risk behaviors. PLoS ONE 2014, 9, e88859. [Google Scholar] [CrossRef]
- Smith, K.J.; McNaughton, S.A.; Cleland, V.J.; Crawford, D.; Ball, K. Health, Behavioral, Cognitive, and Social Correlates of Breakfast Skipping among Women Living in Socioeconomically Disadvantaged Neighborhoods. J. Nutr. 2013, 143, 1774–1784. [Google Scholar] [CrossRef]
- Yasuda, J.; Asako, M.; Arimitsu, T.; Fujita, S. Skipping breakfast is associated with lower fat-free mass in healthy young subjects: A cross-sectional study. Nutr. Res. 2018, 60, 26–32. [Google Scholar] [CrossRef]
- Wannamethee, S.G.; Field, A.E.; Colditz, G.A.; Rimm, E.B. Alcohol Intake and 8-Year Weight Gain in Women: A Prospective Study. Obes. Res. 2004, 4, 1386–1396. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Knüppel, S.; Schwedhelm, C.; Hoffmann, G.; Missbach, B.; Stelmach-Mardas, M.; Dietrich, S.; Eichelmann, F.; Kontopantelis, E.; Iqbal, K.; et al. Perspective: NutriGrade: A Scoring System to Assess and Judge the Meta-Evidence of Randomized Controlled Trials and Cohort Studies in Nutrition Research. Adv. Nutr. 2016, 7, 994–1004. [Google Scholar] [CrossRef]
- Borenstein, M.; Hedges, L.V.; Higgins, J.; Rothstein, H.R. Introduction to Meta-Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Cleophas, T.J.; Zwinderman, A.H. Modern Meta-Analysis. Review and Update of Methodologies; Springer: Cham, Switzerland, 2017; ISBN 978-3-319-55895-0. [Google Scholar]
- DerSimonian, R.; Laird, N. Meta-Analysis in Clinical Trials. Control. Clin. Trials 1986, 177–188. [Google Scholar] [CrossRef]
- Bender, R.; Friede, T.; Koch, A.; Kuss, O.; Schlattmann, P.; Schwarzer, G.; Skipka, G. Methods for evidence synthesis in the case of very few studies. Res. Synth. Methods 2018, 382–392. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.; Green, S. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; Version 5.1.0; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Guinter, M.A.; Park, Y.-M.; Steck, S.E.; Sandler, D.P. Day-to-day regularity in breakfast consumption is associated with weight status in a prospective cohort of women. Int. J. Obes. 2020, 44, 186–194. [Google Scholar] [CrossRef]
- Odegaard, A.O.; Jacobs, D.R.; Steffen, L.M.; Van Horn, L.; Ludwig, D.S.; Pereira, M.A. Breakfast Frequency and Development of Metabolic Risk. Diabetes Care 2013, 36, 3100–3106. [Google Scholar] [CrossRef]
- Nooyens, A.C.J.; Visscher, T.L.S.; Schuit, A.J.; van Rossum, C.T.M.; Verschuren, W.M.M.; van Mechelen, W.; Seidell, J.C. Effects of retirement on lifestyle in relation to changes in weight and waist circumference in Dutch men: A prospective study. Public Health Nutr. 2005, 8, 1266–1274. [Google Scholar] [CrossRef]
- Kahleova, H.; Lloren, J.I.; Mashchak, A.; Hill, M.; Fraser, G.E. Meal frequency and timing are associated with changes in body mass index in adventist health study 2. J. Nutr. 2017, 147, 1722–1728. [Google Scholar] [CrossRef]
- Van der Heijden, A.A.; Hu, F.B.; Rimm, E.B.; van Dam, R.M. A prospective study of breakfast consumption and weight gain among U.S. men. Obesity (Silver Spring) 2007, 15, 2463–2469. [Google Scholar] [CrossRef]
- Horikawa, C.; Kodama, S.; Yachi, Y.; Heianza, Y.; Hirasawa, R.; Ibe, Y.; Saito, K.; Shimano, H.; Yamada, N.; Sone, H. Skipping breakfast and prevalence of overweight and obesity in Asian and Pacific regions: A meta-analysis. Prev. Med. 2011, 53, 260–267. [Google Scholar] [CrossRef]
- Sievert, K.; Hussain, S.M.; Page, M.J.; Wang, Y.; Hughes, H.J.; Malek, M.; Cicuttini, F.M. Effect of breakfast on weight and energy intake: Systematic review and meta-analysis of randomised controlled trials. BMJ 2019, 364, l42. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, G.M.; La Bounty, P.M. Effects of intermittent fasting on body composition and clinical health markers in humans. Nutr. Rev. 2015, 73, 661–674. [Google Scholar] [CrossRef]
- Santos, H.O.; Macedo, R.C.O. Impact of intermittent fasting on the lipid profile: Assessment associated with diet and weight loss. Clin. Nutr. ESPEN 2018, 24, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Deutsche Gesellschaft für Ernährung e.V. Heilfasten, Basenfasten, Intervallfasten—Ein Überblick. DGEinfo 2018, 2, 18–25. [Google Scholar]
- Stote, K.S.; Baer, D.J.; Spears, K.; Paul, D.R.; Harris, G.K.; Rumpler, W.F. A controlled trial of reduced meal frequency without caloric restriction in healthy, normal-weight, middle-aged adults. Am. J. Clin. Nutr. 2007, 4, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Moro, T.; Tinsley, G.; Bianco, A.; Marcolin, G.; Pacelli, Q.F.; Battaglia, G.; Palma, A.; Gentil, P.; Neri, M.; Paoli, A. Effects of eight weeks of time-restricted feeding (16/8) on basal metabolism, maximal strength, body composition, inflammation, and cardiovascular risk factors in resistance-trained males. J. Transl. Med. 2016, 14, 57. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.M.B.; Milanes, J.E.; Allison, D.B.; Brown, A.W. Eating compared to skipping breakfast has no discernible benefit for obesity-related anthropometrics: Systematic review and meta-analysis of randomized controlled trials. FASEB J. 2017, 31, lb363. [Google Scholar]
- Goldstone, A.P.; De Hernandez, C.G.P.; Beaver, J.D.; Muhammed, K.; Croese, C.; Bell, G.; Durighel, G.; Hughes, E.; Waldman, A.D.; Frost, G.; et al. Fasting biases brain reward systems towards high-calorie foods. Eur. J. Neurosci. 2009, 30, 1625–1635. [Google Scholar] [CrossRef]
- De Castro, J.M. The time of day of food intake influences overall intake in humans. J. Nutr. 2004, 134, 104–111. [Google Scholar] [CrossRef]
- De Castro, J.M. When, how much and what foods are eaten are related to total daily food intake. Br. J. Nutr. 2009, 102, 1228–1237. [Google Scholar] [CrossRef]
- Azadbakht, L.; Haghighatdoost, F.; Feizi, A.; Esmaillzadeh, A. Breakfast eating pattern and its association with dietary quality indices and anthropometric measurements in young women in Isfahan. Nutrition 2013, 29, 420–425. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Saito, I.; Henmi, I.; Yoshimura, K.; Maruyama, K.; Yamauchi, K.; Matsuo, T.; Kato, T.; Tanigawa, T.; Kishida, T.; et al. Skipping Breakfast is Correlated with Obesity. J. Rural Med. 2014, 9, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Dietrich, M.; Brown, C.J.P.; Clark, C.A.; Block, G. The effect of breakfast type on total daily energy intake and body mass index: Results from the Third National Health and Nutrition Examination Survey (NHANES III). J. Am. Coll. Nutr. 2003, 22, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Kent, L.M.; Worsley, A. Breakfast size is related to body mass index for men, but not women. Nutr. Res. 2010, 30, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh-Taskar, P.R.; Radcliffe, J.D.; Liu, Y.; Nicklas, T.A. Do Breakfast Skipping and Breakfast Type Affect Energy Intake, Nutrient Intake, Nutrient Adequacy, and Diet Quality in Young Adults? NHANES 1999–2002. J. Am. Coll. Nutr. 2010, 29, 407–418. [Google Scholar] [CrossRef]
- Albertson, A.M.; Wold, A.C.; Joshi, N. Ready-to-Eat Cereal Consumption Patterns: The Relationship to Nutrient Intake, Whole Grain Intake, and Body Mass Index in an Older American Population. J. Aging Res. 2012, 2012, 631310. [Google Scholar] [CrossRef]
- Aparicio, A.; Rodriguez-Rodriguez, E.E.; Aranceta-Bartrina, J.; Gil, A.; Gonzalez-Gross, M.; Serra-Majem, L.; Varela-Moreiras, G.; Ortega, R.M. Differences in meal patterns and timing with regard to central obesity in the ANIBES (‘Anthropometric data, macronutrients and micronutrients intake, practice of physical activity, socioeconomic data and lifestyles in Spain’) Study. Public Health Nutr. 2017, 20, 2364–2373. [Google Scholar] [CrossRef]
- Nishida, C.; Ko, G.T.; Kumanyika, S. Body fat distribution and noncommunicable diseases in populations: Overview of the 2008 WHO Expert Consultation on Waist Circumference and Waist-Hip Ratio. Eur. J. Clin. Nutr. 2010, 64, 2–5. [Google Scholar] [CrossRef]
- Mekary, R.A.; Feskanich, D.; Hu, F.B.; Willett, W.C.; Field, A.E. Physical activity in relation to long-term weight maintenance after intentional weight loss in premenopausal women. Obesity (Silver Spring) 2010, 18, 167–174. [Google Scholar] [CrossRef]
- Seo, D.-C.; Li, K. Leisure-time physical activity dose-response effects on obesity among US adults: Results from the 1999–2006 National Health and Nutrition Examination Survey. J. Epidemiol. Community Health 2010, 64, 426–431. [Google Scholar] [CrossRef]
- Abdel-Megeid, F.Y.; Abdelkarem, H.M.; El-Fetouh, A.M. Unhealthy nutritional habits in university students are a risk factor for cardiovascular diseases. Saudi Med. J. 2011, 32, 621–627. [Google Scholar] [PubMed]
- Ogden, C.L.; Lamb, M.M.; Carroll, M.D.; Flegal, K.M. Obesity and socioeconomic status in adults: United States, 2005–2008. NCHS Data Brief 2010, 50, 1–8. [Google Scholar]
- World Health Organization. Global Status Report on Noncommunicable Diseases; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Brikou, D.; Zannidi, D.; Karfopoulou, E.; Anastasiou, C.A.; Yannakoulia, M. Breakfast consumption and weight-loss maintenance: Results from the MedWeight study. Br. J. Nutr. 2016, 115, 2246–2251. [Google Scholar] [CrossRef] [PubMed]
- Kreienbrock, L.; Pigeot, I.; Ahrens, W. Epidemiologische Methoden, 5th ed.; Springer Spektrum: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
| Reference | Country | Study Design and Follow-Up Period | Participants | Exposure vs. Comparison, Measurement | Outcome, Measurement | Results | |
|---|---|---|---|---|---|---|---|
| n Total n Cases * | Sex ** Age *** | Estimated Effect Sizes (95%CI LL; UL) | |||||
| Goto et al., 2008 [36] | Japan | retrospective, check-up data, 2000–2007 | 4634 598 | 100% 21.5 | Skipping ≥ 2 vs. ≤ 1 d/wk, self-administered questionnaire | >5% increased BMI, weight and height measurement | OR = 1.34 (1.12; 1.62) |
| Guinter et al., 2020 [90] | USA, Puerto Rico | Sisters Study, prospective cohort, 2003–2015 | 46,037 2797, 2383, 6807 | 0% 55.3 | Eating 3–4 d/wk vs. 0, 1–2, 5–6, 7 d/wk, FFQ | 5-year incident BMI ≥ 25 kg/2, ≥ 30 kg/2, ≥ 5 kg weight gain, weight and height measurement and self-reported weight | 5-yr incident BMI ≥ 25kg/2: 0 d/wk RR = 0.74 (0.62; 0.89), 1–2 d/wk RR = 0.91 (0.78; 1.07), 5–6 d/wk RR = 0.97 (0.85; 1.09), 7 d/wk RR = 0.88 (0.78; 0.99) 5-yr incident BMI ≥ 30kg/2: 0 d/wk RR = 0.72 (0.59; 0.87), 1–2 d/wk RR = 0.75 (0.62; 0.89), 5–6 d/wk RR = 0.91 (0.80; 1.04), 7 d/wk RR = 0.79 (0.70; 0.90) 5-yr incident ≥ 5 kg weight gain: 0 d/wk RR = 1.00 (0.90; 1.11), 1–2 d/wk RR = 0.98 (0.89; 1.08), 5–6 d/wk RR = 0.99 (0.92; 1.06), 7 d/wk RR = 0.97 (0.91; 1.04) |
| Hurst and Fukuda, 2018 [37] | Japan | secondary analysis of insurance and health check-up data, 2008–2013 | 59,717 20,671 | 66% 47.4 | Skipping ≤ 2 vs. ≥ 3 d/wk, Health check-up question | BMI ≥ 25 kg/2, BMI and WC change, BMI and WC data from check-up | BMI ≥ 25kg/2: OR = 0.92 (0.87; 0.97) BMI change (in kg/2): β = 0.00 (−0.03; 0.04) WC change (in cm): β = 0.03 (−0.11; 0.16) |
| Kahleova et al., 2017 [93] | North America Canada | AHS-2, prospective cohort, 2002–2010 | 50,660 n.g. ++ | 36% 58 | Eating vs. skipping, Hospital History Form | BMI change/year, weight and height measurement and self-report | BMI change (in kg/2): β = −0.03 (−0.04; -0.01) |
| Kito et al., 2019 [38] | Japan | retrospective cohort, 2008/09–2012 | 45,524 5093 | 100% 34 | Skipping ≥ 3 vs. ≤ 2 d/wk, Health check-up question | BMI ≥ 25 kg/2, weight and height measurement | OR = 1.18 (1.04; 1.33) |
| Nooyens et al., 2005 [92] | The Netherlands | Doetinchem Cohort Study, prospective, 1987–2002 | 288 n.g. ++ | 100% 54.9 | Eating 0–7 d/wk Dutch version of EPIC FFQ | Weight and WC change/year, weight, height and WC measurement | Weight change (in kg): β = 0.04 (n.g. ++) WC change (in cm): β = 0.10 (n.g. ++) |
| Odegaard et al., 2013 [91] | USA | CARDIA Study, prospective cohort, 1992/93–2011 | 3598 972 WC 783 BMI | 44% 32.1 | Eating ≤ 3 vs. 4–6, 7 d/wk, interviewer-administered CARDIA DHQ | BMI ≥ 30 kg/2, WC > 88 cm for women and > 102 cm for men, weight, height and WC measurement | BMI ≥ 30 kg/2: 4–6 d/wk HR = 0.75 (0.62; 0.90), 7 d/wk HR = 0.57 (0.47; 0.68) WC > 88 or 120 cm: 4–6 d/wk HR = 0.84 (0.70; 0.99), 7 d/wk HR = 0.78 (0.66; 0.91) |
| Smith et al., 2017 [39] | Australia | CDAH Study, prospective cohort, baseline 2002/04–2011 | 1155 410 | 43% 31.5 | Met guidelines # consistently vs. met not, postal questionnaire | 5-year weight change, weight and height measurement and self-report | 5-yr weight change (in kg): β = 1.5 (0.5; 2.8) |
| van der Heijden et al., 2007 [94] | USA | HPFS, prospective cohort, 1992–2002 | 20,064 5857 | 100% 57.3 | Eating vs. skipping, semi-quantitative FFQ | ≥ 5 kg weight gain, self-reported weight | HR = 0.87 (0.82; 0.93) |
| Reference | Adjustment for Important Variables | All 7 | Adjustment for Other Variables | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Age | Sex | Education | Smoking | Physical Activity | Alcohol | TEI | |||
| Goto et al., 2008 [36] |  |  | fatty food, living alone | ||||||
| Guinter et al., 2020 [90] |  |  |  |  |  |  |  |  | race/ethnicity, Healthy Eating Index 2015, weight loss dieting, average sleep hours, perceived level of stress |
| Hurst and Fukuda, 2018 [37] |  |  | baseline BMI, obesity status, antidiabetic medication | ||||||
| Kahleova et al., 2017 [93] |  |  |  |  |  |  | ethnicity, dietary pattern, marital status, sleep, tv watching, high blood pressure medication | ||
| Kito et al., 2019 [38] |  |  |  | BMI, eating speed, late-night meals/ snacking, drinking, sleep, interactions | |||||
| Nooyens et al., 2005 [92] |  |  |  | retirement, type of job, diet, sugared soft drinks, fiber density, interactions | |||||
| Odegaard et al., 2013 [91] |  |  |  |  |  |  |  |  | race, fast food, dietary quality, meal frequency, baseline BMI and WC |
| Smith et al., 2017 [39] |  |  |  |  | baseline weight, time to follow-up, meal pattern, weekday of follow-up | ||||
| van der Heijden et al., 2007 [94] |  |  |  |  | baseline BMI, marital status, weightlifting | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wicherski, J.; Schlesinger, S.; Fischer, F. Association between Breakfast Skipping and Body Weight—A Systematic Review and Meta-Analysis of Observational Longitudinal Studies. Nutrients 2021, 13, 272. https://doi.org/10.3390/nu13010272
Wicherski J, Schlesinger S, Fischer F. Association between Breakfast Skipping and Body Weight—A Systematic Review and Meta-Analysis of Observational Longitudinal Studies. Nutrients. 2021; 13(1):272. https://doi.org/10.3390/nu13010272
Chicago/Turabian StyleWicherski, Julia, Sabrina Schlesinger, and Florian Fischer. 2021. "Association between Breakfast Skipping and Body Weight—A Systematic Review and Meta-Analysis of Observational Longitudinal Studies" Nutrients 13, no. 1: 272. https://doi.org/10.3390/nu13010272
APA StyleWicherski, J., Schlesinger, S., & Fischer, F. (2021). Association between Breakfast Skipping and Body Weight—A Systematic Review and Meta-Analysis of Observational Longitudinal Studies. Nutrients, 13(1), 272. https://doi.org/10.3390/nu13010272