Long-Chain Polyunsaturated Fatty Acids (LCPUFAs) and the Developing Immune System: A Narrative Review
Abstract
1. Introduction
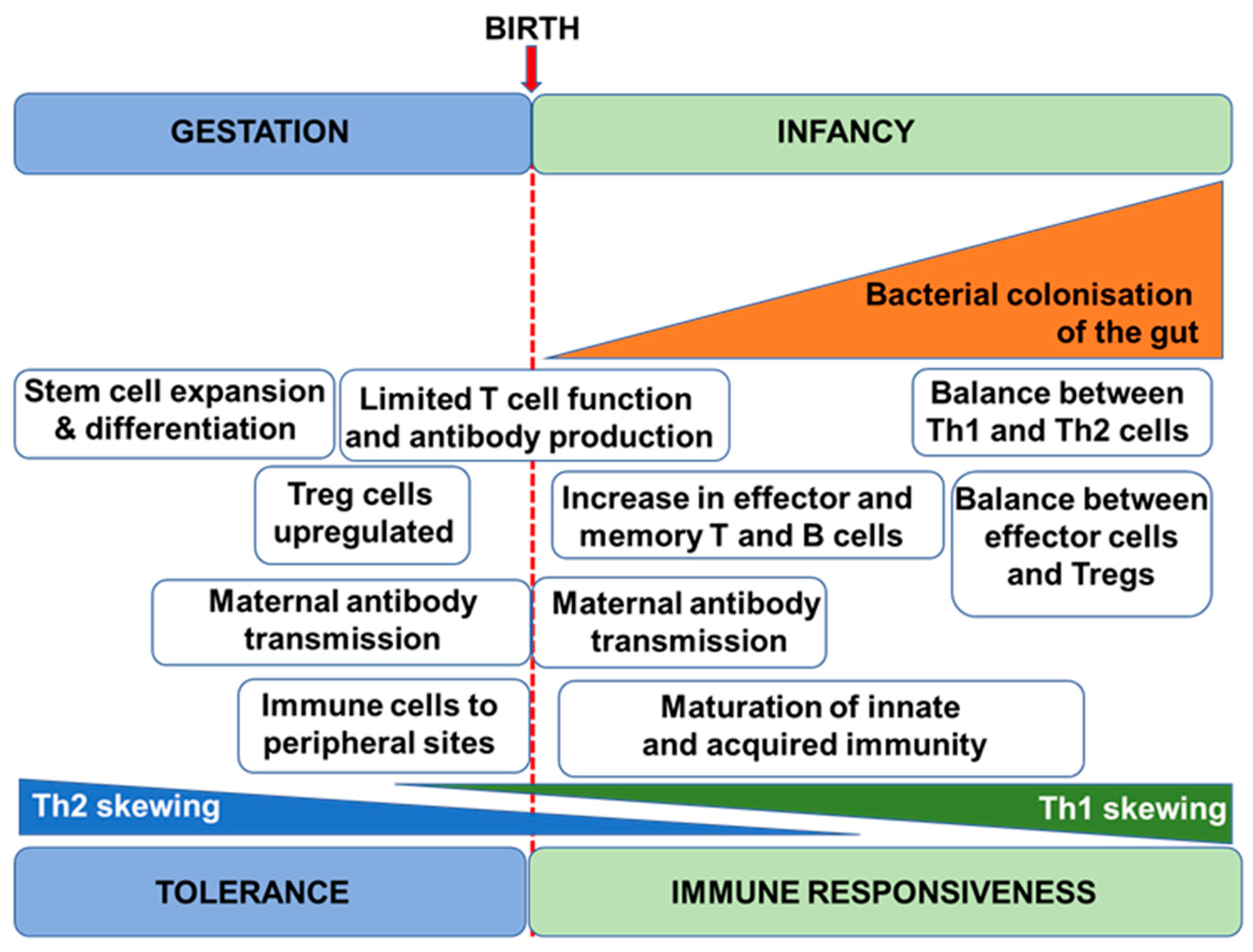
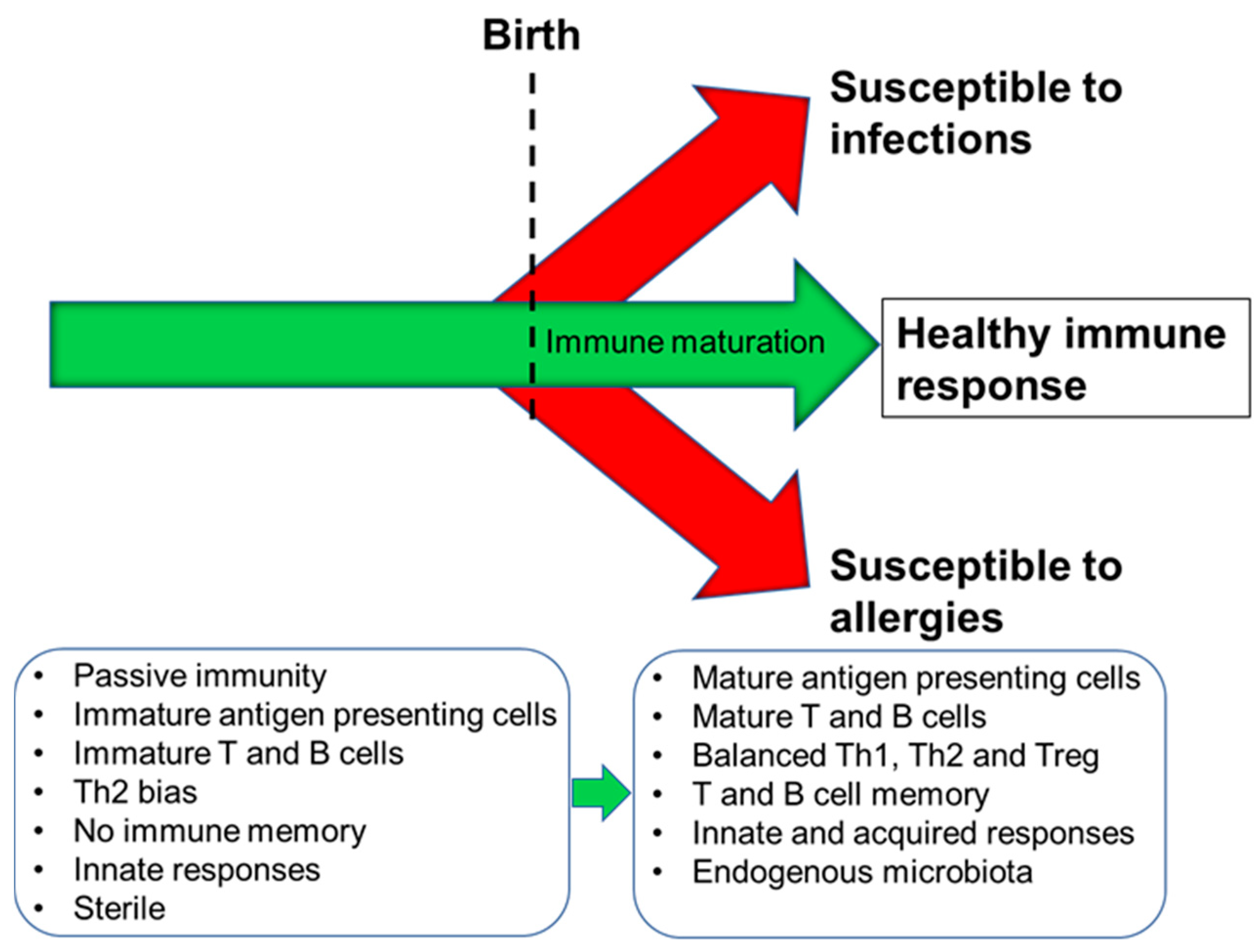
2. Immune Development in Humans
3. Importance of Breast Milk Factors to Immune Development
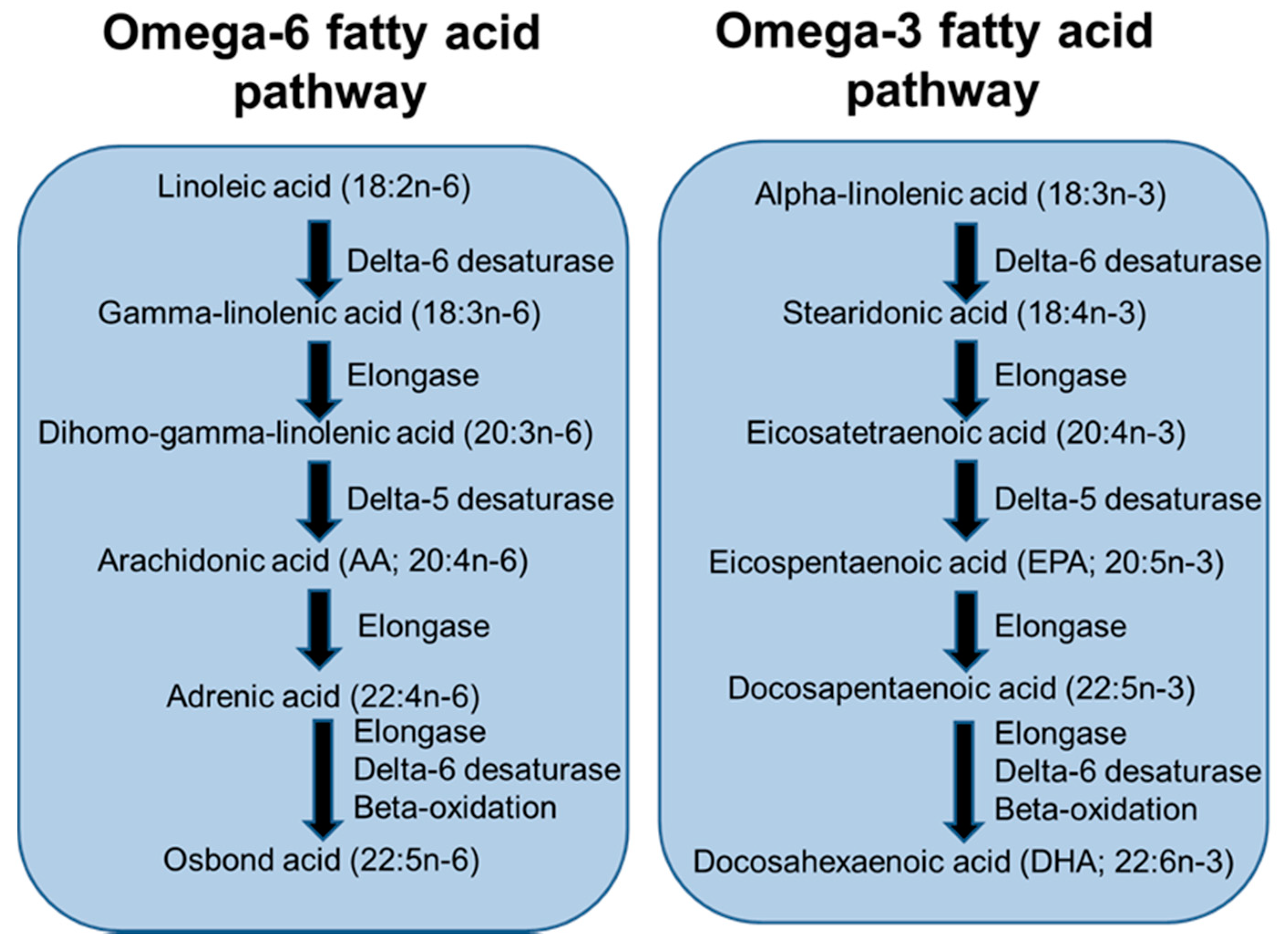
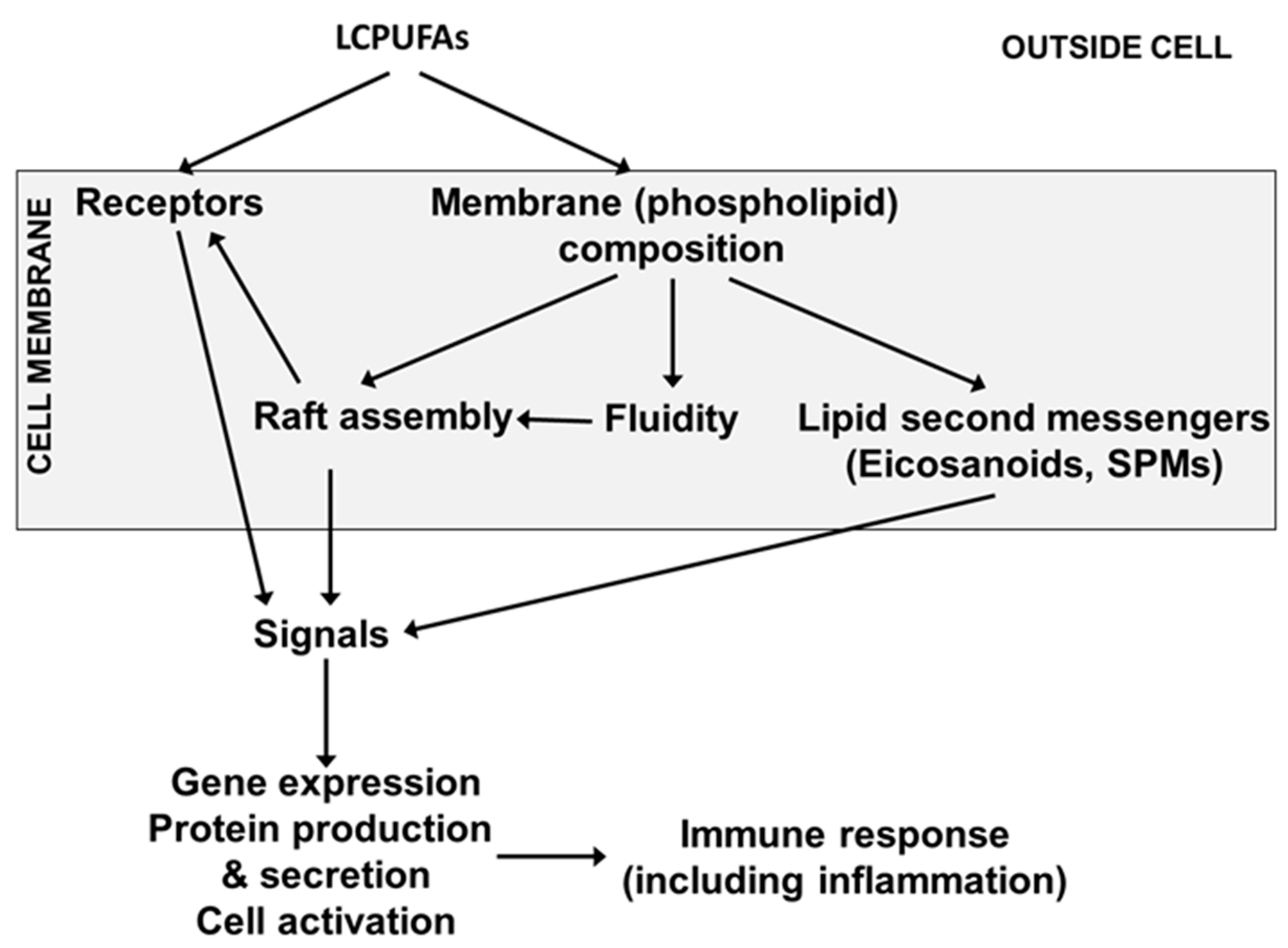
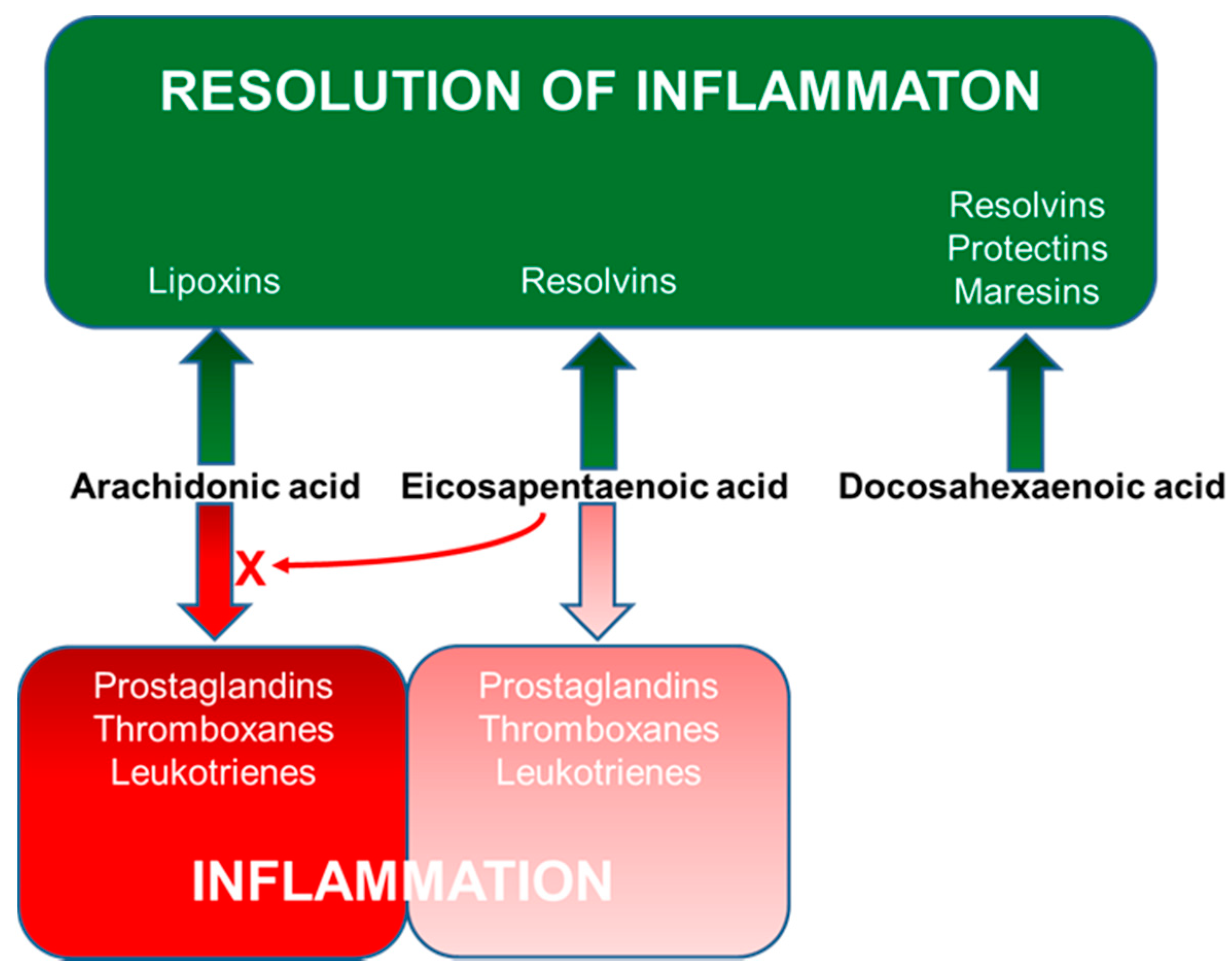
4. Long-Chain Polyunsaturated Fatty Acids, Lipid Mediators, Immunity and Inflammation
5. LCPUFAs, Immune Development, Allergic Disease and Infection
5.1. Trials of n-3 LCPUFAS in Pregnant and Lactating Women
5.2. Trials of LCPUFAs in Infants
6. Summary, Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AA | arachidonic acid |
| COX | cyclooxygenase |
| DHA | docosahexaenoic acid |
| EPA | eicosapentaenoic acid |
| GI | gastrointestinal |
| IFN | interferon |
| Ig | immunoglobulin |
| IL | interleukin |
| LCPUFA | long-chain polyunsaturated fatty acid |
| LOX | lipoxygenase |
| LT | leukotriene |
| PG | prostaglandin |
| PUFA | polyunsaturated fatty acid |
| SPM | specialised pro-resolving mediator |
| SPT | skin prick test |
| Th1 | T-helper 1 |
| Th2 | T-helper 2 |
References
- Caminero, A.; Pinto-Sanchez, M.I. Host immune interactions in chronic inflammatory gastrointestinal conditions. Curr. Opin. Gastroenterol. 2020, 36, 479–484. [Google Scholar] [PubMed]
- Yokanovich, L.T.; Newberry, R.D.; Knoop, K.A. Regulation of oral antigen delivery early in life: Implications for oral tolerance and food allergy. Clin. Exp. Allergy 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Mowat, A.M. Anatomical basis of tolerance and immunity to intestinal antigens. Nat. Rev. Immunol. 2003, 3, 331–341. [Google Scholar] [CrossRef] [PubMed]
- Goenka, A.; Kollmann, T.R. Development of immunity in early life. J. Infect. 2015, 71 (Suppl. 1), S112–S120. [Google Scholar] [CrossRef]
- Kane, S.V.; Acquah, L.A. Placental transport of immunoglobulins: A clinical review for gastroenterologists who prescribe therapeutic monoclonal antibodies to women during conception and pregnancy. Am. J. Gastroenterol. 2009, 104, 228–233. [Google Scholar] [CrossRef]
- Sykes, L.; MacIntyre, D.A.; Yap, X.J.; Teoh, T.G.; Bennett, P.R. The Th1:Th2 dichotomy of pregnancy and preterm labour. Mediat. Inflamm. 2012, 2012, 96762. [Google Scholar] [CrossRef]
- Wang, W.; Sung, N.; Gilman-Sachs, A.; Kwak-Kim, J. T helper (Th) cell profiles in pregnancy and recurrent pregnancy losses: Th1/Th2/Th9/Th17/Th22/Tfh cells. Front. Immunol. 2020, 11, 2025. [Google Scholar] [CrossRef]
- Lewis, E.D.; Richard, C.; Larsen, B.M.; Field, C.J. The importance of human milk for immunity in preterm infants. Clin. Perinatol. 2017, 44, 23–47. [Google Scholar] [CrossRef]
- Simon, A.K.; Hollander, G.A.; McMichael, A. Evolution of the immune system in humans from infancy to old age. Proc. Biol. Sci. 2015, 282, 20143085. [Google Scholar] [CrossRef]
- Ximenez, C.; Torres, J. Development of microbiota in infants and its role in maturation of gut mucosa and immune system. Arch. Med. Res. 2017, 48, 666–680. [Google Scholar] [CrossRef]
- Gopalakrishna, K.P.; Hand, T.W. Influence of maternal milk on the neonatal intestinal microbiome. Nutrients 2020, 12, 823. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, F.; O’Sullivan, A.; Smilowitz, J.T.; Freeman, S.L. Lactation and intestinal microbiota: How early diet shapes the infant gut. J. Mammary Gland Biol. Neoplasia 2015, 20, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Andreas, N.J.; Kampmann, B.; Mehring Le-Doare, K. Human breast milk: A review on its composition and bioactivity. Early Hum. Dev. 2015, 91, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Ogra, P.L. Immunology of human milk and lactation: Historical overview. Nestle Nutr. Inst. Workshop Ser. 2020, 94, 11–26. [Google Scholar] [PubMed]
- Weström, B.; Arévalo Sureda, E.; Pierzynowska, K.; Pierzynowski, S.G.; Pérez-Cano, F.J. The immature gut barrier and its importance in establishing immunity in newborn mammals. Front. Immunol. 2020, 11, 1153. [Google Scholar] [CrossRef] [PubMed]
- Rogier, E.W.; Frantz, A.L.; Bruno, M.E.; Wedlund, L.; Cohen, D.A.; Stromberg, A.J.; Kaetzel, C.S. Lessons from mother: Long-term impact of antibodies in breast milk on the gut microbiota and intestinal immune system of breastfed offspring. Gut Microbes 2014, 5, 663–668. [Google Scholar] [CrossRef]
- Sankar, M.J.; Sinha, B.; Chowdhury, R.; Bhandari, N.; Taneja, S.; Martines, J.; Bahl, R. Optimal breastfeeding practices and infant and child mortality: A systematic review and meta-analysis. Acta Paediatr. 2015, 104, 3–13. [Google Scholar] [CrossRef]
- Oddy, W.H. Breastfeeding, childhood asthma, and allergic disease. Ann. Nutr. Metab. 2017, 70 (Suppl. 2), 26–36. [Google Scholar] [CrossRef]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-Schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and arachidonic acid concentrations in human breast milk worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [CrossRef]
- Demmelmair, H.; Koletzko, B. Importance of fatty acids in the perinatal period. World Rev. Nutr. Diet. 2015, 112, 31–47. [Google Scholar]
- Yaqoob, P.; Pala, H.S.; Cortina-Borja, M.; Newsholme, E.A.; Calder, P.C. Encapsulated fish oil enriched in α-tocopherol alters plasma phospholipid and mononuclear cell fatty acid compositions but not mononuclear cell functions. Eur. J. Clin. Investig. 2000, 30, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Rees, D.; Miles, E.A.; Banerjee, T.; Wells, S.J.; Roynette, C.E.; Wahle, K.W.J.W.; Calder, P.C. Dose-related effects of eicosapentaenoic acid on innate immune function in healthy humans: A comparison of young and older men. Am. J. Clin. Nutr. 2006, 83, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Functional roles of fatty acids and their effects on human health. J. Parent. Enter. Nutr. 2015, 39 (Suppl. 1), 18S–32S. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Dietary lipids and the immune system. Nutr. Rev. 1998, 56, S70–S83. [Google Scholar] [CrossRef]
- Christie, W.W.; Harwood, J.L. Oxidation of polyunsaturated fatty acids to produce lipid mediators. Essays Biochem. 2020, 64, 401–421. [Google Scholar]
- Calder, P.C. Eicosanoids. Essays Biochem. 2020, 64, 423–441. [Google Scholar]
- Tilley, S.L.; Cofman, T.M.; Koller, B.H. Mixed messages: Modulation of inflammation and immune responses by prostaglandins and thromboxanes. J. Clin. Investig. 2001, 108, 15–23. [Google Scholar] [CrossRef]
- Samuchiwal, S.K.; Boyce, J.A. Role of lipid mediators and control of lymphocyte responses in type 2 immunopathology. J. Allergy Clin. Immunol. 2018, 141, 1182–1190. [Google Scholar] [CrossRef]
- Kalinski, P. Regulation of immune responses by prostaglandin E2. J. Immunol. 2012, 188, 21–28. [Google Scholar] [CrossRef]
- Brundvik, K.W.; Tasken, K. Modulation of T cell immune functions by the prostaglandin E2—cAMP pathway in chronic inflammatory states. Br. J. Pharmacol. 2012, 166, 411–419. [Google Scholar] [CrossRef]
- Gao, Y.; Zhao, C.; Wang, W.; Jin, R.; Li, Q.; Ge, Q.; Guan, Y.; Zhang, Y. Prostaglandins E2 signal mediated by receptor subtype EP2 promotes IgE production in vivo and contributes to asthma development. Sci. Rep. 2016, 6, 20505. [Google Scholar] [CrossRef] [PubMed]
- Tsuge, K.; Inazumi, T.; Shimamoto, A.; Sugimoto, Y. Molecular mechanisms underlying prostaglandin E2-exacerbated inflammation and immune diseases. Int. Immunol. 2019, 31, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Levy, B.D.; Clish, C.B.; Schmidt, B.; Gronert, K.; Serhan, C.N. Lipid mediator class switching during acute inflammation: Signals in resolution. Nat. Immunol. 2001, 2, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Vachier, I.; Chanez, P.; Bonnans, C.; Godard, P.; Bousquet, J.; Chavis, C. Endogenous anti-inflammatory mediators from arachidonate in human neutrophils. Biochem. Biophys. Res. Commun. 2002, 290, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, M.; Mitsui, C.; Hayashi, H.; Ono, E.; Kajiwara, K.; Mita, H.; Watai, K.; Kamide, Y.; Fukutomi, Y.; Sekiya, K.; et al. Aspirin-exacerbated respiratory disease (AERD): Current understanding of AERD. Allergol. Int. 2019, 68, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Rusznak, M.; Peebles, R.S., Jr. Prostaglandin E2 in NSAID-exacerbated respiratory disease: Protection against cysteinyl leukotrienes and group 2 innate lymphoid cells. Curr. Opin. Allergy Clin. Immunol. 2019, 19, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, S.; Willmes, D.M.; Nassiri, M.; Babina, M.; Worm, M. PGE 2 deficiency predisposes to anaphylaxis by causing mast cell hyperresponsiveness. J. Allergy Clin. Immunol. 2020, 146, 1387–1396. [Google Scholar] [CrossRef] [PubMed]
- Joo, M.; Sasikot, R.T. PGD synthase and PGD2 in immune response. Mediat. Inflamm. 2012, 2012, 503128. [Google Scholar] [CrossRef]
- Domoingo, C.; Palomares, O.; Sandham, D.A.; Erpenbeck, V.J.; Altman, P. The prostaglandin D2 receptor pathway in asthma: A key player in airway inflammation. Respir. Res. 2018, 19, 189. [Google Scholar] [CrossRef]
- Honda, T.; Kabashima, K. Prostanoids and leukotrienes in pathophysiology of atopic dermatitis and psoriasis. Int. Immunol. 2019, 31, 589–595. [Google Scholar] [CrossRef]
- Radmark, O.; Werz, O.; Steinhilber, D.; Samuelsson, B. 5-Lipoxygenase, a key enzyme for leukotriene biosynthesis in health and disease. Biochim. Biophys. Acta 2015, 1851, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Haeggstrom, J.Z.; Funk, C.D. Lipoxygenase and leukotriene pathways: Biochemistry, biology, and roles in disease. Chem. Rev. 2011, 111, 5866–5898. [Google Scholar] [CrossRef] [PubMed]
- Brandt, S.L.; Serezani, C.H. Too much of a good thing: How modulating LTB4 actions restore host defense in homeostasis or disease. Semin. Immunol. 2017, 33, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Theron, A.J.; Steel, H.C.; Tintinger, G.R.; Gravett, C.M.; Anderson, R.; Feldman, C. Cysteinyl leukotriene receptor-1 antagonists as modulators of innate immune cell function. J. Immunol. Res. 2014, 2014, 608930. [Google Scholar] [CrossRef] [PubMed]
- Jo-Watanabe, A.; Okuna, T.; Yokomizo, T. The role of leukotrienes as potential targets in allergic disease. Int. J. Mol. Sci. 2019, 20, 3580. [Google Scholar] [CrossRef] [PubMed]
- Gelfrand, E.W. Importance of the leukotriene B4-BLT1 and LTB4-BLT2 pathways in asthma. Semin. Immunol. 2017, 33, 44–51. [Google Scholar] [CrossRef]
- Laidlaw, T.M.; Boyce, J.A. Cysteinyl leukotriene receptors, old and new; implications for asthma. Clin. Exp. Allergy 2012, 42, 1313–1320. [Google Scholar] [CrossRef]
- Lee, T.H.; Hoover, R.L.; Williams, J.D.; Sperling, R.I.; Ravalese, J., III; Spur, B.W.; Robinson, D.R.; Corey, E.; Lewis, R.A.; Austen, K.F. Effects of dietary enrichment with eicosapentaenoic acid and docosahexaenoic acid on in vitro neutrophil and monocyte leukotriene generation and neutrophil function. N. Engl. J. Med. 1985, 312, 1217–1224. [Google Scholar] [CrossRef]
- Endres, S.; Ghorbani, R.; Kelley, V.E.; Georgilis, K.; Lonnemann, G.; van der Meer, L.M.W.; Cannon, J.G.; Rogers, T.S.; Klempner, M.S.; Weber, P.C. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N. Engl. J. Med. 1989, 320, 265–271. [Google Scholar] [CrossRef]
- Sperling, R.I.; Benincaso, A.I.; Knoell, C.T.; Larkin, J.K.; Austen, K.F.; Robinson, D.R. Dietary ω-3 polyunsaturated fatty acids inhibit phosphoinositide formation and chemotaxis in neutrophils. J. Clin. Investig. 1993, 91, 651–660. [Google Scholar] [CrossRef]
- Goodnight, S.H., Jr.; Harris, W.S.; Connor, W.E. The effects of dietary omega 3 fatty acids on platelet composition and function in man: A prospective, controlled study. Blood 1981, 58, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Bevan, S.J.; Newsholme, E.A. The inhibition of T-lymphocyte proliferation by fatty acids is via an eicosanoid-independent mechanism. Immunology 1992, 75, 108–115. [Google Scholar] [PubMed]
- Goldman, D.W.; Pickett, W.C.; Goetzl, E.J. Human neutrophil chemotactic and degranulating activities of leukotriene B5 (LTB5) derived from eicosapentaenoic acid. Biochem. Biophys. Res. Commun. 1983, 117, 282–288. [Google Scholar] [CrossRef]
- Lee, T.H.; Mencia-Huerta, J.M.; Shih, C.; Corey, E.J.; Lewis, R.A.; Austen, K.F. Characterization and biologic properties of 5,12-dihydroxy derivatives of eicosapentaenoic acid, including leukotriene-B5 and the double lipoxygenase product. J. Biol. Chem. 1984, 259, 2383–2389. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N. Resolution phase lipid mediators of inflammation: Agonists of resolution. Curr. Opin. Pharmacol. 2013, 13, 632–640. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N.; Dalli, J. The resolution code of acute inflammation: Novel pro-resolving lipid mediators in resolution. Semin. Immunol. 2015, 27, 200–215. [Google Scholar] [CrossRef]
- Serhan, C.N. Discovery of specialized pro-resolving mediators marks the dawn of resolution physiology and pharmacology. Mol. Asp. Med. 2017, 58, 1–11. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N.; Dalli, J. New pro-resolving n-3 mediators bridge resolution of infectious inflammation to tissue regeneration. Mol. Asp. Med. 2018, 64, 1–17. [Google Scholar] [CrossRef]
- Nordgren, T.M.; Anderson Berry, A.; Van Ormer, M.; Zoucha, S.; Elliott, E.; Johnson, R.; McGinn, E.; Cave, C.; Rilett, K.; Weishaar, K.; et al. Omega-3 fatty acid supplementation, pro-resolving mediators, and clinical outcomes in maternal-infant pairs. Nutrients 2019, 11, 98. [Google Scholar] [CrossRef]
- See, V.H.L.; Mas, E.; Prescott, S.L.; Beilin, L.J.; Burrows, S.; Barden, A.E.; Huang, R.C.; Mori, T.A. Effects of postnatal omega-3 fatty acid supplementation on offspring pro-resolving mediators of inflammation at 6 months and 5 years of age: A double blind, randomized controlled clinical trial. Prostaglandins Leukot. Essent. Fatty Acids 2017, 126, 126–132. [Google Scholar] [CrossRef] [PubMed]
- See, V.H.L.; Mas, E.; Prescott, S.L.; Beilin, L.J.; Burrows, S.; Barden, A.E.; Huang, R.C.; Mori, T.A. Effects of prenatal n-3 fatty acid supplementation on offspring resolvins at birth and 12 years of age: A double-blind, randomised controlled clinical trial. Br. J. Nutr. 2017, 118, 971–980. [Google Scholar] [CrossRef] [PubMed]
- Winkler, J.W.; Orr, S.K.; Dalli, J.; Cheng, C.Y.; Sanger, J.M.; Chiang, N.; Petasis, N.A.; Serhan, C.N. Resolvin D4 stereoassignment and its novel actions in host protection and bacterial clearance. Sci. Rep. 2016, 6, 18972. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G.A.; Troxler, H.; Klinke, G.; Rogler, D.; Braegger, C.; Hersberger, M. High levels of anti-inflammatory and pro-resolving lipid mediators lipoxins and resolvins and declining docosahexaenoic acid levels in human milk during the first month of lactation. Lipids Health Dis. 2013, 12, 89. [Google Scholar] [CrossRef] [PubMed]
- Arnardottir, H.; Orr, S.K.; Dalli, J.; Serhan, C.N. Human milk proresolving mediators stimulate resolution of acute inflammation. Mucosal Immunol. 2016, 9, 757–766. [Google Scholar] [CrossRef]
- Healy, D.A.; Wallace, F.A.; Miles, E.A.; Calder, P.C.; Newsholme, P. Effect of low-to-moderate amounts of dietary fish oil on neutrophil lipid composition and function. Lipids 2000, 35, 763–768. [Google Scholar] [CrossRef]
- Browning, L.M.; Walker, C.G.; Mander, A.P.; West, A.L.; Madden, J.; Gambell, J.M.; Young, S.; Wang, L.; Jebb, S.A.; Calder, P.C. Incorporation of eicosapentaenoic and docosahexaenoic acids into lipid pools when given as supplements providing doses equivalent to typical intakes of oily fish. Am. J. Clin. Nutr. 2012, 96, 748–758. [Google Scholar] [CrossRef]
- Calder, P.C. Eicosapentaenoic and docosahexaenoic acid derived specialised pro-resolving mediators: Concentrations in humans and the effects of age, sex, disease and increased omega-3 fatty acid intake. Biochimie 2020, 178, 105–123. [Google Scholar] [CrossRef]
- Krauss-Etschmann, S.; Hartl, D.; Rzehak, P.; Heinrich, J.; Shadid, R.; del Carmen Ramírez-Tortosa, M.; Campoy, C.; Pardillo, S.; Schendel, D.J.; Decsi, T.; et al. Nutraceuticals for Healthier Life Study Group. Decreased cord blood IL-4, IL-13, and CCR4 and increased TGF-beta levels after fish oil supplementation of pregnant women. J. Allergy Clin. Immunol. 2008, 121, 464–470. [Google Scholar] [CrossRef]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Taylor, A.L.; Holt, P.G.; Prescott, S.L. Maternal fish oil supplementation in pregnancy reduces interleukin-13 levels in cord blood of infants at high risk of atopy. Clin. Exp. Allergy 2003, 33, 442–448. [Google Scholar] [CrossRef]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Taylor, A.L.; Holt, P.G.; Prescott, S.L. Fish oil supplementation in pregnancy modifies neonatal allergen-specific immune responses and clinical outcomes in infants at high risk of atopy: A randomized, controlled trial. J. Allergy Clin. Immunol. 2003, 112, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Prescott, S.L.; Barden, A.E.; Mori, T.A.; Dunstan, J.A. Maternal fish oil supplementation in pregnancy modifies neonatal leukotriene production by cord-blood-derived neutrophils. Clin. Sci. 2006, 113, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Denburg, J.A.; Hatfield, H.M.; Cyr, M.M.; Hayes, L.; Holt, P.G.; Sehmi, R.; Dunstan, J.A.; Prescott, S.L. Fish oil supplementation in pregnancy modifies neonatal progenitors at birth in infants at risk of atopy. Pediatric Res. 2005, 57, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Furuhjelm, C.; Warstedt, K.; Larsson, J.; Fredriksson, M.; Böttcher, M.F.; Fälth-Magnusson, K.; Duchén, K. Fish oil supplementation in pregnancy and lactation may decrease the risk of infant allergy. Acta Paediatr. 2009, 98, 1461–1467. [Google Scholar] [CrossRef]
- Furuhjelm, C.; Warstedt, K.; Fagerås, M.; Fälth-Magnusson, K.; Larsson, J.; Fredriksson, M.; Duchén, K. Allergic disease in infants up to 2 years of age in relation to plasma omega-3 fatty acids and maternal fish oil supplementation in pregnancy and lactation. Pediatric Allergy Immunol. 2011, 22, 505–514. [Google Scholar] [CrossRef]
- Mozurkewich, E.L.; Berman, D.R.; Vahratian, A.; Clinton, C.M.; Romero, V.C.; Chilimigras, J.L.; Vazquez, D.; Qualls, C.; Djuric, Z. Effect of prenatal EPA and DHA on maternal and umbilical cord blood cytokines. BMC Pregnancy Childbirth 2018, 18, 261. [Google Scholar] [CrossRef]
- Mozurkewich, E.L.; Greenwood, M.; Clinton, C.; Berman, D.; Romero, V.; Djuric, Z.; Qualls, C.; Gronert, K. Pathway markers for pro-resolving lipid mediators in maternal and umbilical cord blood: A secondary analysis of the Mothers, Omega-3, and Mental Health Study. Front. Pharmacol. 2016, 7, 274. [Google Scholar] [CrossRef]
- Romero, V.C.; Somers, E.C.; Stolberg, V.; Clinton, C.; Chensue, S.; Djuric, Z.; Berman, D.R.; Treadwell, M.C.; Vahratian, A.M.; Mozurkewich, E. Developmental programming for allergy: A secondary analysis of the Mothers, Omega-3, and Mental Health Study. Am. J. Obstet. Gynecol. 2013, 208, 316. [Google Scholar] [CrossRef]
- Berman, D.; Clinton, C.; Limb, R.; Somers, E.C.; Romero, V.; Mozurkewich, E. Prenatal omega-3 supplementation and eczema risk among offspring at age 36 months. Insights Allergy Asthma Bronchitis 2016, 2, 1. [Google Scholar] [CrossRef]
- Granot, E.; Jakobovich, E.; Rabinowitz, R.; Levy, P.; Schlesinger, M. DHA supplementation during pregnancy and lactation affects infants’ cellular but not humoral immune response. Mediat. Inflamm. 2011, 2011, 493925. [Google Scholar] [CrossRef]
- Olsen, S.F.; Osterdal, M.L.; Salvig, J.D.; Mortensen, L.M.; Rytter, D.; Secher, N.J.; Henriksen, T.B. Fish oil intake compared with olive oil intake in late pregnancy and asthma in the offspring: 16 y of registry-based follow-up from a randomized controlled trial. Am. J. Clin. Nutr. 2008, 88, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.; Strøm, M.; Maslova, E.; Dahl, R.; Hoffmann, H.J.; Rytter, D.; Bech, B.H.; Henriksen, T.B.; Granström, C.; Halldorsson, T.I.; et al. Fish oil supplementation during pregnancy and allergic respiratory disease in the adult offspring. J. Allergy Clin. Immunol. 2017, 139, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Palmer, D.J.; Sullivan, T.; Gold, M.S.; Prescott, S.L.; Heddle, R.; Gibson, R.A.; Makrides, M. Effect of n-3 long chain polyunsaturated fatty acid supplementation in pregnancy on infants’ allergies in first year of life: Randomised controlled trial. Br. Med. J. 2012, 344, e184. [Google Scholar] [CrossRef] [PubMed]
- Palmer, D.J.; Sullivan, T.; Gold, M.S.; Prescott, S.L.; Heddle, R.; Gibson, R.A.; Makrides, M. Randomized controlled trial of fish oil supplementation in pregnancy on childhood allergies. Allergy 2013, 68, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- Best, K.P.; Sullivan, T.; Palmer, D.; Gold, M.; Kennedy, D.J.; Martin, J.; Makrides, M. Prenatal fish oil supplementation and allergy: 6-year follow-up of a randomized controlled trial. Pediatrics 2016, 137, e20154443. [Google Scholar] [CrossRef] [PubMed]
- Bisgaard, H.; Stokholm, J.; Chawes, B.L.; Vissing, N.H.; Bjarnadóttir, E.; Schoos, A.M.; Wolsk, H.M.; Pedersen, T.M.; Vinding, R.K.; Thorsteinsdóttir, S.; et al. Fish oil-derived fatty acids in pregnancy and wheeze and asthma in offspring. N. Engl. J. Med. 2016, 375, 2530–2539. [Google Scholar] [CrossRef]
- Imhoff-Kunsch, B.; Stein, A.D.; Martorell, R.; Parra-Cabrera, S.; Romieu, I.; Ramakrishnan, U. Prenatal docosahexaenoic acid supplementation and infant morbidity: Randomized controlled trial. Pediatrics 2011, 128, e505–e512. [Google Scholar] [CrossRef]
- Escamilla-Nuñez, M.C.; Barraza-Villarreal, A.; Hernández-Cadena, L.; Navarro-Olivos, E.; Sly, P.D.; Romieu, I. Omega-3 fatty acid supplementation during pregnancy and respiratory symptoms in children. Chest 2014, 146, 373–382. [Google Scholar] [CrossRef]
- Lee, H.S.; Barraza-Villarreal, A.; Hernandez-Vargas, H.; Sly, P.D.; Biessy, C.; Ramakrishnan, U.; Romieu, I.; Herceg, Z. Modulation of DNA methylation states and infant immune system by dietary supplementation with ω-3 PUFA during pregnancy in an intervention study. Am. J. Clin. Nutr. 2013, 98, 480–487. [Google Scholar] [CrossRef]
- Amarasekera, M.; Noakes, P.; Strickland, D.; Saffery, R.; Martino, D.J.; Prescott, S.L. Epigenome-wide analysis of neonatal CD4+ T-cell DNA methylation sites potentially affected by maternal fish oil supplementation. Epigenetics 2014, 9, 1570–1576. [Google Scholar] [CrossRef]
- Lauritzen, L.; Kjaer, T.M.R.; Fruekilde, M.B.; Michaelsen, K.F.; Frokiaer, H. Fish oil supplementation of lactating mothers affects cytokine production in 2 1/2-year-old children. Lipids 2005, 40, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Warstedt, K.; Furuhjelm, C.; Duchen, K.; Falth-Magnusson, K.; Fageras, M. The effects of omega-3 fatty acid supplementation in pregnancy on maternal eicosanoid, cytokine, and chemokine secretion. Pediatric Res. 2009, 66, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Field, C.J.; Thomson, C.A.; Van Aerde, J.E.; Parrott, A.; Euler, A.; Lien, E.; Clandinin, M.T. Lower proportion of CD45R0+ cells and deficient interleukin-10 production by formula-fed infants, compared with human-fed, is corrected with supplementation of long-chain polyunsaturated fatty acids. J. Pediatric Gastroenterol. Nutr. 2000, 31, 291–299. [Google Scholar] [CrossRef] [PubMed]
- Field, C.J.; Van Aerde, J.E.; Robinson, L.E.; Clandinin, M.T. Effect of providing a formula supplemented with long-chain polyunsaturated fatty acids on immunity in full-term neonates. Br. J. Nutr. 2008, 99, 91–99. [Google Scholar] [CrossRef]
- Birch, E.E.; Khoury, J.C.; Berseth, C.L.; Castañeda, Y.S.; Couch, J.M.; Bean, J.; Tamer, R.; Harris, C.L.; Mitmesser, S.H.; Scalabrin, D.M. The impact of early nutrition on incidence of allergic manifestations and common respiratory illnesses in children. J. Pediatrics 2010, 156, 902–906. [Google Scholar] [CrossRef]
- Pastor, N.; Soler, B.; Mitmesser, S.H.; Ferguson, P.; Lifschitz, C. Infants fed docosahexaenoic acid- and arachidonic acid-supplemented formula have decreased incidence of bronchiolitis/bronchitis the first year of life. Clin. Pediatrics 2006, 45, 850–855. [Google Scholar] [CrossRef]
- Lapillonne, A.; Pastor, N.; Zhuang, W.; Scalabrin, D.M.F. Infants fed formula with added long chain polyunsaturated fatty acids have reduced incidence of respiratory illnesses and diarrhea during the first year of life. BMC Prediatr. 2014, 14, 168. [Google Scholar] [CrossRef]
- Foiles, A.M.; Kerling, E.H.; Wick, J.A.; Scalabrin, D.M.F.; Colombo, J.; Carlson, S.E. Formula with long-chain polyunsaturated fatty acids reduces incidence of allergy in early childhood. Pediatr. Allergy Immunol. 2016, 27, 156–161. [Google Scholar] [CrossRef]
- D’Vaz, N.; Meldrum, S.J.; Dunstan, J.A.; Lee-Pullen, T.F.; Metcalfe, J.; Holt, B.J.; Serralha, M.; Tulic, M.K.; Mori, T.A.; Prescott, S.L. Fish oil supplementation in early infancy modulates developing infant immune responses. Clin. Exp. Allergy 2012, 42, 1206–1216. [Google Scholar] [CrossRef]
- D’Vaz, N.; Meldrum, S.J.; Dunstan, J.A.; Martino, D.; McCarthy, S.; Metcalfe, J.; Tulic, M.K.; Mori, T.A.; Prescott, S.A. Postnatal fish oil supplementation in high-risk infants to prevent allergy: Randomized controlled trial. Pediatrics 2012, 130, 674–682. [Google Scholar] [CrossRef]
- Damsgaard, C.T.; Lauritzen, L.; Kjaer, T.M.; Holm, P.M.; Fruekilde, M.B.; Michaelsen, K.F.; Frøkiaer, H. Fish oil supplementation modulates immune function in healthy infants. J. Nutr. 2007, 137, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Mihrshahi, S.; Peat, J.K.; Marks, G.B.; Mellis, C.M.; Tovey, E.R.; Webb, K.; Britton, W.J.; Leeder, S.R. Childhood Asthma Prevention Study. Eighteen-month outcomes of house dust mite avoidance and dietary fatty acid modification in the Childhood Asthma Prevention Study (CAPS). J. Allergy Clin. Immunol. 2003, 111, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Mihrshahi, S.; Peat, J.K.; Webb, K.; Oddy, W.; Marks, G.B.; Mellis, C.M. Effect of omega-3 fatty acid concentrations in plasma on symptoms of asthma at 18 months of age. Pediatric Allergy. Immunol. 2004, 15, 517–522. [Google Scholar] [CrossRef] [PubMed]
- Peat, J.K.; Mihrshahi, S.; Kemp, A.S.; Marks, G.B.; Tovey, E.R.; Webb, K.; Mellis, C.M.; Leeder, S.R. Three-year outcomes of dietary fatty acid modification and house dust mite reduction in the Childhood Asthma Prevention Study. J. Allergy Clin. Immunol. 2004, 114, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Marks, G.B.; Mihrshahi, S.; Kemp, A.S.; Tovey, E.R.; Webb, K.; Almqvist, C.; Ampon, R.D.; Crisafulli, D.; Belousova, E.G.; Mellis, C.M.; et al. Prevention of asthma during the first 5 years of life: A randomized controlled trial. J. Allergy Clin. Immunol. 2006, 118, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Almqvist, C.; Garden, F.; Xuan, W.; Mihrshahi, S.; Leeder, S.R.; Oddy, W.; Webb, K.; Marks, G.B.; CAPS team. Omega-3 and omega-6 fatty acid exposure from early life does not affect atopy and asthma at age 5 years. J. Allergy Clin. Immunol. 2007, 119, 1438–1444. [Google Scholar] [CrossRef]
- Clausen, M.; Jonasson, K.; Keil, T.; Beyer, K.; Sigurdardottir, S.T. Fish oil in infancy protects against food allergy in Iceland—Results from a birth cohort study. Allergy 2018, 73, 1305–1312. [Google Scholar] [CrossRef]
- Food and Agricultural Organisation of the United Nations. Fat and Fatty Acids in Human Nutrition: Report of an Expert Consultation; Food and Agricultural Organisation of the United Nations: Rome, Italy, 2010. [Google Scholar]
- European Food Safety Authority. Scientific opinion on dietary reference values for fats, including saturated fatty acids, polyunsaturated fatty acids, monounsaturated fatty acids, trans fatty acids and cholesterol. EFSA J. 2010, 8, 1461. [Google Scholar]
- Scientific Advisory Committee on Nutrition/Committee on Toxicity. Advice on Fish Consumption: Benefits and Risks; TSO: London, UK, 2004. [Google Scholar]
- Calder, P.C. Very long-chain n-3 fatty acids and human health: Fact, fiction and the future. Proc. Nutr. Soc. 2018, 77, 52–72. [Google Scholar] [CrossRef]
| Innate (Natural) Immunity | Acquired (Adaptive) Immunity | ||
|---|---|---|---|
| Barriers | Cellular Components | Cell-Mediated Immunity | Humoral Immunity |
| Skin Mucosal surfaces Mucus Antimicrobial proteins in secretions Acid pH of stomach | Granulocytes (Neutrophils, Basophils, Eosinophils, Mast cells) Phagocytes (Neutrophils, Macrophages, Dendritic cells) Inflammatory response Natural killer cells Other innate cells (includes Innate lymphoid cells, Mucosal associated invariant T cells) | T lymphocytes (Helper, Cytotoxic, Regulatory, Others) Cytokines | B lymphocytes Antibodies |
| Memory response | |||
| Reference(s) | Period of Intervention | Intervention (g/day) | Control | Effect of n-3 LCPUFAs on Infant Immune Outcomes | Effect of n-3 LCPUFAs on Infant/Child Clinical Outcomes |
|---|---|---|---|---|---|
| [69] | From week 22 of pregnancy until birth | EPA 0.15 plus DHA 0.5 in milk-based supplement | Milk-based supplement without n-3 LCPUFA | Cord blood: Lower CCR4, IL-4 and IL-13 mRNA; Higher TGF-β mRNA; No effect on CRTH2, CXCR3, IL-1 or IFN-γ mRNA | - |
| [70,71,72,73] | From week 20 of pregnancy until birth | EPA 1.02 plus DHA 2.07 in capsules | Olive oil capsules | Cord blood: Lower IL-13; No effect on numbers of T, helper T, cytotoxic T, B or natural killer cells; Lower LTB4 production by neutrophils; Higher percentage of CD34+ progenitor cells (but no effect on cytokine or chemokine receptor expression on these cells); Lower IL-10 production by mononuclear cells stimulated with cat allergen or house dust mite allergen (trend with egg allergen); Trends to lower IL-5, IL-13 and IFN-γ production by mononuclear cells stimulated with cat, egg or housedust mite allergen | At one year of age: Less likelihood of severe atopic dermatitis; Trends for less likelihood of SPT positivity to egg and less likelihood of asthma; No effect on likelihood of food allergy, atopic dermatitis, wheeze, any SPT positivity and SPT positivity to allergens other than egg |
| [74,75] | From week 25 of pregnancy until week 15 of lactation | EPA 1.6 plus DHA 1.1 in capsules | Soybean oil capsules | - | At one year of age: Less likelihood of SPT positivity, SPT positivity to egg, having IgE-associated atopic dermatitis and having food allergy At two years of age: Less likelihood of SPT positivity, SPT positivity to egg or to food, having IgE-mediated food allergy, IgE-associated atopic dermatitis or IgE-associated disease |
| [76,77,78,79] | From week 12 to 20 until week 34 to 36 of pregnancy | EPA 1.06 plus DHA 0.274 or EPA 0.18 plus DHA 0.9 in capsules | Soybean oil capsules | Cord blood: Lower ratios of Th2 to Th1 chemokines (CCL22:CXCL10 and CCL17:CXCL10) in both n-3 LCPUFA groups; No effect on multiple cytokines; Higher 17-hydroxyDHA in both n-3 LCPUFA groups | At three years of age: Greater likelihood of atopic dermatitis in both n-3 LCPUFA groups |
| [80] | From week 12 of pregnancy until week 16 of lactation | DHA 0.4 from capsules | None | Blood at 4 months: No effect on IgA, IgM or IgG; No effect on lymphocyte or T helper, memory T helper or natural killer cell numbers; Lower percentage of cytotoxic T cells; Higher percentage of naïve T helper and memory cytotoxic T cells; Lower percentage of IFN-γ and IL-4 producing T helper and cytotoxic T cells in response to phorbol ester and ionomycin stimulation | - |
| [81,82] | From week 30 of pregnancy until birth | EPA 1.28 plus DHA 0.92 from capsules | Olive oil capsules | - | At 16 years of age: Less likelihood of asthma; allergic asthma; asthma, atopic dermatitis or allergic rhinitis; and allergic asthma, atopic dermatitis or allergic rhinitis At 24 years of age: Less likelihood of allergic asthma, diagnosed asthma, and requiring medication of asthma; Trend to less likelihood of requiring medication of allergic rhinitis; No effect on allergic sensitisation (allergen-specific IgE) |
| [83,84,85] | From week 21 of pregnancy until birth | EPA 0.1 plus DHA 0.8 from capsules | Blended vegetable oil capsules | - | At one year of age: Trend to less likelihood of IgE-associated allergic disease; Less likelihood of atopic dermatitis and sensitisation to egg; No effect on food allergy or respiratory infections In the first three years of life: Trend to less likelihood of IgE-associated allergic disease and to less likelihood of atopic dermatitis; No effect on food allergy or any sensitisation At three years of age: No effects on any outcome related to allergic disease At six years of age: No effects on any outcome related to allergic disease or on sensitisation to most allergens; Decreased likelihood to be sensitised to one species of house dust mite |
| [86] | From week 24 of pregnancy until birth | EPA 1.32 plus DHA 0.88 from capsules | Olive oil capsules | - | In the first five years of life: Less likelihood of persistent wheeze or asthma; Less likelihood of respiratory tract infections No effect on atopic dermatitis |
| [87,88] | From weeks 18 to 22 of pregnancy until birth | DHA 0.4 from capsules | Blended vegetable oil capsules | - | At one month of age: Shorter duration of cough, phlegm or wheezing At three months of age: Shorter duration of nasal congestion and all illnesses At six months of age: Shorter duration of fever, nasal secretion, difficulty breathing, rash and “other” illnesses Over the first 18 months of age: Less likelihood of some respiratory symptoms |
| [91] | First 4 months of lactation | EPA plus DHA 1.5 incorporated into muesli bars | Olive oil incorporated into muesli bars | Blood at 2.5 years of age: No effect on plasma IgE or IL-10 production by whole blood in response to lipopolysaccharide; Higher IFN-γ production by whole blood in response to lipopolysaccharide and a higher ratio of IFN-γ to IL-10 | At 2.5 years of age: No effect on atopic dermatitis, wheezing or food allergy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miles, E.A.; Childs, C.E.; Calder, P.C. Long-Chain Polyunsaturated Fatty Acids (LCPUFAs) and the Developing Immune System: A Narrative Review. Nutrients 2021, 13, 247. https://doi.org/10.3390/nu13010247
Miles EA, Childs CE, Calder PC. Long-Chain Polyunsaturated Fatty Acids (LCPUFAs) and the Developing Immune System: A Narrative Review. Nutrients. 2021; 13(1):247. https://doi.org/10.3390/nu13010247
Chicago/Turabian StyleMiles, Elizabeth A., Caroline E. Childs, and Philip C. Calder. 2021. "Long-Chain Polyunsaturated Fatty Acids (LCPUFAs) and the Developing Immune System: A Narrative Review" Nutrients 13, no. 1: 247. https://doi.org/10.3390/nu13010247
APA StyleMiles, E. A., Childs, C. E., & Calder, P. C. (2021). Long-Chain Polyunsaturated Fatty Acids (LCPUFAs) and the Developing Immune System: A Narrative Review. Nutrients, 13(1), 247. https://doi.org/10.3390/nu13010247





