Bioactive Compounds for Skin Health: A Review
Abstract
1. Introduction
2. Selected Substances of Importance for Skin Function
2.1. Vitamins
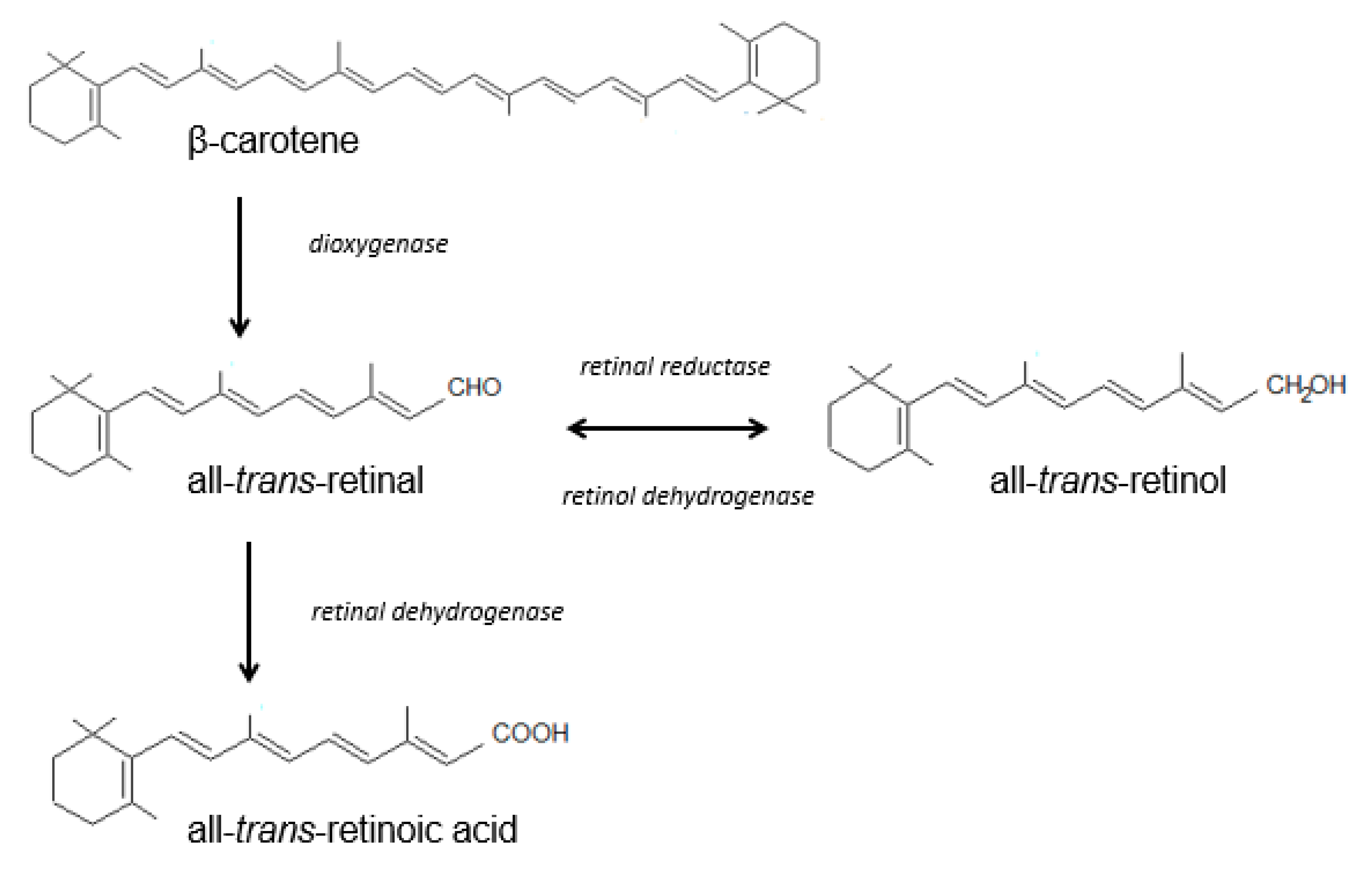
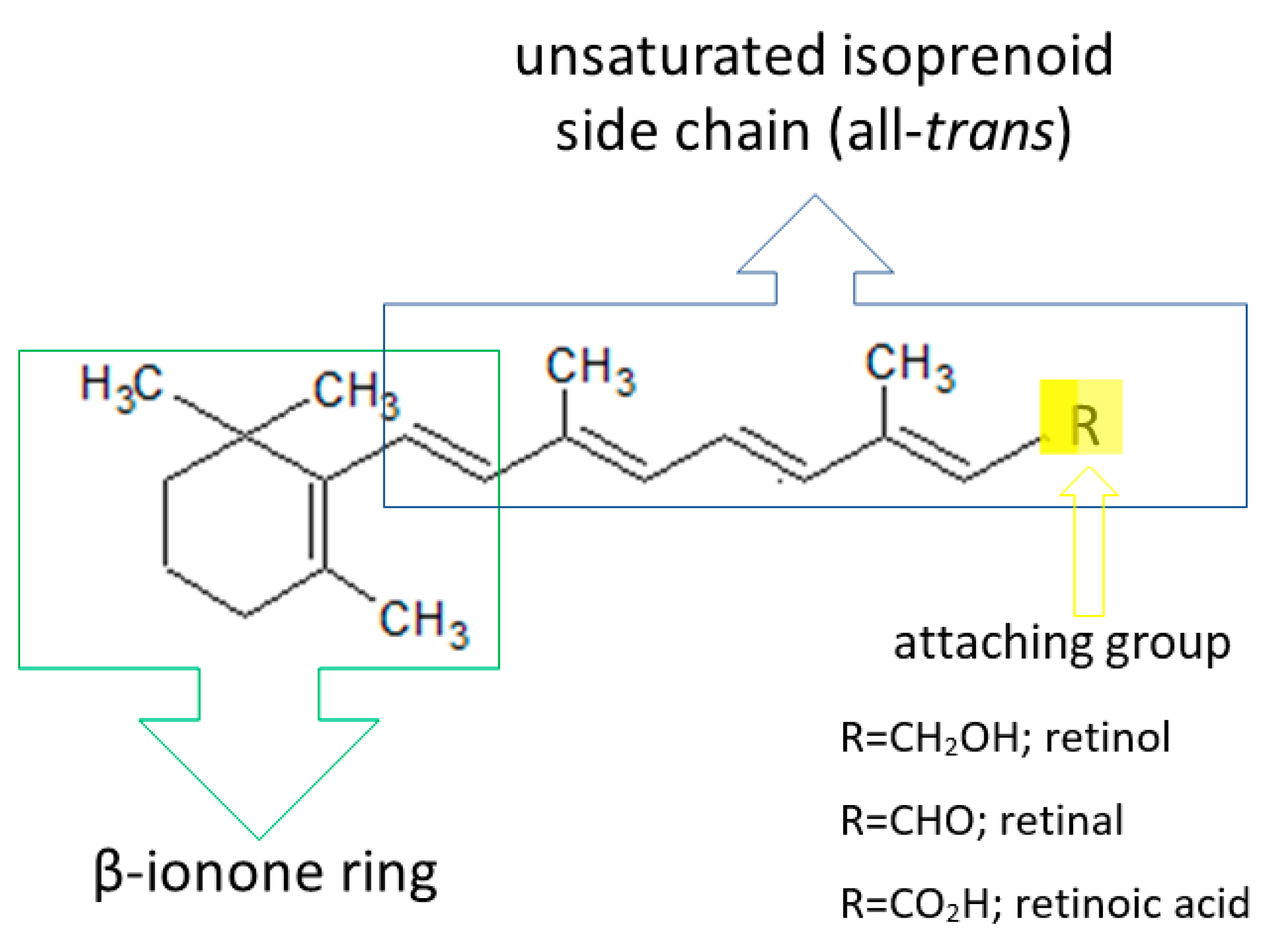
2.1.1. Vitamin A
2.1.2. Vitamin C
2.1.3. Vitamin E
2.2. Minerals
2.2.1. Selenium
2.2.2. Zinc
2.2.3. Copper
2.2.4. Silicon
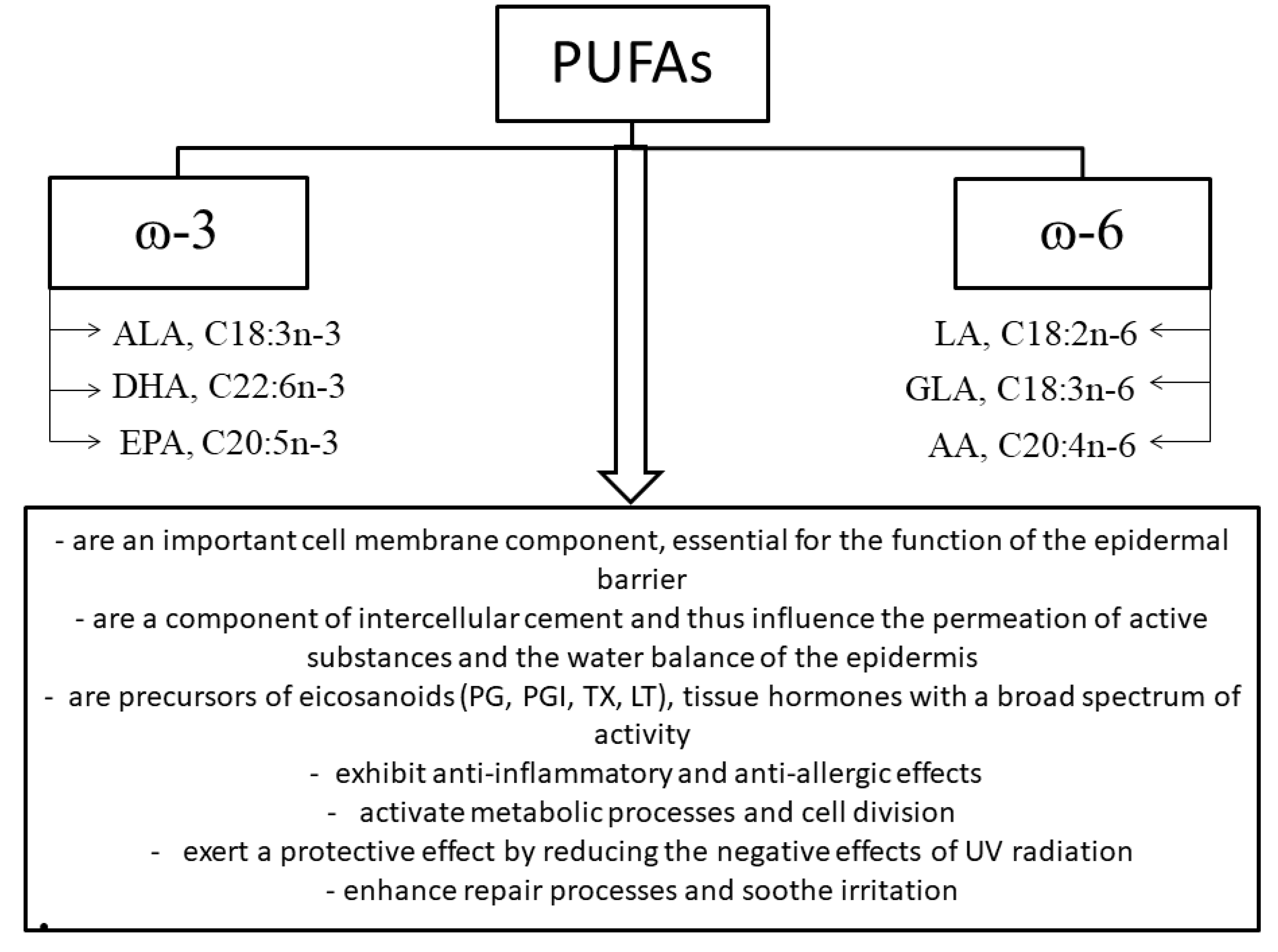
2.3. Fatty Acids
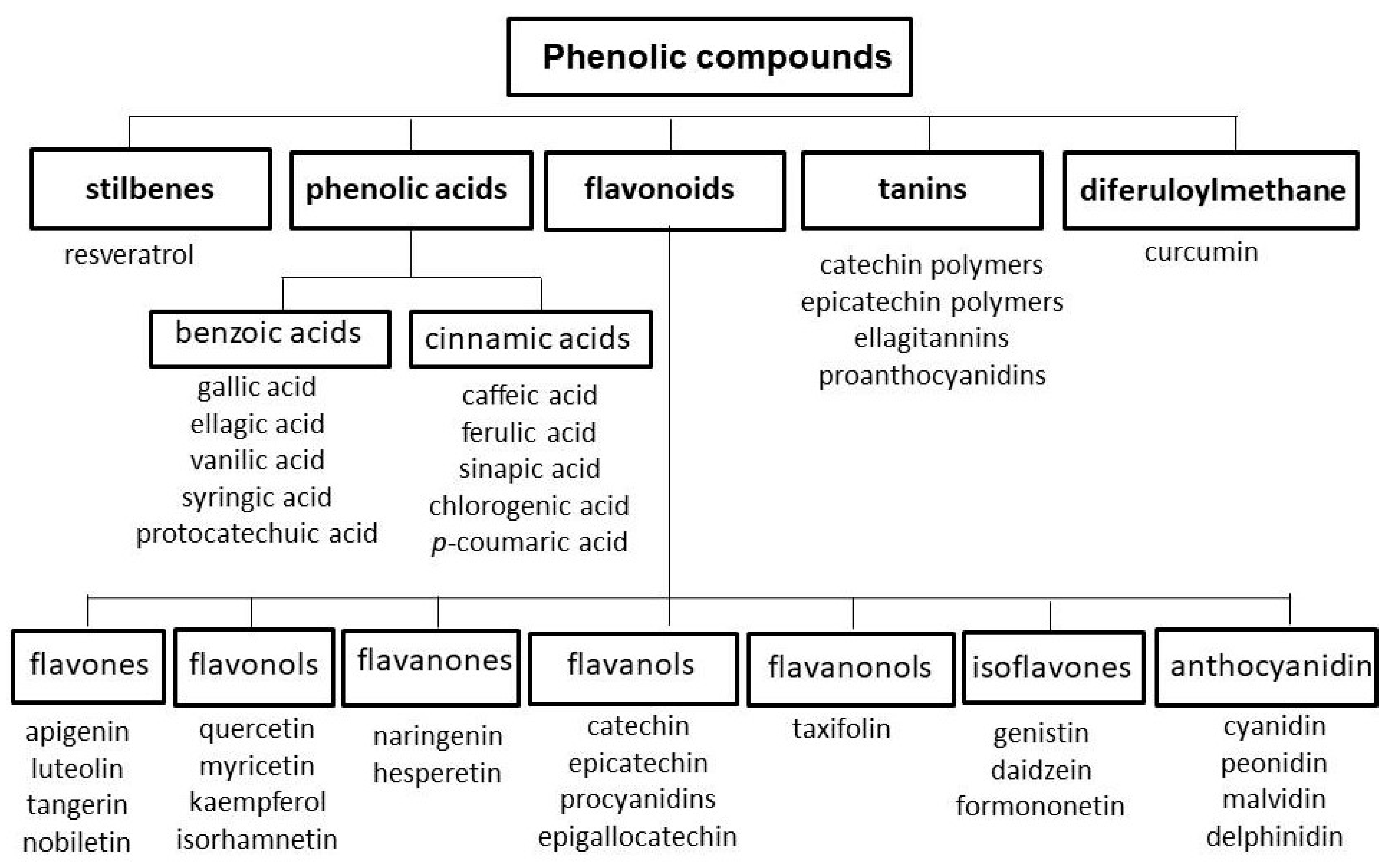
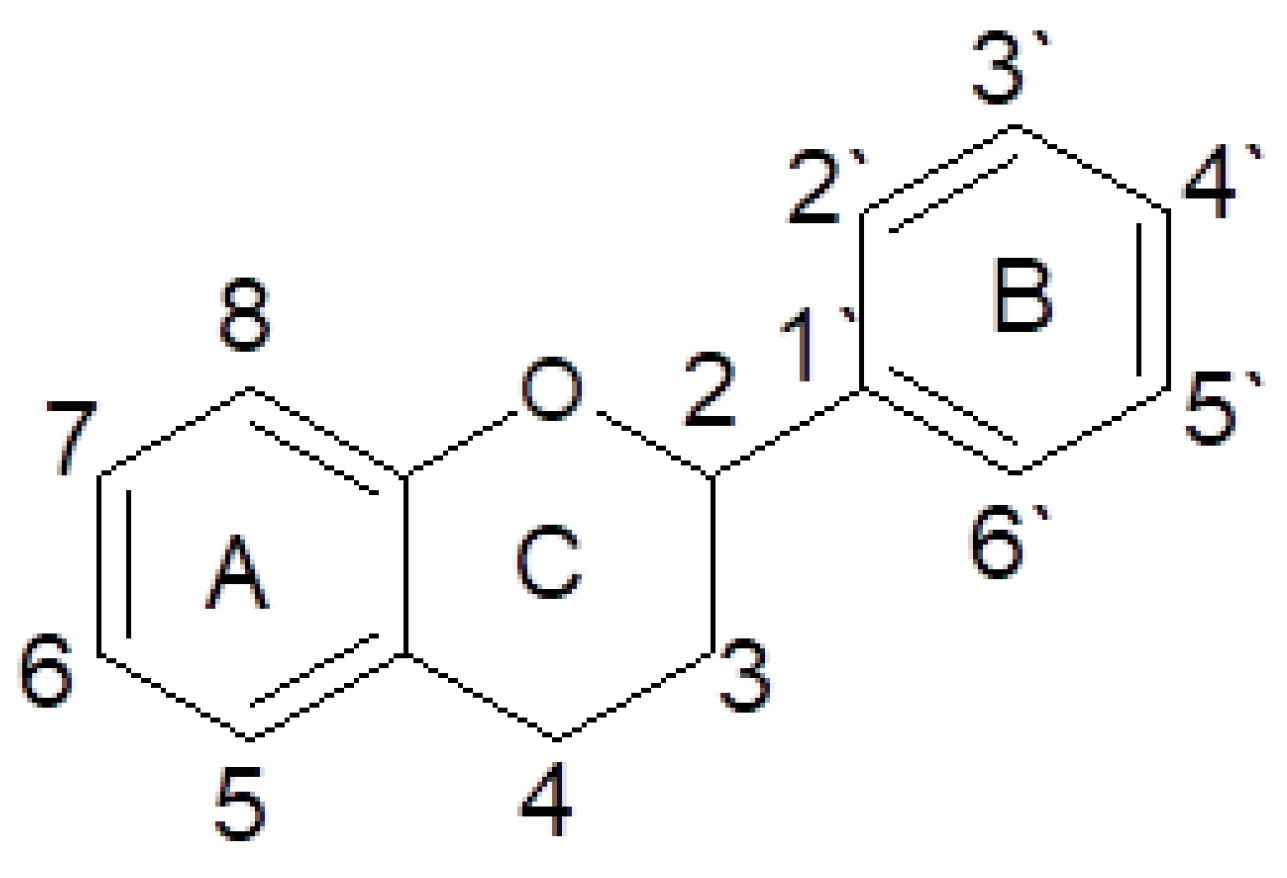
2.4. Polyphenols
2.5. Carotenoids
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ndiaye, M.A.; Nihal, M.; Wood, G.S.; Ahmad, N. Skin, reactive oxygen species, and circadian clocks. Antioxid. Redox Signal. 2014, 20, 2982–2996. [Google Scholar] [CrossRef]
- Tagami, H. Location-related differences in structure and function of the stratum corneum with special emphasis on those of the facial skin. Int. J. Cosmet. Sci. 2008, 30, 413–434. [Google Scholar] [CrossRef]
- Driskell, R.; Jahoda, C.; Chuong, C.M.; Watt, F.; Horsley, V. Defining dermal adipose tissue. Exp. Dermatol. 2014, 23, 629–631. [Google Scholar] [CrossRef]
- Venus, M.; Waterman, J.; McNab, I. Basic physiology of the skin. Surgery 2011, 29, 471–474. [Google Scholar]
- Wölfle, U.; Bauer, G.; Meinke, M.C.; Lademann, J.; Schempp, C.M. Reactive molecule species and antioxidative mechanisms in normal skin and skin aging. Skin Pharmacol. Physiol. 2014, 27, 316–332. [Google Scholar] [CrossRef]
- Poljšak, B.; Dahmane, R.G.; Godić, A. Intrinsic skin aging: The role of oxidative stress. Acta Dermatovener. 2012, 21, 33–36. [Google Scholar]
- Nishigori, C.; Hattori, Y.; Arima, Y.; Miyachi, Y. Photoaging and oxidative stress. Exp. Dermatol. 2003, 12, 18–21. [Google Scholar] [CrossRef]
- Binic, I.; Lazarevic, V.; Ljubenovic, M.; Mojsa, J.; Sokolovic, D. Skin ageing: Natural weapons and strategies. Evid. Based Complement. Alternat. Med. 2013. [Google Scholar] [CrossRef]
- Poljšak, B.; Dahmane, R.G. Free radicals and extrinsic skin aging. Dermatol. Res. Pract. 2012, 1–10. [Google Scholar] [CrossRef]
- Biesalski, H.K.; Berneburg, M.; Grune, T.; Kerscher, M.; Krutmann, J.; Raab, W.; Reimann, J.; Reuther, T.; Robert, L.; Schwarz, T. Hohenheimer Consensus Talk. Oxidative and premature skin ageing. Exp. Dermatol. 2003, 12, 3–15. [Google Scholar] [CrossRef]
- Pinnel, S.R. Cutaneous photodamage, oxidative stress, and topical antioxidant protection. J. Am. Acad. Dermatol. 2003, 48, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Masaki, H. Role of antioxidants in the skin: Anti-aging effects. J. Dermatol. Sci. 2010, 58, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Kang, S.; Varani, J.; Bata-Csorgo, Z.; Wan, Y.; Datta, S.; Voorhees, J.J. Mechanisms of photoaging and chronological skin aging. Arch. Dermatol. 2002, 138, 1462–1470. [Google Scholar] [CrossRef] [PubMed]
- Kaimal, S.; Thappa, D.M. Diet in dermatology: Revisited. IndianJ. Dermatol. Venereol. Leprol. 2010, 76, 103–115. [Google Scholar]
- Boelsma, E.; van de Vijver, L.P.L.; Goldbohm, R.A.; Klöpping-Ketelaars, I.A.A.; Hendriks, H.F.J.; Roza, L. Human skin condition and its associations with nutrient concentrations in serum and diet. Am. J. Clin. Nutr. 2003, 77, 348–355. [Google Scholar] [CrossRef]
- Divya, S.A.; Sriharsha, M.; Narotham, R.K.; Krupa, S.N.; Siva, T.R.K. Role of diet in dermatological conditions. J. Nutr. Food Sci. 2015, 5, 1–7. [Google Scholar]
- Rezaković, S.; Pavlić, M.; Navratil, M.; Počanić, L.; Žužul, K.; Kostović, K. The impact of diet on common skin disorders. J. Nutr. Ther. 2014, 3, 149–155. [Google Scholar] [CrossRef]
- Cao, C.; Xiao, Z.; Wu, Y.; Ge, C. Diet and skin aging—from the perspective of food nutrition. Nutrients 2020, 12, 870. [Google Scholar] [CrossRef]
- Strasser, B.; Volaklis, K.; Fuchs, D.; Burtscher, M. Role of dietary protein and muscular fitness on longevity and aging. Aging Dis. 2018, 9, 119–132. [Google Scholar] [CrossRef]
- Balić, A.; Vlašić, D.; Žužul, K.; Marinović, B.; Bukvić Mokos, Z. Omega-3 versus omega-6 polyunsaturated fatty acids in the prevention and treatment of inflammatory skin diseases. Int. J. Mol. Sci. 2020, 21, 741. [Google Scholar] [CrossRef]
- Rawlings, A.V.; Scott, I.R.; Harding, C.R.; Bowser, P.A. Stratum corneum moisturization at the molecular level. J. Investig. Dermatol. 1994, 103, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Schurer, N.Y.; Plewig, G.; Elias, P.W. Stratum corneum lipid function. Dermatologica 1991, 183, 77–94. [Google Scholar] [CrossRef] [PubMed]
- Reuter, J.; Merfort, I.; Schempp, C.M. Botanicals in dermatology: An evidence-Based review. Am. J. Clin. Dermatol. 2010, 11, 247–267. [Google Scholar] [CrossRef] [PubMed]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Anunciato, T.P.; da Rocha Filho, P.A. Carotenoids and polyphenols in nutricosmetics, nutraceuticals, and cosmeceuticals. J. Cosmet. Dermatol. 2012, 11, 51–54. [Google Scholar] [CrossRef]
- Godswill, A.G.; Somtochukwu, I.V.; Ikechukwu, A.O.; Kate, E.C. Health benefits of micronutrients (vitamins and minerals) and their associated deficiency diseases: A systematic review. Int. J. Food. Sci. 2020, 3, 1–32. [Google Scholar]
- Khali, S.; Bardawil, T.; Stephan, C.; Darwiche, N.; Abbas, O.; Kibbi, A.G. Retinoids: A journey from the molecular structures and mechanisms of action to clinical uses in dermatology and adverse effects. J. Dermatolog. Treat. 2017, 8, 684–696. [Google Scholar] [CrossRef]
- Sorg, O.; Kuenzli, S.; Kaya, S.; Saurat, J.H. Proposed mechanisms of action for retinoid derivatives in the treatment of skin aging. J. Cosmetic. Dermato. 2005, 4, 237–244. [Google Scholar] [CrossRef]
- Mukherjee, S.; Date, A.; Patravale, P.; Korting, H.C.; Roeder, A.; Weindel, G. Retinoids in the treatment of skin aging: An overview of clinical efficacy and safety. Clin. Interv.Aging 2006, 1, 327–348. [Google Scholar] [CrossRef]
- Polcz, E.M.; Barbul, A. The Role of Vitamin A in Wound Healing. Nutr. Clin. Pract. 2019, 34, 695–700. [Google Scholar] [CrossRef]
- Bono, M.R.; Tejon, G.; Flores-Santibañez, F.; Fernandez, G.; Rosemblatt, M.; Sauma, D. Retinoic acid as a modulator of T cell immunity. Nutrients 2016, 8, 349. [Google Scholar] [CrossRef] [PubMed]
- Zasada, M.; Budzisz, E. Retinoids: Active molecules influencing skin structure formation in cosmetic and dermatological treatments. Postepy Dermatol. Alergol. 2019, 36, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Erkelens, M.N.; Mebius, R.E. Retinoic acid and immune homeostasis: A balancing act. Trends Immunol. 2017, 38, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, L.M.; Teixeira, F.M.E.; Sato, M. N Impact of retinoic acid on immune cells and inflammatory diseases. Mediat. Inflamm. 2018. [Google Scholar] [CrossRef]
- Ferreira, R.; Napoli, J.; Enver, T.; Bernardino, L.; Ferrrira, L. Advances and challenges in retinoid delivery systems in regenerative and therapeutic medicine. Nat. Commun. 2020, 1, 4265. [Google Scholar] [CrossRef]
- Sorg, O.; Antille, C.; Kaya, G.; Saurat, J.H. Retinoids in cosmeceuticals. Dermatol. Ther. 2006, 19, 289–296. [Google Scholar] [CrossRef]
- Chaudhuri, R.K.; Bojanowski, K. Bakuchiol: A retinol-like functional compound revealed by gene expression profiling and clinically proven to have anti-aging effects. Int. J. Cosmet. Sci. 2014, 36, 221–230. [Google Scholar] [CrossRef]
- Shao, Y.; Te, H.; Fisher, G.J.; Voorhees, J.J.; Quan, T. Molecular basis of retinol anti-ageing properties in naturally aged human skin in vivo. Int. J. Cosmet. Sci. 2017, 39, 56–65. [Google Scholar] [CrossRef]
- Törmä, H.; Brunnberg, L.; Vahlquist, A. Age-related variations in acyl-CoA: Retinol acyltransferase activity and vitamin A concentration in the liver and epidermis of hairless mice. Biochim. Biophys. Acta. 1987, 921, 254–258. [Google Scholar] [CrossRef]
- Manela-Azulay, M.; Bagatin, E. Cosmeceuticals vitamins. Clin. Dermatol. 2009, 27, 469–474. [Google Scholar] [CrossRef]
- Kang, S.; Duell, E.A.; Fisher, G.J. Application of retinol to human skin in vivo induces epidermal hyperplasia and cellular retinoid binding proteins characteristic of retinoic acid but without measurable retinoic acid levels or irritation. J. Investig. Dermatol. 1995, 105, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Varani, J.; Warner, R.L.; Gharaee-Kermani, M.; Phan, S.H.; Kang, S.; Chung, J.H.; Wang, Z.Q.; Datta, S.C.; Fisher, G.J.; Voorhees, J.J. Vitamin A antagonizes decreased cell growth and elevated collagen degrading matrix metaloproteinases and stimulates collagen accumulation in naturally aged human skin. J. Investig. Dermatol. 2000, 114, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Piérard-Franchimont, C.; Castelli, D.; Cromphaut, I.V. Tensile properties and contours of aging facial skin. A controlled double-blind comparative study of the effects of retinol, melibose-lactose and their association. Skin Res. Technol. 1998, 4, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Antille, C.; Tran, C.; Sorg, O.; Carraux, P.; Didierjean, L.; Saurat, J.H. Vitamin A exerts a photoprotective action in skin by absorbing ultraviolet B radiation. J. Investig. Dermatol. 2003, 121, 1163–1167. [Google Scholar] [CrossRef]
- Fisher, G.J.; Talwar, H.S.; Lin, J.; Voorhees, J.J. Molecular mechanisms of photoaging in human skin in vivo and their prevention by all-trans retinoic acid. Photochem. Photobiol. 1999, 69, 154–157. [Google Scholar] [CrossRef]
- Hacışevki, A. An overview of ascorbic acid biochemistry. J. Fac. Pharm. Ankara. 2009, 38, 233–255. [Google Scholar]
- Lykkesfeldt, J.; Tveden-Nyborg, P. The pharmacokinetics of vitamin C. Nutrients 2019, 11, 2412. [Google Scholar] [CrossRef]
- Janda, K.; Kasprzak, M.; Wolska, J. Vitamin C – structure, properties, occurrence and functions. Pom. J. Life Sci. 2015, 61, 419–425. [Google Scholar]
- Pullar, J.M.; Carr, A.C.; Vissers, M.C.M. The roles of vitamin C in skin health. Nutrients 2017, 9, 866–892. [Google Scholar]
- Rhie, G.; Shin, M.H.; Seo, J.Y.; Choi, W.W.; Cho, K.H.; Kim, K.H.; Park, K.C.; Eun, H.C.; Chung, J.H. Aging-and photoaging-dependent changes of enzymic and nonenzymic antioxidants in the epidermis and dermis of human skin in vivo. J. Investig. Dermatol. 2001, 117, 1212–1217. [Google Scholar] [CrossRef]
- Shindo, Y.; Witt, E.; Han, D.; Epstein, W.; Packer, L. Enzymic and non-enzymic antioxidants in epidermis and dermis of human skin. J. Investig. Dermatol. 1994, 102, 122–124. [Google Scholar] [CrossRef] [PubMed]
- McArdle, F.; Rhodes, L.E.; Parslew, R.; Jack, C.I.; Friedmann, P.S.; Jackson, M.J. UVR-induced oxidative stress in human skin in vivo: Effects of oral vitamin C supplementation. Free Radic. Biol. Med. 2002, 33, 1355–1362. [Google Scholar] [CrossRef]
- Steiling, H.; Longet, K.; Moodycliffe, A.; Mansourian, R.; Bertschy, E.; Smola, H.; Mauch, C.; Williamson, G. Sodium-dependent vitamin C transporter isoforms in skin: Distribution, kinetics, and effect of UVB-induced oxidative stress. Free Radic. Biol. Med. 2007, 43, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Butler, J.D.; Bergsten, P.; Welch, R.W.; Levine, M. Ascorbic acid accumulation in human skin fibroblasts. Am. Soc. Clin. Nutr. 1991, 54, 1144–1146. [Google Scholar] [CrossRef]
- Ponec, M.; Weerheim, A.; Kempenaar, J.; Mulder, A.; Gooris, G.S.; Bouwstra, J.; Mommaas, A.M. The formation of competent barrier lipids in reconstructed human epidermis requires the presence of vitamin C. J. Investig. Dermatol. 1997, 109, 348–355. [Google Scholar] [CrossRef]
- Wang, K.; Jiang, H.; Li, W.; Qiang, M.; Li, H. Role of Vitamin C in Skin Diseases. Front. Physiol. 2018, 9, 819. [Google Scholar] [CrossRef]
- Savini, I.; Valeria, M.; Duranti, G.; Avigliano, L.; Catani, M.V.; Melino, G. Characterization of keratinocyte differentiation induced by ascorbic acid: Protein kinase C involvement and vitamin C homeostasis. J. Investig. Derm. 2002, 118, 372–379. [Google Scholar] [CrossRef]
- Pasonen-Seppanen, S.; Suhonen, T.M.; Kirjavainen, M.; Suihko, E.; Urtti, A.; Miettinen, M.; Hyttinen, M.; Tammi, M.; Tammi, R. Vitamin C enhances differentiation of a continuous keratinocyte cell line (REK) into epidermis with normal stratum corneum ultrastructure and functional permeability barrier. Histochem. Cell Biol. 2001, 116, 287–297. [Google Scholar] [CrossRef]
- Cosgrove, M.C.; Franco, O.H.; Granger, S.P.; Murray, P.G.; Mayes, A.E. Dietary nutrient intakes and skin-aging appearance among middle-aged American women. Am. J. Clin. Nutr. 2007, 86, 1225–1231. [Google Scholar] [CrossRef]
- Jones, D.P.; Kagan, V.E.; Aust, S.D.; Reed, D.J.; Omaye, S.T. Impact of nutrients on cellular lipid peroxidation and antioxidant defense system. Fundam. Appl. Toxicol. 1995, 26, 1–7. [Google Scholar] [CrossRef]
- Ou-Yang, H.; Stamatas, G.; Saliou, C.; Kollias, N. A chemiluminescence study of UVA-induced oxidative stress in human skin in vivo. J. Investig. Dermatol. 2004, 122, 1020–1029. [Google Scholar] [CrossRef] [PubMed]
- Offord, E.A.; Gautier, J.C.; Avanti, O.; Scaletta, C.; Runge, F.; Krämer, K.; Applegate, L.A. Photoprotective potential of lycopene, beta-carotene, vitamin E, vitamin C and carnosic acid in UVA-irradiated human skin fibroblasts. Free Radic. Biol. Med. 2002, 32, 1293–1303. [Google Scholar] [CrossRef]
- Darr, D.; Dunston, H.; Faust, H.; Pinnell, S. Effectiveness of antioxidants (vitamin C and E) with and without sunscreens as topical photoprotectants. Acta Derm. Venereol. 1996, 76, 264–268. [Google Scholar] [PubMed]
- Placzek, M.; Gaube, S.; Kerkmann, U.; Gilbertz, K.P.; Herzinger, T.; Haen, E.; Przybilla, B. Ultraviolet B-induced DNA damage in human epidermis is modified by the antioxidants ascorbic acid and D-alpha-tocopherol. J. Investig. Dermatol. 2005, 124, 304–307. [Google Scholar] [CrossRef]
- Eberlein-König, B.; Ring, J. Relevance of vitamins C and E in cutaneous photoprotection. J. Cosmet. Dermatol. 2005, 4, 4–9. [Google Scholar] [CrossRef]
- d’Ischia, M.; Wakamatsu, K.; Cicoira, F.; Di, M.E.; Garcia-Borron, J.C.; Commo, S.; Galván, I.; Ghanem, G.; Kenzo, K.; Meredith, P.; et al. Melanins and melanogenesis: From pigment cells to human health and technological applications. Pigment Cell Melanoma Res. 2015, 28, 520–544. [Google Scholar] [CrossRef]
- Panich, U.; Tangsupa-a-nan, V.; Onkoksoong, T.; Kongtaphan, K.; Kasetsinsombat, K.; Akarasereenont, P.; Wongkajornsilp, A. Inhibition of UVA-mediated Melanogenesis by Ascorbic Acid through Modulation of Antioxidant Defense and Nitric Oxide System. Arch. Pharm. Res. 2011, 34, 811–820. [Google Scholar] [CrossRef]
- Choi, Y.K.; Rho, Y.K.; Yoo, K.H.; Lim, Y.Y.; Li, K.; Kim, B.J.; Seo, S.J.; Kim, M.N.; Hong, C.K.; Kim, D.S. Effects of vitamin C vs. multivitamin on melanogenesis: Comparative study in vitro and in vivo. Int. J. Dermatol. 2010, 49, 218–226. [Google Scholar] [CrossRef]
- Shimada, Y.; Tai, H.; Tanaka, A.; Ikezawa-Suzuki, I.; Takagi, K.; Yoshida, Y.; Yoshie, H. Effects of ascorbic acid on gingival melanin pigmentation in vitro and in vivo. J. Periodontol. 2009, 80, 317–323. [Google Scholar] [CrossRef]
- Chawla, S. Split face comparative study of microneedling with PRP versus microneedling with vitamin C in treating atrophic post acne scars. J. Cutan. Aesthet. Surg. 2014, 7, 209–212. [Google Scholar] [CrossRef]
- Basketter, D.A.; White, I.R.; Kullavanijaya, P.; Tresukosol, P.; Wichaidit, M.; McFadden, J.P. Influence of vitamin C on the elicitation of allergiccontact dermatitis top-phenylenediamine. Contact Dermat. 2016, 74, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Soodgupta, D.; Kaul, D.; Kanwar, A.J.; Parsad, D. Modulation of LXR-αand the effector genes by Ascorbic acid and Statins in psoriatic keratinocytes. Mol. Cell. Biochem. 2014, 397, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Schober, S.M.; Peitsch, W.K.; Bonsmann, G.; Metze, D.; Thomas, K.; Goerge, T.; Luger, T.A.; Schneider, S.W. Early treatment with rutoside and ascorbic acid is highly effectivefor progressive pigmented purpuric dermatosis. J. Dtsch. Dermatol. Ges. 2016, 12, 1112–1119. [Google Scholar]
- Zervoudis, S.; Iatrakis, G.; Peitsidis, P.; Peitsidou, A.; Papandonopolos, L.; Nikolopoulou, M.K.; Papadopoulos, L.; Vladareanu, R. Complementary treatment with oralpidotimod plus vitamin C after laser vaporization for female genital warts: Aprospective study. J. Med. Life 2010, 3, 286–288. [Google Scholar]
- Don, P.; Iuga, A.; Dacko, A.; Hardick, K. Treatment of vitiligo with broadband ultraviolet B and vitamins. Int. J. Dermatol. 2006, 45, 63–65. [Google Scholar] [CrossRef]
- Khadangi, F.; Azzi, A. Vitamin-The Next 100 Years. IUBMB Life 2019, 71, 411–415. [Google Scholar] [CrossRef]
- Abraham, A.; Kattoor, A.J.; Saldeen, T.; Mehta, J.L. Vitamin E and its anticancer effects. Crit. Rev. Food. Sci. Nutr. 2019, 59, 2831–2838. [Google Scholar] [CrossRef]
- Traber, M.G.; Atkinson, J. Vitamin E, antioxidant and nothing more. Free. Rad. Biol. Med. 2007, 43, 4–15. [Google Scholar] [CrossRef]
- Mustacich, D.J.; Bruno, R.S.; Traber, M.G. Vitamin E. Vitam. Horm. 2007, 76, 1–21. [Google Scholar]
- Lee, G.Y.; Han, S.N. The role of Vitamin E in Immunity. Nutrients 2018, 10, 1614. [Google Scholar] [CrossRef]
- Ichihashi, M.; Funasaka, Y.; Ohashi, A.; Chacraborty, A.; Ahmed, N.U.; Ueda, M.; Osawa, T. The inhibitory effect of DL-alpha-tocopheryl ferulate in lecithin on melanogenesis. Anticancer Res. 1999, 19, 3769–3774. [Google Scholar] [PubMed]
- Keen, M.A.; Hassan, I. Vitamin E in dermatology. Indian. Dermatol. Online J. 2016, 7, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Dahiaya, A.; Romano, J.F. Cosmeceuticals: A review of their use for aging and photoaged skin. Cosmet. Dermatol. 2006, 19, 479–484. [Google Scholar]
- Tsoureli-Nikita, E.; Hercogova, J.; Lotti, T.; Menchini, G. Evaluation of dietary intake of vitamin E in the treatment of atopic dermatitis: A study of the clinical course and evaluation of the immunoglobulin E serum levels. Int. J. Dermatol. 2002, 41, 146–150. [Google Scholar] [CrossRef]
- Butt, H.; Mehmood, A.; Ali, M.; Tasneem, S.; Tarar, M.N.; Riazuddin, S. Vitamin E preconditioning alleviates in vitro thermal stress in cultured human epidermal keratinocytes. Life Sci. 2019, 239, 116–972. [Google Scholar] [CrossRef]
- Baumann, L.S.; Spencer, J. The Effects of Topical Vitamin E on the Cosmetic Appearance of Scars. Dermatol. Surg. 1999, 25, 311–315. [Google Scholar] [CrossRef]
- Park, K. Role of micronutrients in skin health and function. Biomol. Ther. 2015, 23, 207–217. [Google Scholar] [CrossRef]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R.; Collings, R.; Ford, D.; Hesketh, J.H.; Hurst, R. Selenium in human health and disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef]
- Cai, Z.; Zhang, J.; Li, H. Selenium, aging and aging-related diseases. Aging Clin. Exp. Res. 2019, 31, 1035–1047. [Google Scholar] [CrossRef]
- Zhu, X.; Jiang, M.; Song, E.; Jiang, X.; Song, Y. Selenium deficiency sensitizes the skin for UVB-induced oxidative damage and inflammation which involved the activation of p38 MAPK signaling. Food Chem. Toxicol. 2015, 75, 139–145. [Google Scholar] [CrossRef]
- Jobeili, L.; Rousselle, P.; Béal, D.; Blouin, E.; Roussel, AM.; Damour, O.; Rachidi, W. Selenium preserves keratinocyte stemness and delays senescence by maintaining epidermal adhesion. Aging 2017, 9, 2302–2315. [Google Scholar] [CrossRef] [PubMed]
- Favrot, C.; Beal, D.; Blouin, E.; Leccia, M.E.; Roussel, A.M. Age-dependent protective effect of selenium against UVA irradiation in primary human keratinocytes and the associated DNA repair signature. Oxid. Med. Cell. Longev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Jung, H.J.; Choi, J.S.; Nam, T.J. Anti-wrinkle effects of a tuna heart H2O fraction on Hs27 human fibroblasts. Int. J. Mol. Med. 2016, 37, 92–98. [Google Scholar] [CrossRef] [PubMed]
- van der Pols, J.C.; Heinen, M.M.; Hughes, M.C.; Ibiebele, T.I.; Marks, G.C.; Green, A.C. Serum antioxidants and skin cancer risk: An 8-year community-based follow-up study. Cancer Epidemiol. Biomark. Prev. 2009, 18, 1167–1173. [Google Scholar] [CrossRef]
- Combos, G.F.; Clark, L.C.; Turnbull, B.W. Reduction of cancer mortality and incidence by selenium supplementation. Med Klin 1997, 92, 42–45. [Google Scholar] [CrossRef]
- Duffield-Lillico, A.J.; Slate, E.H.; Reid, M.E.; Turnbull, B.W.; Wilkins, P.A.; Jr, G.F.C.; Park, H.K.; Gross, E.G.; Graham, G.F.; Stratton, M.S.; et al. Selenium supplementation and secondary prevention of nonmelanoma skin cancer in a randomized trial. J. Natl. Cancer. Inst. 2003, 95, 1477–1481. [Google Scholar] [CrossRef]
- Devi, C.B.; Nandakishore, Th.; Sangeeta, N.; Basar, G.; Deni, N.O.; Sungdirenla, J.; Singh, M.A. Zinc in human health. IOSR-JDMS 2014, 13, 18–23. [Google Scholar] [CrossRef]
- Gupta, M.; Mahajan, V.K.; Mehta, K.S.; Chauhan, P.S. Zinc therapy in dermatology: A review. Dermatol. Res. Pract. 2014. [CrossRef]
- Ogawa, Y.; Kinoshita, M.; Shimada, S.; Kawamura, T. Zinc and skin disorders. Nutrients 2018, 10, 199. [Google Scholar] [CrossRef]
- Manet, W. Zinc biochemistry: From a single zinc enzyme to a key element of life. Adv. Nutr. 2013, 4, 82–91. [Google Scholar]
- Fraga, C.G. Relevance, essentiality and toxicity of trace elements in human health. Mol. Aspects Med. 2005, 26, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Michaelsson, G.; Ljunghall, K.; Danielson, B.G. Zinc in epidermis and dermis in healthy subjects. Acta Derm. Venereol. 1980, 60, 295–299. [Google Scholar] [PubMed]
- Inoue, Y.; Hasegawa, S.; Ban, S.; Yamada, T.; Date, Y. ZIP2 protein, a zinc transporter, is associated with keratinocyte differentiation. J. Biol. Chem. 2014, 289, 21451–21462. [Google Scholar] [CrossRef] [PubMed]
- Vallee, B.L.; Galdes, A. The metallobiochemistry of zinc enzymes. Adv. Enzymol. Relat. Areas Mol. Biol. 1984, 56, 283–430. [Google Scholar]
- Klug, A. The discovery of zinc fingers and their applications in gene regulation and genome manipulation. Annu. Rev. Biochem. 2010, 79, 213–231. [Google Scholar] [CrossRef]
- Vollmer, D.L.; West, V.A.; Lephart, E.D. Enhancing skin health: By oral administration of natural compounds and minerals with implications to the dermal microbiome. Int. J. Mol. Sci. 2018, 19, 3059. [Google Scholar] [CrossRef]
- Nitzan, Y.B.; Cohen, A.D. Zinc in skin pathology and care. J. Dermatolog. Treat. 2006, 17, 205–210. [Google Scholar] [CrossRef]
- Tang, X.; Shay, N.F. Zinc has an insulin-like effect on glucose transport mediated by phosphoinositol-3-kinaseand Akt in 3T3-L1 fibroblasts and adipocytes. J. Nutr. 2001, 131, 1414–1420. [Google Scholar] [CrossRef]
- Lin, P.H.; Sermersheim, M.; Li, H.; Lee, P.H.U.; Steinberg, S.M.; Ma, J. Zinc in wound healing modulation. Nutrients 2018, 10, 16. [Google Scholar] [CrossRef]
- Kim, J.E.; Yoo, S.R.; Jeong, M.G.; Ko, J.Y.; Ro, Y.S. Hair zinc levels and the efficacy of oral zinc supplementation in patients with atopic dermatitis. Acta Derm. Venereol. 2014, 94, 558–562. [Google Scholar] [CrossRef]
- Mahoney, M.G.; Brennan, D.; Starcher, B.; Faryniarz, J.; Ramirez, J.; Parr, L.; Uitto, J. Extracellular matrix in cutaneous ageing: The effects of 0.1% copper-zincmalonate-containing cream on elastin biosynthesis. Exp. Dermatol. 2009, 18, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Sharquie, K.E.; Al-Mashhadani, S.A.; Salman, H.A. Topical 10% zinc sulfate solution for treatment of melisma. Dermatol. Surg. 2008, 34, 1346–1349. [Google Scholar] [PubMed]
- Sharquie, K.E.; Al-Mashhadani, S.A.; Noaimi, A.A.; Hasan, A.A. Topical zinc sulphate (25%) solution: A new therapy for actinic keratosis. J. Cutan. Aesthet. Surg. 2012, 5, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Sharquie, K.E. Topical therapy of xeroderma pigmentosa with 20% zinc sulfate solution. IASJ 2008, 7, 231–236. [Google Scholar]
- Faghihi, G.; Iraji, F.; Shahingohar, A.; Saidat, A.H. The efficacy of 0.05% Clobetasol + 2.5% zinc sulphate cream versus 0.05% Clobetasol alone cream in the treatment of the chronich and eczema: A double-blind study. J. Eur. Acad. Dermatol. Venereol. 2008, 22, 531–536. [Google Scholar] [CrossRef]
- Sharquie, K.E.; Najim, R.A.; Al-Salman, H.N. Oral zinc sulfate in the treatment of rosacea: A double-blind, placebo-controlled study. Int. J. Dermatol. 2006, 45, 857–861. [Google Scholar] [CrossRef]
- Sharquie, K.E.; Noaimi, A.A.; Shwail, E.R. Oral zinc sulphate in treatment of alopecia areata (Double Blind; Cross-Over Study). J. Clin. Exp. Dermatol. Res. 2012, 3, 150. [Google Scholar] [CrossRef]
- Borkow, G. Using Copper to Improve the Well-Being of the Skin. Curr. Chem. Biol. 2014, 8, 89–102. [Google Scholar] [CrossRef]
- Nishito, Y.; Kambe, T. Absorption Mechanisms of Iron, Copper, and Zinc: An Overview. J. Nutr. Sci. Vitaminol. 2018, 64, 1–7. [Google Scholar] [CrossRef]
- Linder, M.C.; Wooten, L.; Cerveza, P.; Cotton, S.; Shulze, R.; Lomeli, N. Copper transport. Am. J. Clin. Nutr. 1998, 67, 965–971. [Google Scholar] [CrossRef]
- Karlin, K.D. Metalloenzymes, structural motifs, and inorganic models. Science 1993, 261, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Uauy, R.; Olivares, M.; Gonzalez, M. Essentiality of copper in humans. Am. J. Clin. Nutr. 1998, 67, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Borkow, G.; Gabbay, J. Copper as a biocidal tool. Curr. Med. Chem. 2005, 12, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Santo, C.E.; Quaranta, D.; Grass, G. Antimicrobial metallic copper surface kill Staphylococcus haemolyticus via membrane demage. Microbiol. Open 2012, 1, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Grass, G.; Rensing, C.; Solioz, M. Metallic copper as an antimicrobial surface. Appl. Environ. Microbiol. 2011, 77, 1541–1547. [Google Scholar] [CrossRef] [PubMed]
- Philips, N.; Hwang, H.; Chauhan, S.; Leonardi, D.; Gonzalez, S. Stimulation of cell proliferation and expression of matrix metalloproteinase-1 and interluekin-8 genes in dermal fibroblasts by copper. Connect. Tissue Res. 2010, 51, 224–229. [Google Scholar] [CrossRef]
- Wołonciej, M.; Milewska, E.; Roszkowska-Jakimiec, W. Trace elements as an activator of antioxidant enzymes. Postepy. Hig. Med. Dosw. 2016, 70, 1483–1498. [Google Scholar] [CrossRef]
- Olivares, C.; Solano, F. New insights into the active site structure and catalytic mechanism of tyrosinase and its related proteins. Pigment Cell Melanoma Res. 2009, 22, 750–760. [Google Scholar] [CrossRef]
- Chang, T.S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef]
- Pickart, L.; Margolina, A. Skin regenerative and anti-cancer actions of copper peptides. Cosmetics 2018, 5, 29. [Google Scholar] [CrossRef]
- Mazurowska, L.; Mojski, M. Biological activities of selected peptides: Skin penetration ability of copper complexes with peptides. J. Cosmet. Sci. 2008, 59, 59–69. [Google Scholar] [PubMed]
- Hostynek, J.J.; Maibach, H.I. Copper hypersensitivity: Dermatologic aspects-an overview. Rev. Environ. Health 2003, 18, 153–183. [Google Scholar] [CrossRef] [PubMed]
- Fage, S.W.; Faurschou, A.; Thyssen, J.P. Copper hypersensitivity. Contact Dermat. 2014, 71, 191–201. [Google Scholar] [CrossRef] [PubMed]
- De Araújo, L.A.; Addor, F.; Campos, P.M.B.G.M. Use of silicon for skin and hair care: An approach of chemical forms available and efficacy. An. Bras. Dermatol. 2016, 91, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.O.; Freire, É.S.; Polonini, H.C.; da Silva, P.J.L.C.; Brandão, M.A.F.; Raposo, N.R.B. Anti-aging effects of monomethylsilanetriol and maltodextrin-stabilized orthosilicic acid on nails, skin and hair. Cosmetics 2018, 5, 41. [Google Scholar] [CrossRef]
- Harlim, A.; Aisah, S.; Sihombing, R. Silicon level in skin tissues of normal female individuals. J. Pakistan Assoc. Dermatologists. 2018, 28, 134–138. [Google Scholar]
- Boguszewska-Czubara, A.; Pasternak, K. Silicon in medicine and therapy. J. Elem. 2011, 3, 489–497. [Google Scholar] [CrossRef]
- Barel, A.; Calomme, M.; Timchenko, A.; de Paepe, K.; Demeester, N.; Rogiers, V.; Clarys, P.; Vandenberghe, D. Effect of oral intake of choline-stabilized orthosilicic acid on skin, nails and hair in women with photodamaged skin. Arch. Dermatol. Res. 2005, 297, 147–153. [Google Scholar] [CrossRef]
- Mancinella, A. Silicon, a trace element essential for living organisms. Recent knowledge on its preventive role in atherosclerotic process, aging and neoplasms. Clin. Ter. 1991, 137, 343–350. [Google Scholar]
- Reffitt, D.M.; Ogston, N.; Jugdaohsingh, R.; Cheung, H.F.; Evans, B.A.; Thompson, R.P.; Powell, J.J.; Hampson, G.N. Orthosilicic acid stimulates collagen type 1 synthesis and osteoblastic differentiation in human osteoblast-like cells in vitro. Bone 2003, 32, 127–135. [Google Scholar] [CrossRef]
- Kalil, C.L.P.V.; Campos, V.; Cignachi, S.; Izidoro, J.V.; Reinehr, C.P.H.; Chaves, C. Evaluation of cutaneous rejuvenation associated with the use of ortho-silicic acid stabilized by hydrolyzed marine collagen. J. Cosmet. Dermatol. 2017, 17, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Jacob, S.; Al-Dhublab, B.; Attimarad, M.; Harsha, S. Basic considerations in the dermatokinetics of topical formulations. Bras. J. Pharmaceut. Sci. 2013, 49, 423–434. [Google Scholar] [CrossRef]
- Penniston, K.L.; Tanumihardjo, S.A. The acute and chronic toxic effects of vitamin A. Am. J. Clin. Nutr. 2006, 83, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.A.; Russell, R.M.; Stephensen, C.H.B.; Gannon, B.M.; Craft, N.E.; Haskell, M.J.; Lietz, G.; Schulze, K.; Raiten, D.J. Biomarkers of nutrition for development (BOND)-vitamin A overview. J. Nutr. 2016, 146, 1816–1848. [Google Scholar] [CrossRef] [PubMed]
- Arscott, S.A.; Howe, J.A.; Davis, C.R.; Tanumihardjo, S.A. Carotenoid profiles in provitamin A-containing fruits and vegetables affect the bioefficacy in Mongolian gerbils. Exp. Biol. Med. 2010, 235, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Szymańska, R.; Nowicka, B.; Kruk, J. Vitamin E-occurrence, biosynthesis by plants and functions in human nutrition. Mini. Rev. Med. Chem. 2017, 17, 1039–1052. [Google Scholar] [CrossRef]
- Lv, J.; Ai, P.; Lei, S.; Zhou, F.; Chen, S.; Zhang, Y. Selenium levels and skin diseases: Systematic review and meta-analysis. J. Trace. Elem. Med. Biol. 2020, 62, 126548. [Google Scholar] [CrossRef]
- Fine, J.D.; Tamura, T.; Johnson, L. Blood vitamin and trace metal levels in epidermolysis bullosa. Arch. Dermatol. 1989, 125, 374–379. [Google Scholar] [CrossRef]
- Ewing, C.I.; Gibbs, A.C.; Ashcroft, C.; David, T.J. Failure of oral zinc supplementation in atopic eczema. Eur. J. Clin. Nutr. 1991, 45, 507–510. [Google Scholar]
- Choudhary, S.V.; Gadegone, R.W.; Koley, S. Menkes Kinky hair disease. Indian J. Dermatol. 2012, 57, 407–409. [Google Scholar] [CrossRef]
- Hornych, A.; Oravec, S.; Girault, F.; Forette, B.; Horrobin, D.F. The effect of gamma-linolenic acid on plasma and membranelipids and renal prostaglandin synthesis in older subjects. Bratisl. Lek. Listy. 2002, 103, 101–107. [Google Scholar] [PubMed]
- Michalak, M.; Glinka, R. Plant oils in cosmetology and dermatology. Pol. J. Cosmetol. 2018, 21, 2–9. [Google Scholar]
- Correa, M.C.; Mao, G.; Saad, P.; Flach, C.R.; Mendelsohn, R.; Walters, R.M. Molecular interactions of plant oil components with stratum corneum lipids correlate with clinical measures of skin barrier function. Exp. Dermatol. 2014, 23, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Feingold, K.R.; Elias, P.M. Role of lipids in the formation and maintenance of the cutaneous permeability barier. Biochim. Biophys. Acta. 2014, 1841, 280–294. [Google Scholar] [CrossRef]
- Kapoor, R.; Huang, Y.-S. Gamma linolenic acid: An antiinflammatory omega-6 fatty acid. Curr. Pharm. Biotechno. 2006, 7, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Michalak, M.; Kiełtyka-Dadasiewicz, A. Oils from fruit seeds and their dietetic and cosmetic significance. Herba Pol. 2018, 64, 63–70. [Google Scholar] [CrossRef]
- Burris, J.; Rietkerk, W.; Woolf, K. Acne: The role of medical nutrition therapy. J. Acad. Nutr. Diet. 2013, 113, 416–430. [Google Scholar] [CrossRef]
- Cakir, A. Essential oil and fatty acid composition of the fruits of Hippophae rhamnoides L. (Sea Buckthorn) and Myrtus communis L. from Turkey. Biochem. Syst. Ecol. 2004, 32, 809–816. [Google Scholar] [CrossRef]
- Van Smeden, J.; Janssens, M.; Gooris, G.S.; Bouwstra, J.A. The important role of stratum corneum lipids for the cutaneous barrier function. Biochim. Biophys. Acta. 2014, 1841, 295–313. [Google Scholar] [CrossRef]
- Sabikhi, L.; Kumar, M.H.S. Fatty acid profile of unconventional oil seeds. Adv. Food. Nutr. Res. 2012, 67, 141–184. [Google Scholar]
- Simon, D.; Eng, P.A.; Borelli, S.; Kägi, R.; Zimmermann, C.; Zahner, C.; Drewe, J.; Hess, L.; Ferrari, G.; Lautenschlager, S.; et al. Gamma-linolenic acid levels correlate with clinical efficacy of evening primrose oil in patients with atopic dermatitis. Adv. Ther. 2014, 31, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Larmo, P.; Ulvinen, T.; Määttä, P.; Judin, V.-P. CO2-extracted blackcurrant seed oil for well-being of the skin. Innov. Food Technol. 2011, 68–69. [Google Scholar]
- Asadi-Samani, M.; Bahmani, M.; Rafieian-Kopaei, M. The chemical composition, botanical characteristic and biological activities of Borago officinalis: A review. Asian. Pac. J. Trop. Med. 2014, 7, 22–28. [Google Scholar] [CrossRef]
- Muggli, R. Systemic evening primrose oil improves the biophysical skin parameters of healthy adults. Int. J. Cosmet. Sci. 2005, 27, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Linnamaa, P.; Savolainen, J.; Koulu, L.; Tuomasjukka, S.; Kallio, H.; Yang, B.; Vahlberg, T.; Tahvonen, R. Blackcurrant seed oil for prevention of atopic dermatitis in newborns: A randomized, double-blind, placebo-controlled trial. Clin. Exp. Allergy 2010, 40, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Oomah, B.D.; Busson, M.; Godfrey, D.V.; Drover, J.C.G. Characteristics of hemp (Cannabis sativa L.) seed oil. Food Chem. 2002, 76, 33–43. [Google Scholar] [CrossRef]
- Montserrat-de la Paz, S.; Marín-Aguilar, F.; García-Giménez, M.D. Fernández-Arche, M.A. Hemp (Cannabis sativa L.) seed oil: Analytical and phytochemical characterization of the unsaponifiable fraction. J. Agric. Food Chem. 2014, 62, 1105–1110. [Google Scholar] [CrossRef]
- Razafimamonjison, G.; Tsy, J.M.L.P.; Randriamiarinarivo, M.; Ramanoelina, P.; Rasoarahona, J.; Fawbush, F.; Danthu, P. Fatty acid composition of baobab seed and its relationship with the genus Adansonia taxonomy. Chem. Biodiversity 2017, 14. [Google Scholar] [CrossRef]
- Osman, M.A. Chemical and nutrient analysis of baobab (Adansonia digitata) fruit and seed protein solubility. Plant Foods Hum. Nutrients 2004, 59, 29–33. [Google Scholar]
- Komane, B.M.; Vermaak, I.; Kamatou, G.P.P.; Summers, B.; Viljoen, A.M. Beauty in baobab: A pilot study of the safety and efficacy of Adansonia digitata seed oil. Rev. Bras. Farmacogn. 2017, 27, 1–8. [Google Scholar] [CrossRef]
- Momchilova, S.; Antonova, D.; Marekov, I.; Kuleva, L.; Nikolova-Damyanova, B. Fatty acids, triacylglycerols, and sterols in neem oil (Azadirachta indica A. Juss) as determined by a combination of chromatographic and spectral techniques. J. Liq. Chromatogr. Relat. Technol. 2007, 30, 11–25. [Google Scholar] [CrossRef]
- Campos, E.V.R.; de Oliveira, J.L.; Pascoli, M.; de Lima, R.; Fraceto, L.F. Neem oil and crop protection: From now to the future. Front. Plant Sci. 2016, 7, 1494. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, A.; Husain, A.; Mujeeb, M.; Khan, S.A.; Najmi, A.K.; Siddique, N.A.; Damanhouri, Z.A.; Anwar, F. A review on therapeutic potential of Nigella sativa: A miracle herb. Asian Pac. J. Trop. Biomed. 2013, 3, 337–352. [Google Scholar] [CrossRef]
- Ghorbanibirgani, A.; Khalili, A.; Rokhafrooz, D. Comparing Nigella sativa oil and fish oil in treatment of vitiligo. Iran Red. Crescent. Med. J. 2014, 16, 4515. [Google Scholar] [CrossRef]
- DebMandal, M.; Mandal, S. Coconut (Cocos nucifera L.: Arecaceae): In health promotion and disease prevention. Asian Pac. J. Trop. Med. 2011, 4, 241–247. [Google Scholar] [CrossRef]
- Yang, B.; Kalimo, K.O.; Mattila, L.M.; Kallio, S.E.; Katajisto, J.K.; Peltola, O.J.; Kallio, H.P. Effects of dietary supplementation with sea buckthorn (Hippophae¨rhamnoides) seed and pulp oils on atopic dermatitis. J. Nutr. Biochem. 1999, 10, 622–630. [Google Scholar] [CrossRef]
- Christaki, E. Hippophae rhamnoides L. (Sea Buckthorn): A potential source of nutraceuticals. Food Pub. Health 2012, 2, 69–72. [Google Scholar] [CrossRef]
- Wagemaker, T.A.; Rijo, P.; Rodrigues, L.M.; Campos, P.M.B.G.M.; Fernandes, A.S.; Rosado, C. Integrated approach in the assessment of skin compatibility of cosmetic formulations with green coffee oil. Int. J. Cosmet. Sci. 2015, 37, 506–510. [Google Scholar] [CrossRef]
- Oliveira, L.S.; Franca, A.S.; Mendonça, J.C.F.; Barros-Júnior, M.C. Proximate composition and fatty acids profile of green and roasted defective coffee beans. Food Sci. Technol. 2006, 39, 235–239. [Google Scholar] [CrossRef]
- Attia, R.S.; Abou-Gharbia, H.A. Evaluation and stabilization of wheat germ and its oil characteristics. Alex. J. Fd. Sci. Technol. 2011, 8, 31–39. [Google Scholar]
- Matthäus, B.; Özcan, M.M. Habitat effects on yield, fatty acid composition and tocopherol contents of prickly pear (Opuntiaficus-indica L.) seed oils. Sci. Horticulturae 2011, 131, 95–98. [Google Scholar] [CrossRef]
- Boucetta, K.Q.; Charrouf, Z.; Aguenaou, H.; Derouiche, A.; Bensouda, Y. Does argan oil have a moisturizing effect on the skin of postmenopausal women? Skin Res. Technol. 2013, 19, 356–357. [Google Scholar] [CrossRef]
- Guillaume, D.; Charrouf, Z. Argan oil and other argan products: Use in dermocosmetology. Eur. J. Lipid Sci. Technol. 2011, 113, 403–408. [Google Scholar] [CrossRef]
- Boucetta, K.Q.; Charrouf, Z.; Aguenaou, H.; Derouiche, A.; Bensouda, Y. The effect of dietary and/or cosmetic argan oil on postmenopausal skin elasticity. Clin. Interv. Aging 2015, 30, 339–349. [Google Scholar]
- Ziboh, V.A. The significance of polyunsaturated fatty acids in cutaneous biology. Lipids 1996, 31, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.E.; O’Farrell, S.; Jackson, M.J.; Friedmann, P.S. Dietary fish-oil supplementation in humans reduces UVB-erythemal sensitivity but increases epidermal lipid peroxidation. J. Investig. Derm. 1994, 103, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.E.; Shahbakhti, H.; Azurdia, M.R.; Moison, R.M.W.; Steenwinkel, M.-J.S.T.; Homburg, M.I.; Dean, M.P.; McArdle, F.; van Henegouwen, G.M.J.B.; Epe, B.; et al. Effect of eicosapentaenoic acid, an omega-3 polyunsaturated fatty acid, on UVR-related cancer risk in humans. An assessment of early genotoxic markers. Carcinogenesis 2003, 24, 919–925. [Google Scholar] [CrossRef]
- Michalak, M.; Glinka, R. Sources of vegetable dyes and their use in cosmetology. Pol. J. Cosmetol. 2017, 20, 196–205. [Google Scholar]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010, 64, 112–120. [Google Scholar] [CrossRef]
- Han, X.; Shen, T.; Lou, H. Dietary polyphenols and their biological significance. Int. J. Mol. Sci. 2007, 8, 950–988. [Google Scholar] [CrossRef]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects –A review. J. Funct. Foods 2015, 18, 820–897. [Google Scholar] [CrossRef]
- Jain, P.K.; Kharya, M.D.; Gajbhiye, A.; Sara, U.V.S.; Sharma, V.K. Flavonoids as nutraceuticals. A review. Herba Pol. 2010, 56, 105–117. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef]
- Epstein, H. Cosmeceuticals and polyphenols. Clin. Dermatol. 2009, 27, 475–478. [Google Scholar] [CrossRef]
- Nichols, J.A.; Katiyar, S.K. Skin photoprotection by natural polyphenols: Anti-inflammatory, antioxidant and DNA repair mechanisms. Arch. Dermatol. Res. 2010, 302, 71–83. [Google Scholar] [CrossRef]
- Zillich, O.V.; Schweiggert-Weisz, U.; Eisner, P.; Kerscher, M. Polyphenols as active ingredients for cosmetic products. Int. J. Cosmet. Sci. 2015, 37, 455–464. [Google Scholar] [CrossRef]
- Chuarienthong, P.; Lourith, N.; Leelapornpisid, P. Clinical efficacy comparison of anti-wrinkle cosmetics containing herbal flavonoids. Int. J. Cosmet. Sci. 2010, 32, 99–106. [Google Scholar] [CrossRef]
- Chen, L.; Hu, J.Y.; Wang, S.Q. The role of antioxidants in photoprotection: A critical review. J. Am. Acad. Dermatol. 2012, 67, 1013–1024. [Google Scholar] [CrossRef]
- Afag, F.; Mukhtar, H. Botanical antioxidants in the prevention of photocarcinogenesis and photoaging. Exp. Derm. 2006, 15, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Kumria, R.; Garg, M.; Gupta, M. Recent updates on free radicals scavenging flavonoids: An overview. Asian. J. Plant Sci. 2010, 9, 108–117. [Google Scholar] [CrossRef]
- Cho, H.S.; Lee, M.H.; Lee, J.W.; No, K.O.; Park, S.K.; Lee, H.S. Anti-wrinkling effects of the mixture of vitamin C, vitamin E, pycnogenol and evening primrose oil, and molecular mechanisms on hairless mouse skin caused by chronic ultraviolet B irradiation. Photodermatol. Photoimmunol. Photomed 2007, 23, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Svobodová, A.; Zdarilová, A.; Malisková, J.; Mikulková, H.; Walterová, D.; Vostalová, J. Attenuation of UVA-induced damage to human keratinocytes by silymarin. J. Dermatol. Sci. 2007, 46, 21–30. [Google Scholar] [CrossRef]
- Moore, J.O.; Yongyin, W.; Stebbins, W.G.; Gao, D.; Zhou, X.; Phelps, R.; Lebwohl, M.; Wei, H. Photoprotective effect of isoflavone genistein on ultraviolet B-induced pyrimidine dimer formation and PCNA expression in human reconstituted skin and its implications in dermatology and prevention of cutaneous carcinogenesis. Carcinogenesis 2006, 27, 1627–1635. [Google Scholar] [CrossRef]
- Lima, C.F.; Pereira-Wilson, C.; Rattan, S.I. Curcumin induces heme oxygenase-1 in normal human skin fibroblasts through redox signaling: Relevance for anti-aging intervention. Mol. Nutr. Food Res. 2011, 55, 430–442. [Google Scholar] [CrossRef]
- Afaq, F.; Adhami, V.M.; Ahmad, N. Prevention of short-term ultraviolet B radiation-mediated damages by resveratrol in SKH-1 hairless mice. Toxicol. Appl. Pharmacol. 2003, 186, 28–37. [Google Scholar] [CrossRef]
- Afaq, F.; Saleem, M.; Krueger, C.G.; Reed, J.D.; Mukhtar, H. Anthocyanin- and hydrolyzable tannin-rich pomegranate fruit extract modulates MAPK and NF-kappa B pathways and inhibits skin tumorigenesis in CD-1 mice. Int. J. Cancer 2005, 113, 423–433. [Google Scholar] [CrossRef]
- Khan, N.; Syed, D.N.; Pal, C.H.; Mukhtar, H.; Afag, F. Pomegranate fruit extract inhibits UVB-induced inflammation and proliferation by modulating NF-κB and MAPK signaling pathways in mouse skin. Photochem. Photobiol. 2012, 88, 1126–1134. [Google Scholar] [CrossRef]
- Kasai, K.; Yoshimura, M.; Koga, T.; Arii, M.; Kawasaki, S. Effects of oral administration of ellagic acid-rich pomegranate extract on ultraviolet-induced pigmentation in the human skin. J. Nutr. Sci. Vitaminol. 2006, 52, 383–388. [Google Scholar] [CrossRef]
- Henning, S.M.T.; Niua, Y.; Liua, Y.; Leea, N.H.; Harac, Y.; Thamesa, G.D.; Minuttia, R.R.; Carpentera, C.L.; Wang, H.; Heber, D. Bioavailability and antioxidant effect of epigallocatechin gallate administered in purified form versus as green tea extract in healthy individuals. J. Nutr. Biochem. 2005, 16, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Katiyar, S.K.; Afaq, F.; Perez, A.; Mukhtar, H. Green tea polyphenol (-)-epigallocatechin-3-gallate treatment of human skin inhibits ultraviolet radiation-induced oxidative stress. Carcinogenesis 2001, 22, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Vayalil, P.K.; Mittal, A.; Hara, Y.; Elmets, C.E.; Katiyar, S.K. Green tea polyphenols prevent ultraviolet light-induced oxidative damage and matrix metalloproteinases expression in mouse skin. J. Investig. Dermatol. 2004, 122, 1480–1487. [Google Scholar] [CrossRef] [PubMed]
- Chiu, A.E.; Chan, J.L.; Kern, D.G.; Kohler, S.; Rehmus, W.E.; Kimball, A.B. Double-blinded, placebo-controlled trial of green tea extracts in the clinical and histologic appearance of photoaging skin. Dermatol. Surg. 2005, 31, 855–860. [Google Scholar] [CrossRef]
- Higdon, J.V.; Frei, B. Tea catechins and polyphenols: Health effects, metabolism, and antioxidant functions. Crit. Rev. Food Sci. Nutr. 2003, 43, 89–143. [Google Scholar] [CrossRef]
- Rutter, K.; David, R.S.; Nalani, F.; Obrenovich, M.; Zito, M.; Starke-Reed, P.; Monnier, V.M. Green tea extract suppresses the age-related increase in collagen crosslinking and fluorescent products in C57BL/6Mice. Int. J. Vitam. Nutr. Res. 2003, 73, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Tuong, W.; Walker, L.; Sivamani, R.K. Polyphenols as novel treatment options for dermatological diseases: A systematic review of clinical trials. J. Dermatolog. Treat. 2015, 26, 381–388. [Google Scholar] [CrossRef]
- Beutner, S.; Bloedorn, B.; Frixel, S.; Hermandez, B.; Hoffmann, T.; Martin, H.; Mayer, B.; Noack, P.; Ruck, C.; Schmidt, M.; et al. Quantitative assessment of antioxidant properties of natural carotene in antioxidant functions. J. Sci. Food Argic. 2001, 81, 559–568. [Google Scholar] [CrossRef]
- Delgado-Vargas, F.; Jiménez, A.R.; Paredes-López, O. Natural pigments: Carotenoids, anthocyanins, and betalains-characteristics, biosynthesis, processing, and stability. Crit. Rev. Food Sci. Nutr. 2000, 40, 173–289. [Google Scholar] [CrossRef] [PubMed]
- Balić, A.; Mokos, M. Do we utilize our knowledge of the skin protective effects of carotenoids enough? Antioxidants 2019, 8, 259. [Google Scholar] [CrossRef] [PubMed]
- Eldahshan, O.A.; Singab, A.N.B. Carotenoids. J. Pharmacogn. Phytochem. 2013, 2, 225–234. [Google Scholar]
- Davinelli, S.; Nielsen, M.E.; Scapagnini, G. Astaxanthin in skin health, repair, and disease: A comprehensive review. Nutrients 2018, 10, 522. [Google Scholar] [CrossRef] [PubMed]
- Mathews-Roth, M.M. Plasma concentration of carotenoids after large doses of beta-carotene. Am. J. Clin. Nutr. 1990, 52, 500–501. [Google Scholar] [CrossRef]
- Bendich, A. Biological functions of dietary carotenoids. Ann. N. Y. Acad. Sci. 1993, 691, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Mezzomo, N.; Ferreira, S.R.S. Carotenoids functionality, sources, and processing by supercritical technology: A review. J. Chem. 2016, 2016, 1–16. [Google Scholar] [CrossRef]
- Bayerl, C. Beta-carotene in dermatology-does it help? Acta. Dermatovenerol. Alp. Pannonica. Adriat. 2008, 17, 160–166. [Google Scholar] [PubMed]
- Astner, S.; Wu, A.; Chen, J.; Philips, N.; Rius-Diaz, F.; Parrado, C.; Mihm, M.C.; Goukassian, D.A.; Pathak, M.A.; González, S. Dietary lutein/zeaxanthin partially reduces photoaging and photocarcinogenesis in chronically UVB-irradiated Skh-1 hairless mice. Skin. Pharmacol. Physiol. 2007, 20, 283–291. [Google Scholar] [CrossRef]
- Wertz, K.; Seifert, N.; Hunziker, P.B.; Riss, G.; Wyss, A.; Lankin, C.; Goralczyk, R. β-carotene inhibits UVA-induced matrix metalloprotease 1 and 10 expression in keratinocytes by a singlet oxygen-dependent mechanism. Free. Radic. Biolol. Med. 2004, 37, 654–670. [Google Scholar] [CrossRef]
- Darvin, M.E.; Sterry, W.; Lademann, J.; Vergou, T. The role of carotenoids in human skin. Molecules 2011, 16, 10491–10506. [Google Scholar] [CrossRef]
- Darvin, M.E.; Fluhr, J.W.; Schanzer, S.; Richter, H.; Patzelt, A.; Meinke, M.C.; Zastrow, L.; Golz, K.; Doucet, O.; Sterry, W. Dermal carotenoid level and kinetics after topical and systemic administration of antioxidants: Enrichment strategies in a controlled in vivo study. J. Dermatol. Sci. 2011, 64, 53–58. [Google Scholar] [CrossRef]
- Tominaga, K.; Hongo, N.; Karato, M.; Yamashita, E. Cosmetic benefits of astaxanthin on human subjects. Acta. Biochim. Pol. 2012, 59, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, E. Cosmetic benefit of dietary supplements including astaxanthin and tocotrienol on human skin. Food Style 2002, 216, 112–117. [Google Scholar]
- Yoon, H.-S.; Cho, H.H.; Cho, S.; Lee, S.-R.; Shin, M.-H.; Chung, J.H. Supplementing with dietary astaxanthin combined with collagen hydrolysate improves facial elasticity and decreases matrix metalloproteinase-1and-12 expression: A comparative study with placebo. J. Med. Food 2014, 17, 810–816. [Google Scholar] [CrossRef] [PubMed]
- Juturu, V.; Bowman, J.P.; Deshpande, J. Overall skin tone and skin-lightening-improving effects with oral supplementation of lutein and zeaxanthin isomers: A double-blind, placebo-controlled clinical trial. Clin. Cosmet. Investig. Dermatol. 2016, 9, 325–332. [Google Scholar] [CrossRef]
- Schwartz, S.; Frank, E.; Gierhart, D.; Simpson, P.; Frumento, R. Zeaxanthin-based dietary supplement and topical serum improve hydration and reduce wrinkle count in female subjects. J. Cosmet. Dermatol. 2016, 15, 13–20. [Google Scholar] [CrossRef]
- Meephansan, J.; Rungjang, A.; Yingmema, W.; Deenonpoe, R.; Ponnikorn, S. Effect of astaxanthin on cutaneous wound healing. Clin. Cosmet. Investig. Dermatol. 2017, 10, 259–265. [Google Scholar] [CrossRef]
- Palombo, P.; Fabrizi, G.; Ruocco, V.; Ruocco, E.; Fluhr, J.; Roberts, R.; Morganti, P. Beneficial long-term effects of combined oral/topical antioxidant treatment with the carotenoids lutein and zeaxanthin on human skin: A double-blind, placebo-controlled study. Skin Pharmacol. Physiol. 2007, 20, 199–210. [Google Scholar] [CrossRef]
- Césarini, J.P.; Michel, L.; Maurette, J.M.; Adhoute, H.; Béjot, M. Immediate effects of UV radiation on the skin: Modification by an antioxidant complex containing carotenoids. Photodermatol. Photoimmunol. Photomed. 2003, 19, 182–189. [Google Scholar] [CrossRef]
- Heinrich, U.; Gärtner, C.; Wiebusch, M.; Eichler, O.; Sies, H.; Tronnier, H.; Stahl, W. Supplementation with beta-carotene or a similar amount of mixed carotenoids protects humans from UV-induced erythema. J. Nutr. 2003, 133, 98–101. [Google Scholar] [CrossRef]
- Aust, O.; Stahl, W.; Sies, H.; Tronnier, H.; Heinrich, U. Supplementation with tomato-based products increases lycopene, phytofluene, and phytoene levels in human serum and protects against UV-light-induced erythema. Int. J. Vitam. Nutr. Res. 2005, 75, 54–60. [Google Scholar] [CrossRef]
- Heinen, M.M.; Hughes, M.C.; Ibiebele, T.I.; Marks, G.C.; Green, A.C.; van der Pols, J.C. Intake of antioxidant nutrients and the risk of skin cancer. Eur. J. Cancer. 2007, 43, 2707–2716. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, E.R.; Baron, J.A.; Stukel, T.A.; Stevens, M.M.; Mandei, J.S.; Spencer, S.K. A clinical trial of β-carotene to prevent basal-cell and squamous-cell cancers of the skin. N. Engl. J. Med. 1990, 323, 789–795. [Google Scholar] [CrossRef] [PubMed]
| Micronutrient | Source | Functions in Skin | Hypovitaminosis and Skin Disorders | References |
|---|---|---|---|---|
| Vitamin A (retinol) | liver, fish liver oils, dairy products, butter, cheese, egg yolk, meat products, certain saltwater fish, plant products (β-carotene) | -stimulates synthesis of epidermal proteins -stimulates proliferation of epidermal cells and regulates exfoliation of keratinocytes -stimulates fibroblasts to synthesize collagen and elastin, protects collagen against MMP-induced degradation -takes part in angiogenesis in the dermis -influences skin thickness -influences melanin degradation and skin color -protects the skin against UV radiation -reduces sebum and limits the development of Propionibacterium acnes | -abnormal keratinization of the epidermis -excessive epidermal exfoliation -excessive skin dryness -reduced sebum secretion -follicular keratosis | [27,34,35,143,144,145] |
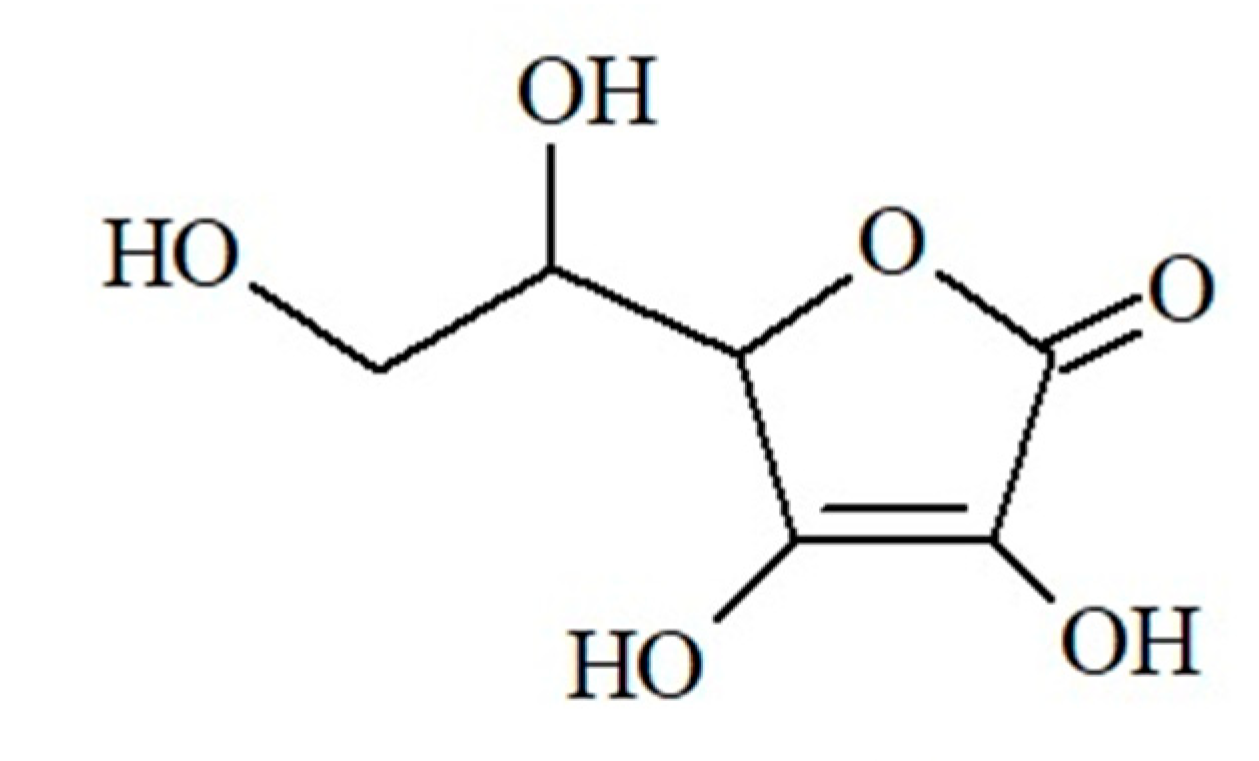
| Vitamin C (ascorbic acid) | citrus fruits, seaberry, dog-rose, blackcurrant, strawberry, raspberry, kiwi, hawthorn, rowan, cruciferous vegetables (Brussels sprouts, kohlrabi, broccoli, cabbage, cauliflower), spinach, chicory, green bean, red pepper, chives, parsley, nettle, five-flavor berry, oregano | -takes part in differentiation of keratinocytes -stimulates ceramide synthesis -influences skin hydration -takes part in collagen biosynthesis and the formation of the extracellular matrix, increases synthesis of GAGs -inhibits melanogenesis -protects against UV radiation, photoaging | -skin fragility -impaired collagen biosynthesis -thickening of the stratum corneum -impaired wound healing -subcutaneous bleeding -scurvy | [48,49,50,58,62] |
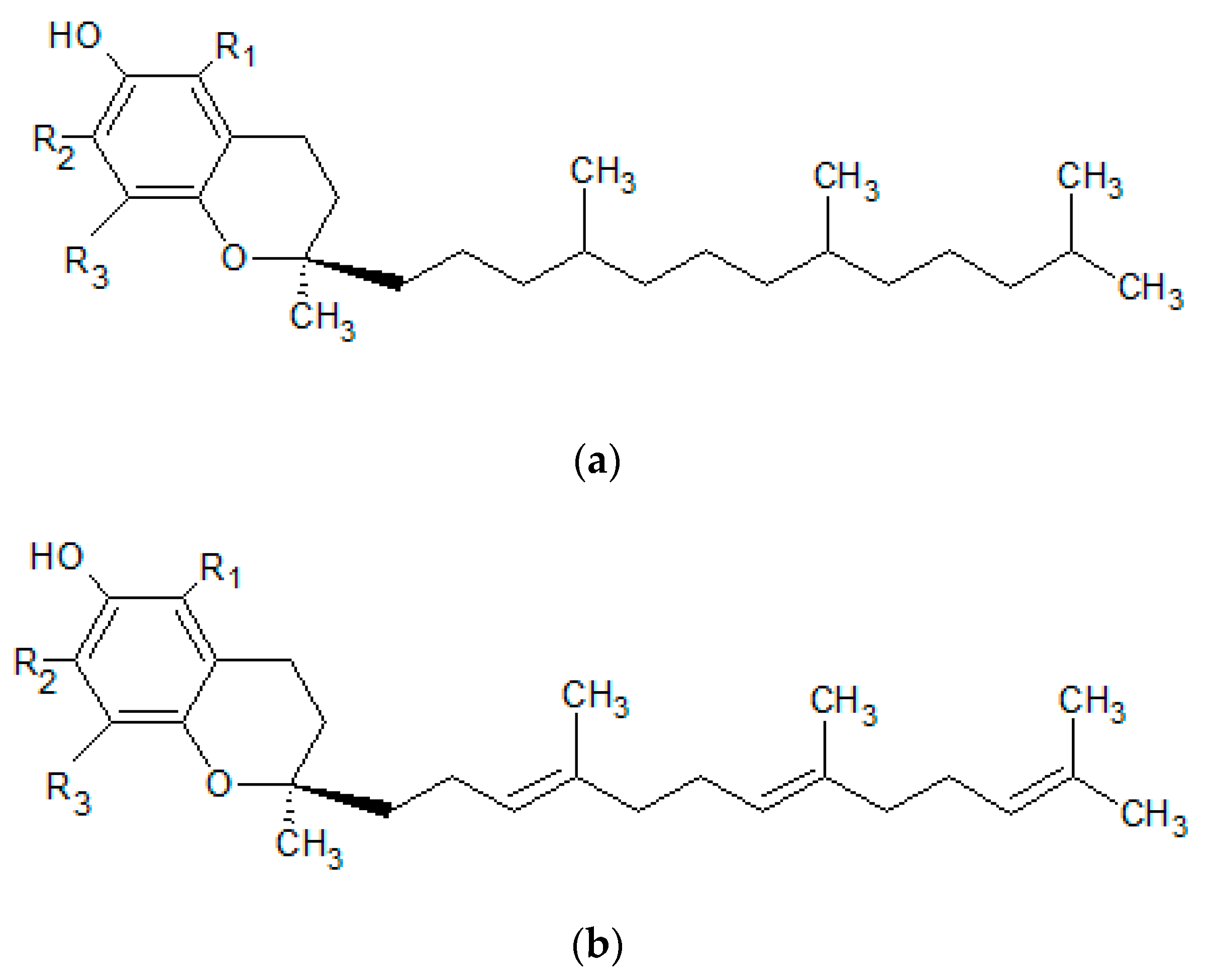
| Vitamin E (tocopherol) | wheat germ oil, sunflower oil, safflower oil, soybean oil, maize oil, cottonseed oil, palm oil, cereal products, nuts | -takes part in biosynthesis of collagen, elastin and GAGs -protects lipid structures of the stratum corneum -prevents the development of UV-induced erythema -has strong antioxidant properties | -exfoliative dermatitis -skin inflammation | [76,78,80,82,84,85,146] |
| Selenium (Se) | Brazil nuts, saltwater fish (yellowfin tuna, halibut, sardines), seafood, sea salt, meats, poultry, eggs, dairy products, cereals, cereal products, broccoli, white cabbage, asparagus, kohlrabi, garlic, onion, legumes, mushrooms | -acts as an antioxidant -is a cofactor for GPx, which removes harmful peroxides -reduces ROS-mediated inflammation and DNA damage -protects the skin from UV-induced oxidative stress -plays roles in fighting ageing and preventing ageing-related diseases | -several skin diseases, including psoriasis, acne vulgaris, and atopic dermatitis | [26,88,89,90,106,147] |
| Zinc (Zn) | red meat, fish, poultry, seafood (shrimp, oysters), nuts, pumpkin seeds, sunflower seeds, legumes, whole grains, dairy products | -prevents UV-induced skin damage -protects against photodamage -plays role in skin morphogenesis, repair and maintenance -modulates activity of 5α-reductase | -acrodermatitis enteropathica -atopic dermatitis, epidermolysis bullosa | [26,97,98,106,107,109,148,149] |
| Copper (Cu) | liver, seafood, nuts, oysters, seeds, some whole grains and legumes, chocolate, cocoa | -exhibits antimicrobial activity -stimulates collagen maturation -modulates melanin synthesis | -Menkes kinky hair disease (hypopigmented skin and hair, sparse, short, brittle scalp hair, pale, mottled, doughy skin) | [118,126,128,150] |
| Silicon (Si) | grains (rice, barley, oat, wheat), grain products (breakfast cereals, bread, pasta), root vegetables (carrots, beetroot, radish, onion, potatoes), beans, maize, fruit (bananas), dried fruit (raisins), nuts, field horsetail | -plays role in synthesis of collagen, elastin and GAGs -improves skin surface and its mechanical properties -improves keratin structure -increases resistance and thickness of nails and hair | -faster appearance of wrinkles -soft and brittle nails -slower wound healing | [135,137,138] |
| Oil Example | Source | Fatty Acids Composition | Reference |
|---|---|---|---|
| Dry, sensitive and atopic skin | |||
| Borage oil | Borago officinalis | γ-linolenic (26–38%), linoleic (35–38%), oleic (16–20%), palmitic (10–11%), stearic (3.5–4.5%), eicosenoic (3.5–5.5%), erucic (1.5–3.5%) acid | [163] |
| Evening primrose oil | Oenothera biennis | linoleic (70–75%), γ-linolenic (8–14%) acid | [161,164] |
| Blackcurrant seed oil | Ribes nigrum | linoleic (>40%), linolenic (10–20%), γ-linolenic (13–15%), stearidonic (2–3%) acid | [162,165] |
| Hemp oil | Canabis sativa | α-linolenic (16–19%), linoleic (55–58%), oleic (11–13%), γ-linolenic (4%), palmitic (5–6%), stearic (2.3–2.5%) acid | [166,167] |
| Oily, combination and acne skin | |||
| Baobab oil | Adansonia digitata | linoleic (36%), palmitic (28.8%), oleic (25.1%) acid | [168,169,170] |
| Neem oil | Azadirachta indica | oleic (40%), stearic (16%), palmitic (14%), linoleic (21%) acid | [171,172] |
| Black cumin oil | Nigella sativa | γ-linolenic (50–60%), oleic (20%), eicosadienoic (3%), dihomo-γ-linolenic (10%), palmitic and stearic (˃30%) acid | [173,174] |
| Coconut oil | Cocos nucifera | capric (7%), lauric (49%), myristic (18%), palmitic (9%), oleic (6%), stearic (2%), linoleic (2%) acid | [175] |
| Mature skin, photoaging, UV protection | |||
| Sea buckthorn oil | Hippophaë rhamnoides | linoleic (34–40%), α-linolenic (23–36%), palmitic (26.3%), oleic (15–20%) acid | [158,176,177] |
| Green coffee oil | Coffea arabica | linoleic (44%), palmitic (34%), oleic (9%), stearic (7%), arachidonic (3%), linolenic (1.5%) acid | [178,179] |
| Wheat germ oil | Triticum aestivum | linoleic (58.6%), α-linolenic (9.6%), oleic (11.2%), palmitic (17.9%), stearic (1.2%), palmitoleic (0.2%) acid | [180] |
| Barbary fig oil | Opuntia ficus-indica | linoleic (62–49%), palmitic (10.6–12.8%), oleic (13–23.5%), stearic (3.3–5.4%) acid | [181] |
| Argan oil | Argania spinosa | oleic (43–49%), linoleic (29–36%), palmitic (11–15%), stearic (4–7%) acid | [182,183,184] |
| Classification | Example | Source | Function |
|---|---|---|---|
| Carotenes—the hydrocarbon carotenoids | β-carotene | beet root, apricots, cantaloupe, carrots, pumpkin, sweet potato, pink grapefruit, tomatoes, watermelon, mango, papaya, peaches, prunes, oranges, bilberry green fruits and vegetables such as green beans, broccoli, brussels sprouts, cabbage, kale, kiwi, lettuce, peas, spinach, acerola, nuts, oil palm, buriti (Mauritia vinifera), sea buckthorn (Hippophaë rhamnoides), camu-camu (Myrciaria dubia), rose hip (Rosa canina), alga Dunaliella salina | -conversion to retinoids (provitamin A carotenoids: α- and β-carotene, β-cryptoxanthin) -modulation of the enzymatic activity of lipoxygenases -enhancement of immune system function -antioxidant activity (ability to quench singlet oxygen, free radical scavenging (including superoxide anions and hydroxyl radicals) -reduction of lipid peroxidation in human skin -promotion of secretion of transcription factors and growth factors -suppression of UVA-mediated induction of MMP-1, MMP-3, and MMP-10 -protection of dermal collagen against UV-induced oxidation of proline -promotion of synthesis of procollagen type I -anti-inflammatory properties -protection of cell membranes and tissues against effects of UV light -protection of skin against UVB-induced photoaging and photocarcinogenesis -reduction of sensitivity to UV radiation-induced erythema -protection from sunburn -skin-lightening effects -improvement of skin condition (hydration, skin texture, radiance, elasticity, reduction in furrows and wrinkles) |
| lycopene | tomato and its derivatives, such as juices, soups, sauces, and ketchup, cherry, guava, watermelon, papaya, peaches, grapefruit, asparagus, rose hip | ||
| Xanthophylls—the oxygenated carotenoids | lutein | green and dark green leafy vegetables, like broccoli, brussels sprouts, spinach, parsley, pumpkin, acerola, rose hip, Tropaeolum majus, caja (Spondias lutea), camu-camu (Myrciaria dubia), microalgae Chlorella vulgaris, eggs | |
| zeaxanthin | broccoli, brussels sprouts, spinach, parsley, maize, pequi (Caryocar villosum), microalgae Chlorella saccharophila | ||
| astaxanthin | aquatic animals, such as lobster, crab, salmon, trout, krill, shrimp, crayfish and crustacea, microalgae Phaffia rhodozyma, Chlorella vulgaris, Haematococcus pluvialis, yeast | ||
| antheraxanthin | many plants, especially maize | ||
| α-, β-cryptoxanthin | many colored plants as maize and papaya, sea buckthorn | ||
| capsanthin | peppers, paprika |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michalak, M.; Pierzak, M.; Kręcisz, B.; Suliga, E. Bioactive Compounds for Skin Health: A Review. Nutrients 2021, 13, 203. https://doi.org/10.3390/nu13010203
Michalak M, Pierzak M, Kręcisz B, Suliga E. Bioactive Compounds for Skin Health: A Review. Nutrients. 2021; 13(1):203. https://doi.org/10.3390/nu13010203
Chicago/Turabian StyleMichalak, Monika, Monika Pierzak, Beata Kręcisz, and Edyta Suliga. 2021. "Bioactive Compounds for Skin Health: A Review" Nutrients 13, no. 1: 203. https://doi.org/10.3390/nu13010203
APA StyleMichalak, M., Pierzak, M., Kręcisz, B., & Suliga, E. (2021). Bioactive Compounds for Skin Health: A Review. Nutrients, 13(1), 203. https://doi.org/10.3390/nu13010203