Effects of Different Doses of Caffeinated Coffee on Muscular Endurance, Cognitive Performance, and Cardiac Autonomic Modulation in Caffeine Naive Female Athletes
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
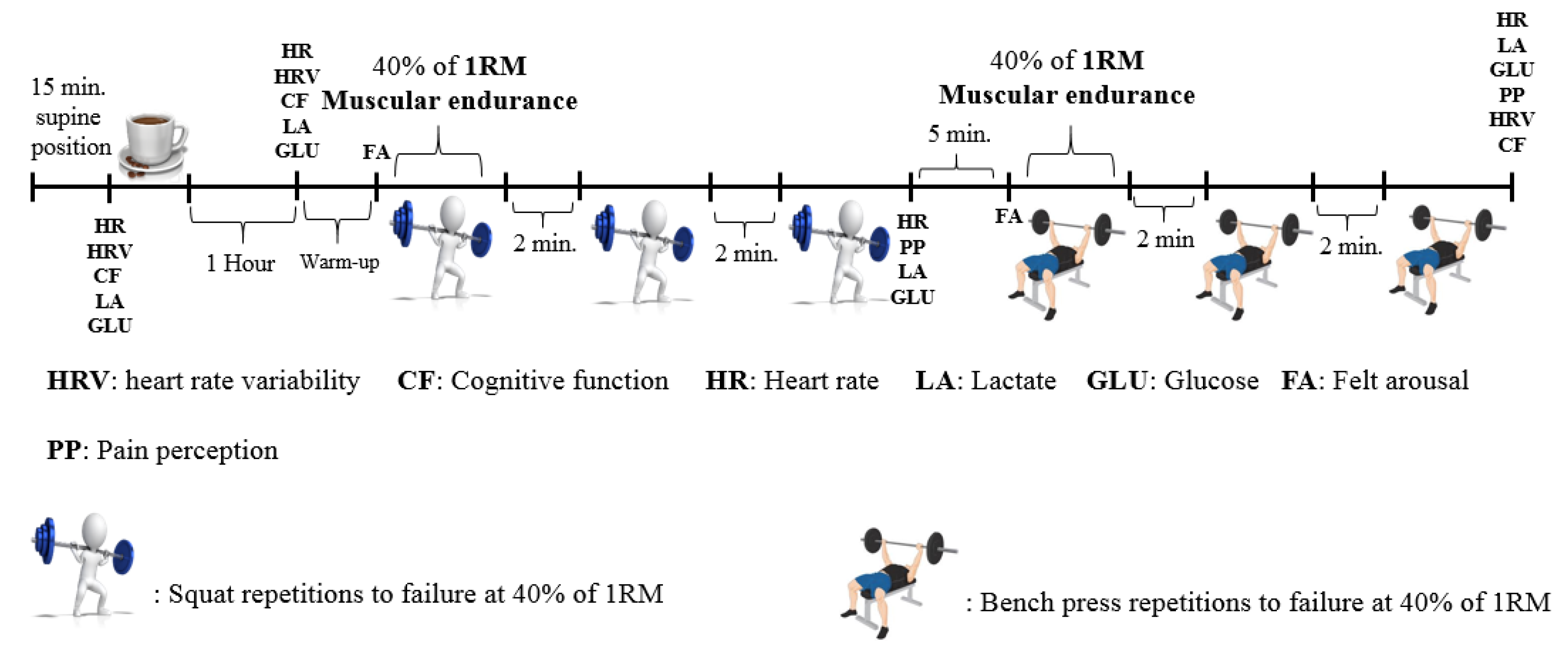
2.2. Study Design
2.3. Strength (1 RM) and Muscular Endurance Test Protocol
2.4. Caffeinated Coffee Protocol
2.5. Heart Rate Variability
2.6. Cognitive Function
2.7. Statistical Analysis
3. Results
3.1. Strength (1 RM) Performance during Familiarization Sessions
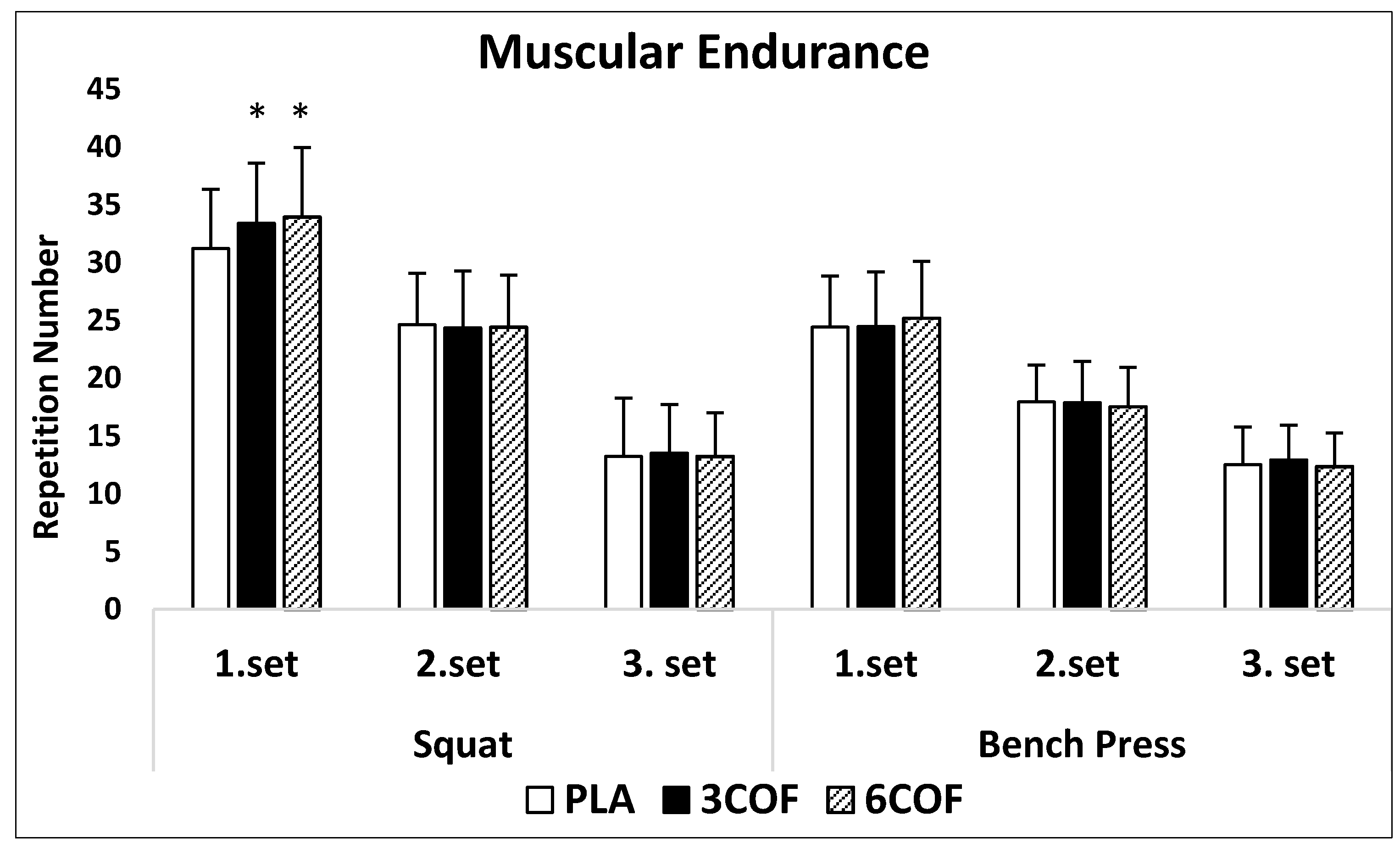
3.2. Muscular Endurance Performance
3.3. HRV
3.4. Cognitive Performance
3.5. Lactate, Glucose, Heart Rate, Pain Perception, Arousal
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pickering, C.; Grgic, J. Is coffee a useful source of caffeine preexercise? Int. J. Sports Nutr. Exerc. Metab. 2020, 30, 69–82. [Google Scholar] [CrossRef]
- Astorino, T.A.; Roberson, D.W. Efficacy of acute caffeine ingestion for short-term high-intensity exercise performance: A systematic review. J. Strength Cond. Res. 2010, 24, 257–265. [Google Scholar] [CrossRef]
- Higgins, S.; Straight, C.R.; Lewis, R.D. The effects of preexercise caffeinated coffee ingestion on endurance performance: An evidence-based review. Int. J. Sports Nutr. Exerc. Metab. 2016, 26, 221–239. [Google Scholar] [CrossRef]
- Duncan, M.J.; Stanley, M.; Parkhouse, N.; Cook, K.; Smith, M. Acute caffeine ingestion enhances strength performance and reduces perceived exertion and muscle pain perception during resistance exercise. Eur. J. Sport Sci. 2013, 13, 392–399. [Google Scholar] [CrossRef]
- Graham, T.E.; Hibbert, E.; Sathasivam, P. Metabolic and exercise endurance effects of coffee and caffeine ingestion. J. Appl. Physiol. 1998, 85, 883–889. [Google Scholar] [CrossRef]
- Hodgson, A.B.; Randell, R.K.; Jeukendrup, A.E. The metabolic and performance effects of caffeine compared to coffee during endurance exercise. PLoS ONE. 2013, 8, e59561. [Google Scholar] [CrossRef]
- Mclellan, T.M.; Bell, D.G. The impact of prior coffee consumption on the subsequent ergogenic effect of anhydrous caffeine. Int. J. Sports Nutr. Exerc. Metab. 2004, 14, 698–708. [Google Scholar] [CrossRef]
- Clarke, N.D.; Kirwan, N.A.; Richardson, D.L. Coffee ingestion improves 5 km cycling performance in men and women by a similar magnitude. Nutrients 2019, 11, 2575. [Google Scholar] [CrossRef]
- Clarke, N.D.; Richardson, D.L.; Thie, J.; Taylor, R. Coffee ingestion enhances 1-mile running race performance. Int. J. Sports Physiol. Perform. 2018, 13, 789–794. [Google Scholar] [CrossRef]
- Richardson, D.L.; Clarke, N.D. Effect of coffee and caffeine ingestion on resistance exercise performance. J. Strength Cond. Res. 2016, 30, 2892–2900. [Google Scholar] [CrossRef]
- Trexler, E.T.; Smith-Ryan, A.E.; Roellofs, E.J.; Hirsch, K.R.; Mock, M.G. Effects of coffee and caffeine anhydrous on strength and sprint performance. Eur. J. Sport Sci. 2016, 16, 702–710. [Google Scholar] [CrossRef]
- Gonçalves, L.S.; Painelli, V.S.; Yamaguchi, G.; Oliveira, L.F.; Saunders, B.; da Silva, R.P.; Maciel, E.; Artilo, G.G.; Roschel, H.; Gualano, B. Dispelling the myth that habitual caffeine consumption influences the performance response to acute caffeine supplementation. J. Appl. Physiol. 2017, 123, 213–220. [Google Scholar] [CrossRef]
- Astorino, T.A.; Martin, B.J.; Schachtsiek, L.; Wong, K.; NG, K. Minimal effect of acute caffeine ingestion on intense resistance training performance. J. Strength Cond. Res. 2011, 25, 1752–1758. [Google Scholar] [CrossRef]
- Duncan, M.J.; Samuel, W.O. The effect of caffeine ingestion on mood state and bench press performance to failure. J. Strength Cond. Res. 2011, 25, 178–185. [Google Scholar] [CrossRef]
- Grgic, J.; Mikulic, P. Caffeine ingestion acutely enhances muscular strength and power but not muscular endurance in resistance-trained men. Eur. J. Sport Sci. 2017, 17, 1029–1036. [Google Scholar] [CrossRef]
- Timmins, T.D.; Saunders, D.H. Effect of caffeine ingestion on maximal voluntary contraction strength in upper- and lower-body muscle groups. J. Strength Cond. Res. 2014, 28, 3239–3244. [Google Scholar] [CrossRef]
- Warren, G.L.; Park, N.D.; Maresca, R.D.; Mckibans, K.I.; Millard-Stafford, M.L. Effect of caffeine ingestion on muscular strength and endurance: A meta-analysis. Med. Sci. Sports Exerc. 2010, 42, 1375–1387. [Google Scholar] [CrossRef]
- Pallares, J.G.; Fernandez-Elias, V.E.; Ortega, J.F.; Munoz, G.; Munoz-Guerra, J.; Mora-Rodriguez, R. Neuromuscular responses to incremental caffeine doses: Performance and side effects. Med. Sci. Sports Exerc. 2013, 45, 2184–2192. [Google Scholar] [CrossRef]
- Wilk, M.; Filip, A.; Krzysztofik, M.; Gepfert, M.; Zajac, A.; Del Coso, J. Acute Caffeine Intake Enhances Mean Power Output and Bar Velocity during the Bench Press Throw in Athletes Habituated to Caffeine. Nutrients 2020, 12, 406. [Google Scholar] [CrossRef]
- Arazi, H.; Hoseinihaji, M.; Eghbali, E. The effects of different doses of caffeine on performance, rating of perceived exertion and pain perception in teenagers female karate athletes. Braz. J. Pharm. Sci. 2016, 52, 685–692. [Google Scholar] [CrossRef]
- Sabblah, S.; Dixon, D.; Bottoms, L. Sex differences on the acute effects of caffeine o maximal strength and muscular endurance. Comp. Exerc. Physiol. 2015, 11, 89–94. [Google Scholar] [CrossRef]
- Wilk, M.; Krzysztofik, M.; Filip, A.; Zajac, A.; Del Coso, J. The effects of high doses of caffeine on maximal strength and muscular endurance in athletes habituated to caffeine. Nutrients 2019, 11, 1912. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Marquez-Jimenez, D.; Refoyo, I.; Del Coso, J.; Leon-Guereno, P.; Calleja-Gonzales, J. Effect of caffeine supplementation on sports performance based on a differences between sexes: A systematic review. Nutrients 2019, 11, 2313. [Google Scholar] [CrossRef]
- Tallis, J.; Yavuz, H.C.M. The effects of low and moderate doses of caffeine supplementation on upper and lower body maximal voluntary concentric and eccentric muscle force. Appl. Physiol. Nutr. Metab. 2017, 43, 274–281. [Google Scholar] [CrossRef]
- Facer-Childs, E.R.; Boiling, S.; Balanos, G.M. The effects of time of day and chronotype on cognitive and physical performance in healthy volunteers. Sports Med. Open. 2018, 4. [Google Scholar] [CrossRef]
- Mora-Rodriguez, R.; Pallares, J.G.; Lopez-Gullon, J.M.; Lopez-Samanes, A.; Fernandez-Elias, V.E.; Ortega, J.F. Improments on neuromuscular performance with caffeine ingestion depend on the time-of-day. J. Sci. Med. Sport. 2015, 18, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Hogervorst, E.; Bandelow, S.; Schmitt, J.; Jentjens, R.; Oliveira, M.; Allgrove, J.; Carter, T.; Gleeson, M. Caffeine improves physical and cognitive performance during exhaustive exercise. Med. Sci. Sports Exerc. 2008, 40, 1841–1851. [Google Scholar] [CrossRef]
- Duncan, M.J.; Dobell, A.P.; Caygill, C.L.; Eyre, E.; Tallis, J. The effect of acute caffeine ingestion on repeated upper body anaerobic exercise and cognitive performance. Eur. J. Sport Sci. 2019, 19, 103–111. [Google Scholar] [CrossRef]
- Ali, A.; O’Donnell, J.; Foskett, A.; Rutherfurd-Markwick, K. The influence of caffeine ingestion on strength and power performance in female team-sport players. J. Int. Soc. Sports Nutr. 2016, 5, 46–54. [Google Scholar] [CrossRef]
- Romero-Moraleda, B.; Del Coso, J.; Gutierrez-Hellin, J.; Lara, B. The effect of caffeine on the velocity of half-squat exercise during the menstrual cycle: A randomized controlled study. Nutrients 2019, 11, 2662. [Google Scholar] [CrossRef]
- Lara, B.; Gutierrez-Hellin, J.; Garcia-Bataller, A.; Rodriguez-Fernandez, P.; Romero-Moraleda, B.; Del Coso, J. Ergogenic effects of caffeine on peak aerobic cycling power during the menstrual cycle. Eur. J. Nutr. 2020, 59, 2525–2534. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Mufti, M.; Kisan, R. Variation of reaction time in different phases of menstrual cycle. J. Clin. Diagn. Res. 2013, 7, 1604–1605. [Google Scholar] [PubMed]
- Gonzaga, L.A.; Vanderlei, L.C.; Gomes, R.L.; Valenti, V.E. Caffeine affects autonomic control of heart rate and blood pressure recovery after aerobic exercise in young adults: A crossover study. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bunsawat, K.; White, W.D.; Kappus, M.R.; Baynard, T. Caffeine delays autonomic recovery following acute exercise. Eur. J. Prev. Cardiol. 2015, 22, 1473–1479. [Google Scholar] [CrossRef]
- Sarshin, A.; Naderi, A.; Gomez da Cruz, C.J.; Feizolahi, F.; Forbes, S.C.; Candow, D.G.; Mohammadgholian, E.; Amiri, M.; Jafari, N.; Rahimi, A.; et al. The effect of varying doses of caffeine on cardiac parasympathetic reactivation following an acute bout of anaerobic exercise in recreational athletes. J. Int. Soc. Sports. Nutr. 2020, 17, 44. [Google Scholar] [CrossRef]
- Bühler, R.; Lachenmeier, D.W.; Schlegel, K.; Winkler, G. Development of a tool to assess the caffeine intake among teenagers and young adults. Ernahrungs Umschau. 2014, 61, 58–63. [Google Scholar]
- Turkish Food Composition Data. Available online: http://www.turkomp.gov.tr (accessed on 7 December 2019).
- Filip, A.; Wilk, M.; Krzysztofik, M.; Del Coso, J. Inconsistency in the ergogenic effect of caffeine in athletes who regularly consume caffeine: Is it due to the disparity in the criteria that defines habitual caffeine intake? Nutrients 2020, 12, 1087. [Google Scholar] [CrossRef]
- Baechle, T.R.; Earle, R.W. Essentials of Strength Training and Conditioning; Human Kinetics: Leeds, UK, 2008. [Google Scholar]
- Svebak, S.; Murgatroyd, S. Metamotivational dominance: A multimethod validation of reversal theory constructs. J. Pers. Soc. Psychol. 1985, 48, 107–116. [Google Scholar] [CrossRef]
- Cook, D.B.; O’Connor, P.J.; Oliver, S.E.; Lee, Y. Sex differences in naturally occurring leg muscle pain and exertion during maximal cycle ergometry. Int. J. Neurosci. 1998, 95, 183–202. [Google Scholar] [CrossRef]
- Beedle, B.; Rytter, S.J.; Healy, R.C.; Ward, T.R. Pretesting static and dynamic stretching does not affect maximum strength. J. Strength Cond. Res. 2008, 22, 1838–1843. [Google Scholar] [CrossRef]
- Tommaso, M. Pain perception during menstrual cycle. Curr. Pain Headache Rep. 2011, 15, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Wilk, M.; Gepfert, M.; Krzysztofik, M.; Golas, A.; Mostowik, A.; Maszczyk, A.; Zajac, A. The influence of grip width on training volume during the bench press with different movement tempos. J. Hum. Kinet. 2019, 21, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Wilk, M.; Tufano, J.J.; Zajac, A. The influence of movement tempo on acute neuromuscular, hormonal, and mechanical responses to resistance exercise- a mini review. J. Strength Cond. Res. 2020, 34, 2369–2383. [Google Scholar] [CrossRef] [PubMed]
- Wilk, M.; Golas, A.; Zmijewski, P.; Krzysztofik, M.; Filip, A.; Del Coso, J.; Tufano, J.J. The effects of the movement tempo on the one-repetition maximum bench press results. J. Hum. Kinet. 2020, 31, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, E.; Jacobs, P.L.; Whitehurst, M.; Penhollow, T.; Antonio, J. Caffeine enhances upper body strength in resistance-trained women. J. Int. Soc. Sports Nutr. 2010, 7, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Aras, D.; Karakoc, B.; Koz, M.; Bizati, O. The effects of active recovery and carbohydrate intake on HRV during 48 hours in athletes after a vigorous-intensity physical activity. Sci. Sports 2017, 32, 295–302. [Google Scholar] [CrossRef]
- Eriksen, B.A.; Eriksen, C.W. Effects of noise letters upon the identification of a target letter in a nonsearch task. Percept. Psychophys. 1974, 16, 143–149. [Google Scholar] [CrossRef]
- Cohen, J.A. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef]
- Grgic, J.; Mikulic, P.; Schoenfeld, B.J.; Bishop, D.J.; Pedisic, Z. The influence of caffeine supplementation on resistance exercise: A review. Sports Med. 2019, 49, 17–30. [Google Scholar] [CrossRef]
- Astorino, T.A.; Rohmann, R.L.; Firth, K. Effect of caffeine ingestion on one-repetition maximum muscular strength. Eur. J. Appl. Physiol. 2008, 102, 127–132. [Google Scholar] [CrossRef]
- Green, J.M.; Wickwire, P.J.; Mclester, J.R.; Gendle, S.; Hudson, G.; Pritchett, R.C.; Laurent, C.M. Effects of caffeine on repetitions to failure and ratings of perceived exertion during resistance training. Int. J. Sports Physiol. Perform. 2007, 2, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Polito, M.D.; Souza, D.B.; Casonatto, J.; Farinatti, P. Acute effect of caffeine consumption on isotonic muscular strength and endurance: A systemic review and meta-analysis. Sci. Sports 2016, 31, 119–128. [Google Scholar] [CrossRef]
- Davis, J.K.; Green, J.M. Caffeine and anaerobic performance: Ergogenic value and mechanisms of action. Sports Med. 2009, 39, 813–832. [Google Scholar] [CrossRef] [PubMed]
- Waller, G.; Dolby, M.; Steele, J.; Fisher, J.P. A low caffeine dose improves maximal strength, but not relative muscular endurance in either heavier-or lighter-loads, or perception of effort or discomfort at task failure in females. PeerJ 2020, 14, e9144. [Google Scholar] [CrossRef]
- Grgic, J.; Sabol, F.; Venier, S.; Mikulic, I.; Bratkovic, N.; Schoenfeld, B.J.; Pickering, C.; Bishop, D.J.; Pedisic, Z.; Mikulic, P. What doses of caffeine to use: Acute effects of 3 doses of caffeine on muscle endurance and strength. Int. J. Sports Physiol. Perform. 2020, 15, 470–477. [Google Scholar] [CrossRef]
- Karayigit, R.; Yildiz, H.; Sahin, M.A.; Sisman, A.; Sari, C.; Buyukcelebi, H.; Ersöz, G. The effects of low-dose caffeinated coffee ingestion on strength and muscular endurance performance in male athletes. Progr. Nutr. 2020. ahead of print. [Google Scholar]
- Rahimi, R. The effect of CYP1A2 genotype on the ergogenic properties of caffeine during resistance exercise: A randomized, double-blind, placebo-controlled, crossover study. Ir. J. Med. Sci. 2019, 188, 337–345. [Google Scholar] [CrossRef]
- Sabol, F.; Grgic, J.; Mikulic, P. The effects of 3 different doses of caffeine on jumping and throwing performance: A randomized, double-blind, crossover study. Int. J. Sports Physiol. Perform. 2019, 22, 1170–1177. [Google Scholar] [CrossRef]
- Grgic, J.; Mikulic, P. Acute effects of caffeine supplementation on resistance exercise, jumping, and Wingate performance: No influence of habitual caffeine intake. Eur. J. Sport Sci. 2020, 2, 1–11. [Google Scholar] [CrossRef]
- Silva Rolim, P.; Costa Matos, R.A.; Melo Keene Von Koenig, E.; Molina, G.E.; Cruz, C.J.G. Caffeine increases parasympathetic reactivation without altering resting and exercise cardiac parasympathetic modulation: A balanced placebo design. Eur. J. Sport Sci. 2019, 19, 490–498. [Google Scholar] [CrossRef]
| Variables | Test | Retest | p | ICC |
|---|---|---|---|---|
| Squat (kg) | 91.2 ± 7.4 (87.3–94.9) | 92.0 ± 7.1 (88.3–95.7) | 0.26 | 0.95 (0.86–0.98) |
| Bench Press (kg) | 63.5 ± 6.7 (60.0–66.9) | 64.5 ± 8.3 (60.2–68.8) | 0.23 | 0.94 (0.85–0.98) |
| Pre Coffee | Post Coffee | Post Test | |||||||
|---|---|---|---|---|---|---|---|---|---|
| M | SD | 95% CI | M | SD | 95% CI | M | SD | 95% CI | |
| SDNN | |||||||||
| PLA | 75.2 | 33.7 | 57.8–92.6 | 81.5 | 29.1 | 66.5–96.5 | 26.1 | 15.3 | 18.2–33.9 |
| 3COF | 78.5 | 41.8 | 57.0–100.0 | 76.1 | 35.7 | 57.7–94.4 | 25.5 | 12.2 | 19.2–31.8 |
| 6COF | 80.9 | 44.2 | 58.2–103.6 | 82.5 | 36.1 | 63.9–101.1 | 23.5 | 19.3 | 13.6–33.5 |
| SDSD | |||||||||
| PLA | 109.3 | 66.8 | 74.9–143.7 | 106.3 | 60.2 | 75.3–137.3 | 19.2 | 14.5 | 11.7–26.6 |
| 3COF | 118.8 | 84.4 | 75.4–162.2 | 107.1 | 66.3 | 73.0–141.3 | 20.1 | 9.9 | 15.0–25.2 |
| 6COF | 122.9 | 86.3 | 78.5–167.3 | 119.1 | 76.1 | 80.0–158.3 | 20.2 | 13.1 | 13.5–27.0 |
| RMSSD | |||||||||
| PLA | 86.4 | 53.1 | 59.0–113.7 | 82.9 | 46.9 | 58.8 -107.0 | 20.0 | 13.8 | 12.9–27.1 |
| 3COF | 93.0 | 66.7 | 58.7–127.3 | 86.9 | 54.6 | 58.8–115.0 | 17.9 | 13.6 | 10.9–24.9 |
| 6COF | 96.0 | 67.6 | 61.2–130.8 | 94.1 | 59.1 | 63.7–124.5 | 19.8 | 13.8 | 12.6–26.9 |
| TP | |||||||||
| PLA | 2227.9 | 1549.2 | 1931.3–3524.4 | 2244.4 | 1268.3 | 1592.3–2896.5 | 243.5 | 138.1 | 172.5–314.5 |
| 3COF | 2922.7 | 1637.2 | 2080.9–3764.5 | 2090.4 | 1278.6 | 1433.0–2747.8 | 251.1 | 105.2 | 196.9–305.2 |
| 6COF | 2961.5 | 1404.4 | 2239.4–3683.6 | 2246.2 | 853.6 | 1807.3–2685.1 | 278.1 | 189.1 | 180.9–375.4 |
| LF | |||||||||
| PLA | 932.7 | 437.0 | 708.0–1157.4 | 688.5 | 379.0 | 493.6–883.4 | 116.9 | 104.0 | 63.4–170.4 |
| 3COF | 977.0 | 499.2 | 720.3–1233.6 | 739.3 | 464.1 | 500.7–977.9 | 124.0 | 77.4 | 84.2–163.8 |
| 6COF | 1082.7 | 669.4 | 738.5–1426.9 | 759.8 | 413.8 | 547.1–972.6 | 126.6 | 112.5 | 68.8–184.4 |
| HF | |||||||||
| PLA | 1637.5 | 1412.6 | 911.2–2363.8 | 1125.1 | 765.4 | 731.6–1518.7 | 89.4 | 50.0 | 63.6–115.1 |
| 3COF | 1684.9 | 1291.4 | 1020.9–2348.9 | 1296.1 | 876.9 | 845.2–1747.0 | 98.9 | 51.8 | 72.3–125.5 |
| 6COF | 1547.7 | 894.3 | 1087.8–2007.5 | 1318.7 | 737.6 | 939.4–1697.9 | 82.8 | 65.9 | 48.9–116.7 |
| LF/HF | |||||||||
| PLA | 0.91 | 0.65 | 0.57–1.25 | 1.31 | 1.15 | 0.72–1.91 | 5.75 | 1.91 | 4.76–6.74 |
| 3COF | 1.23 | 0.65 | 0.89–1.57 | 1.07 | 0.99 | 0.56–1.58 | 5.55 | 1.82 | 4.61–6.49 |
| 6COF | 0.94 | 0.40 | 0.74–1.15 | 0.86 | 0.63 | 0.53–1.18 | 5.46 | 2.44 | 4.21–6.72 |
| HFnu | |||||||||
| PLA | 57.8 | 17.6 | 48.7–66.9 | 57.0 | 21.3 | 46.0–68.0 | 21.8 | 17.7 | 12.7–30.9 |
| 3COF | 53.3 | 20.6 | 42.7–63.9 | 59.5 | 20.2 | 49.0–69.9 | 29.2 | 13.1 | 22.5–36.0 |
| 6COF | 54.9 | 14.7 | 47.4–62.5 | 58.7 | 21.1 | 47.8–62.5 | 25.9 | 17.4 | 16.9–34.8 |
| LFnu | |||||||||
| PLA | 39.9 | 17.4 | 31.0–48.9 | 39.3 | 20.1 | 28.9–49.7 | 74.0 | 16.5 | 65.4–82.5 |
| 3COF | 44.7 | 20.4 | 34.2–55.2 | 40.3 | 22.3 | 28.8–51.8 | 61.6 | 20.9 | 50.8–72.4 |
| 6COF | 44.1 | 13.4 | 37.2–51.0 | 47.9 | 31.9 | 31.5–64.4 | 73.6 | 17.9 | 64.4–82.8 |
| Pre Coffee | Post Coffee | Post Test | |||||||
|---|---|---|---|---|---|---|---|---|---|
| M | SD | 95% CI | M | SD | 95% CI | M | SD | 95% CI | |
| Response Accuracy (%)—Congruent Task | |||||||||
| PLA | 94.1 | 2.7 | 92.7–95.5 | 94.2 | 2.9 | 92.7–95.7 | 93.4 | 2.3 | 92.2–94.6 |
| 3COF | 94.5 | 2.4 | 93.2–95.7 | 94.2 | 2.7 | 92.8–95.6 | 93.7 | 2.6 | 92.3–95.0 |
| 6COF | 94.5 | 2.5 | 93.2–95.8 | 95.2 | 2.4 | 94.0–96.5 | 94.1 | 2.8 | 92.6–95.6 |
| Response Accuracy (%)—Incongruent Task | |||||||||
| PLA | 92.2 | 2.27 | 91.0–93.4 | 92.7 | 2.8 | 91.2–94.1 | 92.2 | 2.7 | 90.8–93.6 |
| 3COF | 92.3 | 2.4 | 91.0–93.6 | 92.0 | 2.1 | 90.9–93.1 | 93.3 | 2.2 | 92.2–94.5 |
| 6COF | 91.1 | 2.0 | 90.1–92.2 | 91.8 | 2.6 | 90.5–93.2 | 93.1 | 2.7 | 91.6–94.5 |
| Reaction Time (ms)—Congruent Task | |||||||||
| PLA | 481.2 | 41.1 | 460.1–502.4 | 466.9 | 38.4 | 447.2–486.7 | 486.8 | 42.5 | 465.0–508.7 |
| 3COF | 482.4 | 43.6 | 459.9–504.9 | 462.9 | 47.6 | 438.3–487.4 | 459.6 | 51.6 | 433.0–486.2 |
| 6COF | 480.2 | 42.6 | 458.3–502.2 | 439.9 | 54.2 | 412.0–467.8 | 436.3 | 44.4 | 413.4–459.1 |
| Reaction Time (ms)—Incongruent Task | |||||||||
| PLA | 528.8 | 44.0 | 506.2–551.4 | 513.5 | 37.8 | 494.1–533.0 | 520.2 | 39.0 | 500.1–540.3 |
| 3COF | 530.7 | 39.0 | 510.6–550.7 | 482.5 | 46.2 | 458.7–506.2 | 482.3 | 57.1 | 452.9–511.7 |
| 6COF | 539.1 | 39.6 | 518.7–559.5 | 469.4 | 45.1 | 446.2–492.6 | 475.7 | 62.6 | 443.4–507.9 |
| PLA | 3COF | 6COF | |||||||
|---|---|---|---|---|---|---|---|---|---|
| M | SD | 95% CI | M | SD | 95% CI | M | SD | 95% CI | |
| Lactate | |||||||||
| PreCof | 1.16 | 0.22 | 1.05–1.28 | 1.19 | 0.19 | 1.09–1.29 | 1.30 | 0.21 | 1.19–1.41 |
| PostCof | 1.22 | 0.24 | 1.09–1.35 | 1.13 | 0.21 | 1.02–1.25 | 1.21 | 0.18 | 1.11–1.31 |
| PostSqu | 7.31 | 1.78 | 6.36–8.27 | 7.85 | 1.70 | 6.94–8.76 | 7.91 | 1.52 | 7.09–8.72 |
| PostBch | 7.89 | 1.65 | 7.01–8.77 | 8.77 | 1.97 | 7.72–9.82 | 8.98 | 2.17 | 7.82–10.14 |
| Glucose | |||||||||
| PreCof | 87.52 | 8.20 | 83.30–91.75 | 90.35 | 7.50 | 86.49–94.21 | 89.17 | 6.01 | 86.08–92.26 |
| PostCof | 90.29 | 9.20 | 85.56–95.02 | 91.23 | 9.09 | 86.56–95.91 | 83.64 | 7.38 | 79.85–87.44 |
| PostSqu | 88.58 | 7.90 | 84.52–92.65 | 89.47 | 9.80 | 84.42–94.51 | 87.58 | 6.71 | 84.13–91.04 |
| PostBch | 90.29 | 7.53 | 86.41–94.17 | 92.47 | 9.50 | 87.58–97.35 | 90.35 | 10.82 | 84.78–95.92 |
| Heart Rate | |||||||||
| PreCof | 65.94 | 6.84 | 62.42–69.45 | 65.52 | 4.96 | 62.97–68.08 | 66.23 | 4.69 | 63.82–68.65 |
| PostCof | 64.29 | 5.87 | 61.27–67.31 | 66.94 | 6.49 | 63.60–70.28 | 64.76 | 5.81 | 61.77–67.75 |
| PostSqu | 163.47 | 10.35 | 158.14–168.79 | 164.11 | 10.43 | 158.75- 169.48 | 163.58 | 12.19 | 157.32–169.85 |
| PostBch | 164.64 | 11.52 | 158.72–170.57 | 165.88 | 11.70 | 159.86–171.90 | 164.47 | 10.85 | 158.88–170.05 |
| Pain Perception | |||||||||
| PostSqu | 8.23 | 1.09 | 7.64–8.79 | 8.05 | 0.89 | 7.59–8.52 | 7.29 | 1.26 | 6.64–7.94 |
| PostBch | 6.76 | 2.04 | 5.71–7.81 | 6.58 | 2.12 | 5.49–7.68 | 6.94 | 1.47 | 6.18–7.70 |
| Felt Arousal | |||||||||
| PreSqu | 1.64 | 0.70 | 1.28–2.00 | 1.58 | 0.87 | 1.27–1.90 | 1.47 | 0.62 | 1.15–1.79 |
| PreBch | 2.52 | 1.06 | 1.98–3.07 | 3.47 | 0.62 | 3.02–3.92 | 3.64 | 1.11 | 3.07–4.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karayigit, R.; Naderi, A.; Akca, F.; Cruz, C.J.G.d.; Sarshin, A.; Yasli, B.C.; Ersoz, G.; Kaviani, M. Effects of Different Doses of Caffeinated Coffee on Muscular Endurance, Cognitive Performance, and Cardiac Autonomic Modulation in Caffeine Naive Female Athletes. Nutrients 2021, 13, 2. https://doi.org/10.3390/nu13010002
Karayigit R, Naderi A, Akca F, Cruz CJGd, Sarshin A, Yasli BC, Ersoz G, Kaviani M. Effects of Different Doses of Caffeinated Coffee on Muscular Endurance, Cognitive Performance, and Cardiac Autonomic Modulation in Caffeine Naive Female Athletes. Nutrients. 2021; 13(1):2. https://doi.org/10.3390/nu13010002
Chicago/Turabian StyleKarayigit, Raci, Alireza Naderi, Firat Akca, Carlos Janssen Gomes da Cruz, Amir Sarshin, Burak Caglar Yasli, Gulfem Ersoz, and Mojtaba Kaviani. 2021. "Effects of Different Doses of Caffeinated Coffee on Muscular Endurance, Cognitive Performance, and Cardiac Autonomic Modulation in Caffeine Naive Female Athletes" Nutrients 13, no. 1: 2. https://doi.org/10.3390/nu13010002
APA StyleKarayigit, R., Naderi, A., Akca, F., Cruz, C. J. G. d., Sarshin, A., Yasli, B. C., Ersoz, G., & Kaviani, M. (2021). Effects of Different Doses of Caffeinated Coffee on Muscular Endurance, Cognitive Performance, and Cardiac Autonomic Modulation in Caffeine Naive Female Athletes. Nutrients, 13(1), 2. https://doi.org/10.3390/nu13010002







