Vitamin D and Immunological Patterns of Allergic Diseases in Children
Abstract
1. Introduction
2. Materials and Methods
2.1. Analyzed Patients
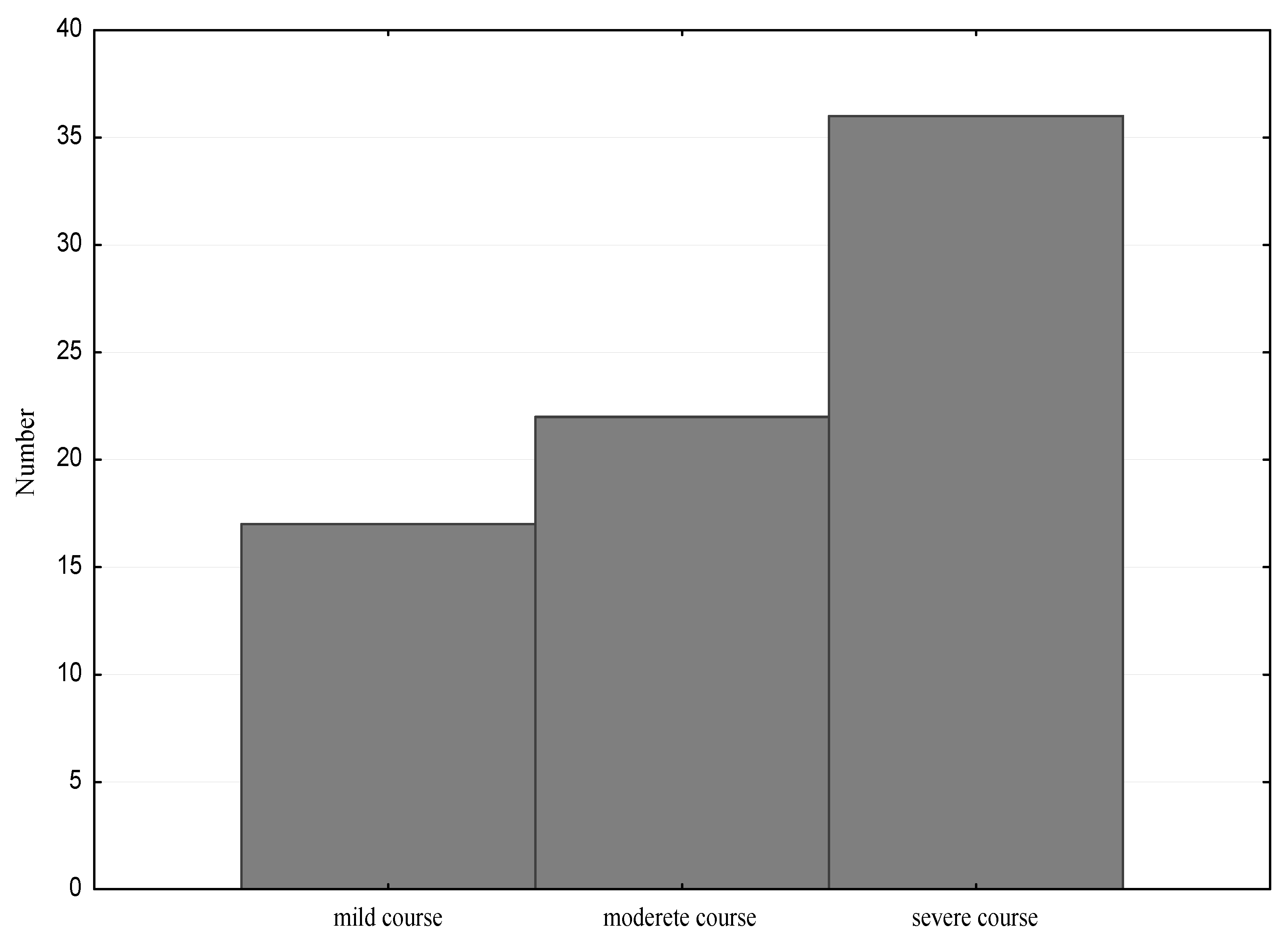
2.2. Course of the Disease
- Mild asthma—asthma controlled with Step 1 or Step 2 treatment;
- Moderate asthma—asthma controlled with Step 3 treatment;
- Severe asthma—asthma controlled with Step 4 or Step 5 treatment [17].
- Mild atopic dermatitis—SCORAD < 20;
- Moderate atopic dermatitis—SCORAD 20–40;
- Severe atopic dermatitis—SCORAD > 40.
2.3. Vitamin D Blood Concentration
2.4. Phenotype of Peripheral Blood Lymphocytes
2.4.1. Phenotype of Peripheral Blood Lymphocytes
2.4.2. Natural T-Regulatory Lymphocytes
2.4.3. Cytokines
2.5. Statistical Analysis
3. Results
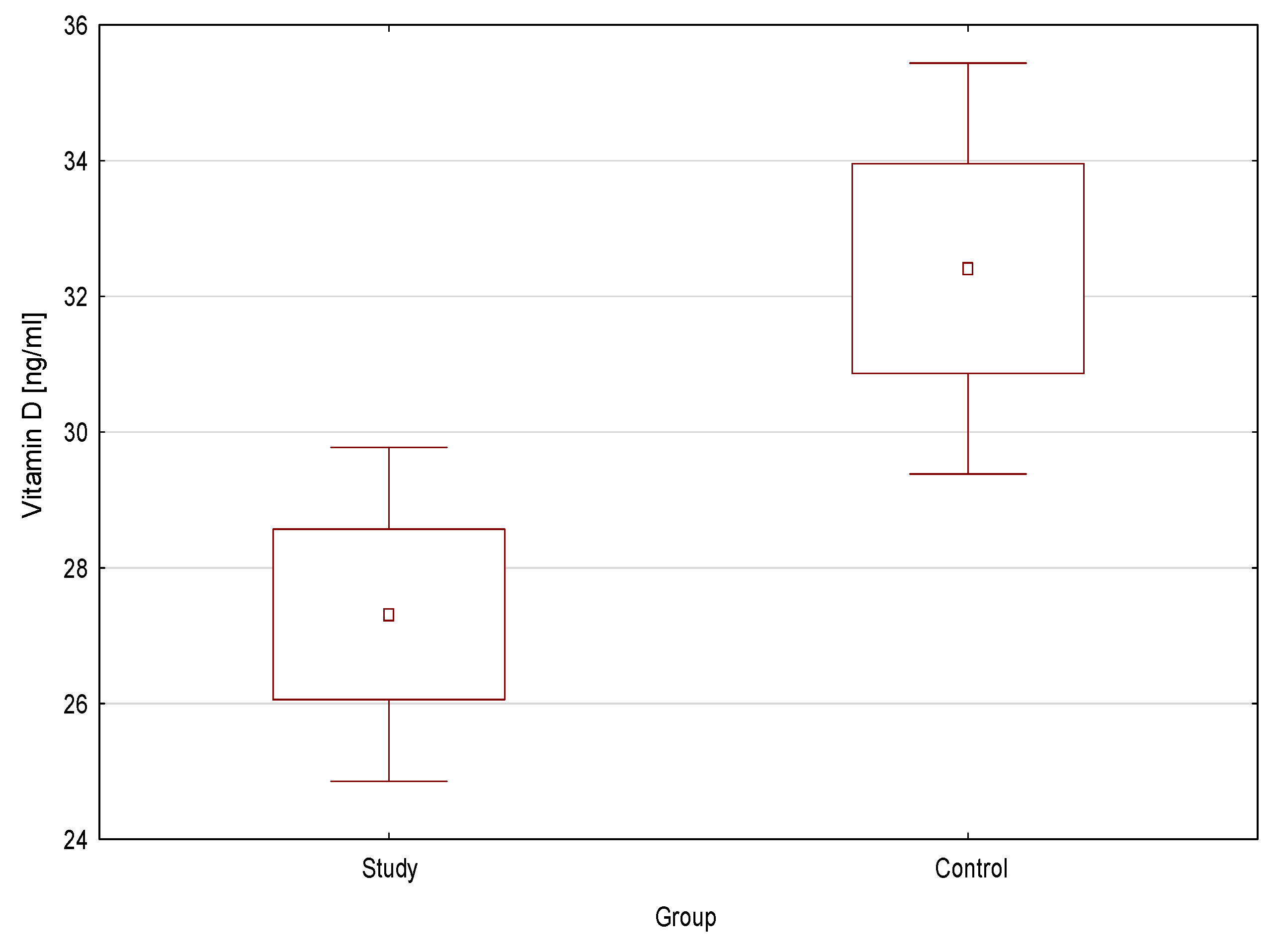
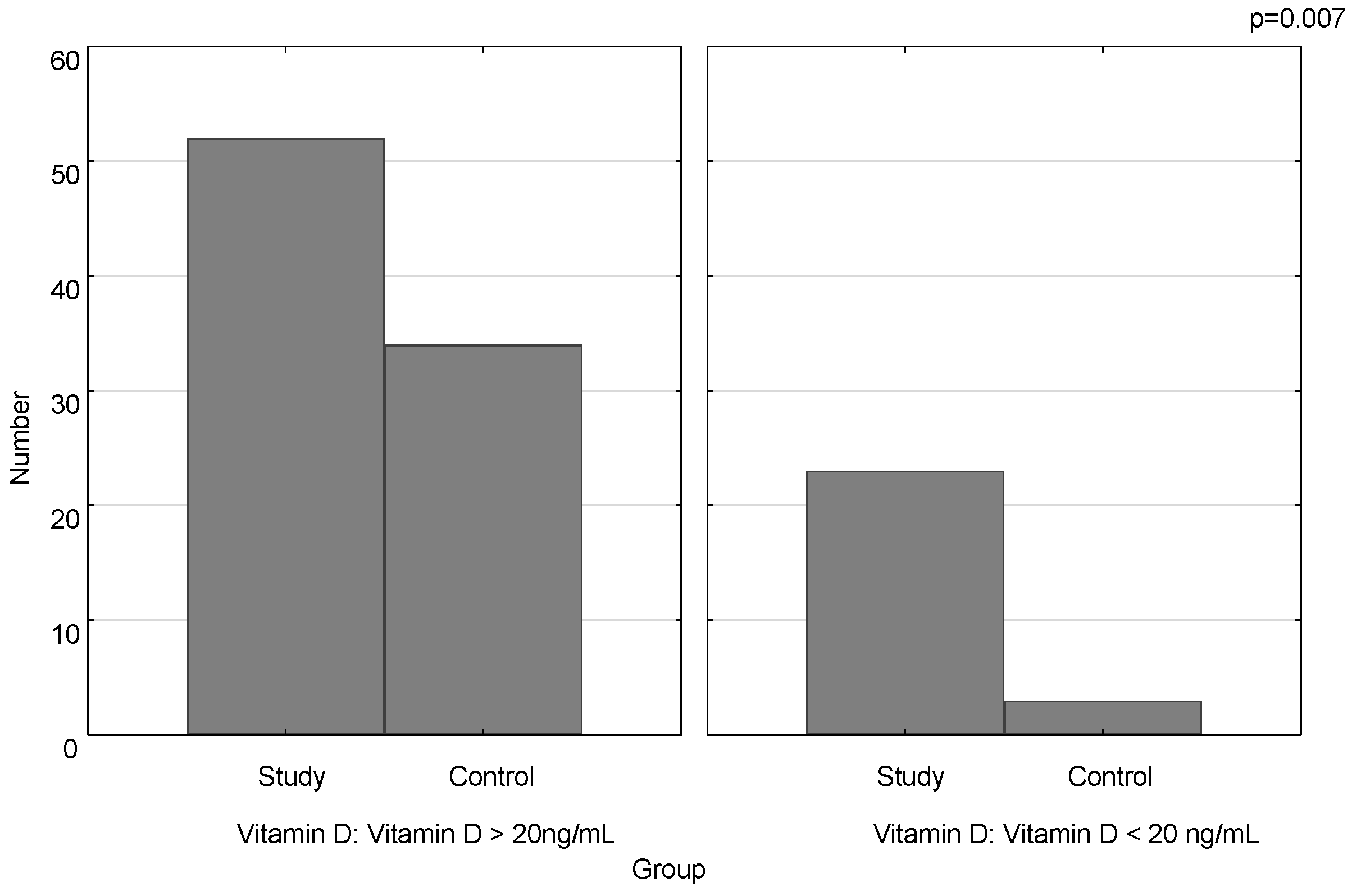
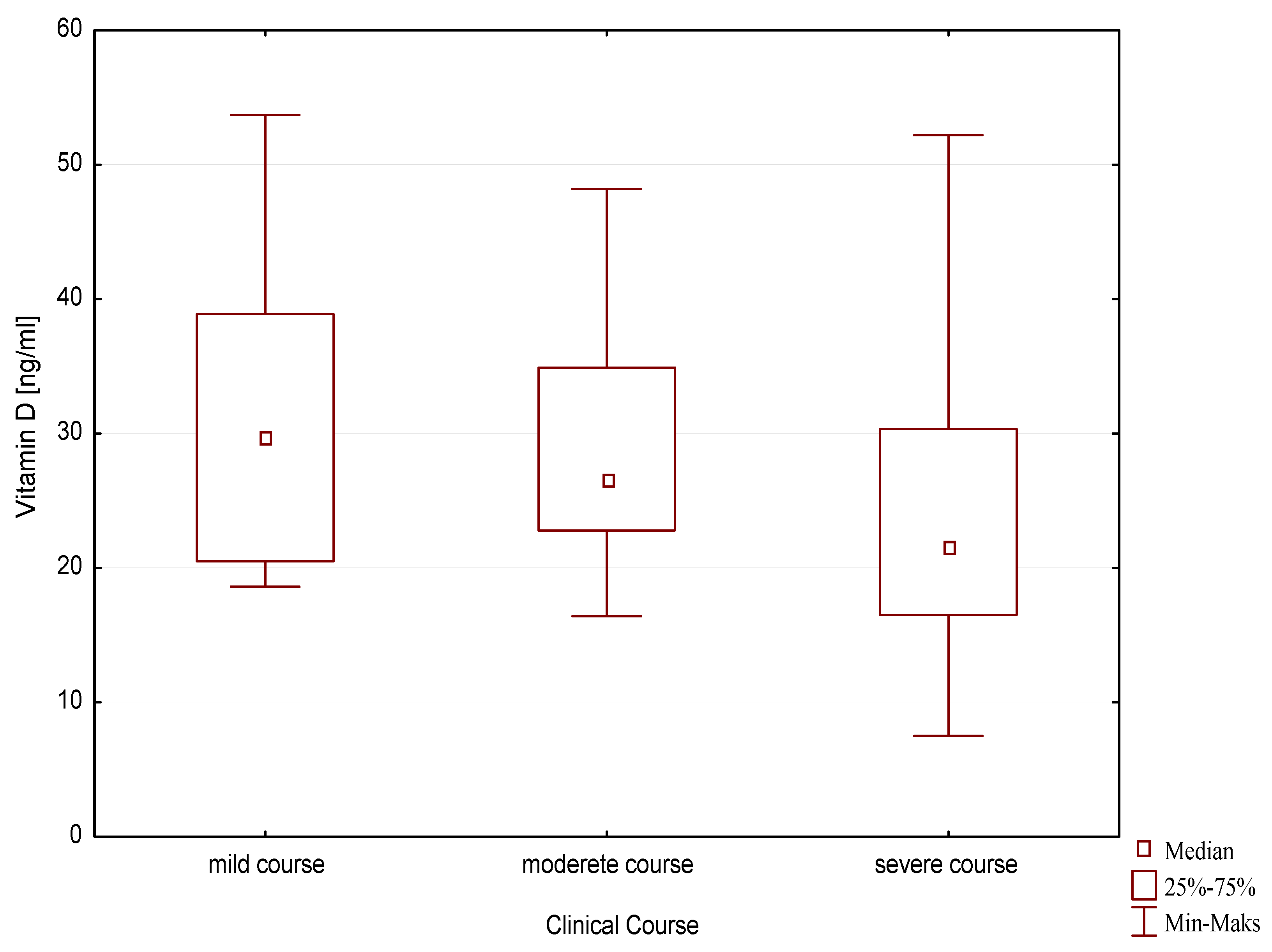
3.1. Vitamin D and the Course of the Disease
3.2. Vitamin D and the Immune Profile
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Marino, R.; Misra, M. Extra-Skeletal Effects of Vitamin D. Nutrients 2019, 11, 1460. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef] [PubMed]
- Wacker, M.; Holick, M.F. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinology 2013, 5, 51–108. [Google Scholar] [CrossRef] [PubMed]
- Chaiprasongsuk, A.; Janjetovic, Z.; Kim, T.-K.; Schwartz, C.J.; Tuckey, R.C.; Tang, E.K.Y.; Raman, C.; Panich, U.; Slominski, A.T. Hydroxylumisterols, Photoproducts of Pre-Vitamin D3, Protect Human Keratinocytes against UVB-Induced Damage. Int. J. Mol. Sci. 2020, 21, 9374. [Google Scholar] [CrossRef]
- Slominski, R.; Tuckey, R.C.; Manna, P.R.; Jetten, A.M.; Postlethwaite, A.; Raman, C.; Slominski, A.T. Extra-adrenal glucocorticoid biosynthesis: Implications for autoimmune and inflammatory disorders. Genes Immun. 2020, 21, 150–168. [Google Scholar] [CrossRef]
- Slominski, A.T.; Kim, T.-K.; Shehabi, H.Z.; Semak, I.; Tang, E.K.Y.; Nguyen, M.N.; Benson, H.E.A.; Korik, E.; Janjetovic, Z.; Chen, J.; et al. In vivo evidence for a novel pathway of vitamin D3 metabolism initiated by P450scc and modified by CYP27B1. FASEB J. 2012, 26, 3901–3915. [Google Scholar] [CrossRef]
- Slominski, A.T.; Chaiprasongsuk, A.; Janjetovic, Z.; Kim, T.-K.; Stefan, J.; Slominski, R.M.; Hanumanthu, V.S.; Raman, C.; Qayyum, S.; Song, Y.; et al. Photoprotective Properties of Vitamin D and Lumisterol Hydroxyderivatives. Cell Biochem. Biophys. 2020, 78, 165–180. [Google Scholar] [CrossRef]
- Slominski, A.T.; Kim, T.-K.; Takeda, Y.; Janjetovic, Z.; Brożyna, A.A.; Skobowiat, C.; Wang, J.; Postlethwaite, A.; Li, W.; Tuckey, R.C.; et al. ROR α and ROR γ are expressed in human skin and serve as receptors for endogenously produced noncalcemic 20-hydroxy- and 20,23-dihydroxyvitamin D. FASEB J. 2014, 28, 2775–2789. [Google Scholar] [CrossRef]
- Slominski, A.T.; Kim, T.-K.; Janjetovic, Z.; Brożyna, A.A.; Żmijewski, M.A.; Xu, H.; Sutter, T.R.; Tuckey, R.C.; Jetten, A.M.; Crossman, D.K. Differential and Overlapping Effects of 20,23(OH)2D3 and 1,25(OH)2D3 on Gene Expression in Human Epidermal Keratinocytes: Identification of AhR as an Alternative Receptor for 20,23(OH)2D3. Int. J. Mol. Sci. 2018, 19, 3072. [Google Scholar] [CrossRef]
- Kidney Disease. Improving Global Outcomes (KDIGO) CKD-MBD Work Group: KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2009, 113, 1–130.
- Buczkowski, K.; Chlabicz, S.; Dytfeld, J.; Horst-Sikorska, W.; Jaroszyński, A.; Kardas, P.; Marcinkowska, M.; Siebert, J.; Tałałaj, M. Wytyczne dla lekarzy rodzinnych dotyczące suplementacji witaminy D. Forum. Med. Rodz. 2013, 7, 55–58. [Google Scholar]
- Nowicki, R.J.; Trzeciak, M.; Kaczmarski, M.; Wilkowska, A.; Czarnecka-Operacz, M.; Kowalewski, C.; Rudnicka, L.; Kulus, M.; Mastalerz-Migas, A.; Peregud-Pogorzelski, J.; et al. Atopic dermatitis. Interdisciplinary diagnostic and therapeutic recommendations of the Polish Dermatological Society, Polish Society of Allergology, Polish Pediatric Society and Polish Society of Family Medicine. Part, I. Prophylaxis, topical treatment and phototherapy. Postepy Dermatol. Alergol 2020, 37, 1–10. [Google Scholar] [PubMed]
- Smoliński, B.; Raciborski, F.; Lipiec, A.; Tomaszewska, A.; Krzych-Fałta, E.; Samel-Kowalik, P.; Walkiewicz, A.; Lusawa, A.; Borowicz, J.; Komorowski, J.; et al. Epidemiologia Chorób Alergicznych w Polsce (ECAP). Pol. J. Allergol. 2014, 1, 10–18. [Google Scholar] [CrossRef]
- Murdaca, G.; Tonacci, A.; Negrini, S.; Greco, M.; Borro, M.; Puppo, F.; Gangemi, S. Emerging role of vitamin D in autoimmune diseases: An update on evidence and therapeutic implications. Autoimmun. Rev. 2019, 18, 102350. [Google Scholar] [CrossRef] [PubMed]
- Muehleisen, B.; Gallo, R.L. Vitamin D in allergic disease: Shedding light on a complex problem. J. Allergy Clin. Immunol. 2012, 131, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, J.M.; Rajka, G. Diagnostic features of atopic dermatitis. Acta Derm Venereol. 1980, 92, 44–47. [Google Scholar]
- From the Global Strategy for Asthma Management and Prevention, Global Initiative for Asthma (GINA) 2014. Available online: http://www.ginasthma.org/ (accessed on 15 November 2020).
- van Schoor, N.M.; Lips, P. Worldwide vitamin D status. Best Pr. Res. Clin. Endocrinol. Metab. 2011, 25, 671–680. [Google Scholar] [CrossRef]
- Datta, S.; Mondal, M.K.; Pal, M. Blood vitamin D concentration in atopic dermatitis patients. World J. Pharm. Pharm. Sci. 2014, 3, 1371–1379. [Google Scholar]
- Kim, M.J.; Kim, S.N.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Vitamin D Status and Efficacy of Vitamin D Supplementation in Atopic Dermatitis: A Systematic Review and Meta-Analysis. Nutrients 2016, 8, 789. [Google Scholar] [CrossRef]
- Peroni, D.G.; Piacentini, G.L.; Cametti, E.; Chinellato, I.; Boner, A.L. Correlation between serum 25-hydroxyvitamin D levels and severity of atopic dermatitis in children. Br. J. Derm. 2011, 164, 1078–1082. [Google Scholar] [CrossRef]
- Sharma, S.; Kaur, T.; Malhotra, S.K.; Rai, J.; Chaudhari, S. Correlation of Vitamin D3 Levels and SCORAD Index in Atopic Dermatitis: A Case Control Study. J. Clin. Diagn. Res. 2017, 11, WC01–WC03. [Google Scholar] [PubMed]
- Wang, S.S.; Hon, K.L.; Kong, A.P.; Pong, H.N.; Wong, G.W.; Leung, T.F. Vitamin D deficiency is associated with diagnosis and severity of childhood atopic dermatitis. Pediatr. Allergy Immunol. 2014, 25, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Su, O.; Bahalı, A.G.; Demir, A.D.; Ozkaya, D.B.; Uzuner, S.; Dizman, D.; Onsun, N. The relationship between severity of disease and vitamin D levels in children with atopic dermatitis. Adv. Derm. Alergol. 2017, 34, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Ozkars, M.Y.; Keskin, O.; Almacioglu, M.; Kucukosmanoglu, E.; Keskin, M.; Balc, O. The relationship between serum vitamin D level and asthma. North Clin. Istanb. 2019, 6, 334–340. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T. Why do T cells express the vitamin D receptor? Ann. N. Y. Acad. Sci. 2011, 1217, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Maalmi, H.; Berraïes, A.; Tangour, E.; Ammar, J.; Abid, H.; Hamzaoui, K.; Hamzaoui, A. The impact of vitamin D deficiency on immune T cells in asthmatic children: A case-control study. J. Asthma Allergy 2012, 5, 11–19. [Google Scholar]
- Mabrouk, R.R.; Amer, H.A.; Soliman, D.A.; Mohamed, N.A.; El-Ghoneimy, D.H.; Hamdy, A.M.; Atef, S.A. Vitamin D Increases Percentages of Interleukin-10 Secreting Regulatory T Cells in Children With Cow’s Milk Allergy. Egypt J. Immunol. 2019, 26, 15–29. [Google Scholar]
- Yu, S.; Cantorna, M.T. Epigenetic reduction in invariant NKT cells following in utero vitamin D deficiency in mice. J. Immunol. 2011, 186, 1384–1390. [Google Scholar] [CrossRef]
- Singh, A.K.; Wilson, M.T.; Hong, S.; Olivares-Villagómez, D.; Du, C.; Stanic, A.K.; Joyce, S.; Sriram, S.; Koezuka, Y.; Van Kaer, L. Natural killer T cell activation protects mice against experimental autoimmune encephalomyelitis. J. Exp. Med. 2001, 194, 1801–1811. [Google Scholar] [CrossRef]
- Boonpiyathad, T.; Sözener, Z.C.; Satitsuksanoa, P.; Akdis, C.A. Immunologic mechanisms in asthma. Semin. Immunol. 2019, 46, 101333. [Google Scholar] [CrossRef]
- Iwamura, C.; Nakayama, T. Role of CD1d- and MR1-Restricted t cells in asthma. Front. Immunol. 2018, 9, 1942. [Google Scholar] [CrossRef] [PubMed]
- Carpio-Pedroza, J.C.; Vaughan, G.; del Rio-Navarro, B.E.; del Rio-Chivardi, J.M.; Vergara-Castaneda, A.; Jimenez-Zamudio, L.A.; Morales-Flores, A.; Rodriguez-Moreno, G.; Ruiz-Tovar, K.; Fonseca-Coronado, S.; et al. Participation of CD161(+) and invariant natural killer T cells in pediatric asthma exacerbations. Allergy Asthma Proc. 2013, 34, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.; Kozlowski, E.; Simon, H. Natural killer T cells expressing IFN-gamma and IL-4 in lesional skin of atopic eczema. Allergy 2009, 64, 1681–1684. [Google Scholar] [CrossRef] [PubMed]
- Gyimesi, E.; Nagy, G.; Remenyik, É.; Sipka, A.; Zeher, M.; Bíró, T.; Szegedi, A. Altered peripheral invariant natural killer T cells in atopic dermatitis. J. Clin. Immunol. 2011, 31, 864–872. [Google Scholar] [CrossRef]
- Eyerich, S.; Traidl-Hoffmann, C.; Behrendt, H.; Cavani, A.; Schmidt-Weber, C.B.; Ring, J.; Eyerich, K. Novel key cytokines in allergy: IL-17, IL-22. Allergol. Sel. 2017, 1, 71–76. [Google Scholar] [CrossRef]
- Kanda, N.; Hau, C.S.; Tada, Y.; Sato, S.; Watanabe, S. Decreased serum LL-37 and vitamin D3 levels in atopic dermatitis: Relationship between IL-31 and oncostatin M. Allergy 2012, 67, 804–812. [Google Scholar] [CrossRef]
- Anderson, J.; Do, L.A.H.; Toh, Z.Q.; Hoe, E.; Reitsma, A.; Mulholland, K.; Licciardi, P.V. Vitamin D Induces Differential Effects on Inflammatory Responses During Bacterial and/or Viral Stimulation of Human Peripheral Blood Mononuclear Cells. Front. Immunol. 2020, 11, 602. [Google Scholar] [CrossRef]
- Colin, E.M.; Asmawidjaja, P.S.; van Hamburg, J.P.; Mus, A.M.C.; van Driel, M.; Hazes, J.M.W.; van Leeuwen, J.P.T.M.; Lubberts, E. 1,25-Dihydroxyvitamin D3 Modulates Th17 Polarization and Interleukin-22 Expression by Memory T Cells from Patients with Early Rheumatoid Arthritis. Arthritis Rheum 2010, 62, 132–142. [Google Scholar] [CrossRef]
| Vitamin D Concentration Range | Serum 25 (OH) D Concentration | |
|---|---|---|
| nmol/L | ng/mL | |
| Severe deficiency | <25 | <10 |
| Deficiency | 25–50 | 10–20 |
| Insufficiency (suboptimal level) | >50–70 | >20–30 |
| Optimal level | >75–200 | >30–80 |
| Toxic level | >250 | >100 |
| Vitamin D > 20 ng/mL [Me, q25–q75] | Vitamin D < 20 ng/mL [Me, q25–q75] | p | |
|---|---|---|---|
| IgE total [IU/mL] | 329.50 (193.0–636.50) | 277.00 (151.00–812.00) | ns. |
| Eosinophil [×103/uL] | 0.55 (0.29–1.04) | 0.45 (0.26–0.86) | ns. |
| Eosinophil [%] | 7.57 (4.00–9.60) | 5.50 (3.00–8.90) | ns. |
| Vitamin D > 20 ng/mL [Me, q25–q75] | Vitamin D < 20 ng/mL [Me, q25–q75] | p | |
|---|---|---|---|
| Study Group | |||
| CD3 [%] | 65.32 (63.33–69.20) | 64.09 (60.29–70.10) | ns. |
| CD 19 [%] | 15.88 (12.98–17.87) | 18.13 (14.64–23.16) | ns. |
| CD4 [%] | 35.88 (34.28–38.94) | 33.85 (30.61–39.46) | ns. |
| CD8 [%] | 28.30 (26.78–31.84) | 26.31 (22.29–31.58) | ns. |
| CD4/CD8 | 1.28 (1.06–1.45) | 1.33 (0.97–1.63) | ns. |
| CD16/56 [%] | 12.61 (9.64–17.37) | 10.83 (7.30–14.36) | ns. |
| NKT [%] | 3.16 (1.45–5.23) | 1.62 (0.79–3.14) | 0.02 |
| CD3 anti-HLADR [%] | 5.23 (3.03–6.81) | 5.38 (3.83–8.19) | ns. |
| T-regulatory [%] | 0.76 (0.43–1.07) | 0.55 (0.32–0.83) | 0.05 |
| Control Group | |||
| CD3 [%] | 64.77 (60.05–68.03) | 63.81 (23.34–65.17) | ns. |
| CD 19 [%] | 15.64 (12.65–18.82) | 11.63 (5.90–18.84) | ns. |
| CD4 [%] | 34.24 (30.01–37.90) | 28.59 (26.69–39.05) | ns. |
| CD8 [%] | 26.13 (23.29–30.87) | 32.54 (22.16–36.28) | ns. |
| CD4/CD8 | 1.32 (1.05–1.47) | 0.82 (0.79–1.76) | ns. |
| CD16/56 [%] | 11.61 (8.25–15.11) | 19.00 (14.41–27.30) | ns. |
| NKT [%] | 2.25 (1.27–3.27) | 2.32 (2.15–4.59) | ns. |
| CD3 anti-HLADR [%] | 4.87 (2.58–8.61) | 7.53 (4.86–8.09) | ns. |
| T-regulatory [%] | 0.87 (0.76–1.29) | 0.97 (0.65–1.16) | ns. |
| Vitamin D > 20 ng/mL [Me, q25–q75] | Vitamin D < 20 ng/mL [Me, q25–q75] | p | |
|---|---|---|---|
| Study Group | |||
| IL-2 [pg/mL] | 2.05 (0.00–3.19) | 2.25 (1.09–3.13) | ns. |
| IL-4 [pg/mL] | 1.19 (0.00–1.38) | 1.42 (0.00–1.69) | ns. |
| IL-6 [pg/mL] | 2.34 (1.81–3.23) | 2.50 (1.95–3.36) | ns. |
| TNF-α [pg/mL] | 1.53 (1.35–1.88) | 1.54 (1.26–1.79) | ns. |
| IL-10 [pg/mL] | 1.92 (1.66–2.52) | 2.25 (1.83–2.93) | ns. |
| IFN-γ [pg/mL] | 0.00 (0.00–1.19) | 0.00 (0.00–1.35) | ns. |
| IL-17A [pg/mL] | 6.07 (2.02–10.33) | 5.997 (2.55–9.53) | ns. |
| IL-1 [pg/mL] | 0.00 (0.00–0.90) | 0.00 (0.00–0.96) | ns. |
| IL-9 [pg/mL] | 2.87 (1.22–3.91) | 3.01 (1.28–5.49) | ns. |
| IL-22 [pg/mL] | 3.25 (0.00–11.47) | 10.35 (3.88–34.75) | 0.01 |
| TGF-β [pg/mL] | 400.53 (346.27–546.50) | 419.25 (342.88–508.37) | ns. |
| Control Group | |||
| IL-2 [pg/mL] | 1.29 (0.00–1.91) | 2.25 (0.00–2.53) | ns. |
| IL-4 [pg/mL] | 1.41 (1.14–1.69) | 1.28 (0.00–1.51) | ns. |
| IL-6 [pg/mL] | 2.18 (1.63–3.26) | 4.23 (1.63–6.53) | ns. |
| TNF-α [pg/mL] | 1.59 (1.26–2.33) | 1.90 (0.00–2.37) | ns. |
| IL-10 [pg/mL] | 1.94 (1.78–2.3) | 1.52 (1.35–1.94) | ns. |
| IFN-γ [pg/mL] | 1.30 (0.00–1.58) | 0.00 (0.00–1.56) | ns. |
| IL-17A [pg/mL] | 5.38 (2.37–7.47) | 0.00 (0.00–4.91) | ns. |
| IL-1 [pg/mL] | 0.00 (0.00–0.13) | 2.01 (0.00–2.38) | ns. |
| IL-9 [pg/mL] | 1.15 (0.01–4.80) | 3.23 (0.00–4.03) | ns. |
| IL-22 [pg/mL] | 5.39 (0.33–20.58) | 0.88 (0.84–6.09) | ns. |
| TGF-β [pg/mL] | 452.14 (358.81–535.93) | 397.66 (397.04–450.20) | ns. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lipińska-Opałka, A.; Tomaszewska, A.; Kubiak, J.Z.; Kalicki, B. Vitamin D and Immunological Patterns of Allergic Diseases in Children. Nutrients 2021, 13, 177. https://doi.org/10.3390/nu13010177
Lipińska-Opałka A, Tomaszewska A, Kubiak JZ, Kalicki B. Vitamin D and Immunological Patterns of Allergic Diseases in Children. Nutrients. 2021; 13(1):177. https://doi.org/10.3390/nu13010177
Chicago/Turabian StyleLipińska-Opałka, Agnieszka, Agata Tomaszewska, Jacek Z. Kubiak, and Bolesław Kalicki. 2021. "Vitamin D and Immunological Patterns of Allergic Diseases in Children" Nutrients 13, no. 1: 177. https://doi.org/10.3390/nu13010177
APA StyleLipińska-Opałka, A., Tomaszewska, A., Kubiak, J. Z., & Kalicki, B. (2021). Vitamin D and Immunological Patterns of Allergic Diseases in Children. Nutrients, 13(1), 177. https://doi.org/10.3390/nu13010177






