The Role of Diet in the Pathogenesis and Management of Inflammatory Bowel Disease: A Review
Abstract
1. Background
2. Treatment of IBD
3. Diet–Microbial–Immune System Interactions
3.1. Breastfeeding
3.2. Dietary Fibre
3.3. FODMAPs
3.4. Sugar
3.5. Gluten
3.6. Red Meat
3.7. Zinc
3.8. Vitamin D and Calcium
3.9. Fat
3.10. Emulsifiers and Nanoparticles
3.11. Mediterranean Style Diet
4. Diet as Prescribed Therapy for Active Disease in IBD
4.1. Exclusive Enteral Nutrition (EEN)
4.2. Partial Enteral Nutrition (PEN)
4.3. Real Food Diet
4.4. Specific Carbohydrate Diet
4.5. Nanoparticles
5. Future Directions of Diet and IBD—Personalised Nutrition
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Torres, J.; Mehandru, S.; Colomebel, J.; Peyrin-Biroulet, L. Crohn’s Disease. Lancet 2017, 389, 1741–1755. [Google Scholar] [CrossRef]
- Ungaro, R.; Mehandru, S.; Allen, P.; Peyrin-Biroulet, L. Ulcerative Colitis. Lancet 2017, 389, 1756–1770. [Google Scholar] [CrossRef]
- Ruemmele, G.; Veres, G.; Kolho, K.; Griffiths, A.; Levine, A.; Escher, J.; Amil Dias, J.; Barabino, A.; Braegger, C.; Bronsky, J.; et al. Consensus guidelines of ECCO/ESPGHAN on the medical management of pediatric Crohn’s disease. J. Crohn’s Colitis 2014, 8, 1179–1207. [Google Scholar] [CrossRef] [PubMed]
- Forbes, A.; Escher, J.; Hebuterne, X.; Kiek, S.; Krznaric, Z.; Schneider, S.; Shamir, R.; Stardelova, K.; Wierdsma, N.; Wiskin, A.; et al. ESPEN guideline: Clinical nutrition in inflamamtory bowel disease. Clin. Nutr. 2017, 36, 321–347. [Google Scholar] [CrossRef]
- Wright, E.; Ding, N.; Niewiadomski, O. Management of inflammatory bowel disease Medical. J. Aust. 2018, 209, 318–323. [Google Scholar] [CrossRef]
- Sartor, R.B.; Wu, G.D. Roles for Intestinal Bacteria, Viruses, and Fungi in Pathogenesis of Inflammatory Bowel Diseases and Therapeutic Approaches. Gastroenterology 2017, 152, 327–339.e4. [Google Scholar] [CrossRef]
- Magro, F.; Gionchetti, P.; Eliakim, R.; Ardizzone, S.; Armuzzi, A.; Barreiro-de Acosta, M.; Burisch, J.; Gecse, K.; Hart, A.; Hindryckx, P.; et al. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. J. Crohn’s Colitis 2017, 11, 649–670. [Google Scholar] [CrossRef]
- Torres, J.; Bonovas, S.; Doherty, G.; Kucharzik, T.; Gisbert, J.; Raine, A.; Adamina, M.; Armuzzi, A.; Bachmann, O.; Bager, P.; et al. ECCO Guidelines on Therapeutics in Crohn’s Disease: Medical Treatment. J. Crohn’s Colitis 2020, 14, 4–22. [Google Scholar] [CrossRef]
- Halme, L.; Paavola-Sakki, P.; Turunen, U.; Lappalainen, M.; Farkkila, M. Family and twin studies in IBD- WJG 2006. World J. Gastroenterol. 2006, 12, 3668–3672. [Google Scholar] [CrossRef]
- Gordon, H.; Trier Moller, F.; Andersen, V.; Harbord, M. Heritability in Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2015, 21, 1428–1434. [Google Scholar] [CrossRef]
- Molodecky, N.A.; Soon, I.S.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W.; et al. Increasing Incidence and Prevalence of the Inflammatory Bowel Diseases With Time, Based on Systematic Review. Gastroenterology 2012, 142, 46–54.e42. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.; Shui, H.; Hamidi, N.; Underwood, F.E.; Tang, W.; Benchimol, E.; Panaccione, R.; Ghosh, S.; Wu, J.C.; Chan, F.K.L.; et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: A systematic review of population based studies. Lancet 2017, 390, 2769–2778. [Google Scholar] [CrossRef]
- Kaplan, G.; Ng, S. Understanding and Preventing the Global Increase of Inflamamtory Bowel Disease. Gastroenterology 2017, 152, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Abegunde, A.; Muhammad, B.; Bhatti, O.; Ali, T. Environmental risk factors for inflammatory diseases: Evidence based literature review. World J. Gastroenterol. 2016, 22, 6296–6317. [Google Scholar] [CrossRef] [PubMed]
- M’Koma, A.E. Inflammatory Bowel Disease: An Expanding Global Health Problem. Clin. Med. Insights Gastroenterol. 2013, 6, CGast.S12731. [Google Scholar] [CrossRef]
- Ungaro, R.; Bernstein, C.; Gearry, R.; Hviid, A.; Kolho, K.; Kronman, M.; Shaw, S.; Van Kruiningen, H.; Colombel, J.F.; Atreja, A. Antibiotics associated with increased risk of Crohn’s disease but not ulcerative colitis: A meta-analysis. Am. J. Gastroenterol. 2014, 109, 1728–1738. [Google Scholar] [CrossRef]
- Mahid, S.; Minor, K.; Soto, R.; Hornung, C.; Galandiuk, S. Smoking and Inflammatory Bowel Disease: A meta-analysis. Mayo Clin. Proc. 2006, 81, 1462–1471. [Google Scholar] [CrossRef]
- Cornish, J.; Tan, E.; Simillis, C.; Clark, S.; Tekkis, P. The risk of oral contraceptives in the etiology of inflammatory bowel disease: A meta-analysis. Gastroenterology 2008, 103, 2394–2400. [Google Scholar] [CrossRef]
- Rubin, D.; Ananthkrishnan, A.; Siegel, C.; Sauer, B.; Long, M. ACG Clinical Guideline: Ulcerative Colitis in Adults. Am. J. Gastroenterol. 2019, 114, 384–413. [Google Scholar] [CrossRef]
- Celiberto, L.S.; Graef, F.A.; Healey, G.R.; Bosman, E.S.; Jacobson, K.; Sly, L.M.; Vallance, B.A. Inflammatory bowel disease and immunonutrition: Novel therapeutic approaches through modulation of diet and the gut microbiome. Immunology 2018, 155, 36–52. [Google Scholar] [CrossRef]
- Limdi, J.K.; Aggarwal, D.; McLaughlin, J.T. Dietary practices and beliefs in patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2016, 22, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.; Walker, J.; Carr, R.; Graff, L.A.; Clara, I.; Promislow, S.; Rogala, L.; Miller, N.; Rawsthorne, P.; Bernstein, C.N. The information needs and preferences of persons with longstanding inflammatory bowel disease. Can. J. Gastroenterol. Hepatol. 2012, 26, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Owczarek, D.; Rodacki, T.; Domagala-Rodacka, R.; Cibor, D.; Mach, T. Diet and nutritional factors in inflammatory bowel diseases. World J. Gastroenterol. 2016, 22, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Marion-Letellier, R.; Savoye, G.; Ghosh, S. IBD: In Food We Trust. J. Crohn’s Colitis 2016, 10, 1351–1361. [Google Scholar] [CrossRef] [PubMed]
- Marchesi, J.; Adams, D.; Fava, F.; Hermes, G.D.; Hirschfield, G.M.; Hold, G.; Nabil Quraishi, M.; Kinross, G.; Smidt, H.; Tuohy, K.M.; et al. The gut microbiota and host health: A new clinical frontier. Gut 2016, 65, 330–339. [Google Scholar] [CrossRef] [PubMed]
- Kostic, A.D.; Xavier, R.J.; Gevers, D. The microbiome in inflammatory bowel disease: Current status and the future ahead. Gastroenterology 2014, 146, 1489–1499. [Google Scholar] [CrossRef] [PubMed]
- Franzosa, E.A.; Sirota-Madi, A.; Avila-Pacheco, J.; Fornelos, N.; Haiser, H.J.; Reinker, S.; Vatanen, T.; Hall, A.B.; Mallick, H.; McIver, L.J.; et al. Gut microbiome structure and metabolic activity in inflammatory bowel disease. Nat. Microbiol. 2019, 4, 293–305. [Google Scholar] [CrossRef]
- Strauss, J.; Kaplan, G.G.; Beck, P.L.; Rioux, K.; Panaccione, R.; Devinney, R.; Lynch, T.; Allen-Vercoe, E. Invasive potential of gut mucosa-derived Fusobacterium nucleatum positively correlates with IBD status of the host. Inflamm. Bowel Dis. 2011, 17, 1971–1978. [Google Scholar] [CrossRef]
- Rowan, F.; Docherty, N.G.; Murphy, M.; Murphy, B.; Coffey, J.C.; O’Connell, P.R. Desulfovibrio Bacterial Species Are Increased in Ulcerative Colitis. Dis. Colon Rectum 2010, 53, 1530–1536. [Google Scholar] [CrossRef]
- McIlroy, J.; Ianiro, G.; Mukhopadhya, I.; Hansen, R.; Hold, G.L. Review article: The gut microbiome in inflammatory bowel disease- avenues for microbial management. Aliment. Pharmacol. Ther. 2017, 47, 26–42. [Google Scholar] [CrossRef]
- Santoru, M.L.; Piras, C.; Murgia, A.; Palmas, V.; Camboni, T.; Liggi, S.; Ibba, I.; Lai, M.A.; Orrù, S.; Blois, S.; et al. Cross sectional evaluation of the gut-microbiome metabolome axis in an Italian cohort of IBD patients. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Levine, A.; Sigall Boneh, R.; Wine, E. Evolving role of diet in the pathogenesis and treatment of Iinflammatory bowel diseases. Gut 2018, 67, 1726–1738. [Google Scholar] [CrossRef] [PubMed]
- David, L.; Maurice, C.; Carmody, R.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Sloan Devlin, A.; Varma, Y.; Fischbach, M.A.; et al. Diet Rapidly and reproducibly alters the human gut microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.D.; Abreu, M.T. Diet as a Trigger or Therapy for Iinflammatory bowel diseases. Gastroenterology 2017, 152, 398–414.e6. [Google Scholar] [CrossRef] [PubMed]
- Castro, F.; De Souza, H.S. Dietary Composition and Effects in Inflammatory Bowel Disease. Nutrients 2019, 11, 1398. [Google Scholar] [CrossRef] [PubMed]
- Zmora, N.; Suez, J.; Elinav, E. You are what you eat: Diet, health and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 35–56. [Google Scholar] [CrossRef]
- Johnson, C.C.; Ownby, D.R. The Infant Gut Bacterial Microbiota and RIsk of Pediatric Asthma and Allergic Diseases. Transl. Res. 2017, 179, 60–70. [Google Scholar] [CrossRef]
- Stewart, C.; Ajami, N.; O’Brien, J.L.; Hutchinson, D.S.; Smith, D.P.; Wong, M.C.; Ross, M.C.; Lloyd, R.E.; Doddapaneni, H.; Metcalf, G.A.; et al. Temporal development of the gut microbiome in early childhood from the TEDDY study. Nature 2018, 562, 583–588. [Google Scholar] [CrossRef]
- Xu, L.; Lochhead, P.; Ko, Y.; Claggett, B.; Leong, R.W.; Ananthakrishnan, A.N. Systematic Review with meta-analysis: Breastfeeding and the risk of Crohn’s Disease and Ulcerative Colitis. Aliment. Pharmacol. Ther. 2017, 46, 780–789. [Google Scholar] [CrossRef]
- Shoda, R.; Matsueda, K.; Yamato, S.; Umeda, N. Epidemiologic analysis of Crohn disease in Japan: Increased dietary intake of n-6 polyunsatirated fatty acids and animal protein relates to the increased incidence of Crohn disease in Japan. Am. J. Clin. Nutr. 1996, 63, 741–745. [Google Scholar] [CrossRef]
- Amre, D.; D’Souza, S.; Morgan, K.; Seidman, G.; Lambrette, P.; Girimard, G.; Israel, D.; Mack, D.; Ghadirian, P.; Deslandres, C.; et al. Imbalances in dietary consumption of fatty acids, vegetables and druits are associated with risk for Crohn’s disease in children. Am. J. Gastroenterol. 2007, 102, 2016–2025. [Google Scholar] [CrossRef]
- Jantchou, P.; Morois, S.; Clavel-Chapelon, F.; Boutron-Ruault, M.C.; Carbonnel, F. Animal protein intake and risk of inflammatory bowel disease: The E3N prospective study. Am. J. Gastroenterol. 2010, 105, 2195–2201. [Google Scholar] [CrossRef] [PubMed]
- John, S.; Luben, R.; Shrestha, S.; Welch, A.; Khaw, K.; Hart, A. Dietary n-3 polyunsaturated fatty acids and the aetiology of ulcerative colitis: A UK prospective cohort study. Eur. J. Gastroenterol. Hepatol. 2010, 22, 602–606. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.; Khalili, H.; Higuchi, L.; Bao, Y.; Krozenik, J.; Giovannucci, E.; Richter, J.M.; Fuchs, C.S.; Chan, A.T. Higher Predicted Vitamin D Status Is Associated With Reduced Risk of Crohn’s Disease. Gastroenterology 2012, 142, 482–489. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; Khalili, H.; Konijeti, G.G.; Higuchi, L.M.; De Silva, P.; Korzenik, J.R.; Fuchs, C.S.; Willett, W.C.; Richter, J.M.; Chan, A.T. A prospective study of long-term intake of dietary fiber and risk of Crohn’s disease and ulcerative colitis. Gastroenterology 2013, 145, 970–977. [Google Scholar] [CrossRef]
- Ananthakrishnan, A.; Khalili, H.; Song, M.; Higuchi, L.M.; Richter, J.M.; Nimotsch, K.; Chan, A.T. High School Diet and Risk of Crohn’s Disease and Ulcerative Colitis. Inflamm. Bowel Dis. 2015, 21, 2311–2319. [Google Scholar]
- Ananthakrishnan, A.; Khalili, H.; Song, M.; Higuchi, L.M.; Richter, J.M.; Chan, A.T. Zinc intake and risk of Crohn’s disease and ulcerative colitis: A prospective cohort study. Int. J. Epidemiol. 2015, 44, 1995–2005. [Google Scholar] [CrossRef]
- Racine, A.; Carbonnel, F.; Chan, S.S.M.; Hart, A.R.; Bueno-de-Mesquita, H.B.; Oldenburg, B.; Van Schaik, F.D.M.; Tjønneland, A.; Olsen, A.; Dahm, C.C.; et al. Dietary Patterns and Risk of Inflammatory Bowel Disease in Europe. Inflamm. Bowel Dis. 2016, 22, 345–354. [Google Scholar] [CrossRef]
- Andersen, V.; Chan, S.; Luben, R.; Khaw, K.; Olsen, A.; Tjonneland, A.; Kaaks, R.; Grip, O.; Bergmann, M.M.; Boeing, H.; et al. Fibre intake and the development of inflammatory bowel disease. J. Crohn’s Colitis 2018, 12, 129–136. [Google Scholar] [CrossRef]
- Opstelten, J.L.; Chan, S.S.; Hart, A.R.; Van Schaik, F.D.; Siersema, P.D.; Lentjes, E.G.; Khaw, K.T.; Luben, R.; Key, T.J.; Boeing, H.; et al. Prediagnostic Serum Vitamin D Levels and the Risk of Crohn’s Disease and Ulcerative Colitis in European Populations: A Nested Case-Control Study. Inflamm. Bowel Dis. 2018, 24, 633–640. [Google Scholar] [CrossRef]
- Dong, C.; Mahamat-Saleh, Y.; Racine, A.; Jantchou, P.; Chan, S.; Hart, A.; Carbonnel, F.; Boutron-Ruault, M.C. Protein intakes and risk of inflammatory bowel disease in the European Prospective Investigation into Cancer and Nutrition Cohort (EPIC-IBD). J. Crohn’s Colitis 2020, 14, S015. [Google Scholar] [CrossRef]
- Khalili, H.; Håkansson, N.; Chan, S.S.; Chen, Y.; Lochhead, P.; Ludvigsson, J.F.; Chan, A.T.; Hart, A.R.; Olén, O.; Wolk, A. Adherence to a Mediterranean diet is associated with a lower risk of later-onset Crohn’s disease: Results from two large prospective cohort studies. Gut 2020, 69, 1637–1644. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, P.; Araújo, J.R.; Di Santo, J.P. A Cross-Talk Between Microbiota-Derived Short-Chain Fatty Acids and the Host Mucosal Immune System Regulates Intestinal Homeostasis and Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2018, 24, 558–572. [Google Scholar] [CrossRef] [PubMed]
- Silveira, A.L.M.; Ferreira, A.V.M.; De Oliveira, M.C.; Rachid, M.A.; Da Cunha Sousa, L.F.; Dos Santos Martins, F.; Gomes-Santos, A.C.; Vieira, A.T.; Teixeira, M.M. Preventive rather than therapeutic treatment with high fiber diet attenuates clinical and inflammatory markers of acute and chronic DSS-induced colitis in mice. Eur. J. Nutr. 2017, 56, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.; Gutafsson, G.; Holmen-Larsson, J.; Jabbar, K.; Xia, L.; Xu, H.; Ghishan, F.; Carvalho, F.A.; Gewirtz, A.T.; Sjovall, H.; et al. Bacteria penetrate the normally impenetrable inner colon mucus layer in both murine colitis models and patients with ulcerative colitis. Gut 2014, 63, 281–291. [Google Scholar] [CrossRef]
- Desai, M.; Seekatz, A.; Koropatkin, N.; Kamada, N.; Hickey, C.A.; Wolder, M.; Pudlo, N.A.; Kitamoto, S.; Terrapon, N.; Muller, A.; et al. Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell 2016, 167, 1339–1353. [Google Scholar] [CrossRef]
- Luhrs, H.; Gerke, T.; Muller, J. Butyrate inhibits NF-κB activation in lamina propria macrophages of patients with ulcerative colitis. Scand. J. Gastroenterol. 2002, 37, 458–466. [Google Scholar] [CrossRef]
- Milajerdi, A.; Ebrahimi-Daryani, N.; Dieleman, L.; Larijani, B.; Esmaillzadeh, A. Association of Dietary Fiber, Fruit and Vegetable Consumption with Risk of Inflammatory Bowel DIsease: A Systematic Review and Meta-Analysis. Adv. Nutr. 2020, nmaa145. [Google Scholar] [CrossRef]
- Brotherton, C.; Martin, C.; Long, M.; Kappleman, M.D.; Sandler, R.S. Avoidance of Fiber is Associated With Greater Risk of Crohn’s Disease Flare in a 6-Month Period. Clin. Gastroenterol. Hepatol. 2016, 14, 1130–1136. [Google Scholar] [CrossRef]
- Wedlake, L.; Slack, N.; Andreyev, H.J.N.; Whelan, K. Fiber in the Treatment and Maintenance of Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2014, 20, 576–586. [Google Scholar] [CrossRef]
- Altobelli, E.; Del Negro, V.; Matteo Angeletti, P.; Latella, G. Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis. Nutrients 2017, 9, 940. [Google Scholar] [CrossRef]
- Cox, S.; Prince, A.; Myers, C.; Irving, P.M.; Lindsay, J.O.; Lomer, M.C.; Whelan, K. Fermentable carbohydrates [FODMAPs] exaccerbate functional gastrointestinal symptoms in patients with inflammatory bowel disease: A randomised, double-blind placebo-controlled, cross-over, re-challange trial. J. Crohn’s Colitis 2017, 11, 1420–1429. [Google Scholar] [CrossRef]
- Cox, S.; Lindsay, J.; Fromentin, S.; Stagg, A.J.; McCarthy, N.E.; Galleron, N.; Ibraim, S.B.; Roume, H.; Levenez, F.; Pons, N.; et al. Effects of Low FODMAP Diet on symptoms, fecal mirobiome and markers of inflammation in patients with quiescent inflamamtory bowel disease in a randomised trial. Gastroenterology 2020, 158, 176–188. [Google Scholar] [CrossRef] [PubMed]
- Prince, A.C.; Myers, C.E.; Joyce, T.; Irving, P.; Lomer, M.; Whelan, K. Fermentable carbohydrate restriction (Low FODMAP Diet) in clinical practice improves functional gastrointestinal symptoms in patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2016, 22, 1129–1135. [Google Scholar] [CrossRef]
- Bodini, G.; Zanella, C.; Crespi, M.; Pumo, S.L.; Demarzo, M.G.; Savarino, E.; Savarino, V.; Giannini, E.G. A randomised, 6-wk trial of low FODMAP diet in patients with inflammatory bowel disease. Nutrition 2019, 67–68, 110542. [Google Scholar] [CrossRef] [PubMed]
- Laffin, M.; Fedorak, R.; Zalasky, A.; Park, H.; Gill, A.; Agrawal, A.; Ammar, K.; Hotte, N.; Madsen, K.L. A high-sugar diet rapidly enhances susceptibility to colitis via depletion of luminal short-chain fatty acids in mice. Nat. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Montrose, D.C.; Nishiguchi, R.; Basu, S.; Staab, H.A.; Zhou, X.K.; Wang, H.; Meng, L.; Johncilla, M.; Cubillos-Ruiz, J.R.; Morales, D.K.; et al. Dietary Frictose Alters the Composition, Localization and Metabolism of Gut Microbiota in Association with Worsening of Colitis. Cell. Mol. Gastroenterol. Hepatol. 2020, in press October 2020. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Feng, J.; Gao, Q.; Ma, M.; Lin, X.; Liu, J.; Li, J.; Zhao, Q. Carbohydrate and protein intake and risk of ulcerative colitis: Systemic review and dose-response meta-analysis of epidemiological studies. Clin. Nutr. 2017, 36, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, K.; Neumann, J.; Liu, F.; Frohlich-Nowoisky, J.; Cremer, C.; Saloga, J.; Reinmuth-Selzle, K.; Poschl, U.; Schuppan, D.; Bellinghausen, I.; et al. Nitration of Wheat Amylase Inhibitors Increases Their Innate and Adaptive Immunostimulatory Potential in vitro. Front. Immunol. 2019, 9, 3174. [Google Scholar] [CrossRef] [PubMed]
- Junker, Y.; Zeissing, S.; Kim, S.; Barisani, D.; Wieser, H.; Leffler, D.A.; Zevallos, V.; Libermann, T.A.; Dillon, S.; Freitag, T.L.; et al. Wheat amylase trypsin inhibitors drive intestinal inflammation via activation of toll-like receptor 4. J. Exp. Med. 2012, 201, 2395–2408. [Google Scholar] [CrossRef]
- Aziz, I.; Branchi, F.; Pearson, K.; Priest, J.; Sanders, D.S. A Study Evaluating the Bidirectional Relationship Between Inflammatory Bowel Disease and Self-Reported Non-Coeliac Gluten Senstivity. Inflamm. Bowel Dis. 2015, 21, 847–853. [Google Scholar] [CrossRef] [PubMed]
- Herfarth, H.; Martin, C.; Sandler, R.; Kappelman, M.D.; Long, M.D. Prevalence of a gluten-free diet and improvement of clinical symptoms in patients with Iinflammatory bowel diseases. Inflamm. Bowel Dis. 2014, 2014, 1194–1197. [Google Scholar] [CrossRef] [PubMed]
- Limketkai, B.N.; Sepulveda, R.; Hing, T.; Shah, N.D.; Choe, M.; Limsui, D.; Shah, S. Prevalence and factors associated wheat gluten sensitivity in inflammatory bowel disease. Scand. J. Gastroenterol. 2018, 53, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Han, T.; Liu, J.; Shan Li, J.; Hua Zhang, Z.; Wang, Y.; Yan Li, Q.; Zhu, Q.; Yang, C.M. Meat intake and risk of inflammatory bowel disease: A meta-analysis. Turk. J. Gastroenterol. 2015, 26, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Lewin, M.H.; Bailey, N.; Bandaletova, T.; Bowman, R.; Cross, A.J.; Pollock, J.; Shuker, D.E.G.; Bingham, S. Red Meat Enhances the Colonic Formation of the DNA Adduct O6-Carboxymethyl Guanine: Implications for Colorectal Cancer Risk. Cancer Res. 2006, 66, 1859–1865. [Google Scholar] [CrossRef] [PubMed]
- Le Leu, R.; Young, G. Fermentation of Starh and Protein in the Colon. Cancer Biol. Ther. 2007, 6, 259–260. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Le Leu, R.; Young, G.P.; Hu, Y.; Winter, J.; Conlon, M.A. Dietary Red Meat Aggravates Dextran Sulfate Sodium-Induced Colitis in Mice Wheras Resistant Starch Attenuates Inflammation. Dig. Dis. Sci. 2013, 58, 3475–3482. [Google Scholar] [CrossRef]
- Jowett, S.L.; Seal, C.J.; Pearce, M.S.; Phillips, E.; Gregory, W.; Barton, J.R.; Welfare, M.R. Influence of dietary factors on the clinical course of ulcerative colitis: A prospective cohort study. Gut 2004, 53, 1479–1484. [Google Scholar] [CrossRef]
- Albenberg, L.; Brensinger, C.M.; Wu, Q.; Gilroy, E.; Kappelman, M.D.; Sandler, R.S.; Lewis, J.D. A Diet Low in Red and Processed Meat Does Not Reduce Rate of Crohn’s Disease Flares. Gastroenterology 2019, 157, 128–136. [Google Scholar] [CrossRef]
- Barollo, M.; Medici, V.; D’Inca, R.; Banergee, A.; Ingravallo, G.; Scarpa, M.; Patak, S.; Ruffolo, C.; Cardin, R.; Carlo Sturniolo, G. Antioxidative potential of a combined therapy of anti TNFalpha and Zn acetate in experimental colitis. World J. Gastroenterol. 2011, 17, 4099–4103. [Google Scholar] [CrossRef]
- Iwaya, H.; Kashiwaya, M.; Shinoki, A.; Lee, J.; Hayashi, K.; Hara, H.; Ishizuka, S. Marginal zinc deficiency exacerbates experimental colitis induced by dextran sulfate sodium in rats. J. Nutr. 2011, 141, 1077–1082. [Google Scholar] [CrossRef] [PubMed]
- Lahiri, A.; Abraham, C. Activation of Pattern Recognition Receptors Up-Regulates Metallothioneins, Thereby Increasing Intracellular Accumulation of Zinc, Autophagy, and Bacterial Clearance by Macrophages. Gastroenterology 2014, 147, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Nutrient Reference Values for Australia and New Zeland Council. 2014. Available online: https://www.nrv.gov.au (accessed on 30 December 2020).
- Taylor, L.; Almutairdi, A.; Shommu, N.; Fedorak, R.; Ghosh, S.; Reimer, R.A.; Panaccione, R.; Raman, M. Cross-Sectional Analysis of Overall Dietary Intake and Mediterranean Dietary Pattern in Patients with Crohn’s Disease. Nutrients 2018, 10, 1761. [Google Scholar] [CrossRef] [PubMed]
- Siva, S.; Rubin, D.; Gulotta, G.; Wroblewski, K.; Pekow, J. ZInc Deficiency is Associated with Poor Clinical Outcomes in Patients with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2017, 23, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Harbord, M.; Annese, V.; Vavrika, S.R.; Allez, M.; Barreiro-de Acosta, M.; Muri Boberg, K.; Burisch, J.; De Vos, M.; De Vries, A.; Dick, A.D.; et al. THe First European Evidence-based Consensus on Extra-intestinal Manfestations of Inflammatory Bowel Disease. J. Crohn’s Colitis 2015, 10, 239–254. [Google Scholar] [CrossRef] [PubMed]
- Polzonetti, V.; Pucciarelli, S.; Vincenzetti, S.; Polidori, P. Dietary Intake of VItamin D from Dairy Products Reduces the Risk of Osteoporosis. Nutrients 2020, 16, 1743. [Google Scholar] [CrossRef]
- Zhang, Y.G.; Wu, S.; Sun, J.; Vitamin, D. Vitamin D receptor and tissue barriers. Tissue Barriers 2013, 1, e23118. [Google Scholar] [CrossRef]
- Penna, G.; Adorini, L. 1α 25-dihydroxyvitamin D3 inhibits differentiation, maturation, activation and survival of dendritic cells leading to impaired alloreactive T cell activation. J. Immunol. 2000, 164, 2405–2411. [Google Scholar] [CrossRef]
- Ooi, J.; Rogers, C.; Cantorna, M. Vitamin D Regulates the Gut Microbiome and Protects Mice from Dextran Sodium Sulfate-Induced Colitis. J. Nutr. 2013, 143, 1679–1686. [Google Scholar] [CrossRef]
- Assa, A.; Vong, L.; Pinnell, L.J.; Avitzur, N.; Johnson-Henry, K.C.; Sherman, P.M. Vitamin D Deficiency Promotes Epithelial Barrier Dysfunction and Intestinal Inflammation. J. Infect. Dis. 2014, 210, 1296–1305. [Google Scholar] [CrossRef]
- Lagishetty, V.; Misharin, A.; Lu, N.; Lisse, T.; Chun, R.; Ouyang, Y.; McLachlan, S.M.; Adams, J.S.; Hewison, M. Vitamin D deficiency in mice impairs colonic antibacterial activity and predisposes to colitis. Endocrinology 2010, 151, 2423–2432. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, S.; Hvas, C.; Agnholt, J.; Ambrosius Christensen, L.; Heickendorff, L.; Dahlerup, J.F. Active Crohn’s disease is associated with low vitamin D levels. J. Crohn’s Colitis 2013, 7, e407–e413. [Google Scholar] [CrossRef] [PubMed]
- Ulitsky, A.; Ananthakrishnan, A.N.; Naik, A.; Skaros, S.; Zadvornova, Y.; Binion, D.G.; Issa, M. Vitamin D deficiency in patients with inflammatory bowel disease: Association with disease activity and quality of life. J. Parenter. Enter. Nutr. 2011, 35, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Ananthakrishnan, A.N.; Cagan, A.; Gainer, V.; Cai, T.; Cheng, S.; Gavova, G.; Chen, P.; Szolovits, P.; Xia, Z.; De Jager, P. Normalization of Plasma 25-Hydroxy Vitamin D Is Associated with Reduced Risk of Surgery in Crohn’s Disease. Inflamm. Bowel Dis. 2013, 19, 1921–1927. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, O.H.; Hansen, T.I.; Gubatan, J.M.; Jensen, K.B.; Rejnmark, L. Managing vitamin D deficiency in Inflammatory Bowel Disease. Frontline Gastroenterol. 2019, 10, 394–400. [Google Scholar] [CrossRef]
- Verina, P.; Loizos, P.; Di Giuseppantonio, I.; Amore, B.; Chiappini, A.; Cannizzaro, S. Dietary calcium intake in patients with inflammatory bowel disease. J. Crohn’s Colitis 2014, 8, 312–317. [Google Scholar] [CrossRef]
- Szilagyi, A.; Galiatsatos, P.; Xue, X. Systematic review and meta analysis of lactose digestion, its impact on intolerance and nutritional effects of dairy food restriction in Iinflammatory bowel diseases. Nutr. J. 2016, 15, 67. [Google Scholar] [CrossRef]
- Lomer, M.C.E.; Grainger, S.L.; Ede, R.; Catterall, A.P.; Greenfield, S.M.; Cowan, R.E.; Vicary, F.R.; Jenkins, A.P.; Fidler, H.; Harvey, R.S. Lack of efficacy of a reduced microparticle diet in a multi-centred trial of patients with active Crohn’s Disease. Eur. J. Gastroenterol. Hepatol. 2005, 17, 377–384. [Google Scholar] [CrossRef]
- Strisciuglio, C.; Gianetti, E.; Martinelli, M.; Sciorio, E.; Staiano, A.; Miele, E. Does cow’s milk protein elimination diet have a role on induction and maintenance of remission in children with ulcerative colitis? Acta Paediatr. 2013, 102, e273–e278. [Google Scholar] [CrossRef]
- Cohen, S.A.; Gold, B.D.; Oliva, S.; Lewis, J.; Stallworth, A.; Koch, B.; Eshee, L.; Mason, D. Clinical and Mucosal Improvement with Specific Carbohydrate Diet in Pediatric Crohn Disease. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 516–521. [Google Scholar] [CrossRef]
- Devkota, S.; Wang, Y.; Musch, M.W.; Leone, V.; Fehlner-Peach, H.; Nadimpalli, A.; Antonopoulos, D.A.; Jabri, B.; Chang, E.B. Dietary-fat-induced taurocholic acid promotes pathobiont expansion and colitis in Il10-/- mice. Nature 2012, 487, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Agus, A.; Denizot, J.; Thevenot, J.; Martinez-Medina, M.; Massier, S.; Sauvanet, P.; Bernalier-Donadille, A.; Denis, S.; Hofman, P.; Bonnet, R.; et al. Western diet indices a shift in the microbiome composition enhancing susceptibility to Adherent-Invastive E coli infection and intestinal inflammation. Sci. Rep. 2016, 6, 19032. [Google Scholar] [CrossRef] [PubMed]
- Muhomah, T.; Nishino, N.; Katsumata, E.; Haoming, W.; Tsuruta, T. High-fat diet reduces the level of secretory immunoglobulin A coating of commensal gut microbiota. Biosci. Microb. Food Health 2019, 38, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Chapkin, R.S.; Davidson, L.A.; Ly, L.; Weeks, B.R.; Lupton, J.R.; McMurray, D.N. Immunomodulatory Effects of n-3 Fatty Acids: Putative Link to Inflammation and Colon Cancer. J. Nutr. 2007, 137, 200s–204s. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.; Wall, R.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. Health Implications of High Dietary Omega-6 Polyunsaturated Fatty Acids. J. Nutr. Metab. 2011, 539426. [Google Scholar] [CrossRef]
- Schreiner, P.; Martinho-Grueber, M.; Struderus, D.; Vavricka, S.R.; Tilg, H.; Biedermann, L. Nutrition in Inflammatory Bowel Disease. Digestion 2019, 101, 120–135. [Google Scholar] [CrossRef]
- Uchiyama, K.; Nakamura, M.; Odahara, S.; Kiodo, S.; Katahira, K.; Shiraishi, H.; Ohkusa, T.; Fujise, K.; Tajiri, H. N-3 Polyunsaturated Fatty Acid Diet Therapy for Patients with Inflammatory Bowel Disease. Inflamm. Bowel Dis. 2010, 16, 1696–1707. [Google Scholar] [CrossRef]
- Barnes, E.; Nestor, M.; Onyewadume, L.; De Silva, P.; Korzenik, J. High Dietary Intake of Specific Fatty Acids Increases Risk of Flares in Patients with Ulcerative Colitis in Remission During Treatment with Aminosalicylates. Clin. Gastroenterol. Hepatol. 2017, 15, 1390–1396. [Google Scholar] [CrossRef]
- Chassaing, B.; Koren, O.; Goodrich, J.K.; Poole, A.C.; Srinivasan, S.; Ley, R.E.; Gewirtz, A.T. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 2015, 519, 92–96. [Google Scholar] [CrossRef]
- Roberts, C.L.; Keita, A.V.; Duncan, S.H.; O’Kennedy, N.; Soderholm, J.D.; Rhodes, J.M.; Campbell, B.J. Translocation of Crohn’s disease Escherichia coli across M-cells: Contrasting effects of soluble plant fibres and emulsifiers. Gut 2010, 59, 1331–1339. [Google Scholar] [CrossRef]
- Ruiz, P.; Moron, B.; Becker, H.; Lang, S.; Atrott, K.; Spalinger, M.R.; Scharl, M.; Wotjtal, K.A.; Fischbeck-Terhalle, A.; Frey-Wagner, I.; et al. Titanium dioxide nanoparticles exacerbate DSS-induced colitis: Role of the NLRP3 inflammasome. Gut 2017, 66, 1216–1224. [Google Scholar] [CrossRef] [PubMed]
- Lomer, M.C.; Harvey, R.S.; Evans, S.M.; Thompson, R.P.; Powell, J.J. Efficacy and tolerability of a low microparticle diet in a double blind, randomised, pilor study in Crohn’s Disease. Eur. J. Gastroenterol. Hepatol. 2001, 13, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Thangaraju, M.; Cresci, G.; Liu, K.; Anath, S.; Gnanaprakasam, J.P.; Browning, D.D.; Mellinger, J.D.; Smith, S.B.; Digby, G.J.; Lambert, N.A. GPR109A Is a G-Protein-Coupled Receptor for the Bacterial Fermentation Product Butyrate and Functions as a Tumor Suppressor in Colon. Cancer Res. 2009, 69, 2826–2832. [Google Scholar] [CrossRef] [PubMed]
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.; Lannoy, V.; Decobecq, M.; Brezillon, S.; Dupriez, V.; Vassart, G.; Van Damme, J.; et al. Functional Characterization of Human Receptors for Short Chain Fatty Acids and Their Role in Polyporphonuclear Cell Activation. J. Biol. Chem. 2003, 287, 25481–25489. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Thangaraju, M.; Prasad, P.; Martin, P.M.; Lambert, N.A.; Boettger, T.; Offermanns, S.; Ganapathy, V. Blockade of dendritic cell development by bacterial fermentation products butyrate and propionate through transporter (Slc5a8(- dependent inhibition of histone deacetylases. J. Biol. Chem. 2010, 285, 27601–27608. [Google Scholar] [CrossRef]
- Kurita-Ochiai, T.; Hashizume, T.; Yonezawa, H.; Ochiai, K.; Yamamoto, M. Characterization of the effects of butyric acid on cell proliferation, cell cycle distribution and apoptosis. Pathog. Dis. 2006, 47, 67–74. [Google Scholar] [CrossRef]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; Van der Veeken, J.; DeRoos, P.; Lui, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef]
- Smith, P.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly-Y, M.; Glickman, J.; Garrett, W. The Microbial Metabolites, Short-Chain Fatty Acids, Regulate Colonic Treg Cell Homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef]
- Raqib, R.; Sarker, P.; Bergman, P.; Ara, G.; Lindh, M.; Sack, D.A.; Nasirul Islam, K.M.; Gudmundsson, G.H.; Andersson, J.; Agerberth, B. Improved outcome of shigellosis associated with butyrate induction of an engogenous peptide antibiotic. Proc. Natl. Acad. Sci. USA 2006, 103, 9178–9183. [Google Scholar] [CrossRef]
- Xiong, H.; Guo, B.; Gan, Z.; Song, D.; Lu, Z.; Yi, H.; Wu, Y.; Wang, Y.; Du, H. Butyrate upregulates endogenous host defence peptides to enhance disease resistance in piglets via histone deacetylase inhibition. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Finnie, I.; Dwarakanath, A.D.; Taylor, B.A.; Rhodes, J.M. Colonic mucin synthesis is increased my sodium butyrate. Gut 1995, 36, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.J.; Zheng, L.; Campbell, E.L.; Saeedi, B.; Scholz, C.C.; Bayless, A.J.; Wilson, K.E.; Glove, L.E.; Kominsky, D.J.; Magnuson, A.; et al. Crosstalk between microbiota-derived short-chain fatty acids and intestinal epithelial hif augments tissue barrier function. Cell Host Microb. 2015, 17, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Macia, L.; Tran, J.; Vieira, A.; Leach, K.; Stanley, D.; Luong, S.; Maruya, M.; McKenzie, C.I.; Hijikata, A.; Wong, C.; et al. Metabolite-sensing receptors gpr43 and gpr109a facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat. Commun. 2015, 6, 6734. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.K.; Kim, T.I.; Kim, Y.; Choi, C.H.; Yang, K.M.; Chae, B.; Kim, W.H. Cellular differentiation-induced attenuation of LPS response in HT-29 Cells is related to the down-regulation of TLR4 expression. Biochem. Biophys. Res. Commun. 2005, 337, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Qian, J.; Wang, Q.; Wang, F.; Ma, Z.; Qiao, Y. Butyrate protects rat liver agains total hepatic ischemic reperfusion injury with bowel congestion. PLoS ONE 2014, 9, e106184. [Google Scholar]
- Asarat, M.; Vasiljevic, T.; Apostolopoulos, V.; Donkor, O. Short-chain fatty acids regulare secretion of IL-8 from human intestinal epithelial cell lines in vitro. Immunol. Investig. 2015, 44, 678–693. [Google Scholar] [CrossRef]
- Blais, M.; Seidman, E.; Asselin, C. Duel effect of butyrate on IL-1 Beta-Mediated Intestinal Epithelial Cell Inflammatory Response. DNA Cell Biol. 2007, 26, 133–147. [Google Scholar] [CrossRef]
- Zapolska-Downar, D.; Siennicka, A.; Kaczmarczyk, M.; Kolodziej, B.; Naruszewicz, M. Butyrate inhibits cytokine-induced vcam-1 and icam-1 expression in cultured endothelial cells: The role of nf-kppab and pparalpha. J. Nutr. Biochem. 2004, 15, 220–228. [Google Scholar] [CrossRef]
- Vinolo, M.; Rodrigues, H.; Hatanaka, E.; Sato, F.T.; Sampaio, S.C.; Curi, R. Suppressive effect of short-chain fatty acids on production of proinflammatory mediators by neutrophils. J. Nutr. Biochem. 2011, 22, 849–855. [Google Scholar] [CrossRef]
- NHMRC. Fats: Total Fat & Fatty Acids. 2014. Available online: https://www.nrv.gov.au/nutrients/fats-total-fat-fatty-acids (accessed on 15 November 2020).
- Wang, F.; Lin, X.; Zhao, Q.; Li, J. Fat intake and risk of ulcerative colitis: Systematic review and dose-response meta-analysis of epidemiological studies. J. Gastroenterol. Hepatol. 2017, 31, 19–27. [Google Scholar] [CrossRef]
- Ananthakrishnan, A.; Khalili, H.; Konijeti, G.G.; Higuchi, L.M.; De Silva, P.; Fuchs, C.S.; Willett, W.C.; Richter, J.M.; Chan, A.T. Long-term intake of dietary fat and risk of ulcerative colitis and Crohn’s disease. Gut 2014, 63, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.; Rhodes, J.M.; Lindsay, J.O.; Aberu, M.T.; Kamm, M.A.; Gibson, P.R.; Gasche, C.; Silverberg, M.S.; Mahadevan, U.; Sigall Boneh, R.; et al. Dietary Guidance From the International Organization for the Study of Iinflammatory bowel diseases. Clin. Gastroenterol. Hepatol. 2020, 18, 1381–1392. [Google Scholar] [CrossRef] [PubMed]
- Dolan, K.T.; Chang, E.B. Diet, gut microbes, and the pathogenesis of Iinflammatory bowel diseases. Mol. Nutr. Food Res. 2017, 61, 1600129. [Google Scholar] [CrossRef] [PubMed]
- Chassaing, B.; Van de Wiele, T.; De Bodt, J.; Marzorati, M.; Gewirtz, A. Dietary emulsifiers directly alter human microbiota composition and gene expression ex vivo potentiating intestinal inflammation. Gut 2017, 66, 1414–1427. [Google Scholar] [CrossRef] [PubMed]
- Hajam, I.A.; Dar, P.A.; Shahnawaz, I.; Jaume, J.C.; Lee, J.H. Bacterial flagellin-a potent immunomodulatory agent. Exp. Mol. Med. 2017, 49, e373. [Google Scholar] [CrossRef]
- Tobacman, J. Review of harmful gastrointestinal effects of carrageenan in animal experiments. Environ. Health Perspect. 2001, 109, 983. [Google Scholar] [CrossRef]
- Nickerson, K.; Homer, C.; Kessler, S.P.; Dixon, L.J.; Kabi, A.; Gordon, I.O.; Johnson, E.E.; De la Motte, C.A.; Mc Donald, C. The dietary polysaccharide maltodextrin promotes Salmonella survival and mucosal colonization in mice. PLoS ONE 2014, 9, e101789. [Google Scholar] [CrossRef]
- Nickerson, K.; McDonald, C. Crohn’s disease-associated adherent-invasive Escherichia coli adhesion is enhances by esposure to the ubiquitous dietary polysaccharide maltodextrin. PLoS ONE 2012, 7, e52132. [Google Scholar] [CrossRef]
- Roberts, C.L.; Rushworth, S.L.; Richman, E.; Rhodes, J.M. Hypothesis: Increased consumption of emulsifiers as an explanation for the rising incidence of Crohn’s Disease. J. Crohn’s Colitis 2013, 7, 338–341. [Google Scholar] [CrossRef]
- Aljebab, F.; Choonara, I.; Conroy, S. Systematic Reveiw of the Toxicity of Long-Course Oral Corticosteroids in Children. PLoS ONE 2017, 12, e0170259. [Google Scholar] [CrossRef]
- Borrelli, O.; Cordischi, L.; Cirulli, M.; Paganelli, M.; Labalestra, V.; Uccini, S.; Russo, P.M.; Cucchiara, S. Polymeric Diet Alone Versus Corticosteroids in the Treatment of Active Pediatric Crohn’s Disease: A Randomized Controlled Open-Label Trial. Clin. Gastroenterol. Hepatol. 2006, 4, 744–753. [Google Scholar] [CrossRef] [PubMed]
- Wall, C.; Gearry, R.; Day, A. Treatment of Active Crohn’s Disease with Exclusive and Partial Enteral Nutrition: A Pilot Study in Adults. Inflamm. Intest. Dis. 2018, 2, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.; Macdonald, S.; Hill, S.M.; Thomas, A.; Murphy, M.S. Treatment of active Crohn’s disease in children using partial enteral nutrition with liquid formula: A randomised controlled trial. Gut 2005, 55, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.; Wine, E.; Assa, A.; Sigall Boneh, R.; Shaoul, R.; Kori, M.; Cohen, S.; Peleg, S.; Shamaly, H.; On, A. Crohn’s Disease Exclusion Diet Plus Partial Enteral Nutrition Induces Sustained Remission in a Randomised Controlled Trial. Gastroenterology 2019, 157, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Svolos, V.; Hansen, R.; Nichols, B.; Quince, C.; Ijaz, U.Z.; Papadopoulou, R.T.; Edwards, C.A.; Watson, D.; Alghamdi, A.; Brejnrod, A. Treatment of Active Crohn’s Disease with an Ordinary Food-based Diet That Replicates Exclusive Enteral Nutrition. Gastroenterology 2019, 156, 1354–1367. [Google Scholar] [CrossRef]
- Suskind, D.L.; Cohen, S.A.; Brittnacher, M.J.; Wahbeh, G.; Lee, D.; Shaffer, M.; Braly, K.; Hayden, H.; Klein, J.; Gold, B.; et al. Clinical and Fecal Microbial Changes With Diet Therapy in Active Inflammatory Bowel Disease. J. Clin. Gastroenterol. 2018, 52, 155–163. [Google Scholar] [CrossRef]
- Hansen, T.; Duerksen, D. Enteral Nutrition in the Management of Pediatric and Adult Crohn’s Disease. Nutrients 2018, 10, 537. [Google Scholar] [CrossRef]
- Gatti, S.; Galeazzi, T.; Franceschini, E.; Annibali, R.; Albano, V.; Verma, A.K.; De Angelis, M.; Lionetti, M.E.; Catassi, C. Effects of the Exclusive Enteral Nutrition on the Microbiota Profile of Patients with Crohn’s Disease: A Systematic Review. Nutrients 2017, 9, 832. [Google Scholar] [CrossRef]
- Narula, N.; Dhillon, A.; Zhang, D.; Sherlock, M.E.; Tondeur, M.; Zachos, M. Enteral nutritional therapy for induction of remission in Crohn’s disease (Review). Cochrane Database Syst. Rev. 2018. [Google Scholar] [CrossRef]
- Tsertsvadze, A.; Gurung, T.; Court, R.; Clarke, A.; Sutcliffe, P. Clinical effectiveness and cost-effectiveness of elemental nutrition for the maintenance of remission in Crohn’s disease: A systematic review and meta-analysis. Health Technol. Assess. 2015, 19, 1–138. [Google Scholar] [CrossRef]
- Logan, M.; Gklkas, K.; Svolos, V.; Nichols, B.; Milling, S.; Gaya, D.R.; Seenan, J.P.; Macdonald, J.; Hansen, R.; Ijaz, U.Z. Analysis of 61 exclusive enteral nutritional formulas used in the management of active Crohn’s disease-new insights into dietary disease triggers. Aliment. Pharmacol. Ther. 2020, 51, 935–947. [Google Scholar] [CrossRef] [PubMed]
- Suskind, D.; Wahbeh, G.; Cohen, S.; Damman, C.; Klein, J.; Brlay, K.; Shaffer, M.; Lee, D. Patients percieve clinical benefit with specific carbohydrate diet for inflammatory bowel disease. Dig. Dis. Sci. 2016, 61, 3255–3260. [Google Scholar] [CrossRef] [PubMed]
- Kakodkar, S.; Farooqui, A.; Mikolaitis, S.; Mutlu, E.A. THe specific carbohydrate diet for inflammatory bowel disease. J. Acad. Nutr. Diet. 2015, 115, 1226–1232. [Google Scholar] [CrossRef] [PubMed]
- Denson, L.; Curran, M.; McGovern, D.; Koltun, W.A.; Duerr, R.; Kim, S.C.; Sartor, R.B.; Sylvester, F.A.; Abraham, C.; De Zoeten, E.F. Challenges in IBD Reserach: Precision Medicine. Inflamm. Bowel Dis. 2019, 25, S31–S39. [Google Scholar] [CrossRef]
- Borg-Bartolo, S.; Boyapati, R.; Satsangi, J.; Kalla, R. Precision medicine in inflammatory bowel disease: Concept, progress and challenges. F100 Res. 2020, 9. [Google Scholar] [CrossRef]
- Liaing, B.B.; Lim, A.G.; Ferguson, L.R. A Personalised Dietary Approach- A Way Forward to Manage Nutrient Deficiency, Effects of the Western Diet, and Food Intolerances in Inflammatory Bowel Disease. Nutrients 2019, 11, 1532. [Google Scholar] [CrossRef]
- Zeevi, D.; Korem, T.; Zomra, N.; Israeli, D.; Rothschild, D.; Weinberger, A.; Ben-Yacov, O.; Lador, D.; Avnit-Sagi, T.; Lotan-Pompan, M. Personalised Nutrition by Prediction of Glycaemic Responses. Cell 2015, 163, 1079–1094. [Google Scholar] [CrossRef]
- Mendes-Soares, H.; Raveh-Sadka, T.; Azulay, S.; Ben-Shlomo, Y.; Cohen, Y.; Ofek, T.; Stevens, J.; Bachrach, D.; Kashyap, P.; Segal, L.; et al. Model of personalised post prandial glycemic response to food for an Israeli cohort predicts responses in Midwestern American individuals. Am. J. Clin. Nutr. 2019, 110, 63–75. [Google Scholar] [CrossRef]
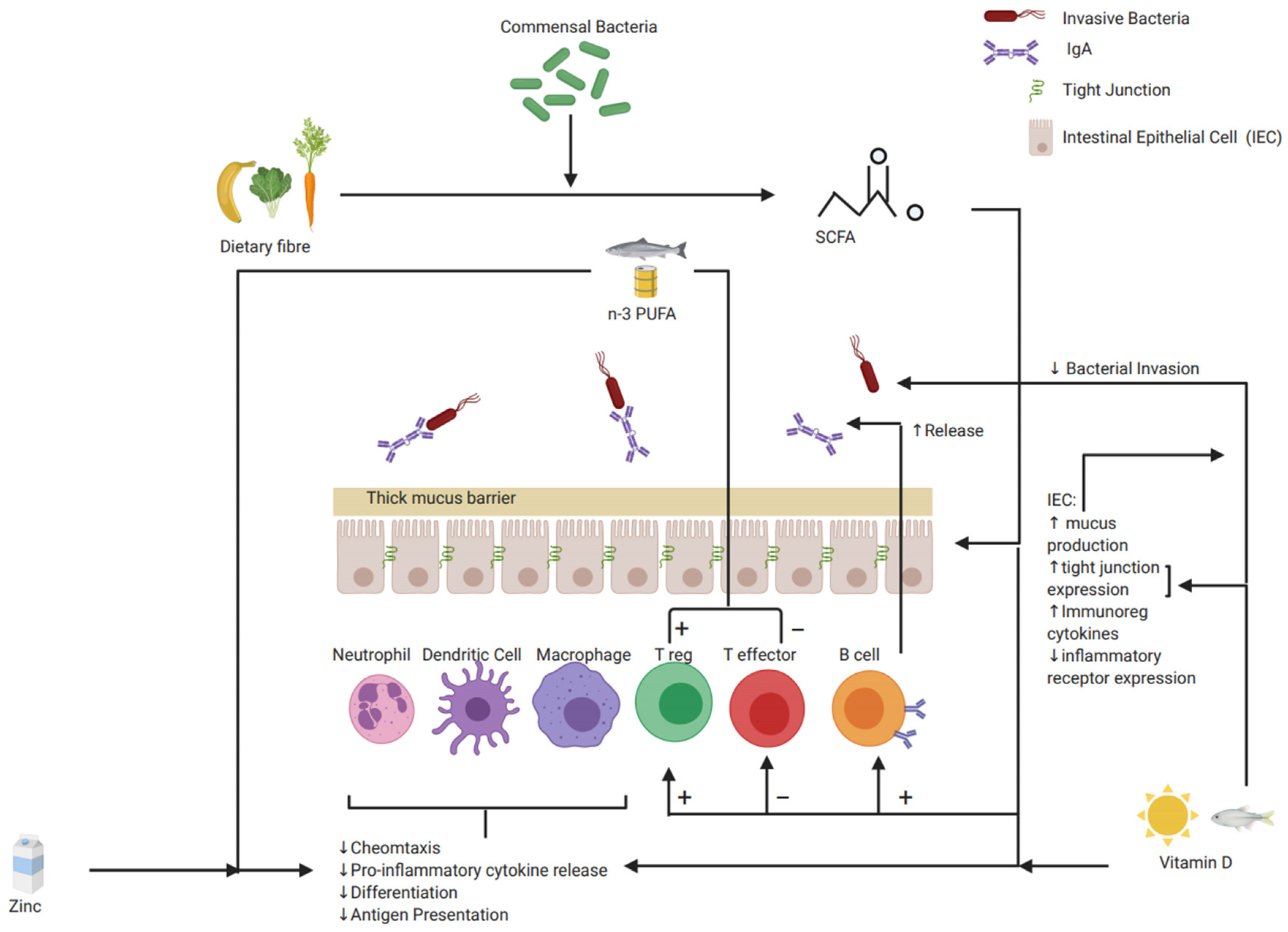
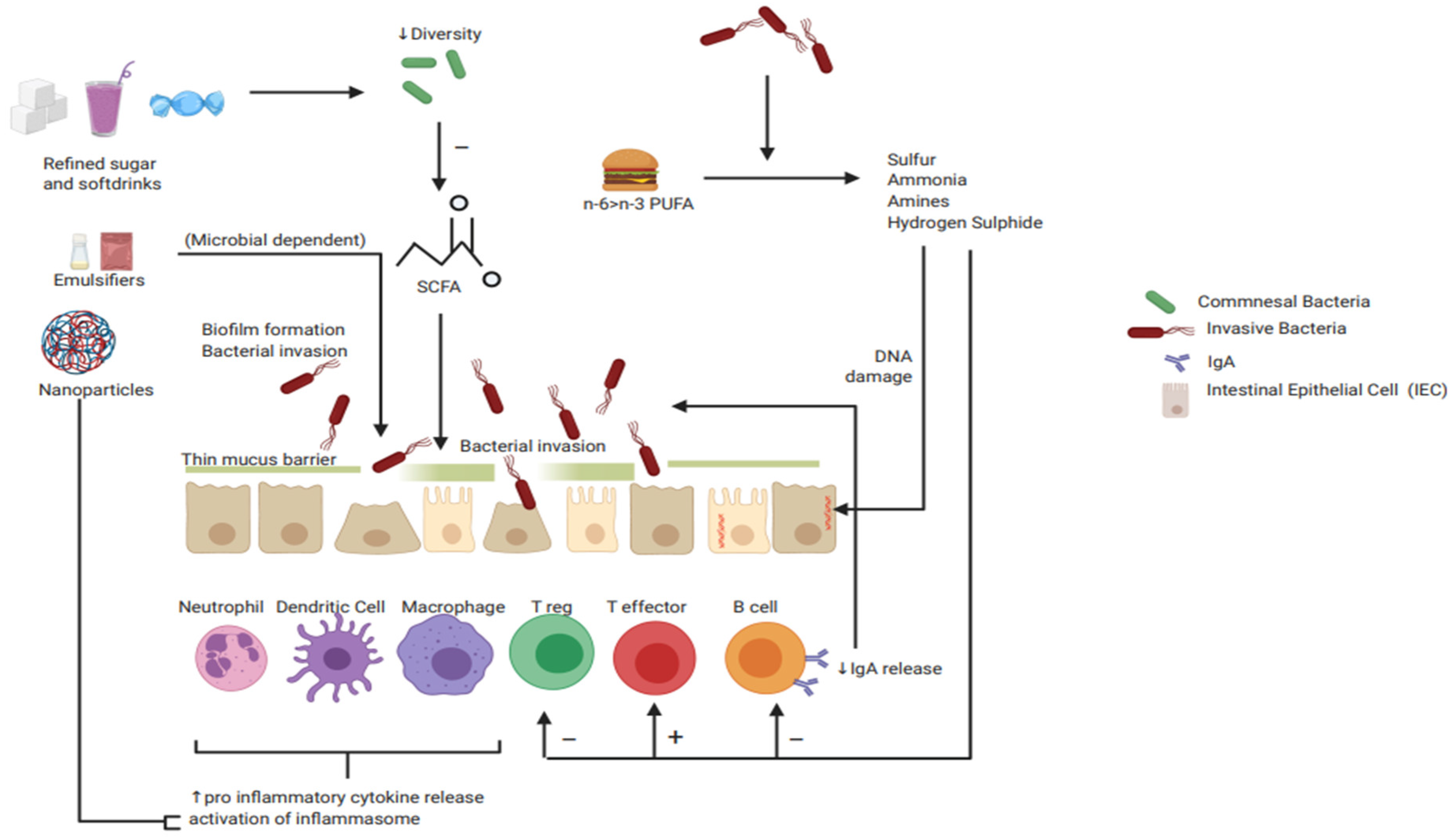
| Study | Year | Design | Sample Size | Follow up (Years) | IBD Type | Exposure | Impact on Risk |
|---|---|---|---|---|---|---|---|
| Shoda [40] | 1996 | Epidemiologic | 68,000 | 12 | CD | Correlations between dietary intake and CD risk | Total fat r 0.919 (p < 0.01) * Animal fat r 0.88 (p < 0.01) * Ratio n-6/n-3 PUFA 0.792 (p < 0.01) * |
| Amre [41] | 2007 | Case control | 202 controls 120 CD | Dietary consumption 1 year prior to diagnosis | CD | n-3 PUFA, highest vs. lowest quartile; Ratio n-3 PUFA/n-6 PUFA, highest vs. lowest quartile | OR 0.44 (95% CI 0.19–1.0) OR 0.32 (95% CI 0.14–0.71) * |
| Jantchou [42] | 2010 | Prospective cohort | 67,581 participants Incident IBD cases: 77 | 10.4 | CD and UC combined | Animal protein, 3rd vs. 1st tertile | HR 3.03 (1.45–6.34) * |
| John [43] | 2010 | Prospective cohort | 25,639 participants Incident UC cases: 22 | Mean 4.2 (1.8–4.2) | UC | Intake of DHA in the highest tertile | UC OR 0.43 (95% CI 0.22–0.86) * |
| Anathkrishnan [44] | 2012 | Prospective cohort | 72,719 Incident cases: 122 CD 123 UC | 22 | CD and UC | Validated prediction score of serum vitamin D level | CD HR (95% CI 0.3–0.99) UC 0.65 (95% CI 0.34–1.25) |
| Ananthakrishnan [45] | 2014 | Prospective cohort | 170,776 Incident cases: CD 269 UC 338 | 26 | CD and UC | n-3 PUFA intake in highest quintile; transfat intake in highest quintile | UC HR 0.72 (95% CI 0.51–1.01) UC HR 1.34 (95% CI 0.94–1.92) |
| Ananthakrishnan [45] | 2014 | Prospective cohort | 170,776 Incident cases: CD 269 UC 338 | 26 | CD and UC | Fibre intake, highest quintile | CD HR 0.59 (95% CI 0.39–0.9) * UC HR 0.82 (95% CI 0.58–1.17) |
| Ananthakrishnan [46] | 2015 | Prospective cohort | 39,511 Incident cases: CD 70 UC 103 | 26 | CD and UC | Fibre intake, highest quartile | CD HR 0.47 (95% CI 0.23–0.98) * UC: not significant |
| Anathkrishnan [47] | 2015 | Prospective cohort | 170 776 Incident cases CD 269 UC 338 | 26 | CD and UC | Dietary zinc intake, highest quartile vs. lowest quartile | CD HR 0.63 (95% CI 0.43–0.93) * UC HR 0.96 (95% CI 0.68–1.34) |
| Ananthakrishnan [46] | 2015 | Prospective cohort | 116,686 Incident cases CD 70 UC 103 | 19 | CD and UC | Fish intake, highest vs. lowest quartile | CD HR 0.43 (95% CI 0.21–0.90) * UC HR 0.99 (95% CI 0.55–1.77) |
| Racine [48] | 2016 | Prospective cohort | 366,351 participants Incident cases CD 117 UC 256 | 18 | CD and UC | High sugar and soft drinks intake, 5th vs. 1st quintile (>2 years post-dietary assessment) | UC IRR 1.68 (1.00–2.82) * No impact on CD risk |
| Anderson [49] | 2018 | Case control (from prospective cohort) | 401,326 Incident cases CD 104 UC 221 | 19 | CD and UC | Dietary fibre intake, quartile 4 vs. 1 | CD non-smokers OR 0.5 (95% CI 0.29–0.86) * CD OR 0.83 (0.38–1.81) UC OR 1.22 (0.71–2.08) |
| Opstelten [50] | 2018 | Prospective cohort | 359,728 Incident cases: CD 72 UC 169 | Up to 15.7 years | CD and UC | Serum vitamin D at baseline, highest vs. lowest quartile | CD OR 0.69 (95% CI 0.29–1.6) UC OR 1.22 (95% CI 0.67–2.2) |
| Dong [51] | 2020 | Prospective cohort | 413,953 participants CD 177 UC 595 | 16 | CD and UC | Animal protein intake increase/10 g/day; red meat, 4th vs. 1st quartile | HR 1.1 (95% CI 1.004–1.21) * HR 1.41 (95% CI 1.03–1.92) * (Only significant for UC) |
| Khalili [52] | 2020 | Prospective cohort | 83,147 Incident cases: CD 164 UC 395 | 17 | CD and UC | Adherence to Mediterranean diet (highest vs. lowest Med score) | CD HR 0.42 (95% CI 0.22–0.80) * UC HR 1.08 (95% CI 0.74–1.58) |
| Dietary Component | Proposed Mechanism | Impact on Disease Control | |
|---|---|---|---|
| Crohn’s Disease | Ulcerative Colitis | ||
| Fibre |
Fermented to SCFA by colonic bacteria. Downstream effects include
| Reduces risk of flares [59] | Improves disease activity [60] |
| FODMAPs | Fermented by commensal bacteria in distal gut to produce gas and distension [62] | Exclusion improves symptoms [63,65] | Exclusion improves symptoms [63,65] |
| Wheat/Gluten | ATI proteins stimulate release of inflammatory cytokines, activate Toll-Like Receptors and induce a T-Cell response [69,70] | Improved symptoms, especially in stricturing CD [71,72,73] | Improved symptoms [71,72,73] |
| Sugar | Increased gut permeability Decreased microbial diversity Increased pro-inflammatory cytokines [66,67] | Restriction improved clinical indices and mucosal healing in a small paediatric study [101] | Reported by patients but no trials to support [21] |
| Fat | Saturated fat>increased taurine conjugation of hepatic bile acids>increases sulphate reducing microorganisms>promote pro-inflammatory T-cell response [102,103,104] n-3 PUFA anti-inflammatory vs. n-6 PUFA pro-inflammatory [105,106,107] | Higher n-3 PUFA: n-6 PUFA ratio improves remission rates [108] | High dietary fat intake [78], in particular myristic acid (found in palm oil, coconut oil and dairy fat) associated with flares [109] |
| Emulsifiers | Erode mucus barrier [110] Increase translocation of invasive E. Coli [111] Increase pro-inflammatory cytokines [110] | No studies in humans | No studies in humans |
| Nanoparticles | Activate inflammasome [112] | Small pilot study showed improved remission rates [113], not replicated in larger study [99] | No studies in humans |
| Meat | Fermentation produces by-products that promote DNA instability [75,76] Also high in fat (see above) | No impact demonstrated [79] | Red meat increased risk of disease flares [78] |
| Zinc | Modulates TNF expression which decreases myeloperoxidase enzyme activity [80,81,82] | Low zinc levels associated with increased hospitlaisation and surgery [85] | Low zinc levels associated with increased hospitlaisation and surgery [85] |
| Vitamin D | Strengthens epithelial barrier and tight junction protein expression [88] Decreases maturation and function of Dendritic cells with subsequent decreased T-cell activation [89] | Inverse relationship between serum vitamin D and CDAI and HBI as well as poorer health-related QOL scores [93,94] | Inverse relationship between serum vitamin D and UCDI [94] |
| Calcium | Low-calcium diet did not improve disease control [99] | Cow’s milk protein exclusion did not improve disease control [100] | |
| Immune Cells | Intestinal Epithelial Cells: Barrier | Pro Inflammatory Mediators | Anti-Inflammatory Mediators |
|---|---|---|---|
|
|
| Diet | Description | Evidence |
|---|---|---|
| Exclusive Enteral Nutrition | 100% of nutritional requirements delivered via liquid formula orally or via nasogastric tube | Equivalent to corticosteroids in achieving remission in paediatric population [143] Adherence is challenging in adults [144] |
| Partial Enteral Nutrition | 50% Enteral nutrition plus 50% unrestricted diet | Inferior to EEN [145] |
| 50% Enteral nutrition plus 50% select whole foods | Equivalent to EEN in response rates Improved patient acceptability compared to EEN [146] | |
| Ordinary Food Diet (CD-TREAT) | Food based diet with exclusion of specific components in common with EEN such as gluten, lactose and alcohol | Reduced inflammatory markers and disease activity at week 12 and better tolerated than EEN (though n = 5) [147] |
| Specific Carbohydrate Diet | Excludes grains, sugars, processed food and most dairy | Reduced inflammation at 12 weeks [148] |
| Reduced microparticles diet | Dietary advice to avoid food containing micro particles | Pilot study of 20 patients demonstrated improvement in CDAI at 4 months [113], not replicated in larger multicentre 82 patient cohort [99] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wark, G.; Samocha-Bonet, D.; Ghaly, S.; Danta, M. The Role of Diet in the Pathogenesis and Management of Inflammatory Bowel Disease: A Review. Nutrients 2021, 13, 135. https://doi.org/10.3390/nu13010135
Wark G, Samocha-Bonet D, Ghaly S, Danta M. The Role of Diet in the Pathogenesis and Management of Inflammatory Bowel Disease: A Review. Nutrients. 2021; 13(1):135. https://doi.org/10.3390/nu13010135
Chicago/Turabian StyleWark, Gabrielle, Dorit Samocha-Bonet, Simon Ghaly, and Mark Danta. 2021. "The Role of Diet in the Pathogenesis and Management of Inflammatory Bowel Disease: A Review" Nutrients 13, no. 1: 135. https://doi.org/10.3390/nu13010135
APA StyleWark, G., Samocha-Bonet, D., Ghaly, S., & Danta, M. (2021). The Role of Diet in the Pathogenesis and Management of Inflammatory Bowel Disease: A Review. Nutrients, 13(1), 135. https://doi.org/10.3390/nu13010135






