Abstract
Evidence of synergic health effects of co-supplementation with vitamin D and probiotics is emerging. Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses PRISMA statement, scientific databases and the grey literature were searched, and a narrative review and risk of bias assessment were conducted. Seven randomized controlled trials were included, which had low risk of bias. Six studies were double-blind, and once single-blind, extended over 6–12 weeks, and included 50–105 participants. Conditions explored included schizophrenia, gestational diabetes, type 2 diabetes and coronary heart disease, polycystic ovarian syndrome, osteopenia, irritable bowel syndrome (IBS), and infantile colic. Supplementation frequency was daily or bi-monthly, with mainly vitamin D3, and Lactobacillus, Bifidobacterium, and Streptococcus. Comparators were placebo, vitamin D, lower vitamin D dose, and probiotics and lower vitamin D dose. The co-supplementation yielded greater health benefits than its comparators did in all studies except in one assessing IBS. Beneficial effects included decreased disease severity, improved mental health, metabolic parameters, mainly insulin sensitivity, dyslipidemia, inflammation, and antioxidative capacity, and lower use of healthcare. Co-supplementation of vitamin D and probiotics generated greater health benefits than its comparators did. More studies in other diseases and various populations are needed to confirm these findings and to elucidate the optimal form, composition, and frequency of this co-supplementation.
1. Introduction
The gut microbiota refers to the assemblage of microorganisms, including bacteria, viruses, and fungi, located in the gastrointestinal (GI) tract [1]. There has been increasing emphasis on the role of the microbiota in physiology, suggesting that it can be considered as another human organ [2]. Furthermore, emerging evidence suggests that this invisible organ is a key driver of human health and disease. Gut microbiota plays a critical role in maintaining metabolic and immune health, synthesis of vitamins, obtaining inaccessible nutrients from the diet, renewal of epithelial cells, fat storage, maintaining intestinal barrier integrity, and brain development [3,4]. Dysbiosis, or alteration in the gut microbiota composition, is a crucial risk factor for the development of several disorders such as inflammatory bowel disease, obesity, diabetes, asthma, and allergies [5,6]. The gut microbiota composition is affected by intrinsic and extrinsic factors like genetics, age, dietary changes, in addition to physiological and psychological stress [2,7].
Specifically, vitamin D and the vitamin D receptor (VDR) were shown to modulate the gut microbiota [8]. Increased VDR expression may decrease microbial dysbiosis, enhance barrier function, increase the expression of antimicrobial peptides, decrease pro-inflammatory cytokines, and increase the commensal production of short-chain fatty acids [2,8]. Likewise, probiotics, which are ingestible nonpathogenic living microorganisms, were also shown to improve the balance of intestinal microbiota by regulating microbial components and metabolites [9]. Probiotics simulate the immune system, balance commensal and pathogenic bacteria, and reestablish homeostasis. They protect barrier integrity, alter toxic compounds, and host products. Thus, they ameliorate inflammation and prevent and repair cell damage [9].
Vitamin D deficiency and defects in VDR signaling have been related to several metabolic, cardiovascular, neurodevelopmental and cancer diseases [10,11]. Yet, interventional studies have conflicting evidence on the effect of vitamin D supplementation in their treatment [12,13,14,15,16]. Similarly, human probiotic supplementation studies generated conflicting evidence regarding the effectiveness of probiotics in the treatment of several health conditions such as allergies, GI disorders, metabolic syndrome, and obesity [17,18,19,20].
Recently, a promising evidence of synergic effects of combined supplementation with vitamin D and probiotics in modulating the gut microbiota and metabolome, in addition to fostering healthy microbe–host interactions, is emerging [9,21,22]. This co-supplementation holds a preventive and therapeutic potential with crucial clinical implications. Biologically plausible mechanisms support this interplay. Probiotics were shown to increase vitamin D intestinal absorption, and increase VDR protein expression and transcriptional activity [9]. Likewise, VDR status seems to be crucial in regulating the mechanisms of action of probiotics and modulating their anti-inflammatory, immunomodulatory and anti-infective benefits, suggesting a two-sided pathway [6,8].
The aim of this systematic review is to investigate the literature and summarize the available evidence of randomized controlled trials (RCTs) on the various health effects of a combined supplementation of vitamin D and probiotics among children and adults.
2. Materials and Methods
2.1. Review Design
The reporting of this systematic review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [23]. A predefined protocol for this systematic review was registered at the OSF registries.
2.2. Criteria for Study Inclusion
Randomized controlled trials (RCTs) conducted on adults or children, healthy or with disease other than those known to influence vitamin D metabolism, and including an intervention group that received a co-supplementation of vitamin D and probiotics, and a control group of placebo, or a lower dose of vitamin D or probiotics, or a different form of vitamin D, or different strains of probiotics, were included in this systematic review. RCTs with a duration of a minimum of 1 month were included; this duration was deemed sufficient for the intervention to produce an effect. Additionally, RCTs involving other co-interventions were included, only if both arms received the same co-intervention.
Studies were excluded if they were non-randomized, uncontrolled, involving participants taking medication known to influence vitamin D metabolism or with conditions affecting vitamin D metabolism such as chronic kidney disease, chronic liver disease, or malabsorption states, or entailing a supplementation with either vitamin D or probiotics.
2.3. Search Strategy
The systematic search included Medical Subject Headings (MeSH) and keywords for three concepts: (1) vitamin D, (2) probiotics, and (3) randomized controlled trial, and was conducted in PubMed, MEDLINE, CINAHL, EMBASE, the Cochrane Library, ClinicalTrials.gov, and the International Clinical Trials Registry Platform (ICTRP), from inception until 4 November 2020, without language restrictions. The electronic search strategy, detailed in the Tables S1 and S2, was validated by a medical information specialist. Reference lists of included RCTs and relevant reviews were also hand-searched for eligible studies.
2.4. Study Selection
The titles and/or abstracts retrieved by the search were screened by two pairs of authors, and the full text of all relevant papers was assessed for eligibility independently and in duplicate. A calibration exercise was conducted before study selection to ensure the validity of the process. Inconsistencies were discussed amongst reviewers, and unresolved discrepancies were settled by a third reviewer.
2.5. Data Extraction
Data from the selected articles were extracted by two pairs of authors using a data extraction form. Changes from baseline for the intervention were compared with the control in all the parameters analyzed. A calibration exercise was first conducted. Disagreements were resolved through discussion or with the help of a third reviewer.
2.6. Quality Assessment
The risk of bias for the included studies was assessed using the Cochrane criteria (sequence generation, allocation concealment, blinding of participants and outcome assessors, incomplete outcome data, and selective outcome reporting) [24], whereby each potential source of bias was graded as low, high, or unclear risk. The process was carried out by two pairs of authors independently and in duplicate. They underwent a calibration exercise before performing the assessment of risk of bias. Conflicts were resolved through discussion amongst the pair of reviewers or through consultation with a third reviewer.
2.7. Data Synthesis
A narrative review of the findings was performed and is included in Table S2.
3. Results
3.1. Search Results
Study selection process is detailed in Figure 1, whereby seven studies meeting the inclusion criteria were included in the systematic review.
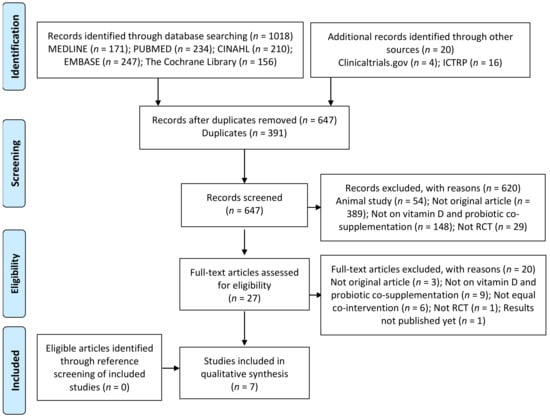
Figure 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Diagram of Study Selection.
3.2. Characteristics of Included Studies
Characteristics of included RCTs are detailed in Table 1. The studies were published between 2015 [25,26] and 2019 [27,28]. Five studies were conducted in Iran [27,28,29,30,31], one in Italy [25], and one in the United Kingdom [26]. All the studies were randomized double-blind [26,27,28,29,30,31], except for Savino et al. [25], which was single-blind. The duration of the studies ranged between 6 [29,31] to 12 weeks [25,26,27,28,30]. The number of participants ranged between 50 [31] and 105 [25]. The studies were conducted on infants [25], pregnant women [29], and other adults with diseases [26,27,28,30,31]. Health conditions that were studied included schizophrenia [27], gestational diabetes mellitus (GDM) [29], type 2 diabetes mellitus (T2DM) and coronary heart disease (CHD) [30], polycystic ovarian syndrome (PCOS) [28], osteopenia [31], irritable bowel syndrome [26], and infantile colic [25].
In the studies by Ghaderi et al. [27], Ostadmohammadi et al. [28], Raygan et al. [30] interventions consisted of a co-supplementation with vitamin D and probiotics, and the control group received placebo only [27,28,30]. In the study by Tazzyman et al. [26], the intervention group received a co-supplementation with vitamin D and probiotics, one of the control groups received a placebo, and the other one received placebo and vitamin D [26]. In Savino et al. [25], the intervention group received vitamin D and probiotics, but the control group received vitamin D only. In the study by Jafarnejad et al. [31], the intervention group received probiotics, yet vitamin D was supplemented in all groups. This co-intervention rendered the comparison between the intervention group receiving probiotics and vitamin D and the control group receiving placebo and a similar dose of vitamin D. Additionally, in the study by Jamilian et al. [29] the intervention consisted of a co-supplementation with vitamin D and probiotics; one of the control groups received probiotics, and the other one received placebo. Yet, in this study [29], all the groups also received a lower dose of vitamin D. This co-intervention rendered the comparison between the intervention group receiving probiotics and a high dose of vitamin D, the first control group receiving probiotics and a lower dose of vitamin D, and the second control group receiving placebo and a lower dose of vitamin D [29].

Table 1.
Characteristics of included studies.
Table 1.
Characteristics of included studies.
| First Author, Year, Country | Study Design | Duration | Study Population | Intervention | Control | Co-Intervention | Compliance/Drop-out |
|---|---|---|---|---|---|---|---|
| Ghaderi, 2019, Iran [27] | Randomized, double-blind, placebo-controlled trial | 12 weeks | n = 60, aged 25–65, 93.33% men, diagnosed with schizophrenia using DSM-IV-TR criteria with disease duration ≥2 years, PANSS score ≥55, treated with chlorpromazine (300–1000 mg/day, except clozapine) and anticholinergic agents (Trihexyphenidyl, 4–8 mg/day) during the last 6 months | Vitamin D3 and probiotic supplement:
| Placebo similar shape and packaging | None | Compliance: >90%Drop out: I: 13.33% C: 13.33% (Intention-to-treat analysis) |
| Jafarnejad, 2017, Iran [31] | Randomized, double-blind, placebo-controlled clinical trial | 6 weeks | n = 50, age 50–72 years, women with mild bone loss (osteopenia) diagnosed based on the World Health Organization criteria (T-score between −1.0 and −2.5) | Probiotic supplement: Lactobacillus casei 1.3 × 1010 CFU, Bifidobacterium longum 5 × 1010 CFU, Lactobacillus acidophilus 1.5 × 1010 CFU, Lactobacillus rhamnosus 3.5 × 109 CFU, Lactobacillus bulgaricus 2.5 × 108 CFU, Bifidobacterium breve 1 × 1010 CFU, and Streptococcus thermophilus 1.5 × 108 CFU/500 mg | Placebo similar in shape, size, odor, color and packaging | Vitamin D (200 IU daily) and Calcium (500 mg daily) | Compliance 100% Drop out: I: 20% C: 16% |
| Jamilian, 2018, Iran [29] | Randomized, double-blind, placebo-controlled clinical trial | 6 weeks | n = 87, women with GDM diagnosed by a “one-step” 2-h 75-g oral glucose tolerance test based on the ADA guidelines | Vitamin D and probiotic supplement:
| C1: 8 × 109 CFU/day of probiotic supplements C2: Placebo Similar in appearance, color, shape, size, odor, taste and packaging | Vitamin D3: 1000 IU and Vitamin B9: 400 mg, daily from the beginning of pregnancy, and Ferrous sulfate: 60 mg, daily from the secondtrimester | Compliance: 100% Drop out: I: 0% C1: 6.66% C2: 10% |
| Ostadmohammadi, 2019, Iran [28] | Randomized, double-blind, placebo-controlled clinical trial | 12 weeks | n = 60, aged 18–40 years, women with PCOS, diagnosed based on the Rotterdam criteria with BMI: 17–34 kg/m2 and insulin resistance: 1.4–4 | Vitamin D and probiotic supplement:
| Placebo similar in appearance, color, shape, size, odor, taste and packaging | None | Compliance 100%; No drop out |
| Raygan, 2018, Iran [30] | Randomized, double-blind, placebo-controlled clinical trial | 12 weeks | n = 60, age 45–85 years, 50% men, with T2DM diagnosed based on the criteria of the ADA and with CHD diagnosed as per the AHA with 2- and 3-vessel CHD | Vitamin D3 and probiotic supplement:
| Placebo similar in appearance, color, shape, size, odor, taste and packaging | None | Compliance > 90% Drop out: I: 13.33% C: 13.33% (Intention-to-treat analysis) |
| Savino, 2015, Italy [25] | Single-blind, randomized controlled, parallel-group trial | 12 weeks | n = 105, newborns aged less than 10 days of life, 48.5% boys, with gestational age between 37 and 42 weeks, birth weight from 2500 to 4300 g, and normal physical examination | Vitamin D and probiotic supplement:
| Vitamin D (400 IU daily) | None | No infants lost to follow- ups |
| Tazzyman, 2015, United Kingdom [26] | Double-blind, randomized, three-arm parallel design trial | 12 weeks | n = 51, 7.8% men, with previous clinical diagnosis of IBS and met the Rome III criteria and stratified according to vitamin D status at baseline (deficient: 25(OH)D <20 ng/mL; repleted: 25(OH)D >20 ng/mL) | Vitamin D3 and probiotic supplement:
| C1: Double placebo C2: Placebo and Vitamin D3 (400 IU daily) Similar in form, containing identical buffers | None | Compliance: 98% Drop out: 0% |
25(OH)D: 25-hydroxyvitamin D; ADA: American Diabetes Association; AHA: American Heart Association; BMI: Body Mass Index; C: Control; CFU: Colony Forming Units; CHD: Coronary Heart Disease; DDE: Daily Dose Equivalent; DSM-IV-TR: Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision; GDM: Gestational Diabetes Mellitus; I: Intervention; IBS: Irritable Bowel Syndrome; IU: International Unit; PANSS: The Positive and Negative Syndrome Scale; PCOS: Polycystic Ovary Syndrome; T2DM: Type 2 Diabetes Mellitus; TDD: Total Daily Dose.
The frequency of supplement administration ranged between daily [25,26,31] and bi-monthly [27,28,29,30]. Probiotic supplementation was given in the form of a capsule in all studies [25,26,27,28,29,30,31], whereas supplementation of vitamin D was either in the form of a capsule [26,27,28,29,30,31] or sublingual liquid spray [25]. The form of vitamin D supplemented was not specified in the studies by Jamilian et al. [29], Ostadmohammadi et al. [28], and Jafarnejad et al. [31], and studies by Ghaderi et al. [27], Raygan et al. [30], Tazzyman et al. [26], and Savino et al. [25] used vitamin D3, and the daily dose equivalent ranged from 200 International Units (IU) [31] to 4571.4 IU [29]. Probiotic strains that were investigated included Lactobacillus in all the studies [25,26,27,28,29,30,31], Bifidobacterium in all the studies [26,27,28,29,30,31] except for the one by Savino et al. [25], and Streptococcus only in Jafarnejad et al. [31]. The supplemented doses greatly varied across studies, and in the majority of the studies, it consisted of 8 × 109 Colony Forming Units (CFU) per day.
There was a high rate of compliance in all studies [25,26,27,28,29,30,31], and the drop-out rate ranged from 0% [25,26,28,29] to 20% [31], and was almost equal between the compared groups in all studies [25,26,27,28,30,31], except in Jamilian et al. [29].
3.3. Assessment of Risk of Bias
Risk of bias assessment of included RCTs is available in Table 2. In general, the quality of the RCTs design and reporting was high. In all studies [25,26,27,28,29,30,31], random allocation of participants was adequate, and allocation was concealed. Blinding of participants and personnel was reported in all of the included studies [26,27,28,29,30,31], except in the one by Saviano et al. [25], where both patients and physicians, except outcome assessors, were aware of their allocation. All studies reported complete outcome data [25,26,28,29,31], except for the studies conducted by Ghaderi et al. [27] and Raygan et al. [30] who did not mention how missing data were dealt with. Finally, in all studies [25,26,27,28,29,30,31], all pre-specified outcomes were reported on.

Table 2.
Risk of bias of included studies from consensus between a pair of raters.
3.4. Results of Included Studies
The outcomes assessed and the findings of included RCTs are presented in Table 3. In Ghaderi et al. [27], Ostadmohammadi et al. [28], Raygan et al. [30], and Savino et al. [25], co-supplementation with probiotics and vitamin D yielded greater health benefits than either placebo [27,28,30] or vitamin D on its own [25]. Specifically, in Ghaderi et al. [27], the co-supplementation, compared with placebo, had a favorable effect on schizophrenia symptoms severity, as well as other metabolic outcomes, mainly insulin sensitivity, inflammation, and antioxidative capacity. In Ostadmohammadi et al. [28], vitamin D and probiotic co-supplementation in women with PCOS, compared with placebo, had beneficial effects on mental health parameters, namely depression, anxiety and stress, as well as hormonal, inflammatory, and antioxidative parameters, and on the symptoms of PCOS, specifically, hirsutism. However, the co-supplementation was not associated with improvements in sex hormone-binding globulin, nor with other symptoms of PCOS, namely acne and alopecia, nor were there improvement in sleep quality [28]. In Raygan et al. [30], combined supplementation with vitamin D and probiotics for people with T2DM and CHD, compared with placebo, improved anxiety and depression, insulin sensitivity, inflammatory markers, antioxidative capacity and dyslipidemia, specifically high-density lipoprotein-cholesterol. However, this intervention did not result in a better control of fasting glucose, other markers of dyslipidemia, specifically triglycerides, very low and low lipoprotein-cholesterol, nor with blood pressures [30]. In the study by Savino et al. [25], compared with vitamin D supplementation alone, vitamin D and probiotic co-supplementation to newborns was associated with a reduction of more than two pediatric consultations and phone calls regarding infantile colic over a 12-week period. The co-supplementation was also associated with a lower use of pain-relieving agents and of infant formula [25].

Table 3.
Outcomes and results of included studies.
In the study by Jamilian et al. [29], all women with GDM in all groups were being supplemented with 1000 IU (low dose) vitamin D. The group supplemented with probiotics and high dose vitamin D, compared with placebo and low dose vitamin D, showed greater improvement in glucose control, insulin sensitivity, dyslipidemia, inflammatory markers, and antioxidative capacity [29]. Additionally, upon birth, newborns of mothers in this arm had lower incidence of both hyperbilirubinemia and hospitalization [29]. Moreover, the group supplemented with probiotics and high dose vitamin D, compared with probiotics and low dose vitamin D, exhibited a greater improvement in dyslipidemia, inflammation and antioxidative capacity [29]. Furthermore, newborns had better health outcomes [29]. Similarly, in the study by Jafarnejad et al. [31], all groups received 200 IU of vitamin D, and the group receiving probiotics had improvement in osteopenia markers (bone resorption and turnover), namely, bone-specific alkaline phosphatase, collagen type 1 cross-linked C-telopeptide, tumor necrosis factor α, and parathyroid hormone, but did not show an improvement in bone mineral density nor other serum indicators of osteopenia [31], compared with the group receiving placebo and vitamin D.
The only study where the co-supplementation was not found to be more effective than its comparators was the one conducted by Tazzyman et al. [26], where no significant difference in the symptoms of irritable bowel syndrome (IBS) was evident, between co-supplementation with probiotics and vitamin D, compared with vitamin D alone, or with placebo.
4. Discussion
So far, probiotic or vitamin D trials have shown major inconsistency in preventive or therapeutic effects on various health outcomes. The emergence of promising experimental studies on the interplay between vitamin D/VDR and probiotics in modulating the gut microbiota and influencing health and disease has led to several clinical trials of a combined supplementation in human subjects. Our exhaustive search identified seven eligible studies, which were included in our review. Our results show that a combined supplementation with vitamin D and probiotics was mostly more beneficial than placebo, vitamin D or probiotics alone in improving health outcomes in various populations, and suggest a dose-dependent effect.
Vitamin D deficiency had long been seen as a concern in metabolic and inflammatory disorders [32,33,34]. In the included studies, the majority of inflammatory markers improved with the co-supplementation. It is now evident that VDR expression regulates responses to inflammation through numerous mechanisms, such as inhibiting the nuclear factor-kappa B (NF-ĸB) pathway and activating autophagy [6]. VDR has an essential role for innate immune cells in intestinal inflammation, whereby the deletion of VDR in macrophages and granulocytes significantly increases the expression of pro-inflammatory cytokines in the colon [35]. In contrast, VDR signaling stimulates anti-inflammatory cytokine secretion [36]. Being a transcription factor, VDR can regulate the expression and signaling of target genes involved in intestinal inflammation and dysbiosis, such as Atg16l1 [6]. A genome-wide association study of the gut microbiota showed that VDR gene variation in humans influences the intestinal microbiota [37]. Genetic variation at the VDR locus significantly influences microbial co-metabolism and the gut–liver axis [37]. Another study in VDR knockout mice found that the lack of VDR in the intestine leads to dysbiosis, with profound alterations in the gut microbiome profile characterized by an increased abundance of Bacteroidaceae [38]. However, to date, the mechanisms behind the change of human VDR protein after using vitamin D supplementation and its role in regulating the gut microbiome in health and inflammation are not entirely known [6]. In parallel, the anti-inflammatory markers and properties of probiotics are reliant on VDR expression [39]. There are data showing that probiotic treatment enhances VDR expression and activity in the host. In a probiotic mono-associated pig model, treatment with Lactobacillus plantarum in cultured intestinal epithelial cells resulted in an increase in VDR expression and cathelicidin mRNA [39]. Other data show that probiotics did not inhibit inflammation in mice lacking VDR [39]. Future research is needed to enhance our understanding of the complex interplay of nuclear receptors and probiotics, specifically VDR’s contribution to probiotic-induced anti-inflammation and its potential role in inflammatory conditions such as inflammatory bowel diseases [39].
Besides, our review documented improvement in insulin sensitivity, anti-oxidative patterns, and dyslipidemia markers with co-supplementation of vitamin D and probiotics. The same positive direction was also highlighted elsewhere [6,8]. Previous research documented a functional link existing between probiotic metabolism and nuclear receptors involved in regulating insulin sensitivity [22]. In a mice model of genetic dyslipidemia and intestinal inflammation, supplementation with a mixture of probiotic strains, including Streptococcus thermophiles, Bifidobacterium breve, Bifidobacterium lactis, Lactobacillus acidophilus, Lactobacillus plantarum, Lactobacillus paracasei, and Lactobacillus helveticus modified the nuclear receptors’ expression including VDR, and caused their direct transactivation, leading to reversing insulin resistance in liver and fat tissues and protecting against steatohepatitis and atherosclerosis [40]. Yet, these results although emanating from high-quality studies, are far from being conclusive, and future trials are needed before we can confidently establish the effectiveness and superiority of this co-supplementation.
More human experimental studies are needed to fully elucidate the interplay between nuclear receptors and probiotics in metabolic diseases. Shaping our understanding of this unexplored path might pave the way for multi-target preventive and therapeutic strategies, especially in situations where dietary and lifestyle changes have failed [22].
Additionally, improvement in mental health has been reported in this review. Vitamin D is involved in numerous brain processes including neuroimmunomodulation, neuroprotection, as well as brain development; all of which suggests a link between vitamin D and mental health [41,42]. Vitamin D may positively affect mental health through up-regulating tyrosine hydroxylase gene expression and increasing bioavailability of key neurotransmitters, such as norepinephrine and dopamine [43]. In parallel, mechanisms through which gut bacteria can affect mental status include microflora biosynthesis and the regulation of neurotransmitters, including serotonin [44] and gamma aminobutyric acid (GABA) [45]. Existing evidence also pinpoint an association between mood disorders and gut microbiota, and specify a role of the gut–brain axis in the physiopathology of clinical depression [46]. It is highly plausible that the synergism in vitamin D and probiotics’ anti-inflammatory, antioxidant, and immunomodulatory effects might augment their impact on mental health. This is yet to be confirmed by future interventional human studies.
The only study in this review that reported null results with the co-supplementation was a trial by Tazzyman et al. [26] which did not show any improvement in the symptoms of patient with IBS whose vitamin D was repleted. This study had a limited sample size (underpowered trial), and a limited duration of follow-up. Additionally, in that study, the group receiving placebo showed an improvement in vitamin D levels, which might be due to seasonal differences in sun exposure, and a placebo effect was observed on symptom scores. The authors speculated that increased sunlight exposure had increased vitamin D levels which in turn improved IBS symptoms. All of these limitations may have prevented the authors from detecting a significant difference in symptom scores between the placebo and supplemented groups. Additionally, individuals might need higher doses of vitamin D plus probiotic supplementation for a longer period of time to provide appropriate circulating levels for improving symptoms.
Understanding the mechanisms of the interplay between vitamin D and probiotics in modulating the gut microbiota and regulating host responses, and exploring the effectiveness of this form of supplementation in high-quality human studies are crucial before applying it to prevent and manage disease. Studies included in this review had revealed thoroughly the superiority of co-supplementing with vitamin D and probiotics. Vitamin D has shown benefits in cellular restoration and reducing inflammation. The latter has been implicated in the pathophysiology of an unlimited number of conditions and diseases. VDR expression and transcriptional activity can be a research focus for future genetic studies. In parallel, data about probiotics and their role in optimizing microbiota and absorption pathways would be very useful not only for vitamin D but for many other nutrients or enzymes involved to boost immunity and host response.
5. Strengths and Limitations
To our knowledge, this is the first review to systematically compile human interventional evidence on the effectiveness of a combined supplementation of vitamin D and probiotics. Our review has numerous strengths [47]. It was conducted following standard methods for reporting systematic reviews [23], and according to a pre-defined protocol, which was published a priori. To increase the comprehensiveness of our search, we searched multiple scientific databases and the grey literature, and did not limit our search to any publication language or time. All the steps of study inclusion, data extraction and quality assessment were conducted in duplicate. We only included RCTs, and assessed their risk of bias using a validated tool; and, in general, the included studies were of high quality. However, included trials were limited in number, and conditions assessed. They were also limited by the small sample size, and short duration of follow-up. Moreover, only two studies [25,26] provided details regarding the strain of bacteria in the used probiotics. None of the studies provided analyses of the gut microbiota, disabling us from establishing whether the co-supplementation changed the composition of the microbiota, or ascertaining whether the observed changes were due to changes in the gut microbiota. Furthermore, we could not pool the studies in a meta-analysis due to the heterogeneity in the populations, conditions assessed, outcomes, doses and forms of vitamin D supplemented, and doses and strains of probiotics supplemented.
6. Conclusions
A combined supplementation with vitamin D and probiotics seems to play a role on the physiological and psychological attributes of the human body, and represents a novel insight in the management of chronic diseases. The findings of this systematic review suggest a superiority of vitamin D and probiotics supplementation over placebo, vitamin D or probiotics alone, and propose a dose-dependent effect. However, solid conclusions cannot be drawn at this level, and these findings remain certainly not robust enough and should be interpreted with caution. Future high-quality studies in other disease areas and various populations are needed to confirm these findings and to inform on the form, composition, and frequency of this co-supplementation for optimal outcomes.
Supplementary Materials
The following are available online at https://www.mdpi.com/2072-6643/13/1/111/s1, Table S1: Characteristics of included studies, Table S2: Outcomes and results of included studies.
Author Contributions
Conceptualization M.A. and R.R.; design M.A. and R.R.; methodology M.A. and R.R.; S.H. performed the searches; project administration F.A. and D.P.; funding acquisition S.H., R.R. and M.A. writing-review and editing S.H., R.R. and M.A.; writing-original draft N.M. All authors have read and agreed to the published version of the manuscript.
Funding
College of Natural and Health Sciences, Zayed University, Dubai, United Arab Emirates. The funding body will not be involved in the design of the study and collection, analysis, and interpretation of data or in writing the manuscript. Cluster grant R18030.
Institutional Review Board Statement
Not applicable to this review.
Informed Consent Statement
Not applicable to this review.
Acknowledgments
We would like to thank Aida Farha for her assistance in developing the search strategy.
Conflicts of Interest
The authors declare that they have no competing interests.
References
- Quigley, E.M.M. Microbiota-Brain-Gut Axis and Neurodegenerative Diseases. Curr. Neurol. Neurosci. Rep. 2017, 17, 94. [Google Scholar] [CrossRef]
- Ogbu, D.; Xia, E.; Sun, J. Gut instincts: Vitamin D/vitamin D receptor and microbiome in neurodevelopment disorders. Open Biol. 2020, 10, 200063. [Google Scholar] [CrossRef]
- Sun, J. Dietary Vitamin D, Vitamin D Receptor, and Microbiome. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 471–474. [Google Scholar] [CrossRef] [PubMed]
- Strandwitz, P. Neurotransmitter modulation by the gut microbiota. Brain Res. 2018, 1693, 128–133. [Google Scholar] [CrossRef] [PubMed]
- Butel, M.-J.; Waligora-Dupriet, A.-J.; Wydau-Dematteis, S. The developing gut microbiota and its consequences for health. J. Dev. Orig. Health Dis. 2018, 9, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Bakke, D.; Chatterjee, I.; Agrawal, A.; Dai, Y.; Sun, J. Regulation of Microbiota by Vitamin D Receptor: A Nuclear Weapon in Metabolic Diseases. Nucl. Recept. Res. 2018, 5. [Google Scholar] [CrossRef] [PubMed]
- Hawrelak, J.A.; Myers, S.P. The Causes of Intestinal Dysbiosis: A Review. Altern. Med. Rev. 2004, 9, 180–197. [Google Scholar]
- Battistini, C.; Nassani, N.; Saad, S.M.; Sun, J. Probiotics, Vitamin D, and Vitamin D Receptor in Health and Disease. In Lactic Acid Bacteria; Cavalcanti de Albuquerque, M.A., de Moreno de LeBlanc, A., LeBlanc, J.G., Bedani, R., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 93–105. ISBN 978-0-429-42259-1. [Google Scholar]
- Shang, M.; Sun, J. Vitamin D/VDR, Probiotics, and Gastrointestinal Diseases. Curr. Med. Chem. 2017, 24, 876–887. [Google Scholar] [CrossRef]
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: A worldwide problem with health consequences. Am. J. Clin. Nutr. 2008, 87, 1080S–1086S. [Google Scholar] [CrossRef]
- Trehan, N.; Afonso, L.; Levine, D.L.; Levy, P.D. Vitamin D Deficiency, Supplementation, and Cardiovascular Health. Crit. Pathw. Cardiol. 2017, 16, 109–118. [Google Scholar] [CrossRef]
- AlAnouti, F.; Abboud, M.; Papandreou, D.; Mahboub, N.; Haidar, S.; Rizk, R. Effects of Vitamin D Supplementation on Lipid Profile in Adults with the Metabolic Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2020, 12, 3352. [Google Scholar] [CrossRef] [PubMed]
- Abboud, M. Vitamin D Supplementation and Blood Pressure in Children and Adolescents: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 1163. [Google Scholar] [CrossRef] [PubMed]
- Spedding, S. Vitamin D and Depression: A Systematic Review and Meta-Analysis Comparing Studies with and without Biological Flaws. Nutrients 2014, 6, 1501–1518. [Google Scholar] [CrossRef] [PubMed]
- Zmijewski, M.A. Vitamin D and human health. Int. J. Mol. Sci. 2019, 20, 145. [Google Scholar] [CrossRef]
- Batacchi, Z.; Robinson-Cohen, C.; Hoofnagle, A.N.; Isakova, T.; Kestenbaum, B.; Martin, K.J.; Wolf, M.S.; De Boer, I.H. Effects of vitamin D2 supplementation on vitamin D3 metabolism in health and CKD. Clin. J. Am. Soc. Nephrol. 2017, 7, 1498–1506. [Google Scholar] [CrossRef]
- Sivamaruthi, B.S.; Kesika, P.; Suganthy, N.; Chaiyasut, C. A Review on Role of Microbiome in Obesity and Antiobesity Properties of Probiotic Supplements. BioMed Res. Int. 2019, 1–20. [Google Scholar] [CrossRef]
- Isolauri, E. Probiotics in the Development and Treatment of Allergic Disease. Gastroenterol. Clin. 2012, 41, 747–762. [Google Scholar] [CrossRef]
- Tomaro-Duchesneau, C.; Saha, S.; Malhotra, M.; Jones, M.L.; Labbé, A.; Rodes, L.; Kahouli, I.; Prakash, S. Effect of orally administered L. fermentum NCIMB 5221 on markers of metabolic syndrome: An in vivo analysis using ZDF rats. Appl. Microbiol. Biotechnol. 2014, 98, 115–126. [Google Scholar] [CrossRef]
- Varankovich, N.V.; Nickerson, M.T.; Korber, D.R. Probiotic-based strategies for therapeutic and prophylactic use against multiple gastrointestinal diseases. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- Mohammadi-Sartang, M.; Bellissimo, N.; Mazloomi, S.M.; Fararouie, M.; Bedeltavana, A.; Famouri, M.; Mazloom, Z. The effect of daily fortified yogurt consumption on weight loss in adults with metabolic syndrome: A 10-week randomized controlled trial. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 565–574. [Google Scholar] [CrossRef]
- Shang, M.; Sun, J. Vitamin D, VDR, and probiotics in health and disease: A mini review. CAB Rev. 2017, 24, 876–887. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: New York, NY, USA, 2019. [Google Scholar]
- Savino, F.; Ceratto, S.; Poggi, E.; Cartosio, M.E.; Cordero di Montezemolo, L.; Giannattasio, A. Preventive effects of oral probiotic on infantile colic: A prospective, randomised, blinded, controlled trial using Lactobacillus reuteri DSM 17938. Benef. Microbes 2015, 6, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Tazzyman, S.; Richards, N.; Trueman, A.R.; Evans, A.L.; Grant, V.A.; Garaiova, I.; Plummer, S.F.; Williams, E.A.; Corfe, B.M. Vitamin D associates with improved quality of life in participants with irritable bowel syndrome: Outcomes from a pilot trial. BMJ Open Gastroenterol. 2015, 2, e000052. [Google Scholar] [CrossRef] [PubMed]
- Ghaderi, A.; Banafshe, H.R.; Mirhosseini, N.; Moradi, M.; Karimi, M.-A.; Mehrzad, F.; Bahmani, F.; Asemi, Z. Clinical and metabolic response to vitamin D plus probiotic in schizophrenia patients. BMC Psychiatry 2019, 19, 77. [Google Scholar] [CrossRef] [PubMed]
- Ostadmohammadi, V. Vitamin D and probiotic co-supplementation affects mental health, hormonal, inflammatory and oxidative stress parameters in women with polycystic ovary syndrome. J. Ovarian. Res. 2019, 12. [Google Scholar] [CrossRef]
- Jamilian, M.; Amirani, E.; Asemi, Z. The effects of vitamin D and probiotic co-supplementation on glucose homeostasis, inflammation, oxidative stress and pregnancy outcomes in gestational diabetes: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. 2019, 38, 2098–2105. [Google Scholar] [CrossRef]
- Raygan, F. The effects of vitamin D and probiotic co-supplementation on mental health parameters and metabolic status in type 2 diabetic patients with coronary heart disease: A randomized, double-blind, placebo-controlled trial. Prog. Neuropsychopharmacol. 2018, 8, 50–55. [Google Scholar] [CrossRef]
- Jafarnejad, S.; Djafarian, K.; Fazeli, M.R.; Yekaninejad, M.S.; Rostamian, A.; Keshavarz, S.A. Effects of a Multispecies Probiotic Supplement on Bone Health in Osteopenic Postmenopausal Women: A Randomized, Double-blind, Controlled Trial. J. Am. Coll. Nutr. 2017, 36, 497–506. [Google Scholar] [CrossRef]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Schippa, S.; Conte, M.P. Dysbiotic Events in Gut Microbiota: Impact on Human Health. Nutrients 2014, 6, 5786–5805. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.Y.; Inohara, N.; Nuñez, G. Mechanisms of inflammation-driven bacterial dysbiosis in the gut. Mucosal Immunol. 2017, 10, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Leyssens, C.; Verlinden, L.; De Hertogh, G.; Kato, S.; Gysemans, C.; Mathieu, C.; Carmeliet, G.; Verstuyf, A. Impact on Experimental Colitis of Vitamin D Receptor Deletion in Intestinal Epithelial or Myeloid Cells. Endocrinology 2017, 158, 2354–2366. [Google Scholar] [CrossRef] [PubMed]
- Barragan, M.; Good, M.; Kolls, J. Regulation of Dendritic Cell Function by Vitamin D. Nutrients 2015, 7, 8127–8151. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Thingholm, L.B.; Skiecevičienė, J.; Rausch, P.; Kummen, M.; Hov, J.R.; Degenhardt, F.; Heinsen, F.-A.; Rühlemann, M.C.; Szymczak, S.; et al. Genome-wide association analysis identifies variation in vitamin D receptor and other host factors influencing the gut microbiota. Nat. Genet. 2016, 48, 1396–1406. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zhang, Y.; Lu, R.; Xia, Y.; Zhou, D.; Petrof, E.O.; Claud, E.C.; Chen, D.; Chang, E.B.; Carmeliet, G.; et al. Intestinal epithelial vitamin D receptor deletion leads to defective autophagy in colitis. Gut 2015, 64, 1082–1094. [Google Scholar] [CrossRef]
- Yoon, S.S.; Sun, J. Probiotics, Nuclear Receptor Signaling, and Anti-Inflammatory Pathways. Gastroenterol. Res. Pract. 2011, 2011, 1–16. [Google Scholar] [CrossRef]
- Mencarelli, A.; Cipriani, S.; Renga, B.; Bruno, A.; D’Amore, C.; Distrutti, E.; Fiorucci, S. VSL#3 Resets Insulin Signaling and Protects against NASH and Atherosclerosis in a Model of Genetic Dyslipidemia and Intestinal Inflammation. PLoS ONE 2012, 7, e45425. [Google Scholar] [CrossRef]
- Fernandes de Abreu, D.A.; Eyles, D.; Féron, F. Vitamin D, a neuro-immunomodulator: Implications for neurodegenerative and autoimmune diseases. Psychoneuroendocrinology 2009, 34, S265–S277. [Google Scholar] [CrossRef]
- Bertone-Johnson, E.R. Vitamin D and the occurrence of depression: Causal association or circumstantial evidence? Nutr. Rev. 2009, 67, 481–492. [Google Scholar] [CrossRef]
- Humble, M.B. Vitamin D, light and mental health. J. Photochem. Photobiol. B 2010, 101, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Yano, J.M.; Yu, K.; Donaldson, G.P.; Shastri, G.G.; Ann, P.; Ma, L.; Nagler, C.R.; Ismagilov, R.F.; Mazmanian, S.K.; Hsiao, E.Y. Indigenous Bacteria from the Gut Microbiota Regulate Host Serotonin Biosynthesis. Cell 2015, 161, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Barrett, E.; Ross, R.P.; O’Toole, P.W.; Fitzgerald, G.F.; Stanton, C. γ-Aminobutyric acid production by culturable bacteria from the human intestine. J. Appl. Microbiol. 2012, 113, 411–417. [Google Scholar] [CrossRef]
- Schmidt, C. Mental health: Thinking from the gut. Nature 2015, 25, S12–S15. [Google Scholar] [CrossRef] [PubMed]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, j4008. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).


