Oxidative Stress in Rats is Modulated by Seasonal Consumption of Sweet Cherries from Different Geographical Origins: Local vs. Non-Local
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Fruit Material
2.3. Proximate Composition
2.4. Extraction and Quantification of the Phenolic Compounds
2.5. Experimental Procedure in Rats
2.6. Biochemical Analysis
2.6.1. Antioxidant Status
Oxygen Radical Absorbance Capacity
Reduced Glutathione Concentration
2.6.2. Oxidative Stress Biomarkers
Alanine Aminotransferase and Aspartate Aminotransferase Activity
Malondialdehyde Production
ROS Generation
2.7. Statistical Analysis
3. Results
3.1. Proximate Composition of Local and Non-Local Brooks Sweet Cherry
3.2. Phenolic Profiles of Local and Non-Local Brooks Sweet Cherry
3.3. Body Composition and Feeding Test
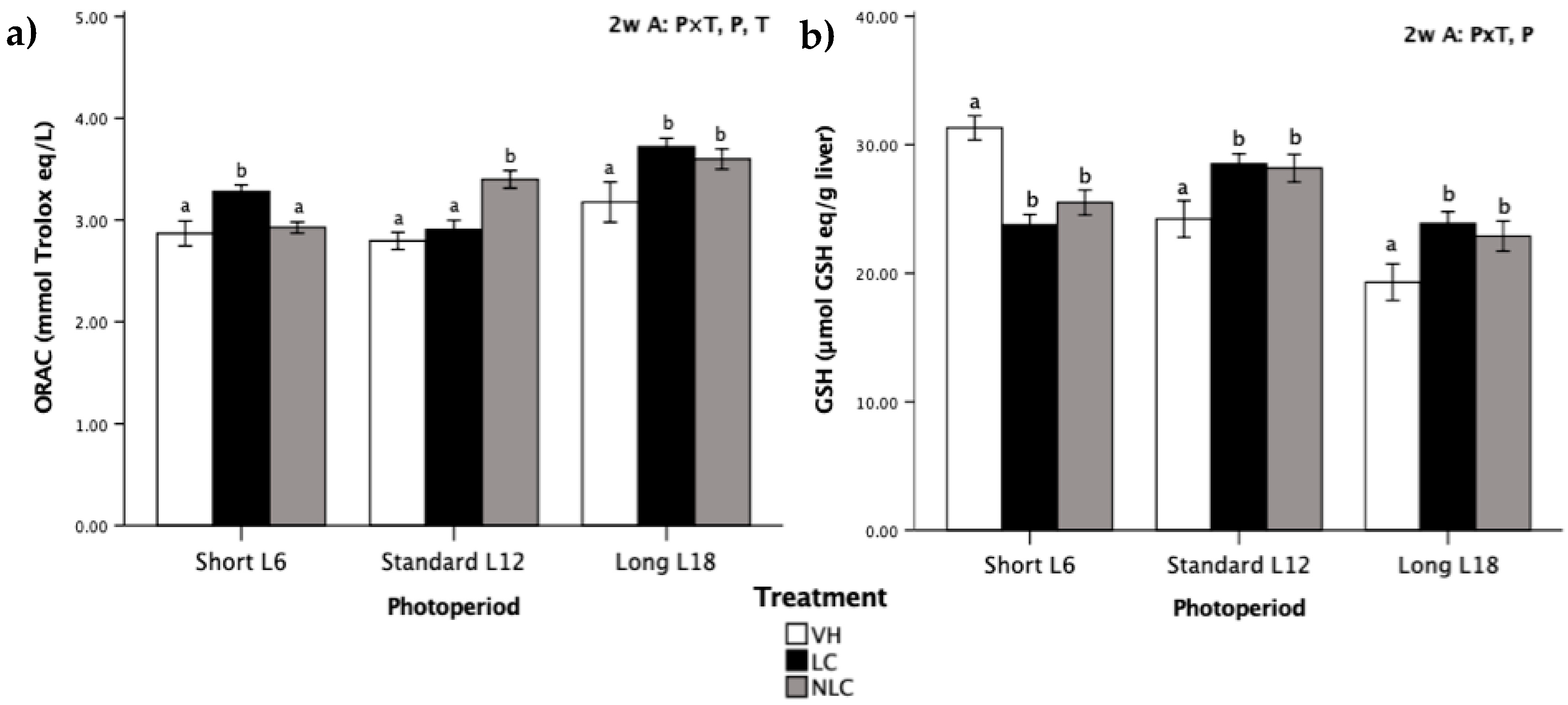
3.4. Antioxidant Status
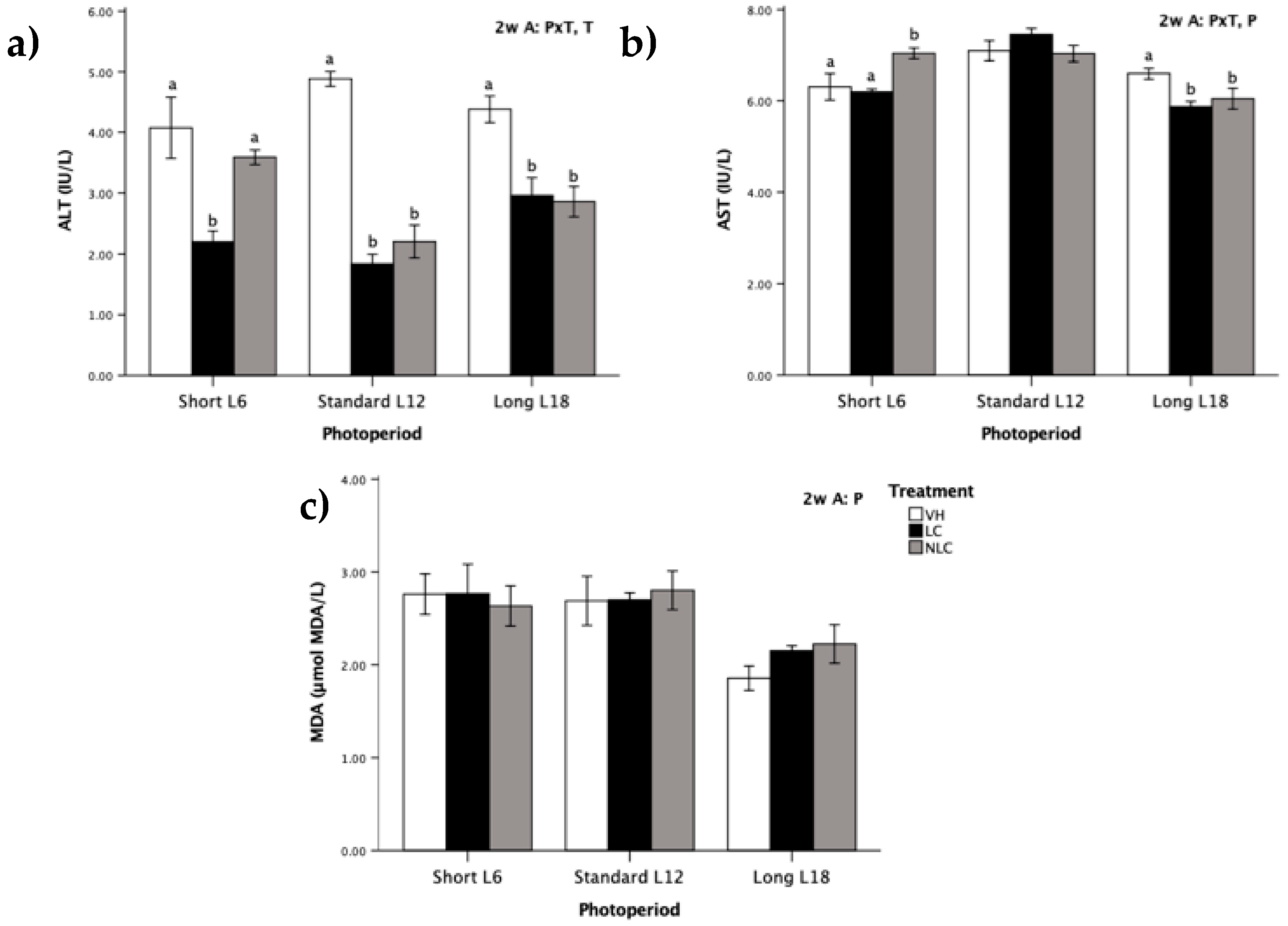
3.5. Serum Oxidative Stress Biomarkers
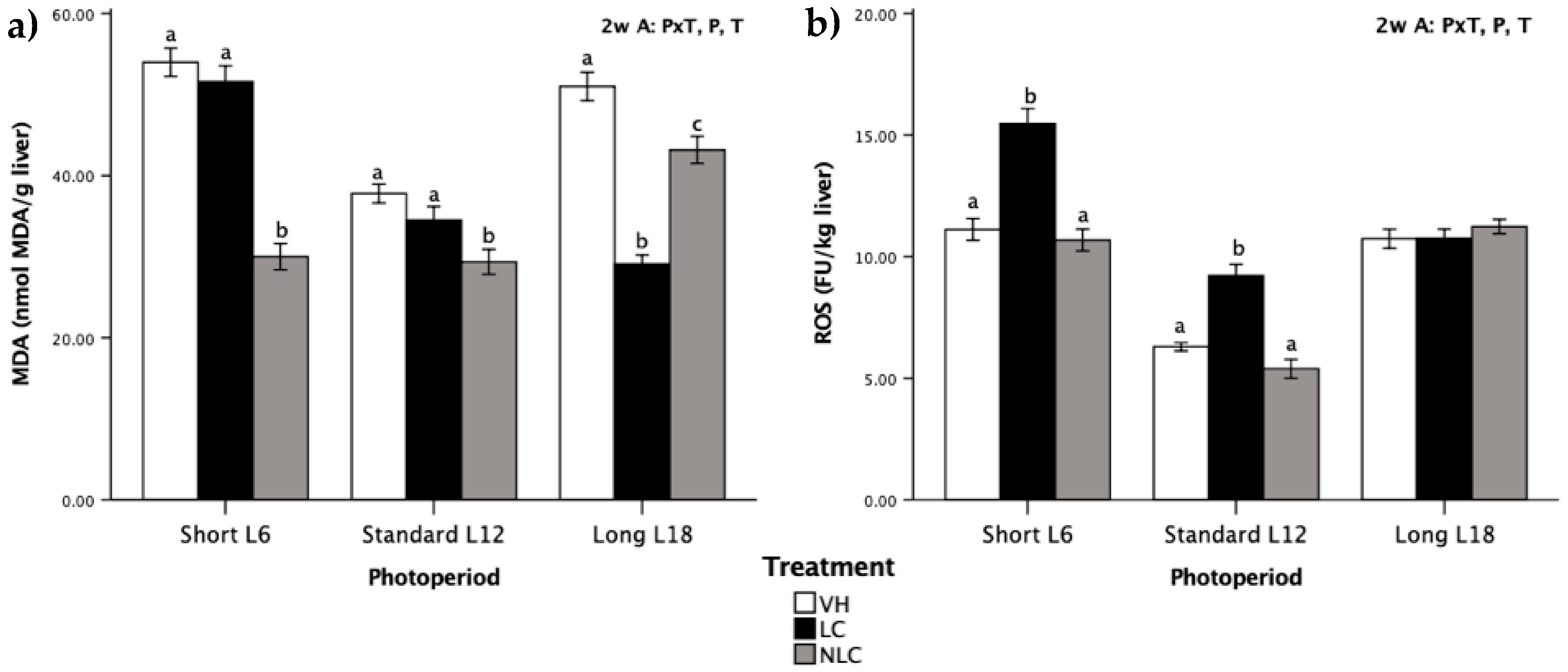
3.6. Liver Oxidative Stress Biomarkers
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oda, H.; Kato, H.; Seki, T. (Eds.) Health Nutrition: Nutritional Physiology as Health Science; Kyoritsu Shuppan: Tokyo, Japan, 2005; ISBN 9784320061538. (In Japanese) [Google Scholar]
- Hooper, P.L.; Hooper, P.L.; Tytell, M.; Vígh, L. Xenohormesis: Health benefits from an eon of plant stress response evolution. Cell Stress Chaperones 2010, 15, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Tahara, Y.; Shibata, S. Chrono-biology, chrono-pharmacology, and chrono-nutrition. J. Pharmacol. Sci. 2014, 124, 320–335. [Google Scholar] [CrossRef] [PubMed]
- Mariné-Casadó, R.; Domenech-Coca, C.; Del Bas, J.M.; Bladé, C.; Arola, L.; Caimari, A. The Exposure to Different Photoperiods Strongly Modulates the Glucose and Lipid Metabolisms of Normoweight Fischer 344 Rats. Front. Physiol. 2018, 9, 416. [Google Scholar] [CrossRef] [PubMed]
- European Commission ECHI European Core Health Indicators. Available online: http://ec.europa.eu/health/dyna/echi/datatool/index.cfm?indlist=49 (accessed on 10 April 2020).
- Blando, F.; Oomah, B.D. Sweet and sour cherries: Origin, distribution, nutritional composition and health benefits. Trends Food Sci. Technol. 2019, 86, 517–529. [Google Scholar] [CrossRef]
- Kelley, D.S.; Adkins, Y.; Laugero, K.D. A review of the health benefits of cherries. Nutrients 2018, 10, 368. [Google Scholar] [CrossRef]
- Chockchaisawasdee, S.; Golding, J.B.; Vuong, Q.V.; Papoutsis, K.; Stathopoulos, C.E. Sweet cherry: Composition, postharvest preservation, processing and trends for its future use. Trends Food Sci. Technol. 2016, 55, 72–83. [Google Scholar] [CrossRef]
- Serradilla, M.J.; Hernández, A.; López-Corrales, M.; Ruiz-Moyano, S.; de Guía Córdoba, M.; Martín, A. Composition of the Cherry (Prunus avium L. and Prunus cerasus L.; Rosaceae). In Nutritional Composition of Fruit Cultivars; Elsevier Inc.: Amsterdam, The Netherlands, 2015; pp. 127–147. ISBN 9780124081178. [Google Scholar]
- Escribano, B.M.; Moreno, A.; Tasset, I.; Túnez, I. Impact of light/dark cycle patterns on oxidative stress in an adriamycin-induced nephropathy model in rats. PLoS ONE 2014, 9, 1–10. [Google Scholar] [CrossRef]
- Arola-Arnal, A.; Cruz-Carrión, Á.; Torres-Fuentes, C.; Ávila-Román, J.; Aragonès, G.; Mulero, M.; Bravo, F.I.; Muguerza, B.; Arola, L.; Suárez, M. Chrononutrition and polyphenols: Roles and diseases. Nutrients 2019, 11, 2602. [Google Scholar] [CrossRef]
- Luisa Fanjul-Moles, M.; Durán-Lizarraga, M.E.; Gonsebatt, M.E.; Prieto-Sagredo, J. The Effect of Photoperiod and Light Irradiance on the Antioxidant Circadian System of Two Species of Crayfish from Different Latitudes: Procambarus clarkii and P. digueti. Photochem. Photobiol. 2003, 77, 210. [Google Scholar] [CrossRef]
- Bernabucci, U.; Ronchi, B.; Lacetera, N.; Nardone, A. Markers of oxidative status in plasma and erythrocytes of transition dairy cows during hot season. J. Dairy Sci. 2002, 85, 2173–2179. [Google Scholar] [CrossRef]
- Alila-Johansson, A.; Eriksson, L.; Soveri, T.; Laakso, M.L. Serum cortisol levels in goats exhibit seasonal but not daily rhythmicity. Chronobiol. Int. 2003, 20, 65–79. [Google Scholar] [CrossRef]
- Crisosto, C.H.; Crisosto, G.M.; Ritenour, M.A. Testing the reliability of skin color as an indicator of quality for early season “Brooks” (Prunus avium L.) cherry. Postharvest Biol. Technol. 2002, 24, 147–154. [Google Scholar] [CrossRef]
- AOAC International. Official Methods of Analysis, 15th ed.; Helrich, K., Ed.; Association of Official Analytical Chemists, Inc.: Arlington, VA, USA, 1990; Volume 1. [Google Scholar]
- Iglesias-Carres, L.; Mas-Capdevila, A.; Bravo, F.I.; Mulero, M.; Muguerza, B.; Arola-Arnal, A. Optimization and characterization of Royal Dawn cherry (Prunus avium) phenolics extraction. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Iglesias-Carres, L.; Mas-Capdevila, A.; Bravo, F.I.; Bladé, C.; Arola-Arnal, A.; Muguerza, B. Optimization of extraction methods for characterization of phenolic compounds in apricot fruit (Prunus armeniaca). Food Funct. 2019, 10, 6492–6502. [Google Scholar] [CrossRef] [PubMed]
- Iglesias-Carres, L.; Mas-Capdevila, A.; Bravo, F.I.; Arola, L.; Muguerza, B.; Arola-Arnal, A. Exposure of Fischer 344 rats to distinct photoperiods influences the bioavailability of red grape polyphenols. J. Photochem. Photobiol. B Biol. 2019, 199, 111623. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.A.; Prior, R.L. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 2002, 50, 4437–4444. [Google Scholar] [CrossRef] [PubMed]
- Kamencic, H.; Lyon, A.; Paterson, P.G.; Juurlink, B.H.J. Monochlorobimane fluorometric method to measure tissue glutathione. Anal. Biochem. 2000, 286, 35–37. [Google Scholar] [CrossRef]
- Pons, Z.; Guerrero, L.; Margalef, M.; Arola, L.; Arola-Arnal, A.; Muguerza, B. Effect of low molecular grape seed proanthocyanidins on blood pressure and lipid homeostasis in cafeteria diet-fed rats. J. Physiol. Biochem. 2014, 70, 629–637. [Google Scholar] [CrossRef]
- Gabbia, D.; Pozzo, L.; Zigiotto, G.; Roverso, M.; Sacchi, D.; Pozza, A.D.; Carrara, M.; Bogialli, S.; Floreani, A.; Guido, M.; et al. Dexamethasone counteracts hepatic inflammation and oxidative stress in cholestatic rats via CAR activation. PLoS ONE 2018, 13, 1–21. [Google Scholar] [CrossRef]
- Wei, H.; Cai, W.J.; Liu, H.K.; Han, D.; Zhu, X.M.; Yang, Y.X.; Jin, J.Y.; Xie, S.Q. Effects of photoperiod on growth, lipid metabolism and oxidative stress of juvenile gibel carp (Carassius auratus). J. Photochem. Photobiol. B Biol. 2019, 198, 111552. [Google Scholar] [CrossRef]
- Ponce, I.T.; Rezza, I.G.; Delgado, S.M.; Navigatore, L.S.; Bonomi, M.R.; Golini, R.L.; Gimenez, M.S.; Anzulovich, A.C. Daily oscillation of glutathione redox cycle is dampened in the nutritional vitamin A deficiency. Biol. Rhythm Res. 2012, 43, 351–372. [Google Scholar] [CrossRef] [PubMed]
- Morera-Fumero, A.L.; Abreu-Gonzalez, P.; Henry-Benitez, M.; Fernandez-Lopez, L.; Diaz-Mesa, E.; del Rosario Cejas-Mendez, M.; Guillen-Pino, F. Day/Night and Summer/Winter Changes in Serum Total Antioxidant Capacity. Med. Chem. 2018, 14, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Ho, E.; Karimi Galougahi, K.; Liu, C.-C.; Bhindi, R.; Figtree, G.A. Biological markers of oxidative stress: Applications to cardiovascular research and practice. Redox Biol. 2013, 1, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Baydas, G.; Gursu, M.F.; Yilmaz, S.; Canpolat, S.; Yasar, A.; Cikim, G.; Canatan, H. Daily rhythm of glutathione peroxidase activity, lipid peroxidation and glutathione levels in tissues of pinealectomized rats. Neurosci. Lett. 2002, 323, 195–198. [Google Scholar] [CrossRef]
- Coto-Montes, A.; Hardeland, R. Diurnal rhythm of lipid peroxidation in Drosophila melanogaster. In Studies on Antioxidants and Their Metabolites; Hardeland, R., Ed.; Cuvillier: Göttingen, Germany, 1999; pp. 110–113. [Google Scholar]
- Hardeland, R.; Hohmann, D.; Rensing, L. The rhythmic organization of rodent liver. A review. J. Interdiscipl. Cycle Res. 1973, 4, 89–118. [Google Scholar] [CrossRef]
- Çevik, C.; Aslan, R. Effects of photoperiod variations and alpha-lipoic acid treatment on melatonin, cortisol, and oxidative stress levels in the blood of rats. Turkish J. Biol. 2015, 39, 941–949. [Google Scholar] [CrossRef]
- Olayaki, L.; Abduraheem, T.; Mbukanma, O.; Agbede, O.; Salman, T.; Ojo, O. Short day photoperiod protects against acetaminophen-induced heptotoxicity in rats. Niger. J. Biotechnol. 2015, 29, 56. [Google Scholar] [CrossRef]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Promoting Fruit and Vegetable Consumption around the World. Available online: https://www.who.int/dietphysicalactivity/fruit/en/ (accessed on 12 June 2020).
- Sies, H. Oxidative stress: Oxidants and antioxidants. Exp. Physiol. 1997, 82, 291–295. [Google Scholar] [CrossRef]
- Garrido, M.; Paredes, S.D.; Cubero, J.; Lozano, M.; Toribio-Delgado, A.F.; Muñoz, J.L.; Reiter, R.J.; Barriga, C.; Rodríguez, A.B. Jerte valley cherry-enriched diets improve nocturnal rest and increase 6-sulfatoxymelatonin and total antioxidant capacity in the urine of middle-aged and elderly humans. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2010, 65, 909–914. [Google Scholar] [CrossRef]
- Levers, K.; Dalton, R.; Galvan, E.; Goodenough, C.; O’Connor, A.; Simbo, S.; Barringer, N.; Mertens-Talcott, S.U.; Rasmussen, C.; Greenwood, M.; et al. Effects of powdered Montmorency tart cherry supplementation on an acute bout of intense lower body strength exercise in resistance trained males. J. Int. Soc. Sports Nutr. 2015, 12, 41. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; McHugh, M.P.; Hill, J.A.; Brouner, J.; Jewell, A.P.; Van Someren, K.A.; Shave, R.E.; Howatson, S.A. Influence of tart cherry juice on indices of recovery following marathon running. Scand. J. Med. Sci. Sports 2010, 20, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Levers, K.; Dalton, R.; Galvan, E.; O’Connor, A.; Goodenough, C.; Simbo, S.; Mertens-Talcott, S.U.; Rasmussen, C.; Greenwood, M.; Riechman, S.; et al. Effects of powdered Montmorency tart cherry supplementation on acute endurance exercise performance in aerobically trained individuals. J. Int. Soc. Sports Nutr. 2016, 13, 22. [Google Scholar] [CrossRef]
- Dziadek, K.; Kopeć, A.; Piątkowska, E. Intake of fruit and leaves of sweet cherry beneficially affects lipid metabolism, oxidative stress and inflammation in Wistar rats fed with high fat-cholesterol diet. J. Funct. Foods 2019, 57, 31–39. [Google Scholar] [CrossRef]
- Viljevac, M.; Dugalić, K.; Mihaljević, I.; Tomaš, V.; Vuković, D.; Zdunić, Z.; Puškar, B.; Jurković, Z. Season, location and cultivar influence on bioactive compounds of sour cherry fruits. Plant Soil Environ. 2017, 63, 389–395. [Google Scholar] [CrossRef]
- Rieger, G.; Müller, M.; Guttenberger, H.; Bucar, F. Influence of altitudinal variation on the content of phenolic compounds in wild populations of Calluna vulgaris, Sambucus nigra, and Vaccinium myrtillus. J. Agric. Food Chem. 2008, 56, 9080–9086. [Google Scholar] [CrossRef]
- Cedar Lake Ventures Inc. Comparison of the Average Weather in Tarragona and Santiago Weather Spark. Available online: https://weatherspark.com/compare/y/45958~26525/Comparison-of-the-Average-Weather-in-Tarragona-and-Santiago (accessed on 12 April 2020).
- Josuttis, M.; Carlen, C.; Crespo, P.; Nestby, R.; Toldam-Andersen, T.B.; Dietrich, H.; Krüger, E. A comparison of bioactive compounds of strawberry fruit from Europe affected by genotype and latitude. J. Berry Res. 2012, 2, 73–95. [Google Scholar] [CrossRef]
- Saura-Calixto, F. Dietary fiber as a carrier of dietary antioxidants: An essential physiological function. J. Agric. Food Chem. 2011, 59, 43–49. [Google Scholar] [CrossRef]
- Mariné-Casadó, R.; Domenech-Coca, C.; del Bas, J.M.; Bladé, C.; Caimari, A.; Arola, L. Cherry consumption out of season alters lipid and glucose homeostasis in normoweight and cafeteria-fed obese Fischer 344 rats. J. Nutr. Biochem. 2019, 63, 72–86. [Google Scholar] [CrossRef]
- Gibert-Ramos, A.; Crescenti, A.; Salvadó, M.J. Consumption of cherry out of season changes white adipose tissue gene expression and morphology to a phenotype prone to fat accumulation. Nutrients 2018, 10, 1102. [Google Scholar] [CrossRef]
- Ibars, M.; Aragonès, G.; Ardid-Ruiz, A.; Gibert-Ramos, A.; Arola-Arnal, A.; Suárez, M.; Bladé, C. Seasonal consumption of polyphenol-rich fruits affects the hypothalamic leptin signaling system in a photoperiod-dependent mode. Sci. Rep. 2018, 8, 13572. [Google Scholar] [CrossRef] [PubMed]
| Nutrients | LC | NLC | p Value |
|---|---|---|---|
| Ash | 2.05 ± 0.15 | 2.11 ± 0.35 | 0.81 |
| Protein | 6.41 ± 0.11 | 5.51± 0.07 | 0.01 |
| Total lipids (fat) | 1.01 ± 0.01 | 0.70 ± 0.07 | 0.03 |
| Fiber, total dietary | 10.95 ± 0.03 | 10.74 ± 0.06 | 0.04 |
| Total carbohydrates, by difference | 79.50 ± 0.18 | 80.78 ± 0.34 | 0.04 |
| Sugars, total | 43.30 ± 2.31 | 50.08 ± 0.68 | 0.02 |
| Phenolic Compounds | LC | NLC | p Value |
|---|---|---|---|
| TPC (mg GA eq/g dw) | 8.17 ± 0.20 | 7.64 ± 0.41 | 0.03 |
| TAC (mg Cy3R eq/g dw) | 1.31 ± 0.02 | 1.23 ± 0.03 | 0.02 |
| TFaC (mg Cat eq/g dw) | 0.44 ± 0.02 | 0.38 ± 0.03 | 0.02 |
| TFoC (mg Quer eq/g dw) | 0.55 ± 0.00 | 0.63 ± 0.05 | 0.04 |
| Short L6 | Standard L12 | Long L18 | 2wA | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LC | NLC | VH | LC | NLC | VH | LC | NLC | VH | ||
| Biometric parameters | ||||||||||
| Body weight gain (g) | 57.1 ± 5.14 | 52.9 ± 3.15 | 44.6 ± 4.12 | 55.3 ± 1.24 | 52.9 ± 3.30 | 54.0 ± 3.50 | 50.0 ± 1.55 | 53.5 ± 4.00 | 51.5 ± 0.72 | n.s. |
| Skeletal muscle (g) | 3.64 ± 0.28 | 3.92 ± 0.16 | 4.13 ± 0.10 | 4.40 ± 0.07 | 4.18 ± 0.09 | 4.30 ± 0.10 | 4.24 ± 0.12 | 4.33 ± 0.15 | 4.43 ± 0.07 | P |
| BAT (g) | 0.45 ± 0.03 | 0.45 ± 0.05 | 0.40 ± 0.06 | 0.47 ± 0.03 | 0.39 ± 0.01 | 0.39 ± 0.03 | 0.34 ± 0.03 | 0.36 ± 0.05 | 0.44 ± 0.03 | n.s. |
| EWAT (g) | 8.61 ± 0.23 | 8.73 ± 0.45 | 9.01 ± 0.44 | 10.10 ± 0.93 | 8.94 ± 0.50 | 9.49 ± 0.47 | 9.20 ± 0.58 | 9.21 ± 0.57 | 9.88 ± 0.74 | n.s. |
| IWAT (g) | 2.87 ± 0.36 | 2.46 ± 0.21 | 2.83 ± 0.37 | 2.80 ± 0.21 | 2.50 ± 0.27 | 2.78 ± 0.19 | 2.52 ± 0.27 | 2.42 ± 0.22 * | 3.35 ± 0.37 | n.s. |
| MWAT (g) | 4.69 ± 0.26 | 4.64 ± 0.27 | 5.56 ± 0.50 | 6.05 ± 0.36 | 5.22 ± 0.38 | 5.52 ± 0.30 | 5.39 ± 0.37 | 4.51 ± 0.24 | 5.12 ± 0.30 | P |
| Body fat (%) | 4.30 ± 0.19 | 4.43 ± 0.11 | 4.37 ± 0.16 | 4.68 ± 0.29 | 4.71 ± 0.20 | 4.78 ± 0.20 | 4.74 ± 0.23 | 5.11 ± 0.31 | 4.62 ± 0.16 | P × T, P |
| Feeding tests | ||||||||||
| Food intake (kcal/day) | 56.9 ± 1.23 | 55.1 ± 1.62 | 54.1 ± 0.29 | 56.2 ± 1.13 | 53.8 ± 1.06 | 55.4 ± 1.15 | 54.0 ± 0.40 | 55.1 ± 1.00 | 54.9 ± 0.47 | n.s. |
| FE (g/100 kcal) | 2.76 ± 0.16 * | 2.64 ± 0.14 * | 2.16 ± 0.14 | 2.50 ± 0.04 | 2.54 ± 0.14 | 2.46 ± 0.18 | 2.64 ± 0.11 | 2.89 ± 0.16 | 2.75 ± 0.20 | n.s. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz-Carrión, Á.; Ruiz de Azua, M.J.; Mulero, M.; Arola-Arnal, A.; Suárez, M. Oxidative Stress in Rats is Modulated by Seasonal Consumption of Sweet Cherries from Different Geographical Origins: Local vs. Non-Local. Nutrients 2020, 12, 2854. https://doi.org/10.3390/nu12092854
Cruz-Carrión Á, Ruiz de Azua MJ, Mulero M, Arola-Arnal A, Suárez M. Oxidative Stress in Rats is Modulated by Seasonal Consumption of Sweet Cherries from Different Geographical Origins: Local vs. Non-Local. Nutrients. 2020; 12(9):2854. https://doi.org/10.3390/nu12092854
Chicago/Turabian StyleCruz-Carrión, Álvaro, Ma. Josefina Ruiz de Azua, Miquel Mulero, Anna Arola-Arnal, and Manuel Suárez. 2020. "Oxidative Stress in Rats is Modulated by Seasonal Consumption of Sweet Cherries from Different Geographical Origins: Local vs. Non-Local" Nutrients 12, no. 9: 2854. https://doi.org/10.3390/nu12092854
APA StyleCruz-Carrión, Á., Ruiz de Azua, M. J., Mulero, M., Arola-Arnal, A., & Suárez, M. (2020). Oxidative Stress in Rats is Modulated by Seasonal Consumption of Sweet Cherries from Different Geographical Origins: Local vs. Non-Local. Nutrients, 12(9), 2854. https://doi.org/10.3390/nu12092854








