Risk Reduction for End-Stage Renal Disease by Dietary Guidance Using the Gustatory Threshold Test for Salty Taste
Abstract
1. Introduction
2. Materials and Methods
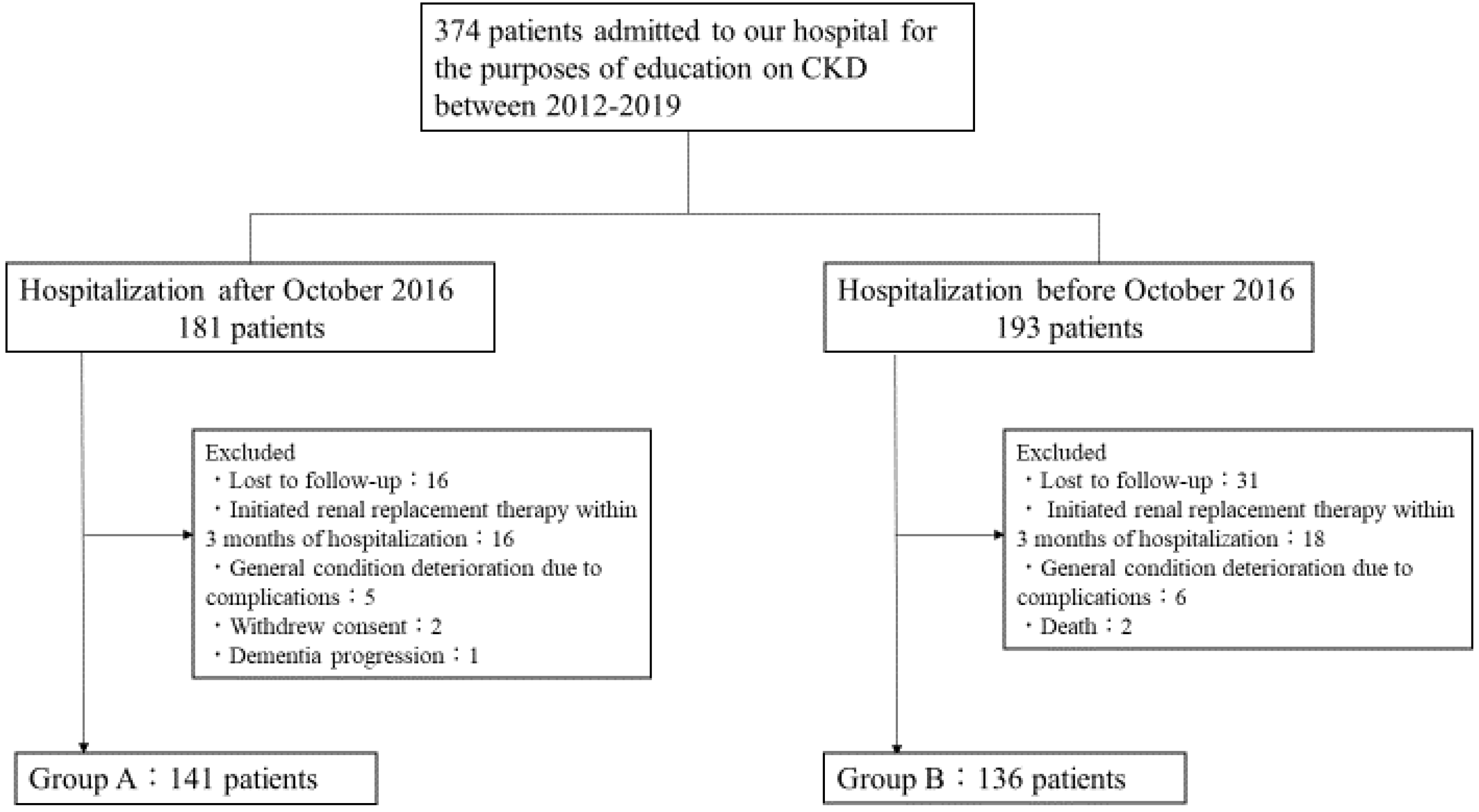
2.1. Subjects and Study Design
2.2. Assessments
2.2.1. Estimated Glomerular Filtration Rate: eGFR
2.2.2. Gustatory Threshold for Salty Taste
(mg/dL) × 10) × (−2.04 × age + 14.89 × body weight (kg) + 16.14 × height (cm) −
2244.45)}0.392
2.3. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bikbov, B.; Purcell, C.A.; Levey, A.S.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.M.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef]
- He, F.J.; Tan, M.; Ma, Y.; MacGregor, G.A. Salt Reduction to Prevent Hypertension and Cardiovascular Disease: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2020, 75, 632–647. [Google Scholar] [CrossRef]
- Saran, R.; Padilla, R.L.; Gillespie, B.W.; Heung, M.; Hummel, S.L.; Derebail, V.K.; Pitt, B.; Levin, N.W.; Zhu, F.; Abbas, S.R.; et al. A randomized crossover trial of dietary sodium restriction in stage 3–4 CKD. Clin. J. Am. Soc. Nephrol. 2017, 12, 399–407. [Google Scholar] [CrossRef]
- Garofalo, C.; Borrelli, S.; Provenzano, M.; De Stefano, T.; Vita, C.; Chiodini, P.; Minutolo, R.; Nicola, L.; Conte, G. Dietary Salt Restriction in Chronic Kidney Disease: A Meta-Analysis of Randomized Clinical Trials. Nutrients 2018, 10, 732. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.A.M.; Nguyen, H.A. Nutrition education in the care of patients with chronic kidney disease and end-stage renal disease. Semin. Dial. 2018, 31, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Machida, S.; Shibagaki, Y.; Sakurada, T. An inpatient educational program for chronic kidney disease. Clin. Exp. Nephrol. 2019, 23, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Kusaba, T.; Mori, Y.; Masami, O.; Hiroko, N.; Adachi, T.; Sugishita, C.; Sonomura, K.; Kimura, T.; Kishimoto, N.; Nakagawa, H.; et al. Sodium restriction improves the gustatory threshold for salty taste in patients with chronic kidney disease. Kidney Int. 2009, 76, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Eckardt, K.U.; Tsukamoto, Y.; Levin, A.; Coresh, J.; Rossert, J.; De Zeeuw, D.; Hostetter, T.H.; Lameire, N.; Eknoyan, G.; et al. Definition and classification of chronic kidney disease: A position statement from Kidney Disease: Improving Global Outcomes (KDIGO)z. Kidney Int. 2005, 67, 2089–2100. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, S.; Imai, E.; Horio, M.; Yasuda, Y.; Tomita, K.; Nitta, K.; Yamagata, K.; Tomino, Y.; Yokoyama, H.; Hishida, A.; et al. Revised Equations for Estimated GFR From Serum Creatinine in Japan. Am. J. Kidney Dis. 2009, 53, 982–992. [Google Scholar] [CrossRef] [PubMed]
- Michikawa, T.; Nishiwaki, Y.; Okamura, T.; Asakura, K.; Nakano, M.; Takebayashi, T. The taste of salt measured by a simple test and blood pressure in Japanese women and men. Hypertens. Res. 2009, 32, 399–403. [Google Scholar] [CrossRef]
- Nishimoto, K.; Hirota, R.; Egawa, M.; Furuta, S. Clinical evaluation of taste dysfunction using a salt-impregnated taste strip. ORL 1996, 58, 258–261. [Google Scholar] [CrossRef]
- Tanaka, T.; Okamura, T.; Miura, K.; Kadowaki, T.; Ueshima, H.; Nakagawa, H. A simple method to estimate populational 24-h urinary sodium and potassium. J. Hum. Hypertens. 2002, 16, 97–103. [Google Scholar] [CrossRef]
- Ogura, M.; Kimura, A.; Takane, K.; Nakao, M.; Hamaguchi, A.; Terawaki, H.; Hosoya, T. Estimation of salt intake from spot urine samples in patients with chronic kidney disease. BMC Nephrol. 2012, 13, 36. [Google Scholar] [CrossRef] [PubMed]
- Imai, E.; Yasuda, Y.; Horio, M.; Shibata, K.; Kato, S.; Mizutani, Y.; Imai, J.; Hayashi, M.; Kamiya, H.; Oiso, Y.; et al. Validation of the equations for estimating daily sodium excretion from spot urine in patients with chronic kidney disease. Clin. Exp. Nephrol. 2011, 15, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Bayliss, E.A.; Bhardwaja, B.; Ross, C.; Beck, A.; Lanese, D.M. Multidisciplinary team care may slow the rate of decline in renal function. Clin. J. Am. Soc. Nephrol. 2011, 6, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.T.; Chen, J.; Yang, W.; Appel, L.J.; Kusek, J.W.; Alper, A.; Delafontaine, P.; Keane, M.G.; Mohler, E.; Ojo, A.; et al. Sodium excretion and the risk of cardiovascular disease in patients with chronic kidney disease. JAMA J. Am. Med. Assoc. 2016, 315, 2200–2210. [Google Scholar] [CrossRef] [PubMed]
- Campbell, K.L.; Johnson, D.W.; Bauer, J.D.; Hawley, C.M.; Isbel, N.M.; Stowasser, M.; Whitehead, J.P.; Dimeski, G.; McMahon, E. A randomized trial of sodium-restriction on kidney function, fluid volume and adipokines in CKD patients. BMC Nephrol. 2014, 15, 57. [Google Scholar] [CrossRef] [PubMed]
- Mazarova, A.; Molnar, A.O.; Akbari, A.; Sood, M.M.; Hiremath, S.; Burns, K.D.; Ramsay, T.O.; Mallick, R.; Knoll, G.A.; Ruzicka, M. The association of urinary sodium excretion and the need for renal replacement therapy in advanced chronic kidney disease: A cohort study. BMC Nephrol. 2016, 17, 123. [Google Scholar] [CrossRef] [PubMed]
- McQuarrie, E.P.; Traynor, J.P.; Taylor, A.H.; Freel, E.M.; Fox, J.G.; Jardine, A.G.; Mark, P.B. Association between urinary sodium, creatinine, albumin, and long-term survival in chronic kidney disease. Hypertension 2014, 64, 111–117. [Google Scholar] [CrossRef]
- O’Donnell, M.; Mente, A.; Rangarajan, S.; McQueen, M.J.; Wang, X.; Liu, L.; Yan, H.; Lee, S.F.; Mony, P.; Devanath, A.; et al. Urinary sodium and potassium excretion, mortality, and cardiovascular events. N. Engl. J. Med. 2014, 371, 612–623. [Google Scholar] [CrossRef]
- Yoon, C.; Noh, J.; Lee, J.; Kee, Y.K.; Seo, C.; Lee, M.; Cha, M.; Kim, H.; Park, S.; Yun, H.; et al. High and low sodium intakes are associated with incident chronic kidney disease in patients with normal renal function and hypertension. Kidney Int. 93, 921–931. [CrossRef] [PubMed]
- Kim, T.H.; Kim, Y.H.; Bae, N.Y.; Kang, S.S.; Lee, J.B.; Kim, S.B. Salty taste thresholds and preference in patients with chronic kidney disease according to disease stage: A cross-sectional study. Nutr. Diet. 2018, 75, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Fernström, A.; Hylander, B.; Rössner, S. Taste acuity in patients with chronic renal failure. Clin. Nephrol. 1996, 45, 169–174. [Google Scholar]
- Nomura, K.; Nakanishi, M.; Ishidate, F.; Iwata, K.; Taruno, A. All-Electrical Ca2+-Independent Signal Transduction Mediates Attractive Sodium Taste in Taste Buds. Neuron 2020. [Google Scholar] [CrossRef] [PubMed]
- Shigemura, N.; Iwata, S.; Yasumatsu, K.; Ohkuri, T.; Horio, N.; Sanematsu, K.; Yoshida, R.; Margolskee, R.F.; Ninomiya, Y. Angiotensin II modulates salty and sweet taste sensitivities. J. Neurosci. 2013, 33, 6267–6277. [Google Scholar] [CrossRef] [PubMed]

| All (277) | Group A (141) | Group B (136) | p-Value | |
|---|---|---|---|---|
| Age, years | 69.2 ± 12.2 | 67.7 ± 13.2 | 70.7 ± 10.8 | 0.045 |
| Male, % | 62.1% (172) | 64.5% (91) | 59.6% (81) | 0.46 |
| Height, cm | 160.0 ± 9.3 | 161.3 ± 61.3 | 158.6 ± 58.6 | 0.02 |
| Weight, kg | 60.2 ± 13.4 | 62.9 ± 14.3 | 57.5 ± 11.8 | <0.001 |
| BMI, kg/m2 | 23.4 ± 4.2 | 24.1 ± 4.8 | 22.7 ± 3.5 | 0.007 |
| CKD stage, % | G3: 18.0% (50) G4: 49.1% (136) G5: 32.9% (91) | G3: 24.1% (34) G4: 49.7% (70) G5: 26.2% (37) | G3: 11.8% (16) G4: 48.5% (66) G5: 39.7% (54) | |
| Primary disease, % | Diabetic nephropathy: 35.7% (99) Nephrosclerosis: 31.0% (86) Glomerulonephritis: 15.5% (43) ADPKD: 4.3% (12) One kidney: 4.3% (12) Others: 9.0% (25) | Diabetic nephropathy: 39.7% (56) Nephrosclerosis: 27.0% (38) Glomerulonephritis: 17.0% (24) ADPKD: 4.3% (6) One kidney: 5.7% (8) Others: 6.4% (9) | Diabetic nephropathy: 31.6% (43) Nephrosclerosis: 35.3% (48) Glomerulonephritis: 14.0% (19) ADPKD: 4.4% (6) One kidney: 2.9% (4) Others: 12.5% (16) | |
| Diabetes mellitus, % | 46.9% (130) | 51.1% (72) | 42.7% (58) | 0.23 |
| BUN, mg/dL | 39.7 ± 15.7 | 39.1 ± 15.8 | 40.3 ± 15.7 | 0.55 |
| Cr, mg/dL | 2.6 ± 1.1 | 2.6 ± 1.1 | 2.5 ± 1.1 | 0.51 |
| eGFR, mL/min/1.73 m2 | 20.8 ± 10.1 | 22.4 ± 10.3 | 19.1 ± 9.6 | 0.005 |
| Na, mmol/L | 139.8 ± 2.5 | 139.6 ± 2.3 | 140.2 ± 2.6 | 0.05 |
| K, mmol/L | 4.5 ± 0.5 | 4.4 ± 0.5 | 4.6 ± 0.5 | 0.06 |
| Ca, mg/dL | 9.1 ± 0.6 | 9.1 ± 0.6 | 9.0 ± 0.6 | 0.10 |
| P, mg/dL | 3.7 ± 0.8 | 3.7 ± 0.9 | 3.7 ± 0.8 | 0.99 |
| UA, mg/dL | 7.1 ± 1.6 | 7.1 ± 1.6 | 7.1 ± 1.7 | 0.87 |
| Intact-PTH a, pg/mL | 111 (67–177) | 104 (63.9–151) | 131 (71.7–217) | 0.004 |
| HCO3-, mmol/L | 23.1 ± 3.3 | 23.1 ± 3.4 | 23.1 ± 3.0 | 0.98 |
| Alb, g/dL | 3.8 ± 0.5 | 3.8 ± 0.5 | 3.7 ± 0.5 | 0.51 |
| LDL-C, mg/dL | 96.6 ± 30.1 | 96.8 ± 28.4 | 96.4 ± 32.3 | 0.91 |
| HbA1c, % | 6.1 ± 1.0 | 6.2 ± 1.1 | 6.1 ± 1.0 | 0.86 |
| Ferritin a, ng/mL | 133 (68–248) | 148 (82–273) | 111 (49–196) | 0.14 |
| Hb, g/dL | 11.4 ± 1.7 | 11.7 ± 1.8 | 11.1 ± 1.6 | 0.004 |
| UPCR a, g/gCr | 1.5 (0.4–3.3) | 1.5 (0.5–3.0) | 1.5 (0.3–3.5) | 0.58 |
| SBP, mmHg | 138 ± 21 | 138 ± 22 | 137 ± 20 | 0.72 |
| DBP, mmHg | 76 ± 13 | 77 ± 12 | 74 ± 13 | 0.06 |
| On Admission | 3 Months after Admission | p-Value | |
|---|---|---|---|
| Gustatory threshold for salty taste (mg/cm2) | 1.02 ± 0.35 | 0.85 ± 0.28 | <0.001 |
| Salt intake (g/day) | 8.5 ± 2.0 | 8.1 ± 2.5 | 0.04 |
| SBP, mmHg | 137 ± 21 | 133 ± 16 | 0.003 |
| DBP, mmHg | 76 ± 13 | 74 ± 11 | 0.02 |
| Group A (105) | Group B (105) | HR | p-Value | |
|---|---|---|---|---|
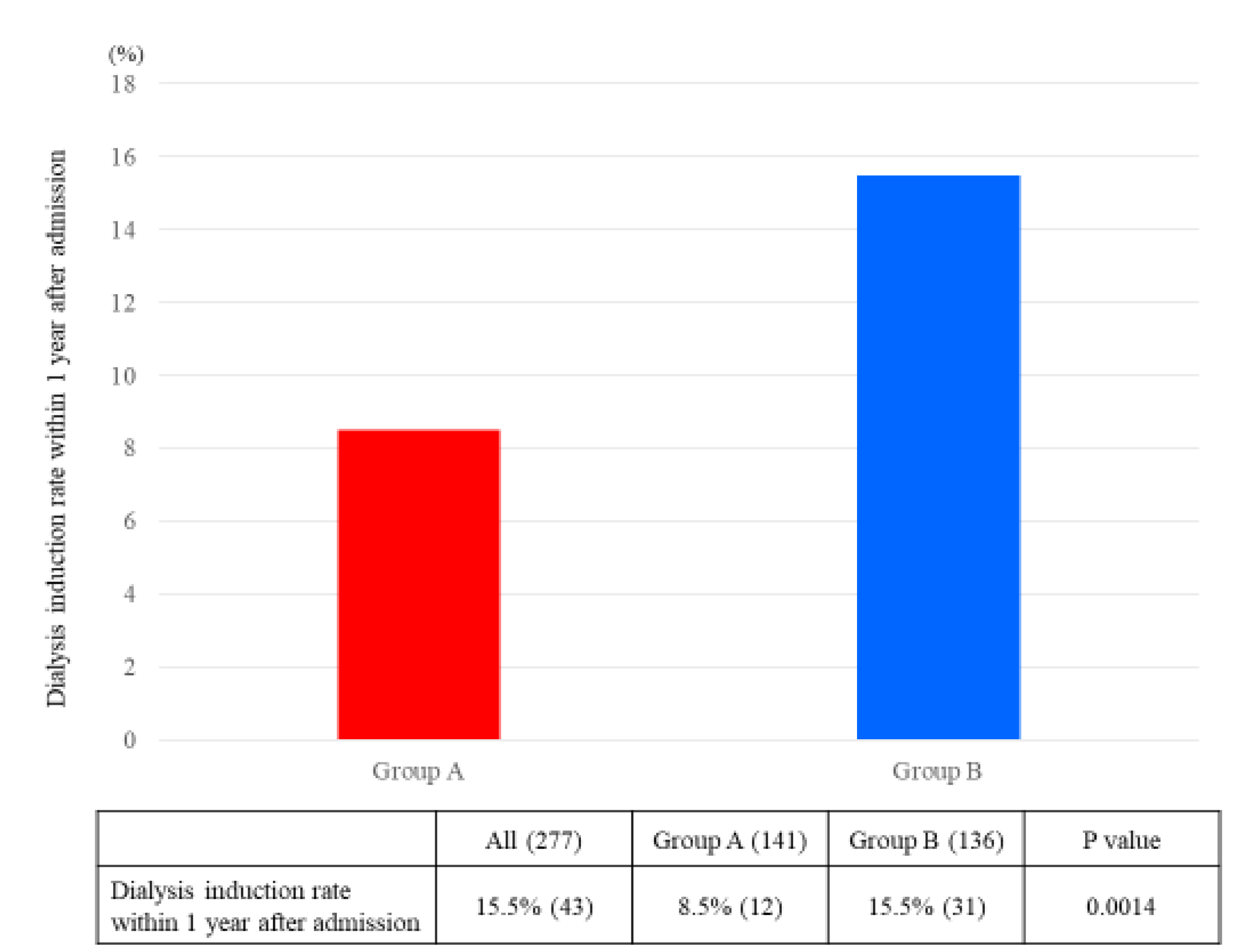
| Dialysis initiation rate, % | 9.5% (10) | 21.0% (22) | 0.40 (0.18–0.89) | 0.03 |
| ΔeGFR at admission and 6 months after admission (mL/min/1.73 m2/month) | 0.94 ± 1.79 | 0.49 ± 1.50 | 0.048 |
| Univariate | Multivariate (Model 1) | Multivariate (Model 2) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | HR | 95% CI | p-Value | |
| Age, year | 0.99 | 0.96–1.01 | 0.33 | 0.98 | 0.95–1.01 | 0.13 | 0.96 | 0.93–0.99 | 0.01 |
| Male, % | 0.65 | 0.34–1.26 | 0.21 | 0.66 | 0.34–1.30 | 0.24 | 1.15 | 0.53–2.48 | 0.72 |
| Enhanced salt reduction guidance | 0.32 | 0.15–0.64 | 0.001 | 0.30 | 0.14–0.61 | <0.001 | 0.35 | 0.16–0.81 | 0.01 |
| BMI, kg/m2 | 0.91 | 0.83–1.00 | 0.03 | 0.94 | 0.85–1.04 | 0.23 | |||
| eGFR, mL/min/1.73 m2 | 0.82 | 0.77–0.88 | <0.001 | 0.81 | 0.75–0.88 | <0.001 | |||
| Diabetes mellitus, % | 0.98 | 0.51–1.88 | 0.95 | ||||||
| K, mmol/L | 1.37 | 0.81–2.33 | 0.24 | ||||||
| Ca, mg/dL | 0.38 | 0.21–0.69 | 0.002 | ||||||
| P, mg/dL | 1.91 | 1.28–2.86 | 0.001 | ||||||
| UA, mg/dL | 1.03 | 0.83–1.26 | 0.80 | ||||||
| Alb, g/dL | 0.31 | 0.17–0.59 | <0.001 | ||||||
| LDL-C, mg/dL | 0.99 | 0.98–1.00 | 0.18 | ||||||
| HbA1c, % | 0.87 | 0.58–1.30 | 0.48 | ||||||
| Hb, g/dL | 0.61 | 0.49–0.76 | <0.001 | ||||||
| UPCR, g/gCr | 1.37 | 1.21–1.55 | <0.001 | ||||||
| SBP, mmHg | 1.03 | 1.01–1.05 | <0.001 | ||||||
| DBP, mmHg | 1.04 | 1.01–1.07 | 0.002 | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ota, Y.; Kitamura, M.; Tsuji, K.; Torigoe, K.; Yamashita, A.; Abe, S.; Muta, K.; Uramatsu, T.; Obata, Y.; Furutani, J.; et al. Risk Reduction for End-Stage Renal Disease by Dietary Guidance Using the Gustatory Threshold Test for Salty Taste. Nutrients 2020, 12, 2703. https://doi.org/10.3390/nu12092703
Ota Y, Kitamura M, Tsuji K, Torigoe K, Yamashita A, Abe S, Muta K, Uramatsu T, Obata Y, Furutani J, et al. Risk Reduction for End-Stage Renal Disease by Dietary Guidance Using the Gustatory Threshold Test for Salty Taste. Nutrients. 2020; 12(9):2703. https://doi.org/10.3390/nu12092703
Chicago/Turabian StyleOta, Yuki, Mineaki Kitamura, Kiyokazu Tsuji, Kenta Torigoe, Ayuko Yamashita, Shinichi Abe, Kumiko Muta, Tadashi Uramatsu, Yoko Obata, Junya Furutani, and et al. 2020. "Risk Reduction for End-Stage Renal Disease by Dietary Guidance Using the Gustatory Threshold Test for Salty Taste" Nutrients 12, no. 9: 2703. https://doi.org/10.3390/nu12092703
APA StyleOta, Y., Kitamura, M., Tsuji, K., Torigoe, K., Yamashita, A., Abe, S., Muta, K., Uramatsu, T., Obata, Y., Furutani, J., Takashima, M., Mukae, H., & Nishino, T. (2020). Risk Reduction for End-Stage Renal Disease by Dietary Guidance Using the Gustatory Threshold Test for Salty Taste. Nutrients, 12(9), 2703. https://doi.org/10.3390/nu12092703





