Potential of Chlorella as a Dietary Supplement to Promote Human Health
Abstract
1. Introduction
2. Nutrients in Commercial Chlorella Products
2.1. Macronutrients
2.2. Micronutrients
2.2.1. Vitamins
2.2.2. Minerals
2.3. Pigments
3. Pharmacological Activities of Chlorella Products
3.1. Antihypertensive Effects
3.2. Antihypercholesterolemic and Antihyperlipemic Effects
3.3. Antidiabetic Effect
3.4. Hepatoprotective Effect
3.5. Detoxification Effect
3.6. Immunomodulatory Effects
3.7. Antioxidant Effects
3.8. Other Effects
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yan, N.; Fan, C.; Chen, Y.; Hu, Z. The potential for microalgae as bioreactors to produce pharmaceuticals. Int. J. Mol. Sci. 2016, 17, 962. [Google Scholar] [CrossRef] [PubMed]
- Barkia, I.; Saari, N.; Manning, S.R. Microalgae for high-value products towards human health and nutrition. Mar. Drugs 2019, 17, 304. [Google Scholar] [CrossRef] [PubMed]
- Beijerinck, M.W. Culturversuche mit Zoochlorellen, Lichenengonidien und anderen niederen Algen. Botanische Zeitung 1890, 47, 725–739. [Google Scholar]
- Chick, H. A study of a unicellular green alga, occurring in polluted water, with especial reference to its nitrogenous metabolism. Proc. Royal Soc. Biol. Sci. Ser. B 1903, 71, 458–476. [Google Scholar]
- Wu, H.L.; Hseu, R.S.; Lin, L.P. Identification of Chlorella spp. isolates using ribosomal DNA sequences. Bot. Bull. Acad. Sin. 2001, 42, 115–121. [Google Scholar]
- Krienitz, L.; Hegewald, E.H.; Hepperle, D.; Huss, V.A.R.; Rohr, T.; Wolf, M. Phylogenetic relationship of Chlorella and Parachlorella gen. nov. (Chloropyta, Trebouxiophyceae). Phycologia 2004, 43, 529–542. [Google Scholar] [CrossRef]
- Sorokin, C.; Myers, J. A high-temperature strain of Chlorella. Science 1953, 117, 330–331. [Google Scholar] [CrossRef]
- Lizzul, A.M.; Lekuona-Amundarain, A.; Purton, S.; Campos, L.C. Characterization of Chlorella sorokiniana, UTEX 1230. Biology 2018, 7, 25. [Google Scholar] [CrossRef]
- Montoya, E.Y.O.; Casazza, A.A.; Aliakbarian, B.; Perego, P.; Converti, A.; Carvalho, M. Production of Chlorella vulgaris as a source of essential fatty acids in a tubular photobioreactor continuously fed with air enriched with CO2 at different concentrations. Biotechnol. Prog. 2014, 30, 916–922. [Google Scholar] [CrossRef]
- Rani, K.; Sandal, N.; Sahoo, P.K. A comprehensive review on chlorella-its composition, health benefits, market and regulation scenario. Pharma Innov. J. 2018, 7, 583–589. [Google Scholar]
- De Ortega, A.R.; Roux, J.C. Production of Chlorella biomass in different types of Flat bioreactors in temperate zones. Biomass 1986, 10, 141–156. [Google Scholar] [CrossRef]
- Ru, I.T.K.; Sung, Y.Y.; Jusoh, M.; Wahid, M.E.A. Chlorella vulgaris: A perspective on its potential for combining high biomass with high value bioproducts. App. Phycol. 2020, 1, 2–11. [Google Scholar] [CrossRef]
- Shukla, S.P.; Kvíderová, J.; Tríska, J.; Elster, J. Chlorella mirabilis as a potential species for biomass production in low-temperature environment. Front. Microbiol. 2013, 4, 97. [Google Scholar] [CrossRef] [PubMed]
- Ward, V.C.A.; Rehmann, L. Rast media optimization for mixotrophic cultivation of Chlorella vulgaris. Sci. Rep. 2019, 9, 19262. [Google Scholar] [CrossRef]
- Komaki, H.; Yamashita, M.; Niwa, Y.; Tanabe, Y.; Kamiya, N.; Ando, Y.; Furuse, M. The effect of processing of Chlorella vulgaris: K-5 on in vitro and in vivo digestibility in rats. Anim. Feed Sci. Technol. 1998, 70, 363–366. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef]
- Becker, E.W. Micro-algae as a sourse of protein. Biotechnol. Adv. 2007, 25, 207–210. [Google Scholar] [CrossRef]
- Waghmare, A.G.; Salve, M.K.; LeBlanc, J.G.; Arya, S.S. Concentration and characterization of microalgae proteins from Chlorella pynenoidosa. Bioresour. Bioprocess. 2016, 3, 16. [Google Scholar] [CrossRef]
- Kent, M.; Welladsen, H.M.; Mangott, A.; Li, Y. Nutritional evaluation of Australian microalgae as potential human health supplements. PLoS ONE 2015, 10, e0118985. [Google Scholar] [CrossRef]
- Morris, S.M., Jr. Recent advances in arginine metabolism: Roles and regulation of the arginases. Br. J. Pharm. 2009, 157, 922–930. [Google Scholar] [CrossRef]
- Bansal, V.; Ochoa, J.B. Arginine availability, arginase, and the immune response. Curr. Opin. Clin. Nutr. Metab. Care 2003, 6, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Olaitan, S.A.; Northcote, D.H. Polysaccharides of Chlorella pyrenoidosa. Biochem. J. 1962, 82, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Sui, Z.; Gizaw, Y.; BeMiller, J.N. Extraction of polysaccharides from a species of Chlorella. Carbohydr. Polym. 2012, 90, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Chen, M.; Gui, J.; Huang, S.; Liu, Y.; Shentu, H.; He, J.; Fang, Z.; Wang, W.; Zhag, Y. Preparation of Chlorella vulgaris polysaccharides and their antioxidant activity in vitro and in vivo. Int. J. Biol. Macromol. 2019, 137, 139–150. [Google Scholar] [CrossRef] [PubMed]
- El-Naggar, N.E.A.; Hussein, M.H.; Shaaban-Dessuuki, S.A.; Dalal, S.R. Production, extraction and characterization of Chlorella vulgaris soluble polysaccharaides and their applications in AgNPs biosynthesis and biostimulation of plant growth. Sci. Rep. 2020, 10, 3011. [Google Scholar] [CrossRef]
- Tabarsa, M.; Shin, I.S.; Lee, J.H.; Surayot, U.; Park, W.J.; You, S.G. An immune-enhancing water-soluble a-glucan from Chlorella vulgaris and structural characteristics. Food Sci. Biotechnol. 2015, 24, 1933–1941. [Google Scholar] [CrossRef]
- Ötles, S.; Pire, R. Fatty acid composition of Chlorella and Spirulina microalgae species. J. AOAC Int. 2001, 84, 1708–1714. [Google Scholar] [CrossRef]
- Panahi, Y.; Khosroshahi, A.Y.; Sahebkar, A.; Heidari, H.R. Impact of cultivation condition and media content on Chlorella vulgaris composition. Adv. Pharm. Bull. 2019, 9, 182–194. [Google Scholar] [CrossRef]
- Chiu, S.Y.; Kao, C.Y.; Chen, T.Y.; Chang, Y.B.; Kuo, C.M.; Lin, C.S. Cultivation of microalgal Chlorella for biomass and lipid production using wastewater as nutrient resource. Bioresour. Technol. 2015, 184, 179–189. [Google Scholar] [CrossRef]
- Mao, X.; Wu, T.; Sun, D.; Zhang, Z.; Chen, F. Differential responses of the green microalga Chlorella zofingiensis to the starvation of various nutrients for oil and astaxanthin production. Bioresour. Technol. 2018, 249, 791–798. [Google Scholar] [CrossRef]
- Woortman, D.V.; Fuchs, T.; Striegel, L.; Fuchs, M.; Weber, N.; Brück, T.B.; Rychlik, M. Microalae a superior source of folates: Quantification of folates in halophile microalgae by stable isotope dilution assay. Front. Bioeng. Biotechnol. 2020, 7, 481. [Google Scholar] [CrossRef] [PubMed]
- Lips, P. Vitamin D physiology. Prog. Biophys. Mol. Biol. 2006, 92, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Taofiq, O.; Fernandes, A.; Barreiro, M.F.; Ferreira, I.C.F.R. UV-irradiated mushrooms as a source of vitamin D2: A review. Trends Food Sci. Technol. 2017, 70, 82–94. [Google Scholar] [CrossRef]
- Holman, E.H.; Martin, B.R.; Lackcik, P.L.; Godeon, D.T.; Fleet, J.C.; Weaver, C.M. Bioavailability and efficacy of vitamin D2 from UV-irradiated yeast in growing, vitamin D-deficient rats. J. Agric. Food Chem. 2011, 59, 2341–2346. [Google Scholar]
- Ložnjak, P.; Jakobsen, J. Stability of vitamin D3 and vitamin D2 in oil, fish and mushrooms after household cooking. Food Chem. 2018, 254, 144–149. [Google Scholar] [CrossRef]
- Cardwell, G.C.; Bornman, J.F.; James, A.P.; Black, L.J. A review of mushrooms as a potential source of dietary vitamin D. Nutrients 2018, 10, 1498. [Google Scholar] [CrossRef]
- Weete, J.D.; Abril, M.; Blackwell, M. Phylogenetic distribution of fungal sterols. PLoS ONE 2010, 5, e10899. [Google Scholar] [CrossRef]
- Huang, G.; Cai, W.; Xu, B. Vitamin D2, ergosterol, and vitamin B2 content in commercially dried mushrooms. Int. J. Vitam. Nutr. Res. 2016, 1, 1–10. [Google Scholar]
- Urbain, P.; Singler, F.; Ihorst, G.; Biesalski, H.K.; Bertz, H. Bioavailability of vitamin D2 from UV-B-irradiated button mushrooms in healthy adults deficient in serum 25-hydroxyvitamin D: A randomized controlled trial. Eur. J. Clin. Nutr. 2011, 65, 965–971. [Google Scholar] [CrossRef]
- Cashman, K.D.; Kiely, M.; Seamans, K.M.; Urbain, P. Effect of Ultraviolet light-exposed mushrooms on vitamin D status: Liquid chromatography-tandem mass spectrometry reanalysis of biobanked sera from a randomized controlled trial and a systematic review plus meta-analysis. J. Nutr. 2016, 146, 565–575. [Google Scholar] [CrossRef]
- Klosty, M.; Bergmann, W. Sterols of algae. III. The occurrence of ergosterol in Chlorella pyranoidosa. J. Am. Chem. Soc. 1952, 74, 1601. [Google Scholar] [CrossRef]
- Patterson, G.W. Sterols of Chlorella. II. The occurrence of an unusual sterol mixture in Chlorella vulgaris. Plant. Physiol. 1967, 42, 1457–1459. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, C.; Lu, Y. Preparative separation of phytosterol analogues from green alga Chlorella vulgaris using recycling counter-current chromatography. J. Separ. Sci. 2017, 40, 2326–2334. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.R.; Tripkovic, L.; Hart, K.H.; Lanham-New, S.A. Vitamin D deficiency as a public health issue: Using vitamin D2 or vitamin D3 in future fortification strategies. Proc. Nutr. Soc. 2017, 106, 481–490. [Google Scholar] [CrossRef]
- Selhub, J.; Jacques, P.F.; Bostom, A.G.; D’Agostino, R.B.; Wilson, P.W.F.; Belanger, A.J.; O’Leary, D.H.; Wolf, P.A.; Schaffer, E.J.; Rosenberg, I.H. Association between plasma homocysteine concentrations and extracranial carotid-artery stenosis. N. Engl. J. Med. 1995, 332, 286–291. [Google Scholar] [CrossRef]
- Ganguly, P.; Alam, S.F. Role of homocysteine in the develop of cardiovascular disease. Nutr. J. 2015, 14, 6. [Google Scholar] [CrossRef]
- Green, R.; Allen, L.H.; Bjorke-Mercado, A.L.; Brto, A.; Gueant, J.L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.H.; et al. Vitamin B12 deficiency. Nat. Rev. Dis. Prim. 2017, 3, 17040. [Google Scholar] [CrossRef]
- Miller, J.W.; Ribaya-Meercado, J.D.; Russell, R.M.; Shepard, D.C.; Morrow, F.D.; Cochary, E.F.; Sadowski, J.A.; Gershoff, S.N.; Selhub, J. Effect of vitamin B6-deficiency on fasting plasma homocysteine concentrations. Am. J. Clin. Nutr. 1992, 55, 1154–1160. [Google Scholar] [CrossRef]
- Forges, T.; Monnier-Barbarino, P.; Alberto, J.M.; Gueant-Rodriguez, R.M.; Daval, J.L.; Gueant, J.L. Impact of folate and homocysteine metabolism on human reproductive health. Hum. Reprod. Update 2007, 13, 225–238. [Google Scholar] [CrossRef]
- Celik, S.F.; Celik, E. Subclinical atherosclerosis and impaired cardiac autonomic control in pediatric patients with vitamin B12 deficiency. Niger. J. Clin. Pract. 2018, 21, 1012–1016. [Google Scholar]
- Bunout, D.; Petermann, M.; Hirsch, S. Low serum folate but normal homocysteine levels in patients with atherosclerotic vascular disease and matched healthy controls. Nutrition 2000, 16, 434–438. [Google Scholar] [CrossRef]
- Czeizel, A.E.; Dudás, I.; Vereczkey, A.; Bránhidy, F. The prevention of neural-tube defects and congenital heart defects. Nutrients 2013, 5, 4760–4775. [Google Scholar] [CrossRef] [PubMed]
- Gorelova, V.; Ambach, L.; Rébeillé, F.; Stove, C.; Van der Straeten, D. Fotes in plants: Research advances and progress in crop biofortification. Front. Chem. 2017, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Delchier, N.; Herbig, A.L.; Rychlik, M.; Renard, M.G.C. Folates in fruits and vegetables: Contents, processing and stability. Comp. Rev. Food Sci. Food Saf. 2016, 15, 506–528. [Google Scholar] [CrossRef]
- Watanabe, F.; Bito, T. Corrinoids in food and biological samples. In Frontiers in Natural Product Chemistry; Atta-ur-Rahman, Ed.; Bentham Science Publishers: Dubai, UAE, 2016; Volume 2, pp. 229–244. [Google Scholar]
- Watanabe, F.; Bito, T. Vitamin B12 sources and microbial interaction. Exp. Biol. Med. 2018, 243, 148–158. [Google Scholar] [CrossRef]
- Bito, T.; Tanioka, Y.; Watanabe, F. Characterization of vitamin B12 compounds from marine foods. Fish. Sci. 2018, 84, 747–755. [Google Scholar] [CrossRef]
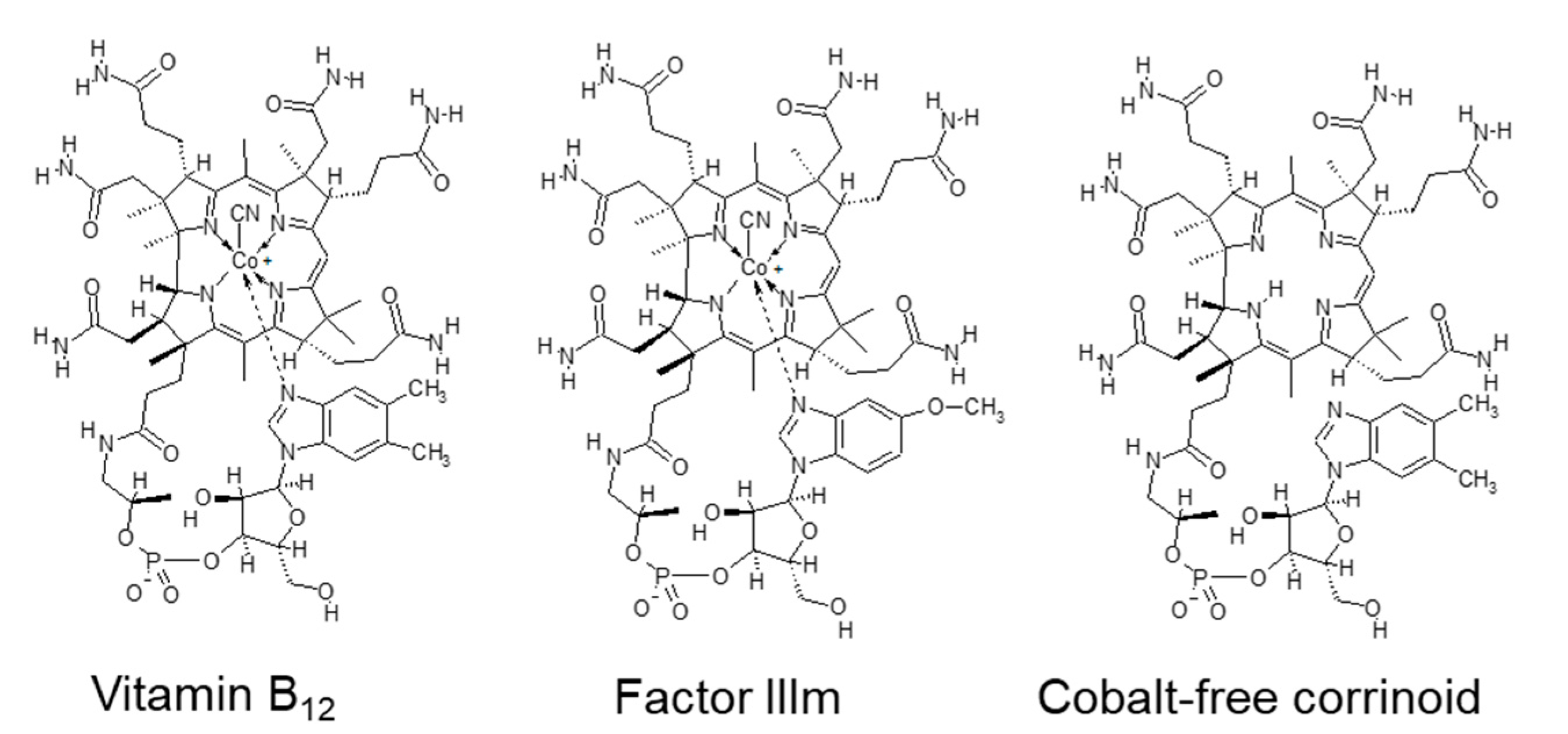
- Kittaka-Katsura, H.; Fujita, T.; Watanabe, F.; Nakano, Y. Purification and characterization of a corrinoid compound from Chlorella Tablets as an algal health food. J. Agric. Food Chem. 2002, 50, 4994–4997. [Google Scholar] [CrossRef]
- Bito, T.; Bito, M.; Asai, Y.; Takenaka, S.; Yabuta, Y.; Tago, K.; Ohnishi, M.; Mizoguchi, T.; Watanabe, F. Characterization and quantitation of vitamin B12 compounds in various Chlorella supplements. J. Agric. Food Chem. 2016, 64, 8516–8524. [Google Scholar] [CrossRef]
- Watanabe, F.; Abe, K.; Takenaka, S.; Tamura, Y.; Maruyama, I.; Nakano, Y. Occurrence of cobalamin coenzymes in the photosynthetic green alga, Chlorella vulgaris. Biosci. Biotechnol. Biochem. 1997, 61, 896–897. [Google Scholar] [CrossRef]
- Rauma, A.L.; Törrönen, R.; Hänninen, O.; Mykkänen, H. Vitamin B12 status of long-term adherents of strict uncooked vegan diet (“living food diet”) is comprised. J. Nutr. 1995, 125, 2511–2515. [Google Scholar]
- Merchant, R.E.; Phillips, T.W.; Udani, J. Nutritional supplementation with Chlorella pyrenoidosa lowers serum methylmalonic acid in vegans and vegetarians with a suspected vitamin B12 deficiency. J. Med. Food 2015, 18, 1357–1362. [Google Scholar] [CrossRef] [PubMed]
- Camaschella, C. Iron-deficiency anemia. N. Engl. J. Med. 2015, 372, 1832–1843. [Google Scholar] [CrossRef] [PubMed]
- Adrogué, H.J.; Madias, N.E. Sodium and potassium in pathogenesis of hypertension. N. Engl. J. Med. 2007, 356, 1966–1978. [Google Scholar] [CrossRef] [PubMed]
- Hentze, M.W.; Muckenthaler, M.U.; Galy, B.; Camaschella, C. Two to tango: Regulation of mammalian iron metabolism. Cell 2010, 142, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.S.; Raboy, V.; King, J.C. Implication of phytate in plant-based foods for iron and zinc bioavailability, setting dietary requirements, and formulating programs and policies. Nutr. Rev. 2018, 76, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Guo, W.; Zeng, M.; Feng, Y.; Feng, G. Effect of microalgae as iron supplements on ion-deficiency anemia in rats. Food Funct. 2019, 10, 723–732. [Google Scholar] [CrossRef]
- Nakano, S.; Takekoshi, H.; Nakano, M. Chlorella pyrenoidosa supplementation reduces the risk of anemia, proteinuria and edema in pregnant women. Plant Food Hum. Nutr. 2010, 65, 25–30. [Google Scholar] [CrossRef]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Allen, C.B.; Lacourciere, G.M.; Stadtman, T.C. Responsiveness of selenoproteins to dietary selenium. Ann. Rev. Nutr. 1999, 19, 1–16. [Google Scholar] [CrossRef]
- Sunde, R.A. Handbook of nutritionally essential mineral elements. In Handbook of Nutritionally Essential Mineral Elements; O’Dell, B.L., Sunde, R.A., Eds.; Marcel Dekker Inc: New York, NY, USA, 1997; pp. 493–556. [Google Scholar]
- White, P.J.; Brown, H.C.; Parmaguru, P.; Fritz, M.; Spracklen, W.P.; Spiby, R.E.; Meacham, M.C.; Trueman, L.J.; Smith, B.M.; Thomas, B.; et al. Interactions between selenium and Sulphur nutrition in Arabidopsis thaliana. J. Exp. Bot. 2004, 55, 1927–1937. [Google Scholar] [CrossRef]
- Doucha, J.; Lívanský, K.; Kotrbáček, V.; Zachleder, V. Production of Chlorella biomass enriched by selenium and its use in animal nutrition: A review. Appl. Microbiol. Biotechnol. 2009, 83, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Zhong, Y.; Huang, Z.; Yang, Y. Selenium accumulation in unicellular green alga Chlorella vulgaris and its effects on antioxidant enzymes and content of photosynthetic pigments. PLoS ONE 2014, 9, e112270. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.F.; Zheng, W.J.; Wong, Y.S.; Yang, F. Selenium-induced changes in activities of antioxidant enzymes and content of photosynthetic pigments in Spirulina platensis. J. Integr. Plant. Biol. 2008, 50, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Nisar, N.; Li, L.; Lu, S.; Khin, N.C.; Pogson, B.J. Carotenoid metabolism in plants. Mol. Plant. 2015, 8, 68–82. [Google Scholar] [CrossRef]
- Sathasivam, R.; Radhakrishnan, R.; Hashem, A.; Abd_Allah, E.F. Microalgae metabolites: A rich source for food and medicine. Saudi J. Biol. Sci. 2019, 26, 709–722. [Google Scholar] [CrossRef]
- Raja, R.; Hemaiswarya, S.; Rengasamy, R. Exploitation of Dunaliella for β-carotene production. Appl. Microbiol. Biotechnol. 2007, 74, 517–523. [Google Scholar] [CrossRef]
- Boussiba, S.; Bing, W.; Yuan, J.P.; Zarka, A.; Chen, F. Changes in pigments profile in the green alga Haeamtococcus pluvialis exposed to environmental stresses. Biotechnol. Lett. 1999, 20, 601–604. [Google Scholar] [CrossRef]
- O’Sullivan, A.M.; O’Callaghan, Y.C.; O’Connor, T.P.; O’Brien, N.M. The content and bioaccessibility of carotenoids from selected commercially available health supplements. Proc. Nutr. Soc. 2011, 70, E62. [Google Scholar] [CrossRef]
- Guedes, A.C.; Amaro, H.M.; Malcata, F.X. Microalgae as sources of carotenoids. Mar. Drugs 2011, 9, 625–644. [Google Scholar] [CrossRef]
- Plaza, M.; Herrero, M.; Cifuentes, A.; Ibάañez, E. Innovative natural functional ingredients from microalgae. J. Agric. Food Chem. 2009, 57, 7159–7170. [Google Scholar] [CrossRef]
- Lui, J.; Sun, Z.; Gerken, H.; Liu, S.; Jiang, Y.; Chen, F. Chlorella zofingiensis as an alternative microalgal producer of astaxanthin: Biology and industrial potential. Mar. Drugs 2014, 12, 3487–3515. [Google Scholar]
- McFarlane, S.I.; Jean-Lours, G.; Zizi, F.; Whaley-Connel, A.T.; Ogedegbe, O.; Nakaryus, A.N.; Maraj, I. Hypertension in the high-cardiovascular-risk populations. Int. J. Hypertens. 2012, 2011, 746369. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yamori, Y. Experimental evidence for dietary prevention of cardiovascular disease. Clin. Exp. Pharm. Physiol. 1989, 16, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Sansawa, H.; Takahashi, M.; Tsuchikura, S.; Endo, H. Effect of Chlorella and its fractions on blood pressure, cerebral stroke lesions, and life-span in stroke-prone spontaneously hypertensive rats. J. Nutr. Sci. Vitam. 2006, 52, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Merchant, R.E.; Andre, C.A.; Sica, D.A. Nutritional supplementation with Chlorella pyrenoidosa for mild to moderate hypertension. J. Med. Food 2002, 5, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Aznaouridis, K.; Terentes-Printzios, D.; Ioakeimidis, N.; Stefanadis, C. Prediction of cardiovascular events and all-cause mortality with brachial-ankle elasticity index: A systematic review and meta-analysis. Hypertension 2012, 60, 556–562. [Google Scholar] [CrossRef]
- Plantinga, Y.; Ghiadoni, L.; Magagna, A.; Giannarelli, C.; Franzoni, F.; Salvetti, A. Supplementation with vitamins C and E improves arterial stiffness and endothelial function in essential hypertensive patients. Am. J. Hypertens. 2007, 20, 392–397. [Google Scholar] [CrossRef]
- Nestel, P.J.; Pomeroy, S.E.; Sasahara, T.; Yamashita, T.; Ling, Y.L.; Dart, A.M.; Jennings, G.L.; Abbey, M.; Cameron, J.D. Arterial compliance in obese subjects is improved with dietary plant n-3 fatty acid from flaxseed oil despite increased LDL oxidizability. Arter. Thromb. Vasc. Biol. 1997, 17, 1163–1170. [Google Scholar] [CrossRef]
- He, F.J.; Marciniak, M.; Carney, C.; Markandu, N.D.; Anand, V.; Fraser, W.D.; Dalton, R.N.; Kaski, J.C.; MacGregor, G.A. Effects of potassium chloride and potassium bicarbonate on endothelial function, cardiovascular risk factors, and bone turnover in mild hypertensives. Hypertension 2010, 55, 681–688. [Google Scholar] [CrossRef]
- Kinlay, S.; Creager, M.A.; Fukumoto, M.; Hikita, H.; Fang, J.C.; Selwyn, A.P.; Ganz, P. Endothelium-derived nitric oxide regulates arterial elasticity in human arteries in vivo. Hypertension 2001, 38, 1049–1053. [Google Scholar] [CrossRef]
- Osuki, T.; Shimizu, K.; Iemitsu, M.; Kono, I. Multicomponent supplement containing Chlorella decreases arterial stiffness in healthy young men. J. Clin. Biochem. Nutr. 2013, 53, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Otuski, T.; Shimizu, K.; Maeda, S. Changes in arterial stiffness and nitric oxide production with Chlorella-derived multicomponent supplementation in middle-aged and older individuals. J. Clin. Biochem. Nutr. 2015, 57, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Fallah, A.A.; Sarmast, E.D.; Dehkordi, S.H.; Engardeh, J.; Mahmoodnia, L.; Khaledifar, A.; Jafari, T. Effect of Chlorella supplementation on cardiovascular risk factors: A meta-analysis of randomized controlled trials. Clin. Nutr. 2018, 37, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Mora, S.; Otvos, J.D.; Rifai, N.; Rosenson, R.S.; Buring, J.E.; Ridker, P.M. Lipoprotein particle profiles by nuclear magnetic resonance compared with standard lipids and apolipoproteins in predicting incident cardiovascular disease in women. Circulation 2009, 119, 931–939. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Benn, M.; Schnohr, P.; Tybjaerg-Hansen, A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. J. Am. Med Assoc. 2007, 298, 299–308. [Google Scholar] [CrossRef]
- Sacks, F.M.; Alaupovic, P.; Moye, L.A.; Cole, T.G.; Sussex, B.; Stampfer, M.J.; Pfeffer, M.A.; Braunwald, E. VLDL, apolipoproteins B, CIII and E, and risk of recurrent coronary events in the cholesterol and recurrent events (CARE) trial. Circulation 2000, 102, 1886–1892. [Google Scholar] [CrossRef]
- Adnerson, J.W.; Deakins, D.A.; Bridges, S.R. Soluble fiber: Hypocholesteromic effects and proposed mechanisms. In Dietary Fiber: Chemistry, Physiology and Health Effects; Kritchevsky, D., Bonfield, C., Anderson, J.W., Eds.; Plenum Press: New York, NY, USA, 1990; pp. 339–363. [Google Scholar]
- Cherng, J.Y.; Shih, M.F. Preventing dyslipidemia by Chlorella pyrenoidosa in rats and hamsters after chronic high fat diet treatment. Life Sci. 2005, 76, 3001–3013. [Google Scholar] [CrossRef]
- Shibata, S.; Oda, K.; Onodera-Masuoka, N.; Matsubara, S.; Kikuchi-Hayakawa, H.; Ishikawa, F.; Iwabuchi, A.; Sansawa, H. Hypocholesteromic effect of indigestible fraction of Chlorella regularis in cholesterol-fed rats. J. Nutr. Sci. Vitam. 2001, 47, 373–377. [Google Scholar] [CrossRef]
- Shibata, S.; Hayakawa, K.; Egashira, Y.; Sanada, H. Hypocholesteromic mechanism of Chlorella: Chlorella and its indigestible fraction enhance hepatic cholesterol catabolism through up-regulation of cholesterol 7α-hydroxylase in rats. Biosci. Biotechnol. Biochem. 2007, 71, 916–925. [Google Scholar] [CrossRef]
- Sansawa, H.; Inoue, K.; Shirai, T. Effect of Chlorella tablet ingestion on mild hypercholesterolemic patients. J. Jpn. Soc. Food Sci. Technol. 2002, 49, 167–173. [Google Scholar] [CrossRef][Green Version]
- Ryu, N.H.; Lim, Y.; Park, J.E.; Kim, J.; Kim, J.Y.; Kwon, S.W.; Kwon, O. Impact of daily Chlorella consumption on serum lipid and carotenoid profiles in mild hypercholesterolemic adults: A double-blinded, randomized, placebo-controlled study. Nutr. J. 2014, 13, 57. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, J.; Lim, Y.; Kim, Y.J.; Kim, J.Y.; Kwon, O. A dietary cholesterol challenge study to assess Chlorella supplementation in maintaining healthy lipid levels in adults: A double-blinded, randomized, placebo-controlled study. Nutr. J. 2016, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Caruso, R.; Magon, A.; Baroni, I.; Dellafiore, F.; Arrigoni, C.; Pittella, F.; Ausili, D. Health literacy in type 2 diabetes patients: A systematic review of systematic reviews. Acta Diabetol. 2018, 55, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Chaimani, A.; Hoffmann, G.; Schwedhelm, C.; Boeing, H. Impact of different dietary approaches on glycemic control and cardiovascular risk factors in patients with type 2 diabetes: A protocol for a systematic review and network meta-analysis. Sys. Rev. 2017, 6, 57. [Google Scholar] [CrossRef]
- De Groot, M.; Anderson, R.; Freedland, K.E.; Clouse, R.E.; Lustman, P.J. Association for depression and diabetes complications: A meta-analysis. Psychosom. Med. 2001, 63, 619–630. [Google Scholar] [CrossRef]
- Shibata, S.; Natori, Y.; Nishihara, T.; Tomisaka, K.; Matsumoto, K.; Sansawa, H.; Nguyen, V.C. Antioxidant and anti-cataract effects of Chlorella on rats with streptozotocin-induced diabetes. J. Nutr. Sci. Vitam. 2003, 49, 334–339. [Google Scholar] [CrossRef]
- Cherng, J.Y.; Shih, M.F. Potential hypoglycemic effects of Chlorella in streptozotocin-induced diabetic mice. Life Sci. 2005, 77, 980–990. [Google Scholar]
- Cherng, J.Y.; Shih, M.F. Improving glycogenesis in streptozotocin (STZ) diabetic mice after administration of green algae Chlorella. Life Sci. 2006, 78, 1181–1186. [Google Scholar] [CrossRef]
- Vecina, J.F.; Oliveira, A.G.; Araujo, T.G.; Baggio, S.R.; Torello, C.O.; Saad, M.J.A.; Queiroz, M.L.S. Chlorella modulates insulin signaling pathway and prevents high-fat diet-induced insulin resistance in mice. Life Sci. 2014, 95, 45–52. [Google Scholar] [CrossRef]
- Horii, N.; Hasegawa, N.; Fujii, S.; Uchida, M.; Iemitsu, K.; Inoue, K.; Iemitsu, M. Effect of combination of chlorella intake and aerobic exercise training on glycemic control in type 2 diabetic rats. Nutrition 2019, 63–64, 45–50. [Google Scholar] [CrossRef]
- Itakura, H.; Kobayashi, M.; Nakamura, S. Chlorella ingestion suppresses resistin gene expression in peripheral blood cells of borderline diabetics. Clin. Nutr. ESPEN 2015, 10, e95–e101. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Kim, Y.; Lee, Y.W. Chlorella vulgaris extract ameliorates carbon tetrachloride-induced acute hepatic injury in mice. Exp. Toxicol. Pathol. 2013, 65, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Paradis, V.; Bedossa, P. Definition and natural history of metabolic steatosis: Histology and cellular aspects. Diabetes Metab. 2008, 34, 638–642. [Google Scholar] [CrossRef]
- Schwimmer, J.B.; Deutsch, R.; Kahen, T.; Lavine, J.E.; Stanley, C.; Behling, C. Prevalence of fatty liver in children and adolescents. Pediatrics 2006, 118, 1388–1393. [Google Scholar] [CrossRef] [PubMed]
- Ben, M.D.; Polimeni, L.; Baratta, F.; Pastori, D.; Loffredo, L.; Angelico, F. Modern approach to the clinical management of non-alcoholic fatty liver disease. World J. Gastroentrol. 2014, 20, 8341–8350. [Google Scholar] [CrossRef]
- Yki-Järvinen, H. Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014, 2, 901–910. [Google Scholar] [CrossRef]
- Ebrahimi-Mameghani, M.; Sadeghi, Z.; Farhangi, M.A.; Vaghef-Mehrabany, E.; Aliashrafi, S. Glucose homeostasis, insulin resistance and inflammatory biomarkers in patients with non-alcoholic fatty liver disease: Beneficial effects of supplementation with microalgae Chlorella vulgaris: A double-blind placebo-controlled randomized clinical trial. Clin. Nutr. 2017, 36, 1001–1006. [Google Scholar] [CrossRef]
- Ebrahimi-Mameghani, M.; Aliashrafi, S.; Javadzadeh, Y.; AsghariJafarabadi, M. The effect of Chlorella vulgaris supplementation on liver enzymes, serum glucose and lipid profile in patients with non-alcoholic fatty liver disease. Health Promot. Perspect. 2014, 4, 107–115. [Google Scholar]
- Azocar, J.; Diaz, A. Efficacy and safety of Chlorella supplementation in adults with chronic hepatitis C virus infection. World J. Gastroenterol. 2013, 19, 1085–1090. [Google Scholar] [CrossRef]
- Schecter, A.; Startin, J.; Wright, C.; Kelly, M.; Papke, O.; Lis, A.; Ball, M.; Olson, J.R. Dioxins in, U.S. food and estimated daily intake. Chemosphere 1994, 29, 2261–2265. [Google Scholar] [CrossRef]
- Ven den Berg, M.; Jongh, J.; Poiger, H.; Olson, J.R. The toxicokinetics and metabolism of polychlorinated dibenzo-p-dioxins (PCDDs) and dibenzofurans (PCDFs) and their relevance for toxicity. Crit. Rev. Toxicol. 1994, 24, 1–74. [Google Scholar] [CrossRef] [PubMed]
- Schecter, A.; Papke, O.; Lis, A.; Ball, M.; Ryan, J.J.; Olson, J.R.; Li, L.; Kessler, H. Decrease in milk and blood dioxin levels over two years in a mother nursing twins: Estimates of decreased maternal and increased infant dioxin body burden from nursing. Chemosphere 1996, 32, 543–549. [Google Scholar] [CrossRef]
- Masuda, Y.; Kuroki, H.; Haraguchi, H.; Nagayama, J. PCB and PCDF congeners in the blood and tissues of Yusho and Yu-cheng patients. Envion. Health Perspect. 1985, 59, 53–58. [Google Scholar]
- Morita, K.; Matsueda, T.; Iida, T.; Hasegawa, T. Chlorella accelerates dioxin excretion in rats. J. Nutr. 1999, 129, 1731–1736. [Google Scholar] [CrossRef] [PubMed]
- Kinze, M.G.; Dolbere, F.A.; Carroll, K.L.; Moore, D.H., II; Felton, J.S. Effect of cooking time and temperature on the heterocyclic amine content of fried beef patties. Food Chem. Toxicol. 1994, 32, 595–603. [Google Scholar]
- Lee, I.; Tran, M.; Evans-Nguyen, T.; Stickle, D.; Kim, S.; Han, J.; Park, J.Y.; Yang, M. Detoxification of chlorella supplement on heterocyclic amines in Korean young adults. Environ. Toxicol. Pharm. 2015, 39, 441–446. [Google Scholar] [CrossRef]
- Harding, G.; Dalziel, J.; Vass, P. Bioaccumulation of methylmercury within the marine food web of the outer bay of Fundy, gulf of maine. PLoS ONE 2018, 13, e0197220. [Google Scholar] [CrossRef]
- His, H.C.; Hsu, Y.W.; Chang, T.C.; Chien, L.C. Methylmercury concentration in fish and risk-benefit assessment of fish intake among pregnant versus infertile women in Taiwan. PLoS ONE 2016, 11, e0155704. [Google Scholar]
- Uchikawa, T.; Yasutake, A.; Kumamoto, Y.; Maruyama, I.; Kumamoto, S.; Ando, Y. The influence of Parachlorella beijerinckii CK-5 on the absorption and excretion of methylmercury (MeHg) in mice. J. Toxicol. Sci. 2010, 35, 101–105. [Google Scholar] [CrossRef]
- Maruyama, I.; Uchikawa, T.; Kanno, T.; Ando, Y.; Kitsuki, H.; Yasutake, A. Chlorella supplementation decreases methylmercury concentrations of hair and blood in healthy volunteers. Fundam. Toxicol. Sci. 2018, 5, 117–122. [Google Scholar] [CrossRef]
- Miettinen, J.K.; Rahola, T.; Hattula, T.; Rissanen, K.; Tillander, M. Elimination of 203Hg-methylmercury in man. Ann. Clin. Res. 1971, 3, 116–122. [Google Scholar] [PubMed]
- Ballatori, N.; Clarkson, T.W. Biliary transport of glutathione and methylmercury. Am. J. Physiol 1983, 244, G435–G441. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, Y.; Sinpo, K.; Imae, Y.; Nonomura, M.; Hirakawa, K. Effect of Chlorella vulgaris strain CK-5 on the frequency of bowel movement in humans. Ipn. J. Nutr. 1998, 56, 253–263. [Google Scholar]
- Nauta, A.; Engels, F.; Knippples, L.M.; Garssen, J.; Nijkamp, F.; Redegeld, F.A. Mechanisms of allergy and asthma. Eur. J. Pharmcol. 2008, 585, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Paul, W.E. CD4 T cells: Fates, functions, and faults. Blood 2008, 112, 1557–1569. [Google Scholar] [CrossRef] [PubMed]
- Luckheeram, R.V.; Zhou, R.; Verma, A.D.; Xia, B. CD4+ cells: Differentiation and functions. Clin. Dev. Immunol. 2012, 2012, 925135. [Google Scholar] [CrossRef]
- Bancroft, G.J.; Schreiber, R.D.; Unanue, E.R. Natural immunity: A T-cell-independent pathway of macrophage activation, defined in the scid mouse. Immunol. Rev. 1991, 124, 5–24. [Google Scholar] [CrossRef]
- Wynn, T.A.; Jankovic, D.; Hieny, S.; Cheever, A.W.; Sher, A. IL-12 enhances vaccine induced immunity to Schistosomiasia mansoni in mice and decreases T helper 2 cytokine expression, IgE production, and tissue eosinophilia. J. Immumol. 1995, 154, 4701–4709. [Google Scholar]
- Galli, S.J.; Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 2012, 18, 693–704. [Google Scholar] [CrossRef]
- Hasegawa, T.; Ito, K.; Ueno, S.; Kumamoto, S.; Ando, Y.; Yamada, A.; Nomoto, K.; Yasunobu, Y. Oral administration of hot water extracts of Chlorella vulgaris reduces IgE production against milk casein in mice. Int. J. Immunopharmacol. 1999, 21, 311–323. [Google Scholar] [CrossRef]
- Hsu, H.Y.; Jeyashoke, N.; Yeh, C.H.; Song, Y.J.; Hua, K.F.; Chao, L.K. Immunostimulatory bioactivity of algal polysaccharides from Chlorella pyrenoidosa activates macrophages via Toll-like receptor 4. J. Agric. Food Chem. 2010, 58, 927–936. [Google Scholar] [CrossRef] [PubMed]
- Eder, C. Mechanisms of interkeukin-1β release. Immnobiology 2009, 214, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Halperin, S.A.; Smith, B.; Nolan, C.; Shay, J.; Kralovec, J. Safety and immunoenhancing effect of a Chlorella-derived dietary supplement in healthy adults undergoing influenza vaccination: Randomized, double-bind, placebo-controlled trial. Can. Med Assoc. J. 2003, 169, 111–117. [Google Scholar]
- Lamm, M.E.; Nedrud, J.G.; Kaetzel, C.S.; Mazanec, M.B. IgA and mucosal defense. Acta Pathol. Microbiol. Immunol. Scand. 1995, 103, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Otsuki, T.; Shimizu, K.; Iemitsu, M.; Kono, I. Salivary secretory immunoglobulin a secretion increases after 4-weeks ingestion of chlorella-derived multicomponent supplement in humans: A randomized cross over study. Nutr. J. 2011, 10, 91. [Google Scholar] [CrossRef] [PubMed]
- Abel, A.M.; Yang, C.; Thakar, M.S.; Malarkannan, S. Natural killer cells: Development, maturation, and clinical utilization. Front. Immunol. 2018, 9, 1869. [Google Scholar] [CrossRef]
- Kwak, J.H.; Baek, S.H.; Woo, Y.; Han, J.K.; Kim, B.G.; Kim, O.Y. Beneficial immunostimulatory effect of short-term Chlorella supplementation: Enhancement of Natural Killer cell activity and early inflammatory response (randomized, double-blinded, placebo-controlled trial). Nutr. J. 2012, 11, 53. [Google Scholar] [CrossRef]
- Konishi, F.; Tanaka, K.; Himeno, K.; Taniguchi, K.; Nomoto, K. Antitumor effect induced by a hot water extract of Chlorella vultaris (CE): Resistance to Meth-A tumor growth mediated by CE-induced polymorphonuclear leukocytes. Cancer Immunol. Immunother. 1985, 19, 73–78. [Google Scholar] [CrossRef]
- Tanaka, K.; Tomita, Y.; Tsuruta, M.; Konishi, F.; Okuda, M.; Himeno, K. Oral administration of Chlorella vulgaris augments concomitant antitumor immunity. Immunopharmacol. Immunotoxicol. 1990, 12, 277–291. [Google Scholar] [CrossRef]
- Wu, L.C.; Ho, J.A.; Shieh, M.C.; Lu, I.W. Antioxidant and antiproliferatives of Spirulina and Chlorella water extracts. J. Agric. Food Chem. 2005, 53, 4207–4212. [Google Scholar] [CrossRef]
- Cha, K.H.; Koo, S.Y.; Lee, D.U. Antiproliferative effects of carotenoids extracted from Chlorella ellipsoidea and Chlorella vulgaris on human colon cancer cells. J. Agric. Food Chem. 2008, 56, 10521–10526. [Google Scholar] [CrossRef] [PubMed]
- Romos, A.L.; Torello, C.O.; Queiroz, M.L.S. Chlorella vulgaris modulates immunomyelopoietic activity and enhances the resistance of tumor-bearing mice. Nutr. Cancer 2010, 62, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- Noda, K.; Tanaka, K. A water-soluble glycoprotein from Chlorella vulgaris. Planta Med. 1996, 62, 423–426. [Google Scholar] [CrossRef] [PubMed]
- Noda, K.; Ohno, N.; Tanaka, K. A new type of biological response modifier from Chlorella vulgaris which needs protein moiety to show antitumor activity. Phytother. Res. 1998, 12, 309–319. [Google Scholar] [CrossRef]
- Hasegawa, T.; Matsuguchi, T.; Noda, K.; Tanaka, K.; Kumamoto, S.; Shoyama, Y.; Yoshikai, Y. Toll-like receptor 2 is at least partly involved in the antitumor activity of glycoprotein from Chlorella vulgaris. Int. Immunopharmacol. 2002, 2, 579–589. [Google Scholar] [CrossRef]
- Selkoe, D.J. Alzheimer’s disease: Genes, proteins, and therapy. Physiol. Rev. 2001, 81, 741–766. [Google Scholar] [CrossRef]
- Bosman, G.J.; Barholomeus, I.G.; de Man, A.J.; van Kalmthout, P.J.; de Grip, W.J. Erythrocyte membrane characteristics indicate abnormal cellular aging in patients with Alzheimer’s disease. Neurobiol. Aging. 1991, 12, 13–18. [Google Scholar] [CrossRef]
- Nakagawa, K.; Kiko, T.; Hatade, K.; Asai, A.; Kumra, F.; Sookwong, P.; Tsuduki, T.; Arai, H.; Miyazawa, T. Development of a high-performance liquid chromatography-based assay for carotenoids in human red blood cells: Application to clinical studies. Anal. Biochem. 2008, 381, 129–134. [Google Scholar] [CrossRef]
- Kiko, T.; Nakagawa, K.; Tsuduki, T.; Suzuki, T.; Arai, H.; Miyazawa, T. Significance of lutein in red blood cells of Alzheimer’s disease patients. J. Alzheimers Dis. 2012, 28, 593–600. [Google Scholar] [CrossRef]
- Nakagawa, K.; Kiko, T.; Hatade, K.; Sookwong, P.; Arai, H.; Miyazawa, T. Antioxidant effect of lutein towards phospholipid hydroperoxidation in human erythrocytes. Br. J. Nutr. 2009, 102, 1280–1284. [Google Scholar] [CrossRef]
- Miyazawa, T.; Nakagawa, K.; Takekoshi, H.; Higuchi, O.; Koto, S.; Kondo, M.; Kimura, F.; Miyazawa, T. Ingestion of Chlorella reduced the oxidation of erythrocyte membrance lipids in senior Japanese subjects. J. Oleo Sci. 2013, 62, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Andrade, L.; Caraveo-Anduaga, J.J.; Berglung, P.; Bijl, R.V.; de Graaf, R.; Vollebergh, W.; Dragomirecka, E.; Kohn, R.; Keller, M.; Kessler, R.; et al. The epidemiology of major depressive episodes: Results from the international consortium of psychiatric epidemiology (ICPE) surveys. Int. J. Methods Psychiatry Res. 2003, 12, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Nemeroff, C.B. Prevalence and management of treatment-resistant depression. J. Clin. Psychiatry 2007, 68, s17–s25. [Google Scholar]
- Khawam, E.; Laurence, G.; Malone, J.R.D. Side effects of antidepressants: An overview. Cleve Clin. J. Med. 2006, 73, 351–353. [Google Scholar] [CrossRef]
- Panahi, Y.; Badeli, R.; Karami, G.R.; Badeli, Z.; Sahebkar, A. A randomized controlled trial of 6-week Chlorella vulgaris supplementation in patients with major depressive disorder. Comp. Ther. Med. 2015, 23, 598–602. [Google Scholar] [CrossRef]
- Halliwell, B. Oxidative stress and neurodegeneration: Where are we now? J. Neurochem. 2006, 97, 1634–1658. [Google Scholar] [CrossRef]
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef]
- Vijayavel, K.; Anbuselvam, C.; Balasubramanian, M.P. Antioxidant effect of the marine algae Chlorella vulgaris against naphthalene-induced oxidative stress in the albino rats. Mol. Cell Biochem. 2007, 303, 39–44. [Google Scholar] [CrossRef]
- Konishi, F.; Mitsuyama, M.; Okuda, M.; Tanaka, K.; Hasegawa, T.; Nomoto, K. Protective effect of an acidic glycoprotein obtained from culture of Chlorella vulgaris against myelosuppression by 5-fluorouraci. Cancer Immunol. Immunother. 1996, 42, 268–274. [Google Scholar] [CrossRef]
- De Souza Queiroz, J.; Barbosa, C.M.V.; da Rocha, M.; Bincoletto, C.; Paredes-Gamero, E.J.; de Souza Queiroz, M.L.; Neto, J.P. Chlorella vulgaris treatment ameliorates the suppressive effects of single and repeated stressors on hematopoiesis. Brain Behav. Immun. 2013, 29, 39–50. [Google Scholar] [CrossRef]
- Armario, A. The contribution of immediate early genes to the understanding of brain processing of stressors. In Immediate Early Genes in Sensory Processing Cognitive Performance and Neurological Disorders; Pinaud, R., Termere, L., Eds.; Springer: Berlin, Germany, 2006; pp. 199–221. [Google Scholar]
- Queiroz, J.S.; Blasco, I.M.; Gagliano, H.; Daviu, N.; Román, A.G.; Belda, X.; Carrasco, J.; Rocha, M.C.; Neto, J.P.; Armario, A. Chlorella vulgaris reduces the impact of stress on hypothalamic-pituitary-adrenal axis and brain c-fos expression. Psychoneuroendocrinology 2016, 65, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Inoue, Y.H.; Kohno, N.; Fujishima, M.; Okumura, E.; Sato, K. Phenethylamine in hot water extract of Chlorella pyrenoidosa expands lifespan of SOD1 mutant adults of Drosophila melanogaster at very low dose. J. Food Bioact. 2020, 9, 52–57. [Google Scholar] [CrossRef]
- Qiu, S.; Shen, Y.; Zhang, L.; Ma, B.; Amadu, A.A.; Ge, S. Antioxidant assessment of wastewater-cultivated Chlorella sorokiniana in Drosophila melanogaster. Algal. Res. 2020, 46, 101795. [Google Scholar] [CrossRef]
| Macronutrients (Per 100 g Dry Weight) | A | B | C | D | E | F | G | H | I | J | K | L | M |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Proteins (g) | 50–65 | 61 | 63 | 65 | 57 | 50 | 56–72 | 50–67 | 50–70 | 62 | 60 | 58 | 57 |
| Fats (g) | 7–14 | 10 | 13 | 12 | 11 | 7–20 | 5–15 | 8–15 | 11 | 10 | 10 | 12 | |
| Carbohydrates (g) | 15 | 11 | 5–23 | 8–42 | 8–20 | 20 | 18 | ||||||
| Sugars (g) | 5–21 | 7 | 5 | 0–1 | 0–5 | 2–23 | 1–10 | 11 | |||||
| Dietary fibers (g) | 7–14 | 11 | 10 | 11 | 11 | 5–18 | 7–18 | 8–16 | 10 | ||||
| Remarks | *1 *2 78% | *1 *4 | *2 | *2 | *1 *2 77–82% *3 | *2 75–85% *3 | *1 *2 83% *3 | *2 82% *3 | *4 | *1 *3 *4 |
| Amino Acids (mg/100 g Dry Weight) | C | M |
|---|---|---|
| Essential | ||
| Isoleucine | 1820 | 2030 |
| Leucine | 4180 | 4480 |
| Lysine | 4659 | 3140 |
| Methionine | 1009 | 1240 |
| Phenylalanine | 2230 | 2580 |
| Threonine | 2209 | 2490 |
| Tryptophan | 1030 | 1090 |
| Valine | 2780 | 3090 |
| Histidine | 1141 | 1040 |
| Non-essential | ||
| Tyrosine | 1720 | 1940 |
| Cystine | 659 | 650 |
| Aspartic acid | 4469 | 4710 |
| Serine | 1930 | 2120 |
| Glutamic acid | 6209 | 6030 |
| Proline | 2320 | 2560 |
| Glycine | 2859 | 2990 |
| Alanine | 4009 | 4170 |
| Arginine | 3109 | 3260 |
| Vitamins (Per 100 g Dry Weight) | A | B | C | D | E | F | G | H | I | J | K | L | M |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vitamin B1 (mg) | 1.9 | 2.5 | 6.5 | 1.0–3.0 | 1.0–3.0 | 1.8 | 1.6 | ||||||
| Vitamin B2 (mg) | 3–8 | 5.6 | 5.0 | 5.7 | 5.5 | 2.0–9.0 | 4.0–9.0 | 4.0–8.0 | 5.0 | 5.0 | 4.8 | ||
| Vitamin B6 (mg) | 0.9 | 2.5 | 1.7 | 1.0–3.0 | 1.0–3.0 | 1.0–3.0 | 1.8 | ||||||
| Vitamin B12 (μg) | 20.0 | 6.0–30.0 | 200.0–500.0 | 230.0 | |||||||||
| Niacin (mg) | 20.4 | 50.0 | 40.0–80.0 | 20.0–50.0 | 10.0–40.0 | 45.9 | |||||||
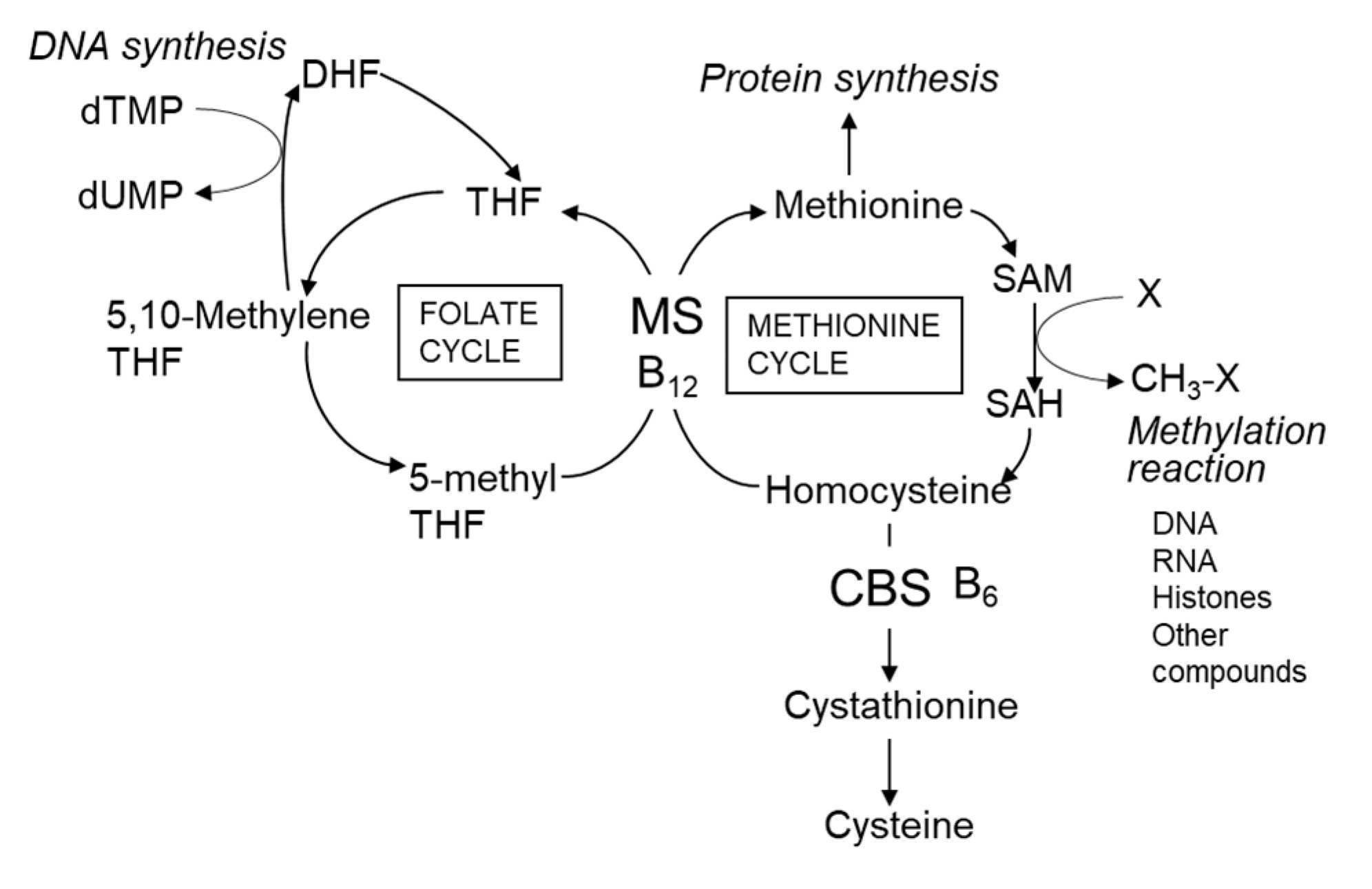
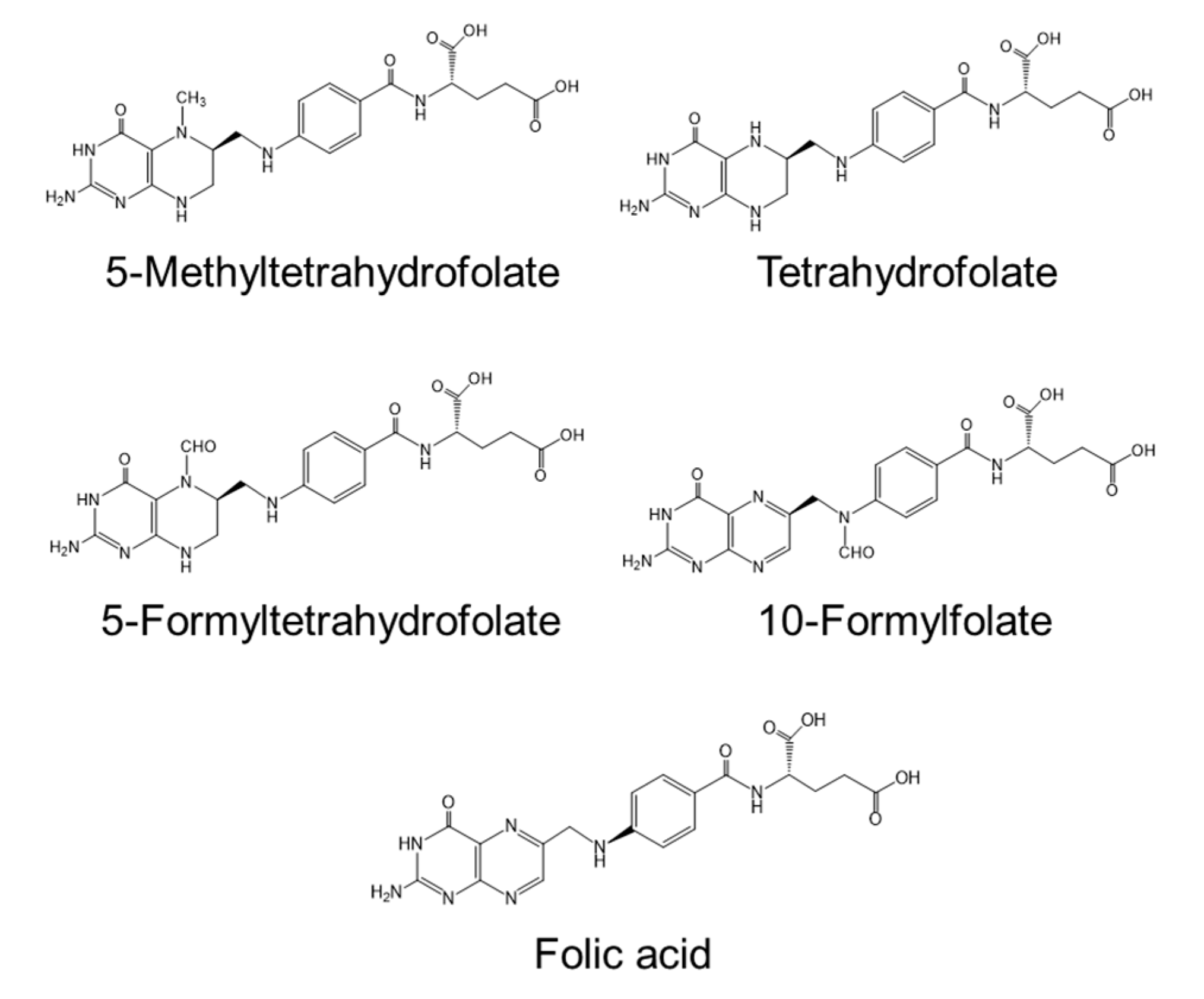
| Folate (mg) | 0.3 | 2.0 | 1.2–3.6 | 1.4 | |||||||||
| Biotin (μg) | 227.0 | ||||||||||||
| Pantothenic acid (mg) | 1.0–6.0 | 1.8 | |||||||||||
| Vitamin C (mg) | 7.0 | 50.0 | 30.0 | 10.0–200.0 | 14.0 | ||||||||
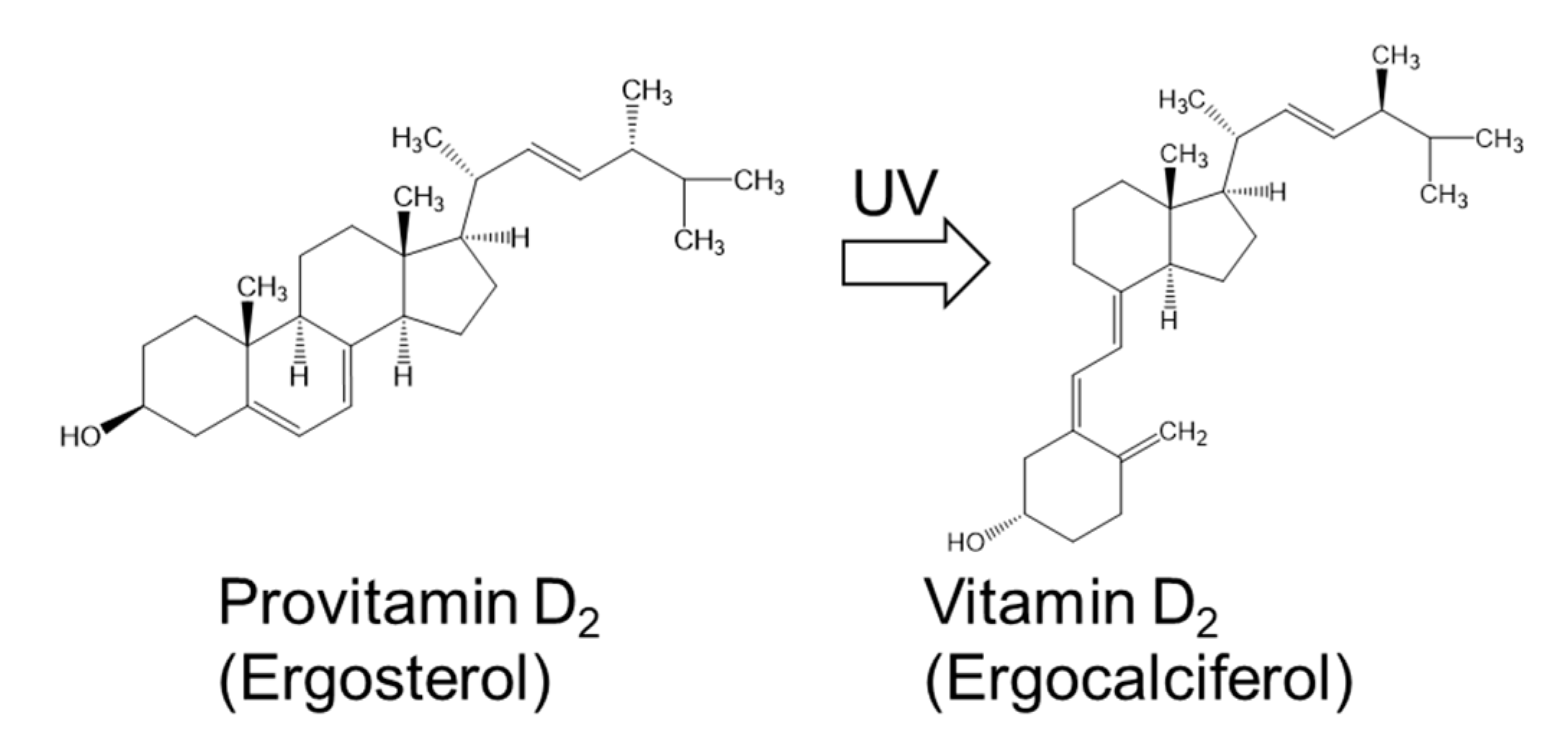
| Vitamin D2 (mg) | 1.4 | ||||||||||||
| Vitamin E (mg) | 3.0 | 25.0 | 10.0–45.0 | 6.2 | |||||||||
| Vitamin K (mg) | 1.4 | 1.1 | 0.3 | 0.5–3.5 *1 | 1.2 *1 | ||||||||
| Carotenoids (mg) | 25.0 *2 | 36.0–150.0 *2 | 100.0–500.0 | 31.5 *3 |
| Minerals (Per 100 g Dry Weight) | A | B | C | D | E | F | G | H | I | J | K | L | M |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sodium (mg) | 5–75 | 65 | 40 | 80–220 | 10–45 | 5–30 | 80 | 65 | 47 | ||||
| Iron (mg) | 10–130 | 160 | 121 | 62 | 350–1600 | 100–200 | 50–100 | 110 | 113 | ||||
| Calcium (mg) | 650 | 513 | 850 | 500–1500 | 100–300 | 433 | |||||||
| Potassium (mg) | 970 | 1075 | 350 | 200–500 | 500–1500 | 1020 | |||||||
| Magnesium (mg) | 350 | 250 | 23–420 | 298 | |||||||||
| Zinc (mg) | 2 | 1 | |||||||||||
| Copper (mg) | 1 | 1 | |||||||||||
| Phosphorus (mg) | 1600 | 1320 | |||||||||||
| Manganese (mg) | 5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bito, T.; Okumura, E.; Fujishima, M.; Watanabe, F. Potential of Chlorella as a Dietary Supplement to Promote Human Health. Nutrients 2020, 12, 2524. https://doi.org/10.3390/nu12092524
Bito T, Okumura E, Fujishima M, Watanabe F. Potential of Chlorella as a Dietary Supplement to Promote Human Health. Nutrients. 2020; 12(9):2524. https://doi.org/10.3390/nu12092524
Chicago/Turabian StyleBito, Tomohiro, Eri Okumura, Masaki Fujishima, and Fumio Watanabe. 2020. "Potential of Chlorella as a Dietary Supplement to Promote Human Health" Nutrients 12, no. 9: 2524. https://doi.org/10.3390/nu12092524
APA StyleBito, T., Okumura, E., Fujishima, M., & Watanabe, F. (2020). Potential of Chlorella as a Dietary Supplement to Promote Human Health. Nutrients, 12(9), 2524. https://doi.org/10.3390/nu12092524





