A Five-Ingredient Nutritional Supplement and Home-Based Resistance Exercise Improve Lean Mass and Strength in Free-Living Elderly
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Participants
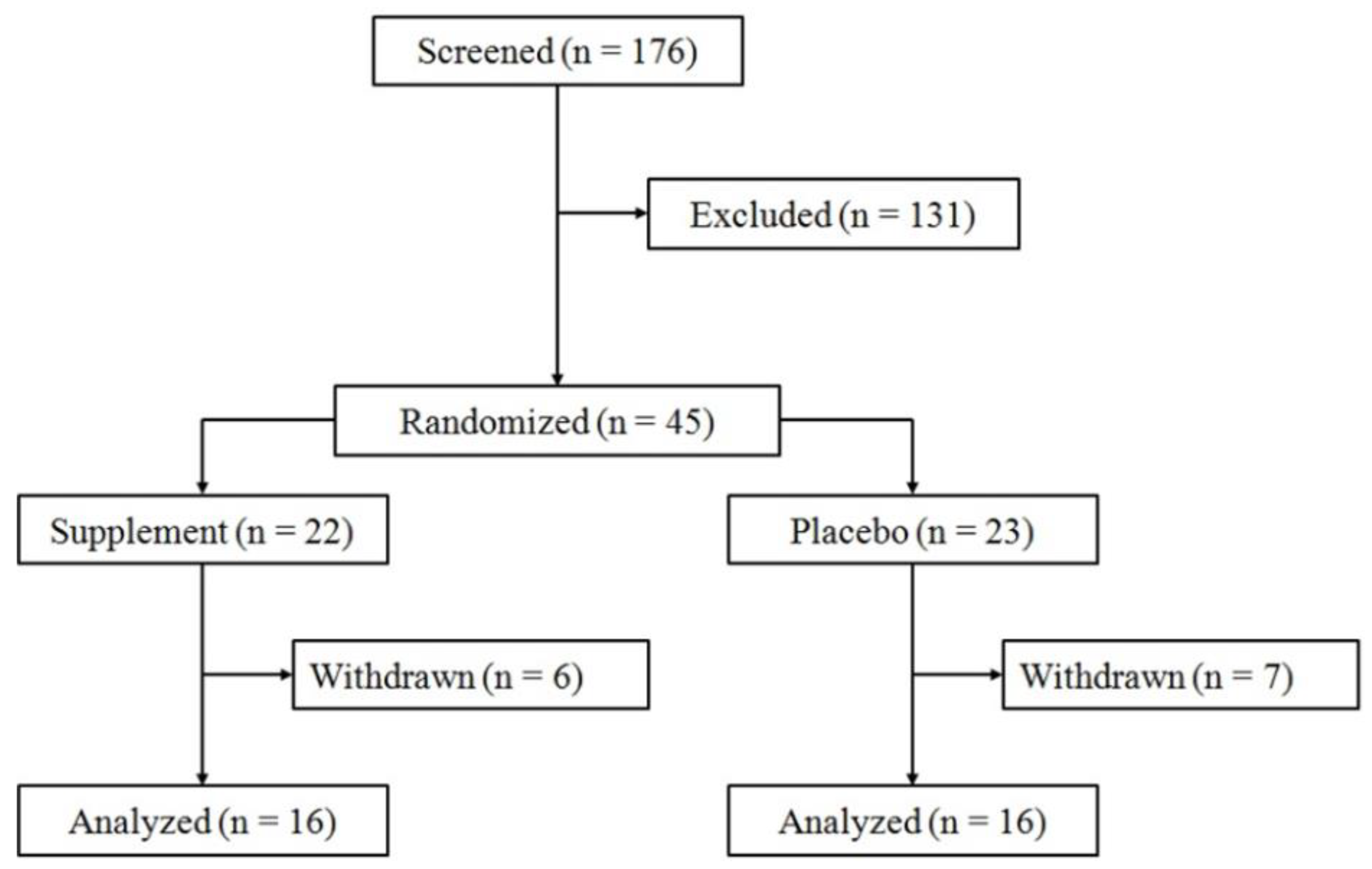
2.2.1. Recruitment, Screening, and Randomization
2.2.2. Exclusion Criteria
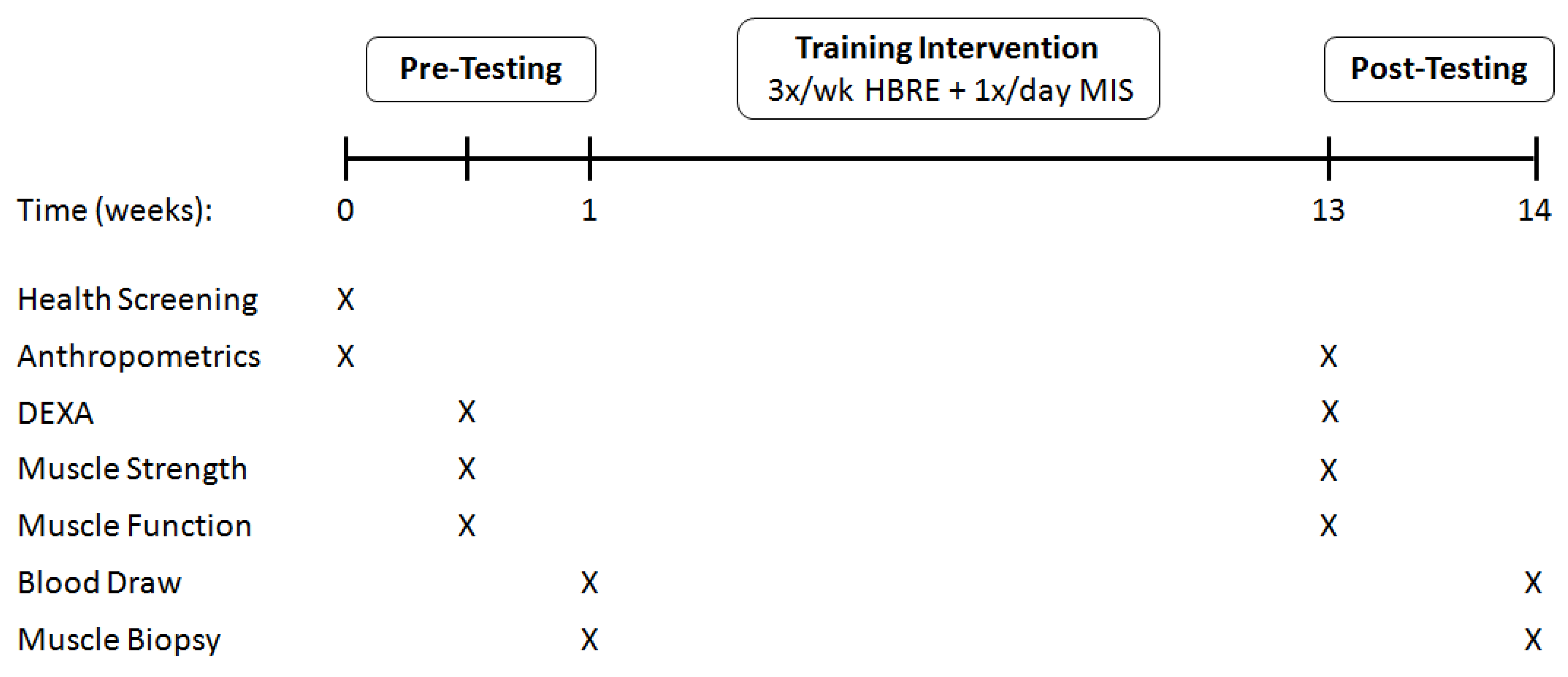
2.3. Experimental Design
2.4. Five-Ingredient Nutritional Supplement-Muscle5 (M5)
2.5. Home-Based Resistance Exercise (HBRE)
2.6. Anthropometry, Vitals, and Body Composition
2.7. Sarcopenia Diagnosis
2.8. Body Composition Testing
2.9. Maximal Strength Testing
2.10. Performance Testing
2.11. Muscle Quality
2.12. Physical Activity
2.13. Dietary Records
2.14. Muscle Biopsies
2.15. Immunofluorescence
2.16. Muscle Fiber Analysis
2.17. Blood Sampling and Analyses
2.18. Statistical Analyses
3. Results
3.1. Withdrawals, Dropouts, and Compliance
3.2. Macronutrient Intake and Daily Physical Activity
3.3. Anthropometry, Vitals, and Descriptives
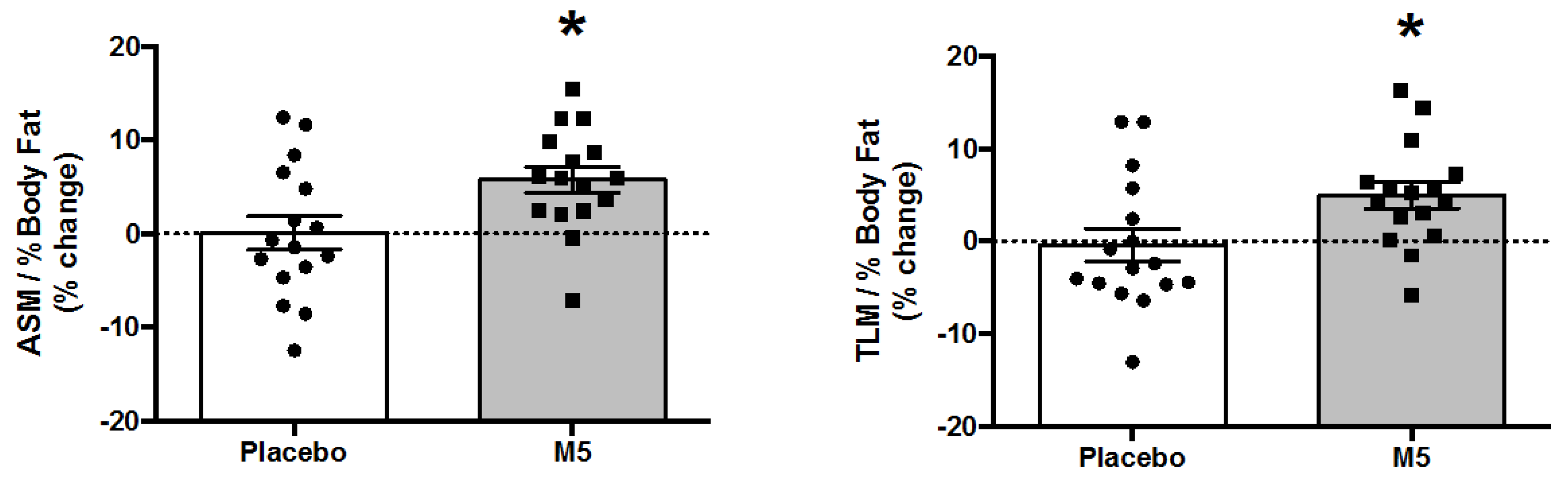
3.4. Body Composition and Muscle Mass
3.5. Muscle Fiber Cross-Sectional Areas
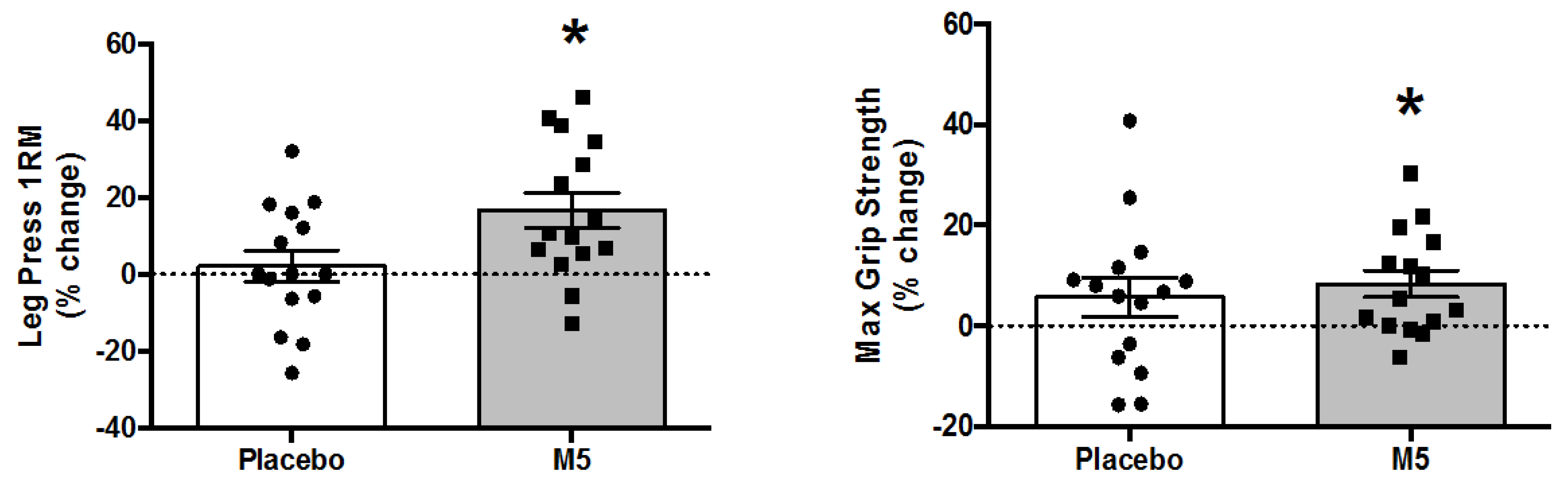
3.6. Muscle Strength
3.7. Performance Tests
3.8. Muscle Quality
3.9. Blood
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nilsson, M.I.; Tarnopolsky, M.A. Mitochondria and Aging—The Role of Exercise as a Countermeasure. Biology 2019, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.P.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.l.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 601. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Liperoti, R.; Russo, A.; Giovannini, S.; Tosato, M.; Capoluongo, E.; Bernabei, R.; Onder, G. Sarcopenia as a risk factor for falls in elderly individuals: Results from the ilSIRENTE study. Clin. Nutr. 2012, 31, 652–658. [Google Scholar] [CrossRef] [PubMed]
- Van Ancum, J.M.; Pijnappels, M.; Jonkman, N.H.; Scheerman, K.; Verlaan, S.; Meskers, C.G.M.; Maier, A.B. Muscle mass and muscle strength are associated with pre- and post-hospitalization falls in older male inpatients: A longitudinal cohort study. BMC Geriatr. 2018, 18, 116. [Google Scholar] [CrossRef] [PubMed]
- Rantanen, T.; Guralnik, J.M.; Foley, D.; Masaki, K.; Leveille, S.; Curb, J.D.; White, L. Midlife Hand Grip Strength as a Predictor of Old Age Disability. JAMA 1999, 281, 558–560. [Google Scholar] [CrossRef] [PubMed]
- Hirani, V.; Blyth, F.; Naganathan, V.; Le Couteur, D.G.; Seibel, M.J.; Waite, L.M.; Handelsman, D.J.; Cumming, R.G. Sarcopenia Is Associated With Incident Disability, Institutionalization, and Mortality in Community-Dwelling Older Men: The Concord Health and Ageing in Men Project. J. Am. Med. Dir. Assoc. 2015, 16, 607–613. [Google Scholar] [CrossRef]
- Landi, F.; Cruz-Jentoft, A.J.; Liperoti, R.; Russo, A.; Giovannini, S.; Tosato, M.; Capoluongo, E.; Bernabei, R.; Onder, G. Sarcopenia and mortality risk in frail older persons aged 80 years and older: Results from ilSIRENTE study. Age Ageing 2013, 42, 203–209. [Google Scholar] [CrossRef]
- Metter, E.J.; Talbot, L.A.; Schrager, M.; Conwit, R. Skeletal Muscle Strength as a Predictor of All-Cause Mortality in Healthy Men. J. Gerontol. Ser. A 2002, 57, B359–B365. [Google Scholar] [CrossRef]
- Lexell, J.; Henriksson-Larsén, K.; Winblad, B.; Sjöström, M. Distribution of different fiber types in human skeletal muscles: Effects of aging studied in whole muscle cross sections. Muscle Nerve 1983, 6, 588–595. [Google Scholar] [CrossRef]
- Lexell, J.; Taylor, C.C.; Sjöström, M. What is the cause of the ageing atrophy?: Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J. Neurol. Sci. 1988, 84, 275–294. [Google Scholar] [CrossRef]
- DeFronzo, R.A. Glucose intolerance and aging. Diabetes Care 1981, 4, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Feldman, H.A.; Longcope, C.; Derby, C.A.; Johannes, C.B.; Araujo, A.B.; Coviello, A.D.; Bremner, W.J.; McKinlay, J.B. Age Trends in the Level of Serum Testosterone and Other Hormones in Middle-Aged Men: Longitudinal Results from the Massachusetts Male Aging Study. J. Clin. Endocrinol. Metab. 2002, 87, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.M.; Tanaka, H. Meta-analysis of the age-associated decline in maximal aerobic capacity in men: Relation to training status. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, H829–H834. [Google Scholar] [CrossRef]
- Santilli, V.; Bernetti, A.; Mangone, M.; Paoloni, M. Clinical definition of sarcopenia. Clin. Cases Miner. Bone Metab. 2014, 11, 177–180. [Google Scholar] [CrossRef]
- Janssen, I.; Shepard, D.S.; Katzmarzyk, P.T.; Roubenoff, R. The healthcare costs of sarcopenia in the United States. J. Am. Geriatr. Soc. 2004, 52, 80–85. [Google Scholar] [CrossRef]
- Fitzgerald, S.; Barlow, C.; Kampert, J.; Morrow, J.; Jackson, A.; Blair, S. Muscular fitness and all-cause mortality: Prospective observations. J. Phys. Act. Health 2004, 1, 7–18. [Google Scholar] [CrossRef]
- Katzmarzyk, P.; Craig, C. Musculoskeletal fitness and risk of mortality. Med. Sci. Sports Exerc. 2002, 34, 740–744. [Google Scholar] [CrossRef]
- Granacher, U.; Gollhofer, A.; Hortobagyi, T.; Kressig, R.; Muehlbauer, T. The importance of trunk muscle strength or balance, functionl performance, and fall prevention in seniors: A systematic review. Sports Med. 2013, 43, 627–641. [Google Scholar] [CrossRef]
- Orr, R.; de Vos, N.; Singh, N.; Ross, D.; Stavrinos, T.; Fiatarone-Singh, M. Power training improves balance in older healthy adults. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 78–85. [Google Scholar] [CrossRef]
- Howe, T.; Rochester, L.; Neil, F.; Skelton, D.; Ballinger, C. Exercise for improving balance in older people (Review). Cochrane Database Syst. Rev. 2011, CD004963. [Google Scholar] [CrossRef]
- Liu, C.; Latham, N. Progressive resistance strength training for improving physical function in older adults. Cochrane Database Syst. Rev. 2009, 2009, CD002759. [Google Scholar] [CrossRef] [PubMed]
- Liu-Ambrose, T.; Khan, K.; Eng, J.; Janssen, P.; Lord, S.; McKay, H. Both resistance and agility training reduce fall risk in 75-85 year old women wih low bone mass: A 6-month randomized controlled trial. J. Am. Geriatr. Soc. 2004, 52, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Fiatarone, M.A.; Marks, E.C.; Ryan, N.D.; Meredith, C.N.; Lipsitz, L.A.; Evans, W.J. High-intensity strength training in nonagenarians. Effects on skeletal muscle. Jama 1990, 263, 3029–3034. [Google Scholar] [CrossRef]
- Fiatarone, M.A.; O’Neill, E.F.; Ryan, N.D.; Clements, K.M.; Solares, G.R.; Nelson, M.E.; Roberts, S.B.; Kehayias, J.J.; Lipsitz, L.A.; Evans, W.J. Exercise training and nutritional supplementation for physical frailty in very elderly people. N. Engl. J. Med. 1994, 330, 1769–1775. [Google Scholar] [CrossRef]
- Howe, T.E.; Shea, B.; Dawson, L.J.; Downie, F.; Murray, A.; Ross, C.; Harbour, R.T.; Caldwell, L.M.; Creed, G. Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst. Rev. 2011, CD000333. [Google Scholar] [CrossRef]
- Kryger, A.I.; Andersen, J.L. Resistance training in the oldest old: Consequences for muscle strength, fiber types, fiber size, and MHC isoforms. Scand. J. Med. Sci. Sports 2007, 17, 422–430. [Google Scholar] [CrossRef]
- Peterson, M.; Sen, A.; Gordon, P. Influence of resistance exercise on lean body mass in aging adults: A meta-analysis. Med. Sci. Sports Exerc. 2011, 43, 249–258. [Google Scholar] [CrossRef]
- Hunter, G.; McCarthy, J.; Bamman, M. Effects of resistance training on older adults. Sports Med. 2004, 34, 329–348. [Google Scholar] [CrossRef]
- Peterson, M.D.; Rhea, M.R.; Sen, A.; Gordon, P.M. Resistance exercise for muscular strength in older adults: A meta-analysis. Ageing Res. Rev. 2010, 9, 226–237. [Google Scholar] [CrossRef]
- Fielding, R.A.; LeBrasseur, N.K.; Cuoco, A.; Bean, J.; Mizer, K.; Singh, M.A.F. High-velocity resistance training increases skeletal muscle peak power in older women. J. Am. Geriatr. Soc. 2002, 50, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Grimby, G.; Aniansson, A.; Hedberg, M.; Henning, G.B.; Grangard, U.; Kvist, H. Training can improve muscle strength and endurance in 78- to 84-yr-old men. J. Appl. Physiol. 1992, 73, 2517–2523. [Google Scholar] [CrossRef] [PubMed]
- Mazzetti, S.A.; Kraemer, W.J.; Volek, J.S.; Duncan, N.D.; Ratamess, N.A.; Gomez, A.L.; Newton, R.U.; Hakkinen, K.; Fleck, S.J. The influence of direct supervision of resistance training on strength performance. Med. Sci. Sports Exerc. 2000, 32, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Tsekoura, M.; Billis, E.; Tsepis, E.; Dimitriadis, Z.; Matzaroglou, C.; Tyllianakis, M.; Panagiotopoulos, E.; Gliatis, J. The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial. J. Clin. Med. 2018, 7, 480. [Google Scholar] [CrossRef]
- Lacroix, A.; Hortobagyi, T.; Beurskens, R.; Granacher, U. Effects of Supervised vs. Unsupervised Training Programs on Balance and Muscle Strength in Older Adults: A Systematic Review and Meta-Analysis. Sports Med. 2017, 47, 2341–2361. [Google Scholar] [CrossRef]
- Maruya, K.; Asakawa, Y.; Ishibashi, H.; Fujita, H.; Arai, T.; Yamaguchi, H. Effect of a simple and adherent home exercise program on the physical function of community dwelling adults sixty years of age and older with pre-sarcopenia or sarcopenia. J. Phys. Ther. Sci. 2016, 28, 3183–3188. [Google Scholar] [CrossRef]
- Timmons, J.F.; Griffin, C.; Cogan, K.E.; Matthews, J.; Egan, B. Exercise Maintenance in Older Adults 1 Year After Completion of a Supervised Training Intervention. J. Am. Geriatr. Soc. 2019, 68, 163–169. [Google Scholar] [CrossRef]
- Bortz, W.M., 2nd. The disuse syndrome. West. J. Med. 1984, 141, 691–694. [Google Scholar]
- Constandt, B.; Thibaut, E.; De Bosscher, V.; Scheerder, J.; Ricour, M.; Willem, A. Exercising in Times of Lockdown: An Analysis of the Impact of COVID-19 on Levels and Patterns of Exercise among Adults in Belgium. Int. J. Environ. Res. Public Health 2020, 17, 4144. [Google Scholar] [CrossRef]
- Breen, L.; Phillips, S.M. Skeletal muscle protein anabolism in elderly: Intervention to counteract the “anabolic resistance” of ageing. Nutr. Metab. 2011, 8, 68. [Google Scholar] [CrossRef]
- Churchward-Venne, T.A.; Breen, L.; Phillips, S.M. Alterations in human muscle protein metabolism with aging: Protein and exercise as countermeasures to offset sarcopenia. BioFactors 2013, 40, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, J.M.; Volpi, E.; Rasmussen, B.B. Exercise and nutrition to target protein synthesis impairments in aging skeletal muscle. Exerc. Sport Sci. Rev. 2013, 41, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Selby, A.; Rankin, D.; Patel, R.; Atherton, P.; Hildebrandt, W.; Williams, J.; Smith, K.; Seynnes, O.; Hiscock, N.; et al. Age-related differences in the dose–response relationship of muscle protein synthesis to resistance exercise in young and old men. J. Physiol. 2009, 587, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Fry, C.S.; Drummond, M.J.; Glynn, E.L.; Dickinson, J.M.; Gundermann, D.M. Aging impairs contraction-induced human skeletal muscle mTORC1 signaling and protein synthesis. Skelet. Muscle 2011, 1, 11. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.; Res, P.; de Groot, L.; Saris, W.; van Loon, L. Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: A meta-analysis. Am. J. Clin. Nutr. 2012, 96, 1454–1464. [Google Scholar] [CrossRef] [PubMed]
- Atherton, P.J.; Smith, K.; Etheridge, T.; Rankin, D.; Rennie, M.J. Distinct anabolic signalling responses to amino acids in C2C12 skeletal muscle cells. Amino Acids 2010, 38, 1533–1539. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.J.; Hossain, T.; Hill, D.S.; Phillips, B.E.; Crossland, H.; Williams, J.; Loughna, P.; Churchward-Venne, T.A.; Breen, L.; Phillips, S.M.; et al. Effects of leucine and its metabolite beta-hydroxy-beta-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 2013, 591, 2911–2923. [Google Scholar] [CrossRef]
- Crozier, S.J.; Kimball, S.R.; Emmert, S.W.; Anthony, J.C.; Jefferson, L.S. Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J. Nutr. 2005, 135, 376–382. [Google Scholar] [CrossRef]
- Reidy, P.; Walker, D.; Dickinson, J.; Gundermann, D.; Drummond, M.; Timmerman, K.; Fry, C.; Borack, M.; Cope, M.; Mukherjea, R.; et al. Protein blend ingestion following resistance exercise promotes human muscle protein synthesis. J. Nutr. 2013, 143, 410–416. [Google Scholar] [CrossRef]
- Symons, T.; Sheffield-Moore, M.; Mamerow, M.; Wolfe, R.; Paddon-Jones, D. The anabolic response to resistance exercise and a protein rich meal is not diminished by age. J. Nutr. Health Aging 2011, 15, 376–381. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Witard, O.C. Characterising the muscle anabolic potential of dairy, meat and plant-based protein sources in older adults. Proc. Nutr. Soc. 2018, 77, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Van Vliet, S.; Burd, N.A.; van Loon, L.J.C. The Skeletal Muscle Anabolic Response to Plant- versus Animal-Based Protein Consumption. J. Nutr. 2015, 145, 1981–1991. [Google Scholar] [CrossRef] [PubMed]
- Bell, K.E.; Snijders, T.; Zulyniak, M.; Kumbhare, D.; Parise, G.; Chabowski, A.; Phillips, S.M. A whey protein-based multi-ingredient nutritional supplement stimulates gains in lean body mass and strength in healthy older men: A randomized controlled trial. PLoS ONE 2017, 12, e0181387. [Google Scholar] [CrossRef] [PubMed]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2018, 52, 376–384. [Google Scholar] [CrossRef]
- O’Bryan, K.R.; Doering, T.M.; Morton, R.W.; Coffey, V.G.; Phillips, S.M.; Cox, G.R. Do multi-ingredient protein supplements augment resistance training-induced gains in skeletal muscle mass and strength? A systematic review and meta-analysis of 35 trials. Br. J. Sports Med. 2019, 54, 573–581. [Google Scholar] [CrossRef]
- Volpi, E.; Campbell, W.W.; Dwyer, J.T.; Johnson, M.A.; Jensen, G.L. Is the optimal level of protein intake for older adults greater than the recommended dietary allowance? J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 677–681. [Google Scholar] [CrossRef]
- Traylor, D.A.; Gorissen, S.H.M.; Phillips, S.M. Perspective: Protein Requirements and Optimal Intakes in Aging: Are We Ready to Recommend More Than the Recommended Daily Allowance? Adv. Nutr. 2018, 9, 171–182. [Google Scholar] [CrossRef]
- Phillips, S.M.; Chevalier, S.; Leidy, H.J. Protein requirements beyond the RDA: Implications for optimizing health. Appl. Physiol. Nutr. Metab. 2016, 41, 565–572. [Google Scholar] [CrossRef]
- ACSM. Nutrition and Athletic Performance. Med. Sci. Sports Exerc. 2016, 48, 543–568. [Google Scholar] [CrossRef]
- Rolland, Y.; Onder, G.; Morley, J.E.; Gillette-Guyonet, S.; Abellan van Kan, G.; Vellas, B. Current and future pharmacologic treatment of sarcopenia. Clin. Geriatr. Med. 2011, 27, 423–447. [Google Scholar] [CrossRef]
- Moreira-Pfrimer, L.; Pedrosa, M.; Teixeira, L. Treatment of vitamin D deficiency increases lower limb muscle strength in institutionalized older people independently of regular physical activity: A randomized double-blind controlled trial. Ann. Nutr. Metab. 2009, 54, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Beaudart, C.; Buckinx, F.; Rabenda, V.; Gillain, S.; Cavalier, E.; Slomian, J.; Petermans, J.; Reginster, J.-Y.; Bruyère, O. The Effects of Vitamin D on Skeletal Muscle Strength, Muscle Mass, and Muscle Power: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Clin. Endocrinol. Metab. 2014, 99, 4336–4345. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.; Atherton, P.; Reeds, D.; Mohammad, B.; Rankin, D.; Rennie, M.; Mittendorfer, B. Dietary omega-3 fatty acid supplementation increases the rate of muscle protein synthesis in older adults: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 93, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.I. The Effects of Dietary Omega-3s on Muscle Composition and Quality in Older Adults. Curr. Nutr. Rep. 2016, 5, 99–105. [Google Scholar] [CrossRef]
- Brose, A.; Parise, G.; Tarnopolsky, M.A. Creatine supplementation enhances isometric strength and body composition improvements following strength exercise training in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2003, 58, 11–19. [Google Scholar] [CrossRef]
- Candow, D.G.; Forbes, S.C.; Chilibeck, P.D.; Cornish, S.M.; Antonio, J.; Kreider, R.B. Variables Influencing the Effectiveness of Creatine Supplementation as a Therapeutic Intervention for Sarcopenia. Front. Nutr. 2019, 6, 124. [Google Scholar] [CrossRef]
- Tarnopolsky, M.; Zimmer, A.; Paikin, J.; Aboud, A.; Pearce, E.; Roy, B.; Doherty, T. Creatine monohydrate and conjugated linoleic acid improve strength and body composition following resistance exercise in older adults. PLoS ONE 2007, 2, e991. [Google Scholar] [CrossRef]
- Tarnopolsky, M.A. The potential benefits of creatine and conjugated linoleic acid as adjuncts to resistance training in older adults. Appl. Physiol. Nutr. Metab. 2008, 33, 213–227. [Google Scholar] [CrossRef][Green Version]
- Bell, K.E.; Snijders, T.; Zulyniak, M.A.; Kumbhare, D.; Parise, G.; Chabowski, A.; Phillips, S.M. A multi-ingredient nutritional supplement enhances exercise training-related reductions in markers of systemic inflammation in healthy older men. Appl. Physiol. Nutr. Metab. 2018, 43, 299–302. [Google Scholar] [CrossRef]
- Bell, K.E.; Fang, H.; Snijders, T.; Allison, D.J.; Zulyniak, M.A.; Chabowski, A.; Parise, G.; Phillips, S.M.; Heisz, J.J. A Multi-Ingredient Nutritional Supplement in Combination With Resistance Exercise and High-Intensity Interval Training Improves Cognitive Function and Increases N-3 Index in Healthy Older Men: A Randomized Controlled Trial. Front. Aging Neurosci. 2019, 11, 107. [Google Scholar] [CrossRef]
- Naclerio, F.; Larumbe-Zabala, E. Effects of Whey Protein Alone or as Part of a Multi-ingredient Formulation on Strength, Fat-Free Mass, or Lean Body Mass in Resistance-Trained Individuals: A Meta-analysis. Sports Med. 2016, 46, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Barazzoni, R.; Bischoff, S.C.; Breda, J.; Wickramasinghe, K.; Krznaric, Z.; Nitzan, D.; Pirlich, M.; Singer, P. Endorsed by the E.C. ESPEN expert statements and practical guidance for nutritional management of individuals with SARS-CoV-2 infection. Clin. Nutr. (Edinb. Scotl.) 2020, 39, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Agborsangaya, C.B.; Ngwakongnwi, E.; Lahtinen, M.; Cooke, T.; Johnson, J.A. Multimorbidity prevalence in the general population: The role of obesity in chronic disease clustering. BMC Public Health 2013, 13, 1161. [Google Scholar] [CrossRef] [PubMed]
- Roberts, K.C.; Rao, D.P.; Bennett, T.L.; Loukine, L.; Jayaraman, G.C. Prevalence and patterns of chronic disease multimorbidity and associated determinants in Canada. Health Promot. Chronic Dis. Prev. Can. Res. Policy Pract. 2015, 35, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Statistics Canada. Table 13-10-0096-20 Body Mass Index, Overweight or Obese, Self-Reported, Adult, Age Groups (18 Years and Older); Canadian Community Health Survey: Ottawa, ON, Canada, 2018.
- Statistics Canada. Table 13-10-0096-13 Physical Activity, Self-Reported, Adult, by Age Group; Canadian Community Health Survey: Ottawa, ON, Canada, 2018.
- Liao, C.-D.; Tsauo, J.-Y.; Lin, L.-F.; Huang, S.-W.; Ku, J.-W.; Chou, L.-C.; Liou, T.-H. Effects of elastic resistance exercise on body composition and physical capacity in older women with sarcopenic obesity: A CONSORT-compliant prospective randomized controlled trial. Medicine 2017, 96, e7115. [Google Scholar] [CrossRef]
- Melov, S.; Tarnopolsky, M.A.; Beckman, K.; Felkey, K.; Hubbard, A. Resistance exercise reverses aging in human skeletal muscle. PLoS ONE 2007, 2, e465. [Google Scholar] [CrossRef]
- Barbat-Artigas, S.; Rolland, Y.; Zamboni, M.; Aubertin-Leheudre, M. How to assess functional status: A new muscle quality index. J. Nutr. Health Aging 2016, 16, 67–77. [Google Scholar] [CrossRef]
- Sasaki, H.; Kasagi, F.; Yamada, M.; Fujita, S. Grip Strength Predicts Cause-Specific Mortality in Middle-Aged and Elderly Persons. Am. J. Med. 2007, 120, 337–342. [Google Scholar] [CrossRef]
- Roberts, H.C.; Denison, H.J.; Martin, H.J.; Patel, H.P.; Syddall, H.; Cooper, C.; Sayer, A.A. A review of the measurement of grip strength in clinical and epidemiological studies: Towards a standardised approach. Age Ageing 2011, 40, 423–429. [Google Scholar] [CrossRef]
- Sousa-Santos, A.R.; Amaral, T.F. Differences in handgrip strength protocols to identify sarcopenia and frailty—A systematic review. BMC Geriatr. 2017, 17, 238. [Google Scholar] [CrossRef]
- Beaudart, C.; McCloskey, E.; Bruyere, O.; Cesari, M.; Rolland, Y.; Rizzoli, R.; Araujo de Carvalho, I.; Amuthavalli Thiyagarajan, J.; Bautmans, I.; Bertière, M.-C.; et al. Sarcopenia in daily practice: Assessment and management. BMC Geriatr. 2016, 16, 170. [Google Scholar] [CrossRef] [PubMed]
- Guralnik, J.; Ferrucci, L.; Pieper, C.; Leveille, S.; Markides, K.; Ostir, G.; Studenski, S.; Berkman, L.; Wallace, R. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J. Gerontol. A Biol. Sci. Med. Sci. 2000, 55, M221–M231. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Kritchevsky, S.B.; Newman, A.B.; Simonsick, E.M.; Harris, T.B.; Penninx, B.W.; Brach, J.S.; Tylavsky, F.A.; Satterfield, S.; Bauer, D.C.; et al. Added value of physical performance measures in predicting adverse health-related events: Results from the Health, Aging And Body Composition Study. J. Am. Geriatr. Soc. 2009, 57, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Kritchevsky, S.B.; Penninx, B.W.H.J.; Nicklas, B.J.; Simonsick, E.M.; Newman, A.B.; Tylavsky, F.A.; Brach, J.S.; Satterfield, S.; Bauer, D.C.; et al. Prognostic Value of Usual Gait Speed in Well-Functioning Older People—Results from the Health, Aging and Body Composition Study. J. Am. Geriatr. Soc. 2005, 53, 1675–1680. [Google Scholar] [CrossRef] [PubMed]
- Fielding, R.A.; Vellas, B.; Evans, W.J.; Bhasin, S.; Morley, J.E.; Newman, A.B.; Abellan van Kan, G.; Andrieu, S.; Bauer, J.; Breuille, D.; et al. Sarcopenia: An undiagnosed condition in older adults. Current consensus definition: Prevalence, etiology, and consequences. International working group on sarcopenia. J. Am. Med. Dir. Assoc. 2011, 12, 249–256. [Google Scholar] [CrossRef]
- Baecke, J.A.; Burema, J.; Frijters, J.E. A short questionnaire for the measurement of habitual physical activity in epidemiological studies. Am. J. Clin. Nutr. 1982, 36, 936–942. [Google Scholar] [CrossRef]
- Tarnopolsky, M.A.; Pearce, E.; Smith, K.; Lach, B. Suction-modified Bergström muscle biopsy technique: Experience with 13,500 procedures. Muscle Nerve 2011, 43, 717–725. [Google Scholar] [CrossRef]
- Bloemberg, D.; Quadrilatero, J. Rapid Determination of Myosin Heavy Chain Expression in Rat, Mouse, and Human Skeletal Muscle Using Multicolor Immunofluorescence Analysis. PLoS ONE 2012, 7, e35273. [Google Scholar] [CrossRef]
- Gupta, S.K. Intention-to-treat concept: A review. Perspect. Clin. Res. 2011, 2, 109–112. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Craig, C.L.; Aoyagi, Y.; Bell, R.C.; Croteau, K.A.; De Bourdeaudhuij, I.; Ewald, B.; Gardner, A.W.; Hatano, Y.; Lutes, L.D.; et al. How many steps/day are enough? For older adults and special populations. Int. J. Behav. Nutr. Phys. Act. 2011, 8, 80. [Google Scholar] [CrossRef]
- Markus, W.; Rima, K.-D. Creatine and Creatinine Metabolism. Physiol. Rev. 2000, 80, 1107–1213. [Google Scholar]
- Lugaresi, R.; Leme, M.; de Salles Painelli, V.; Murai, I.H.; Roschel, H.; Sapienza, M.T.; Lancha Junior, A.H.; Gualano, B. Does long-term creatine supplementation impair kidney function in resistance-trained individuals consuming a high-protein diet? J. Int. Soc. Sports Nutr. 2013, 10, 26. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Roubenoff, R. Origins and clinical relevance of sarcopenia. Can. J. Appl. Physiol. 2001, 26, 78–89. [Google Scholar] [CrossRef]
- Marzetti, E.; Anne Lees, H.; Eva Wohlgemuth, S.; Leeuwenburgh, C. Sarcopenia of aging: Underlying cellular mechanisms and protection by calorie restriction. BioFactors 2009, 35, 28–35. [Google Scholar] [CrossRef]
- Drey, M. Sarcopenia-pathophysiology and clinical relevance. Wien. Med. Wochenschr. 2011, 161, 402–408. [Google Scholar] [CrossRef]
- Doherty, T.J. Invited review: Aging and sarcopenia. J. Appl. Physiol. 2003, 95, 1717–1727. [Google Scholar] [CrossRef]
- Sandri, M.; Barberi, L.; Bijlsma, A.; Blaauw, B.; Dyar, K.; Milan, G. Signalling pathways regulating musclemass in ageing skeletal muscle: The role of the IGF1-Akt-mTOR-FoxO pathway. Biogerontology 2013, 14, 303–323. [Google Scholar] [CrossRef]
- Wilkes, E.A.; Selby, A.L.; Atherton, P.J.; Patel, R.; Rankin, D. Blunting of insulin inhibition of proteolysis in legs of older subjects mayncontribute to age-related sarcopenia. Am. J. Clin. Nutr. 2009, 90, 1343–1350. [Google Scholar] [CrossRef]
- Alway, S.; Myers, M.; Mohamed, J. Regulation of satellite cell function in sarcopenia. Front. Aging Neurosci. 2014, 6, 246. [Google Scholar] [CrossRef]
- Barberi, L.; Scicchitano, B.; De Rossi, M.; Bigot, A.; Duguez, S.; Wielgosik, A.; Stewart, C.; McPhee, J.; Conte, M.; Narici, M.; et al. Age-dependent alteration in muscle regeneration: The critical role of tissue niche. Biogerontology 2013, 14, 273–292. [Google Scholar] [CrossRef] [PubMed]
- Conboy, I.; Conboy, M.; Wagers, A.; Girma, E.; Weissman, I.; Rando, T. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature 2005, 433, 760–764. [Google Scholar] [CrossRef] [PubMed]
- Alway, S.E.; Siu, P.M. Nuclear apoptosis contributes to sarcopenia. Exerc. Sport Sci. Rev. 2008, 36, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, E.; Leeuwenburgh, C. Skeletal muscle apoptosis, sarcopenia and frailty at old age. Exp. Gerontol. 2006, 41, 1234–1238. [Google Scholar] [CrossRef] [PubMed]
- Cimas, M.; Ayala, A.; Sanz, B.; Agullo-Tomas, M.S.; Escobar, A.; Forjaz, M.J. Chronic musculoskeletal pain in European older adults: Cross-national and gender differences. Eur. J. Pain 2017, 22, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Oesen, S.; Halper, B.; Hofmann, M.; Jandrasits, W.; Franzke, B.; Strasser, E.-M.; Graf, A.; Tschan, H.; Bachl, N.; Quittan, M.; et al. Effects of elastic band resistance training and nutritional supplementation on physical performance of institutionalised elderly: A randomized controlled trial. Exp. Gerontol. 2015, 72, 99–108. [Google Scholar] [CrossRef]
- Hofmann, M.; Schober-Halper, B.; Oesen, S.; Franzke, B.; Tschan, H.; Bachl, N.; Strasser, E.-M.; Quittan, M.; Wagner, K.-H.; Wessner, B. Effects of elastic band resistance training and nutritional supplementation on muscle quality and circulating muscle growth and degradation factors of institutionalized elderly women: The Vienna Active Ageing Study (VAAS). Eur. J. Appl. Physiol. 2016, 116, 885–897. [Google Scholar] [CrossRef]
- Argenziano, M.G.; Bruce, S.L.; Slater, C.L.; Tiao, J.R.; Baldwin, M.R.; Barr, R.G.; Chang, B.P.; Chau, K.H.; Choi, J.J.; Gavin, N.; et al. Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: Retrospective case series. BMJ 2020, 369, m1996. [Google Scholar] [CrossRef]
- Bello-Chavolla, O.Y.; Bahena-Lopez, J.P.; Antonio-Villa, N.E.; Vargas-Vazquez, A.; Gonzalez-Diaz, A.; Márquez-Salinas, A.; Fermin-Martinez, C.A.; Naveja, J.J.; Aguilar-Salinas, C.A. Predicting mortality due to SARS-CoV-2: A mechanistic score relating obesity and diabetes to COVID-19 outcomes in Mexico. J. Clin. Endocrinol. Metab. 2020, 105, dgaa346. [Google Scholar] [CrossRef]
- Hamer, M.; Kivimaki, M.; Gale, C.R.; Batty, G.D. Lifestyle risk factors, inflammatory mechanisms, and COVID-19 hospitalization: A community-based cohort study of 387,109 adults in UK. Brain Behav. Immun. 2020, 87, 184–187. [Google Scholar] [CrossRef]
- Nilsson, M.I.; Bourgeois, J.M.; Nederveen, J.P.; Leite, M.R.; Hettinga, B.P.; Bujak, A.L.; May, L.; Lin, E.; Crozier, M.; Rusiecki, D.R.; et al. Lifelong aerobic exercise protects against inflammaging and cancer. PLoS ONE 2019, 14, e0210863. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.C.; Henson, D.A.; Austin, M.D.; Sha, W. Upper respiratory tract infection is reduced in physically fit and active adults. Br. J. Sports Med. 2011, 45, 987. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Spaulding, H.R. Extracellular superoxide dismutase, a molecular transducer of health benefits of exercise. Redox Biol. 2020, 32, 101508. [Google Scholar] [CrossRef]
- Nieman, D.C.; Wentz, L.M. The compelling link between physical activity and the body’s defense system. J. Sport Health Sci. 2019, 8, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Simpson, R.J.; Lowder, T.W.; Spielmann, G.; Bigley, A.B.; LaVoy, E.C.; Kunz, H. Exercise and the aging immune system. Ageing Res. Rev. 2012, 11, 404–420. [Google Scholar] [CrossRef] [PubMed]
- Simpson, R.J.; Kunz, H.; Agha, N.; Graff, R.; Bouchard, C. Chapter Fifteen—Exercise and the Regulation of Immune Functions. In Progress in Molecular Biology and Translational Science; Academic Press: Cambridge, MA, USA, 2015; Volume 135, pp. 355–380. [Google Scholar]
- Turner, J.E.; Brum, P.C. Does Regular Exercise Counter T Cell Immunosenescence Reducing the Risk of Developing Cancer and Promoting Successful Treatment of Malignancies? Oxidative Med. Cell. Longev. 2017, 2017, 4234765. [Google Scholar] [CrossRef] [PubMed]
- Woods, J.A.; Keylock, K.T.; Lowder, T.; Vieira, V.J.; Zelkovich, W.; Dumich, S.; Colantuano, K.; Lyons, K.; Leifheit, K.; Cook, M.; et al. Cardiovascular Exercise Training Extends Influenza Vaccine Seroprotection in Sedentary Older Adults: The Immune Function Intervention Trial. J. Am. Geriatr. Soc. 2009, 57, 2183–2191. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Ren, F.; Sun, D.; Wang, M.; Baker, J.S.; Istvan, B.; Gu, Y. Benefits of Exercise on Influenza or Pneumonia in Older Adults: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 2655. [Google Scholar] [CrossRef]
- Martin, S.A.; Pence, B.D.; Woods, J.A. Exercise and respiratory tract viral infections. Exerc. Sport Sci. Rev. 2009, 37, 157–164. [Google Scholar] [CrossRef]
- Power, S.E.; Jeffery, I.B.; Ross, R.P.; Stanton, C.; O’Toole, P.W.; O’Connor, E.M.; Fitzgerald, G.F. Food and nutrient intake of Irish community-dwelling elderly subjects: Who is at nutritional risk? J. Nutr. Health Aging 2014, 18, 561–572. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Y. Potential interventions for novel coronavirus in China: A systematic review. J. Med. Virol. 2020, 92, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Zabetakis, I.; Lordan, R.; Norton, C.; Tsoupras, A. COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients 2020, 12, 1466. [Google Scholar] [CrossRef] [PubMed]




| Old Placebo + HBRE (O-P; n = 16) | Old Muscle5 + HBRE (O-M5; n = 16) | |||
|---|---|---|---|---|
| Pre | Post | Pre | Post | |
| Anthropometry, Vitals and Descriptive Data | ||||
| Age (years) | 74.4 ± 1.3 | 77.4 ± 2.8 | ||
| Height (cm) | 173.6 ± 1.6 | 173.1 ± 2.1 | ||
| Body Weight (kg) | 82.9 ± 3.3 | 84.1 ± 3.5 § | 87.9 ± 3.7 | 88.9 ± 3.8 |
| BMI (kg/m2) | 27.4 ± 0.9 | 27.8 ± 0.9 * | 29.3 ± 1.1 | 29.6 ± 1.1 |
| Waist-To-Hip Ratio | 1.01 ± 0.02 | 1.00 ± 0.02 | 1.02 ± 0.01 | 1.01 ± 0.01 |
| Resting Heart Rate (bpm) (n.p.) | 68.4 ± 3.3 | 73.6 ± 3.0 | 69.2 ± 3.1 | 69.7 ± 4.0 |
| Systolic Blood Pressure (mmHg) | 131.8 ± 4.7 | 134.8 ± 3.2 | 135.2 ± 4.1 | 139.5 ± 5.6 |
| Diastolic Blood Pressure (mmHg) | 78.1 ± 2.3 | 76.2 ± 1.7 | 74.5 ± 2.0 | 77.7 ± 2.6 |
| HbA1c (%) | 5.8 ± 0.2 | 5.9 ± 0.2 | 6.0 ± 0.2 | 6.0 ± 0.2 |
| Comorbidities | 2.4 ± 0.4 | 2.8 ± 0.5 | ||
| Medications | 3.0 ± 0.6 | 4.7 ± 0.9 | ||
| DEXA | ||||
| Muscle Mass | ||||
| TLM (kg) | 53.56 ± 1.60 | 53.87 ± 1.54 | 55.80 ± 1.66 | 56.89 ± 1.54 * |
| ASM (kg) | 23.66 ± 0.73 | 23.90 ± 0.67 | 24.39 ± 0.79 | 25.08 ± 0.81 * |
| ASM/h2 (kg/m2) | 7.84 ± 0.21 | 7.93 ± 0.18 | 8.13 ± 0.24 | 8.36 ± 0.26 * |
| ASM/BMI (kg/[kg/m2]) | 0.87 ± 0.02 | 0.87 ± 0.03 | 0.84 ± 0.03 | 0.86 ± 0.03 |
| Bone Mass | ||||
| Bone Mineral Density (g/cm2) | 1.22 ± 0.02 | 1.23 ± 0.02 | 1.21 ± 0.02 | 1.20 ± 0.02 |
| Fat Mass | ||||
| Body Fat Mass (kg) | 25.81 ± 2.01 | 26.56 ± 2.18 | 28.82 ± 2.78 | 28.43 ± 2.84 |
| Body Fat (%) | 31.86 ± 1.40 | 32.31 ± 1.51 | 32.98 ± 2.32 | 32.09 ± 2.26 * |
| Muscle: Body Fat Ratios | ||||
| TLM/Body Fat Mass | 2.24 ± 0.16 | 2.21 ± 0.16 | 2.47 ± 0.46 | 2.54 ± 0.45 |
| ASM/Body Fat Mass | 0.99 ± 0.08 | 0.99 ± 0.08 | 1.07 ± 0.20 | 1.12 ± 0.21 * |
| TLM/% Body Fat | 1.73 ± 0.08 | 1.71 ± 0.08 | 1.92 ± 0.24 | 2.02 ± 0.26 * |
| ASM/% Body Fat | 0.76 ± 0.04 | 0.76 ± 0.04 | 0.84 ± 0.11 | 0.89 ± 0.12 * |
| Strength | ||||
| Leg Press 1RM (kg) | 145 ± 10 | 147 ± 11 | 122 ± 7 | 140 ± 7 * |
| Maximal Hand Grip (kg) | 42.2 ± 2.1 | 44.5 ± 2.5 | 38.4 ± 1.6 | 41.4 ± 1.6 * |
| Isometric Knee Extension (Nm) | 183.6 ± 11.5 | 191.5 ± 12.7 | 170.7 ± 10.0 | 179.7 ± 9.4 |
| Performance | ||||
| Timed Up and Go (TUG) (s) | 7.23 ± 0.29 | 7.16 ± 0.41 | 8.28 ± 0.46 | 7.95 ± 0.35 |
| 4-Metre Walk Test (m/s) | 0.98 ± 0.05 | 1.04 ± 0.06 | 0.92 ± 0.06 | 0.95 ± 0.06 |
| 5-Times Sit to Stand (s) | 11.68 ± 0.87 | 10.58 ± 0.73 * | 12.41 ± 0.49 | 11.38 ± 0.74 * |
| 4-Step Stair Climb (s) | 2.73 ± 0.13 | 2.72 ± 0.13 | 3.16 ± 0.17 | 2.92 ± 0.11 |
| SPPB Score | 11.0 ± 0.4 | 11.3 ± 0.4 | 10.3 ± 0.3 | 10.6 ± 0.4 |
| Muscle Quality | ||||
| MQ Index | 48.8 ± 2.2 | 51.3 ± 2.7 § | 44.4 ± 1.6 | 47.7 ± 1.6 * |
| Total Active Ingredient (per Serving) | Calories (kcal) | |
|---|---|---|
| Multi-Ingredient Supplement—‘Muscle5’ | ||
| Ingredients | ||
| 1 Whey protein | 24 g | 96 |
| 2 Micellar casein (Calcium concentration) | 16 g (416 mg) | 64 (0) |
| 3 Creatine | 3 g | 0 |
| 4 Vitamin D3/AM | 1000 IU | 0 |
| 5 Fish oil | 10 mL | 81 |
| Eicosapentaenoic acid (EPA) | 1.51 g | |
| Docosahexaenoic acid (DHA) | 0.95 g | |
| Vitamin E (d-alpha tocopherol) | 0.08 g | |
| Flavour blend | 0.33 g | |
| Flavoring | ||
| Sucrose | 6.4 g | 25 |
| Stevia | 100 mg | 0 |
| Chocolate flavor | 2.8 g | 6 |
| Total | 272 kcal | |
| Placebo | ||
| Ingredients | ||
| Collagen protein | 40 g | 160 |
| Sunflower oil | 10 mL | 81 |
| Vitamin E (d-alpha tocopherol) | 0.08 g | |
| Flavour blend | 0.34 g | |
| Flavoring | ||
| Sucrose | 6.4 g | 25 |
| Stevia | 100 mg | 0 |
| Chocolate flavor | 2.8 g | 6 |
| Xanthan gum | 150 mg | 0 |
| Total | 272 kcal |
| Old Placebo + HBRE (O-P; n = 16) | Old Muscle5 + HBRE (O-M5; n = 16) | |||
|---|---|---|---|---|
| Pre | Post | Pre | Post | |
| Nutrition | ||||
| Calories (kcal) | 1852 ± 107 | 2239 ± 90 * | 2025 ± 131 | 1988 ± 109 |
| Protein (g) | 81.2 ± 5.7 | 127.6 ± 5.2 * | 88.7 ± 7.0 | 115.7 ± 4.5 * |
| Protein (g/kg body weight) | 0.97 ± 0.07 | 1.51 ± 0.08 * | 1.07 ± 0.12 | 1.34 ± 0.08 * |
| Fat (g) | 77.7 ± 5.9 | 88.3 ± 6.5 | 79.6 ± 6.7 | 76.8 ± 8.8 |
| Carb (g) | 188.9 ± 15.0 | 213.7 ± 12.7 | 223.4 ± 20.8 | 199.8 ± 18.5 |
| Vitamin D3 (mcg) | 11.3 ± 5.3 | 5.8 ± 1.0 | 7.3 ± 2.3 | 31.3 ± 2.0 * |
| Calcium (mg) | 782.9 ± 77.7 | 816.7 ± 65.1 | 887.1 ± 116.5 | 1214.4 ± 102.7 * |
| n-3 PUFA (g) | 0.181 ± 0.058 | 0.224 ± 0.106 | 0.104 ± 0.028 | 2.768 ± 0.198 * |
| Physical activity | ||||
| Daily activity (steps/day) | 5899 ± 772 | 5230 ± 483 | 5835 ± 808 | 5659 ± 592 |
| Compliance | ||||
| Supplement (%) | n.a. | 95.4 ± 1.9 | n.a. | 89.3 ± 5.0 |
| Resistance exercise (%) | n.a. | 89.1 ± 8.1 | n.a. | 84.1 ± 6.1 |
| Old Placebo + HBRE (O-P; n = 12) | Old Muscle5 + HBRE (O-M5; n = 16) | |||||
|---|---|---|---|---|---|---|
| Pre | Post | Δ (%) | Pre | Post | Δ (%) | |
| Cross-Sectional Areas | ||||||
| Type I (μm2) | 5264 ± 298 | 6166 ± 558 | 19.4 ± 12.3 | 5163 ± 305 | 5714 ± 537 | 15.8 ± 13.2 |
| Type IIa (μm2) | 5052 ± 276 | 5817 ± 566 | 15.4 ± 9.8 | 4562 ± 236 | 5931 ± 579 * | 30.9 ± 11.8 |
| Type IIx (μm2) | 4240 ± 311 | 4966 ± 658 | 17.4 ± 14.0 | 4409 ± 342 | 5497 ± 477 * | 28.5 ± 9.9 |
| Fiber Type Distribution | ||||||
| Type I (%) | 43.0 ± 3.8 | 41.2 ± 4.5 | - | 46.3 ± 4.9 | 38.8 ± 2.6 | - |
| Type IIa (%) | 39.6 ± 3.8 | 42.6 ± 2.3 | - | 36.3 ± 5.0 | 35.1 ± 3.8 | - |
| Type IIx (%) | 17.3 ± 2.9 | 16.1 ± 3.5 | - | 17.4 ± 3.4 | 26.2 ± 3.6 * | - |
| Old Placebo + HBRE (O-P; n = 16) | Old Muscle5 + HBRE (O-M5; n = 16) | |||
|---|---|---|---|---|
| Pre | Post | Pre | Post | |
| Kidney Function | ||||
| Creatinine (μmol/L) | 95.7 ± 4.0 | 95.6 ± 4.3 | 91.1 ± 4.2 | 100.5 ± 5.9 * |
| Liver Function | ||||
| Bilirubin (μmol/L) | 14.2 ± 1.6 | 11.9 ± 1.5§ | 14.8 ± 2.0 | 13.3 ± 1.4 |
| Alanine Transaminase (U/L) | 22.4 ± 3.0 | 20.9 ± 2.2 | 20.3 ± 3.2 | 20.9 ± 2.3 |
| γ-glutamyltransferase (U/L) | 28.0 ± 4.3 | 26.3 ± 3.1 | 32.3 ± 5.3 | 31.0 ± 4.8 |
| Inflammation | ||||
| C-reactive Protein (mg/L) | 4.0 ± 2.0 | 3.5 ± 1.4 | 2.0 ± 0.5 | 2.7 ± 0.8 |
| Lipid Profile | ||||
| Cholesterol (mmol/L) | 3.9 ± 0.2 | 3.9 ± 0.2 | 4.4 ± 0.3 | 4.2 ± 0.3 |
| Triglycerides (mmol/L) | 1.1 ± 0.1 | 1.2 ± 0.1 | 1.5 ± 0.2 | 1.4 ± 0.2 |
| HDL Cholesterol (mmol/L) | 1.2 ± 0.1 | 1.2 ± 0.1 | 1.1 ± 0.1 | 1.1 ± 0.1 |
| LDL Cholesterol (mmol/L) | 2.1 ± 0.2 | 2.2 ± 0.2 | 2.6 ± 0.3 | 2.4 ± 0.3 |
| Non-HDL Cholesterol (mmol/L) | 2.7 ± 0.2 | 2.7 ± 0.2 | 3.3 ± 0.3 | 3.1 ± 0.3 |
| Total Cholesterol:HDL Ratio | 3.2 ± 0.2 | 3.3 ± 0.2 | 4.1 ± 0.3 § | 3.9 ± 0.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nilsson, M.I.; Mikhail, A.; Lan, L.; Di Carlo, A.; Hamilton, B.; Barnard, K.; Hettinga, B.P.; Hatcher, E.; Tarnopolsky, M.G.; Nederveen, J.P.; et al. A Five-Ingredient Nutritional Supplement and Home-Based Resistance Exercise Improve Lean Mass and Strength in Free-Living Elderly. Nutrients 2020, 12, 2391. https://doi.org/10.3390/nu12082391
Nilsson MI, Mikhail A, Lan L, Di Carlo A, Hamilton B, Barnard K, Hettinga BP, Hatcher E, Tarnopolsky MG, Nederveen JP, et al. A Five-Ingredient Nutritional Supplement and Home-Based Resistance Exercise Improve Lean Mass and Strength in Free-Living Elderly. Nutrients. 2020; 12(8):2391. https://doi.org/10.3390/nu12082391
Chicago/Turabian StyleNilsson, Mats I., Andrew Mikhail, Lucy Lan, Alessia Di Carlo, Bethanie Hamilton, Kristin Barnard, Bart P. Hettinga, Erin Hatcher, Milla G. Tarnopolsky, Joshua P. Nederveen, and et al. 2020. "A Five-Ingredient Nutritional Supplement and Home-Based Resistance Exercise Improve Lean Mass and Strength in Free-Living Elderly" Nutrients 12, no. 8: 2391. https://doi.org/10.3390/nu12082391
APA StyleNilsson, M. I., Mikhail, A., Lan, L., Di Carlo, A., Hamilton, B., Barnard, K., Hettinga, B. P., Hatcher, E., Tarnopolsky, M. G., Nederveen, J. P., Bujak, A. L., May, L., & Tarnopolsky, M. A. (2020). A Five-Ingredient Nutritional Supplement and Home-Based Resistance Exercise Improve Lean Mass and Strength in Free-Living Elderly. Nutrients, 12(8), 2391. https://doi.org/10.3390/nu12082391







