Lactobacillus plantarum PS128 Improves Physiological Adaptation and Performance in Triathletes through Gut Microbiota Modulation
Abstract
1. Introduction
2. Materials and Methods
2.1. Lactic Acid Bacteria
2.2. Participants
2.3. Experimental Design
2.4. VO2 Maximum and Endurance
2.5. Metagenome
2.6. Body Composition
2.7. Blood Sampling and CBC Analysis
2.8. Biochemical Variables
2.9. Short-Chain Fatty Acid (SCFA) Analysis
2.10. Statistics Comparison of Probiotic (Treatment) and Control Groups
3. Results
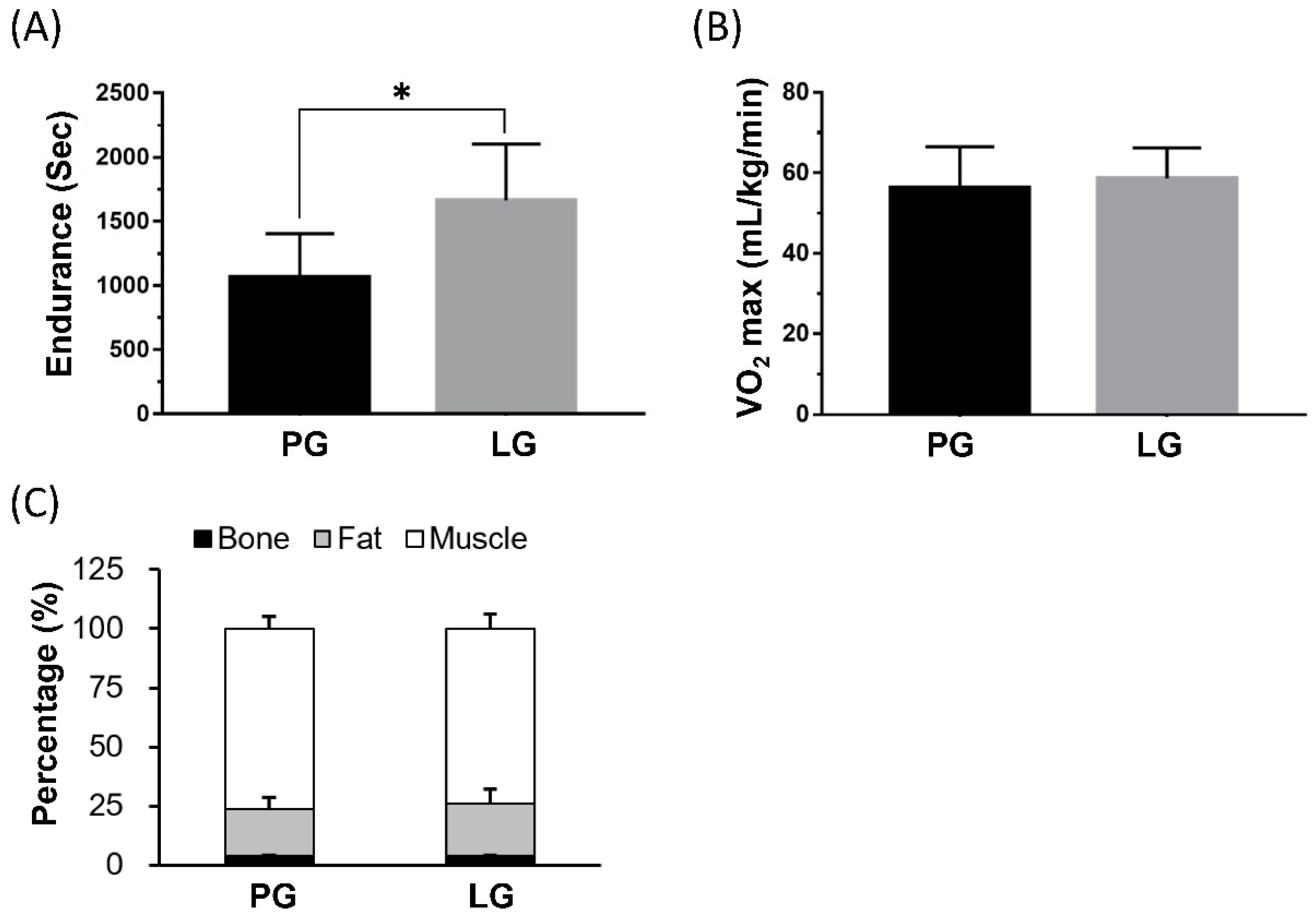
3.1. The Effects of PS128 on Exercise Performance and Body Composition
3.2. The Effects of PS128 on Biochemical Variables and Complete Blood Count
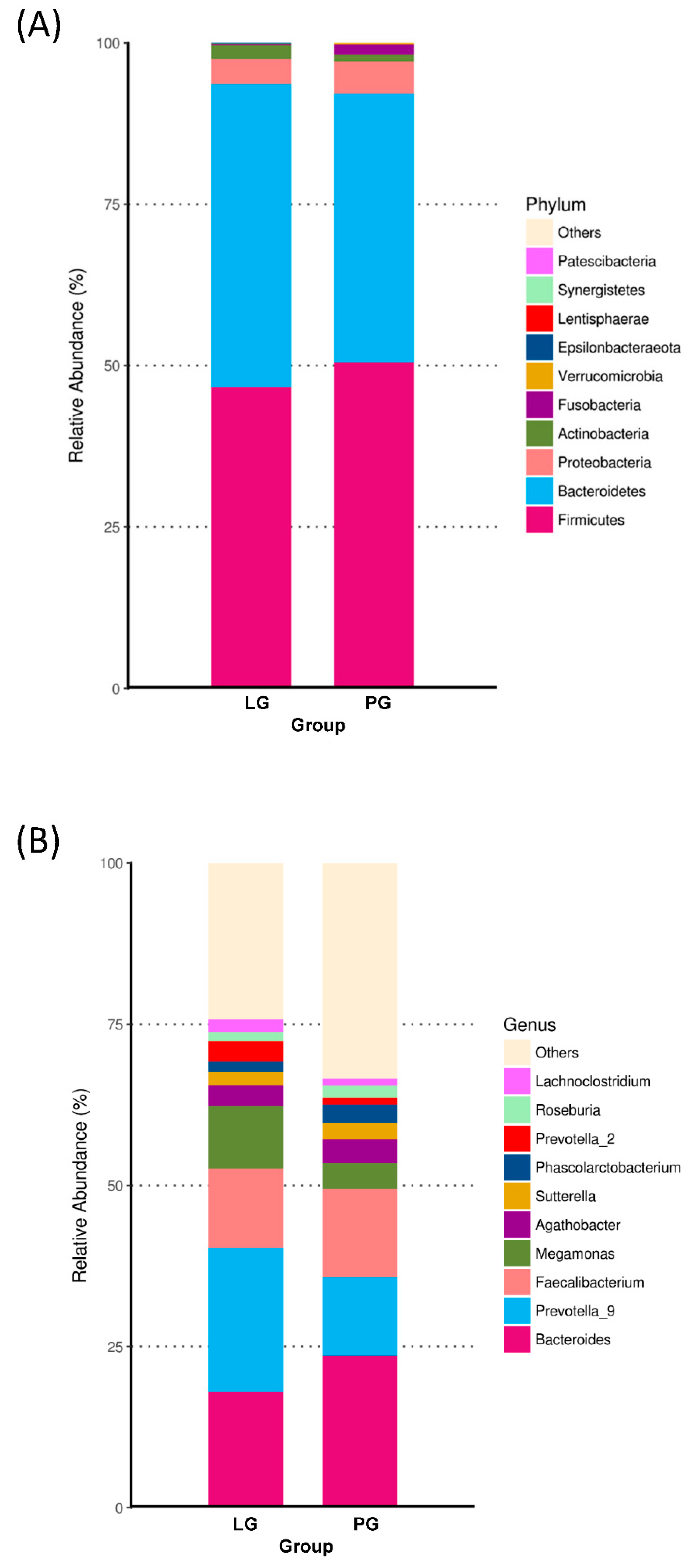
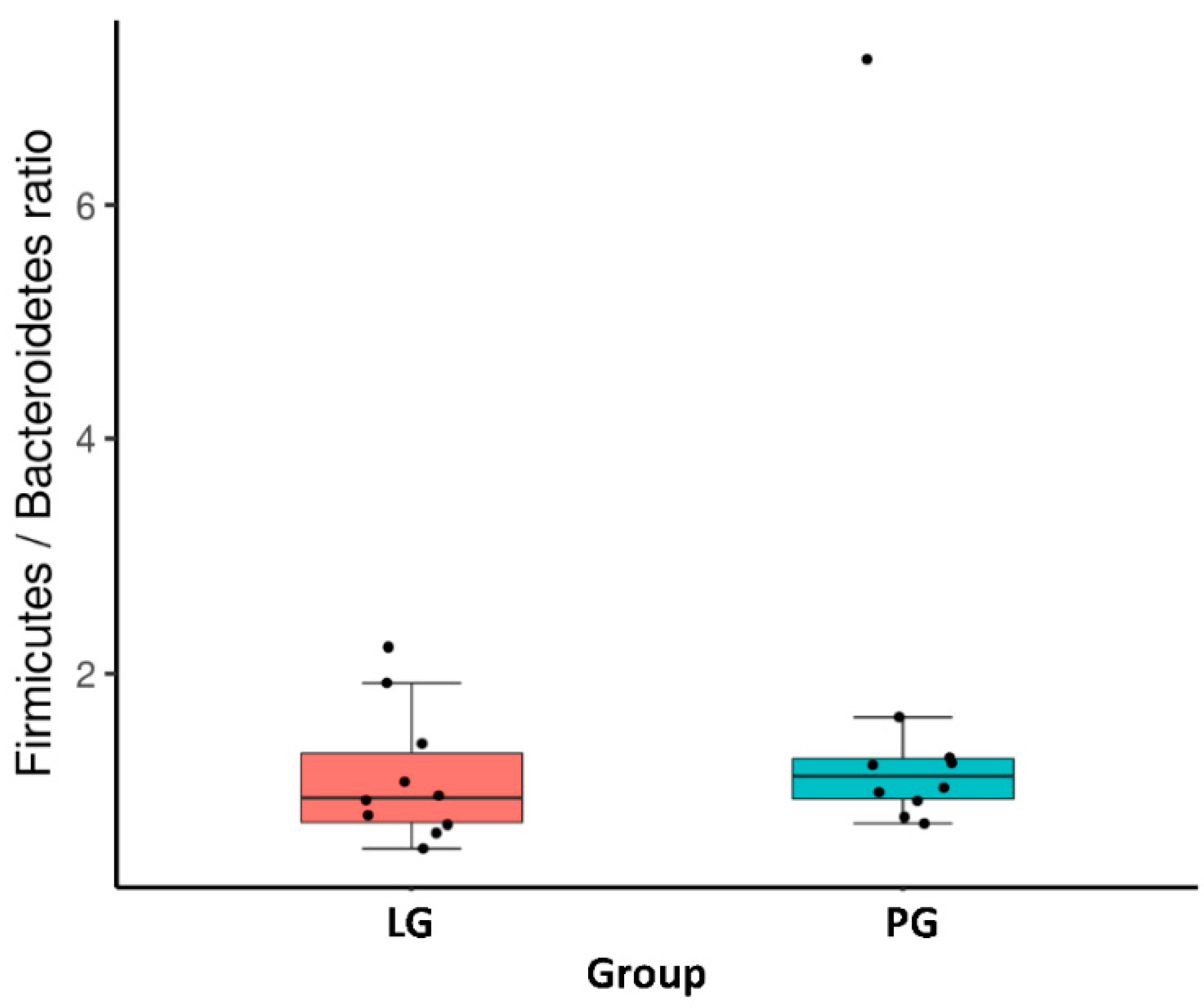
3.3. Changes of the Relative Abundance at the Phylum and Genus Level
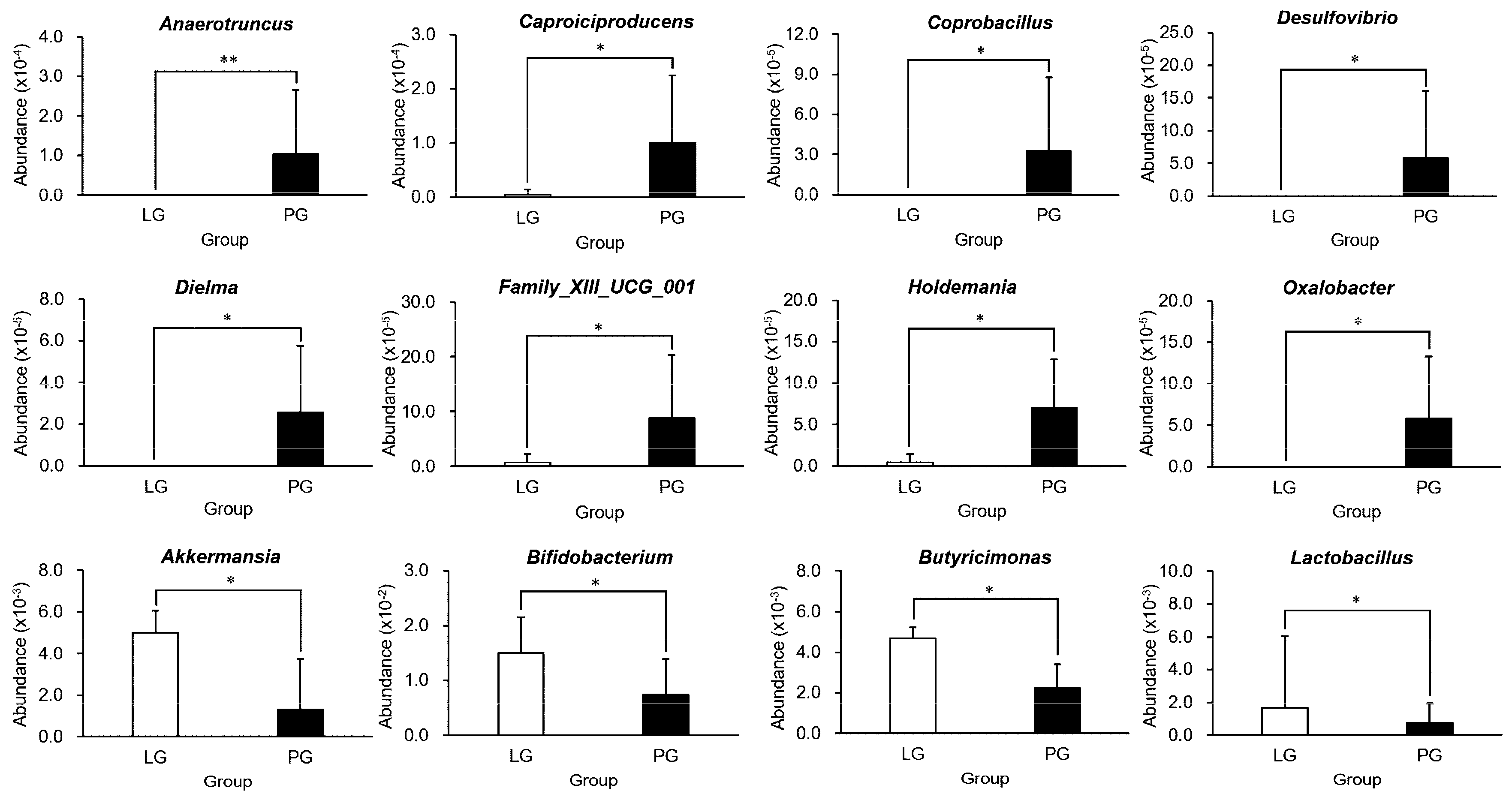
3.4. The Significant Difference of Microbiota between Groups at the Genus Level
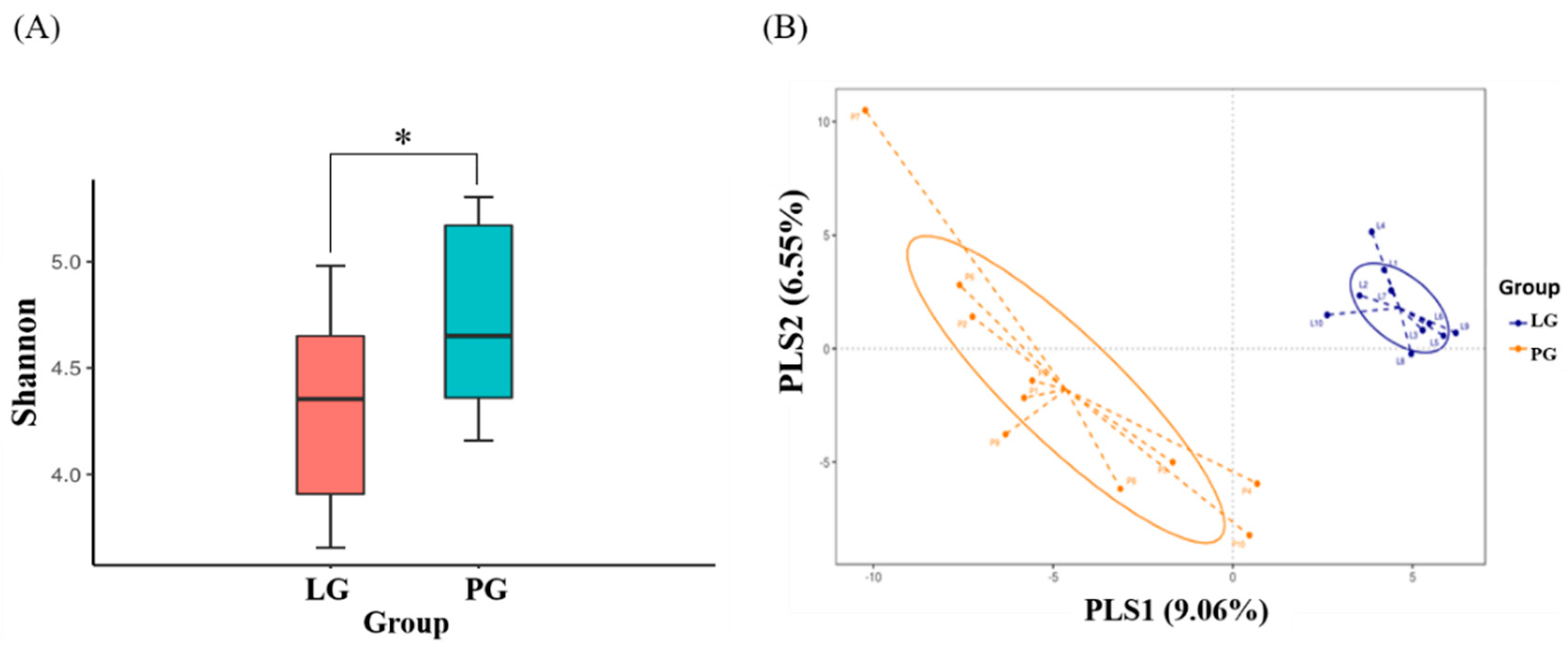
3.5. Changes of Fecal Microbial Diversity
3.6. The Effects of PS128 on Short-Chain Fatty Acids
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O′Toole, M.L.; Douglas, P.S.; Hiller, W.D. Applied physiology of a triathlon. Sports Med. 1989, 8, 201–225. [Google Scholar] [CrossRef] [PubMed]
- Kienstra, C.M.; Asken, T.R.; Garcia, J.D.; Lara, V.; Best, T.M. Triathlon injuries: Transitioning from prevalence to prediction and prevention. Curr. Sports Med. Rep. 2017, 16, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Jamurtas, A.Z. Exercise-induced muscle damage and oxidative stress. Antioxidants 2018, 7, E50. [Google Scholar] [CrossRef] [PubMed]
- Howatson, G.; Van Someren, K.A. The prevention and treatment of exercise-induced muscle damage. Sports Med. 2008, 38, 483–503. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Li, J.; Liu, Z.; Chuang, C.C.; Yang, W.; Zuo, L. Redox mechanism of reactive oxygen species in exercise. Front. Physiol. 2016, 7, 486. [Google Scholar] [CrossRef]
- Sugama, K.; Suzuki, K.; Yoshitani, K.; Shiraishi, K.; Miura, S.; Yoshioka, H.; Mori, Y.; Kometani, T. Changes of thioredoxin, oxidative stress markers, inflammation and muscle/renal damage following intensive endurance exercise. Exerc. Immunol. Rev. 2015, 21, 130–142. [Google Scholar]
- Jeukendrup, A.E. Nutrition for endurance sports: Marathon, triathlon, and road cycling. J. Sports Sci. 2011, 29, 91–99. [Google Scholar] [CrossRef]
- Nybo, L. CNS fatigue and prolonged exercise: Effect of glucose supplementation. Med. Sci. Sports Exerc. 2003, 35, 589–594. [Google Scholar] [CrossRef]
- Pyne, D.B.; West, N.P.; Cox, A.J.; Cripps, A.W. Probiotics supplementation for athletes—Clinical and physiological effects. Eur. J. Sport Sci. 2015, 15, 63–72. [Google Scholar] [CrossRef]
- Pane, M.; Amoruso, A.; Deidda, F.; Graziano, T.; Allesina, S.; Mogna, L. Gut microbiota, probiotics, and sport: From clinical evidence to agonistic performance. J. Clin. Gastroenterol. 2018, 52, S46–S49. [Google Scholar] [CrossRef]
- Ibrahim, N.S.; Ooi, F.K.; Chen, C.K.; Muhamad, A.S. Effects of probiotics supplementation and circuit training on immune responses among sedentary young males. J. Sports Med. Phys. Fit. 2018, 58, 1102–1109. [Google Scholar] [CrossRef]
- Martarelli, D.; Verdenelli, M.C.; Scuri, S.; Cocchioni, M.; Silvi, S.; Cecchini, C.; Pompei, P. Effect of a probiotic intake on oxidant and antioxidant parameters in plasma of athletes during intense exercise training. Curr. Microbiol. 2011, 62, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Hsu, Y.J.; Li, H.; Kan, N.W.; Chen, Y.M.; Lin, J.S.; Hsu, T.K.; Tsai, T.Y.; Chiu, Y.S.; Huang, C.C. Effect of Lactobacillus plantarum twk10 on improving endurance performance in humans. Chin. J. Physiol. 2018, 61, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Hsu, Y.J.; Chuang, H.L.; Hsieh, P.S.; Ho, H.H.; Chen, W.L.; Chiu, Y.S.; Huang, C.C. In vivo ergogenic properties of the bifidobacterium longum olp-01 isolated from a weightlifting gold medalist. Nutrients 2019, 11, E2003. [Google Scholar] [CrossRef]
- Khalesi, S.; Bellissimo, N.; Vandelanotte, C.; Williams, S.; Stanley, D.; Irwin, C. A review of probiotic supplementation in healthy adults: Helpful or hype? Eur. J. Clin. Nutr. 2019, 73, 24–37. [Google Scholar] [CrossRef]
- Kothari, D.; Patel, S.; Kim, S.K. Probiotic supplements might not be universally-effective and safe: A review. Biomed. Pharmacother. 2019, 111, 537–547. [Google Scholar] [CrossRef]
- Huang, W.C.; Wei, C.C.; Huang, C.C.; Chen, W.L.; Huang, H.Y. The beneficial effects of Lactobacillus plantarum ps128 on high-intensity, exercise-induced oxidative stress, inflammation, and performance in triathletes. Nutrients 2019, 11, E353. [Google Scholar] [CrossRef]
- Bruce, R.A.; Kusumi, F.; Hosmer, D. Maximal oxygen intake and nomographic assessment of functional aerobic impairment in cardiovascular disease. Am. Heart J. 1973, 85, 546–562. [Google Scholar] [CrossRef]
- Tung, Y.T.; Hsu, Y.J.; Liao, C.C.; Ho, S.T.; Huang, C.C.; Huang, W.C. Physiological and biochemical effects of intrinsically high and low exercise capacities through multiomics approaches. Front. Physiol. 2019, 10, 1201. [Google Scholar] [CrossRef]
- Bischoff, S.C. ‘Gut health’: A new objective in medicine? BMC Med. 2011, 9, 24. [Google Scholar] [CrossRef]
- Valdes, A.M.; Walter, J.; Segal, E.; Spector, T.D. Role of the gut microbiota in nutrition and health. BMJ 2018, 361, k2179. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.; Stilling, R.M.; Kennedy, P.J.; Stanton, C.; Cryan, J.F.; Dinan, T.G. Minireview: Gut microbiota: The neglected endocrine organ. Mol. Endocrinol. 2014, 28, 1221–1238. [Google Scholar] [CrossRef] [PubMed]
- Turris, S.A.; Lund, A.; Bowles, R.R.; Camporese, M.; Green, T. Patient presentations and medical logistics at full and half ironman distance triathlons. Curr. Sports Med. Rep. 2017, 16, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E. Training the gut for athletes. Sports Med. 2017, 47, 101–110. [Google Scholar] [CrossRef]
- Parker, E.A.; Roy, T.; D′Adamo, C.R.; Wieland, L.S. Probiotics and gastrointestinal conditions: An overview of evidence from the cochrane collaboration. Nutrients 2018, 45, 125–134. [Google Scholar] [CrossRef]
- Chen, L.; Li, H.; Li, J.; Chen, Y.; Yang, Y. Lactobacillus rhamnosus GG treatment improves intestinal permeability and modulates microbiota dysbiosis in an experimental model of sepsis. Int. J. Mol. Med. 2019, 43, 1139–1148. [Google Scholar] [CrossRef]
- Karl, J.P.; Margolis, L.M.; Madslien, E.H.; Murphy, N.E.; Castellani, J.W.; Gundersen, Y.; Hoke, A.V.; Levangie, M.W.; Kumar, R.; Chakraborty, N.; et al. Changes in intestinal microbiota composition and metabolism coincide with increased intestinal permeability in young adults under prolonged physiological stress. Am. J. Physiol. Gastrointest. Liver Physiol. 2017, 312, 559–571. [Google Scholar] [CrossRef]
- Pires, W.; Veneroso, C.E.; Wanner, S.P.; Pacheco, D.A.S.; Vaz, G.C.; Amorim, F.T.; Tonoli, C.; Soares, D.D.; Coimbra, C.C. Association between exercise-induced hyperthermia and intestinal permeability: A systematic review. Sports Med. 2017, 47, 1389–1403. [Google Scholar] [CrossRef]
- Liu, Y.W.; Liu, W.H.; Wu, C.C.; Juan, Y.C.; Wu, Y.C.; Tsai, H.P.; Wang, S.; Tsai, Y.C. Psychotropic effects of Lactobacillus plantarum PS128 in early life-stressed and naïve adult mice. Brain Res. 2016, 1631, 1–12. [Google Scholar] [CrossRef]
- Wang, J.; Ji, H.; Wang, S.; Liu, H.; Zhang, W.; Zhang, D.; Wang, Y. Probiotic Lactobacillus plantarum promotes intestinal barrier function by strengthening the epithelium and modulating gut microbiota. Front. Microbiol. 2018, 9, 1953. [Google Scholar] [CrossRef]
- Ahn, J.E.; Kim, H.; Chung, D.K. Lipoteichoic acid isolated from Lactobacillus plantarum maintains inflammatory homeostasis through regulation of Th1- and Th2-induced cytokines. J. Microbiol. Biotechnol. 2019, 29, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Canfora, E.E.; Jocken, J.W.; Blaak, E.E. Short-chain fatty acids in control of body weight and insulin sensitivity. Nat. Rev. Endocrinol. 2015, 11, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, T.; Morino, K.; Ugi, S.; Nakagawa, F.; Lemecha, M.; Ida, S.; Ohashi, N.; Sato, D.; Fujita, Y.; Maegawa, H. Microbiome potentiates endurance exercise through intestinal acetate production. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E956–E966. [Google Scholar] [CrossRef] [PubMed]
- Waller, A.P.; Geor, R.J.; Spriet, L.L.; Heigenhauser, G.J.; Lindinger, M.I. Oral acetate supplementation after prolonged moderate intensity exercise enhances early muscle glycogen resynthesis in horses. Exp. Physiol. 2009, 94, 888–898. [Google Scholar] [CrossRef]
- Hernández, M.A.G.; Canfora, E.E.; Jocken, J.W.E.; Blaak, E.E. The short-chain fatty acid acetate in body weight control and insulin sensitivity. Nutrients 2019, 11, E1943. [Google Scholar] [CrossRef]
- Iebba, V.; Totino, V.; Gagliardi, A.; Santangelo, F.; Cacciotti, F.; Trancassini, M.; Mancini, C.; Cicerone, C.; Corazziari, E.; Pantanella, F.; et al. Eubiosis and dysbiosis: The two sides of the microbiota. New Microbiol. 2016, 39, 1–12. [Google Scholar]
- Markowiak, P.; Śliżewska, K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Denou, E.; Marcinko, K.; Surette, M.G.; Steinberg, G.R.; Schertzer, J.D. High-intensity exercise training increases the diversity and metabolic capacity of the mouse distal gut microbiota during diet-induced obesity. Am. J. Physiol. Endocrinol. Metab. 2016, 310, E982–E993. [Google Scholar] [CrossRef]
- Mörkl, S.; Lackner, S.; Müller, W.; Gorkiewicz, G.; Kashofer, K.; Oberascher, A.; Painold, A.; Holl, A.; Holzer, P.; Meinitzer, A.; et al. Gut microbiota and body composition in anorexia nervosa inpatients in comparison to athletes, overweight, obese, and normal weight controls. Int. J. Eat. Disord. 2017, 50, 1421–1431. [Google Scholar] [CrossRef]
- Jäger, R.; Mohr, A.E.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Moussa, A.; Townsend, J.R.; Lamprecht, M.; West, N.P.; Black, K.; et al. International society of sports nutrition position stand: Probiotics. J. Int. Soc. Sports Nutr. 2019, 16, 62. [Google Scholar] [CrossRef]
- Shade, A. Diversity is the question, not the answer. ISME J. 2017, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Figliuolo, V.R.; Dos Santos, L.M.; Abalo, A.; Nanini, H.; Santos, A.; Brittes, N.M.; Bernardazzi, C.; de Souza, H.S.P.; Vieira, L.Q.; Coutinho-Silva, R.; et al. Sulfate-reducing bacteria stimulate gut immune responses and contribute to inflammation in experimental colitis. Life Sci. 2017, 189, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Terzo, S.; Mulè, F.; Caldara, G.F.; Baldassano, S.; Puleio, R.; Vitale, M.; Cassata, G.; Ferrantelli, V.; Amato, A. Pistachio consumption alleviates inflammation and improves gut microbiota composition in mice fed a high-fat diet. Int. J. Mol. Sci. 2020, 21, E365. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, T.; Alim, A.; Ren, D.; Zhao, Y.; Yang, X. Regulatory effects of stachyose on colonic and hepatic inflammation, gut microbiota dysbiosis, and peripheral CD4(+) T cell distribution abnormality in high-fat diet-fed mice. J. Agric. Food Chem. 2019, 67, 11665–11674. [Google Scholar] [CrossRef]
- Lun, H.; Yang, W.; Zhao, S.; Jiang, M.; Xu, M.; Liu, F.; Wang, Y. Altered gut microbiota and microbial biomarkers associated with chronic kidney disease. Microbiologyopen 2019, 8, e00678. [Google Scholar] [CrossRef]
- Ouyang, J.; Lin, J.; Isnard, S.; Fombuena, B.; Peng, X.; Marette, A.; Routy, B.; Messaoudene, M.; Chen, Y.; Routy, J.P. The bacterium akkermansia muciniphila: A sentinel for gut permeability and its relevance to HIV-related inflammation. Front. Immunol. 2020, 11, 645. [Google Scholar] [CrossRef]
- Zheng, N.; Gao, Y.; Zhu, W.; Meng, D.; Walker, W.A. Short chain fatty acids produced by colonizing intestinal commensal bacterial interaction with expressed breast milk are anti-inflammatory in human immature enterocytes. PLoS ONE 2020, 15, e0229283. [Google Scholar] [CrossRef]
- Maltz, R.M.; Keirsey, J.; Kim, S.C.; Mackos, A.R.; Gharaibeh, R.Z.; Moore, C.C.; Xu, J.; Somogyi, A.; Bailey, M.T. Social stress affects colonic inflammation, the gut microbiome, and short-chain fatty acid levels and receptors. J. Pediatr. Gastroenterol. Nutr. 2019, 68, 533–540. [Google Scholar] [CrossRef]
- Xia, T.; Zhang, B.; Li, S.; Fang, B.; Duan, W.; Zhang, J.; Song, J.; Wang, M. Vinegar extract ameliorates alcohol-induced liver damage associated with the modulation of gut microbiota in mice. Food Funct. 2020, 11, 2898–2909. [Google Scholar] [CrossRef]
- Batacan, R.B., Jr.; Duncan, M.J.; Dalbo, V.J.; Tucker, P.S.; Fenning, A.S. Effects of high-intensity interval training on cardiometabolic health: A systematic review and meta-analysis of intervention studies. Br. J. Sports Med. 2017, 51, 494–503. [Google Scholar] [CrossRef]
- Held, S.; Behringer, M.; Donath, L. Low intensity rowing with blood flow restriction over 5 weeks increases VO2max in elite rowers: A randomized controlled trial. J. Sci. Med. Sport 2020, 23, 304–308. [Google Scholar] [CrossRef]
- Zinn, C.; Wood, M.; Williden, M.; Chatterton, S.; Maunder, E. Ketogenic diet benefits body composition and well-being but not performance in a pilot case study of New Zealand endurance athletes. J. Int. Soc. Sports Nutr. 2017, 14, 22. [Google Scholar] [CrossRef] [PubMed]
- Menon, A.S.; Anayath, S.; Garg, M.K.; Kapoor, R.; Pisharody, I. The effect of vitamin D supplementation on cardiorespiratory fitness and muscle strength in male adults undergoing basic military training. Med. J. Armed Forces India. 2020, 76, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Brüssow, H. Probiotics and prebiotics in clinical tests: An update. F1000Res. 2019, 8, 1157. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.; Shoenfeld, Y.; Matthias, T. Probiotics: If it does not help it does not do any harm. Really? Microorganisms 2019, 7, 104. [Google Scholar] [CrossRef]
- West, N.P.; Horn, P.L.; Pyne, D.B.; Gebski, V.J.; Lahtinen, S.J.; Fricker, P.A.; Cripps, A.W. Probiotic supplementation for respiratory and gastrointestinal illness symptoms in healthy physically active individuals. Clin. Nutr. 2014, 33, 581–587. [Google Scholar] [CrossRef]

| Parameters | PG | LG | p-Value |
|---|---|---|---|
| Albumin (g/dL) | 4.7 ± 0.2 | 4.8 ± 0.2 | 0.691 |
| Alk-P (U/L) | 70.6 ± 21.3 | 76.7 ± 18.7 | 0.505 |
| BUN (mg/dL) | 16.5 ± 3.2 | 14.8 ± 2.7 | 0.072 |
| Creatinine (mg/dL) | 1.1 ± 0.1 | 1.01 ± 0.1 | 0.109 |
| Free Fatty acid (ng/mL) | 11.0 ± 2.8 | 11.7 ± 2.3 | 0.561 |
| AST (U/L) | 25.7 ± 14.5 | 21.8 ± 4.3 | 0.970 |
| ALT (U/L) | 13.9 ± 6.2 | 12.5 ± 2.8 | 0.970 |
| HDL Cholesterol (mg/dL) | 50.6 ± 4.7 | 50.7 ± 5.1 | 0.964 |
| LDH (U/L) | 268.8 ± 64.9 | 270.6 ± 50.5 | 0.946 |
| LDL Cholesterol (mg/dL) | 101.9 ± 17.8 | 107.5 ± 28.0 | 0.600 |
| Serum AC Sugar (mg/dL) | 82.1 ± 14.6 | 76.3 ± 20.2 | 0.471 |
| T-Cho/HDL-Cho | 3.3 ± 0.5 | 3.4 ± 0.6 | 0.744 |
| Total Cholesterol (mg/dL) | 166.9 ± 18.0 | 173.0 ± 34.8 | 0.629 |
| Total Protein (g/dL) | 7.2 ± 0.4 | 7.3 ± 0.5 | 0.582 |
| Triglyceride (mg/dL) | 111.9 ± 46.1 | 107.5 ± 50.1 | 0.840 |
| Uric Acid (mg/dL) | 5.8 ± 1.1 | 5.9 ± 1.0 | 0.833 |
| Parameters | PG | LG | p-Value |
|---|---|---|---|
| Hb (g/dL) | 15.1 ± 1.6 | 14.8 ± 1.3 | 0.595 |
| Hct (%) | 15.0 ± 4.5 | 44.3 ± 3.9 | 0.571 |
| MCH (pg) | 30.4 ± 1.7 | 30.5 ± 1.2 | 0.988 |
| MCHC (g/dL) | 33.4 ± 0.4 | 33.4 ± 0.2 | 1.000 |
| MCV (fL) | 90.9 ± 4.4 | 92.1 ± 4.0 | 0.632 |
| WBCs (103/μL) | 8.5 ± 2.0 | 8.5 ± 2.2 | 0.731 |
| Basophil (%) | 0.8 ± 0.2 | 0.9 ± 0.3 | 0.349 |
| Eosinophil (%) | 4.4 ± 2.7 | 3.4 ± 2.6 | 0.571 |
| Lymphocyte (%) | 21.0 ± 5.4 | 21.2 ± 4.8 | 0.982 |
| Monocyte (%) | 8.3 ± 2.1 | 8.9 ± 3.4 | 0.674 |
| Neutrophil (%) | 65.5 ± 7.9 | 65.6 ± 7.0 | 0.985 |
| Platelet (103/μL) | 247.2 ± 32.7 | 289.6 ± 50.4 | 0.049 |
| RBCs (106/μL) | 5.0 ± 0.5 | 4.9 ± 0.5 | 0.649 |
| NLR | 3.4 ± 1.4 | 3.3 ± 1.2 | 0.940 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, W.-C.; Pan, C.-H.; Wei, C.-C.; Huang, H.-Y. Lactobacillus plantarum PS128 Improves Physiological Adaptation and Performance in Triathletes through Gut Microbiota Modulation. Nutrients 2020, 12, 2315. https://doi.org/10.3390/nu12082315
Huang W-C, Pan C-H, Wei C-C, Huang H-Y. Lactobacillus plantarum PS128 Improves Physiological Adaptation and Performance in Triathletes through Gut Microbiota Modulation. Nutrients. 2020; 12(8):2315. https://doi.org/10.3390/nu12082315
Chicago/Turabian StyleHuang, Wen-Ching, Chun-Hsu Pan, Chen-Chan Wei, and Hui-Yu Huang. 2020. "Lactobacillus plantarum PS128 Improves Physiological Adaptation and Performance in Triathletes through Gut Microbiota Modulation" Nutrients 12, no. 8: 2315. https://doi.org/10.3390/nu12082315
APA StyleHuang, W.-C., Pan, C.-H., Wei, C.-C., & Huang, H.-Y. (2020). Lactobacillus plantarum PS128 Improves Physiological Adaptation and Performance in Triathletes through Gut Microbiota Modulation. Nutrients, 12(8), 2315. https://doi.org/10.3390/nu12082315





