Exploring Health-Promoting Attributes of Plant Proteins as a Functional Ingredient for the Food Sector: A Systematic Review of Human Interventional Studies
Abstract
1. Introduction
Study Aim
2. Materials and Methods
2.1. Search Strategy and Data Sources
2.2. Study Eligibility Criteria
2.3. Study Selection Process
3. Results
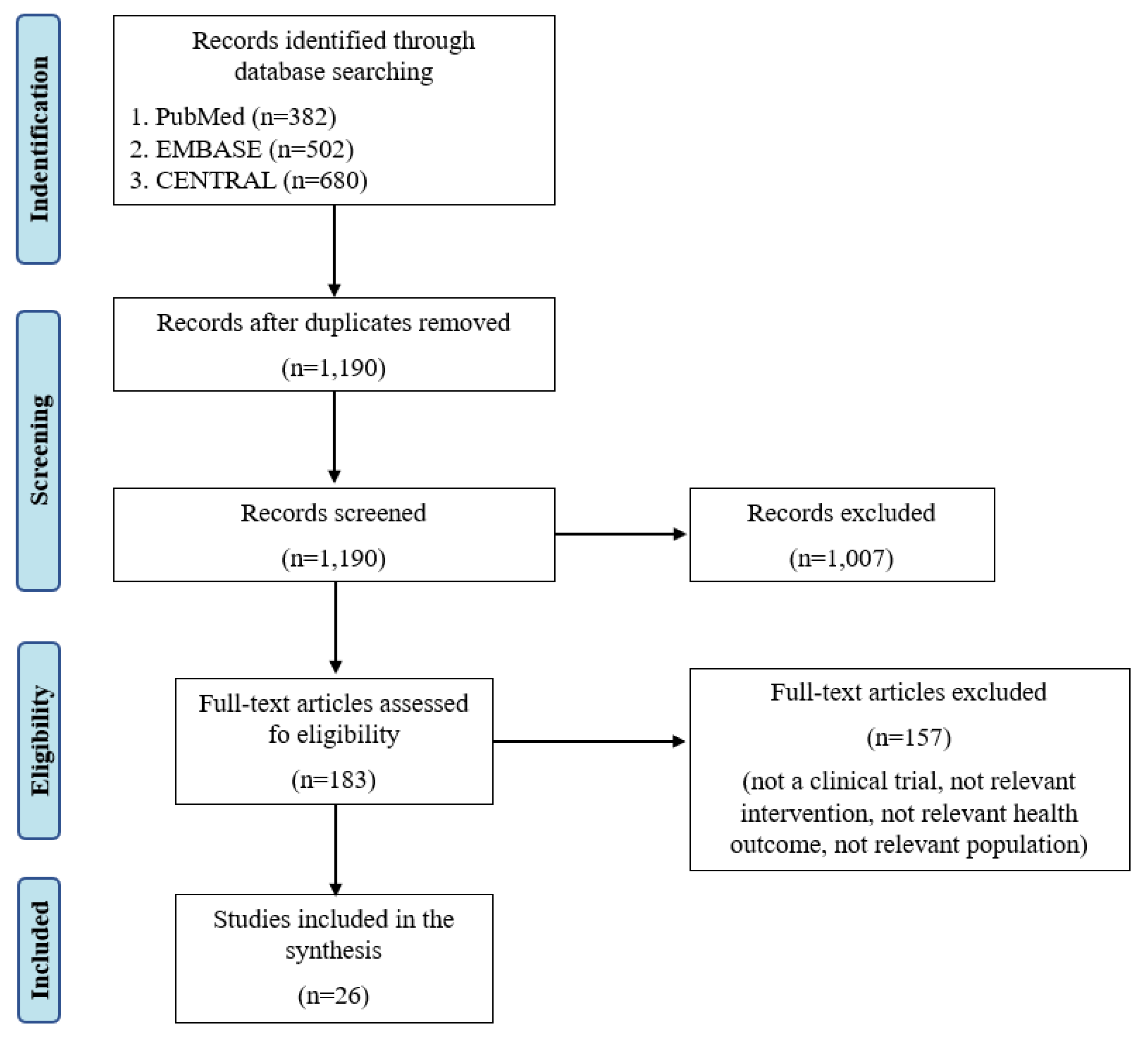
3.1. Search Results
3.2. Trials’ Characteristics
3.3. Satiety, Glycemic and Insulinemic Responses
3.4. Blood Lipids
3.5. Blood Pressure
3.6. Muscle Health
3.7. Body Weight
3.8. Risk of Bias and Quality of the Studies
4. Discussion
5. Industry Perspective—Invited Commentary
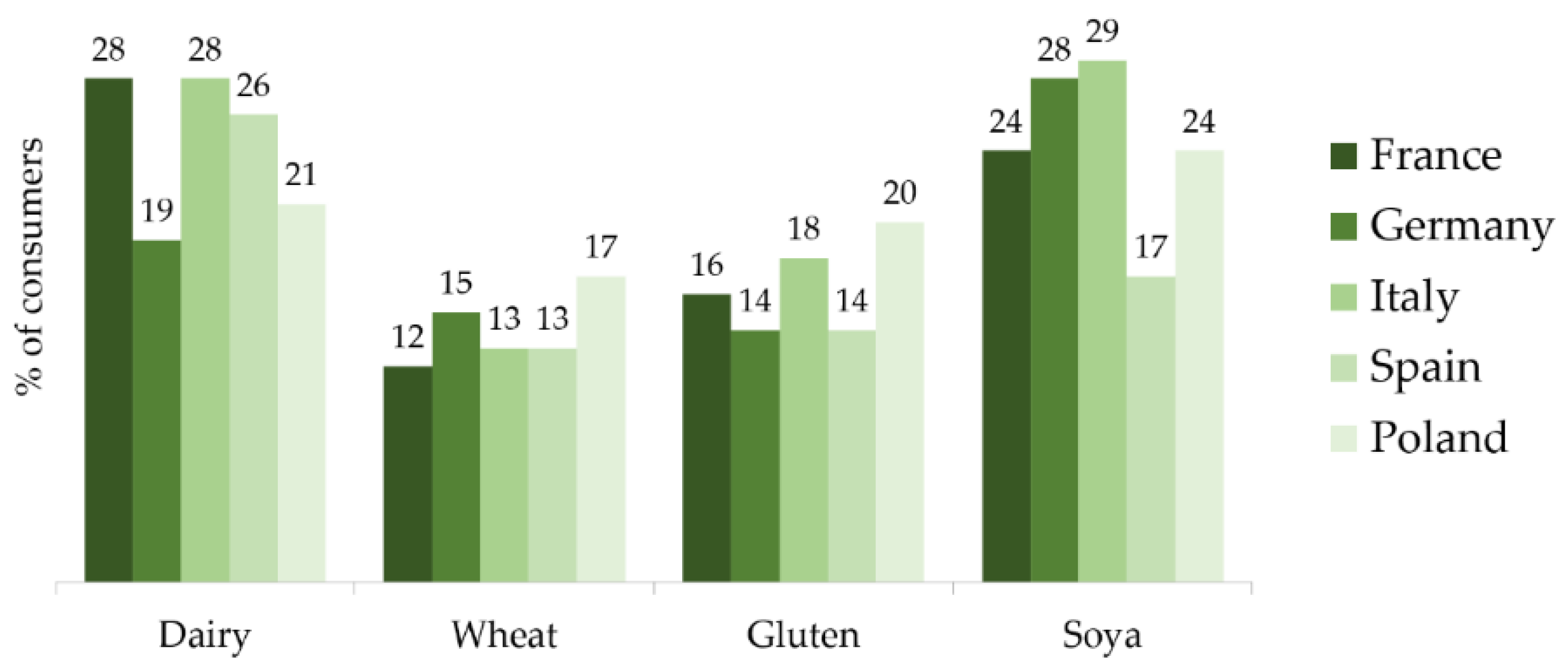
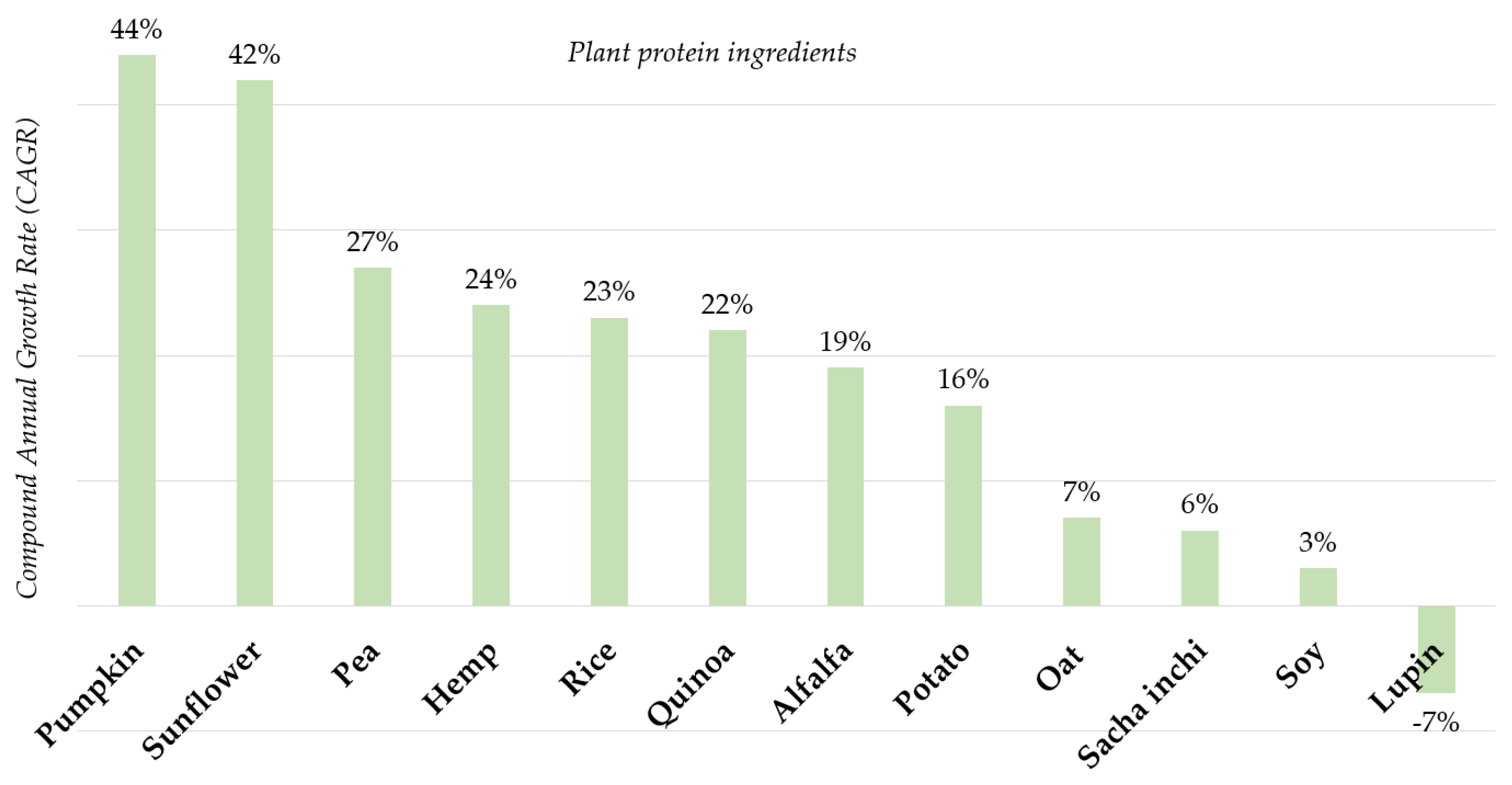
5.1. Changing Consumer Preferences
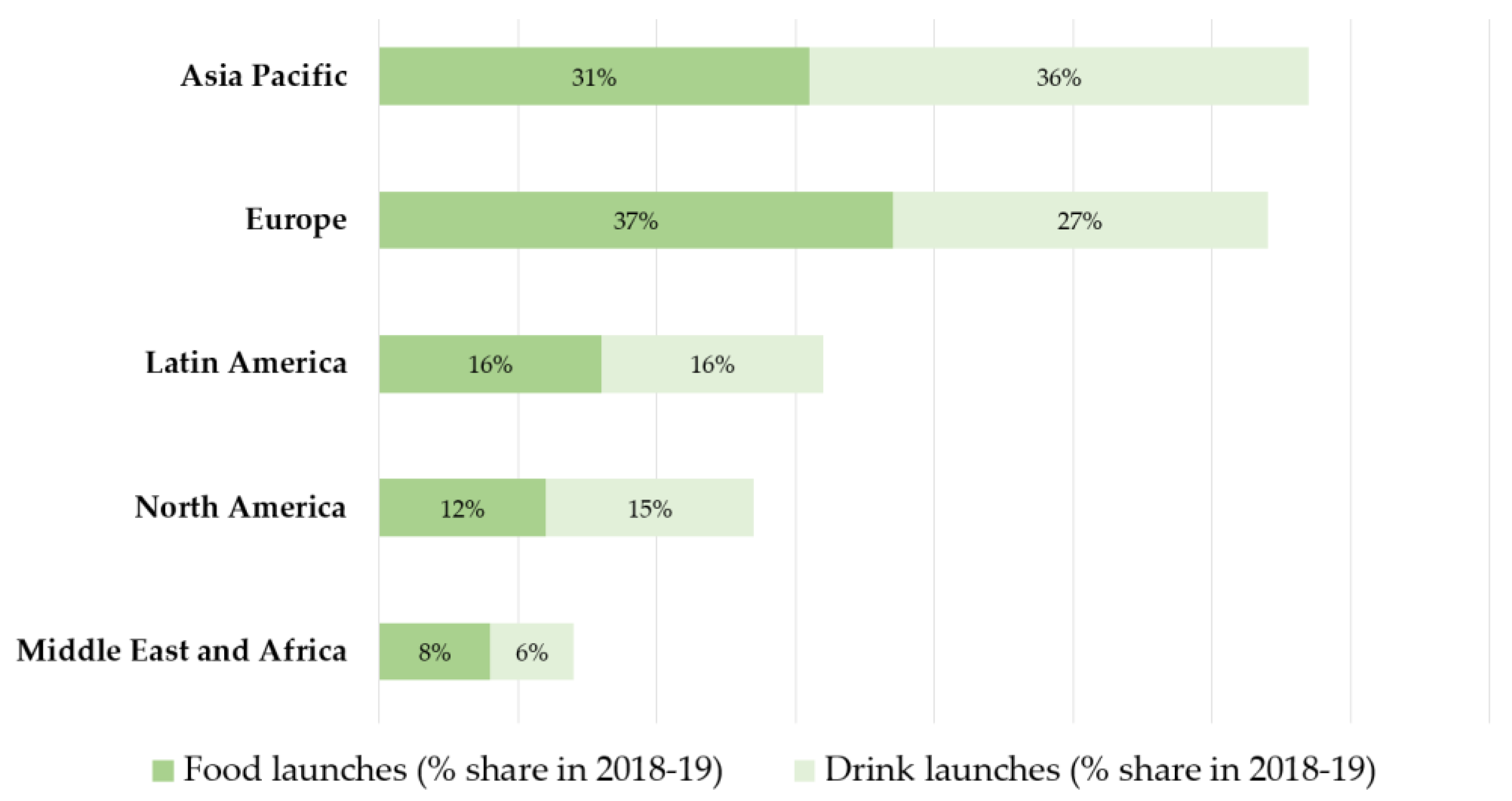
5.2. How Has the Market Responded?
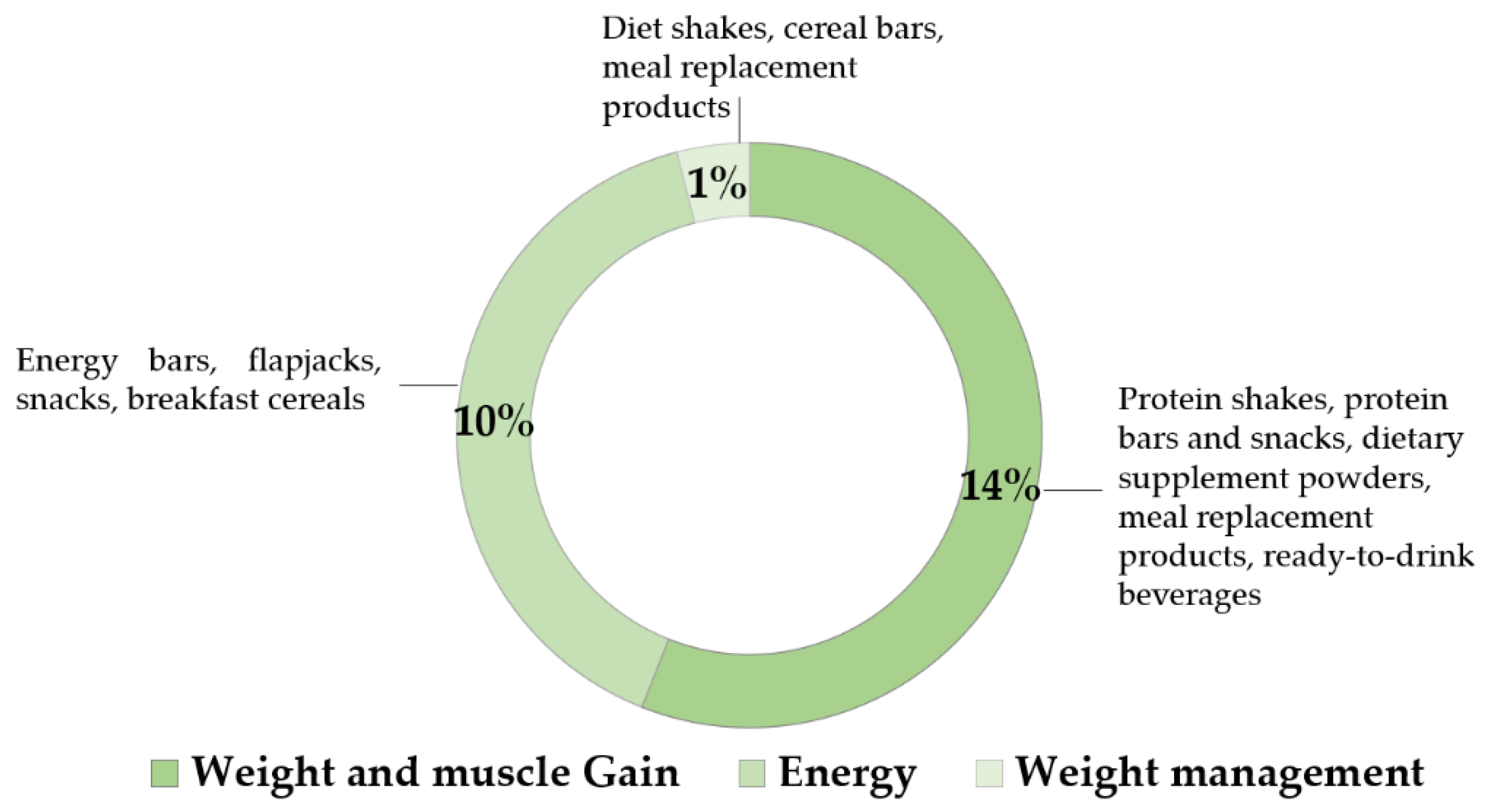
5.3. Innovation in Plant Protein Ingredients Is Changing
5.4. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wu, G.; Fanzo, J.; Miller, D.D.; Pingali, P.; Post, M.; Steiner, J.L.; Thalacker-Mercer, A.E. Production and supply of high-quality food protein for human consumption: Sustainability, challenges, and innovations. Ann. N. Y. Acad. Sci. 2014, 1321, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change through Livestock—A Global Assessment of Emissions and Mitigation Opportunities; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013; p. 115. [Google Scholar]
- Gardner, C.D.; Hartle, J.C.; Garrett, R.D.; Offringa, L.C.; Wasserman, A.S. Maximizing the intersection of human health and the health of the environment with regard to the amount and type of protein produced and consumed in the United States. Nutr. Rev. 2019, 77, 197–215. [Google Scholar] [CrossRef] [PubMed]
- MacDiarmid, J.I.; Kyle, J.; Horgan, G.W.; Loe, J.; Fyfe, C.; Johnstone, A.; McNeill, G. Sustainable diets for the future: Can we contribute to reducing greenhouse gas emissions by eating a healthy diet? Am. J. Clin. Nutr. 2012, 96, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Shu, L.; Zheng, P.F.; Zhang, X.Y.; Si, C.J.; Yu, X.L.; Gao, W.; Zhang, L.; Liao, D. Association between Dietary Patterns and the Indicators of Obesity among Chinese: A Cross-Sectional Study. Nutrients 2015, 7, 7995–8009. [Google Scholar] [CrossRef]
- Jannasch, F.; Kröger, J.; Schulze, M.B. Dietary Patterns and Type 2 Diabetes: A Systematic Literature Review and Meta-Analysis of Prospective Studies. J. Nutr. 2017, 147, 1174–1182. [Google Scholar] [CrossRef]
- Shimazu, T.; Kuriyama, S.; Hozawa, A.; Ohmori, K.; Sato, Y.; Nakaya, N.; Nishino, Y.; Tsubono, Y.; Tsuji, I. Dietary patterns and cardiovascular disease mortality in Japan: A prospective cohort study. Int. J. Epidemiol. 2007, 36, 600–609. [Google Scholar] [CrossRef]
- Rosato, V.; Edefonti, V.; Bravi, F.; Bosetti, C.; Bertuccio, P.; Talamini, R.; Dal Maso, L.; Montella, M.; Ferraroni, M.; La Vecchia, C.; et al. Nutrient-based dietary patterns and prostate cancer risk: A case-control study from Italy. Cancer Causes Control. 2014, 25, 525–532. [Google Scholar] [CrossRef]
- World Cancer Research Fund/American Institute for Cancer Research (WCRF/AICR). Diet, Nutrition, Physical Activity and Cancer: A Global Perspective. Available online: https://www.wcrf.org/dietandcancer/contents (accessed on 6 May 2020).
- Larsson, S.C.; Orsini, N. Red meat and processed meat consumption and all-cause mortality: A meta-analysis. Am. J. Epidemiol. 2014, 17, 282–289. [Google Scholar] [CrossRef]
- Abete, I.; Romaguera, D.; Vieira, A.R.; Lopez de Munain, A.; Norat, T. Association between total, processed, red and white meat consumption and all-cause, CVD and IHD mortality: A meta-analysis of cohort studies. Br. J. Nutr. 2014, 112, 762–775. [Google Scholar] [CrossRef]
- Yang, C.; Pan, L.; Sun, C.; Xi, Y.; Wang, L.; Li, D. Red meat consumption and the risk of stroke: A dose-response meta-analysis of prospective cohort studies. J. Stroke Cereb. Dis. 2016, 25, 1177–1186. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lin, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G.; Pan, A.; Hu, F.B. Red and processed meat consumption and mortality: Dose-response meta-analysis of prospective cohort studies. Public Health Nutr. 2016, 19, 893–905. [Google Scholar] [CrossRef] [PubMed]
- Cui, K.; Liu, Y.; Zhu, L.; Mei, X.; Jin, P.; Luo, Y. Association between intake of red and processed meat and the risk of heart failure: A meta-analysis. BMC Public Health 2019, 19, 354. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Li, Y.; Satija, A.; Pan, A.; Sotos-Prieto, M.; Rimm, E.; Willett, W.C.; Hu, F.B. Association of changes in red meat consumption with total and cause specific mortality among US women and men: Two prospective cohort studies. BMJ 2019, 365, l2110. [Google Scholar] [CrossRef] [PubMed]
- Zhong, V.W.; Van Horn, L.; Greenland, P.; Carnethon, M.R.; Wilkins, J.T.; Lloyd-Jones, M.D.; Allen, N.B. Associations of Processed Meat, Unprocessed Red Meat, Poultry, or Fish Intake With Incident Cardiovascular Disease and All-Cause Mortality. JAMA Intern. Med. 2020, 180, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Mintel. Patent Watch: Next-Gen Plant Protein Ingredients. March 2020. [Google Scholar]
- Mintel. Attitudes towards Healthy Eating, UK. February 2020. [Google Scholar]
- Mintel. Flexitarian Parents Look for More Plant-Based Baby Food. March 2020. [Google Scholar]
- Yu, J.; Ahmedna, M. Functional components of grape pomace: Their composition, biological properties and potential applications. Int. J. Food Sci. Technol. 2013, 48, 221–237. [Google Scholar] [CrossRef]
- Nadathur, S.R.; Wanasundara, J.P.D.; Scanlin, L. Sustainable Protein Sources; Elsevier: London, UK, 2017; pp. 1–9. [Google Scholar]
- Whittaker, R.H. New concepts of kingdoms or organisms. Evolutionary relations are better represented by new classifications than by the traditional two kingdoms. Science 1969, 163, 150–160. [Google Scholar] [CrossRef]
- Kim, S.J.; De Souza, R.J.; Choo, V.L.; Ha, V.; Cozma, A.I.; Chiavaroli, L.; Sievenpiper, J.L. Effects of dietary pulse consumption on body weight: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2016, 103, 1213–1223. [Google Scholar] [CrossRef]
- Jenkins, D.J.; Kendall, C.W.; Marchie, A.; Faulkner, D.; Vidgen, E.; Lapsley, K.G.; Connelly, P.W. The effect of combining plant sterols, soy protein, viscous fibers, and almonds in treating hypercholesterolemia. Metabolism 2003, 52, 1478–1483. [Google Scholar] [CrossRef]
- Sievenpiper, J.L.; Kendall, C.W.; Esfahani, A.; Wong, J.M.; Carleton, A.J.; Jiang, H.Y.; Jenkins, D.J. Effect of non-oil-seed pulses on glycaemic control: A systematic review and meta-analysis of randomised controlled experimental trials in people with and without diabetes. Diabetologia 2009, 52, 1479–1495. [Google Scholar] [CrossRef]
- Jayalath, V.H.; De Souza, R.J.; Sievenpiper, J.L.; Ha, V.; Chiavaroli, L.; Mirrahimi, A.; Jenkins, D.J. Effect of dietary pulses on blood pressure: A systematic review and meta-analysis of controlled feeding trials. Am. J. Hypertens. 2014, 27, 56–64. [Google Scholar] [CrossRef]
- Bazzano, L.A.; Thompson, A.M.; Tees, M.T.; Nguyen, C.H.; Winham, D.M. Non-soy legume consumption lowers cholesterol levels: A meta-analysis of randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Rebello, C.; Greenway, F.L.; Dhurandhar, N.V. Functional foods to promote weight loss and satiety. Curr. Opin. Clin. Nutr. Metab. Care 2014, 17, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Neacsu, M.; Fyfe, C.; Horgan, G.; Johnstone, A.M. Appetite control and biomarkers of satiety with vegetarian (soy) and meat-based high-protein diets for weight loss in obese men: A randomized crossover trial. Am. J. Clin. Nutr. 2014, 100, 548–558. [Google Scholar] [CrossRef] [PubMed]
- Scully, A.; Neacsu, M.; Ruddel, W.; Vaughan, N.; Fyfe, C.; Hudson, K.; Taylor, K.; Johnstone, A.M. Plant protein influence on appetite and food intake in healthy subjects. Proc. Nutr. Soc. 2017, 76, E44. [Google Scholar] [CrossRef]
- Van Vliet, S.; Burd, N.A.; Van Loon, L.J. The Skeletal Muscle Anabolic Response to Plant- versus Animal-Based Protein Consumption. J. Nutr. 2015, 145, 1981–1991. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Witard, O.C. Characterising the muscle anabolic potential of dairy, meat and plant-based protein sources in older adults. Proc. Nutr. Soc. 2018, 77, 20–31. [Google Scholar] [CrossRef]
- CoFID McCance and Widdowson’s Composition of Foods Integrated Dataset. Available online: https://www.gov.uk/government/publications/composition-of-foods-integrated-dataset-cofid (accessed on 14 July 2020).
- Messina, M. Soy and Health Update: Evaluation of the Clinical and Epidemiologic Literature. Nutrients 2016, 8, 754. [Google Scholar] [CrossRef]
- Crowe, K.M.; Francis, C. Position of the academy of nutrition and dietetics: Functional foods. J. Acad. Nutr. Diet 2013, 113, 1096–1103. [Google Scholar] [CrossRef]
- Schaafsma, G. Safety of protein hydrolysates, fractions thereof and bioactive peptides in human nutrition. Eur. J. Clin. Nutr. 2009, 63, 1161–1168. [Google Scholar] [CrossRef]
- Higgins, J.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Version 5.1.0; The Cochrane Collaboration: London, UK, 2017; p. 265. [Google Scholar]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. J. Clin. Epidemiol. 2009, 62, e1000097. [Google Scholar] [CrossRef]
- Centre for Reviews and Dissemination. Systematic Reviews: CRD’s Guidance for Undertaking Reviews in Health Care; University of York: New York, NY, USA, 2006; p. 294. [Google Scholar]
- Mollard, R.C.; Luhovyy, B.L.; Smith, C.; Anderson, G.H. Acute effects of pea protein and hull fibre alone and combined on blood glucose, appetite, and food intake in healthy young men—A randomized crossover trial. Appl. Physiol. Nutr. Metab. 2014, 39, 1360–1365. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.E.; Mollard, R.C.; Luhovyy, B.L.; Anderson, G.H. The effect of yellow pea protein and fibre on short-term food intake, subjective appetite and glycaemic response in healthy young men. Br. J. Nutr. 2012, 108, S74–S80. [Google Scholar] [CrossRef] [PubMed]
- Contaldo, F.; Di Biase, G.; Giacco, A. Evaluation of the hypocholesterolemic effect of vegetable proteins. Prev. Med. 1983, 12, 138–143. [Google Scholar] [CrossRef]
- Li, H.; Prairie, N.; Udenigwe, C.C.; Adebiyi, A.P.; Tappia, P.S.; Aukema, H.M.; Aluko, R.E. Blood pressure lowering effect of a pea protein hydrolysate in hypertensive rats and humans. J. Agric. Food Chem. 2011, 59, 9854–9860. [Google Scholar] [CrossRef]
- Babault, N.; Paizis, C.; Deley, G.; Guerin-Deremaux, L.; Saniez, M.H.; Lefranc-Millot, C.; Allaert, F.A. Pea proteins oral supplementation promotes muscle thickness gains during resistance training: A double-blind, randomized, Placebo-controlled clinical trial vs. Whey protein. J. Int. Soc. Sports Nutr. 2015, 12, 3. [Google Scholar] [CrossRef]
- Abou-Samra, R.; Keersmaekers, L.; Brienza, D.; Mukherjee, R.; Mace, K. Effect of different protein sources on satiation and short-term satiety when consumed as a starter. Nutr. J. 2011, 10, 139. [Google Scholar] [CrossRef]
- Bahr, M.; Fechner, A.; Kiehntopf, M.; Jahreis, G. Consuming a mixed diet enriched with lupin protein beneficially affects plasma lipids in hypercholesterolemic subjects: A randomized controlled trial. Clin. Nutr. 2013, 12, 107. [Google Scholar] [CrossRef]
- Bahr, M.; Fechner, A.; Kramer, J.; Kiehntopf, M.; Jahreis, G. Lupin protein positively affects plasma LDL cholesterol and LDL:HDL cholesterol ratio in hypercholesterolemic adults after four weeks of supplementation: A randomized, controlled crossover study. Nutr. J. 2015, 34, 107. [Google Scholar] [CrossRef]
- Banaszek, A.; Townsend, J.R.; Bender, D.; Vantrease, W.C.; Marshall, A.C.; Johnson, K.D. The Effects of Whey vs. Pea Protein on Physical Adaptations Following 8-Weeks of High-Intensity Functional Training (HIFT): A Pilot Study. Sports 2019, 7, 12. [Google Scholar] [CrossRef]
- Baum, J.I.; Gaines, B.L.; Kubas, G.C.; Mitchell, C.F.; Russell, S.L. Educational nutrition messaging at breakfast reduces snack intake and influences snack preferences in adult men and women. Appetite 2017, 117, 67–73. [Google Scholar] [CrossRef]
- Claessens, M.; Calame, W.; Siemensma, A.D.; Saris, W.H.; Van Baak, M.A. The thermogenic and metabolic effects of protein hydrolysate with or without a carbohydrate load in healthy male subjects. Metabolism 2007, 56, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Claessens, M.; Calame, W.; Siemensma, A.D.; Van Baak, M.A.; Saris, W.H. The effect of different protein hydrolysate/carbohydrate mixtures on postprandial glucagon and insulin responses in healthy subjects. Eur. J. Clin. Nutr. 2009, 63, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Diepvens, K.; Haberer, D.; Westerterp-Plantenga, M. Different proteins and biopeptides differently affect satiety and anorexigenic/orexigenic hormones in healthy humans. Int. J. Obes. 2008, 32, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Fabek, H.; Rajadurai, A.; Arshad, M.U.; Smith, C.E.; Bailo, B.G.; Kubant, R.; Anderson, G.H. Acute effects of lentil fractions on satiety and glycemic responses before and after a meal in healthy young men. FASEB J. 2016, 30, 893. [Google Scholar]
- Geraedts, M.C.; Troost, F.J.; Munsters, M.J.; Stegen, J.H.; De Ridder, R.J.; Conchillo, J.M.; Saris, W.H. Intraduodenal administration of intact pea protein effectively reduces food intake in both lean and obese male subjects. PLoS ONE 2011, 6, e24878. [Google Scholar] [CrossRef]
- Joy, J.M.; Lowery, R.P.; Wilson, J.M.; Purpura, M.; De Souza, E.O.; Wilson, S.M.; Jager, R. The effects of 8 weeks of whey or rice protein supplementation on body composition and exercise performance. Nutr. J. 2013, 12, 86. [Google Scholar] [CrossRef]
- Kaviani, M.; Chilibeck, P.D.; Toles, K.; Farthing, J.P.; Candow, D.H. The benefits of hemp powder supplementation during resistance training Applied Physiology. Nutr. Metab. 2016, 41, S365. [Google Scholar] [CrossRef]
- Lang, V.; Bellisle, F.; Oppert, J.M.; Craplet, C.; Bornet, F.R.; Slama, G.; Guy-Grand, B. Satiating effect of proteins in healthy subjects: A comparison of egg albumin, casein, gelatin, soy protein, pea protein, and wheat gluten. Am. J. Clin. Nutr. 1998, 67, 1197–1204. [Google Scholar] [CrossRef]
- Lefranc-Millot, C.; Rodriguez, B.; Brunger, L.; Re, R.; Guerin-Demraux, L. Impact of the vegetable pea protein NUTRALYS® on Satiety, Food intake and Gut Hormones in humans. In Annals of Nutrition and Metabolism; Karger: Basel, Switzerland, 2015; Volume 67, p. 433. [Google Scholar]
- Mollard, R.C.; Fabek, H.; Anderson, G.H.; Lagorse, C.; Wang, H.; Jones, P.J. The effects of consuming smoothies containing faba bean ingredients on postprandial glycemia in healthy young men. FASEB J. 2017, 31, 33. [Google Scholar]
- Mollard, R.C.; MacKay, D.S.; Wang, H.; Leon, A.S.; Jones, P.J. Acute effects of hemp protein on postprandial glycemia and insulin responses in adults. FASEB J. 2017, 31, 28. [Google Scholar]
- Sirtori, C.R.; Triolo, M.; Bosisio, R.; Bondioli, A.; Calabresi, L.; De Vergori, V.; Arnoldi, A. Hypocholesterolaemic effects of lupin protein and pea protein/fibre combinations in moderately hypercholesterolaemic individuals. Br. J. Nutr. 2012, 107, 1176–1183. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.Y.; Siow, P.C.; Peh, E.; Henry, C.J. Influence of rice, pea and oat proteins in attenuating glycemic response of sugar-sweetened beverages. Eur. J. Nutr. 2018, 57, 2795–2803. [Google Scholar] [CrossRef] [PubMed]
- Teunissen-Beekman, K.F.; Dopheide, J.; Geleijnse, J.M.; Bakker, S.J.; Brink, E.J.; De Leeuw, P.W.; Van Baak, M.A. Dietary proteins improve endothelial function under fasting conditions but not in the postprandial state, with no effects on markers of low-grade inflammation. Br. J. Nutr. 2015, 114, 1819–1828. [Google Scholar] [CrossRef]
- Xia, Z.; Cholewa, J.M.; Dardevet, D.; Huang, T.; Zhao, Y.; Shang, H.; Zanchi, N.E. Effects of oat protein supplementation on skeletal muscle damage, inflammation and performance recovery following downhill running in untrained collegiate men. Food Funct. 2018, 9, 4720–4729. [Google Scholar] [CrossRef]
- Weisse, K.; Brandsch, C.; Zernsdorf, B.; Nkengfack Nembongwe, G.S.; Hofmann, K.; Eder, K.; Stangl, G.I. Lupin protein compared to casein lowers the LDL cholesterol:HDL cholesterol-ratio of hypercholesterolemic adults. Eur. J. Nutr. 2010, 49, 65–71. [Google Scholar] [CrossRef]
- Sterne, J.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. 2: A revised tool for assessing risk of bias in randomised trials. BMJ (Clin. Res. Ed.) 2019, 366, l4898. [Google Scholar] [CrossRef]
- Layman, D.K.; Shiue, H.; Sather, C.; Erickson, D.J.; Baum, J. Increased dietary protein modifies glucose and insulin homeostasis in adult women during weight loss. J. Nutr. 2013, 133, 405–410. [Google Scholar] [CrossRef]
- Markova, M.; Pivovarova, O.; Hornemann, S.; Sucher, S.; Frahnow, T.; Wegner, K.; Machann, J.; Petzke, K.J.; Hierholzer, J.; Lichtinghagen, R.; et al. Isocaloric Diets High in Animal or Plant Protein Reduce Liver Fat and Inflammation in Individuals with Type 2 Diabetes. Gastroenterology 2017, 152, 571–585.e8. [Google Scholar] [CrossRef]
- Layman, D.K.; Anthony, T.G.; Rasmussen, B.B.; Adams, S.H.; Lynch, C.J.; Brinkworth, G.D.; Davis, T.A. Defining meal requirements for protein to optimize metabolic roles of amino acids. Am. J. Clin. Nutr. 2015, 101, 1330S–1338S. [Google Scholar] [CrossRef]
- Chan, C.; Fabek, H.; Mollard, R.C.; Jones, P.; Tulbek, M.C.; Chibbar, R.N.; Gangola, M.P.; Ramadoss, B.R.; Sánchez-Hernández, D.; Anderson, G.H. Faba bean protein flours added to pasta reduce post-ingestion glycaemia, and increase satiety, protein content and quality. Food Funct. 2019, 10, 7476–7488. [Google Scholar] [CrossRef] [PubMed]
- Fabek, H.; Mollard, R.C.; Jones, P.J.; Anderson, G.H. The addition of faba bean ingredients to crackers reduces acute postprandial glycemia in healthy young men. FASEB J. 2017, 31, 26. [Google Scholar]
- Markova, M.; Hornemann, S.; Sucher, S.; Wegner, K.; Pivovarova, O.; Rudovich, N.; Thomann, R.; Schneeweiss, R.; Rohn, S.; Pfeiffer, A. Rate of appearance of amino acids after a meal regulates insulin and glucagon secretion in patients with type 2 diabetes: A randomized clinical trial. Am. J. Clin. Nutr. 2018, 108, 279–291. [Google Scholar] [CrossRef]
- Rajamohan, T.; Kurup, P.A. Lysine: Arginine ratio of a protein influences cholesterol metabolism. Part 1–Studies on sesame protein having low lysine: Arginine ratio. Indian J. Exp. Biol. 1997, 35, 1218–1223. [Google Scholar] [PubMed]
- Hodgson, J.M.; Lee, Y.P.; Puddey, I.B.; Sipsas, S.; Ackland, T.R.; Beilin, L.J.; Belski, R.; Mori, T.A. Effects of increasing dietary protein and fibre intake with lupin on body weight and composition and blood lipids in overweight men and women. Int. J. Obes. 2010, 34, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Belski, R.; Mori, T.A.; Puddey, I.B.; Sipsas, S.; Woodman, R.J.; Ackland, T.R.; Beilin, L.J.; Dove, E.R.; Carlyon, N.B.; Jayaseena, V. Effects of lupin-enriched foods on body composition and cardiovascular disease risk factors: A 12-month randomized controlled weight loss trial. Int. J. Obes. 2011, 35, 810–819. [Google Scholar] [CrossRef]
- Ward, N.C.; Mori, T.A.; Beilin, J.L.; Johnson, S.; Williams, C.; Gan, S.K.; Puddey, I.B.; Woodman, R.; Phillips, M.; Connolly, E. The effect of regular consumption of lupin-containing foods on glycaemic control and blood pressure in people with type 2 diabetes mellitus. Food Funct. 2011, 11, 741–747. [Google Scholar] [CrossRef]
- Berrazaga, I.; Micard, V.; Gueugneau, M.; Walrand, S. The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrients 2019, 11, 1825. [Google Scholar] [CrossRef]
- Bayat, A.; Azizi-Soleiman, F.; Heidari-Beni, M.; Feizi, A.; Iraj, B.; Ghiasvand, R.; Askari, G. Effect of Cucurbita ficifolia and Probiotic Yogurt Consumption on Blood Glucose, Lipid Profile, and Inflammatory Marker in Type 2 Diabetes. Int. J. Prev. Med. 2016, 7, 30. [Google Scholar] [CrossRef]
- Cândido, F.G.; De Oliveira, F.C.E.; Lima, M.F.C.; Pinto, A.C.; Da Silva, L.L.; Martino, S.D.H.; Santos, M.H.D.; De Cássia, R.; Alfenas, G. Addition of pooled pumpkin seed to mixed meals reduced postprandial glycemia: A randomized placebo-controlled clinical trial. Nutr. Res. 2018, 56, 90–97. [Google Scholar] [CrossRef]
- Brennan, J.L.; Keerati-U-Rai, M.; Yin, H.; Daoust, J.; Nonnotte, E.; Quinquis, L.; St-Denis, T.; Bolster, D.R. Differential Responses of Blood Essential Amino Acid Levels Following Ingestion of High-Quality Plant-Based Protein Blends Compared to Whey Protein-A Double-Blind Randomized, Cross-Over, Clinical Trial. Nutrients 2019, 11, 2987. [Google Scholar] [CrossRef] [PubMed]
- Guevara-Cruz, M.; Tovar, A.R.; Aguilar-Salinas, C.A.; Medina-Vera, I.; Gil-Zenteno, L.; Hernández-Viveros, I.; López-Romero, P.; Ordaz-Nava, G.; Canizales-Quinteros, S.; Guillen Pineda, E.L. A dietary pattern including nopal, chia seed, soy protein, and oat reduces serum triglycerides and glucose intolerance in patients with metabolic syndrome. J. Nutr. 2012, 142, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Vuksan, V.; Jenkins, A.L.; Brissette, C.; Choleva, L.; Jovanovski, E.; Gibbs, A.L.; Bazinet, R.P.; Au-Yeung, F.; Zurbau, A.; Ho, H.V.T.; et al. Salba-chia (Salvia hispanica L.) in the treatment of overweight and obese patients with type 2 diabetes: A double-blind randomized controlled trial. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Jokioja, J.; Linderborg, K.M.; Kortesniemi, M.; Nuora, A.; Heinonen, J.; Sainio, T.; Viitanen, M.; Kallio, H.B. Anthocyanin-rich extract from purple potatoes decreases postprandial glycemic response and affects inflammation markers in healthy men. Food Chem. 2020, 310, 125797. [Google Scholar] [CrossRef]
- Qiu, J.; Liu, Y.; Yue, Y.; Qin, Y.; Li, Z. Dietary tartary buckwheat intake attenuates insulin resistance and improves lipid profiles in patients with type 2 diabetes: A randomized controlled trial. Nutr. Res. 2016, 36, 1392–1401. [Google Scholar] [CrossRef]
- Zeinstra, G.G.; Somhorst, D.; Oosterink, E.; Fick, F.; Klopping-Ketelaars, I.; Van Der Meer, I.; Mes, J.J.M. Postprandial amino acid, glucose and insulin responses among healthy adults after a single intake of Lemna minor in comparison with green peas: A randomised trial. J. Nutr. Sci. 2019, 8, e28. [Google Scholar] [CrossRef]
- Zelicha, H.; Kaplan, A.; Yaskolka Meir, A.; Tsaban, G.; Rinott, E.; Shelef, I.; Tirosh, A.; Brikner, D.; Pupkin, E.; Qi, L.; et al. The Effect of Wolffia globosa Mankai, a Green Aquatic Plant, on Postprandial Glycemic Response: A Randomized Crossover Controlled Trial. Diabetes Care 2019, 42, 1162. [Google Scholar] [CrossRef]
- Mintel Global New Product Database (GNPD) 2015–2019. Available online: https://www.mintel.com/global-new-products-database (accessed on 6 May 2020).
- Roland, W.S.U.; Pouvreau, L.; Curran, J.; Van De Velde, F.; De Kok, P.M.T. Flavor Aspects of Pulse Ingredients. CCHEM 2017, 94, 58–65. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Satija, A.; Blondin, S.A.; Janiszewski, M.; Emlen, E.; O’Connor, L.E.; Campbell, W.W.; Hu, F.B.; Willett, W.C.; Stampfer, M.J. Meta-Analysis of Randomized Controlled Trials of Red Meat Consumption in Comparison with Various Comparison Diets on Cardiovascular Risk Factors. Circulation 2017, 139, 1828–1845. [Google Scholar] [CrossRef]
| PICOS Format | Description |
|---|---|
| Population | Adults ≥18 years old; no restrictions regarding sex or health status |
| Intervention | Intake of dietary proteins from the following sources: pea, chickpea, lentil, lupin, fava bean, hemp, sunflower, pumpkin, oat, rice, rapeseed, buckwheat, quinoa, duckweed, alfalfa, potato, sacha inchi, mushrooms. No restrictions regarding the dose or intervention duration were applied. Only extracted forms were considered (concentrates, isolates or hydrolysates). |
| Comparisons | Other protein sources (animal or plant), placebo/control (e.g., water, maltodextrin, other foods with manipulated macronutrient content) or receiving another intervention. |
| Outcomes | Changes in blood glucose, insulin, satiety hormones and subjective satiety, cardiometabolic risk factors (blood lipids, blood pressure), muscle mass and strength and change in body weight |
| Study type | Interventional studies; acute or chronic; parallel or crossover design. |
| Reference | Design | Plant Source | Form | Reported Health Outcomes | |
|---|---|---|---|---|---|
| 1. | Abou-Samra et al. (2011) [45] | Open, single-blind randomised, cross-over | Pea | I | Glycaemia and satiety |
| 2. | Babault et al. (2015) [44] | Double-blind, randomised, placebo-controlled, parallel | Pea | I | Muscle health |
| 3. | Bahr et al. (2013) [46] | Randomised, controlled, cross-over | Lupin | I | Blood lipids, blood pressure, body weight |
| 4. | Bahr et al. (2014) [47] | Randomised, double-blind cross-over | Lupin | I | Blood lipids, blood pressure, body weight |
| 5. | Banaszek et al. (2019) [48] | Randomised, double blind, parallel | Pea | I | Muscle health, body weight |
| 6. | Baum et al. (2017) [49] | Randomised, cross-over | Pea | I | Glycaemia and satiety |
| 7. | Claessens et al. (2007) [50] | Single blind, cross-over | Pea | H | Glycaemia and satiety |
| 8. | Claessens et al. (2009) [51] | Repeated measures with Latin square randomisation, single blind | Pea, rice | H | Glycaemia and satiety |
| 9. | Contaldo et al. (1983) [42] | Cross-over | Fava bean | C | Glycaemia and satiety, blood lipids, body weight |
| 10. | Diepvens et al. (2008) [52] | Randomised, cross over | Pea | H | Glycaemia and satiety |
| 11. | Fabek et al. (2016) [53] * | Two randomised, cross-over, repeated measures | Lentil | C and I | Glycaemia and satiety |
| 12. | Geraedts et al. (2011) [54] | Single blind, randomised, controlled, cross-over | Pea | I | Glycaemia and satiety |
| 13. | Joy et al. (2013) [55] | Randomised, double blind, parallel | Rice | I | Muscle health, body weight |
| 14. | Kaviani et al. (2016) [56] * | Randomised, double blind, parallel | Hemp | C | Muscle health |
| 15. | Lang et al. (1998) [57] | Within-subjects design (two-tail Latin square) | Pea | I | Glycaemia and satiety |
| 16. | Lefranc-Millot et al. (2015) [58] ** | Double blind, randomised, placebo control, cross-over | Pea | I | Glycaemia and satiety |
| 17. | Li et al. (2011) [43] | Randomised, double blind, placebo-controlled, cross-over | Pea | H | Blood pressure |
| 18. | Mollard et al. (2014) [40] | Randomised, cross-over, single-blinded | Pea | I | Glycaemia and satiety |
| 19. | Mollard et al. (2017a) [59] * | Repeated measures, cross-over, randomised | Fava bean | C and I | Glycaemia and satiety |
| 20. | Mollard et al. (2017b) [60] * | Repeated measures, cross-over, randomised | Hemp | C | Glycaemia and satiety |
| 21. | Sirtori et al. (2012) [61] | Randomised, double blind, parallel | Pea, lupin | I | Glycaemia and satiety, blood lipids |
| 22. | Smith et al. (2012) [41] | Single blind, randomised, repeated-measures | Pea | I | Glycaemia and satiety |
| 23. | Tan et al. (2018) [62] | Randomised, controlled, cross-over | Oat, rice, pea | C | Glycaemia and satiety, blood lipids |
| 24. | Teunissen-Beekman et al. (2014) [63] | Double blind, six-arm randomised, cross-over | Pea | I | Glycaemia and satiety, blood pressure |
| 25. | Xia et al. (2018) [64] | Randomised, double-blind, placebo-controlled | Oat | I | Muscle health |
| 26. | Weisse et al. (2010) [65] | Randomised, double blind, placebo controlled, parallel | Lupin | I | Glycaemia and satiety, blood lipids |
| Reference | Protein Source | Population | Intervention | Comparator(s) | Duration | Results | |
|---|---|---|---|---|---|---|---|
| Dose | Form | ||||||
| Abou-Samra et al. (2011) [45] | Pea proteins (isolate; 90% protein) | 32 males, mean age: 25 ± 4 (E1) and 25 ± 0.6 (E2) | ≈20 g | Drink | whey, maltodextrin, casein, egg albumin and water (control) | 1 day (acute) | E1: Ad libitum energy intake ↓ after casein and pea preloads compared to water control; Combined satiety score ↑ after casein and pea compared to other preloads; plasma glucose response to ad libitum meal ↓ when whey protein used as a preload (compared to other preloads); E2: cumulative EI (preload + ad libitum meal) ↑ after pea, casein and whey in comparison to water. |
| Baum et al. (2017) [49] | Pea proteins | 23 females, 10 males, mean age 23.5 | 41 g | Breakfast beverage | Whey protein isolate | 1 day (acute) | No difference in postprandial appetite response between pea and whey protein. |
| Claessens et al. (2007) [50] | Pea protein hydrolysate (PPH) with a CHO afterload | 8 males, mean age 28.5 ± 3.6 | 0.4 g protein/kg of body weight | Drink | Soy protein hydrolysate (SPH), maltodextrin (control) | 5 trials, each 1 day (acute) | Pea protein with a CHO afterload induced ↑ insulin and glucagon responses (area under the curve) than SPH with CHO afterload. |
| Claessens et al. (2009) [51] | Pea protein hydrolysate (PPH) and rice protein hydrolysates (RPH) | 8 males, mean age 32 ± 13.8 | 0.2 g hydrolysate/kg of body weight | Drink | Rice, soy, gluten, whey, egg hydrolysates, and maltodextrin (control) | 7 trials, each 1 day (acute) | No difference in glucose and insulin response at peak (delta 30) between hydrolysates. Glucagon response ↑ only for soy (when compared to gluten). Glucagon AUCs differed only between gluten and egg. |
| Contaldo et al. (1983) [42] | Fava bean concentrate | 4 females, 4 males, age 43.4 | 50 g (F); 70 g(M) daily | Integrated into a diet | Egg whites | 18 days | Fasting BG ↓ after faba bean, compared to baseline. Insulin unchanged in both diets. |
| Diepvens et al. (2008) [52] | Pea protein hydrolysate (PPH) | 20 females, 19 males, mean age 42.3 ± 13.8 | 15 g | Shake | Whey protein (WP), and a blend of WP + PPH, and milk protein (MP, casein + WP) (control) | 1 day in E1 (4 h) and E2 (7 h) | PPH induced ↓ hunger and ↓ desire to eat compared to MP or WP + PPH. A longer intermeal interval and ↑ satiety index after PPH. Both PPH and WP lead to ↑ satiety (E2) and fullness (E2) compared to MP and WP + PPH. GLP-1 and CCK was ↑ after MP, than in four other shakes. No effect on EI was seen. |
| Fabek et al. (2016) [53] Abstract only (FASEB) | Lentil protein concentrate (LPC, 55% protein) and isolate (LPI, 75% protein) | 48 males, young (age not specified) | 20 g of LPC (55% protein) or LPI (75% protein) | Added to tomato soup | Lentil fibre, lentil starch and tomato soup alone (control) | 1 day (acute) | Ad libitum meal after 30 min: only LPI and LPC induced ↓ postprandial glycemia without increasing insulin, and ↓ subjective appetite, compared to the control. Ad libitum meal after 120 min: only lentil starch resulted in ↓ pre-and post-meal subjective appetite, but higher blood glucose than control. |
| Geraedts et al. (2011) [54] | Pea protein | 20 males, mean age: 25 ± 2 (lean), 41 ± 6 (obese) | 250 mg/kg of body weight | Drink (oral or intraduodenal admin.) | Water (placebo) | 1 day (acute) on 4 occasions | Some changes observed in terms of hunger, fullness, CCK, PYY and food intake, with differences regarding method of protein administration (orally vs. intraduodenal) and nutrition status (lean vs. obese). No difference in satiety and ghrelin level. |
| Lang et al. (1998) [57] | Pea protein isolate | 12 males, mean age 22.6 ± 0.6 | 45.3 g ± 1.2 | Protein manipulated meal | Egg albumin, casein, gelatin, soy protein and wheat gluten | 1 day (acute) on 6 occasions | No effect of the type of protein on satiety, on 24-h energy or macronutrient intakes, or on postprandial plasma glucose and insulin concentrations. |
| Lefranc-Millot et al. (2015) [58] Conference poster | Pea protein isolate | 22 females, 11 males, age range 18–65 | 15 g and 30 g | soup | Whey protein and no protein soup (control) | 1 day (acute) | Both PP and WP induced ↓ caloric intake, compared to control. Favorable modulation of glucose, insulin and and some satiety hormones, but mainly when compared to control, rather than other protein treatments. |
| Mollard et al. (2014) [40] | Pea protein (82% protein) | 15 males, mean age 21.5 ± 1.0 | 10 g | Added to meal | Pea hull fibre, pea hull fibre + pea protein, canned yellow peas, noodles with tomato sauce (control) | 1 day (acute), with 1 treatment (or control) per week | No effect of treatment on food intake, pre and post pizza subjective appetite. In terms of PP, between treatments difference was observed only in comparison to fibre (↓ pre-meal BG AUC and ↓ BG cumulative AUC). |
| Mollard et al. (2017a) [59] Abstract | Fava bean concentrate (FBC) and isolate (FBI) | 15 males, young (age not specified) | 32 g FBC 32 g FBI | Smoothies | whole FB flour, high-starch FB flour, corn maltodextrin (control) | 1 day (acute) | All flours had favourable effect on pre-meal glucose and iAUC, in comparison to control. Between treatment differences were observed for FBC and FBI (↓ blood post meal glucose iAUC when compared to FB starch). |
| Mollard et al. (2017b) [60] Abstract | Hemp protein concentrate (HPC) | 16 adults, young (age not specified) | 20 g and 40 g | Fruit shakes | Soybean concentrate (SBC), carbohydrate (control) | 1 day (acute) | Favourable effects (dose-dependent) of all protein treatments (soy and hemp) on glucose and insulin; in general no significant differences between protein type. In addition, HPC (40 g) led to ↑ glucose and insulin responses following a fixed meal 60 min after protein ingestion. |
| Sirtori et al. (2012) [61] | Pea protein isolate (PPI) and cellulose/oat fibre/pectin, Lupin protein isolate (LPI) and cellulose | 93 females, 82 males; mean age 53.9 | Two bars a day (34.6 g day) | Bars | Casein and cellulose, casein and oat fibre, casein and pectin | 4 wk | At 4 wk, glucose ↓ only after pea protein and oat fibre, in comparison to baseline. Insulin ↓ after casein and cellulose, casein and pectin, pea protein and oat fibre, compared to baseline |
| Smith et al. (2012) [41] | Yellow pea protein | E1: 19 males, mean age 23.2 ± 0.5 E2: 20 males, mean age 22.3 ± 0.5 | Two treatments: 10 and 20 g | Added to tomato soup | Tomato soup with 10 and 20 g fibre, tomato soup with no added fibre or protein (control) | 1 day (acute study) | E1: 20 g protein led to ↓ food intake (FI) than control and all other treatments and ↓ cumulative FI compared to 10 g fibre. Both protein doses stipulated ↓ pre-meal glucose (0–30 min) compared to control; only 20 g protein suppressed mean post-meal BG (50–120 min). No effect of on pre-meal or post-meal appetite. E2: no effect of treatment on FI, CFI, or pre-or post-meal BG or appetite. |
| Tan et al. (2018) [62] | Oat, rice and pea concentrates | 20 males, mean age 26 ± 5 | 24 g | Chocolate beverage | Chocolate beverage without protein | 1 day (acute) | Insulin iAUC was ↑ after oat and pea, but not rice ingestions, in comparison to control. No sig. differences in GIP and GLP-1. Sig. effect of time on hunger, fullness, desire to eat, but with no sig. effect of treatment. |
| Teunissen-Beekman et al. (2014) [63] | Pea protein isolate | 31 males, 17 females, mean age 58 ± 1 | 0.6 g of protein/kg of body weight | Drink | Milk protein isolate, an egg-white protein isolate, blend of protein isolates, maltodextrin and sucrose | 1 day (acute study) | The ingestion of all proteins resulted in ↓ plasma glucose concentrations and ↑ insulin (iAUC). No differences in the postprandial BG responses to the ingestion of all the three proteins. Insulin ↓ after egg-white (than milk) after 1–2 h, and ↑ after milk than pea (at 4 h). Glucagon ↑ after all protein, but lowest after egg-white, than other proteins (at 1–3 h). At 1–2 h, highest glucagon after pea protein. GLP-1 highest after all 3 protein, but ↓ after egg-white than of other protein (at 2 h), and ↑ after egg-white than pea (at 4 h). |
| Weisse et al. (2010) [65] | Lupin isolate | 23 females, 20 males; mean age 43.9 ± 11.8 | 17.5 g twice a daily | Bars | Casein | 6 weeks | No changes in plasma glucose from baseline or between the interventions. |
| Reference | Protein Source | Population | Intervention | Comparator(s) | Duration | Results | |
|---|---|---|---|---|---|---|---|
| Dose | Form | ||||||
| Bahr et al. (2013) [46] | Lupin protein isolate (LPI) | 18 females, 15 males, mean age 49.7 ± 12.8 | 25 g/daily | Drink | Milk protein isolate (MPI) | 8 weeks | At wk 4 ↓ in LDL and LDL:HDL in LPI group and ↓ LDL in MPI group. The only difference between treatments in HDL at wk4 (↑ in LPI and ↓ in MPI). After 8 wks ↑ in triglicerides (LPI) and no changes from baseline in MPI group. |
| Bahr et al. (2014) [47] | Lupin protein isolate (LP) | 41 females, 31 males, 18–80 y | 25 g/day | Integrated into a mixed diet | Milk protein (MP), MP + 1.6 g/d arginine (MPA) | 28 days | Compared to baseline, total cholesterol, LDL, HDL was ↓ in LP and MPA groups. Triglycerides were only ↓ in LP group. Between treatments differences only for total cholesterol (↓ in MPA vs. MP) and LDL (↓ in LP vs. MP). The relative changes in total and LDL cholesterol were significantly greater for subjects with severe hypercholesterolemia than those with moderate hypercholesterolemia |
| Contaldo et al. (1983) [42] | Fava bean concentrate | 4 females, 4 males, age 43.4 | 50 g (F); 70 g(M) daily | Integrated into a diet | Egg whites | 18 days | Total cholesterol ↓ in both treatments, but LDL was significantly ↓ only in egg-white diet. HDL decreased only on the fava bean diet. Serum total and VLDL triglyceride showed no changes. |
| Sirtori et al. (2012) [61] | Pea protein isolate (PPI) and cellulose/oat fibre/pectin, Lupin protein isolate (LPI) and cellulose | 93 females, 82 males; mean age 53.9 | Two bars a day (34.6 g day) | Bars | Casein and cellulose, casein and oat fibre, casein and pectin | 4 wk | Lupin protein and cellulose, casein and pectin, pea protein and oat fibre and pea protein and pectin resulted in reduction of total cholesterol. Decrease in LDL only after pea protein + pectin and pea protein + oat fibre. No changes in triglicerides or HDL. |
| Tan et al. (2018) [62] | Oat, rice and pea concentrates | 20 males, mean age 26 ± 5 | 24 g | Chocolate beverage | Chocolate beverage without protein | 1 day (acute) | Serum triglyceride excursions and iAUC did not differ between all test beverages. |
| Weisse et al. (2010) [65] | Lupin isolate | 23 females, 20 males; mean age 43.9 ± 11.8 | 17.5 g twice a daily | Bars | Casein | 6 weeks | Both treatments resulted in ↓ plasma cholesterol from baseline. In addition, LP (reduction of LDL), while casein (reduction of HDL and triglicerides), from baseline. Between the groups difference only for LDL:HDL ratio (↑ in casein, and ↓ in LP group) |
| Reference | Protein Source | Population | Intervention | Comparator(s) | Duration | Results | |
|---|---|---|---|---|---|---|---|
| Dose | Form | ||||||
| Bahr et al. (2013) [46] | Lupin protein isolate (LPI) | 18 females, 15 males, mean age 49.7 ± 12.8 | 25 g/daily | Drink | Milk protein isolate (MPI) | 8 weeks | At 8 wks, ↓ in SBP and DBP in LPI group, and only SBP in MPI (from baseline) but no differences between treatments. |
| Bahr et al. (2014) [47] | Lupin protein isolate (LP) | 41 females, 31 males, 18–80 y | 25 g/day | Integrated into a mixed diet | Milk protein (MP), MP + 1.6 g/d arginine (MPA) | 28 days | No changes in SBP or DBP from baseline or between the treatments |
| Li et al. (2011) [43] | Pea protein hydrolysate (peptides) | 4 females, 3 males; age 30–55 y | Two treatments: 1.5 and 3 g (spread over 3 meals) | Orange juice | Orange juice (placebo) | 3 weeks | In comparison to placebo, a ↓ in SBP after 2 and 3 weeks supplementation of PPH at 3 g/day dose. |
| Teunissen-Beekman et al. (2014) [63] | Pea protein | 17 females, 31 males; mean age: 58 ± 1 | 0.6 g of protein/kg of body weight | Drink | Milk protein isolate, an egg-white protein isolate, mix of protein isolates | 1 day (acute study) | Egg-white protein resulted in ↑ in SBP, while pea protein ↓ DBP. Postprandial BP levels were ↓ after maltodextrin than after protein mix and sucrose meals. |
| Reference | Protein Source | Physical Activity | Population | Intervention | Comparator(s) | Duration | Results | |
|---|---|---|---|---|---|---|---|---|
| Dose | Form | |||||||
| Babault et al. (2015) [44] | Pea proteins | 12-week resistant training | 161 males, mean age: 22 ± 3.5 | 25 g/twice a day | Drink | Whey and placebo | 12 weeks | Thickness increased from baseline in all groups. Highest increase in pea group at 12 wks, compared to placebo. No differences between the groups in terms of muscle circumference at week 12. |
| Banaszek et al. (2019) [48] | Pea proteins | 8-week high-intensity-functional training (HIFT) | 7 females and 8 males, mean age: 38.6 ± 12.7 (M), 38.9 ± 10.9 (F) | 24 g/twice a day | Drink | Whey protein, no control | 8 weeks | Increase in muscle strength, squats and deadlift, from baseline, but no differences between the interventions. |
| Joy et al. (2013) [55] | Rice protein isolate (RPI) | 8-week resistance training | 24 males, mean age: 21.3 ± 1.9 | 48 g | Drink | Whey protein isolate (WPI), no control | 8 weeks | Increase in strength, power and thickness after 8 weeks from the baseline for both RPI and WPI, with no differences between the groups. |
| Kaviani et al. (2016) [56] | Hemp protein (HP) powder | 8-week resistance training | 28 males, 12 females (age not provided) | 60 g powder (40 g protein) | Not specified | Soy protein | 8 weeks | Increased strength and muscle thickness in females, but not males, after HP powder, compared to soy group. |
| Xia et al. (2018) [64] | Oat isolate | Exhaustive downhill running | 16 males, mean age: 19.7 ± 1.1 | 12.5 g/twice a day (14 days before exercise and 4 days thereafter) | Drink | Maltodextrin | 19 days | Compared to placebo, oat protein ↓ inflammatory markers, muscle soreness (VAS) score, and lessen the loss of function associated with damaging exercise. |
| Reference | Protein Source | Population | Intervention | Comparator(s) | Duration | Results | |
|---|---|---|---|---|---|---|---|
| Dose | Form | ||||||
| Bahr et al. (2013) [46] | Lupin protein isolate (LPI) | 18 females, 15 males, mean age 49.7 ± 12.8 | 25 g/daily | Drink | Milk protein isolate (MPI) | 8 weeks | At 8 wks, slight ↑ in body weight, BMI and body fat in LPI and MPI group, compared to baseline, but no differences between intervention |
| Bahr et al. (2014) [47] | Lupin protein isolate (LP) | 41 females, 31 males, 18 −80 y | 25 g/day | Integrated into a mixed diet | Milk protein (MP), MP + 1.6 g/d arginine (MPA) | 28 days | At 28 days, increase in WC in MP group, compared to LP group. No changes in body weight, BMI, or body fat in comparison to baseline and between the treatments. |
| Banaszek et al. (2019) [48] | Pea proteins + PA | 7 females and 8 males, mean age: 38.6 ± 12.7 (M), 38.9 ± 10.9 (F) | 24 g/twice a day | Drink | Whey protein, no control | 8 wk | No change in body mass or body fat from baseline and no differences between the interventions. |
| Contaldo et al. (1983) [42] | Fava bean concentrate | 4 females, 4 males, age 43.4 | 50 g (F); 70 g(M) daily | Integrated into a diet | Egg whites | 18 days | No changes in body weight, except in one patient |
| Joy et al. (2013) [55] | Rice protein isolate + PA | 24 males, mean age: 21.3 ± 1.9 | 48 g | Drink | Whey protein isolate | 48 h (Phase 1) 8 wk (Phase 2) | ↑ in lean body mass ↓ of body fat in both interventions at 8 weeks, with no differences between the interventions; |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lonnie, M.; Laurie, I.; Myers, M.; Horgan, G.; Russell, W.R.; Johnstone, A.M. Exploring Health-Promoting Attributes of Plant Proteins as a Functional Ingredient for the Food Sector: A Systematic Review of Human Interventional Studies. Nutrients 2020, 12, 2291. https://doi.org/10.3390/nu12082291
Lonnie M, Laurie I, Myers M, Horgan G, Russell WR, Johnstone AM. Exploring Health-Promoting Attributes of Plant Proteins as a Functional Ingredient for the Food Sector: A Systematic Review of Human Interventional Studies. Nutrients. 2020; 12(8):2291. https://doi.org/10.3390/nu12082291
Chicago/Turabian StyleLonnie, Marta, Ieva Laurie, Madeleine Myers, Graham Horgan, Wendy R. Russell, and Alexandra M. Johnstone. 2020. "Exploring Health-Promoting Attributes of Plant Proteins as a Functional Ingredient for the Food Sector: A Systematic Review of Human Interventional Studies" Nutrients 12, no. 8: 2291. https://doi.org/10.3390/nu12082291
APA StyleLonnie, M., Laurie, I., Myers, M., Horgan, G., Russell, W. R., & Johnstone, A. M. (2020). Exploring Health-Promoting Attributes of Plant Proteins as a Functional Ingredient for the Food Sector: A Systematic Review of Human Interventional Studies. Nutrients, 12(8), 2291. https://doi.org/10.3390/nu12082291






