Postprandial Metabolic Response to Rapeseed Protein in Healthy Subjects
Abstract
1. Introduction
2. Materials and Methods
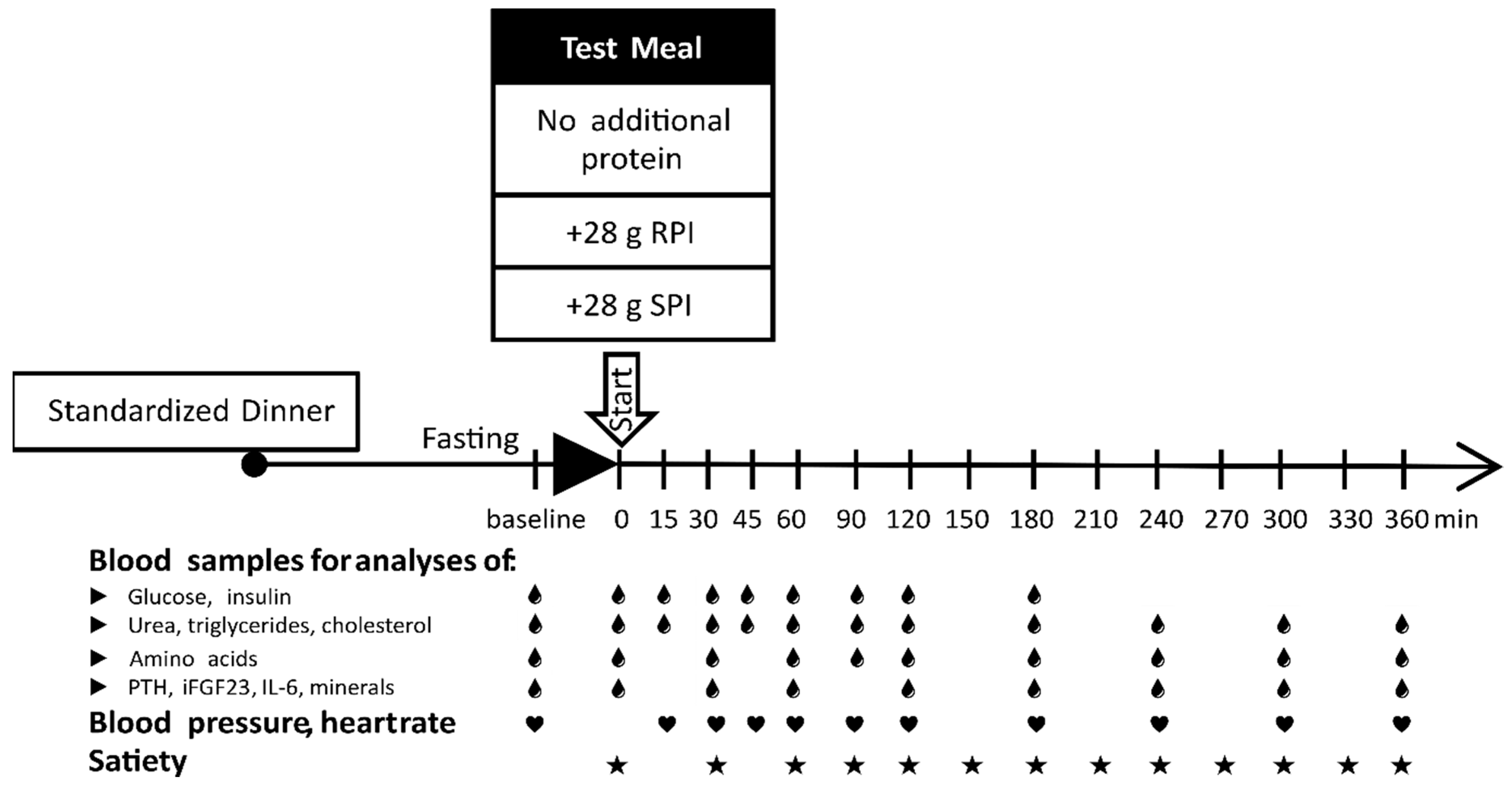
2.1. Study Design and Study Population
2.2. Preparation and Composition of the Meals
2.3. Preparation and Composition of the Proteins
2.4. Satiety and Appetite Assessment
2.5. Blood Sampling and Analysis
2.6. Statistical Analysis
3. Results
3.1. Characteristics of the Test Proteins
3.2. Subjects
3.3. Postprandial Heart Rate and Blood Pressure
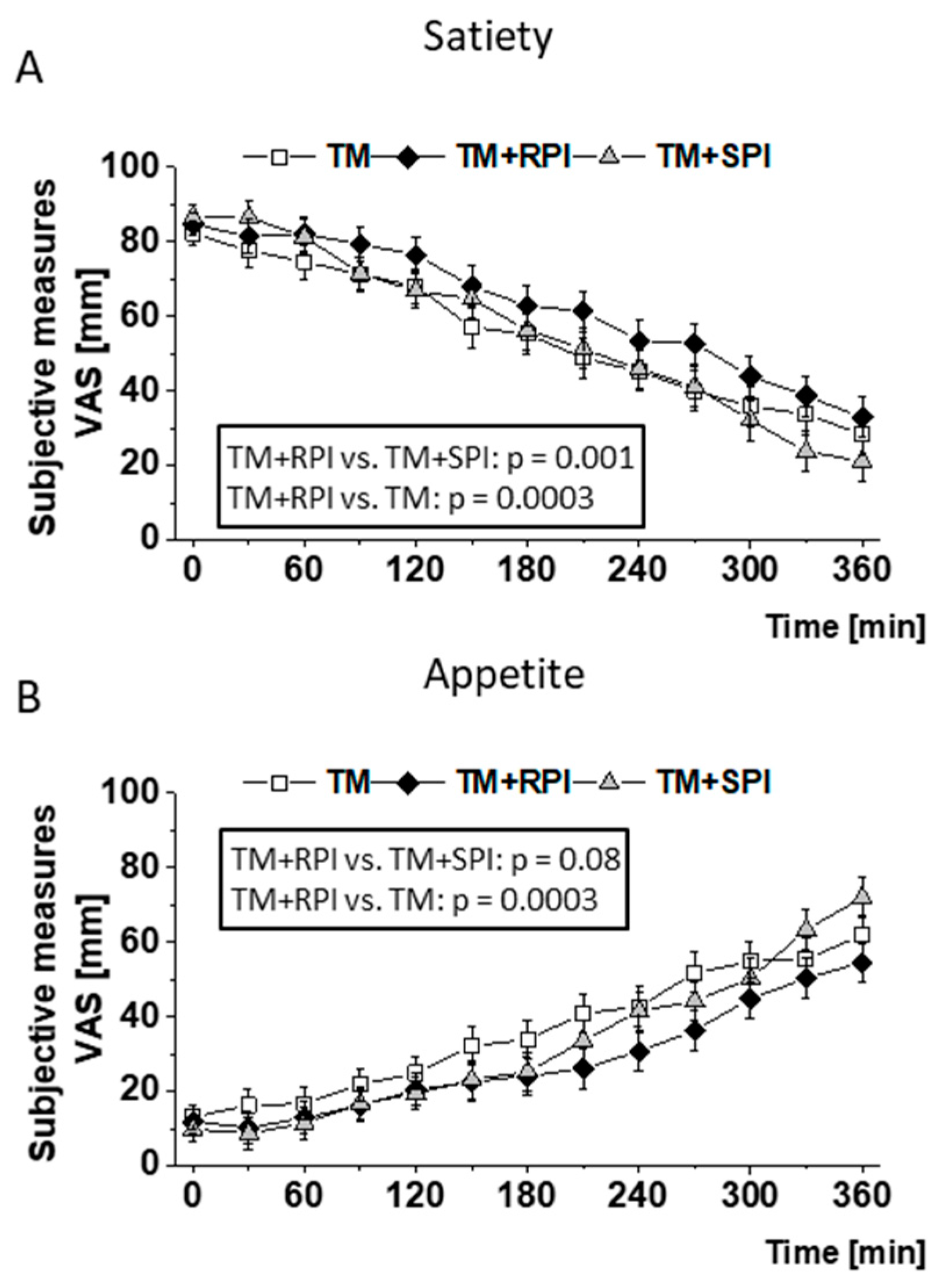
3.4. Postprandial Satiety and Appetite
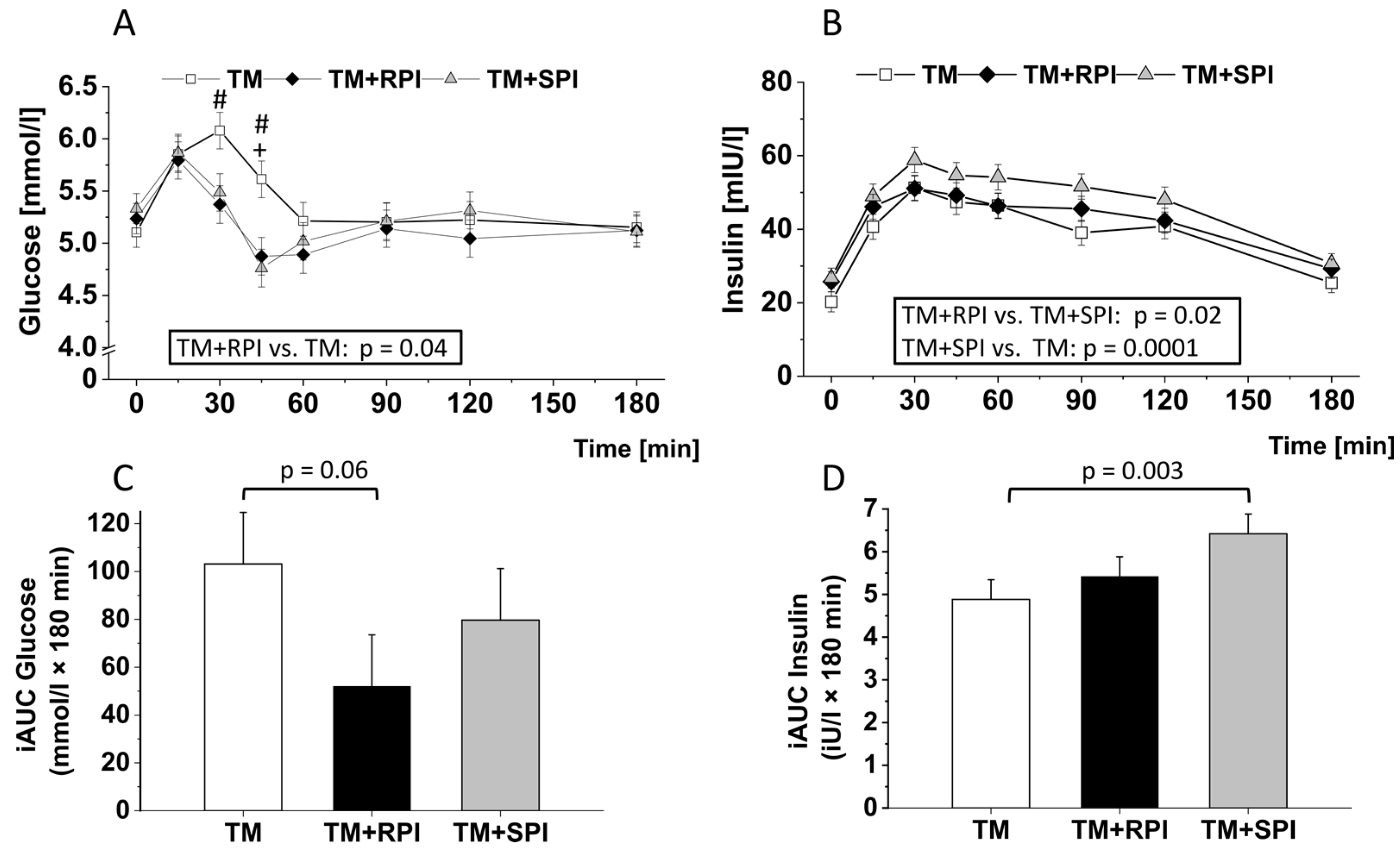
3.5. Postprandial Plasma Glucose and Insulin
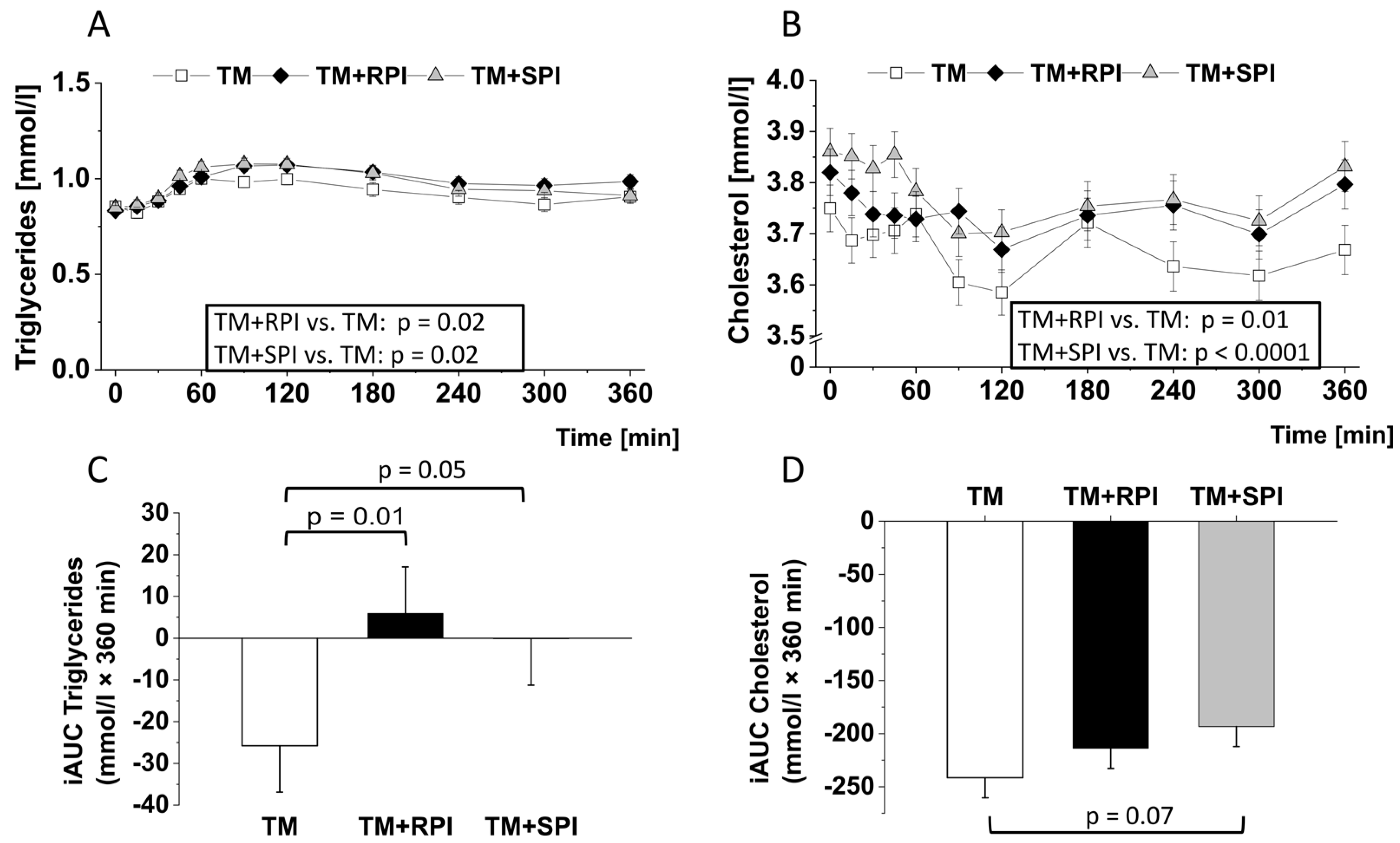
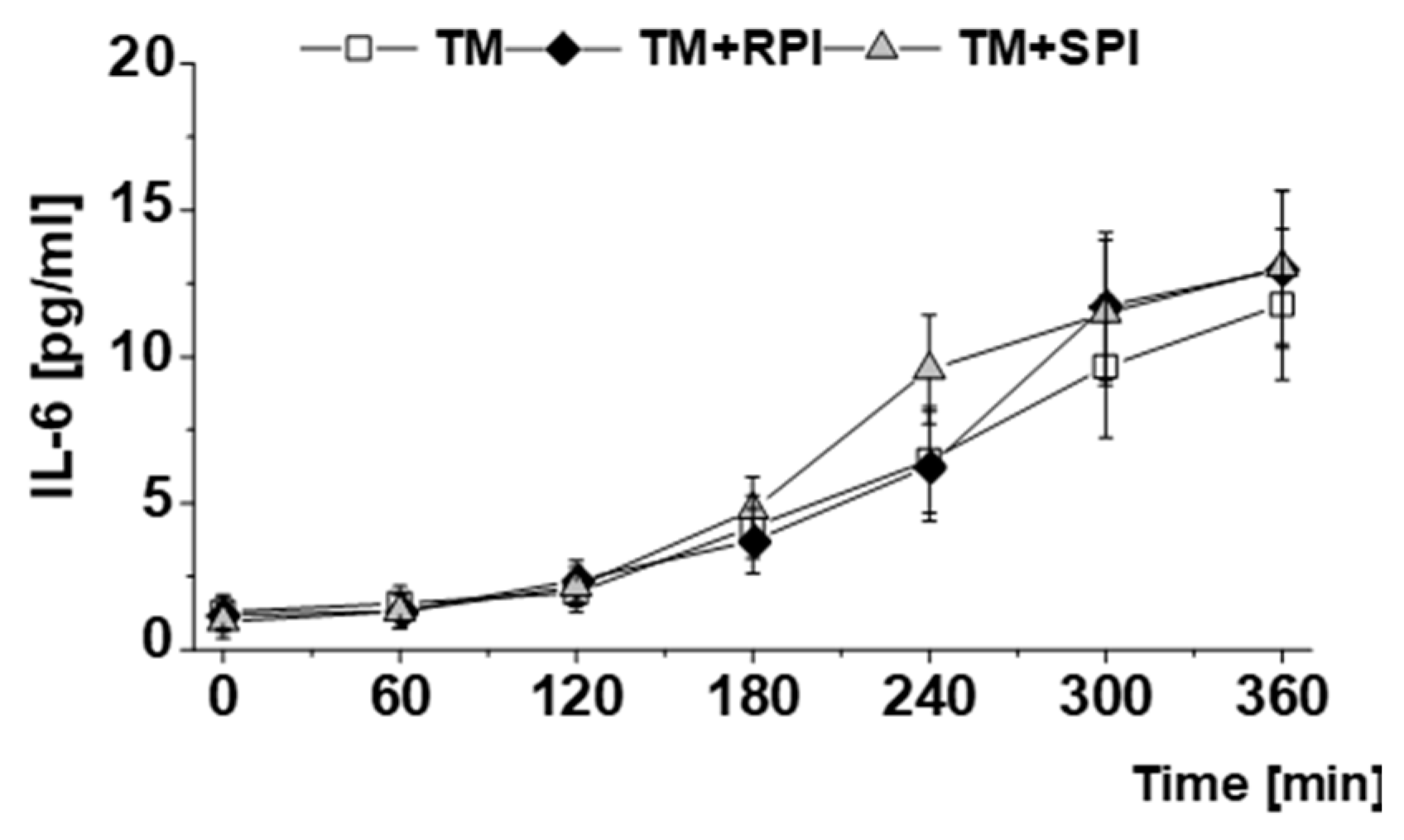
3.6. Postprandial Lipids and Inflammatory Markers
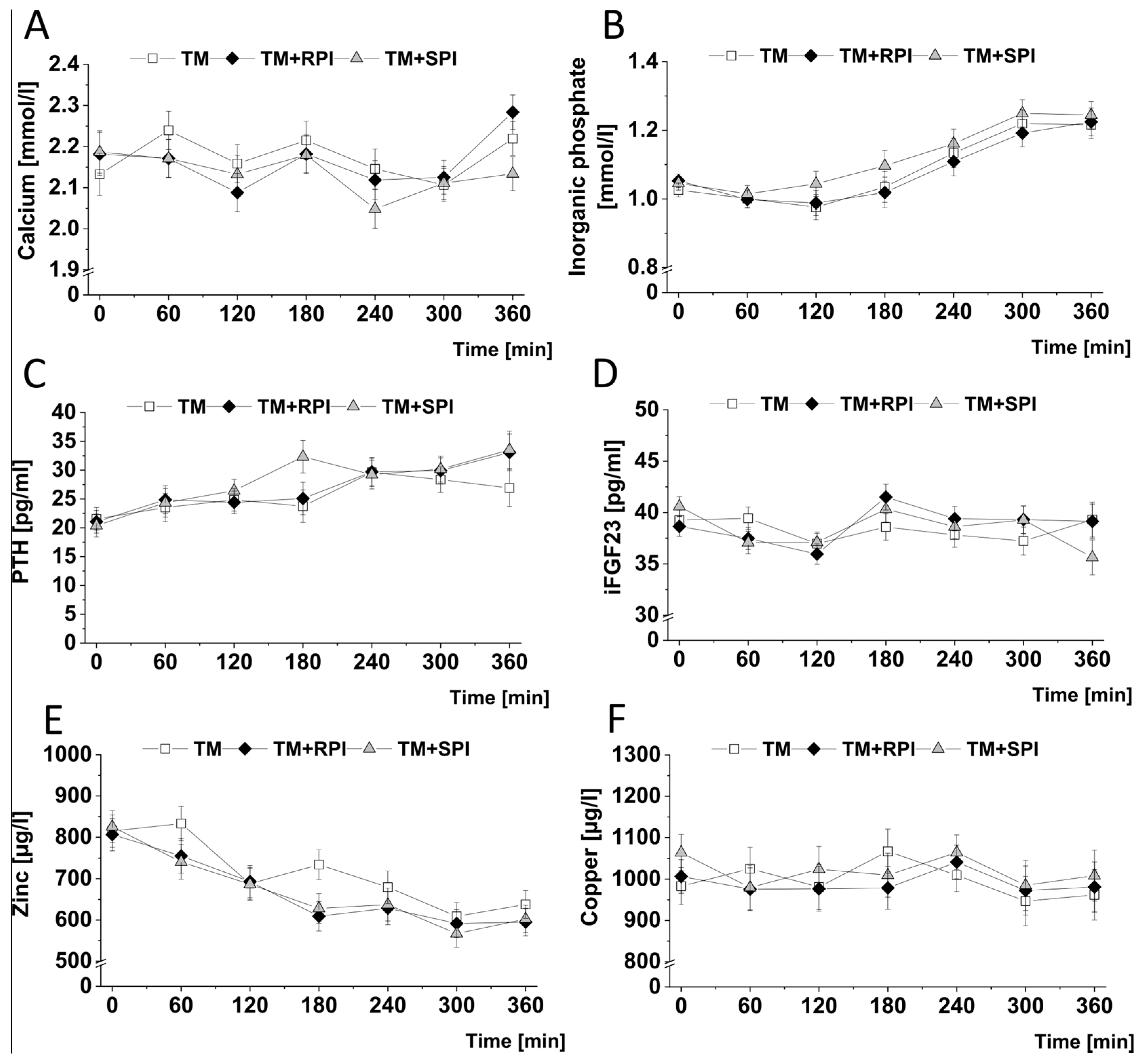
3.7. Postprandial Minerals and Mineral-Regulating Hormones
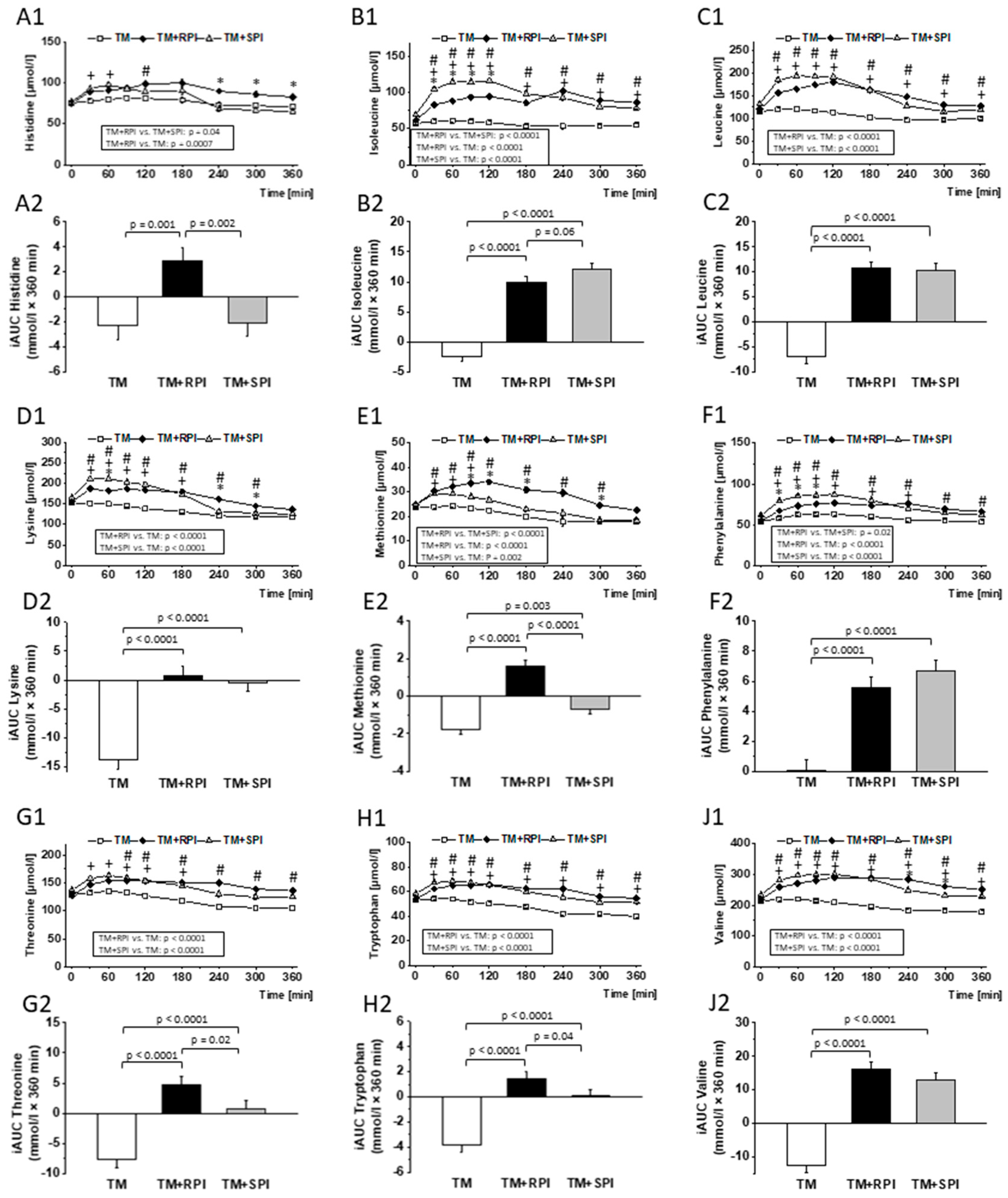
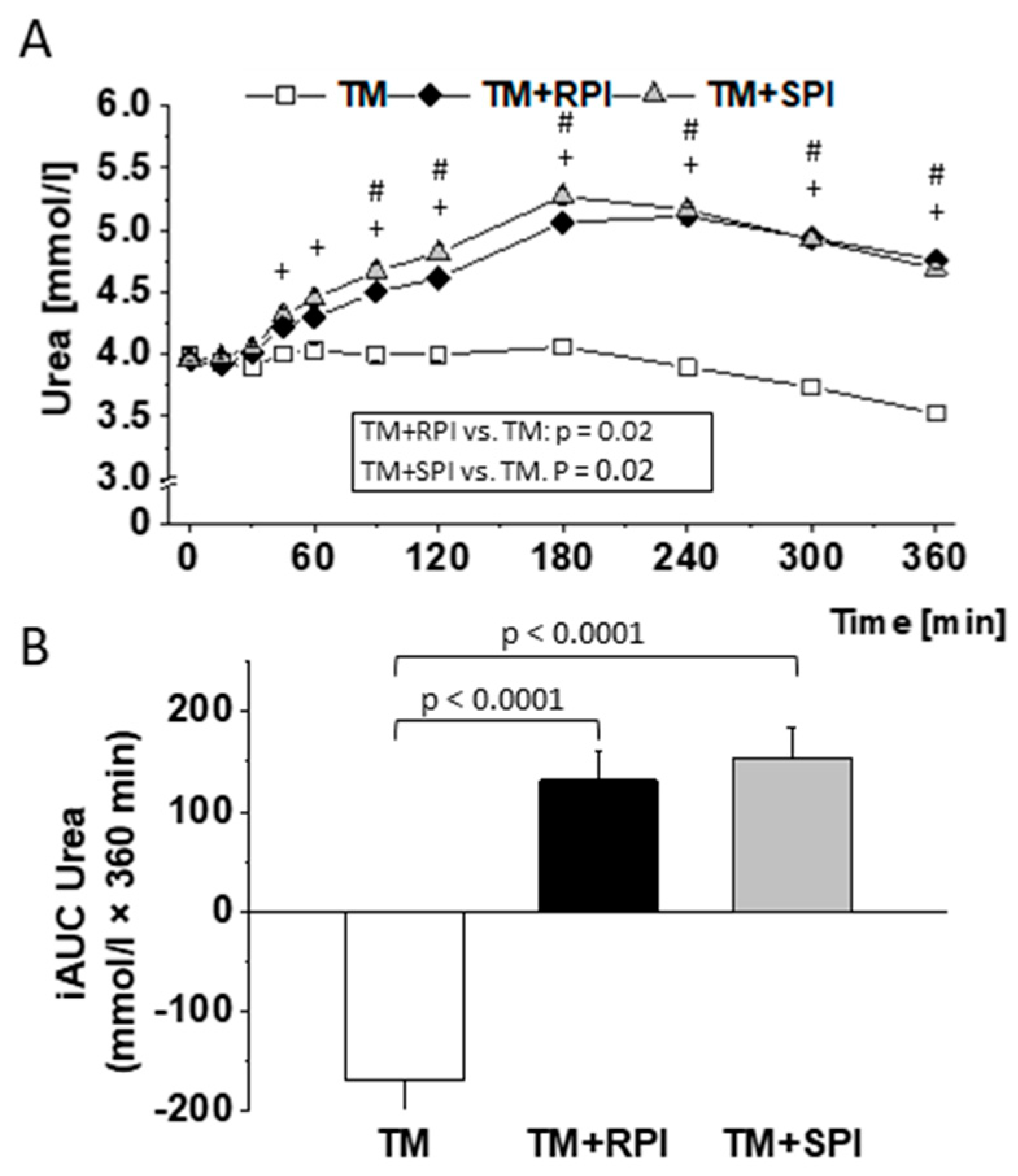
3.8. Postprandial Urea and Amino Acids
4. Discussion
Strengths and Limitations of the Study
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| FAO | Food and Agriculture Organization of the United Nations |
| FDA | Food and Drug Administration |
| iFGF23 | intact fibroblast growth factor 23 |
| hs-CRP | high-sensitivity C-reactive protein |
| iAUC | incremental area under the curve |
| IL-6 | interleukin-6 |
| LSM | least square mean |
| PTH | parathyroid hormone |
| RPI | rapeseed protein isolate |
| SPI | soy protein isolate |
| TM | test meal |
| VAS | visual analog scale |
| WHO | World Health Organization |
References
- White, R.R.; Hall, M.B. Nutritional and greenhouse gas impacts of removing animals from US agriculture. Proc. Natl. Acad. Sci. USA 2017, 114, E10301–E10308. [Google Scholar] [CrossRef]
- Sirtori, C.R.; Eberini, I.; Arnoldi, A. Hypocholesterolaemic effects of soya proteins: Results of recent studies are predictable from the Anderson meta-analysis data. Br. J. Nutr. 2007, 97, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Kashima, H.; Uemoto, S.; Eguchi, K.; Miura, A.; Kobayashi, T.; Fukuba, Y.; Endo, M.Y. Effect of soy protein isolate preload on postprandial glycemic control in healthy humans. Nutrition 2016, 32, 965–969. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Mouratidou, T.; Vereecken, C.; Kersting, M.; Bolca, S.; de Moraes, A.C.F.; Cuenca-García, M.; Moreno, L.A.; González-Gross, M.; Valtueña, J.; et al. Dietary animal and plant protein intakes and their associations with obesity and cardio-metabolic indicators in European adolescents: The HELENA cross-sectional study. Nutr. J. 2015, 14, 10. [Google Scholar] [CrossRef] [PubMed]
- Allison, D.B.; Gadbury, G.; Schwartz, L.G.; Murugesan, R.; Kraker, J.L.; Heshka, S.; Fontaine, K.R.; Heymsfield, S.B. A novel soy-based meal replacement formula for weight loss among obese individuals: A randomized controlled clinical trial. Eur. J. Clin. Nutr. 2003, 57, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Jääskeläinen, P.; Magnussen, C.G.; Pahkala, K.; Mikkilä, V.; Kahonen, M.; Sabin, M.A.; Fogelholm, M.; Hutri-Kähönen, N.; Taittonen, L.; Telama, R.; et al. Childhood nutrition in predicting metabolic syndrome in adults. Diabetes Care 2012, 35, 1937–1943. [Google Scholar] [CrossRef] [PubMed]
- Der Kuil, W.A.-V.; Engberink, M.F.; Brink, E.J.; Van Baak, M.A.; Bakker, S.J.L.; Navis, G.; Veer, P.V.T.; Geleijnse, J.M. Dietary protein and blood pressure: A systematic review. PLoS ONE 2010, 5, e12102. [Google Scholar] [CrossRef]
- Mattila, P.H.; Mäkinen, S.; Eurola, M.; Jalava, T.; Pihlava, J.-M.; Hellström, J.; Pihlanto, A. Nutritional value of commercial protein-rich plant products. Plant Foods Hum. Nutr. 2018, 73, 108–115. [Google Scholar] [CrossRef]
- Barth, C.A.B.; Metges Cornelia, C. Use of rapeseed protein in human nutrition. UFOP Schr. 2008, 1–9. [Google Scholar]
- Fleddermann, M.; Fechner, A.; Rößler, A.; Bähr, M.; Pastor, A.; Liebert, F.; Jahreis, G. Nutritional evaluation of rapeseed protein compared to soy protein for quality, plasma amino acids, and nitrogen balance—A randomized cross-over intervention study in humans. Clin. Nutr. 2013, 32, 519–526. [Google Scholar] [CrossRef]
- Patisaul, H.B.; Jefferson, W. The pros and cons of phytoestrogens. Front. Neuroendocr. 2010, 31, 400–419. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.H.; Lee, J.H.; Ahn, C.-W.; Park, S.-H.; Shim, S.-T.; Song, Y.D.; Han, E.N.; Lee, K.H.; Chae, J.S. Black soy peptide supplementation improves glucose control in subjects with prediabetes and newly diagnosed type 2 diabetes mellitus. J. Med. Food 2010, 13, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Hoie, L.H.; Guldstrand, M.; Sjöholm, Å.; Graubaum, H.J.; Gruenwald, J.; Zunft, H.J.F.; Lueder, W. Cholesterol-lowering effects of a new isolated soy protein with high levels of nondenaturated protein in hypercholesterolemic patients. Adv. Ther. 2007, 24, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Atabak, S.; Esmaillzadeh, A. Soy protein intake, cardiorenal indices, and c-reactive protein in type 2 diabetes with nephropathy: A longitudinal randomized clinical trial. Diabetes Care 2008, 31, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Matthan, N.R.; Jalbert, S.M.; Ausman, L.M.; Kuvin, J.T.; Karas, R.H.; Lichtenstein, A.H. Effect of soy protein from differently processed products on cardiovascular disease risk factors and vascular endothelial function in hypercholesterolemic subjects. Am. J. Clin. Nutr. 2007, 85, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Food Labeling, Health Claims, Soy Protein, and Coronary Heart Disease; Food and Drug Administration: Washington, DC, USA, 1999; pp. 699–733.
- Amarowicz, R.; Raab, B.; Shahidi, F. Antioxidant activity of phenolic fractions of rapeseed. J. Food Lipids 2003, 10, 51–62. [Google Scholar] [CrossRef]
- Marwede, V.; Schierholt, A.; Möllers, C.; Becker, H.C. Genotype × environment interactions and heritability of tocopherol contents in canola. Crop Sci. 2004, 44, 728–731. [Google Scholar] [CrossRef]
- Thiyam, U.; Stöckmann, H.; Felde, T.Z.; Schwarz, K. Antioxidative effect of the main sinapic acid derivatives from rapeseed and mustard oil by-products. Eur. J. Lipid Sci. Technol. 2006, 108, 239–248. [Google Scholar] [CrossRef]
- Gupta, A.K.; Savopoulos, C.; Ahuja, J.; I Hatzitolios, A. Role of phytosterols in lipid-lowering: Current perspectives. QJM: Int. J. Med. 2011, 104, 301–308. [Google Scholar] [CrossRef]
- Felker, P.; Bunch, R.; Leung, A.M. Concentrations of thiocyanate and goitrin in human plasma, their precursor concentrations in brassica vegetables, and associated potential risk for hypothyroidism. Nutr. Rev. 2016, 74, 248–258. [Google Scholar] [CrossRef]
- Jaafaru, M.S.; Karim, N.A.A.; Eliaser, E.M.; Rollin, P.; Mazzon, E.; Razis, A.F.A. Protective effect of glucosinolates hydrolytic products in Neurodegenerative Diseases (NDDs). Nutrients 2018, 10, 580. [Google Scholar] [CrossRef] [PubMed]
- Soundararajan, P.; Kim, J.S. Anti-carcinogenic glucosinolates in cruciferous vegetables and their antagonistic effects on prevention of cancers. Molecules 2018, 23, 2983. [Google Scholar] [CrossRef]
- Couzy, F.; Mansourian, R.; Labate, A.; Guinchard, S.; Montagne, D.H.; Dirren, H. Effect of dietary phytic acid on zinc absorption in the healthy elderly, as assessed by serum concentration curve tests. Br. J. Nutr. 1998, 80, 177–182. [Google Scholar] [CrossRef][Green Version]
- Hummel, M.; Talsma, E.; Taleon, V.; Londoño, L.; Brychkova, G.; Gallego, S.; Raatz, B.; Spillane, C. Iron, zinc and phytic acid retention of biofortified, low phytic acid, and conventional bean varieties when preparing common household recipes. Nutrients 2020, 12, 658. [Google Scholar] [CrossRef] [PubMed]
- Curran, J. The nutritional value and health benefits of pulses in relation to obesity, diabetes, heart disease and cancer. Br. J. Nutr. 2012, 108, S1–S2. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Liu, Z.; Yang, H.; Jue, Y. Acute effects of high-protein versus normal-protein isocaloric meals on satiety and ghrelin. Eur. J. Nutr. 2013, 53, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, J.; Craig, B.; Leidy, H.J.; Amankwaah, A.F.; Anguah, K.O.-B.; Jacobs, A.; Jones, B.L.; Jones, J.B.; Keeler, C.; Keller, C.; et al. The effects of increased protein intake on fullness: A meta-analysis and its limitations. J. Acad. Nutr. Diet. 2016, 116, 968–983. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Westman, E.; Mattes, R.D.; Wolfe, R.R.; Astrup, A.; Westerterp-Plantenga, M. Protein, weight management, and satiety. Am. J. Clin. Nutr. 2008, 87, 1558S–1561S. [Google Scholar] [CrossRef]
- Welborn, T.A.; Wearne, K. Coronary heart disease incidence and cardiovascular mortality in busselton with reference to glucose and insulin concentrations. Diabetes Care 1979, 2, 154–160. [Google Scholar] [CrossRef]
- Ceriello, A. Postprandial hyperglycemia and diabetes complications: Is it time to treat? Diabetes 2005, 54, 1–7. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Benn, M.; Schnohr, P.; Tybjærg-Hansen, A. Nonfasting triglycerides and risk of myocardial infarction, ischemic heart disease, and death in men and women. JAMA 2007, 298, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G.; Langsted, A.; Freiberg, J.J. Nonfasting hyperlipidemia and cardiovascular disease. Curr. Drug Targets 2009, 10, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R.H. Hyperlipidemia as a risk factor for cardiovascular disease. Prim. Care: Clin. Off. Pr. 2012, 40, 195–211. [Google Scholar] [CrossRef]
- Lenth, R.V. Statistical power calculations. J. Anim. Sci. 2007, 85, E24–E29. [Google Scholar] [CrossRef] [PubMed]
- Schopen, K.; Ewald, A.C.; Johannes, B.W.; Bloch, W.; Rittweger, J.; Frings-Meuthen, P. Short-term effects of lupin vs. whey supplementation on glucose and insulin responses to a standardized meal in a randomized cross-over trial. Front. Physiol. 2017, 8, 198. [Google Scholar] [CrossRef]
- Wanasundara, J.; McIntosh, T.C.; Perera, S.P.; Withana-Gamage, T.S.; Mitra, P. Canola/rapeseed protein-functionality and nutrition. OCl 2016, 23, D407. [Google Scholar] [CrossRef]
- Schwarz, M.; Lossow, K.; Kopp, J.F.; Schwerdtle, T.; Kipp, A.P. Crosstalk of Nrf2 with the trace elements selenium, iron, zinc, and copper. Nutrients 2019, 11, 2112. [Google Scholar] [CrossRef]
- Hara, A.; Radin, N.S. Lipid extraction of tissues with a low-toxicity solvent. Anal. Biochem. 1978, 90, 420–426. [Google Scholar] [CrossRef]
- Horwitz, W.; George, W.L. Official Methods of AOAC International: Official Method 993.21, 17th ed.; AOAC International: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Harland, B.F.; Oberleas, D. Phytate in Foods. World Rev. Nutr. Diet. 1987, 52, 235–259. [Google Scholar] [CrossRef]
- Kopp, J.; Müller, S.M.; Pohl, G.; Lossow, K.; Kipp, A.P.; Schwerdtle, T. A quick and simple method for the determination of six trace elements in mammalian serum samples using ICP-MS/MS. J. Trace Elem. Med. Boil. 2019, 54, 221–225. [Google Scholar] [CrossRef]
- Terrlink, T.; A Van Leeuwen, P.; Houdijk, A. Plasma amino acids determined by liquid chromatography within 17 minutes. Clin. Chem. 1994, 40, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Schuster, R. Determination of amino acids in biological, pharmaceutical, plant and food samples by automated precolumn derivatization and high-performance liquid chromatography. J. Chromatogr. B 1988, 431, 271–284. [Google Scholar] [CrossRef]
- Jones, B.; Kenward, M.G. Design and Analysis of Cross-Over Trials, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Kniskern, M.A.; Johnston, C.S. Protein dietary reference intakes may be inadequate for vegetarians if low amounts of animal protein are consumed. Nutrients 2011, 27, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Gorissen, S.H.M.; Crombag, J.J.R.; Senden, J.M.G.; Waterval, W.A.H.; Bierau, J.; Verdijk, L.B.; Van Loon, L.J.C. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acids 2018, 50, 1685–1695. [Google Scholar] [CrossRef]
- Boirie, Y.; Dangin, M.; Gachon, P.; Vasson, M.-P.; Maubois, J.-L.; Beaufrère, B. Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc. Natl. Acad. Sci. USA 1997, 94, 14930–14935. [Google Scholar] [CrossRef]
- Blanco, A.; Blanco, G. Amino acid metabolism. In Medical Biochemistry; Elsevier: Cambridge, MA, USA, 2017; pp. 367–399. [Google Scholar]
- Veldhorst, M.A.B.; Nieuwenhuizen, A.G.; Hochstenbach-Waelen, A.; Westerterp, K.R.; Engelen, M.P.K.J.; Brummer, R.-J.M.; Deutz, N.; Westerterp-Plantenga, M.S. Effects of high and normal soyprotein breakfasts on satiety and subsequent energy intake, including amino acid and ‘satiety’ hormone responses. Eur. J. Nutr. 2009, 48, 92–100. [Google Scholar] [CrossRef]
- Wang, B.; Kammer, L.M.; Ding, Z.; Lassiter, D.G.; Hwang, J.; Nelson, J.L.; Ivy, J.L. Amino acid mixture acutely improves the glucose tolerance of healthy overweight adults. Nutr. Res. 2012, 32, 30–38. [Google Scholar] [CrossRef]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; E Tang, J.; I Glover, E.; Wilkinson, S.B.; Prior, T.; A Tarnopolsky, M.; Phillips, S.M. Ingested protein dose response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2008, 89, 161–168. [Google Scholar] [CrossRef]
- Cuthbertson, D.J.; Smith, K.; Babraj, J.; Leese, G.; Waddell, T.; Atherton, P.; Wackerhage, H.; Taylor, P.M.; Rennie, M.J. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. FASEB J. 2004, 19, 1–22. [Google Scholar] [CrossRef]
- Witard, O.C.; Jackman, S.R.; Breen, L.; Smith, K.; Selby, A.; Tipton, K.D. Myofibrillar muscle protein synthesis rates subsequent to a meal in response to increasing doses of whey protein at rest and after resistance exercise. Am. J. Clin. Nutr. 2013, 99, 86–95. [Google Scholar] [CrossRef]
- Bendtsen, L.Q.; Lorenzen, J.K.; Bendsen, N.T.; Rasmussen, C.; Astrup, A. Effect of dairy proteins on appetite, energy expenditure, body weight, and composition: A review of the evidence from controlled clinical trials1. Adv. Nutr. 2013, 4, 418–438. [Google Scholar] [CrossRef] [PubMed]
- Smeets, A.J.; Soenen, S.; Luscombe-Marsh, N.D.; Ueland, Ø.; Westerterp-Plantenga, M.S. Energy expenditure, satiety, and plasma ghrelin, glucagon-like peptide 1, and peptide tyrosine-tyrosine concentrations following a single high-protein lunch. J. Nutr. 2008, 138, 698–702. [Google Scholar] [CrossRef] [PubMed]
- Bertenshaw, E.J.; Lluch, A.; Yeomans, M.R. Satiating effects of protein but not carbohydrate consumed in a between-meal beverage context. Physiol. Behav. 2008, 93, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Veldhorst, M.A.B.; Nieuwenhuizen, A.G.; Hochstenbach-Waelen, A.; Van Vught, A.J.; Westerterp, K.R.; Engelen, M.P.; Brummer, R.-J.M.; Deutz, N.; Westerterp-Plantenga, M.S. Dose-dependent satiating effect of whey relative to casein or soy. Physiol. Behav. 2009, 96, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Hochstenbach-Waelen, A.; Ab Veldhorst, M.; Nieuwenhuizen, A.G.; Westerterp-Plantenga, M.S.; Westerterp, K.R.; Veldhorst, M.A. Comparison of 2 diets with either 25% or 10% of energy as casein on energy expenditure, substrate balance, and appetite profile. Am. J. Clin. Nutr. 2009, 89, 831–838. [Google Scholar] [CrossRef]
- Akhavan, T.; Luhovyy, B.L.; Panahi, S.; Kubant, R.; Brown, P.H.; Anderson, G.H. Mechanism of action of pre-meal consumption of whey protein on glycemic control in young adults. J. Nutr. Biochem. 2014, 25, 36–43. [Google Scholar] [CrossRef]
- Nilsson, M.; Holst, J.J.; Björck, I.M. Metabolic effects of amino acid mixtures and whey protein in healthy subjects: Studies using glucose-equivalent drinks. Am. J. Clin. Nutr. 2007, 85, 996–1004. [Google Scholar] [CrossRef]
- Stevenson, E.J.; Allerton, D.M. The role of whey protein in postprandial glycaemic control. In Proceedings of the Nutrition Society; Cambridge University Press (CUP): Cambridge, UK, 2017; Volume 77, pp. 42–51. [Google Scholar]
- DECODE Study Group; European Diabetes Epidemiology Group. Glucose tolerance and cardiovascular mortality: Comparison of fasting and 2-hour diagnostic criteria. Arch. Intern. Med. 2001, 161, 397–405. [Google Scholar] [CrossRef]
- Balkau, B.; Shipley, M.; Jarrett, R.J.; Pyörälä, K.; Pyörälä, M.; Forhan, A.; Eschwège, E. High blood glucose concentration is a risk factor for mortality in middle-aged nondiabetic men. 20-year follow-up in the Whitehall Study, the Paris Prospective Study, and the Helsinki Policemen Study. Diabetes Care 1998, 21, 360–367. [Google Scholar] [CrossRef]
- Monnier, L.; Colette, C. Postprandial and basal hyperglycaemia in type 2 diabetes: Contributions to overall glucose exposure and diabetic complications. Diabetes Metab. 2015, 41, S9–S15. [Google Scholar] [CrossRef]
- Ingelsson, E.; Sundström, J.; Ärnlöv, J.; Zethelius, B.; Lind, L. Insulin resistance and risk of congestive heart failure. JAMA 2005, 294, 334–341. [Google Scholar] [CrossRef] [PubMed]
- Zilversmit, D.B. Atherogenesis: A postprandial phenomenon. Circulation 1979, 60, 473–485. [Google Scholar] [CrossRef]
- Bansal, S.; Buring, J.E.; Rifai, N.; Mora, S.; Sacks, F.M.; Ridker, P.M. Fasting compared with nonfasting triglycerides and risk of cardiovascular events in women. JAMA 2007, 298, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Holmer-Jensen, J.; Mortensen, L.S.; Astrup, A.; De Vrese, M.; Holst, J.J.; Thomsen, C.; Hermansen, K. Acute differential effects of dietary protein quality on postprandial lipemia in obese non-diabetic subjects. Nutr. Res. 2013, 33, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Mariotti, F.; Valette, M.; Lopez, C.; Fouillet, H.; Famelart, M.-H.; Mathé, V.; Airinei, G.; Benamouzig, R.; Gaudichon, C.; Tomé, D.; et al. Casein compared with whey proteins affects the organization of dietary fat during digestion and attenuates the postprandial triglyceride response to a mixed high-fat meal in healthy, overweight men. J. Nutr. 2015, 145, 2657–2664. [Google Scholar] [CrossRef] [PubMed]
- Mook-Kanamori, D.O.; Romisch-Margl, W.; Kastenmüller, G.; Prehn, C.; Petersen, A.K.; Illig, T.; Gieger, C.; Wang-Sattler, R.; Meisinger, C.; Peters, A.; et al. Increased amino acids levels and the risk of developing of hypertriglyceridemia in a 7-year follow-up. J. Endocrinol. Investig. 2014, 37, 369–374. [Google Scholar] [CrossRef]
- Teymoori, F.; Asghari, G.; Salehi, P.; Sadeghian, S.; Mirmiran, P.; Azizi, F. Are dietary amino acids prospectively predicts changes in serum lipid profile? Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 1837–1843. [Google Scholar] [CrossRef]
- Poppitt, S.D.; Keogh, G.F.; Lithander, F.E.; Wang, Y.; Mulvey, T.B.; Chan, Y.-K.; McArdle, B.H.; Cooper, G.J.S. Postprandial response of adiponectin, interleukin-6, tumor necrosis factor-α, and C-reactive protein to a high-fat dietary load. Nutrients 2008, 24, 322–329. [Google Scholar] [CrossRef]
- Willerson, J.T.; Ridker, P.M. Inflammation as a cardiovascular risk factor. Circulation 2004, 109, 2–10. [Google Scholar] [CrossRef]
- Lundman, P.; Boquist, S.; Samnegård, A.; Bennermo, M.; Held, C.; Ericsson, C.-G.; Silveira, A.; Hamsten, A.; Tornvall, P. A high-fat meal is accompanied by increased plasma interleukin-6 concentrations. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 195–202. [Google Scholar] [CrossRef]
- Miglio, C.; Peluso, I.; Raguzzini, A.; Villaño, D.V.; Cesqui, E.; Catasta, G.; Toti, E.; Serafini, M. Antioxidant and inflammatory response following high-fat meal consumption in overweight subjects. Eur. J. Nutr. 2012, 52, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Phillips, L.K.; Peake, J.M.; Zhang, X.; Hickman, I.J.; Briskey, D.R.; Huang, B.E.; Simpson, P.; Li, S.-H.; Whitehead, J.P.; Martin, J.H.; et al. Postprandial total and HMW adiponectin following a high-fat meal in lean, obese and diabetic men. Eur. J. Clin. Nutr. 2013, 67, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Kaisho, T.; Akira, S.; Ezekowitz, R.A.B.; Hoffmann, J.A. Toll-like receptors. Innate Immun. 2003, 21, 177–190. [Google Scholar] [CrossRef]
- Gressner, A.M.; Gressner, O.A. Interleukin-6. In Encyclopedia of Medical Laboratory Diagnostics; Gressner, A.M., Arndt, T., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; Volume 93, pp. 1265–1266. [Google Scholar]
- Cormick, G.; Ciapponi, A.; Cafferata, M.L.; Belizán, J.M. Calcium supplementation for prevention of primary hypertension. Cochrane Database Syst. Rev. 2015, 2015, 010037. [Google Scholar] [CrossRef]
- Bonovas, S.; Fiorino, G.; Lytras, T.; Malesci, A.; Danese, S. Calcium supplementation for the prevention of colorectal adenomas: A systematic review and meta-analysis of randomized controlled trials. World J. Gastroenterol. 2016, 22, 4594–4603. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.; Avenell, A.; Gamble, G.; Reid, I.R. Calcium supplements with or without vitamin D and risk of cardiovascular events: Reanalysis of the Women’s Health Initiative limited access dataset and meta-analysis. BMJ 2011, 342, d2040. [Google Scholar] [CrossRef]
- Ferreira, T.D.S.; Antunes, V.P.; Leal, P.M.; Sanjuliani, A.F.; Klein, M.R.S.T. The influence of dietary and supplemental calcium on postprandial effects of a high-fat meal on lipaemia, glycaemia, C-reactive protein and adiponectin in obese women. Br. J. Nutr. 2017, 118, 607–615. [Google Scholar] [CrossRef]
- Jones, K.W.; Eller, L.K.; Parnell, J.A.; Doyle-Baker, P.K.; Edwards, A.L.; A Reimer, R. Effect of a dairy- and calcium-rich diet on weight loss and appetite during energy restriction in overweight and obese adults: A randomized trial. Eur. J. Clin. Nutr. 2013, 67, 371–376. [Google Scholar] [CrossRef]
- Green, J.H.; Booth, C.; Bunning, R. Acute effect of high-calcium milk with or without additional magnesium, or calcium phosphate on parathyroid hormone and biochemical markers of bone resorption. Eur. J. Clin. Nutr. 2003, 57, 61–68. [Google Scholar] [CrossRef]
| Test Meal | Test Meal + 28 g RPI | Test Meal + 28 g SPI | |
|---|---|---|---|
| Calculated | |||
| Energy [kcal] | 693 | 808 | 802 |
| Protein [g] | 16 | 41 | 41 |
| Carbohydrates [g] | 93 | 93 | 93 |
| Fat [g] | 23.5 | 25.2 | 24.5 |
| Analyzed | |||
| Na [mg] | 519 | 681 | 832 |
| K [mg] | 489 | 500 | 544 |
| Ca [mg] | 92 | 193 | 80 |
| P [mg] | 234 | 364 | 439 |
| Zn [mg] | 2.2 | 2.6 | 2.8 |
| Cu [mg] | 0.6 | 6.2 | 0.9 |
| Rapeseed Protein Isolate | Soy Protein Isolate | |
|---|---|---|
| Dry Matter (DM) [%] | 93.6 | 94.1 |
| g/100 g DM | ||
| Crude protein | 92.6 | 90.6 |
| Crude fat | 6.08 | 3.71 |
| Crude ash | 4.32 | 4.68 |
| Phytic acid | 1.97 | 1.96 |
| Amino Acids | ||
| Alanine | 3.50 | 3.86 |
| Arginine | 5.64 | 7.00 |
| Aspartate | 5.55 | 10.3 |
| Cysteine | 2.68 | 1.05 |
| Glutamate | 20.6 | 18.0 |
| Serine | 3.46 | 4.60 |
| Glycine | 4.21 | 3.72 |
| Histidine | 3.11 | 2.81 |
| Isoleucine | 2.94 | 4.00 |
| Leucine | 6.49 | 7.36 |
| Lysine | 4.87 | 5.58 |
| Methionine | 1.82 | 1.21 |
| Phenylalanine | 3.17 | 4.49 |
| Proline | 6.22 | 4.44 |
| Threonine | 3.14 | 3.31 |
| Tryptophan | 1.01 | 0.98 |
| Tyrosine | 1.26 | 3.34 |
| Valine | 3.72 | 3.81 |
| Rapeseed Isolate | Soy Protein Isolate | |
|---|---|---|
| mg/100 g DM | ||
| Sodium | 650 | 980 |
| Potassium | 100 | 230 |
| Calcium | 490 | 50 |
| Phosphorus | 640 | 910 |
| Zinc | 2.1 | 2.8 |
| Copper | 22.1 | 1.2 |
| Characteristics | |
|---|---|
| Age [years] | 31 ± 3 (18, 64) |
| Male/female [%] | 25/75 |
| Body weight [kg] | 68.4 ± 3.1 (50, 108) |
| Body mass index [kg/m2] | 23.8 ± 0.7 (18.6, 30.1) |
| Blood pressure [mmHg] | |
| Systolic | 113 ± 4 (91, 156) |
| Diastolic | 77 ± 2 (64, 99) |
| Heart rate [beats/min] | 78 ± 2 (59, 94) |
| Glucose [mmol/L] | 4.9 ± 0.1 (4.2, 5.45) |
| Insulin [mIU/L] | 14.4 ± 14.0 (5.5, 25.9) |
| Triglycerides [mmol/L] | 0.9 ± 0.1 (0.5, 2.6) |
| Cholesterol [mmol/L] | 4.2 ± 0.3 (0.5, 6.4) |
| hsCRP [µg/mL] | 1.36 ± 0.40 (0.15, 6.52) |
| IL-6 [pg/mL] | 0.9 ± 0.1 (0.1, 9.0) |
| iFGF23 [pg/mL] | 43.9 ± 3.1 (25.0, 75.1) |
| PTH [pg/mL] | 30.4 ± 5.2 (11.4, 81.0) |
| Inorganic phosphate [mmol/L] | 1.19 ± 0.04 (0.8, 1.6) |
| Calcium [mmol/L] | 2.36 ± 0.06 (1.69, 2.65) |
| Zinc [µg/L] | 841 ± 53 (501, 1467) |
| Copper [µg/L] | 1158 ± 91 (658, 2321) |
| Urea [mmol/L] | 4.2 ± 0.2 (2.1, 5.9) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Volk, C.; Brandsch, C.; Schlegelmilch, U.; Wensch-Dorendorf, M.; Hirche, F.; Simm, A.; Gargum, O.; Wiacek, C.; Braun, P.G.; Kopp, J.F.; et al. Postprandial Metabolic Response to Rapeseed Protein in Healthy Subjects. Nutrients 2020, 12, 2270. https://doi.org/10.3390/nu12082270
Volk C, Brandsch C, Schlegelmilch U, Wensch-Dorendorf M, Hirche F, Simm A, Gargum O, Wiacek C, Braun PG, Kopp JF, et al. Postprandial Metabolic Response to Rapeseed Protein in Healthy Subjects. Nutrients. 2020; 12(8):2270. https://doi.org/10.3390/nu12082270
Chicago/Turabian StyleVolk, Christin, Corinna Brandsch, Ulf Schlegelmilch, Monika Wensch-Dorendorf, Frank Hirche, Andreas Simm, Osama Gargum, Claudia Wiacek, Peggy G. Braun, Johannes F. Kopp, and et al. 2020. "Postprandial Metabolic Response to Rapeseed Protein in Healthy Subjects" Nutrients 12, no. 8: 2270. https://doi.org/10.3390/nu12082270
APA StyleVolk, C., Brandsch, C., Schlegelmilch, U., Wensch-Dorendorf, M., Hirche, F., Simm, A., Gargum, O., Wiacek, C., Braun, P. G., Kopp, J. F., Schwerdtle, T., Treede, H., & Stangl, G. I. (2020). Postprandial Metabolic Response to Rapeseed Protein in Healthy Subjects. Nutrients, 12(8), 2270. https://doi.org/10.3390/nu12082270





