Effects of Dietary Nitrate Supplementation on Weightlifting Exercise Performance in Healthy Adults: A Systematic Review
Abstract
1. Introduction
2. Methodology
- Studies that were published as a full article (i.e., not a conference abstract) and performed in healthy humans (aged 18 to 65 years).
- Studies that included a NO3− and a placebo intervention.
- Studies which assessed voluntary dynamic resistance strength (i.e., not isometric or isokinetic strength and not involuntary muscle contractions evoked by NMES).
- Studies that included any of the following variables: i) one repetition maximum (1RM); ii) power or velocity movement; iii) number of repetitions to failure with submaximal loads.
3. Results and Discussion
3.1. The Effects of Dietary Nitrate Supplementation on Weightlifting Exercise Performance
3.2. Physiological Mechanisms
4. Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Joanisse, S.; Lim, C.; McKendry, J.; Mcleod, J.C.; Stokes, T.; Phillips, S.M. Recent advances in understanding resistance training-induced skeletal muscle hypertrophy in humans. F1000Research 2020, 9, F1000 Faculty-Rev-141. [Google Scholar] [CrossRef]
- Phillips, S.M. A brief review of critical processes in exercise-induced muscular hypertrophy. Sports Med. 2014, 44 (Suppl. S1), S71–S77. [Google Scholar] [CrossRef] [PubMed]
- Grgic, J.; Schoenfeld, B.J.; Davies, T.B.; Lazinica, B.; Krieger, J.W.; Pedisic, Z. Effect of Resistance Training Frequency on Gains in Muscular Strength: A Systematic Review and Meta-Analysis. Sports Med. 2018, 48, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.J.; Ogborn, D.; Krieger, J.W. Effect of repetition duration during resistance training on muscle hypertrophy: A systematic review and meta-analysis. Sports Med. 2015, 45, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.J.; Ogborn, D.; Krieger, J.W. Effects of Resistance Training Frequency on Measures of Muscle Hypertrophy: A Systematic Review and Meta-Analysis. Sports Med. 2016, 46, 1689–1697. [Google Scholar] [CrossRef] [PubMed]
- Kell, R.T.; Bell, G.; Quinney, A. Musculoskeletal fitness, health outcomes and quality of life. Sports Med. 2001, 31, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Swensen, T. Impact of resistance training on endurance performance. A new form of cross-training? Sports Med. 1998, 25, 191–200. [Google Scholar] [CrossRef]
- Baker, D.; Nance, S.; Moore, M. The load that maximizes the average mechanical power output during explosive bench press throws in highly trained athletes. J. Strength Cond. Res. 2001, 15, 20–24. [Google Scholar]
- Cronin, J.B.; Sleivert, G. Challenges in understanding the influence of maximal power training on improving athletic performance. Sports Med. 2005, 35, 213–234. [Google Scholar] [CrossRef]
- Alcaraz-Ibañez, M.; Rodríguez-Pérez, M. Effects of resistance training on performance in previously trained endurance runners: A systematic review. J. Sports Sci. 2018, 36, 613–629. [Google Scholar] [CrossRef]
- Blagrove, R.C.; Howatson, G.; Hayes, P.R. Effects of Strength Training on the Physiological Determinants of Middle- and Long-Distance Running Performance: A Systematic Review. Sports Med. 2018, 48, 1117–1149. [Google Scholar] [CrossRef] [PubMed]
- Bolger, R.; Lyons, M.; Harrison, A.J.; Kenny, I.C. Sprinting performance and resistance-based training interventions: A systematic review. J. Strength Cond. Res. 2015, 29, 1146–1156. [Google Scholar] [CrossRef] [PubMed]
- Muniz-Pardos, B.; Gomez-Bruton, A.; Matute-Llorente, A.; Gonzalez-Aguero, A.; Gomez-Cabello, A.; Gonzalo-Skok, O.; Casajus, J.A.; Vicente-Rodriguez, G. Swim-Specific Resistance Training: A Systematic Review. J. Strength Cond. Res. 2019, 33, 2875–2881. [Google Scholar] [CrossRef] [PubMed]
- Thiele, D.; Prieske, O.; Chaabene, H.; Granacher, U. Effect of strength training on physical fitness and sport-specific performance in recreational, sub-elite, and elite rowers: A systematic review with meta-analysis. J. Sports Sci. 2020. [Google Scholar] [CrossRef]
- Lauersen, J.B.; Andersen, T.E.; Andersen, L.B. Strength training as superior, dose-dependent and safe prevention of acute and overuse sports injuries: A systematic review, qualitative analysis and meta-analysis. Br. J. Sports Med. 2018, 52, 1557–1563. [Google Scholar] [CrossRef]
- Lopez, P.; Pinto, R.S.; Radaelli, R.; Rech, A.; Grazioli, R.; Izquierdo, M.; Cardore, E.L. Benefits of resistance training in physically frail elderly: A systematic review. Aging Clin. Exp. Res. 2018, 30, 889–899. [Google Scholar] [CrossRef]
- McLeod, J.C.; Stokes, T.; Phillips, S.M. Resistance exercise training as a primary countermeasure to age-related chronic disease. Front. Physiol. 2019, 10, 645. [Google Scholar] [CrossRef]
- Nakano, J.; Hashizume, K.; Fukushima, T.; Ueno, K.; Matsuura, E.; Ikio, Y.; Ishii, S.; Morishita, S.; Tanaka, K.; Kusuba, Y. Effects of Aerobic and Resistance Exercises on Physical Symptoms in Cancer Patients: A Meta-analysis. Integr. Cancer Ther. 2018, 17, 1048–1058. [Google Scholar] [CrossRef]
- Nery, C.; Moraes, S.R.A.; Novaes, K.A.; Bezerra, M.A.; Silveira, P.V.C.; Lemos, A. Effectiveness of resistance exercise compared to aerobic exercise without insulin therapy in patients with type 2 diabetes mellitus: A meta-analysis. Braz. J. Phys. Ther. 2017, 21, 400–415. [Google Scholar] [CrossRef]
- Borde, R.; Hortobágyi, T.; Granacher, U. Dose-Response Relationships of Resistance Training in Healthy Old Adults: A Systematic Review and Meta-Analysis. Sports Med. 2015, 45, 1693–1720. [Google Scholar] [CrossRef]
- Peitz, M.; Behringer, M.; Granacher, U. A systematic review on the effects of resistance and plyometric training on physical fitness in youth- What do comparative studies tell us? PLoS ONE 2018, 13, e0205525. [Google Scholar] [CrossRef]
- Schoenfeld, B.J. The mechanisms of muscle hypertrophy and their application to resistance training. J. Strength Cond. Res. 2010, 24, 2857–2872. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports Medicine. American College of Sports Medicine Position Stand. Progression Models in Resistance Training for Healthy Adults. Med. Sci. Sports Exerc. 2009, 41, 687–708. [Google Scholar] [CrossRef] [PubMed]
- Pareja-Blanco, F.; Rodríguez-Rosell, D.; Sánchez-Medina, L.; Gorostiaga, E.M.; González-Badillo, J.J. Effect of movement velocity during resistance training on neuromuscular performance. Int. J. Sports Med. 2014, 35, 916–924. [Google Scholar] [CrossRef]
- Jaric, S. Muscle strength testing: Use of normalisation for body size. Sports Med. 2002, 32, 615–631. [Google Scholar] [CrossRef]
- Tallis, J.; Duncan, M.J.; James, R.S. What can isolated skeletal muscle experiments tell us about the effects of caffeine on exercise performance? Br. J. Pharmacol. 2015, 172, 3703–3713. [Google Scholar] [CrossRef]
- Perrin, D.H. Isokinetic Exercise and Assessment; Human Kinetics Publishers: Champaign, IL, USA, 1993; ISBN 0873224647. [Google Scholar]
- Baker, D.; Wilson, G.; Carlyon, B. Generality versus specificity: A comparison of dynamic and isometric measures of strength and speed-strength. Eur. J. Appl. Physiol. Occup. Physiol. 1994, 68, 350–355. [Google Scholar] [CrossRef]
- Gentil, P.; Del Vecchio, F.B.; Paoli, A.; Schoenfeld, B.J.; Bottaro, M. Isokinetic Dynamometry and 1RM Tests Produce Conflicting Results for Assessing Alterations in Muscle Strength. J. Hum. Kinet. 2017, 56, 19–27. [Google Scholar] [CrossRef]
- Izquierdo, M.; Häkkinen, K.; Gonzalez-Badillo, J.J.; Ibáñez, J.; Gorostiaga, E.M. Effects of long-term training specificity on maximal strength and power of the upper and lower extremities in athletes from different sports. Eur. J. Appl. Physiol. 2002, 87, 264–271. [Google Scholar] [CrossRef]
- González-Badillo, J.J.; Sánchez-Medina, L. Movement velocity as a measure of loading intensity in resistance training. Int. J. Sports Med. 2010, 31, 347–352. [Google Scholar] [CrossRef]
- González-Badillo, J.J.; Marques, M.C.; Sánchez-Medina, L. The importance of movement velocity as a measure to control resistance training intensity. J. Hum. Kinet. 2011, 29A, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Pareja-Blanco, F.; Rodríguez-Rosell, D.; Sánchez-Medina, L.; Sanchis-Moysi, J.; Dorado, C.; Mora-Custodio, R.; Yáñez-García, J.M.; Morales-Alamo, D.; Pérez-Suárez, I.; Calbet, J.A.L.; et al. Effects of velocity loss during resistance training on athletic performance, strength gains and muscle adaptations. Scand. J. Med. Sci. Sports 2017, 27, 724–735. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 104–125. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.M.; Thompson, C.; Wylie, L.J.; Vanhatalo, A. Dietary nitrate and physical performance. Annu. Rev. Nutr. 2018, 38, 303–328. [Google Scholar] [CrossRef]
- Balsalobre-Fernández, C.; Romero-Moraleda, B.; Cupeiro, R.; Peinado, A.B.; Butragueño, J.; Benito, P.J. The effects of beetroot supplementation on exercise economy, rating of perceived exertion and running mechanics in elite distance runners: A double-blinded, randomized study. PLoS ONE 2018, 13, e0200517. [Google Scholar] [CrossRef]
- Lansley, K.E.; Winyard, P.G.; Fulford, J.; Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; DiMenna, F.J.; Gilchrist, M.; Benjamin, N.; Jones, A.M. Dietary nitrate supplementation reduces the O2 cost of walking and running: A place-controlled study. J. Appl. Physiol. 2011, 110, 591–600. [Google Scholar] [CrossRef]
- Murphy, M.; Eliot, K.; Heuertz, R.M.; Weiss, E. Whole beetroot consumption acutely improves running performance. J. Acad Nutr Diet. 2012, 112, 548–552. [Google Scholar] [CrossRef]
- Nyakayiru, J.; Jonvik, K.L.; Pinckaers, P.J.; Senden, J.; van Loon, L.J.C.; Verdijk, L.B. Beetroot juice supplementation improves high-intensity intermittent type exercise performance in trained soccer players. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 11–17. [Google Scholar] [CrossRef]
- Peacock, O.; Tjønna, A.E.; James, P.; Wisløff, U.; Welde, B.; Böhlke, N.; Smith, A.; Stokes, K.; Cook, C.; Sandbakk, O. Dietary nitrate does not enhance running performance in elite cross-country skiers. Med. Sci. Sports Exerc. 2012, 44, 2213–2219. [Google Scholar] [CrossRef]
- Porcelli, S.; Ramaglia, M.; Bellistri, G.; Pavei, G.; Pugliese, L.; Montorsi, M.; Rasica, L.; Mazorati, M. Aerobic fitness affects the exercise performance responses to nitrate supplementation. Med. Sci. Sports Exerc. 2015, 47, 1643–1651. [Google Scholar] [CrossRef]
- Porcelli, S.; Pugliese, L.; Rejc, E.; Pavei, G.; Bonato, M.; Montorsi, M.; La Torre, A.; Rasica, L.; Marzorati, M. Effects of a short-term high-nitrate diet on exercise performance. Nutrients 2016, 8, E534. [Google Scholar] [CrossRef] [PubMed]
- Sandbakk, S.B.; Sandbakk, Ø.; Peacock, O.; James, P.; Welde, B.; Stokes, K.; Böhlke, N.; Tjønna, A.E. Effects of acute supplementation of L-arginine and nitrate on endurance and sprint performance in elite athletes. Nitric Oxide 2015, 48, 10–15. [Google Scholar] [CrossRef]
- Shannon, O.M.; Duckworth, L.; Barlow, M.J.; Woods, D.; Lara, J.; Siervo, M.; O’Hara, J.P. Dietary nitrate supplementation enhances high-intensity running performance in moderate normobaric hypoxia, independent of aerobic fitness. Nitric Oxide 2016, 59, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Thompson, C.; Wylie, L.J.; Fulford, J.; Kelly, J.; Black, M.I.; McDonagh, S.T.; Jeukendrup, A.E.; Vanhatalo, A.; Jones, A.M. Dietary nitrate improves sprint performance and cognitive function during prolonged intermittent exercise. Eur. J. Appl. Physiol. 2015, 115, 1825–1834. [Google Scholar] [CrossRef] [PubMed]
- Thompson, C.; Vanhatalo, A.; Jell, H.; Fulford, J.; Carter, J.; Nyman, L.; Bailey, S.J.; Jones, A.M. Dietary nitrate supplementation improves sprint and high-intensity intermittent running performance. Nitric Oxide 2016, 61, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Wylie, L.J.; Mohr, M.; Krustrup, P.; Jackman, S.R.; Ermidis, G.; Kelly, J.; Black, M.I.; Bailey, S.J.; Vanhatalo, A.; Jones, A.M. Dietary nitrate supplementation improves team sport-specific intense intermittent exercise performance. Eur. J. Appl. Physiol. 2013, 113, 1673–1684. [Google Scholar] [CrossRef] [PubMed]
- Bond, H.; Morton, L.; Braakhuis, A.J. Dietary nitrate supplementation improves rowing performance in well-trained rowers. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 251–256. [Google Scholar] [CrossRef]
- Hoon, M.W.; Jones, A.M.; Johnson, N.A.; Blackwell, J.R.; Broad, E.M.; Lundy, B.; Rice, A.J.; Burke, L.M. The effect of variable doses of inorganic nitrate-rich beetroot juice on simulated 2000-m rowing performance in trained athletes. Int. J. Sports Physiol. Perform. 2014, 9, 615–620. [Google Scholar] [CrossRef]
- Peeling, P.; Cox, G.R.; Bullock, N.; Burke, L.M. Beetroot Juice Improves On-Water 500 M Time-Trial Performance, and Laboratory-Based Paddling Economy in National and International-Level Kayak Athletes. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 278–284. [Google Scholar] [CrossRef]
- Bailey, S.J.; Fulford, J.; Vanhatalo, A.; Winyard, P.G.; Blackwell, J.R.; DiMenna, F.J.; Wilkerson, D.P.; Benjamin, N.; Jones, A.M. Dietary nitrate supplementation enhances muscle contractile efficiency during knee-extensor exercise in humans. J. Appl. Physiol. 2010, 109, 135–148. [Google Scholar] [CrossRef]
- Aucouturier, J.; Boissière, J.; Pawlak-Chaouch, M.; Cuvelier, G.; Gamelin, F.-X. Effect of dietary nitrate supplementation on tolerance to supramaximal intensity intermittent exercise. Nitric Oxide 2015, 49, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Winyard, P.G.; Vanhatalo, A.; Blackwell, J.R.; DiMenna, F.J.; Wilkerson, D.P.; Tarr, J.; Benjamin, N.; Jones, A.M. Dietary nitrate supplementation reduces the O2cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. J. Appl. Physiol. 2009, 107, 1144–1155. [Google Scholar] [CrossRef] [PubMed]
- Bailey, S.J.; Varnham, R.L.; DiMenna, F.J.; Breese, B.C.; Wylie, L.J.; Jones, A.M. Inorganic nitrate supplementation improves muscle oxygenation, O2 uptake kinetics, and exercise tolerance at high but not low pedal rates. J. Appl. Physiol. 2015, 118, 1396–1405. [Google Scholar] [CrossRef]
- Breese, B.C.; McNarry, M.A.; Marwood, S.; Blackwell, J.R.; Bailey, S.J.; Jones, A.M. Beetroot juice supplementation speeds O2 uptake kinetics and improves exercise tolerance during severe-intensity exercise initiated from an elevated metabolic rate. Am. J. Physiol. Integr. Comp. Physiol. 2013, 305, R1441–R1450. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.M.; Gibala, M.J.; Van Loon, L. Nitrate supplementation’s improvement of 10-km time-trial performance in trained cyclists. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 64–71. [Google Scholar] [CrossRef]
- Jonvik, K.; Nyakayiru, J.; Van Dijk, J.-W.; Maase, K.; Ballak, S.; Senden, J.; Van Loon, L.J.C.; Verdijk, L.B. Repeated-sprint performance and plasma responses following beetroot juice supplementation do not differ between recreational, competitive and elite sprint athletes. Eur. J. Sport Sci. 2018, 18, 524–533. [Google Scholar] [CrossRef]
- Kelly, J.; Vanhatalo, A.; Wilkerson, D.P.; Wylie, L.; Jones, A.M. Effects of Nitrate on the Power–Duration Relationship for Severe-Intensity Exercise. Med. Sci. Sports Exerc. 2013, 45, 1798–1806. [Google Scholar] [CrossRef]
- Lansley, K.E.; Winyard, P.; Bailey, S.J.; Vanhatalo, A.; Wilkerson, D.P.; Blackwell, J.R.; Gilchrist, M.; Benjamin, N.; Jones, A.M. Acute Dietary Nitrate Supplementation Improves Cycling Time Trial Performance. Med. Sci. Sports Exerc. 2011, 43, 1125–1131. [Google Scholar] [CrossRef]
- Rimer, E.G.; Peterson, L.R.; Coggan, A.R.; Martin, J.C. Increase in Maximal Cycling Power with Acute Dietary Nitrate Supplementation. Int. J. Sports Physiol. Perform. 2016, 11, 715–720. [Google Scholar] [CrossRef]
- Rokkedal-Lausch, T.; Franch, J.; Poulsen, M.K.; Thomsen, L.P.; Weitzberg, I.; Kamavuako, E.N.; Karbing, D.S.; Larsen, R.G. Chronic high-dose beetroot juice supplementation improves time trial performance of well-trained cyclists in normoxia and hypoxia. Nitric Oxide 2019, 85, 44–52. [Google Scholar] [CrossRef]
- Thompson, C.; Wylie, L.J.; Blackwell, J.R.; Fulford, J.; Black, M.I.; Kelly, J.; McDonagh, S.T.J.; Carter, J.; Bailey, S.J.; Vanhatalo, A.; et al. Influence of dietary nitrate supplementation on physiological and muscle metabolic adaptations to sprint interval training. J. Appl. Physiol. 2016, 122, 642–652. [Google Scholar] [CrossRef] [PubMed]
- Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; DiMenna, F.J.; Pavey, T.; Wilkerson, D.P.; Benjamin, N.; Winyard, P.G.; Jones, A.M. Acute and chronic effects of dietary nitrate supplementation on blood pressure and the physiological responses to moderate-intensity and incremental exercise. Am. J. Physiol. Integr. Comp. Physiol. 2010, 299, R1121–R1131. [Google Scholar] [CrossRef] [PubMed]
- Wylie, L.J.; Kelly, J.; Bailey, S.J.; Blackwell, J.R.; Skiba, P.F.; Winyard, P.G.; Jeukendrup, A.E.; Vanhatalo, A.; Jones, A.M. Beetroot juice and exercise: Pharmacodynamic and dose-response relationships. J. Appl. Physiol. 2013, 115, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Boorsma, R.K.; Whitfield, J.; Spriet, L.L. Beetroot juice supplementation does not improve performance of elite 1500-m runners. Med. Sci. Sports Exerc. 2014, 46, 2326–2334. [Google Scholar] [CrossRef]
- Callahan, M.J.; Parr, E.B.; Hawley, J.A.; Burke, L.M. Single and Combined Effects of Beetroot Crystals and Sodium Bicarbonate on 4-km Cycling Time Trial Performance. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.M.; Res, P.; Stinkens, R.; Lundberg, J.O.; Gibala, M.J.; Van Loon, L. No improvement in endurance performance after a single dose of beetroot juice. Int. J. Sport Nutr. Exerc. Metab. 2012, 22, 470–478. [Google Scholar] [CrossRef]
- De Castro, T.F.; Manoel, F.D.A.; Machado, F. Beetroot juice supplementation does not modify the 3-km running performance in untrained women. Sci. Sports 2018, 33, e167–e170. [Google Scholar] [CrossRef]
- Lowings, S.; Shannon, O.M.; Deighton, K.; Matu, J.; Barlow, M.J. Effect of Dietary Nitrate Supplementation on Swimming Performance in Trained Swimmers. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 377–384. [Google Scholar] [CrossRef]
- McQuillan, J.A.; Dulson, D.K.; Laursen, P.B.; Kilding, A.E. Dietary Nitrate Fails to Improve 1 and 4 km Cycling Performance in Highly Trained Cyclists. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 255–263. [Google Scholar] [CrossRef]
- Mosher, S.L.; Gough, L.A.; Deb, S.; Saunders, B.; McNaughton, L.R.; Brown, D.R.; Sparks, S. High dose Nitrate ingestion does not improve 40 km cycling time trial performance in trained cyclists. Res. Sports Med. 2019, 28, 138–146. [Google Scholar] [CrossRef]
- Oskarsson, J.; McGawley, K. No individual or combined effects of caffeine and beetroot-juice supplementation during submaximal or maximal running. Appl. Physiol. Nutr. Metab. 2018, 43, 697–703. [Google Scholar] [CrossRef]
- Wickham, K.A.; McCarthy, D.G.; Pereira, J.M.; Cervone, D.T.; Verdijk, L.B.; Van Loon, L.J.C.; Power, G.A.; Spriet, L.L. No effect of beetroot juice supplementation on exercise economy and performance in recreationally active females despite increased torque production. Physiol. Rep. 2019, 7, e13982. [Google Scholar] [CrossRef]
- Wilkerson, D.P.; Hayward, G.M.; Bailey, S.J.; Vanhatalo, A.; Blackwell, J.R.; Jones, A.M. Influence of acute dietary nitrate supplementation on 50 mile time trial performance in well-trained cyclists. Graefe’s Arch. Clin. Exp. Ophthalmol. 2012, 112, 4127–4134. [Google Scholar] [CrossRef]
- Domínguez, R.; Cuenca, E.; Maté-Muñoz, J.L.; Garca-Fernández, P.; Paya, N.S.; Lozano-Estevan, M.D.C.; Veiga-Herreros, P.; Garnacho-Castaño, M.V. Effects of Beetroot Juice Supplementation on Cardiorespiratory Endurance in Athletes. A Systematic Review. Nutrition 2017, 9, 43. [Google Scholar] [CrossRef] [PubMed]
- McMahon, N.F.; Leveritt, M.; Pavey, T. The Effect of Dietary Nitrate Supplementation on Endurance Exercise Performance in Healthy Adults: A Systematic Review and Meta-Analysis. Sports Med. 2016, 47, 735–756. [Google Scholar] [CrossRef] [PubMed]
- Van De Walle, G.P.; Vukovich, M.D. The Effect of Nitrate Supplementation on Exercise Tolerance and Performance. J. Strength Cond. Res. 2018, 32, 1796–1808. [Google Scholar] [CrossRef] [PubMed]
- Domínguez, R.; Maté-Muñoz, J.L.; Cuenca, E.; Garca-Fernández, P.; Ordoñez, F.M.; Estevan, M.C.L.; Veiga-Herreros, P.; Da Silva, S.F.; Garnacho-Castaño, M.V. Effects of beetroot juice supplementation on intermittent high-intensity exercise efforts. J. Int. Soc. Sports Nutr. 2018, 15, 2. [Google Scholar] [CrossRef] [PubMed]
- Spiegelhalder, B.; Eisenbrand, G.; Preussmann, R. Influence of dietary nitrate on nitrite content of human saliva: Possible relevance to in vivo formation of N-nitroso compounds. Food Cosmet. Toxicol. 1976, 14, 545–548. [Google Scholar] [CrossRef]
- Cosby, K.; Partovi, K.S.; Crawford, J.H.; Patel, R.P.; Reiter, C.D.; Martyr, S.; Yang, B.K.; Waclawiw, M.A.; Zalos, G.; Xu, X.; et al. Nitrite reduction to nitric oxide by deoxyhemoglobin vasodilates the human circulation. Nat. Med. 2003, 9, 1498–1505. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, A.V.; Dietrich, B.; Nohl, H. Various intracellular compartments cooperate in the release of nitric oxide from glycerol trinitrate in liver. Br. J. Pharmacol. 2003, 139, 989–997. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Cui, H.; Kundu, T.K.; Alzawahra, W.; Zweier, J.L. Nitric Oxide Production from Nitrite Occurs Primarily in Tissues Not in the Blood. J. Biol. Chem. 2008, 283, 17855–17863. [Google Scholar] [CrossRef] [PubMed]
- Shiva, S.; Huang, Z.; Grubina, R.; Sun, J.; Ringwood, L.A.; MacArthur, P.H.; Xu, X.; Murphy, E.; Darley-Usmar, V.; Gladwin, M.T. Deoxymyoglobin is a nitrite reductase that generates nitric oxide and regulates mitochondrial respiration. Circ. Res. 2007, 100, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Naughton, D.P.; Blake, D.R.; Benjamin, N.; Stevens, C.R.; Winyard, P.G.; Symons, M.C.; Harrison, R. Human xanthine oxidase converts nitrite ions into nitric oxide (NO). Biochem. Soc. Trans. 1997, 25, 524S. [Google Scholar] [CrossRef] [PubMed]
- Castello, P.R.; David, P.S.; McClure, T.; Crook, Z.R.; Poyton, R.O. Mitochondrial cytochrome oxidase produces nitric oxide under hypoxic conditions: Implications for oxygen sensing and hypoxic signaling in eukaryotes. Cell Metab. 2006, 3, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Modin, A.; Björne, H.; Herulf, M.; Alving, K.; Weitzberg, E.; Lundberg, J. Nitrite-derived nitric oxide: A possible mediator of ‘acidic-metabolic’ vasodilation. Acta Physiol. Scand. 2001, 171, 9–16. [Google Scholar] [CrossRef]
- Richardson, R.S.; Noyszewski, E.A.; Kendrick, K.F.; Leigh, J.S.; Wagner, P.D. Myoglobin O2 desaturation during exercise. Evidence of limited O2 transport. J. Clin. Investig. 1995, 96, 1916–1926. [Google Scholar] [CrossRef]
- Gilliard, C.N.; Lam, J.K.; Cassel, K.S.; Park, J.W.; Schechter, A.N.; Piknova, B. Effect of dietary nitrate levels on nitrate fluxes in rat skeletal muscle and liver. Nitric Oxide 2018, 75, 1–7. [Google Scholar] [CrossRef]
- Piknova, B.; Park, J.W.; Swanson, K.M.; Dey, S.; Noguchi, C.T.; Schechter, A.N. Skeletal muscle as an endogenous nitrate reservoir. Nitric Oxide 2015, 47, 10–16. [Google Scholar] [CrossRef]
- Piknova, B.; Park, J.W.; Lam, K.K.J.; Schechter, A.N. Nitrate as a source of nitrite and nitric oxide during exercise hyperemia in rat skeletal muscle. Nitric Oxide 2016, 55, 54–61. [Google Scholar] [CrossRef]
- Wylie, L.J.; Park, J.W.; Vanhatalo, A.; Kadach, S.; Black, M.I.; Stoyanov, Z.; Schechter, A.N.; Jones, A.M.; Piknova, B. Human skeletal muscle nitrate store: Influence of dietary nitrate supplementation and exercise. J. Physiol. 2019, 597, 5565–5576. [Google Scholar] [CrossRef]
- Webb, A.J.; Patel, N.; Loukogeorgakis, S.; Okorie, M.; Aboud, Z.; Misra, S.; Rashid, R.; Miall, P.; Deanfield, J.; Benjamin, N.; et al. Acute blood pressure lowering, vasoprotective, and antiplatelet properties of dietary nitrate via bioconversion to nitrite. Hypertension 2008, 51, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Dreißigacker, U.; Wendt, M.; Wittke, T.-C.; Tsikas, D.; Maassen, N. Positive correlation between plasma nitrite and performance during high-intensive exercise but not oxidative stress in healthy men. Nitric Oxide 2010, 23, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Fulford, J.; Winyard, P.G.; Vanhatalo, A.; Bailey, S.J.; Blackwell, J.R.; Jones, A.M. Influence of dietary nitrate supplementation on human skeletal muscle metabolism and force production during maximum voluntary contractions. Pflüg. Arch. 2013, 465, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Haider, G.; Folland, J.P. Nitrate Supplementation Enhances the Contractile Properties of Human Skeletal Muscle. Med. Sci. Sports Exerc. 2014, 46, 2234–2243. [Google Scholar] [CrossRef]
- Whitfield, J.; Gamu, D.; Heigenhauser, G.J.F.; Van Loon, L.; Spriet, L.L.; Tupling, A.R.; Holloway, G.P. Beetroot Juice Increases Human Muscle Force without Changing Ca2+-Handling Proteins. Med. Sci. Sports Exerc. 2017, 49, 2016–2024. [Google Scholar] [CrossRef]
- Coggan, A.R.; Leibowitz, J.L.; Kadkhodayan, A.; Thomas, D.P.; Ramamurthy, S.; Spearie, C.A.; Waller, S.; Farmer, M.; Peterson, L.R. Effect of acute dietary nitrate intake on maximal knee extensor speed and power in healthy men and women. Nitric Oxide 2015, 48, 16–21. [Google Scholar] [CrossRef]
- Hernández, A.; Schiffer, T.A.; Ivarsson, N.; Cheng, A.J.; Bruton, J.D.; Lundberg, J.O.; Weitzberg, I.; Westerblad, H. Dietary nitrate increases tetanic [Ca2+]i and contractile force in mouse fast-twitch muscle. J. Physiol. 2012, 590, 3575–3583. [Google Scholar] [CrossRef]
- Morton, R.; Sonne, M.W.; Zuniga, A.F.; Mohammad, I.; Jones, A.; McGlory, C.; Keir, P.J.; Potvin, J.R.; Phillips, S.M. Muscle fibre activation is unaffected by load and repetition duration when resistance exercise is performed to task failure. J. Physiol. 2019, 597, 4601–4613. [Google Scholar] [CrossRef]
- Bickel, C.S.; Gregory, C.M.; Dean, J.C. Motor unit recruitment during neuromuscular electrical stimulation: A critical appraisal. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 111, 2399–2407. [Google Scholar] [CrossRef]
- Henneman, E. Relation between Size of Neurons and Their Susceptibility to Discharge. Science 1957, 126, 1345–1347. [Google Scholar] [CrossRef]
- Allen, D.G.; Lamb, G.D.; Westerblad, H. Skeletal Muscle Fatigue: Cellular Mechanisms. Physiol. Rev. 2008, 88, 287–332. [Google Scholar] [CrossRef] [PubMed]
- Tillin, N.A.; Moudy, S.; Nourse, K.M.; Tyler, C.J. Nitrate supplement benefits contractile forces in fatigued but not unfatigued muscle. Med. Sci. Sports Exerc. 2018, 50, 2122–2131. [Google Scholar] [CrossRef] [PubMed]
- Bogdanis, G.C.; Nevill, M.E.; Boobis, L.H.; Lakomy, H.K.; Nevill, A. Recovery of power output and muscle metabolites following 30 s of maximal sprint cycling in man. J. Physiol. 1995, 482, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Bogdanis, G.C.; Nevill, M.E.; Boobis, L.H.; Lakomy, H.K. Contribution of phosphocreatine and aerobic metabolism to energy supply during repeated sprint exercise. J. Appl. Physiol. 1996, 80, 876–884. [Google Scholar] [CrossRef]
- Gaitanos, G.C.; Williams, C.; Boobis, L.H.; Brooks, S. Human muscle metabolism during intermittent maximal exercise. J. Appl. Physiol. 1993, 75, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Trump, M.E.; Heigenhauser, G.J.; Putman, C.T.; Spriet, L.L. Importance of muscle phosphocreatine during intermittent maximal cycling. J. Appl. Physiol. 1996, 80, 1574–1580. [Google Scholar] [CrossRef]
- Vanhatalo, A.; Fulford, J.; Bailey, S.J.; Blackwell, J.R.; Winyard, P.G.; Jones, A.M. Dietary nitrate reduces muscle metabolic perturbation and improves exercise tolerance in hypoxia. J. Physiol. 2011, 589, 5517–5528. [Google Scholar] [CrossRef]
- Ferguson, S.K.; Hirai, D.M.; Copp, S.W.; Holdsworth, C.T.; Allen, J.; Jones, A.M.; Musch, T.I.; Poole, D.C. Impact of dietary nitrate supplementation via beetroot juice on exercising muscle vascular control in rats. J. Physiol. 2012, 591, 547–557. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. The PRISMA-P Group Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ 2015, 349, g7647. [Google Scholar] [CrossRef]
- Maher, C.G.; Sherrington, C.; Herbert, R.D.; Moseley, A.M.; Elkins, M.R. Reliability of the PEDro Scale for Rating Quality of Randomized Controlled Trials. Phys. Ther. 2003, 83, 713–721. [Google Scholar] [CrossRef]
- Brown, W.R.; Brunnhuber, K.; Chalkidou, K.; Chalmers, I.; Clarke, M.; Fenton, M.; Forbes, C.; Glanville, J.; Hicks, N.J.; Moody, J.; et al. How to formulate research recommendations. BMJ 2006, 333, 804–806. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.D.; Martin, M.P.; Mintz, J.A.; Rogers, R.R.; Ballmann, C.G. Effect of Acute Beetroot Juice Supplementation on Bench Press Power, Velocity, and Repetition Volume. J. Strength Cond. Res. 2020, 34, 924–928. [Google Scholar] [CrossRef] [PubMed]
- Ranchal-Sanchez, A.; Diaz-Bernier, V.M.; De La Florida-Villagran, C.A.; Llorente-Cantarero, F.J.; Campos-Perez, J.; Jurado-Castro, J.M. Acute Effects of Beetroot Juice Supplements on Resistance Training: A Randomized Double-Blind Crossover. Nutrients 2020, 12, 1912. [Google Scholar] [CrossRef]
- Flanagan, S.D.; Looney, D.P.; Miller, M.J.S.; Dupont, W.H.; Pryor, L.; Creighton, B.C.; Sterczala, A.J.; Szivak, T.K.; Hooper, D.R.; Maresh, C.M.; et al. The Effects of Nitrate-Rich Supplementation on Neuromuscular Efficiency during Heavy Resistance Exercise. J. Am. Coll. Nutr. 2016, 35, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Mosher, S.L.; Sparks, S.A.; Williams, E.L.; Bentley, D.; McNaughton, L.R. Ingestion of a Nitric Oxide Enhancing Supplement Improves Resistance Exercise Performance. J. Strength Cond. Res. 2016, 30, 3520–3524. [Google Scholar] [CrossRef] [PubMed]
- Del Coso, J.; Salinero, J.J.; González-Millán, C.; Abián-Vicén, J.; Pérez-González, B. Dose response effects of a caffeine-containing energy drink on muscle performance: A repeated measures design. J. Int. Soc. Sports Nutr. 2012, 9, 21. [Google Scholar] [CrossRef]
- Mora-Rodriguez, R.; Pallarés, J.G.; López-Gullón, J.M.; López-Samanes, Á.; Fernández-Elías, V.E.; Ortega, J.F. Improvements on neuromuscular performance with caffeine ingestion depend on the time-of-day. J. Sci. Med. Sport 2015, 18, 338–342. [Google Scholar] [CrossRef]
- Pallarés, J.G.; Fernández-Elías, V.E.; Ortega, J.F.; Muñoz, G.; Muñoz-Guerra, J.; Mora-Rodriguez, R. Neuromuscular Responses to Incremental Caffeine Doses. Med. Sci. Sports Exerc. 2013, 45, 2184–2192. [Google Scholar] [CrossRef]
- Maté-Muñoz, J.L.; Lougedo, J.H.; Garnacho-Castaño, M.V.; Veiga-Herreros, P.; Estevan, M.D.C.L.; Garca-Fernández, P.; De Jesús, F.; Guodemar-Pérez, J.; Juan, A.F.S.; Domínguez, R. Effects of β-alanine supplementation during a 5-week strength training program: A randomized, controlled study. J. Int. Soc. Sports Nutr. 2018, 15, 19. [Google Scholar] [CrossRef]
- Venier, S.; Grgic, J.; Mikulic, P.; Veiner, S. Acute Enhancement of Jump Performance, Muscle Strength, and Power in Resistance-Trained Men After Consumption of Caffeinated Chewing Gum. Int. J. Sports Physiol. Perform. 2019, 14, 1415–1421. [Google Scholar] [CrossRef]
- Romero-Moraleda, B.; Del Coso, J.; Gutiérrez-Hellín, J.; Lara, B. The Effect of Caffeine on the Velocity of Half-Squat Exercise during the Menstrual Cycle: A Randomized Controlled Trial. Nutrients 2019, 11, 2662. [Google Scholar] [CrossRef] [PubMed]
- Coggan, A.R.; Broadstreet, S.R.; Mikhalkova, D.; Bole, I.; Leibowitz, J.L.; Kadkhodayan, A.; Park, S.; Thomas, D.P.; Thies, D.; Peterson, L.R. Dietary nitrate-induced increases in human muscle power: High versus low responders. Physiol. Rep. 2018, 6, e13575. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.M.; Ferguson, S.K.; Bailey, S.J.; Vanhatalo, A.; Poole, D.C. Fiber Type-Specific Effects of Dietary Nitrate. Exerc. Sport Sci. Rev. 2016, 44, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Ørtenblad, N.; Nielsen, J.; Boushel, R.; Söderlund, K.; Saltin, B.; Holmberg, H.-C. The Muscle Fiber Profiles, Mitochondrial Content, and Enzyme Activities of the Exceptionally Well-Trained Arm and Leg Muscles of Elite Cross-Country Skiers. Front. Physiol. 2018, 9, 1031. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.L.; Aagaard, P. Myosin heavy chain IIX overshoot in human skeletal muscle. Muscle Nerve 2000, 23, 1095–1104. [Google Scholar] [CrossRef]
- Andersen, L.L.; Andersen, J.L.; Zebis, M.K.; Aagaard, P. Early and late rate of force development: Differential adaptive responses to resistance training? Scand. J. Med. Sci. Sports 2010, 20, e162–e169. [Google Scholar] [CrossRef]
- Coggan, A.R.; Hoffman, R.L.; Gray, D.A.; Moorthi, R.N.; Thomas, D.P.; Leibowitz, J.L.; Thies, D.; Peterson, L.R. A Single Dose of Dietary Nitrate Increases Maximal Knee Extensor Angular Velocity and Power in Healthy Older Men and Women. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2019, 75, 1154–1160. [Google Scholar] [CrossRef]
- Cuenca, E.; Jodra, P.; Pérez-López, A.; Rodríguez, L.G.G.; Da Silva, S.F.; Veiga-Herreros, P.; Domínguez, R. Effects of Beetroot Juice Supplementation on Performance and Fatigue in a 30-s All-Out Sprint Exercise: A Randomized, Double-Blind Cross-Over Study. Nutrients 2018, 10, 1222. [Google Scholar] [CrossRef]
- Domínguez, R.; Garnacho-Castaño, M.V.; Cuenca, E.; García-Fernández, P.; Muñoz-González, A.; De-Jesús-Franco, F.; Estevan, M.C.L.; Da Silva, S.F.; Veiga-Herreros, P.; Maté-Muñoz, J.L. Effects of Beetroot Juice Supplementation on a 30-s High-Intensity Inertial Cycle Ergometer Test. Nutrients 2017, 9, 1360. [Google Scholar] [CrossRef]
- Jodra, P.; Domínguez, R.; Sánchez-Oliver, A.J.; Veiga-Herreros, P.; Bailey, S.J. Effect of Beetroot Juice Supplementation on Mood, Perceived Exertion, and Performance During a 30-Second Wingate Test. Int. J. Sports Physiol. Perform. 2020, 15, 243–248. [Google Scholar] [CrossRef]
- Kramer, S.J.; Baur, D.A.; Spicer, M.T.; Vukovich, M.D.; Ormsbee, M. The effect of six days of dietary nitrate supplementation on performance in trained CrossFit athletes. J. Int. Soc. Sports Nutr. 2016, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wylie, L.J.; Bailey, S.J.; Kelly, J.; Blackwell, J.R.; Vanhatalo, A.; Jones, A.M. Influence of beetroot juice supplementation on intermittent exercise performance. Graefe’s Arch. Clin. Exp. Ophthalmol. 2015, 116, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Andrade, F.H.; Reid, M.B.; Allen, D.G.; Westerblad, H. Effect of hydrogen peroxide and dithiothreitol on contractile function of single skeletal muscle fibres from the mouse. J. Physiol. 1998, 509, 565–575. [Google Scholar] [CrossRef]
- Bailey, S.J.; Gandra, P.G.; Jones, A.M.; Hogan, M.C.; Nogueira, L. Incubation with sodium nitrite attenuates fatigue development in intact single mouse fibres at physiological. J. Physiol. 2019, 597, 5429–5443. [Google Scholar] [CrossRef] [PubMed]
- Nyakayiru, J.; Kouw, I.W.; Cermak, N.M.; Senden, J.M.; Van Loon, L.J.C.; Verdijk, L.B. Sodium nitrate ingestion increases skeletal muscle nitrate content in humans. J. Appl. Physiol. 2017, 123, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Stamler, J.S.; Meissner, G. Physiology of nitric oxide in skeletal muscle. Physiol. Rev. 2001, 81, 209–237. [Google Scholar] [CrossRef]
- Stamler, J.S. Redox signaling: Nitrosylation and related target interactions of nitric oxide. Cell 1994, 78, 931–936. [Google Scholar] [CrossRef]
- Evangelista, A.M.; Rao, V.S.; Filo, A.R.; Marozkina, N.V.; Doctor, A.; Jones, D.R.; Gaston, B.; Guilford, W. Direct Regulation of Striated Muscle Myosins by Nitric Oxide and Endogenous Nitrosothiols. PLoS ONE 2010, 5, e11209. [Google Scholar] [CrossRef]
- Nogueira, L.; Figueiredo-Freitas, C.; Casimiro-Lopes, G.; Magdesian, M.H.; Assreuy, J.; Sorenson, M.M. Myosin is reversibly inhibited by S-nitrosylation. Biochem. J. 2009, 424, 221–231. [Google Scholar] [CrossRef]
- Dutka, T.L.; Mollica, J.P.; Lamboley, C.R.; Weerakkody, V.C.; Greening, D.W.; Posterino, G.S.; Murphy, R.M.; Lamb, G.D. S-nitrosylation and S-glutathionylation of Cys134 on troponin I have opposing competitive actions on Ca2+ sensitivity in rat fast-twitch muscle fibers. Am. J. Physiol. Physiol. 2017, 312, C316–C327. [Google Scholar] [CrossRef]
- Ishii, T.; Sunami, O.; Saitoh, N.; Nishio, H.; Takeuchi, T.; Hata, F. Inhibition of skeletal muscle sarcoplasmic reticulum Ca2+-ATPase by nitric oxide. FEBS Lett. 1998, 440, 218–222. [Google Scholar] [CrossRef]
- Eu, J.P.; Sun, J.; Xu, L.; Stamler, J.S.; Meissner, G. The Skeletal Muscle Calcium Release Channel. Cell 2000, 102, 499–509. [Google Scholar] [CrossRef]
- Stoyanovsky, D.; Murphy, T.; Anno, P.R.; Kim, Y.-M.; Salama, G. Nitric oxide activates skeletal and cardiac ryanodine receptors. Cell Calcium 1997, 21, 19–29. [Google Scholar] [CrossRef]
- Gould, N.; Doulias, P.-T.; Tenopoulou, M.; Raju, K.; Ischiropoulos, H. Regulation of Protein Function and Signaling by Reversible Cysteine S-Nitrosylation*. J. Biol. Chem. 2013, 288, 26473–26479. [Google Scholar] [CrossRef] [PubMed]
- Takeshima, H.; Nishimura, S.; Matsumoto, T.; Ishida, H.; Kangawa, K.; Minamino, N.; Matsuo, H.; Ueda, M.; Hanaoka, M.; Hirose, T.; et al. Primary structure and expression from complementary DNA of skeletal muscle ryanodine receptor. Nature 1989, 339, 439–445. [Google Scholar] [CrossRef] [PubMed]
- Thompson, C.; Vanhatalo, A.; Kadach, S.; Wylie, L.J.; Fulford, J.; Ferguson, S.K.; Blackwell, J.R.; Bailey, S.J.; Jones, A.M. Discrete physiological effects of beetroot juice and potassium nitrate supplementation following 4-wk sprint interval training. J. Appl. Physiol. 2018, 124, 1519–1528. [Google Scholar] [CrossRef]
- Coffey, V.G.; Hawley, J.A. Concurrent exercise training: Do opposites distract? J. Physiol. 2016, 595, 2883–2896. [Google Scholar] [CrossRef]
- Fyfe, J.J.; Bishop, D.; Stepto, N.K. Interference between Concurrent Resistance and Endurance Exercise: Molecular Bases and the Role of Individual Training Variables. Sports Med. 2014, 44, 743–762. [Google Scholar] [CrossRef]
- Pride, C.K.; Mo, L.; Quesnelle, K.M.; Dagda, R.K.; Murillo, D.; Geary, L.; Corey, C.; Portella, R.; Zharikov, S.; Croix, C.S.; et al. Nitrite activates protein kinase A in normoxia to mediate mitochondrial fusion and tolerance to ischaemia/reperfusion. Cardiovasc. Res. 2013, 101, 57–68. [Google Scholar] [CrossRef]
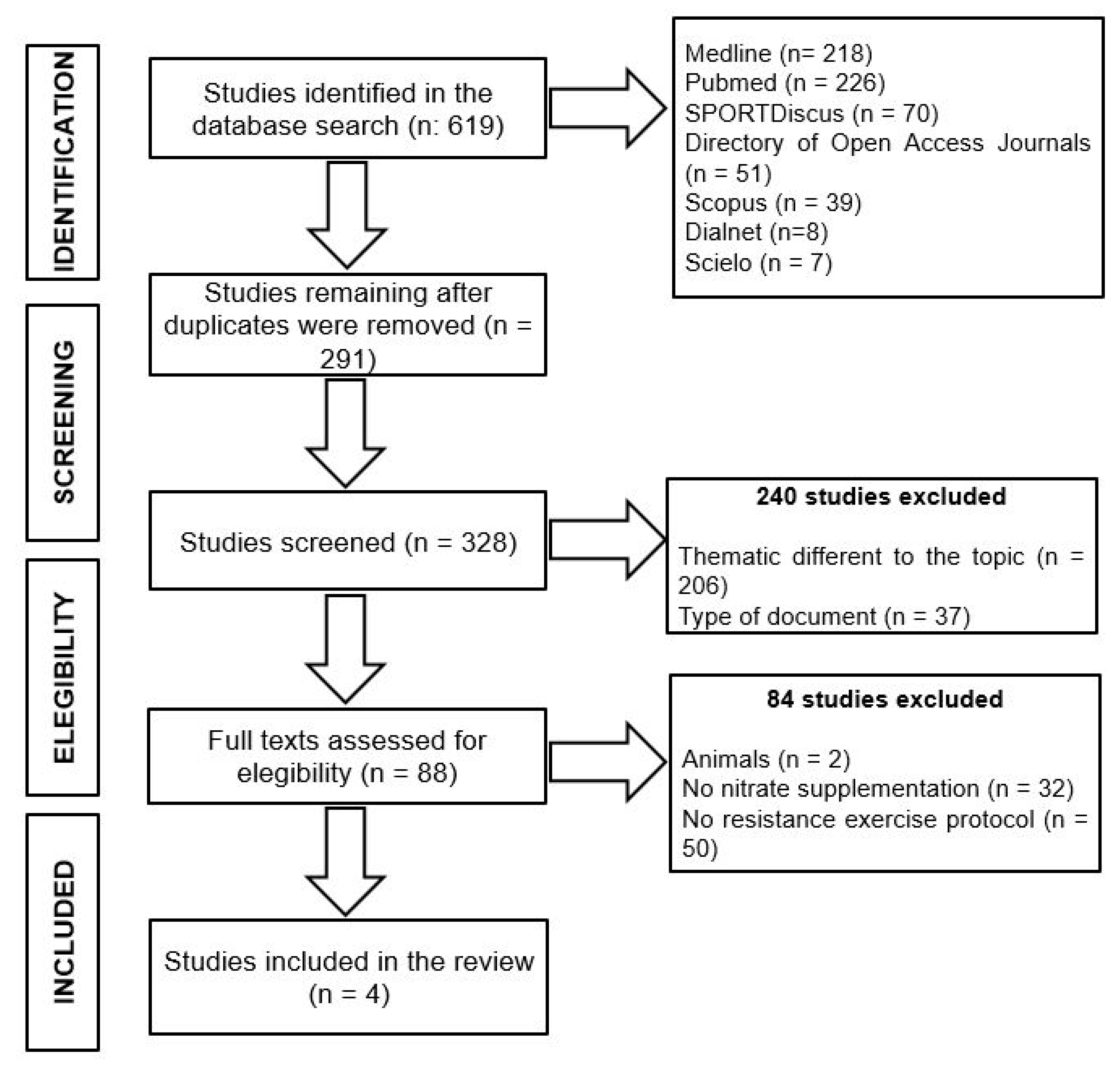
| Reference | Subjects | Supplementation | Exercise Protocol | Findings |
|---|---|---|---|---|
| Flanagan et al. (2016) [115] | Fourteen resistance-trained men | Three days and 60 min prior to exercise ingestion of 2 × NO3−-rich bars (32.5 mg NO3−·d−1) | Smith machine box squats: three sets x 3-s isometric squats interspersed with 120-s rest, then dynamic box squats @ 60%1RM with 10% increases up to 90%1RM, then 10% decreases to 60%1RM, then RTF on last 60%1RM set | ↔ RTF: −1.5% (599 ± 5 vs. 608 ± 5 reps) ↑ EMG amplitude: +5% (83 ± 3 vs. 79 ± 4%) |
| Mosher et al. (2016) [116] | Twelve resistance-trained men | Six days of 1 × 70 mL NO3− rich BR supplementation (~6.4 mmol NO3−·d−1) | Smith machine bench press: three sets of RTF @ 60%1RM interspersed with 2 min of recovery between sets | ↑ RTF: +19.4% ↑ total weight lifted: +18.9% (2583 ± 864 vs. 2172 ± 721 kg) |
| Williams et al. (2020) [113] | Eleven resistance-trained men | Two hours prior to exercise ingestion of 1 × 70 mL NO3− rich BR (~6.4 mmol NO3−) | Free-weight bench press: two sets x 2 explosive reps, 5 min rest, then three sets x RTF @ 70%1RM interspersed with 2 min of recovery between sets | ↑ RTF: +10.7% (31 ± 6 vs. 28 ± 6 reps) ↑ Pmean: +19.5% (607 ± 112 vs. 508 ± 118 W) ↑ Vmean: +6.5% (0.66 ± 0.08 vs. 0.62 ± 0.08 m·s−1) |
| Ranchal-Sanchez et al. (2020) [114] | Twelve resistance-trained men | Two hours prior to exercise ingestion of 1 × 70 mL NO3− rich BR (~6.4 mmol NO3−) | Smith machine bench press and back squat: three sets x RTF @ 60−70−80%1RM with 2 min of recovery between sets. After the eccentric phase of each rep, participants rested for 1.0−1.5 s | ↑ RTF back squat: +23.4% (60 ± 20 vs. 46 ± 16 reps) ↑ RTF total (sum bench press and back squat): +17.7% (89 ± 25 vs. 75 ± 21 reps) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
San Juan, A.F.; Dominguez, R.; Lago-Rodríguez, Á.; Montoya, J.J.; Tan, R.; Bailey, S.J. Effects of Dietary Nitrate Supplementation on Weightlifting Exercise Performance in Healthy Adults: A Systematic Review. Nutrients 2020, 12, 2227. https://doi.org/10.3390/nu12082227
San Juan AF, Dominguez R, Lago-Rodríguez Á, Montoya JJ, Tan R, Bailey SJ. Effects of Dietary Nitrate Supplementation on Weightlifting Exercise Performance in Healthy Adults: A Systematic Review. Nutrients. 2020; 12(8):2227. https://doi.org/10.3390/nu12082227
Chicago/Turabian StyleSan Juan, Alejandro F., Raul Dominguez, Ángel Lago-Rodríguez, Juan José Montoya, Rachel Tan, and Stephen J. Bailey. 2020. "Effects of Dietary Nitrate Supplementation on Weightlifting Exercise Performance in Healthy Adults: A Systematic Review" Nutrients 12, no. 8: 2227. https://doi.org/10.3390/nu12082227
APA StyleSan Juan, A. F., Dominguez, R., Lago-Rodríguez, Á., Montoya, J. J., Tan, R., & Bailey, S. J. (2020). Effects of Dietary Nitrate Supplementation on Weightlifting Exercise Performance in Healthy Adults: A Systematic Review. Nutrients, 12(8), 2227. https://doi.org/10.3390/nu12082227









