Choline: An Essential Nutrient for Skeletal Muscle
Abstract
1. Introduction
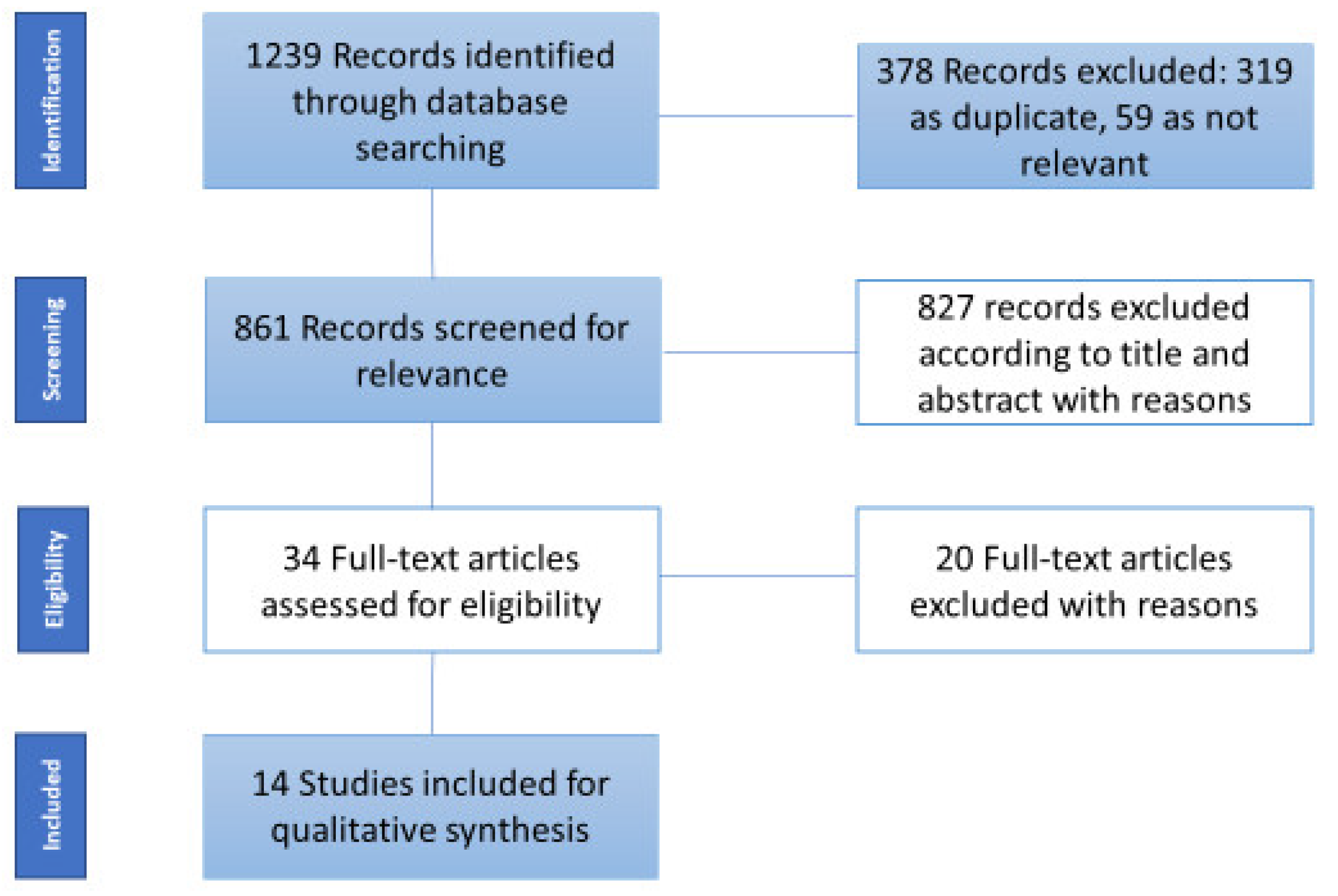
2. Materials and Methods
3. Results
3.1. Pre-Clinical Studies
3.1.1. In Vitro Studies
3.1.2. Animal Studies
3.2. Clinical Studies
4. Discussion
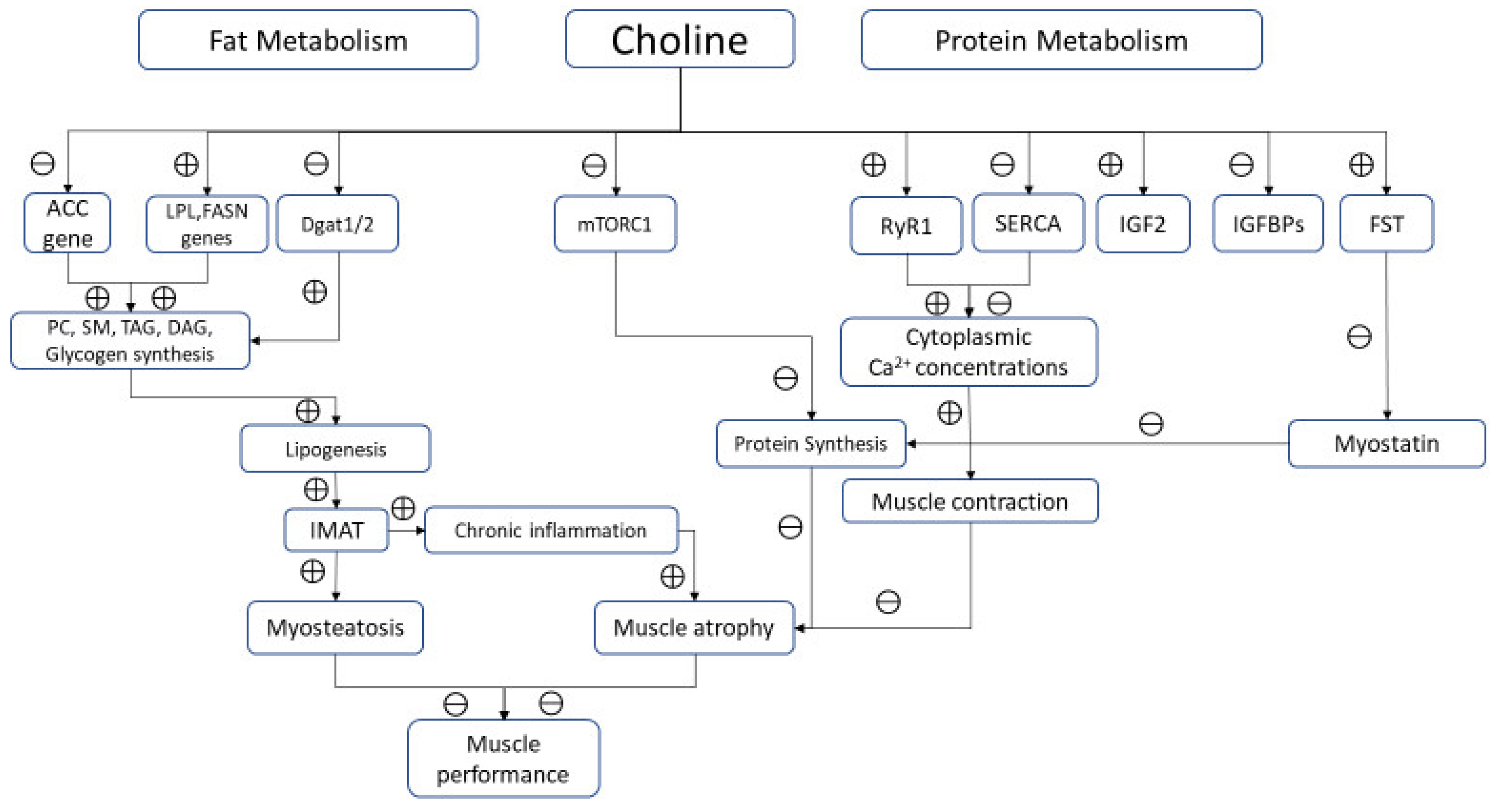
4.1. Choline and Muscle Fat Metabolism
4.2. Choline and Muscle Proteins
4.3. Choline and Inflammation
4.4. Choline, Apoptosis, and Autophagy
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press (US): Washington, DC, USA, 1998; pp. 390–442. [CrossRef]
- Nan, A. Miscellaneous Drugs, Materials, Medical Devices and Techniques. In Side Effects of Drugs Annual, 1st ed.; Sidhartha, D.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2015; Volume 38, pp. 603–619. [Google Scholar]
- EFSA. NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific opinion on Dietary Reference Value for choline. EFSA J. 2016, 14, 1–70. [Google Scholar]
- Cai, C.X.; Carlos, S.; Solaimani, P.; Trivedi, B.J.; Tran, C.; Castelino-Prabhu, S. Nutritional and Dietary Interventions for Nonalcoholic Fatty Liver Disease. Diet. Interv. Liver Dis. 2019, 29, 357–372. [Google Scholar]
- da Costa, K.A.; Badea, M.; Fischer, L.M.; Zeisel, S.H. Elevated serum creatine phosphokinase in choline-deficient humans: Mechanistic studies in C2C12 mouse myoblasts. Am. J. Clin. Nutr. 2004, 80, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Alves, F.M.; Caldow, M.K.; Trieu, J.; Naim, T.; Montgomery, M.K.; Watt, M.J.; Lynch, G.S.; Koopman, R. Choline administration attenuates aspects of the dystrophic pathology in mdx mice. Clin. Nutr. Exp. 2019, 24, 83–91. [Google Scholar] [CrossRef]
- Zhao, D.; Frohman, M.A.; Blusztajn, J.K. Generation of choline for acetylcholine synthesis by phospholipase D isoforms. BMC Neurosci. 2001, 2, 16. [Google Scholar] [CrossRef] [PubMed]
- Penry, J.T.; Manore, M.M. Choline: An important micronutrient for maximal endurance-exercise performance? Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 191–203. [Google Scholar] [CrossRef]
- Hongu, N.; Sachan, D.S. Carnitine and choline supplementation with exercise alter carnitine profiles, biochemical markers of fat metabolism and serum leptin concentration in healthy women. J. Nutr. 2003, 133, 84–89. [Google Scholar] [CrossRef]
- Jäger, R.; Purpura, M.; Kingsley, M. Phospholipids and sports performance. J. Int. Soc. Sports Nutr. 2007, 4, 5. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Beca, S.; Aschar-Sobbi, R.; Ponjevic, D.; Winkfein, R.J.; Kargacin, M.E.; Kargacin, G.J. Effects of monovalent cations on Ca2+ uptake by skeletal and cardiac muscle sarcoplasmic reticulum. Arch. Biochem. Biophys. 2009, 490, 110–117. [Google Scholar] [CrossRef]
- Kovacs, E.; Xu, L.; Pasek, D.A.; Liliom, K.; Meissner, G. Regulation of ryanodine receptors by sphingosylphosphorylcholine: Involvement of both calmodulin-dependent and -independent mechanisms. Biochem. Biophys. Res. Commun. 2010, 401, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Michel, V.; Singh, R.K.; Bakovic, M. The impact of choline availability on muscle lipid metabolism. Food Funct. 2011, 2, 53–62. [Google Scholar] [CrossRef]
- Kenney, J.L.; Carlberg, K.A. The effect of choline and myo-inositol on liver and carcass fat levels in aerobically trained rats. Int. J. Sports Med. 1995, 16, 114–116. [Google Scholar] [CrossRef]
- Eder, K. The effect of a combined dietary treatment with cholesterol and cholic acid on the lipid metabolism of geese at low or high choline concentrations. Arch. Anim. Nutr. 1999, 52, 285–297. [Google Scholar] [CrossRef] [PubMed]
- Ilcol, Y.O.; Yilmaz, Z.; Ulus, I.H. Endotoxin alters serum-free choline and phospholipid-bound choline concentrations, and choline administration attenuates endotoxin-induced organ injury in dogs. Shock 2005, 24, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Schenkel, L.C.; Bakovic, M. Palmitic acid and oleic acid differentially regulate choline transporter-like 1 levels and glycerolipid metabolism in skeletal muscle cells. Lipids 2014, 49, 731–744. [Google Scholar] [CrossRef]
- Li, H.; Wang, H.; Yu, L.; Wang, M.; Liu, S.; Chen, Q. Effects of supplementation of rumen-protected choline on growth performance, meat quality and gene expression in longissimus dorsi muscle of lambs. Arch. Anim. Nutr. 2015, 69, 340–350. [Google Scholar] [CrossRef]
- Oster, M.; Nuchchanart, W.; Trakooljul, N.; Muráni, E.; Zeyner, A.; Wirthgen, E.; Hoeflich, A.; Ponsuksili, S.; Wimmers, K. Methylating micronutrient supplementation during pregnancy influences foetal hepatic gene expression and IGF signalling and increases foetal weight. Eur. J. Nutr. 2016, 55, 1717–1727. [Google Scholar] [CrossRef]
- Robinson, J.L.; Harding, S.V.; Brunton, J.A.; Bertolo, R.F. Dietary Methyl Donors Contribute to Whole-Body Protein Turnover and Protein Synthesis in Skeletal Muscle and the Jejunum in Neonatal Piglets. J. Nutr. 2016, 146, 2007–2012. [Google Scholar] [CrossRef]
- Taylor, A.; Schenkel, L.C.; Yokich, M.; Bakovic, M. Adaptations to excess choline in insulin resistant and Pcyt2 deficient skeletal muscle. Biochem. Cell Biol. 2017, 95, 223–231. [Google Scholar] [CrossRef]
- Jahanian, R.; Ashnagar, M. Effects of dietary supplementation of choline and carnitine on growth performance, meat oxidative stability and carcass composition of broiler chickens fed diets with different metabolisable energy levels. Br. Poult. Sci. 2018, 59, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Fischer, L.M.; DaCosta, K.A.; Kwock, L.; Stewart, P.W.; Lu, T.S.; Stabler, S.P.; Allen, R.H.; Zeisel, S.H. Sex and menopausal status influence human dietary requirements for the nutrient choline. Am. J. Clin. Nutr. 2007, 85, 1275–1285. [Google Scholar] [CrossRef] [PubMed]
- McLean, B.D.; Buttifant, D.; Gore, C.J.; White, K.; Liess, C.; Kemp, J. Physiological and performance responses to a preseason altitude-training camp in elite team-sport athletes. Int. J. Sports Physiol. Perform. 2013, 8, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Vance, D.E. Phosphatidylcholine and choline homeostasis. J. Lipid Res. 2008, 49, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H.; Da Costa, K.A.; Franklin, P.D.; Alexander, E.A.; Lamont, J.T.; Sheard, N.F.; Beiser, A. Choline, an essential nutrient for humans. FASEB J. 1991, 5, 2093–2098. [Google Scholar] [CrossRef]
- Wortmann, S.B.; Mayr, J.A. Choline-related-inherited metabolic diseases-A mini review. J. Inherit. Metab. Dis. 2019, 42, 237–242. [Google Scholar] [CrossRef]
- Zeisel, S.H.; Da Costa, K.A. Choline: An essential nutrient for public health. Nutr. Rev. 2009, 67, 615–623. [Google Scholar] [CrossRef]
- Joseph, G.A.; Wang, S.X.; Jacobs, C.E.; Zhou, W.; Kimble, G.C.; Tse, H.W.; Eash, J.K.; Shavlakadze, T.; Glass, D.J. Partial Inhibition of mTORC1 in Aged Rats Counteracts the Decline in Muscle Mass and Reverses Molecular Signaling Associated with Sarcopenia. Mol. Cell. Biol. 2019, 39, e00141-19. [Google Scholar] [CrossRef]
- Iolascon, G.; Moretti, A. The relationship between serum IGF-1, handgrip strength, physical performance and falls in elderly men and women. Eur. J. Endocrinol. 2018, 3, 103. [Google Scholar] [CrossRef]
- Kalinkovich, A.; Livshits, G. Sarcopenic obesity or obese sarcopenia: A cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of the pathogenesis. Ageing Res. Rev. 2017, 35, 200–221. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wang, Y.; Randell, E.; Pedram, P.; Yi, Y.; Gulliver, W.; Sun, G. Higher Dietary Choline and Betaine Intakes Are Associated with Better Body Composition in the Adult Population of Newfoundland, Canada. PLoS ONE. 2016, 11, e0155403. [Google Scholar] [CrossRef] [PubMed]
- DeVol, D.L.; Rotwein, P.; Sadow, J.L.; Novakofski, J.; Bechtel, P.J. Activation of insulin-like growth factor gene expression during work-induced skeletal muscle growth. Am. J. Physiol. 1990, 259, E89–E95. [Google Scholar] [CrossRef] [PubMed]
- Arazi, H.; Salek, L.; Nikfal, E.; Izadi, M.; Tufano, J.J.; Elliott, B.T.; Brughelli, M. Comparable endocrine and neuromuscular adaptations to variable vs. constant gravity-dependent resistance training among young women. J. Transl. Med. 2020, 18, 239. [Google Scholar] [CrossRef] [PubMed]
- Amthor, H.; Nicholas, G.; McKinnell, I.; Kemp, C.F.; Sharma, M.; Kambadur, R.; Patel, K. Follistatin complexes Myostatin and antagonises Myostatin-mediated inhibition of myogenesis. Dev. Biol. 2004, 270, 19–30. [Google Scholar] [CrossRef]
- Ilcol, Y.O.; Gurun, M.S.; Taga, Y.; Ulus, I.H. Choline increases serum insulin in rat when injected intraperitoneally and augments basal and stimulated aceylcholine release from the rat minced pancreas in vitro. Eur. J. Biochem. 2003, 270, 991–999. [Google Scholar] [CrossRef] [PubMed]
- Downing, J.E.; Miyan, J.A. Neural immunoregulation: Emerging roles for nerves in immune homeostasis and disease. Immunol. Today 2000, 21, 281–289. [Google Scholar] [CrossRef]
- da Costa, K.A.; Niculescu, M.D.; Craciunescu, C.N.; Fischer, L.M.; Zeisel, S.H. Choline deficiency increases lymphocyte apoptosis and DNA damage in humans. Am. J. Clin. Nutr. 2006, 84, 88–94. [Google Scholar] [CrossRef]
- Rabanal-Ruiz, Y.; Otten, E.G.; Korolchuk, V.I. mTORC1 as the main gateway to autophagy. Essays Biochem. 2017, 61, 565–584. [Google Scholar] [CrossRef]
- Shea, L.; Raben, N. Autophagy in skeletal muscle: Implications for Pompe disease. Int. J. Clin. Pharmacol. Ther. 2009, 47, S42–S47. [Google Scholar] [CrossRef]
- Hang, P.; Zhao, J.; Su, Z.; Sun, H.; Chen, T.; Zhao, L.; Du, Z. Choline Inhibits Ischemia-Reperfusion-Induced Cardiomyocyte Autophagy in Rat Myocardium by Activating Akt/mTOR Signaling. Cell. Physiol. Biochem. 2018, 45, 2136–2144. [Google Scholar] [CrossRef]
| Author, Year | Design | Main Results |
|---|---|---|
| Beca et al. 2009 [12] | In vitro | K+ replacement with Choline+ had inhibitory effects on the Vmax of Ca2+ uptake and, directly, on SERCA activity of SR in both canine cardiac and rabbit skeletal muscle. |
| Kovacs et al. 2010 [13] | In vitro | SPC levels below CMC directly inhibited skeletal muscle RyR1. SPC levels above CMC displaced inhibitory CaM from RyR1 increasing its activity. |
| Michel et al. 2011 [14] | In vitro | Choline deficiency adversely influenced incorporation of pre-existing FA and DAG for PC synthesis, increasing TAG synthesis; CD downregulated mRNA level of choline-transporter SLC44A1. |
| Kenney et al. 1995 [15] | Animal study | Choline (0.18%) and myo-inositol (0.5%) consumption compared to control group, determined significant difference on liver fat percent (6.68 ± 2.23% vs. 9.22 ± 2.91%) and no difference on carcass fat percentage (24.04 ± 3.36% vs. 25.122 ± 5.83%), respectively. |
| Eder 1999 [16] | Animal study | Hyperlipidemic diet associated to low choline concentrations diet intake did not influence PC levels suggesting sufficient choline endogenous synthesis in geese. |
| Ilcol et al. 2005 [17] | Animal study | At 2 to 6 h after 0.02 mg/kg intravenous endotoxin infusion, serum-free choline concentration decreased by 14% to 49% and serum phospholipid-bound choline concentrations increased by 19% to 27%; up to 48 h after 1 mg/kg endotoxin, both serum-free and phospholipid-bound choline concentrations increased by 23% to 98% and by 18% to 53% respectively. Intravenous administration of choline (20 mg/kg) 5 min before, and up to 8 h after 1 mg/kg of endotoxin seems to reduce endotoxin-induced tissue injury, in particular on skeletal muscle. |
| Schenkel et al. 2014 [18] | Animal study | Palmitic Acid and Oleic Acid reduced CTL1/SLC44A expression on plasma membrane and mitochondrial membrane, modulating choline cytoplasmatic content and its bioavailability for PC synthesis. |
| Li et al. 2015 [19] | Animal study | Diet supplementation with 0.25% RPC increased growth, intramuscular fat and expression of genes involved in lipogenesis (CD36, ACC and FASN) in lambs. |
| Oster et al. 2015 [20] | Animal study | Diet supplemented with methionine, folate, choline (2230 mg/kg), B6, B12, and zinc increased fetal weight compared to those fed with standard diet at 35- and 91- dpc time points. Methionine diet reduced levels of IGFBP2 and increased IGF2 levels in skeletal muscle at 91 dpc. |
| Robinson et al. 2016 [21] | Animal study | Methyl deficiency diet including low choline content reduced whole-body protein synthesis (−12%, p = 0.01) and skeletal muscle specific protein synthesis (−60%, p = 0.003) compared to methyl sufficient diet. |
| Taylor et al. 2017 [22] | Animal study | Choline supplementation (~240 μg/g/day for 4 weeks) increased content of membrane phospholipids (PC and sphingomyelin), DAG and glycogen levels (+30% and +60%, respectively) in Pcyt2+/− mice. Conversely, choline reduced muscle TAG content of 40%, de novo FAs synthesis and lipogenesis. |
| Jahanian et al. 2018 [23] | Animal study | Dietary supplementation with choline (1000 mg/kg), increased protein content in leg and breast muscle. Diet with +0.42 MJ/kg ME levels plus choline improved protein content compared to ME alone in leg muscle (18.68% vs. 17.80%). |
| Fisher et al. 2007 [24] | Clinical study | Low choline intake provoked fatty liver or muscle damage in 33 subjects (77% of men, 80% of postmenopausal women, and 44% of premenopausal women developing organ dysfunction). During the 550-mg choline diet, also 6 men developed the same signs. All participants presented high levels of CPK. However, these negative effects reversed after choline repletion. |
| Mc Lean et al. 2013 [25] | Clinical study | After a 19-day of ALT an improved carnitine/choline peak (8.8% ± 6.1%) was observed in soleus muscle in athletes, compared to sea level training. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moretti, A.; Paoletta, M.; Liguori, S.; Bertone, M.; Toro, G.; Iolascon, G. Choline: An Essential Nutrient for Skeletal Muscle. Nutrients 2020, 12, 2144. https://doi.org/10.3390/nu12072144
Moretti A, Paoletta M, Liguori S, Bertone M, Toro G, Iolascon G. Choline: An Essential Nutrient for Skeletal Muscle. Nutrients. 2020; 12(7):2144. https://doi.org/10.3390/nu12072144
Chicago/Turabian StyleMoretti, Antimo, Marco Paoletta, Sara Liguori, Matteo Bertone, Giuseppe Toro, and Giovanni Iolascon. 2020. "Choline: An Essential Nutrient for Skeletal Muscle" Nutrients 12, no. 7: 2144. https://doi.org/10.3390/nu12072144
APA StyleMoretti, A., Paoletta, M., Liguori, S., Bertone, M., Toro, G., & Iolascon, G. (2020). Choline: An Essential Nutrient for Skeletal Muscle. Nutrients, 12(7), 2144. https://doi.org/10.3390/nu12072144






