Maternal Supplementation of Food Ingredient (Prebiotic) or Food Contaminant (Mycotoxin) Influences Mucosal Immune System in Piglets
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Diets and Experimental Design
2.2. Immune Cell Dissociation from Jejunum and Ileum
2.3. Flow Cytometry Analysis
2.4. Explant Cultures
2.5. Determination of Cytokine Concentration
2.6. RNA Extraction and Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR) Analysis
2.7. Statistical Analysis
3. Results
3.1. Sow Diet. Prebiotic Supplementation or Mycotoxin Contamination on Sow Performance during Gestation and Lactation
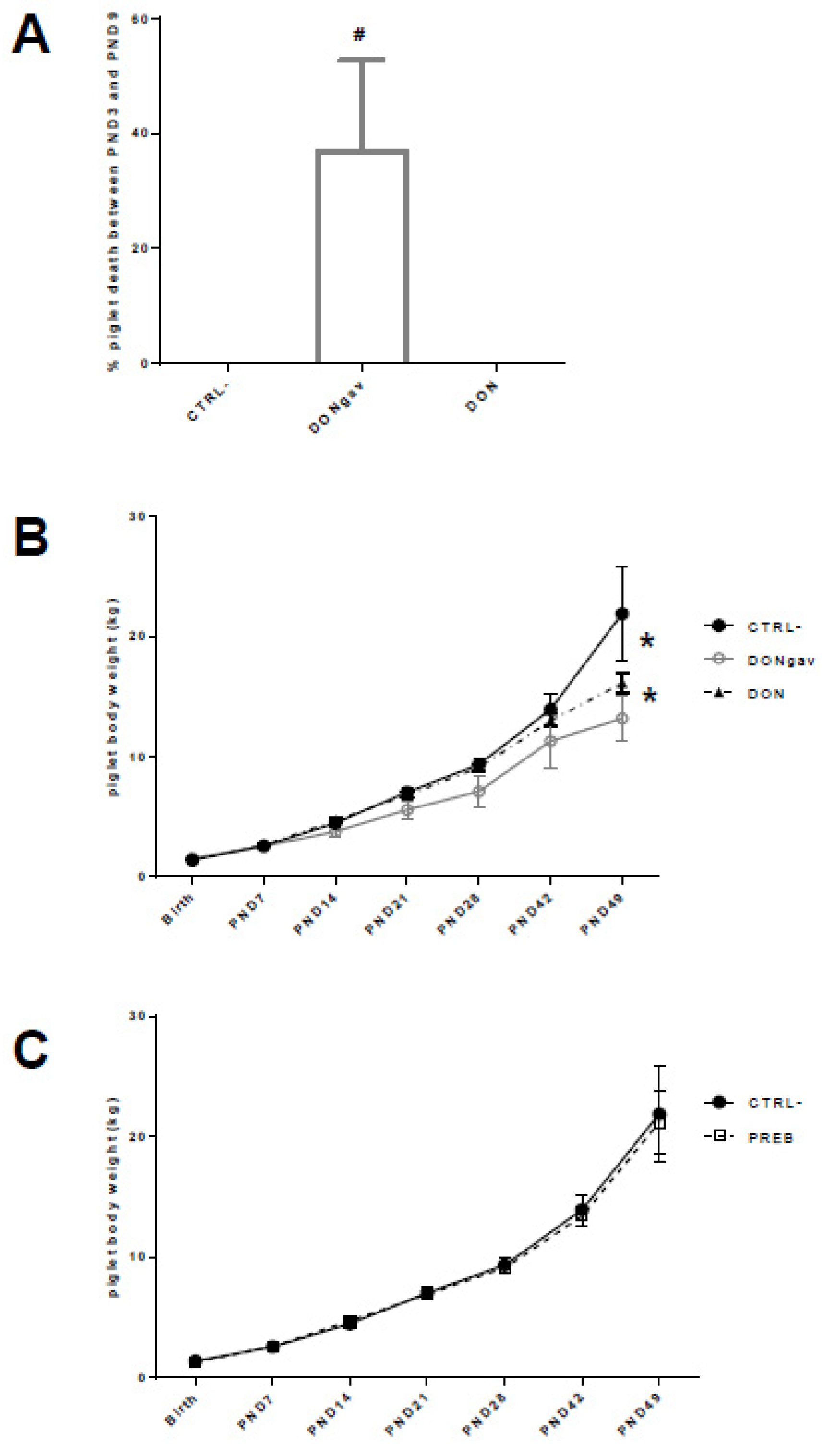
3.2. Sow Diet. Prebiotic Supplementation or Mycotoxin Contamination Differentially Influenced Piglet Performance
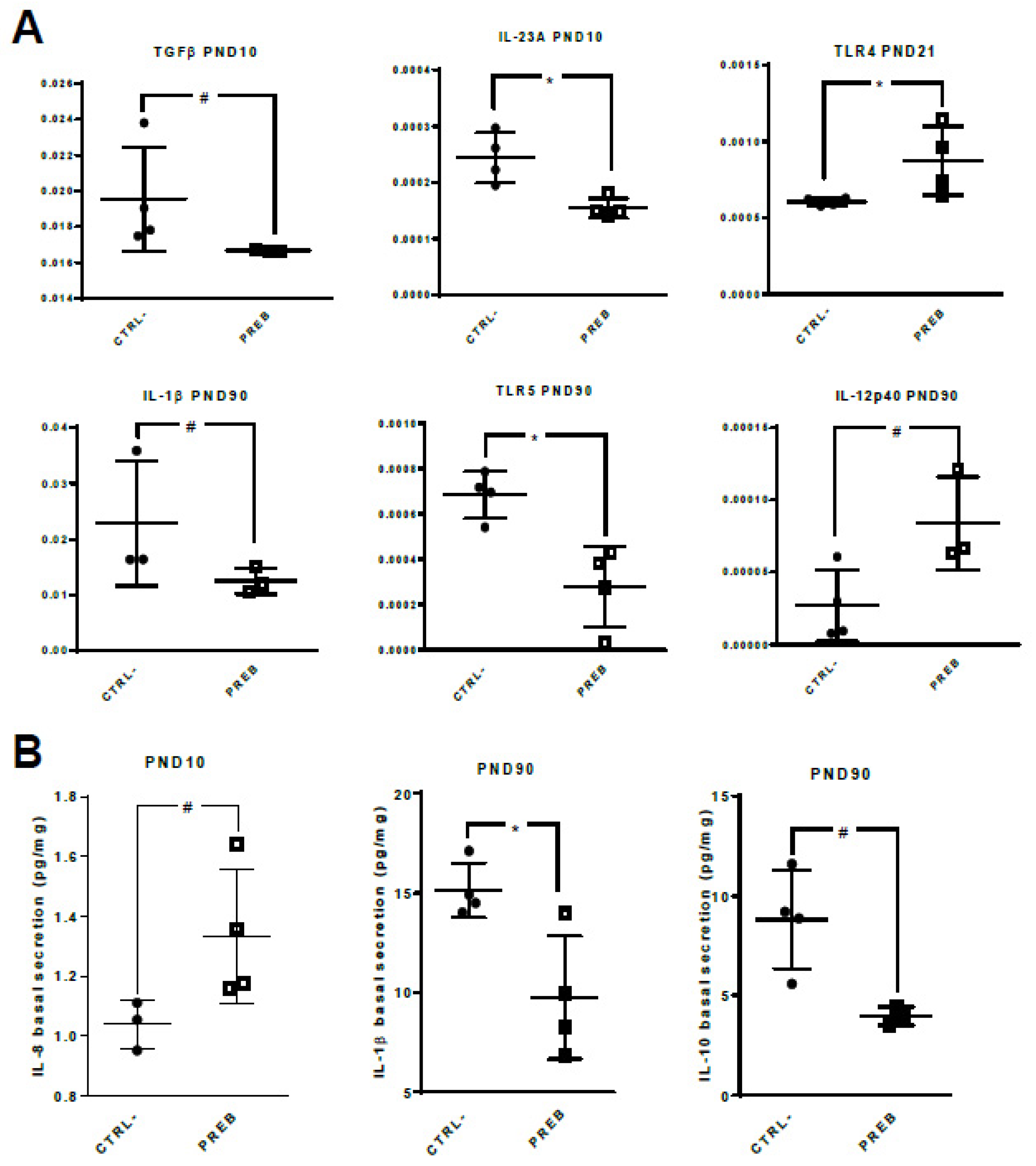
3.3. Sow Diet. Prebiotic Supplementation Changed Gene Expression and Cytokine Patterns of Unstimulated il-PP Explants
3.4. Sow Diet. Prebiotic Supplementation Changed Gene Expression and Cytokine Patterns of il-PP Explants Stimulated with TLR-ligands
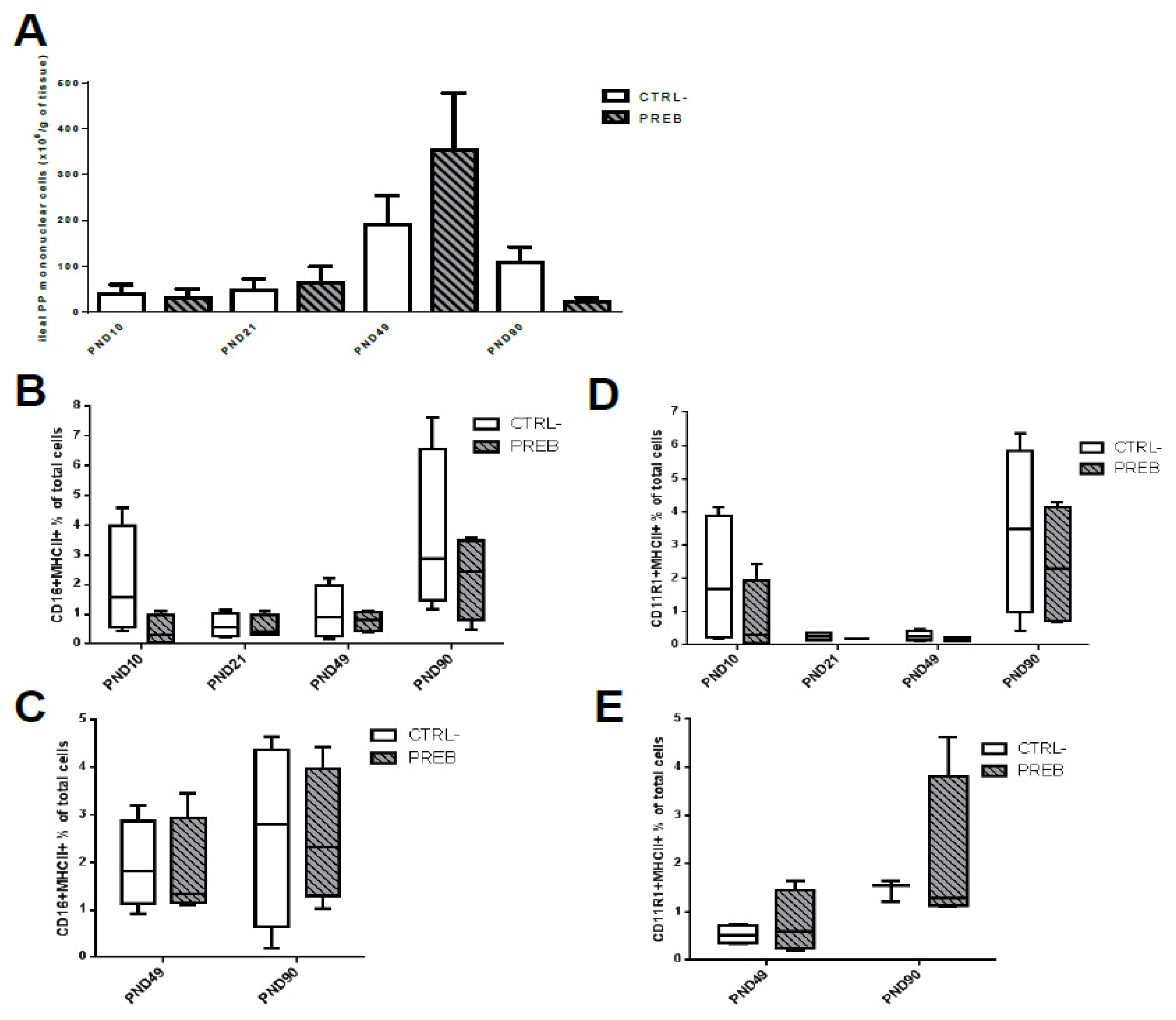
3.5. Sow Diet. Mycotoxin Contamination (DON) and Neonate Mycotoxin Gavage (DONgav) Modified jej-LP Cell Numbers and the Proportion of Resident APC
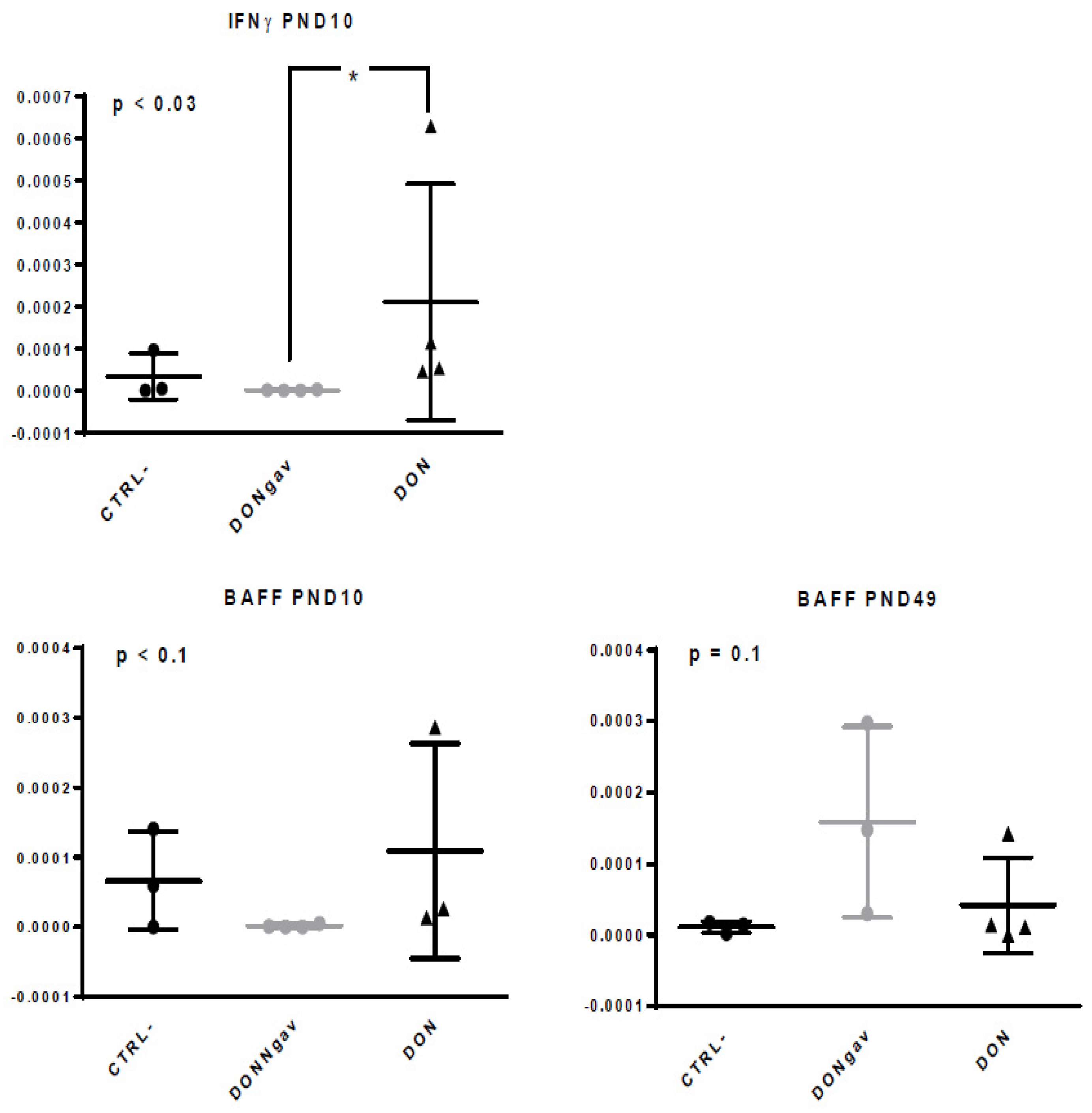
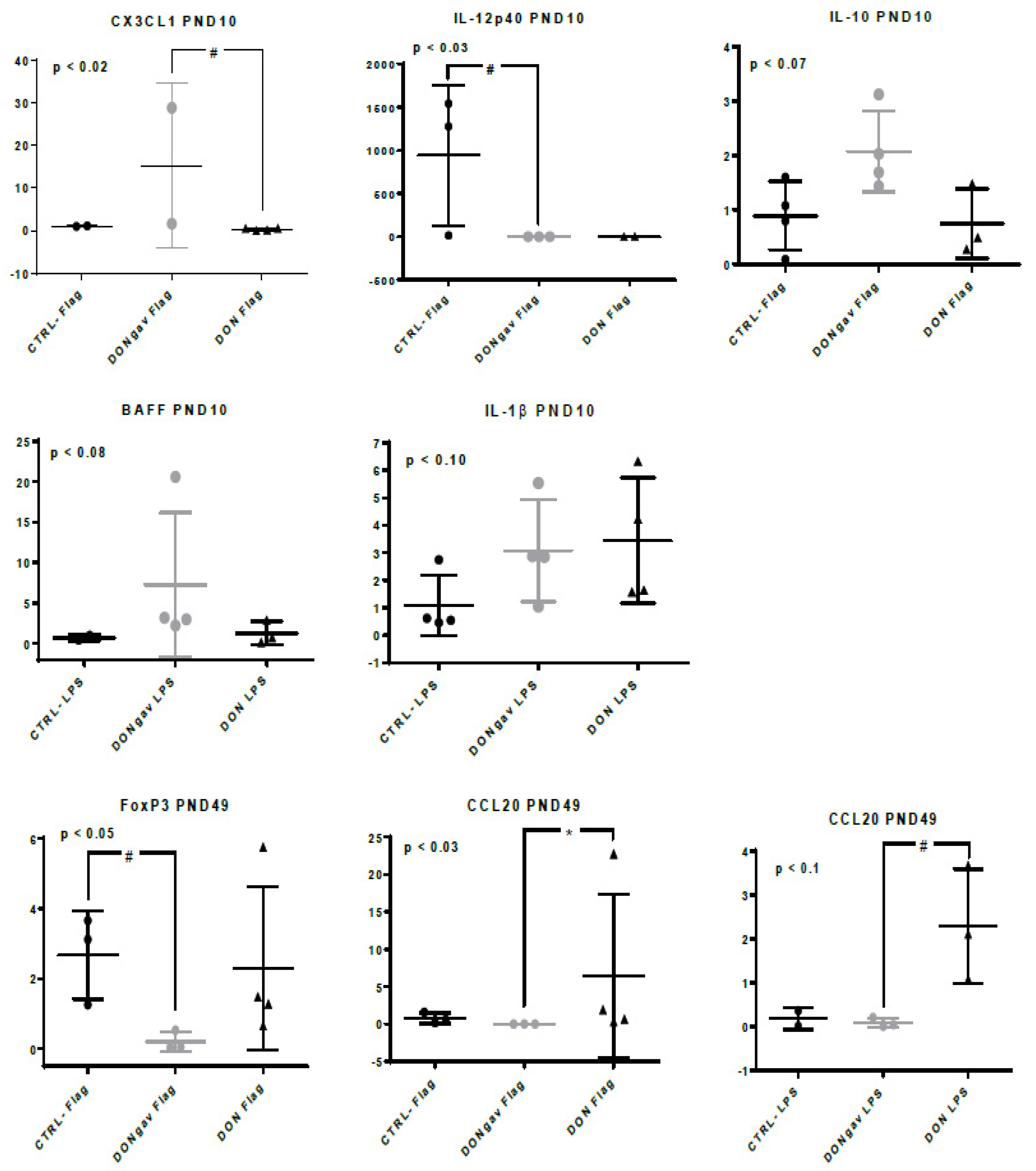
3.6. Sow Diet. Mycotoxin Contamination (DON) and Neonate Mycotoxin Gavage (DONgav) Changed Some Gene Expression of both Unstimulated and Stimulated jej-LP Explants
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bailey, M. The Mucosal Immune System: Recent Developments and Future Directions in the Pig. Dev. Comp. Immunol. 2009, 33, 375–383. [Google Scholar] [CrossRef]
- Bailey, M.; Haverson, K.; Inman, C.; Harris, C.; Jones, P.; Corfield, G.; Miller, B.; Stokes, C. The Influence of Environment on Development of the Mucosal Immune System. Vet. Immunol. Immunopathol. 2005, 108, 189–198. [Google Scholar] [CrossRef]
- Flannigan, K.L.; Geem, D.; Harusato, A.; Denning, T.L. Intestinal Antigen-Presenting Cells: Key Regulators of Immune Homeostasis and Inflammation. Am. J. Pathol. 2015, 185, 1809–1819. [Google Scholar] [CrossRef] [PubMed]
- Desbuards, N.; Gourbeyre, P.; Haure-Mirande, V.; Darmaun, D.; Champ, M.; Bodinier, M. Impact of Perinatal Prebiotic Consumption on Gestating Mice and Their Offspring: A Preliminary Report. Br. J. Nutr. 2012, 107, 1245–1248. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Le Bourgot, C.; Ferret-Bernard, S.; Le Normand, L.; Savary, G.; Menendez-Aparicio, E.; Blat, S.; Appert-Bossard, E.; Respondek, F.; Le Huerou-Luron, I. Maternal Short-Chain Fructooligosaccharide Supplementation Influences Intestinal Immune System Maturation in Piglets. PLoS ONE 2014, 9, e107508. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, M.; Gibson, G.R.; Hoyles, L.; McCartney, A.L.; Rastall, R.; Rowland, I.; Wolvers, D.; Watzl, B.; Szajewska, H.; Stahl, B.; et al. Prebiotic Effects: Metabolic and Health Benefits. Br. J. Nutr. 2010, 104 (Suppl. 2), S1–S63. [Google Scholar] [CrossRef]
- Shadid, R.; Haarman, M.; Knol, J.; Theis, W.; Beermann, C.; Rjosk-Dendorfer, D.; Schendel, D.J.; Koletzko, B.V.; Krauss-Etschmann, S. Effects of Galactooligosaccharide and Long-Chain Fructooligosaccharide Supplementation During Pregnancy on Maternal and Neonatal Microbiota and Immunity—A Randomized, Double-Blind, Placebo-Controlled Study. Am. J. Clin. Nutr. 2007, 86, 1426–1437. [Google Scholar] [CrossRef] [PubMed]
- Le Bourgot, C.; Ferret-Bernard, S.; Apper, E.; Taminiau, B.; Cahu, A.; Le Normand, L.; Respondek, F.; Le Huerou-Luron, I.; Blat, S. Perinatal Short-Chain Fructooligosaccharides Program Intestinal Microbiota and Improve Enteroinsular Axis Function and Inflammatory Status in High-Fat Diet-Fed Adult Pigs. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2019, 33, 301–313. [Google Scholar] [CrossRef]
- Le Bourgot, C.; Ferret-Bernard, S.; Blat, S.; Apper, E.; Le Huërou-Luron, I. Short-Chain Fructooligosaccharide Supplementation During Gestation and Lactation or after Weaning Differentially Impacts Pig Growth and Iga Response to Influenza Vaccination. J. Funct. Foods 2016, 24, 307–315. [Google Scholar] [CrossRef]
- Le Bourgot, C.; Le Normand, L.; Formal, M.; Respondek, F.; Blat, S.; Apper, E.; Ferret-Bernard, S.; Le Huerou-Luron, I. Maternal Short-Chain Fructo-Oligosaccharide Supplementation Increases Intestinal Cytokine Secretion, Goblet Cell Number, Butyrate Concentration and Lawsonia Intracellularis Humoral Vaccine Response in Weaned Pigs. Br. J. Nutr. 2017, 117, 83–92. [Google Scholar] [CrossRef]
- Hosono, A.; Ozawa, A.; Kato, R.; Ohnishi, Y.; Nakanishi, Y.; Kimura, T.; Nakamura, R. Dietary Fructooligosaccharides Induce Immunoregulation of Intestinal Iga Secretion by Murine Peyer’s Patch Cells. Biosci. Biotechnol. Biochem. 2003, 67, 758–764. [Google Scholar] [CrossRef]
- Swanson, K.S.; Grieshop, C.M.; Flickinger, E.A.; Bauer, L.L.; Healy, H.P.; Dawson, K.A.; Merchen, N.R.; Fahey, G.C., Jr. Supplemental Fructooligosaccharides and Mannanoligosaccharides Influence Immune Function, Ileal and Total Tract Nutrient Digestibilities, Microbial Populations and Concentrations of Protein Catabolites in the Large Bowel of Dogs. J. Nutr. 2002, 132, 980–989. [Google Scholar] [CrossRef] [PubMed]
- Benyacoub, J.; Rochat, F.; Saudan, K.Y.; Rochat, I.; Antille, N.; Cherbut, C.; von der Weid, T.; Schiffrin, E.J.; Blum, S. Feeding a Diet Containing a Fructooligosaccharide Mix Can Enhance Salmonella Vaccine Efficacy in Mice. J. Nutr. 2008, 138, 123–129. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bermudez-Brito, M.; Sahasrabudhe, N.M.; Rosch, C.; Schols, H.A.; Faas, M.M.; de Vos, P. The Impact of Dietary Fibers on Dendritic Cell Responses in Vitro Is Dependent on the Differential Effects of the Fibers on Intestinal Epithelial Cells. Mol. Nutr. Food Res. 2015, 59, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Ryz, N.R.; Meddings, J.B.; Taylor, C.G. Long-Chain Inulin Increases Dendritic Cells in the Peyer’s Patches and Increases Ex Vivo Cytokine Secretion in the Spleen and Mesenteric Lymph Nodes of Growing Female Rats, Independent of Zinc Status. Br. J. Nutr. 2009, 101, 1653–1663. [Google Scholar] [CrossRef]
- Vogt, L.; Meyer, D.; Pullens, G.; Faas, M.; Smelt, M.; Venema, K.; Ramasamy, U.; Schols, H.A.; De Vos, P. Immunological Properties of Inulin-Type Fructans. Crit. Rev. Food Sci. Nutr. 2015, 55, 414–436. [Google Scholar] [CrossRef] [PubMed]
- Devriendt, B.; Gallois, M.; Verdonck, F.; Wache, Y.; Bimczok, D.; Oswald, I.P.; Goddeeris, B.M.; Cox, E. The Food Contaminant Fumonisin B(1) Reduces the Maturation of Porcine Cd11r1(+) Intestinal Antigen Presenting Cells and Antigen-Specific Immune Responses, Leading to a Prolonged Intestinal Etec Infection. Vet. Res. 2009, 40, 40. [Google Scholar] [CrossRef] [PubMed]
- Oswald, I.P.; Desautels, C.; Laffitte, J.; Fournout, S.; Peres, S.Y.; Odin, M.; Le Bars, P.; Le Bars, J.; Fairbrother, J.M. Mycotoxin Fumonisin B1 Increases Intestinal Colonization by Pathogenic Escherichia Coli in Pigs. Appl. Environ. Microbiol. 2003, 69, 5870–5874. [Google Scholar] [CrossRef]
- Oswald, I.P.; Marin, D.E.; Bouhet, S.; Pinton, P.; Taranu, I.; Accensi, F. Immunotoxicological Risk of Mycotoxins for Domestic Animals. Food Addit. Contam. 2005, 22, 354–360. [Google Scholar] [CrossRef]
- Vandenbroucke, V.; Croubels, S.; Martel, A.; Verbrugghe, E.; Goossens, J.; van Deun, K.; Boyen, F.; Thompson, A.; Shearer, N.; de Backer, P.; et al. The Mycotoxin Deoxynivalenol Potentiates Intestinal Inflammation by Salmonella Typhimurium in Porcine Ileal Loops. PLoS ONE 2011, 6, e23871. [Google Scholar] [CrossRef]
- Cano, P.M.; Seeboth, J.; Meurens, F.; Cognie, J.; Abrami, R.; Oswald, I.P.; Guzylack-Piriou, L. Deoxynivalenol as a New Factor in the Persistence of Intestinal Inflammatory Diseases: An Emerging Hypothesis through Possible Modulation of Th17-Mediated Response. PLoS ONE 2013, 8, e53647. [Google Scholar] [CrossRef]
- Pestka, J.J.; Smolinski, A.T. Deoxynivalenol: Toxicology and Potential Effects on Humans. J. Toxicol. Environ. Health Part. B Crit. Rev. 2005, 8, 39–69. [Google Scholar] [CrossRef] [PubMed]
- Bimczok, D.; Doll, S.; Rau, H.; Goyarts, T.; Wundrack, N.; Naumann, M.; Danicke, S.; Rothkotter, H.J. The Fusarium Toxin Deoxynivalenol Disrupts Phenotype and Function of Monocyte-Derived Dendritic Cells in Vivo and in Vitro. Immunobiology 2007, 212, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Hymery, N.; Leon, K.; Carpentier, F.G.; Jung, J.L.; Parent-Massin, D. T-2 Toxin Inhibits the Differentiation of Human Monocytes into Dendritic Cells and Macrophages. Toxicol. Vitr. Int. J. Publ. Assoc. BIBRA 2009, 23, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Danicke, S.; Brussow, K.P.; Goyarts, T.; Valenta, H.; Ueberschar, K.H.; Tiemann, U. On the Transfer of the Fusarium Toxins Deoxynivalenol (Don) and Zearalenone (Zon) from the Sow to the Full-Term Piglet During the Last Third of Gestation. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2007, 45, 1565–1574. [Google Scholar] [CrossRef]
- Gutzwiller, A. Effects of Deoxynivalenol (Don) in the Lactation Diet on the Feed Intake and Fertility of Sows. Mycotoxin Res. 2010, 26, 211–215. [Google Scholar] [CrossRef]
- Sayyari, A.; Framstad, T.; Krogenaes, A.K.; Sivertsen, T. Effects of Feeding Naturally Contaminated Deoxynivalenol Diets to Sows During Late Gestation and Lactation in a High-Yield Specific Pathogen-Free Herd. Porc. Health Manag. 2018, 4, 26. [Google Scholar] [CrossRef]
- Sayyari, A.; Uhlig, S.; Faeste, C.K.; Framstad, T.; Sivertsen, T. Transfer of Deoxynivalenol (Don) through Placenta, Colostrum and Milk from Sows to Their Offspring during Late Gestation and Lactation. Toxins 2018, 10, 517. [Google Scholar] [CrossRef]
- Avantaggiato, G.; Havenaar, R.; Visconti, A. Evaluation of the Intestinal Absorption of Deoxynivalenol and Nivalenol by an in Vitro Gastrointestinal Model, and the Binding Efficacy of Activated Carbon and Other Adsorbent Materials. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2004, 42, 817–824. [Google Scholar] [CrossRef]
- Bimczok, D.; Sowa, E.N.; Faber-Zuschratter, H.; Pabst, R.; Rothkotter, H.J. Site-Specific Expression of Cd11b and Sirpalpha (Cd172a) on Dendritic Cells: Implications for Their Migration Patterns in the Gut Immune System. Eur. J. Immunol. 2005, 35, 1418–1427. [Google Scholar] [CrossRef]
- Coeffier, M.; Marion, R.; Ducrotte, P.; Dechelotte, P. Modulating Effect of Glutamine on Il-1beta-Induced Cytokine Production by Human Gut. Clin. Nutr. 2003, 22, 407–413. [Google Scholar] [CrossRef]
- Coeffier, M.; Miralles-Barrachina, O.; Le Pessot, F.; Lalaude, O.; Daveau, M.; Lavoinne, A.; Lerebours, E.; Dechelotte, P. Influence of Glutamine on Cytokine Production by Human Gut in Vitro. Cytokine 2001, 13, 148–154. [Google Scholar] [CrossRef]
- Chatelais, L.; Jamin, A.; Gras-Le Guen, C.; Lallès, J.P.; Le Huërou-Luron, I.; Boudry, G. The Level of Protein in Milk Formula Modifies Ileal Sensitivity to Lps Later in Life in a Piglet Model. PLoS ONE 2011, 6, e19594. [Google Scholar] [CrossRef] [PubMed]
- Pie, S.; Awati, A.; Vida, S.; Falluel, I.; Williams, B.A.; Oswald, I.P. Effects of Added Fermentable Carbohydrates in the Diet on Intestinal Proinflammatory Cytokine-Specific Mrna Content in Weaning Piglets. J. Anim. Sci. 2007, 85, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Levast, B.; de Monte, M.; Chevaleyre, C.; Melo, S.; Berri, M.; Mangin, F.; Zanello, G.; Lantier, I.; Salmon, H.; Meurens, F. Ultra-Early Weaning in Piglets Results in Low Serum Iga Concentration and Il17 Mrna Expression. Vet. Immunol. Immunopathol. 2010, 137, 261–268. [Google Scholar] [CrossRef]
- Meurens, F.; Berri, M.; Auray, G.; Melo, S.; Levast, B.; Virlogeux-Payant, I.; Chevaleyre, C.; Gerdts, V.; Salmon, H. Early Immune Response Following Salmonella Enterica Subspecies Enterica Serovar Typhimurium Infection in Porcine Jejunal Gut Loops. Vet. Res. 2009, 40, 5. [Google Scholar] [CrossRef]
- Pinton, P.; Braicu, C.; Nougayrede, J.P.; Laffitte, J.; Taranu, I.; Oswald, I.P. Deoxynivalenol Impairs Porcine Intestinal Barrier Function and Decreases the Protein Expression of Claudin-4 through a Mitogen-Activated Protein Kinase-Dependent Mechanism. J. Nutr. 2010, 140, 1956–1962. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.; Braber, S.; Akbari, P.; Garssen, J.; Fink-Gremmels, J. Deoxynivalenol Impairs Weight Gain and Affects Markers of Gut Health after Low-Dose, Short-Term Exposure of Growing Pigs. Toxins 2015, 7, 2071–2095. [Google Scholar] [CrossRef] [PubMed]
- Bryden, W.L. Mycotoxins in the Food Chain: Human Health Implications. Asia Pac. J. Clin. Nutr. 2007, 16 (Suppl. 1), 95–101. [Google Scholar]
- Li, R.; Li, Y.; Su, Y.; Shen, D.; Dai, P.; Li, C. Short-Term Ingestion of Deoxynivalenol in Naturally Contaminated Feed Alters Piglet Performance and Gut Hormone Secretion. Anim. Sci. J. Nihon Chikusan Gakkaiho 2018, 89, 1134–1143. [Google Scholar] [CrossRef] [PubMed]
- Bauche, D.; Marie, J.C. Transforming Growth Factor Beta: A Master Regulator of the Gut Microbiota and Immune Cell Interactions. Clin. Transl. Immunol. 2017, 6, e136. [Google Scholar] [CrossRef] [PubMed]
- Kannan, A.K.; Su, Z.; Gauvin, D.M.; Paulsboe, S.E.; Duggan, R.; Lasko, L.M.; Honore, P.; Kort, M.E.; McGaraughty, S.P.; Scott, V.E.; et al. Il-23 Induces Regulatory T Cell Plasticity with Implications for Inflammatory Skin Diseases. Sci. Rep. 2019, 9, 17675. [Google Scholar] [CrossRef] [PubMed]
- Brosseau, C.; Selle, A.; Palmer, D.J.; Prescott, S.L.; Barbarot, S.; Bodinier, M. Prebiotics: Mechanisms and Preventive Effects in Allergy. Nutrients 2019, 11, 1841. [Google Scholar] [CrossRef]
- Ortega-Gonzalez, M.; Ocon, B.; Romero-Calvo, I.; Anzola, A.; Guadix, E.; Zarzuelo, A.; Suarez, M.D.; de Medina, F.S.; Martinez-Augustin, O. Nondigestible Oligosaccharides Exert Nonprebiotic Effects on Intestinal Epithelial Cells Enhancing the Immune Response Via Activation of Tlr4-Nfkappab. Mol. Nutr. Food Res. 2014, 58, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Castillo, N.A.; Perdigon, G.; de Leblanc, A.D. Oral Administration of a Probiotic Lactobacillus Modulates Cytokine Production and Tlr Expression Improving the Immune Response against Salmonella Enterica Serovar Typhimurium Infection in Mice. BMC Microbiol. 2011, 11, 177. [Google Scholar] [CrossRef] [PubMed]
- Chieppa, M.; Rescigno, M.; Huang, A.Y.; Germain, R.N. Dynamic Imaging of Dendritic Cell Extension into the Small Bowel Lumen in Response to Epithelial Cell Tlr Engagement. J. Exp. Med. 2006, 203, 2841–2852. [Google Scholar] [CrossRef]
- Iliev, I.D.; Matteoli, G.; Rescigno, M. The Yin and Yang of Intestinal Epithelial Cells in Controlling Dendritic Cell Function. J. Exp. Med. 2007, 204, 2253–2257. [Google Scholar] [CrossRef]
- Cheat, S.; Gerez, J.R.; Cognie, J.; Alassane-Kpembi, I.; Bracarense, A.P.; Raymond-Letron, I.; Oswald, I.P.; Kolf-Clauw, M. Nivalenol Has a Greater Impact Than Deoxynivalenol on Pig Jejunum Mucosa in Vitro on Explants and in Vivo on Intestinal Loops. Toxins 2015, 7, 1945–1961. [Google Scholar] [CrossRef]
- Nossol, C.; Diesing, A.K.; Kahlert, S.; Kersten, S.; Kluess, J.; Ponsuksili, S.; Hartig, R.; Wimmers, K.; Danicke, S.; Rothkotter, H.J. Deoxynivalenol Affects the Composition of the Basement Membrane Proteins and Influences En Route the Migration of Cd16(+) Cells into the Intestinal Epithelium. Mycotoxin Res. 2013, 29, 245–254. [Google Scholar] [CrossRef]
- Mair, K.H.; Stadler, M.; Talker, S.C.; Forberg, H.; Storset, A.K.; Mullebner, A.; Duvigneau, J.C.; Hammer, S.E.; Saalmuller, A.; Gerner, W. Porcine Cd3(+)Nkp46(+) Lymphocytes Have Nk-Cell Characteristics and Are Present in Increased Frequencies in the Lungs of Influenza-Infected Animals. Front. Immunol. 2016, 7, 263. [Google Scholar] [CrossRef]
- Pestka, J.J.; Amuzie, C.J. Tissue Distribution and Proinflammatory Cytokine Gene Expression Following Acute Oral Exposure to Deoxynivalenol: Comparison of Weanling and Adult Mice. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2008, 46, 2826–2831. [Google Scholar] [CrossRef] [PubMed]
- Diesing, A.K.; Nossol, C.; Danicke, S.; Walk, N.; Post, A.; Kahlert, S.; Rothkotter, H.J.; Kluess, J. Vulnerability of Polarised Intestinal Porcine Epithelial Cells to Mycotoxin Deoxynivalenol Depends on the Route of Application. PLoS ONE 2011, 6, e17472. [Google Scholar] [CrossRef]
- Vignal, C.; Djouina, M.; Pichavant, M.; Caboche, S.; Waxin, C.; Beury, D.; Hot, D.; Gower-Rousseau, C.; Body-Malapel, M. Chronic Ingestion of Deoxynivalenol at Human Dietary Levels Impairs Intestinal Homeostasis and Gut Microbiota in Mice. Arch. Toxicol. 2018, 92, 2327–2338. [Google Scholar] [CrossRef] [PubMed]
- Pestka, J.J. Deoxynivalenol-Induced Iga Production and Iga Nephropathy-Aberrant Mucosal Immune Response with Systemic Repercussions. Toxicol. Lett. 2003, 140–141, 287–295. [Google Scholar] [CrossRef]
- Cerutti, A.; Chen, K.; Chorny, A. Immunoglobulin Responses at the Mucosal Interface. Annu. Rev. Immunol. 2011, 29, 273–293. [Google Scholar] [CrossRef]
- Islam, Z.; Pestka, J.J. Lps Priming Potentiates and Prolongs Proinflammatory Cytokine Response to the Trichothecene Deoxynivalenol in the Mouse. Toxicol. Appl. Pharmacol. 2006, 211, 53–63. [Google Scholar] [CrossRef]
- Scott, C.L.; Aumeunier, A.M.; Mowat, A.M. Intestinal Cd103+ Dendritic Cells: Master Regulators of Tolerance? Trends Immunol. 2011, 32, 412–419. [Google Scholar] [CrossRef]
- Mavin, E.; Nicholson, L.; Ahmed, S.R.; Gao, F.; Dickinson, A.; Wang, X.N. Human Regulatory T Cells Mediate Transcriptional Modulation of Dendritic Cell Function. J. Immunol. 2017, 198, 138–146. [Google Scholar] [CrossRef]
- Ferrer, M.; Manyes, L.; Manes, J.; Meca, G. Influence of Prebiotics, Probiotics and Protein Ingredients on Mycotoxin Bioaccessibility. Food Funct. 2015, 6, 987–994. [Google Scholar] [CrossRef]




© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferret-Bernard, S.; Le Normand, L.; Romé, V.; Le Bourgot, C.; Seeboth, J.; Savary, G.; Laurent, F.; Le Huërou-Luron, I.; Guzylack-Piriou, L. Maternal Supplementation of Food Ingredient (Prebiotic) or Food Contaminant (Mycotoxin) Influences Mucosal Immune System in Piglets. Nutrients 2020, 12, 2115. https://doi.org/10.3390/nu12072115
Ferret-Bernard S, Le Normand L, Romé V, Le Bourgot C, Seeboth J, Savary G, Laurent F, Le Huërou-Luron I, Guzylack-Piriou L. Maternal Supplementation of Food Ingredient (Prebiotic) or Food Contaminant (Mycotoxin) Influences Mucosal Immune System in Piglets. Nutrients. 2020; 12(7):2115. https://doi.org/10.3390/nu12072115
Chicago/Turabian StyleFerret-Bernard, Stéphanie, Laurence Le Normand, Véronique Romé, Cindy Le Bourgot, Julie Seeboth, Gérard Savary, Fabrice Laurent, Isabelle Le Huërou-Luron, and Laurence Guzylack-Piriou. 2020. "Maternal Supplementation of Food Ingredient (Prebiotic) or Food Contaminant (Mycotoxin) Influences Mucosal Immune System in Piglets" Nutrients 12, no. 7: 2115. https://doi.org/10.3390/nu12072115
APA StyleFerret-Bernard, S., Le Normand, L., Romé, V., Le Bourgot, C., Seeboth, J., Savary, G., Laurent, F., Le Huërou-Luron, I., & Guzylack-Piriou, L. (2020). Maternal Supplementation of Food Ingredient (Prebiotic) or Food Contaminant (Mycotoxin) Influences Mucosal Immune System in Piglets. Nutrients, 12(7), 2115. https://doi.org/10.3390/nu12072115






