Dietary Diversity and Micronutrients Adequacy in Women of Childbearing Age: Results from ELANS Study
Abstract
1. Introduction
2. Methods
2.1. Study Population
2.2. Anthropometric Measurements
2.3. Dietary Assessment
2.4. Dietary Diversity Score
2.5. Assessment of Nutrient Adequacy
2.6. Statistical Analyses
3. Results
3.1. Diet Diversity Score (DDS) According to Sociodemographic Variables
3.2. Consumption of Food Groups
3.3. Energy, Nutrients, and Food Groups Intake in Diet Diversity Subgroups
3.4. Nutritional Status and Anthropometric Measurements in Diet Diversity Subgroups
3.5. Nutrient Adequacy Ratio (NAR)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Organización de las Naciones Unidas para la Alimentación y la Agricultura (FAO); Organización Panamericana de la Salud (OPS); Programa Mundial de Alimentos (WSF); Fondo de las Naciones Unidas para la Infancia (UNICEF). Panorama de la Seguridad Alimentaria y Nutricional en América Latina y el Caribe; UNICEF: Santiago, Chile, 2018. [Google Scholar]
- Marangoni, F.; Cetin, I.; Verduci, E.; Canzone, G.; Giovannini, M.; Scollo, P.; Corsello, G.; Poli, A. Maternal diet and nutrient requirements in pregnancy and breastfeeding: An Italian consensus document. Nutrients 2016, 8, 629. [Google Scholar] [CrossRef] [PubMed]
- Preedy, V.; Hunter, L.; Patel, B. Diet Quality: An Evidence-Based Approach; Humana Press: London, UK, 2013. [Google Scholar]
- Lassi, Z.S.; Padhani, Z.A.; Rabbani, A.; Rind, F.; Salam, R.A.; Das, J.K.; Bhutta, Z.A. Impact of dietary interventions during pregnancy on maternal, neonatal, and child outcomes in low- and middle-income countries. Nutrients 2020, 12, 531. [Google Scholar] [CrossRef] [PubMed]
- United Nations System Standing Committee on Nutrition. Sixth Report on the World Nutrition Situation. Available online: https://www.unscn.org/files/Publications/RWNS6/html/ (accessed on 1 June 2020).
- Martin-Prevel, Y.; Arimond, M.; Allemand, P.; Wiesmann, D.; Ballard, T.J.; Deitchler, M.; Dop, M.; Kennedy, G.; Lartey, A.; Lee, W.T.K.; et al. Development of a dichotomous indicator for population-level assessment of dietary diversity in women of reproductive age. Curr. Dev. Nutr. 2017, 1, cdn.117.001701. [Google Scholar] [CrossRef] [PubMed]
- Adubra, L.; Savy, M.; Fortin, S.; Kameli, Y.; Kodjo, N.E.; Fainke, K.; Mahamadou, T.; Le Port, A.; Martin-Prevel, Y. The minimum dietary diversity for women of reproductive age (MDD-W) indicator is related to household food insecurity and farm production diversity: Evidence from rural Mali. Curr. Dev. Nutr. 2019, 3, nzz002. [Google Scholar] [CrossRef]
- Arimond, M.; Wiesmann, D.; Becquey, E.; Carriquiry, A.; Daniels, M.C.; Deitchler, M.; Fanou-Fogny, N.; Joseph, M.L.; Kennedy, G.; Martin-Prevel, Y.; et al. Simple food group diversity indicators predict micronutrient adequacy of women’s diets in 5 diverse, resource-poor settings. J. Nutr. 2010, 140, 2059S–2069S. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, B. Addressing the double burden of malnutrition in developing countries: Need for strategies to improve the lean body mass. Food Nutr. Bull. 2018, 39, S69–S76. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.; Huybregts, L.; Sanghvi, T.G.; Tran, L.M.; Frongillo, E.A.; Menon, P.; Ruel, M.T. Dietary diversity predicts the adequacy of micronutrient intake in pregnant adolescent girls and women in Bangladesh, but use of the 5-group Cutoff Poorly identifies individuals with inadequate intake. J. Nutr. 2018, 148, 790–797. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United State (FAO). Minimum Dietary Diversity for Women—A Guide to Measurement; University of California: Davis, CA, USA, 2016. [Google Scholar]
- Abris, G.P.; Provido, S.M.P.; Hong, S.; Yu, S.H. Association between dietary diversity and obesity in the Filipino Women s Diet and Health Study (FiLWHEL): A cross-sectional study. PLoS ONE 2018, 49, 1–16. [Google Scholar]
- Tavakoli, S.; Dorosty-motlagh, A.R.; Hoshiar-Rad, A.; Eshraghian, M.R.; Sotoudeh, G.; Azadbakht, L.; Karimi, M.; Jalali-Farahani, S. Is dietary diversity a proxy measurement of nutrient adequacy in Iranian elderly women? Appetite 2016, 105, 468–476. [Google Scholar] [CrossRef]
- Liu, C.K.; Huang, Y.C.; Lo, Y.T.C.; Wahlqvist, M.L.; Lee, M.S. Dietary diversity offsets the adverse mortality risk among older indigenous Taiwanese. Asia Pac. J. Clin. Nutr. 2019, 28, 593–600. [Google Scholar]
- Rathnayake, K.; Madushani, P.; Silva, K. Use of dietary diversity score as a proxy indicator of nutrient adequacy of rural elderly people in Sri Lanka. BMC Res. Notes. 2012, 5, 469. [Google Scholar] [CrossRef]
- Oldewage-Theron, W.H.; Kruger, R. Food variety and dietary diversity as indicators of the dietary adequacy and health atatus of an elderly population in Sharpeville, South Africa. J. Nutr. Elder. 2008, 27, 101–133. [Google Scholar] [CrossRef] [PubMed]
- Savy, M.; Martin-Prével, Y.; Danel, P.; Traissac, P.; Dabiré, H.; Delpeuch, F. Are dietary diversity scores related to the socio-economic and anthropometric status of women living in an urban area in Burkina Faso? Public Health Nutr. 2008, 11, 132–141. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Otto, M.C.; Anderson, C.A.M.; Dearborn, J.L.; Ferranti, E.P.; Mozaffarian, D.; Rao, G.; Wylie-Rosett, J.; Lichtenstein, A.H.; American Heart Association Behavioral Change for Improving Health Factors Committee of the Council on Lifestyle and Cardiometabolic Health and Council on Epidemiology and Prevention; Council on Cardiovascular and Stroke Nursing; et al. Dietary diversity: Implications for obesity prevention in adult populations. Circulation 2018, 138, e160–e168. [Google Scholar] [CrossRef] [PubMed]
- Fisberg, M.; Kovalskys, I.; Gómez, G.; Rigotti, A.; Cortés, L.Y.; Herrera-Cuenca, M.; Yépez, M.C.; Pareja, R.G.; Guajardo, V.; Zimberg, I.Z.; et al. Latin American Study of Nutrition and Health (ELANS): Rationale and study design. BMC Public Health 2016, 16, 93. [Google Scholar] [CrossRef] [PubMed]
- Previdelli, A.N.; Gómez, G.; Kovalskys, I.; Fisberg, M.; Cortés, L.Y.; Pareja, R.G.; Liria, M.R.; García, M.; Herrera-Cuenca, M.; Rigotti, A.; et al. Prevalence and determinant of misreporting of energy intake among Latin American populations: Results from ELANS study. Nutr. Res. 2019, 68, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Weiner, J.S. Human Biology: A Guide to Field Methods; International Biological Program by Blackwell Scientific Publications: Oxford, UK, 1981. [Google Scholar]
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Siekmann, J. Development of a WHO growth reference for school-aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Obesity: Preventing and Managing the Global Epidemic; WHO: Geneva, Switzerland, 1998. [Google Scholar]
- Moshfegh, A.J.; Rhodes, D.G.; Baer, D.J.; Murayi, T.; Clemens, J.C.; Rumpler, W.V.; Paul, D.R.; Sebastian, R.S.; Kuczynski, K.J.; Ingwersen, L.A.; et al. The US department of agriculture automated multiple-pass method reduces bias in the collection of energy intakes. Am. J. Clin. Nutr. 2008, 88, 324–332. [Google Scholar] [CrossRef]
- Kovalskys, I.; Fisberg, M.; Gómez, G.; Rigotti, A.; Cortés, L.Y.; Yépez, M.C.; Pareja, R.G.; Herrera-Cuenca, M.; Zimberg, I.Z.; Tucker, K.; et al. Standardization of the food composition database used in the Latin American Nutrition and Health Study (ELANS). Nutrients 2015, 7, 7914–7924. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Food and Nutrition Board; Committee to Review the Dietary Reference Intakes for Sodium and Potassium. Dietary Reference Intakes for Sodium and Potassium; Oria, M., Harrison, M., Stallings, V.A., Eds.; National Academies Press: Washington, DC, USA, 2019. Available online: https://www.ncbi.nlm.nih.gov/books/NBK545436/ (accessed on 1 July 2020).
- Institute of Medicine US (IOM). Dietary Reference Intakes. Food and Nutrition Board; National Academy Press: Washington, DC, USA, 2003. [Google Scholar]
- Bellows, A.L.; Canavan, C.R.; Blakstad, M.M.; Mosha, D.; Noor, R.A.; Webb, P.; Kinabo, J.; Masanja, H.; Fawzi, W.W. The relationship between dietary diversity among women of reproductive age and agricultural diversity in rural Tanzania. Food Nutr. Bull. 2020, 41, 50–60. [Google Scholar] [CrossRef]
- Chakona, G. Minimum dietary diversity scores for women indicate micronutrient adequacy and food insecurity atatus in South African towns. Nutrients 2017, 9, 812. [Google Scholar] [CrossRef] [PubMed]
- Gitagia, M.W.; Ramkat, R.C.; Mituki, D.M.; Termote, C.; Covic, N.; Cheserek, M.J. Determinants of dietary diversity among women of reproductive age in two different agro-ecological zones of Rongai Sub-County, Nakuru, Kenya. Food Nutr. Red. 2019, 18, 63. [Google Scholar] [CrossRef] [PubMed]
- Blackstone, S.; Sanghvi, T. A comparison of minimum dietary diversity in Bangladesh in 2011 and 2014. Matern. Child. Nutr. 2018, 14, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Masa, R.; Chowa, G.; Nyirenda, V. Socioeconomic correlates of dietary diversity and its association with adherence and psychosocial functioning of people living with HIV in rural Zambia. Nutr. Health 2018, 24, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, R.; Byrne, N.M.; Soares, M.J.; Katulanda, P.; Yadav, B.; Hills, A.P. High dietary diversity is associated with obesity in Sri Lankan adults: An evaluation of three dietary scores. BMC Public Health 2013, 13, 314. [Google Scholar] [CrossRef]
- Morseth, M.S.; Grewal, N.K.; Kaasa, I.S.; Hatloy, A.; Barikmo, I.; Henjum, S. Dietary diversity is related to socioeconomic status among adult Saharawi refugees living in Algeria. BMC Public Health 2017, 17, 621. [Google Scholar] [CrossRef]
- Mukherjee, A.; Sourabh, P.; Saha, I.; Som, T.K.; Ghose, G. Dietary diversity and its determinants: A community-based study among adult population of Durgapur, West Bengal. Med. J. Dr. DY Patil Visyapeeth 2018, 11, 94–98. [Google Scholar]
- Chagomoka, T.; Drescher, A.; Glaser, R.; Marschner, B.; Schlesinger, J.; Nyandoro, G. Women’s dietary diversity scores and childhood anthropometric measurements as indices of nutrition insecurity along the urban-rural continuum in Ouagadougou, Burkina Faso. Food Nutr. Res. 2016, 60, 29425. [Google Scholar] [CrossRef]
- Jemal, K.; Awol, M. Minimum dietary diversity score and associated factors among pregnant women at Alamata General Hospital, Raya Azebo Zone, Tigray Region, Ethiopia. J. Nutr. Metab. 2019, 2019, 8314359. [Google Scholar] [CrossRef]
- Larson, J.B.; Castellanos, P.; Jensen, L. Gender, household food security, and dietary diversity in Western Honduras. Glob. Food Sec. 2019, 20, 170–179. [Google Scholar] [CrossRef]
- Zhang, Q.; Chen, X.; Liu, Z.; Varma, D.S.; Wan, R.; Zhao, S. Diet diversity and nutritional status among adults in southwest China. PLoS ONE 2017, 12, e0172406. [Google Scholar] [CrossRef] [PubMed]
- Zongo, U.; Zoungrana, S.L.; Savadogo, A.; Thombiano-Coulibaly, N.; Traoré, A.S. Assessment of dietary diversity and vitamin a-rich foods consumption of pre-school children in rural community in Burkina Faso, an impact study approach. Austin J. Nutr. Food Sci. 2017, 5, 1–5. [Google Scholar]
- Nachvak, S.M.; Abdollahzad, H.; Mostafai, R.; Moradi, S.; Pasdar, Y.; Rezaei, M.; Esksndari, S. Dietary diversity score and its related factors among employees of Kermanshah University of Medical Sciences. Clin. Nutr. Res. 2017, 6, 247. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Mirmiran, P.; Esmaillzadeh, A.; Azizi, F. Dietary diversity score and cardiovascular risk factors in Tehranian adults. Public Health Nutr. 2006, 9, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Mirmiran, P.; Azadbakht, L.; Esmaillzadeh, A.; Azizi, F. Dietary diversity score in adolescents—A good indicator of the nutritional adequacy of diets: Tehran lipid and glucose study. Asia Pac. J. Clin. Nutr. 2004, 13, 56–60. [Google Scholar] [PubMed]
- Fernandez, C.; Kasper, N.M.; Miller, A.L.; Lumeng, J.C.; Peterson, K.E. Association of dietary variety and diversity with body mass index in US preschool children. Pediatrics 2016, 137, e20152307. [Google Scholar] [CrossRef] [PubMed]
- Farhangi, M.A.; Jahangiry, L. Dietary diversity score is associated with cardiovascular risk factors and serum adiponectin concentrations in patients with metabolic syndrome. BMC Cardiovasc. Disord. 2018, 18, 1–6. [Google Scholar] [CrossRef]
- Bezerra, I.N.; Sichieri, R. Household food diversity and nutritional status among adults in Brazil. Int. J. Behav. Nutr. Phys. Act. 2011, 8, 22. [Google Scholar] [CrossRef]
- Ponce, X.; Ramirez, E.; Delisle, H. A more diversified diet among Mexican men may also be more atherogenic. J Nutr. 2006, 136, 2921–2927. [Google Scholar] [CrossRef]
- De Oliveira Otto, M.C.; Padhye, N.S.; Bertoni, A.G.; Jacobs, D.R. Everything in moderation—Dietary diversity and duality, central obesity and risk of diabetes. PLoS ONE 2015, 10, e0141341. [Google Scholar] [CrossRef]
- Foote, J.A.; Murphy, S.P.; Wilkens, L.R.; Basiotis, P.P.; Carlson, A. Dietary variety increases the probability of nutrient adequacy among adults. J. Nutr. 2004, 134, 1779–1785. [Google Scholar] [CrossRef] [PubMed]
- Dhana, K.; Zong, G.; Yuan, C.; Schernhammer, E.; Zhang, C.; Wang, X.; Hu, F.B.; Chavarro, J.E.; Field, A.E.; Sun, Q. Lifestyle of women before pregnancy and the risk of offspring obesity during childhood through early adulhood. Int. J. Obesity 2019, 42, 1275–1284. [Google Scholar] [CrossRef] [PubMed]
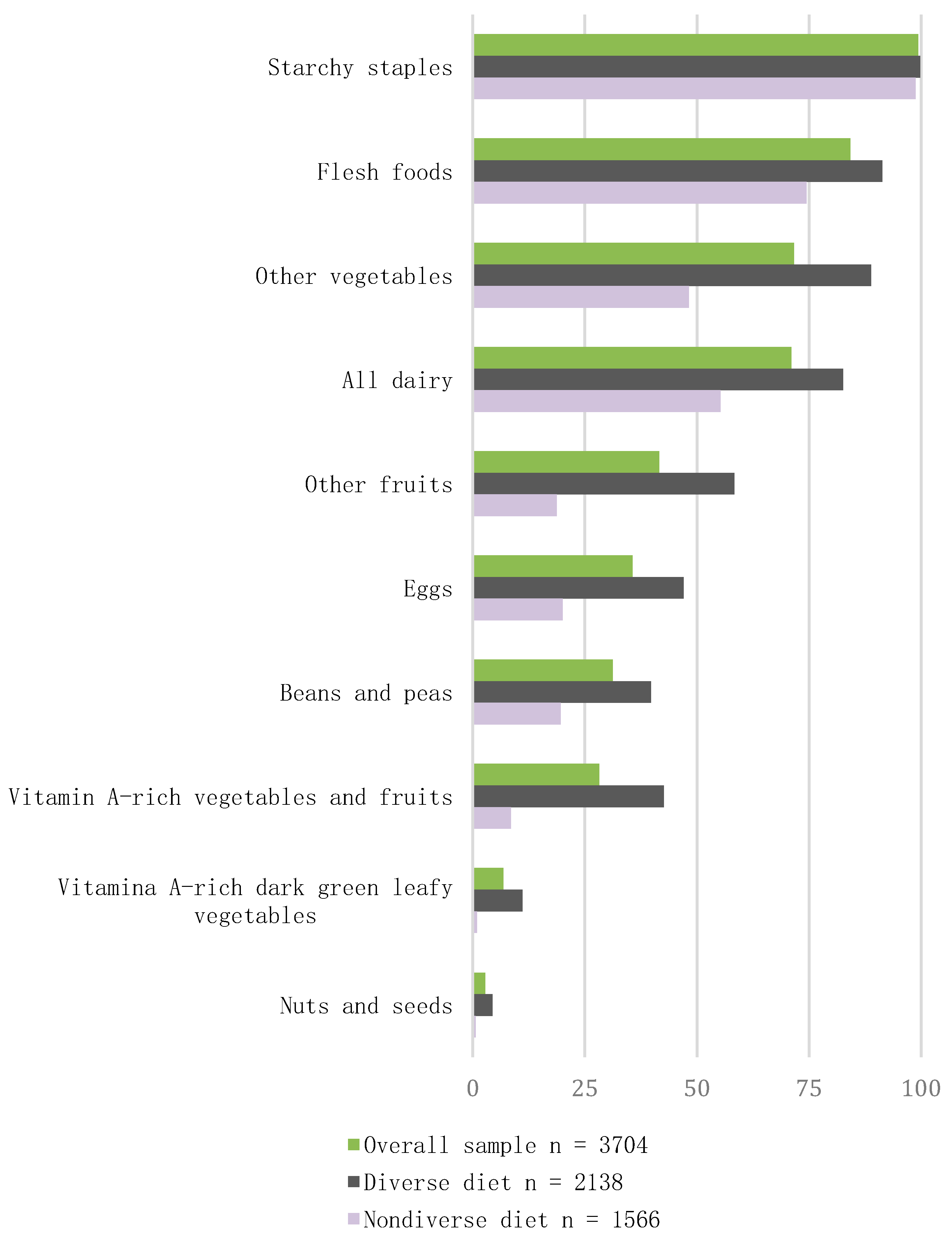
| Food Group | Specific Foods |
|---|---|
| Starchy staples (Grains, White Roots, Tuber, and Plantains) | Rice, Bread, Tortillas, Breakfast Cereals, Maize, Pasta, Cassava, Potatoes, Plantains Rip, and Green |
| Meat Poultry and Fish | Beef, Pork, Lamb, Veal, Chicken, Turkey, Liver, Other Organs, Canned Sardines and Tuna, Fresh Fish, and Seafood |
| Dark Green Leafy Vegetables | Broccoli, Mustard Greens, Turnip Greens, Collards, Spinach |
| Other Vitamin A-rich Fruits and Vegetables | Carrots, Sweet Potatoes, Winter Squash, Pumpkin, Cantaloupe, Mango, Papaya, and Apricot |
| Other Vegetables | Lettuce, Mixed Greens, Tomatoes, Cauliflower, Radish, Okra, Green Peas, Green Pepper, Onions, Shallots, Leeks, String Beans, and Others |
| Other Fruits | Oranges, Grapefruits, Banana, Apples, Pears, Strawberries, Watermelon, Kiwi, Berries, Melons, Avocado, Lemon, Lime, Tangerine, Pineapple, Tamarind, and Others |
| Pulses (Beans, Peas, and Lentils) | Lentils, Beans (Black, Kidney, Pinto, and Others), Chickpeas and Soybeans |
| Dairy | Milk, Yogurt, and Cheese |
| Eggs | Eggs |
| Nuts and Seeds | Pecans, Cashews, Peanuts, Almonds, Walnuts, Sunflower Seeds, other Seeds |
| Dietary Diversity Score (DDS) | Participants Reaching the Minimum DDS | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| n | Mean | SD | p | η2 (%) | n | % | p | η2 (%) | |
| Overall | 3704 | 4.73 | 1.34 | 2138 | 57.7 | ||||
| Age ranges | |||||||||
| 15–19 | 539 | 4.61 | 1.28 | 0.081 | 0.1 | 288 | 13.5 | 0.365 | 0.1 |
| 20–34 | 1771 | 4.73 | 1.34 | 1019 | 47.7 | ||||
| 35–49 | 1394 | 4.76 | 1.35 | 831 | 38.9 | ||||
| Socioeconomic Status | |||||||||
| High | 529 | 4.96 | 1.35 | 0.001 | 0.9 | 339 | 64.1 | 0.028 | 0.3 |
| Medium | 1593 | 4.78 | 1.33 | 956 | 60.0 | ||||
| Low | 1582 | 4.59 | 1.33 | 843 | 53.3 | ||||
| Country | |||||||||
| Argentina | 521 | 4.35 | 1.35 | 0.001 | 5.4 | 236 | 45.3 | 0.001 | 4 |
| Brazil | 798 | 4.61 | 1.34 | 435 | 54.5 | ||||
| Chile | 345 | 4.71 | 1.23 | 205 | 59.4 | ||||
| Colombia | 464 | 4.71 | 1.36 | 270 | 58.2 | ||||
| Costa Rica | 309 | 4.90 | 1.36 | 190 | 61.5 | ||||
| Ecuador | 324 | 5.16 | 1.30 | 228 | 70.4 | ||||
| Peru | 480 | 5.28 | 1.29 | 347 | 72.3 | ||||
| Venezuela | 463 | 4.38 | 1.15 | 227 | 49.0 | ||||
| Nutritional Status | |||||||||
| Underweight | 128 | 4.81 | 1.33 | 0.252 | 0.1 | 77 | 60.2 | 0.764 | 0.1 |
| Normal Weight | 1444 | 4.73 | 1.31 | 834 | 57.8 | ||||
| Overweight | 1177 | 4.77 | 1.33 | 690 | 58.6 | ||||
| Obesity | 952 | 4.66 | 1.38 | 534 | 56.1 | ||||
| Variables | DDS < 5 | DDS ≥ 5 | p | η2 (%) | ||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| Energy (kcal) | 1721.75 | 527.00 | 1902.50 | 505.93 | 0.0001 | 3 |
| Macronutrients * | ||||||
| Omega-3 Fatty Acids from Plants (g) | 0.04 | 0.03 | 0.06 | 0.03 | 0.001 | 4 |
| Added Sugars (g) | 36.86 | 16.12 | 32.53 | 13.10 | 0.001 | 2 |
| Trans Fatty Acids (g) | 1.28 | 0.88 | 1.19 | 0.82 | 0.001 | 0.3 |
| Cholesterol (mg) | 144.83 | 48.25 | 150.30 | 45.32 | 0.001 | 0.3 |
| Monounsaturated Fats (g) | 11.27 | 2.57 | 11.04 | 2.47 | 0.005 | 0.2 |
| Protein (g) | 40.20 | 7.88 | 40.70 | 7.37 | 0.050 | 0.1 |
| Saturated Fats (g) | 11.11 | 2.91 | 10.94 | 2.81 | 0.068 | 0.1 |
| Total Fats (g) | 33.95 | 6.46 | 33.64 | 6.16 | 0.137 | 0.1 |
| Polyunsaturated Fats (g) | 8.68 | 2.26 | 8.59 | 2.04 | 0.193 | 0.01 |
| Carbohydrates (g) | 138.10 | 20.94 | 138.67 | 20.13 | 0.403 | 0.01 |
| Micronutrients * | ||||||
| Vitamin A (mg) | 293.56 | 151.00 | 345.36 | 149.46 | 0.001 | 3 |
| Magnesium (mg) | 117.72 | 25.73 | 126.84 | 25.75 | 0.001 | 3 |
| Pyridoxine (mg) | 0.85 | 0.23 | 0.90 | 0.21 | 0.001 | 2 |
| Vitamin D (mg) | 1.73 | 1.12 | 2.02 | 1.09 | 0.001 | 2 |
| Phosphorous (mg) | 524.29 | 111.01 | 552.35 | 102.24 | 0.001 | 2 |
| Folate Equivalents (mg) | 262.13 | 70.96 | 276.90 | 66.91 | 0.001 | 1 |
| Zinc (mg) | 6.31 | 3.28 | 5.80 | 2.18 | 0.001 | 0.8 |
| Vitamin C (mg) | 48.30 | 75.29 | 57.81 | 47.24 | 0.001 | 0.6 |
| Calcium (mg) | 290.82 | 115.83 | 307.45 | 106.89 | 0.001 | 0.5 |
| Cooper (mg) | 0.82 | 0.76 | 0.73 | 0.47 | 0.001 | 0.004 |
| Cobalamin (mg) | 2.14 | 0.94 | 2.23 | 0.88 | 0.002 | 0.2 |
| Thiamin (mg) | 0.88 | 0.20 | 0.86 | 0.17 | 0.003 | 0.2 |
| Sodium (mg) | 1370.50 | 473.38 | 1331.06 | 555.28 | 0.023 | 0.1 |
| Vitamin E (mg) | 0.20 | 0.09 | 0.21 | 0.09 | 0.059 | 0.1 |
| Iron (mg) | 6.78 | 1.64 | 6.84 | 1.42 | 0.234 | 0.01 |
| Selenium (mg) | 60.81 | 12.41 | 60.39 | 10.75 | 0.275 | 0.01 |
| Niacin (mg) | 11.92 | 2.74 | 11.84 | 2.45 | 0.376 | 0.01 |
| Riboflavin (mg) | 0.82 | 0.21 | 0.82 | 0.20 | 0.515 | 0.01 |
| Food groups * | ||||||
| Fruit (g) | 31.24 | 35.14 | 50.34 | 44.62 | 0.001 | 5 |
| Fiber (g) | 7.66 | 2.48 | 8.74 | 2.59 | 0.001 | 4 |
| Vegetables (g) | 53.34 | 33.62 | 63.39 | 32.37 | 0.001 | 2 |
| Processed Red Meat (g) | 11.89 | 9.09 | 9.74 | 7.84 | 0.001 | 2 |
| Sugar-Sweetened Beverages (g) | 380.57 | 254.48 | 329.68 | 185.59 | 0.001 | 1 |
| Read Meat (g) | 35.93 | 19.40 | 32.62 | 17.58 | 0.001 | 0.8 |
| Dairy (g) | 46.38 | 49.74 | 54.39 | 51.95 | 0.001 | 0.6 |
| Nuts and Seeds (g) | 0.71 | 2.37 | 1.29 | 4.61 | 0.001 | 0.6 |
| Wholegrains (g) | 4.29 | 7.93 | 5.43 | 9.03 | 0.001 | 0.4 |
| Beans and Legumes (g) | 18.20 | 19.14 | 20.17 | 17.82 | 0.001 | 0.3 |
| Fish (g) | 10.08 | 11.40 | 10.43 | 10.82 | 0.340 | 0.01 |
| Variables | DDS < 5 | DDS ≥ 5 | ||||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | p1 | η2 (%) | |
| Body Weight (kg) | 67.32 | 15.46 | 66.69 | 15.26 | 0.217 | 0.0 |
| BMI | 27.02 | 5.89 | 26.88 | 5.76 | 0.464 | 0.0 |
| Waist Circumference (cm) | 85.56 | 14.34 | 85.27 | 13.69 | 0.532 | 0.0 |
| Neck Circumference (cm) | 33.43 | 3.39 | 33.49 | 3.24 | 0.583 | 0.0 |
| Hip Circumference (cm) | 101.62 | 12.08 | 100.95 | 11.63 | 0.090 | 0.0 |
| Nutrient | Overall | DDS < 5 | DDS ≥ 5 | p | η2 (%) | r1 | p | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |||||
| Magnesium | 0.788 | 0.18 | 0.725 | 0.19 | 0.836 | 0.16 | 0.001 | 9 | 0.361 | 0.001 |
| Vitamin A | 0.879 | 0.19 | 0.813 | 0.22 | 0.928 | 0.14 | 0.001 | 9 | 0.352 | 0.001 |
| Vitamin C | 0.873 | 0.21 | 0.799 | 0.25 | 0.928 | 0.15 | 0.001 | 9 | 0.334 | 0.001 |
| Pyridoxine | 0.973 | 0.08 | 0.952 | 0.11 | 0.989 | 0.05 | 0.001 | 5 | 0.273 | 0.001 |
| Vitamin D | 0.342 | 0.20 | 0.292 | 0.18 | 0.380 | 0.21 | 0.001 | 4 | 0.269 | 0.001 |
| Cooper | 0.982 | 0.07 | 0.968 | 0.09 | 0.993 | 0.04 | 0.001 | 3 | 0.216 | 0.001 |
| Zinc | 0.976 | 0.78 | 0.960 | 0.10 | 0.988 | 0.05 | 0.001 | 3 | 0.204 | 0.001 |
| Calcium | 0.684 | 0.45 | 0.594 | 0.49 | 0.750 | 0.43 | 0.001 | 3 | 0.201 | 0.001 |
| Phosphorus | 0.977 | 0.08 | 0.962 | 0.10 | 0.989 | 0.05 | 0.001 | 3 | 0.194 | 0.001 |
| Folate | 0.702 | 0.17 | 0.736 | 0.19 | 0.677 | 0.17 | 0.001 | 3 | −0.205 | 0.001 |
| Riboflavin | 0.987 | 0.06 | 0.978 | 0.08 | 0.994 | 0.04 | 0.001 | 2 | 0.183 | 0.001 |
| Cobalamin | 0.980 | 0.86 | 0.968 | 0.11 | 0.990 | 0.06 | 0.001 | 2 | 0.174 | 0.001 |
| Iron | 0.976 | 0.08 | 0.964 | 0.10 | 0.986 | 0.06 | 0.001 | 2 | 0.162 | 0.001 |
| Vitamin E | 0.031 | 0.02 | 0.029 | 0.02 | 0.033 | 0.02 | 0.001 | 2 | 0.148 | 0.001 |
| Thiamin | 0.989 | 0.06 | 0.982 | 0.07 | 0.994 | 0.04 | 0.001 | 1 | 0.142 | 0.001 |
| Niacin | 0.996 | 0.03 | 0.993 | 0.04 | 0.999 | 0.02 | 0.001 | 0.7 | 0.115 | 0.001 |
| Selenium | 0.998 | 0.02 | 0.998 | 0.02 | 0.999 | 0.02 | 0.135 | 0.1 | 0.047 | 0.004 |
| MAR | 0.806 | 0.06 | 0.807 | 0.07 | 0.850 | 0.05 | 0.0001 | 11 | 0.393 | 0.0001 |
| Predictor 1 | Estimate | SE | Z | p | Odds Ratio 2 | 95% Confidence Interval | |
|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||
| Intercept | −3.689 | 0.131 | −28.26 | 0.001 | 0.03 | 0.02 | 0.03 |
| Vitamin C | 1.589 | 0.115 | 13.78 | 0.001 | 4.9 (7.1) 3 | 3.91 | 6.14 |
| Vitamin A | 1.99 | 0.131 | 15.21 | 0.001 | 7.3 (6.3)3 | 5.66 | 9.45 |
| Magnesium | 0.962 | 0.158 | 6.08 | 0.001 | 2.6 (5.0) 3 | 1.92 | 3.57 |
| Vitamin D | 0.849 | 0.133 | 6.37 | 0.001 | 2.3 (3.4) 3 | 1.8 | 3.03 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez, G.; Nogueira Previdelli, Á.; Fisberg, R.M.; Kovalskys, I.; Fisberg, M.; Herrera-Cuenca, M.; Cortés Sanabria, L.Y.; Yépez García, M.C.; Rigotti, A.; Liria-Domínguez, M.R.; et al. Dietary Diversity and Micronutrients Adequacy in Women of Childbearing Age: Results from ELANS Study. Nutrients 2020, 12, 1994. https://doi.org/10.3390/nu12071994
Gómez G, Nogueira Previdelli Á, Fisberg RM, Kovalskys I, Fisberg M, Herrera-Cuenca M, Cortés Sanabria LY, Yépez García MC, Rigotti A, Liria-Domínguez MR, et al. Dietary Diversity and Micronutrients Adequacy in Women of Childbearing Age: Results from ELANS Study. Nutrients. 2020; 12(7):1994. https://doi.org/10.3390/nu12071994
Chicago/Turabian StyleGómez, Georgina, Ágatha Nogueira Previdelli, Regina Mara Fisberg, Irina Kovalskys, Mauro Fisberg, Marianella Herrera-Cuenca, Lilia Yadira Cortés Sanabria, Martha Cecilia Yépez García, Attilio Rigotti, María Reyna Liria-Domínguez, and et al. 2020. "Dietary Diversity and Micronutrients Adequacy in Women of Childbearing Age: Results from ELANS Study" Nutrients 12, no. 7: 1994. https://doi.org/10.3390/nu12071994
APA StyleGómez, G., Nogueira Previdelli, Á., Fisberg, R. M., Kovalskys, I., Fisberg, M., Herrera-Cuenca, M., Cortés Sanabria, L. Y., Yépez García, M. C., Rigotti, A., Liria-Domínguez, M. R., Guajardo, V., Quesada, D., Murillo, A. G., & Brenes, J. C., on behalf of the ELANS Study Group. (2020). Dietary Diversity and Micronutrients Adequacy in Women of Childbearing Age: Results from ELANS Study. Nutrients, 12(7), 1994. https://doi.org/10.3390/nu12071994






