Effect of the Fat Eaten at Breakfast on Lipid Metabolism: A Crossover Trial in Women with Cardiovascular Risk
Abstract
1. Introduction
2. Materials and Methods
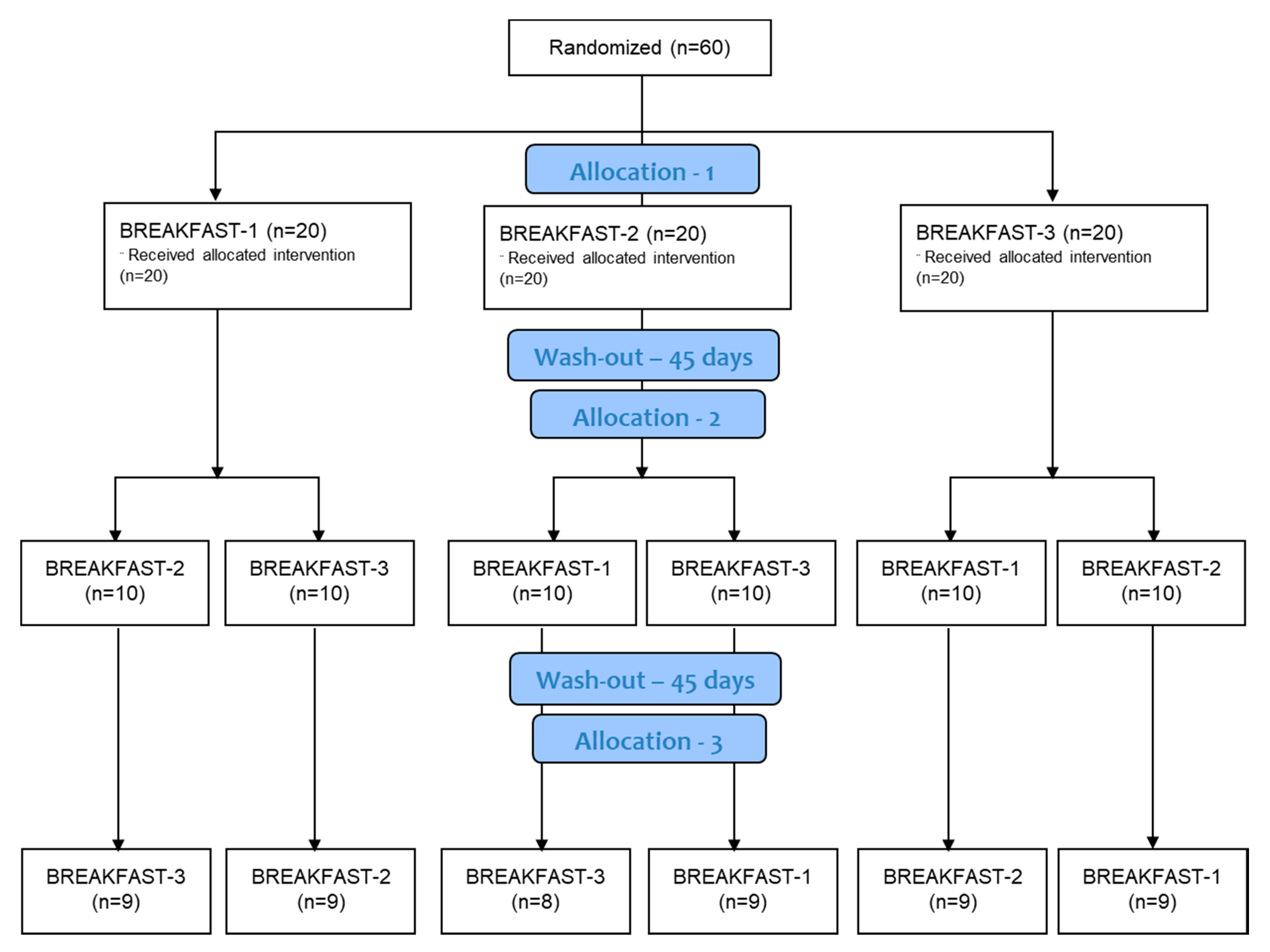
2.1. Study Design
2.2. Participants
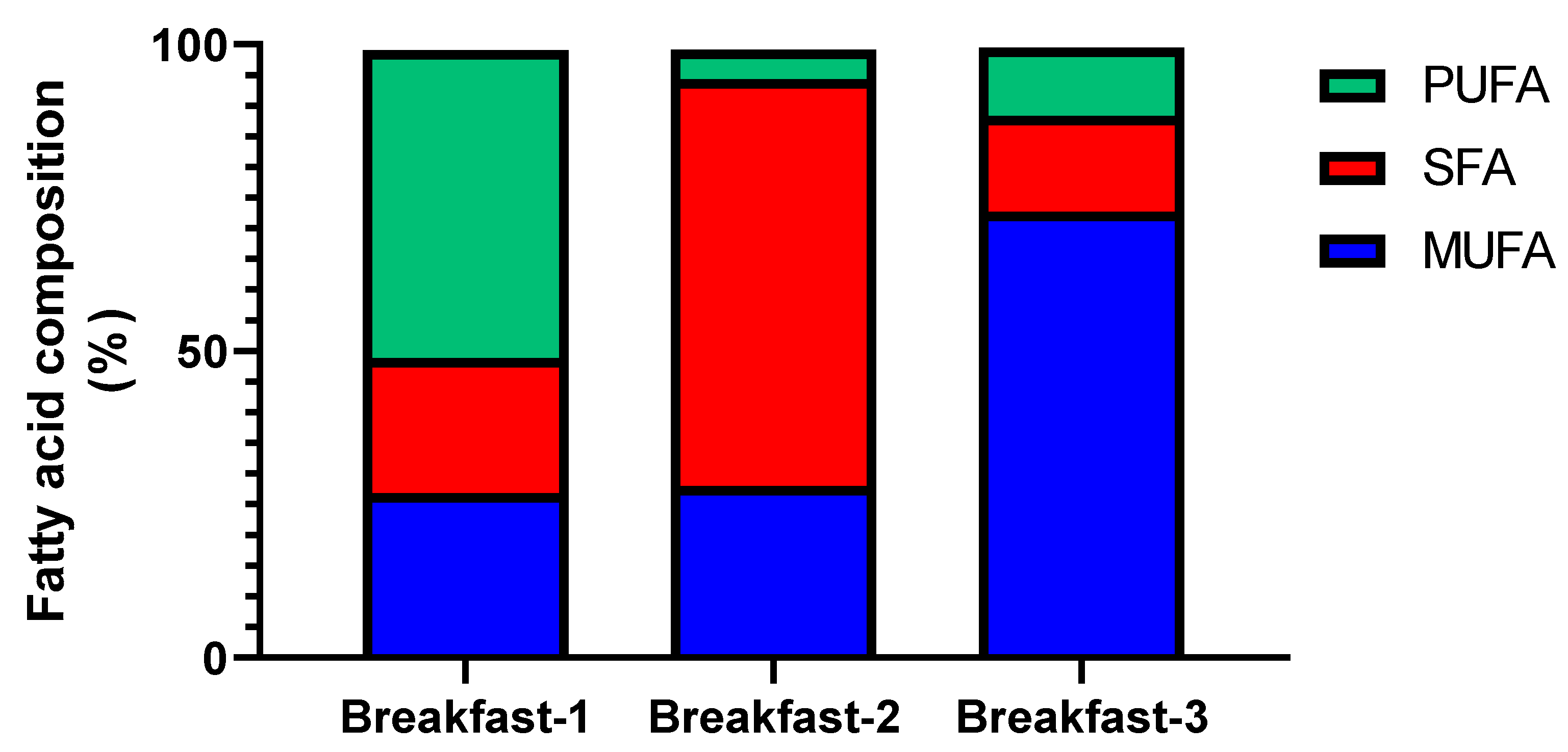
2.3. Intervention
2.4. Lipid Metabolism Parameters
2.5. Statistical Analysis
3. Results
3.1. Clinical Characteristics of the Subjects
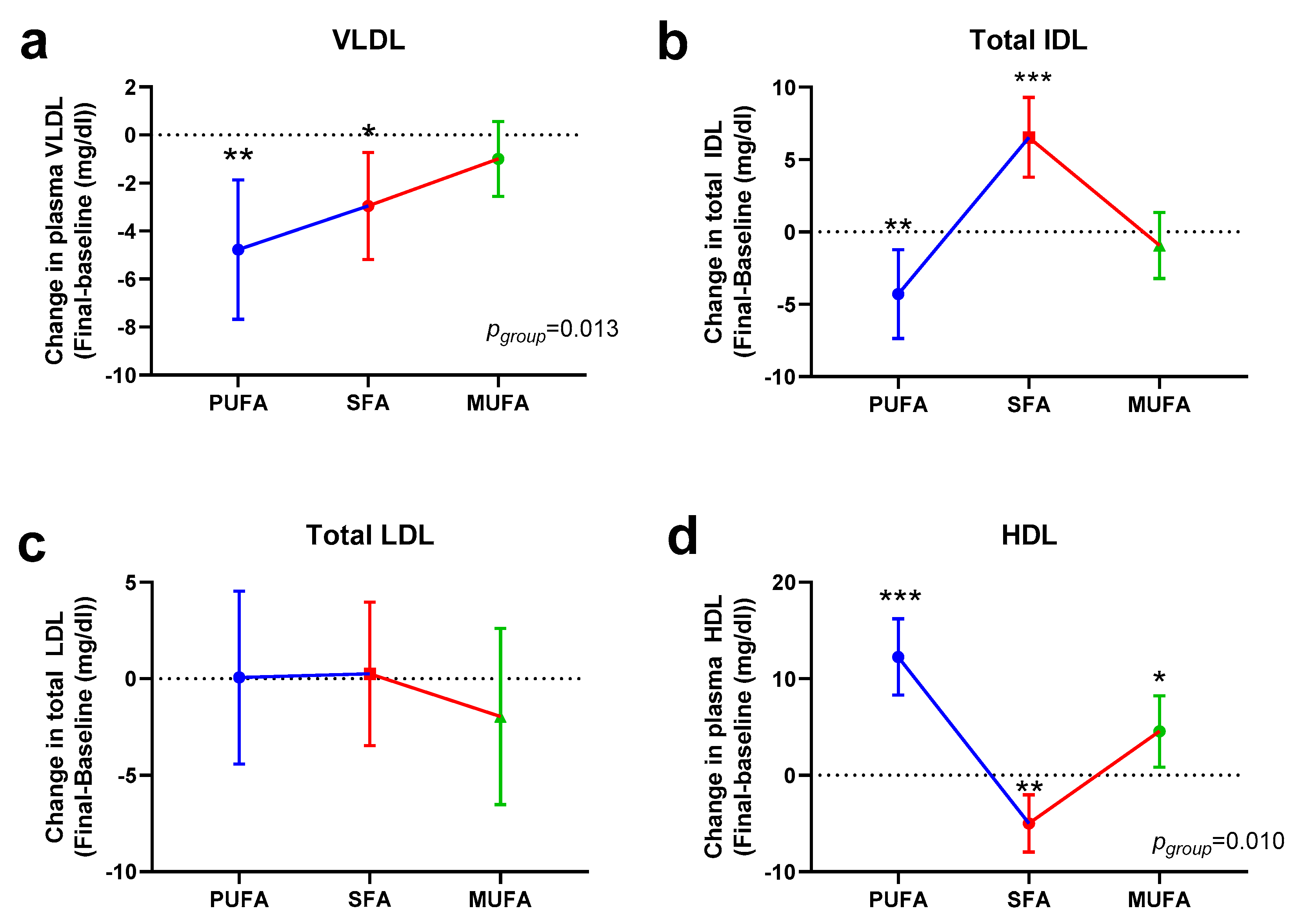
3.2. Effect of the Breakfast Fat on Lipoprotein Subfraction
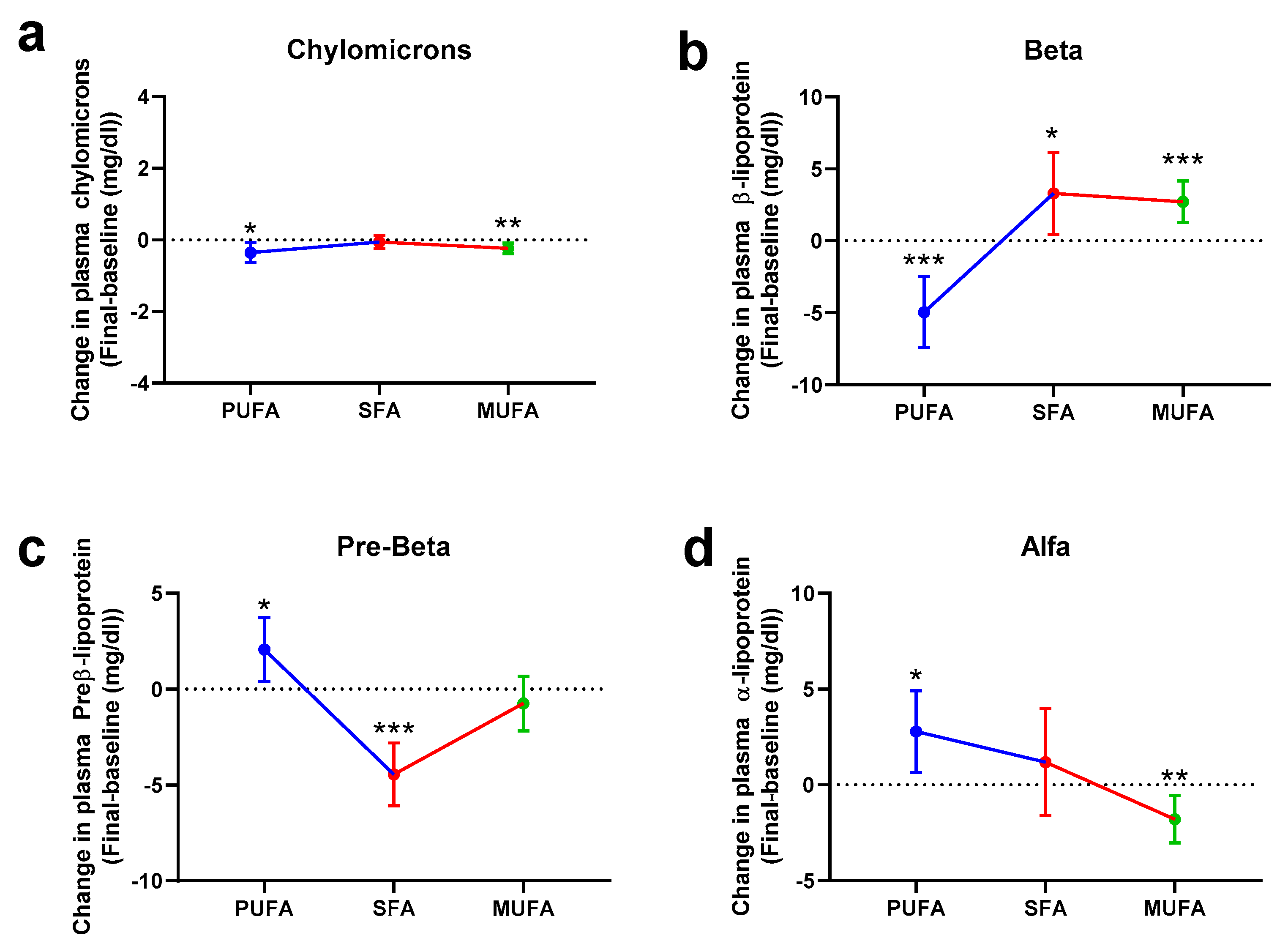
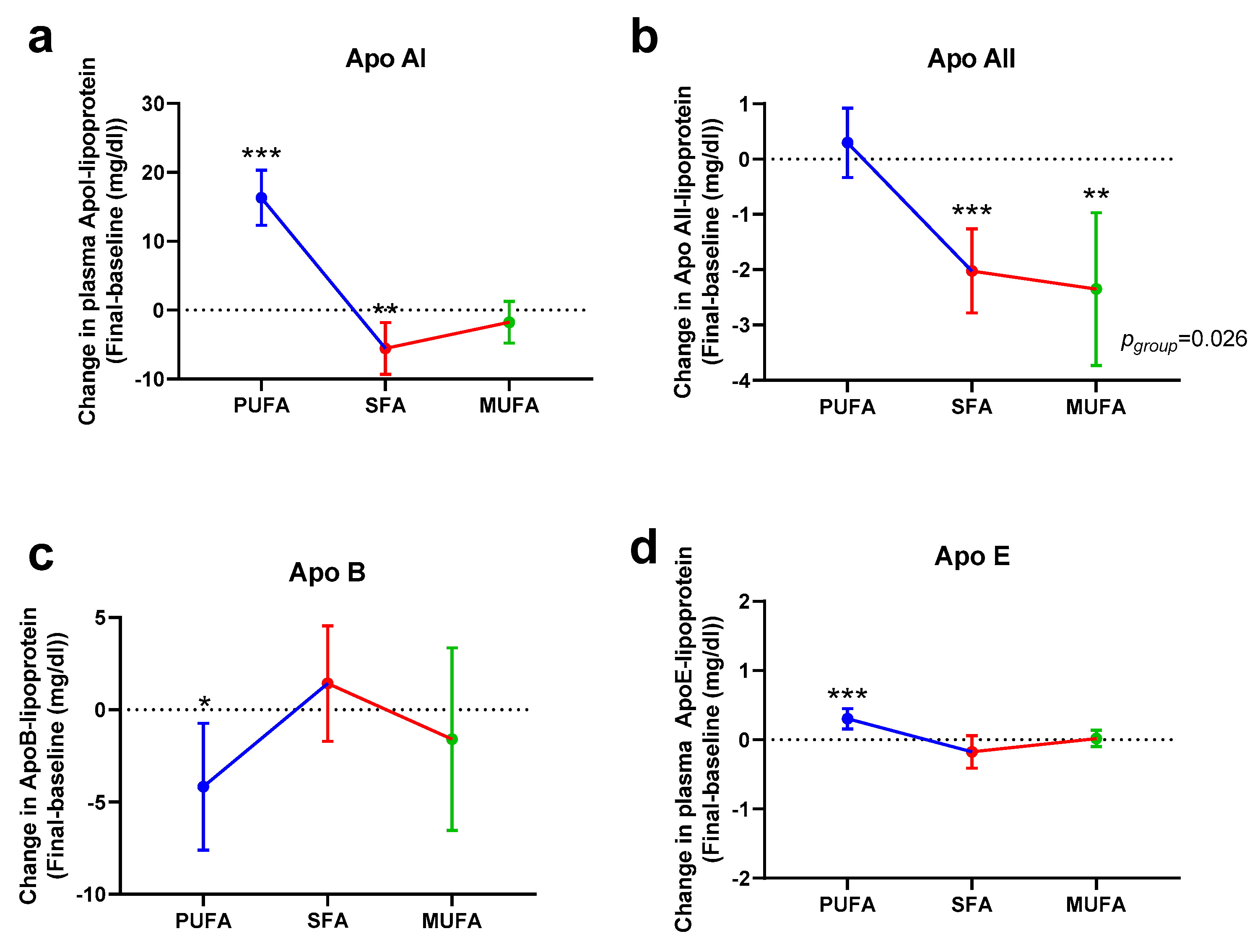
3.3. Effect of the Breakfast Fat on Apolipoproteins
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. The Top 10 Causes of Death. World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 13 March 2020).
- Van Deventer, H.E.; Miller, W.G.; Myers, G.L.; Sakurabayashi, I.; Bachmann, L.M.; Caudill, S.P.; Dziekonski, A.; Edwards, S.; Kimberly, M.M.; Korzun, W.J.; et al. Non-HDL cholesterol shows improved accuracy for cardiovascular risk score classification compared to direct or calculated LDL cholesterol in a dyslipidemic population. Clin. Chem. 2011, 57, 490–501. [Google Scholar] [CrossRef]
- Ridker, P.M.; Rifai, N.; Cook, N.R.; Bradwin, G.; Buring, J.E. Non-HDL cholesterol, apolipoproteins A-I and B 100, standard lipid measures, lipid ratios, and CRP as risk factors for cardiovascular disease in women. J. Am. Med. Assoc. 2005, 294, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Boekholdt, S.M.; Arsenault, B.J.; Mora, S.; Pedersen, T.R.; LaRosa, J.C.; Nestel, P.J.; Simes, R.J.; Durrington, P.; Hitman, G.A.; Welch, K.M.A.; et al. Association of LDL cholesterol, non-HDL cholesterol, and apolipoprotein B levels with risk of cardiovascular events among patients treated with statins: A meta-analysis. JAMA J. Am. Med. Assoc. 2012, 307, 1302–1309. [Google Scholar] [CrossRef] [PubMed]
- Parish, S.; Peto, R.; Palmer, A.; Clarke, R.; Lewington, S.; Offer, A.; Whitlock, G.; Clark, S.; Youngman, L.; Sleight, P.; et al. The joint effects of apolipoprotein B, apolipoprotein A1, LDL cholesterol, and HDL cholesterol on risk: 3510 cases of acute myocardial infarction and 9805 controls. Eur. Heart J. 2009, 30, 2137–2146. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.S.; Chen, Y.C.; Tian, Y.C.; Yang, C.W.; Lien, J.M.; Fang, J.T.; Wu, C.S.; Hung, C.F.; Hwang, T.L.; Tsai, Y.H.; et al. Serum levels of apolipoprotein A-I and high-density lipoprotein can predict organ failure in acute pancreatitis. Crit. Care 2015, 19. [Google Scholar] [CrossRef] [PubMed]
- Chapman, M.J.; ORSONI, A.; Tan, R.; Mellett, N.A.; Nguyen, A.; Robillard, P.; Giral, P.; Therond, P.; Meikle, P.J. LDL subclass lipidomics in atherogenic dyslipidemia:Effect of statin therapy on bioactive lipids and dense LDL. J. Lipid Res. 2020, 61, 911–932. [Google Scholar] [CrossRef] [PubMed]
- Albers, J.J.; Slee, A.; Fleg, J.L.; O’Brien, K.D.; Marcovina, S.M. Relationship of baseline HDL subclasses, small dense LDL and LDL triglyceride to cardiovascular events in the AIM-HIGH clinical trial. Atherosclerosis 2016, 251, 454–459. [Google Scholar] [CrossRef]
- Muggleton, E.; Muggleton, T. LDL cholesterol consists of different subclasses, more precise diagnostics are required. Heart 2019, 105, 1290. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Chapman, M.J.; Ray, K.; Borén, J.; Andreotti, F.; Watts, G.F.; Ginsberg, H.; Amarenco, P.; Catapano, A.; Descamps, O.S.; et al. Lipoprotein(a) as a cardiovascular risk factor: Current status. Eur. Heart J. 2010, 31, 2844–2853. [Google Scholar] [CrossRef]
- Krauss, R.M. Lipoprotein subfractions and cardiovascular disease risk. Curr. Opin. Lipidol. 2010, 21, 305–311. [Google Scholar] [CrossRef]
- Garaulet, M.; Hernandez-Morante, J.J.; Tebar, F.J.; Zamora, S. Relation between degree of obesity and site-specific adipose tissue fatty acid composition in a Mediterranean population. Nutrition 2011. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Hernandez-Morante, J.J.; Lujan, J.; Tebar, F.J.; Zamora, S. Relationship between fat cell size and number and fatty acid composition in adipose tissue from different fat depots in overweight/obese humans. Int. J. Obes. 2006, 30, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Hennig, B.; Reiterer, G.; Toborek, M.; Matveev, S.V.; Daugherty, A.; Smart, E.; Robertson, L.W. Dietary fat interacts with PCBs to induce changes in lipid metabolism in mice deficient in low-density lipoprotein receptor. Environ. Health Perspect. 2005, 113, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.; West, K. Mechanisms by which Dietary Fatty Acids Modulate Plasma Lipids. J. Nutr. 2005, 135, 2075–2078. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.S.G.; Cañavate, R.; Hernández, C.M.M.; Cara-Salmerón, V.; Hernández-Morante, J.J. The association among chronotype, timing of food intake and food preferences depends on body mass status. Eur. J. Clin. Nutr. 2017, 71, 736–742. [Google Scholar] [CrossRef]
- Garaulet, M.; Gomez-Abellan, P.; Alburquerque-Bejar, J.J.; Lee, Y.-C.; Ordovas, J.M.; Scheer, F.A.J.L. Timing of food intake predicts weight loss effectiveness. Int. J. Obes. 2013, 37, 604–611. [Google Scholar] [CrossRef]
- Arora, T.; Taheri, S. Associations among late chronotype, body mass index and dietary behaviors in young adolescents. Int. J. Obes. 2015, 39, 39–44. [Google Scholar] [CrossRef]
- Jakubowicz, D.; Wainstein, J.; Landau, Z.; Raz, I.; Ahren, B.; Chapnik, N.; Ganz, T.; Menaged, M.; Barnea, M.; Bar-Dayan, Y.; et al. Influences of Breakfast on Clock Gene Expression and Postprandial Glycemia in Healthy Individuals and Individuals With Diabetes: A Randomized Clinical Trial. Diabetes Care 2017, 40, 1573–1579. [Google Scholar] [CrossRef]
- Deshmukh-Taskar, P.R.; Nicklas, T.A.; O’Neil, C.E.; Keast, D.R.; Radcliffe, J.D.; Cho, S. The relationship of breakfast skipping and type of breakfast consumption with nutrient intake and weight status in children and adolescents: The National Health and Nutrition Examination Survey 1999–2006. J. Am. Diet. Assoc. 2010, 110, 869–878. [Google Scholar] [CrossRef]
- Spence, C. Breakfast: The most important meal of the day? Int. J. Gastron. Food Sci. 2017, 8, 1–6. [Google Scholar] [CrossRef]
- Gibney, M.J.; Barr, S.I.; Bellisle, F.; Drewnowski, A.; Fagt, S.; Livingstone, B.; Masset, G.; Moreiras, G.V.; Moreno, L.A.; Smith, J.; et al. Breakfast in human nutrition: The international breakfast research initiative. Nutrients 2018, 10, 559. [Google Scholar] [CrossRef] [PubMed]
- Lasserre, M.; Mendy, F.; Spielmann, D.; Jacotot, B. Effects of different dietary intake of essential fatty acids on C20:3ω6 and C20:4ω6 serum levels in human adults. Lipids 1985, 20, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Mayr, S.; Buchner, A.; Erdfelder, E.; Faul, F. A short tutorial of G Power. Tutor. Quant. Methods Psychol. 2007, 3, 51–59. [Google Scholar] [CrossRef]
- Galindo Muñoz, J.S.; Gómez Gallego, M.; Díaz Soler, I.; Barberá Ortega, M.C.; Martínez Cáceres, C.M.; Hernández Morante, J.J. Effect of a chronotype-adjusted diet on weight loss effectiveness: A randomized clinical trial. Clin. Nutr. 2019. [Google Scholar] [CrossRef]
- Tsui, A.K.Y.; Thomas, D.; Hunt, A.; Estey, M.; Christensen, C.-L.; Higgins, T.; Sandhu, I.; Rodriguez-Capote, K. Analytical sensitivity and diagnostic performance of serum protein electrophoresis on the HYDRAGEL 30 PROTEIN(E) β1-β2 Sebia Hydrasys system. Clin. Biochem. 2018, 51, 80–84. [Google Scholar] [CrossRef]
- Weinstock, N.; Bartholome, M.; Seidel, D. Determination of apolipoprotein A-I by kinetic nephelometry. Biochim. Biophys. Acta (BBA)/Lipids Lipid Metab. 1981, 663, 279–288. [Google Scholar] [CrossRef]
- Muñiz, N. Measurement of plasma lipoproteins by electrophoresis on polyacrylamide gel. Clin. Chem. 1977, 23, 1826–1833. [Google Scholar] [CrossRef]
- Chen, M.; Li, Y.; Sun, Q.; Pan, A.; Manson, J.E.; Rexrode, K.M.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Dairy fat and risk of cardiovascular disease in 3 cohorts of US adults. Am. J. Clin. Nutr. 2016, 104, 1209–1217. [Google Scholar] [CrossRef]
- Mensink, R.P. Effects of Saturated Fatty Acids on Serum Lipids and Lipoproteins: A Systematic Review and Regression Analysis; World Health Organization: Maastrich, The Netherlands, 2016; pp. 1–63. ISBN 978-92-4-156534-9. [Google Scholar]
- Yao, Y.; Pek, S.X.; Toh, D.W.K.; Xia, X.; Kim, J.E. Effects of fatty acids composition in a breakfast meal on the postprandial lipid responses: A systematic review and meta-analysis of randomised controlled trials. Int. J. Food Sci. Nutr. 2020. [Google Scholar] [CrossRef]
- Milan, A.M.; Nuora, A.; Pundir, S.; Pileggi, C.A.; Markworth, J.F.; Linderborg, K.M.; Cameron-Smith, D. Older adults have an altered chylomicron response to a high-fat meal. Br. J. Nutr. 2016, 115, 791–799. [Google Scholar] [CrossRef]
- Aneni, E.C.; Osondu, C.U.; De La Cruz, J.; Martin, S.S.; Blaha, M.J.; Younus, A.; Feldman, T.; Agatston, A.S.; Veledar, E.; Nasir, K. Lipoprotein sub-fractions by ion-mobility analysis and its association with subclinical coronary atherosclerosis in high-risk individuals. J. Atheroscler. Thromb. 2019, 26, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, A.C.M.; Maranho, R.C.; Sousa, H.P.D.; Schaefer, E.J.; Santos, R.D. Effects of margarines and butter consumption on lipid profiles, inflammation markers and lipid transfer to HDL particles in free-living subjects with the metabolic syndrome. Eur. J. Clin. Nutr. 2010, 64, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Packard, C.J. Small dense low-density lipoprotein and its role as an independent predictor of cardiovascular disease. Curr. Opin. Lipidol. 2006, 17, 412–417. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, M.; Berneis, K. Low-density lipoprotein size and cardiovascular risk assessment. QJM 2006, 99, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 2002, 106, 3143–3421. [CrossRef]
- Vecka, M.; Dušejovská, M.; Stankova, B.; Zeman, M.; Vavrova, L.; Kodydkova, J.; Slaby, A.; Zak, A. N-3 polyunsaturated fatty acids in the treatment of atherogenic dyslipidemia. Neuro Endocrinol. Lett. 2012, 33, 87–92. [Google Scholar]
- Guay, V.; Lamarche, B.; Charest, A.; Tremblay, A.J.; Couture, P. Effect of short-term low- and high-fat diets on low-density lipoprotein particle size in normolipidemic subjects. Metab.: Clin. Exp. 2012, 61, 76–83. [Google Scholar] [CrossRef]
- Merkel, M.; Velez-Carrasco, W.; Hudgins, L.C.; Breslow, J.L. Compared with saturated fatty acids, dietary monounsaturated fatty acids and carbohydrates increase atherosclerosis and VLDL cholesterol levels in LDL receptor-deficient, but not apolipoprotein E-deficient, mice. Proc. Natl. Acad. Sci. USA 2001, 98, 13294–13299. [Google Scholar] [CrossRef]
- Ito, K.; Yoshida, H.; Yanai, H.; Kurosawa, H.; Sato, R.; Manita, D.; Hirowatari, Y.; Tada, N. Relevance of intermediate-density lipoprotein cholesterol to Framingham risk score of coronary heart disease in middle-aged men with increased non-HDL cholesterol. Int. J. Cardiol. 2013, 168, 3853–3858. [Google Scholar] [CrossRef]
- Walldius, G.; Jungner, I.; Holme, I.; Aastveit, A.H.; Kolar, W.; Steiner, E. High apolipoprotein B, low apolipoprotein A-I, and improvement in the prediction of fatal myocardial infarction (AMORIS study): A prospective study. Lancet 2001, 358, 2026–2033. [Google Scholar] [CrossRef]
- Maïga, S.F.; Kalopissis, A.D.; Chabert, M. Apolipoprotein A-II is a key regulatory factor of HDL metabolism as appears from studies with transgenic animals and clinical outcomes. Biochimie 2014, 96, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Julve, J.; Escolà-Gil, J.C.; Rotllan, N.; Fiévet, C.; Vallez, E.; De La Torre, C.; Ribas, V.; Sloan, J.H.; Blanco-Vaca, F. Human apolipoprotein A-II determines plasma triglycerides by regulating lipoprotein lipase activity and high-density lipoprotein proteome. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Morillas-Ruiz, J.M.; Delgado-Alarcon, J.M.; Rubio-Perez, J.M.; Albaladejo Oton, M.D. The type of fat ingested at breakfast influences the plasma lipid profile of postmenopausal women. Biomed Res. Int. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
| Baseline Characteristics (n = 53) | |
|---|---|
| Age (years) | 63.5 ± 18.4 [31.2–85.4] |
| Weight (kg) | 64.6 ± 8.9 [52.8–78.5] |
| Body Mass Index | 27.79 ± 3.97 [21.75–34.09] |
| SBP (mmHg) | 13.0 ± 2.2 [10.2–17.1] |
| DBP (mmHg) | 7.3 ± 1.0 [6.0–9.0] |
| Personal History of CVD (n,%) | 9, 17% |
| Family History of CVD (n,%) | 17, 32% |
| Diabetes History (n,%) | 7, 13% |
| VLDL (mg/dL) | 24.7 ± 9.2 [22.0–27.4] |
| IDL (mg/dL) | 42.2 ± 8.4 [39.8–44.6] |
| LDL (mg/dL) | 65.0 ± 12.7 [61.3–68.7] |
| HDL (mg/dL) | 62.9 ± 15.4 [58.4–67.4] |
| Apo AI lipoprotein (mg/dL) | 174.4 ± 21.1 [168.2–180.6] |
| Apo AII lipoprotein (mg/dL) | 35.2 ± 5.3 [33.6–36.7] |
| Apo B lipoprotein (mg/dL) | 87.4 ± 16.1 [82.6–92.1] |
| Apo E lipoprotein (mg/dL) | 4.4 ± 0.9 [4.1–4.6] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delgado-Alarcón, J.M.; Hernández Morante, J.J.; Aviles, F.V.; Albaladejo-Otón, M.D.; Morillas-Ruíz, J.M. Effect of the Fat Eaten at Breakfast on Lipid Metabolism: A Crossover Trial in Women with Cardiovascular Risk. Nutrients 2020, 12, 1695. https://doi.org/10.3390/nu12061695
Delgado-Alarcón JM, Hernández Morante JJ, Aviles FV, Albaladejo-Otón MD, Morillas-Ruíz JM. Effect of the Fat Eaten at Breakfast on Lipid Metabolism: A Crossover Trial in Women with Cardiovascular Risk. Nutrients. 2020; 12(6):1695. https://doi.org/10.3390/nu12061695
Chicago/Turabian StyleDelgado-Alarcón, Jessica M., Juan José Hernández Morante, Francisco V. Aviles, María D. Albaladejo-Otón, and Juana M. Morillas-Ruíz. 2020. "Effect of the Fat Eaten at Breakfast on Lipid Metabolism: A Crossover Trial in Women with Cardiovascular Risk" Nutrients 12, no. 6: 1695. https://doi.org/10.3390/nu12061695
APA StyleDelgado-Alarcón, J. M., Hernández Morante, J. J., Aviles, F. V., Albaladejo-Otón, M. D., & Morillas-Ruíz, J. M. (2020). Effect of the Fat Eaten at Breakfast on Lipid Metabolism: A Crossover Trial in Women with Cardiovascular Risk. Nutrients, 12(6), 1695. https://doi.org/10.3390/nu12061695






