Dietary Diversity and Prostate Cancer in a Spanish Adult Population: CAPLIFE Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Setting
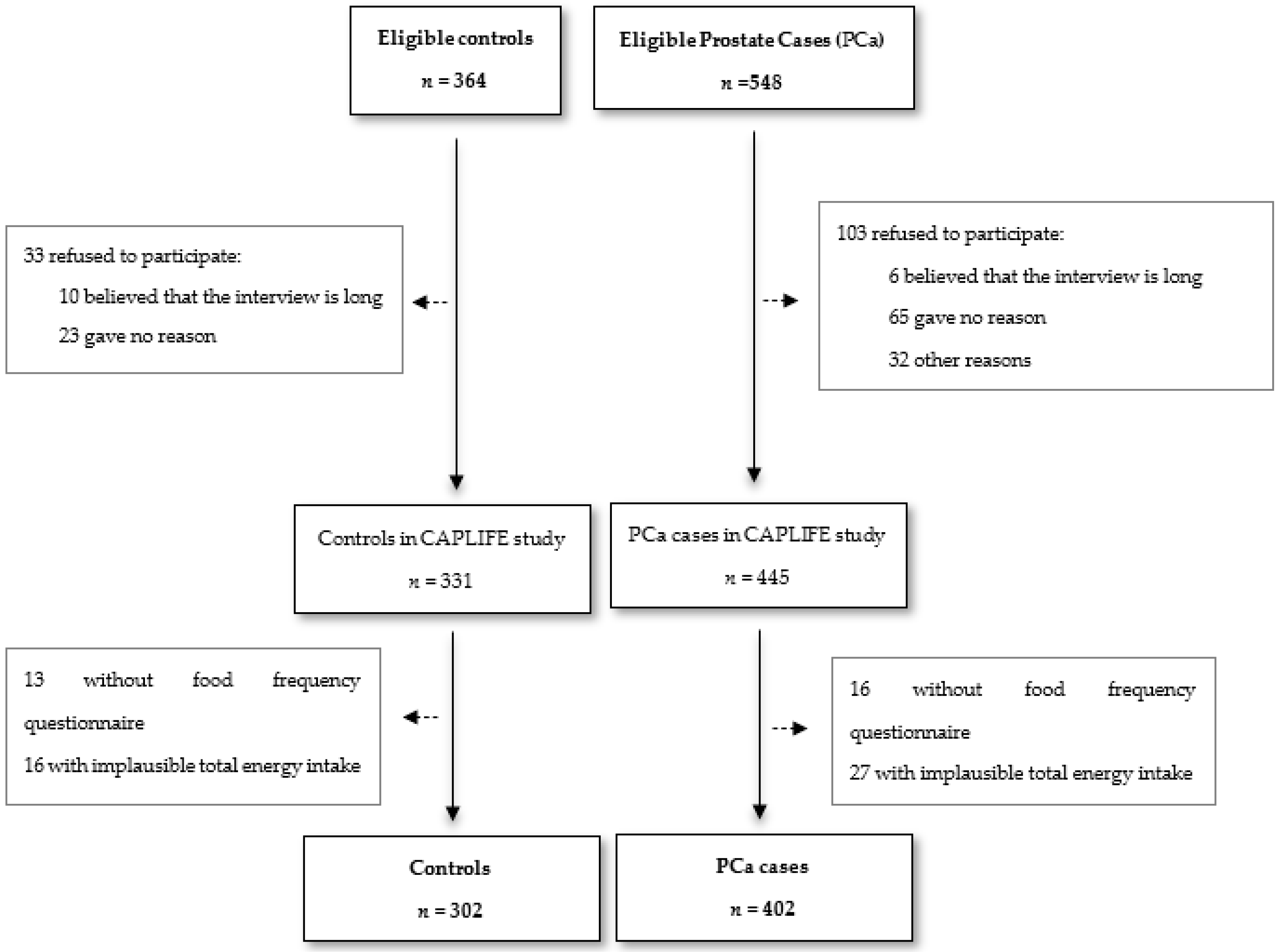
2.2. Participants
2.3. Data Sources and Variables
2.3.1. Dietary Assessment
2.3.2. Dietary Diversity Score Construction
2.3.3. Nutrient Adequate Intake
2.4. Measurement of the Tumor Aggressiveness
2.5. Statistical Analysis
3. Results
3.1. Characteristics of CAPLIFE Study Participants
3.2. DDS and Variety in Each Food Group and Risk of PCa
3.3. Prevalence of Participants with Adequate, Deficient, or Excessive Nutrient Intake
3.4. Relationship between Nutrient Adequate Intake and PCa, According to Tumor Aggressiveness
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rawla, P. Epidemiology of Prostate Cancer. World J. Oncol. 2019, 10, 63–89. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.K.; Check, D.P.; Lortet-Tieulent, J.; Laversanne, M.; Jemal, A.; Ferlay, J.; Bray, F.; Cook, M.B.; Devesa, S.S. Prostate cancer incidence in 43 populations worldwide: An analysis of time trends overall and by age group. Int. J. Cancer 2016, 138, 1388–1400. [Google Scholar] [CrossRef] [PubMed]
- Tangirala, K.; Appukkuttan, S.; Simmons, S. Costs and healthcare resource utilization associated with hospital admissions of patients with metastatic or nonmetastatic prostate cancer. Am. Health Drug Benefits 2019, 12, 306–312. [Google Scholar]
- Thompson, I.M.; Ankerst, D.; Chi, C.; Goodman, P.J.; Tangen, C.M.; Lucia, M.S.; Feng, Z.; Parnes, H.L.; Coltman, C.A., Jr. Assessing prostate cancer risk: Results from the Prostate Cancer Prevention Trial. J. Natl. Cancer Inst. 2006, 98, 529–534. [Google Scholar] [CrossRef]
- Nelson, W.; De Marzo, A.M.; Isaacs, W.B. Prostate cancer. N. Engl. J. Med. 2003, 349, 366–381. [Google Scholar] [CrossRef]
- Arthur, R.S.; Kirsh, V.; Rohan, T.E. Association between dietary energy density and risk of breast, endometrial, ovarian, and colorectal cancer among Canadian women. Cancer Epidemiol. Biomark. Prev. 2018, 27, 338–341. [Google Scholar] [CrossRef]
- Mohsenpour, M.A.; Fallah-Moshkani, R.; Ghiasvand, R.; Khosravi-Boroujeni, H.; Ahmadi, S.M.; Brauer, P.; Salehi-Abargouei, A. Adherence to Dietary Approaches to Stop Hypertension (DASH)-Style Diet and the Risk of Cancer: A Systematic Review and Meta-Analysis of Cohort Studies. J. Am. Coll. Nutr. 2019, 38, 513–525. [Google Scholar] [CrossRef]
- Lin, P.-H.; Aronson, W.; Freedland, S.J. An update of research evidence on nutrition and prostate cancer. Urol. Oncol. Semin. Orig. Investig. 2019, 37, 387–401. [Google Scholar] [CrossRef]
- World Cancer Research Fund/American Institute for Cancer Research. Diet, Nutrition, Physical Activity and Cancer: A Global Perspective. Continuous Update Project Expert Report. 2018. Available online: http://dietandcancerreport.org (accessed on 11 October 2019).
- Lane, J.A.; Oliver, S.E.; Appleby, P.N.; Lentjes, M.; Emmett, P.; Kuh, D.; Stephen, A.; Brunner, E.J.; Shipley, M.J.; Hamdy, F.C.; et al. Prostate cancer risk related to foods, food groups, macronutrients and micronutrients derived from the UK Dietary Cohort Consortium food diaries. Eur. J. Clin. Nutr. 2017, 71, 274–283. [Google Scholar] [CrossRef]
- Willis, M.S.; Wians, F.H. The role of nutrition in preventing prostate cancer: A review of the proposed mechanism of action of various dietary substances. Clin. Chim. Acta 2003, 330, 57–83. [Google Scholar] [CrossRef]
- Huang, H.-Y.; Alberg, A.J.; Norkus, E.P.; Hoffman, S.C.; Comstock, G.W.; Helzlsouer, K.J. Prospective study of antioxidant micronutrients in the blood and the risk of developing prostate cancer. Am. J. Epidemiol. 2003, 157, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Kristal, A.R.; Arnold, K.B.; Neuhouser, M.L.; Goodman, P.; Platz, E.A.; Albanes, D.; Thompson, I.M. Diet, supplement use, and prostate cancer risk: Results from the prostate cancer prevention trial. Am. J. Epidemiol. 2010, 172, 566–577. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.P.; Dwivedi, S.; Dhakad, U.; Murthy, R.C.; Choubey, V.K.; Goel, T.C.; Sankhwar, S.N. Status and Interrelationship of Zinc, Copper, Iron, Calcium and Selenium in Prostate Cancer. Indian J. Clin. Biochem. 2016, 31, 50–56. [Google Scholar] [CrossRef]
- Chan, J.M.; Gann, P.H.; Giovannucci, E.L. Role of diet in prostate cancer development and progression. J. Clin. Oncol. 2005, 23, 8152–8160. [Google Scholar] [CrossRef]
- Turati, F.; Rossi, M.; Pelucchi, C.; Levi, F.; La Vecchia, C. Fruit and vegetables and cancer risk: A review of southern European studies. Br. J. Nutr. 2015, 113, S102–S110. [Google Scholar] [CrossRef]
- Lin, P.-H.; Aronson, W.J.; Freedland, S.J. Nutrition, dietary interventions and prostate cancer: The latest evidence. BMC Med. 2015, 13, 3. [Google Scholar] [CrossRef]
- Umesawa, M.; Iso, H.; Mikami, K.; Kubo, T.; Suzuki, K.; Watanabe, Y.; Mori, M.; Miki, T.; Tamakoshi, A. Relationship between vegetable and carotene intake and risk of prostate cancer: The JACC study. Br. J. Cancer 2014, 110, 792–796. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Galbete, C.; Hoffmann, G. Adherence to mediterranean diet and risk of cancer: An updated systematic review and meta-analysis. Nutrients 2017, 9, 1063. [Google Scholar] [CrossRef]
- Kenfield, S.A.; Dupre, N.C.; Richman, E.L.; Stampfer, M.J.; Chan, J.M.; Giovannucci, E.L. Mediterranean diet and prostate cancer risk and mortality in the health professionals follow-up study. Eur. Urol. 2014, 65, 887–894. [Google Scholar] [CrossRef]
- National Research Council. Diet and health: Implications for reducing chronic disease risk: Abstract of executive summary. Nutr. Today 1989, 24, 36–38. [Google Scholar]
- Kant, A.K.; Thompson, F.E. Measures of overall diet quality from a food frequency questionnaire: National Health interview survey, 1992. Nutr. Res. 1997, 17, 1443–1456. [Google Scholar] [CrossRef]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Aranceta Bartrina, J.; Arija Val, V.V.; Maíz Aldalur, E.; Martínez de Victoria Muñoz, E.; Ortega Anta, R.M.; Pérez-Rodrigo, C.; Quiles Izquierdo, J.; Rodríguez Martín, A.; Román Viñas, B.; Salvador Castell, G.; et al. Dietary Guidelines for the Spanish population (SENC, diciembre 2016); the new graphic icon of healthy food. Nutr. Hosp. 2016, 33, 1–48. [Google Scholar] [CrossRef] [PubMed]
- Cano-Ibáñez, N.; Gea, A.; Martínez-González, M.; Salas-Salvadó, J.; Corella, D.; Zomeño, M.D.; Romaguera, D.; Vioque, J.; Aros, F.; Wärnberg, J.; et al. Dietary diversity and nutritional adequacy among an older Spanish population with metabolic syndrome in the PREDIMED-plus study: A cross-sectional analysis. Nutrients 2019, 11, 958. [Google Scholar] [CrossRef]
- World Health Organization. International Classification of Diseases 11th Revision. 2016. Available online: https://icd.who.int/browse10/2016/en (accessed on 14 February 2020).
- Roman-Viñas, B.; Serra-Majem, L.; Hagströmer, M.; Ribas-Barba, L.; Sjöström, M.; Segura-Cardona, R. International physical activity questionnaire: Reliability and validity in a Spanish population. Eur. J. Sport Sci. 2010, 10, 297–304. [Google Scholar] [CrossRef]
- Martin-Moreno, J.M.; Boyle, P.; Gorgojo, L.; Maisonneuve, P.; Fernandez-Rodriguez, J.C.; Salvini, S.; Willett, W.C. Development and validation of a food frequency questionnaire in Spain. Int. J. Epidemiol. 1993, 22, 512–519. [Google Scholar] [CrossRef]
- Fernandez-Ballart, J.D.; Piñol, J.L.; Zazpe, I.; Corella, D.; Carrasco, P.; Toledo, E.; Perez-Bauer, M.; A Martínez-González, M.; Salas-Salvadó, J.; Martin-Moreno, J.M. Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br. J. Nutr. 2010, 103, 1808–1816. [Google Scholar] [CrossRef]
- Moreiras, O.C.A.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos (Spanish Food Composition Tables), 7th ed.; Pirámide: Madrid, Spain, 2003. [Google Scholar]
- Mataix-Verdú, J.; García, L.; Mañas Almendros, M.; Martínez-Victoria, E.; Llopis González, J. Tabla de Composición de Alimentos Españoles. (Spanish Food Composition Tables), 4th ed.; Universidad de Granada: Granada, Spain, 2003. [Google Scholar]
- Willet, W. Nutritional Epidemiology, 3rd ed.; Oxford University Press: New York, NY, USA, 2013. [Google Scholar]
- Farhangi, M.A.; Jahangiry, L. Dietary diversity score is associated with cardiovascular risk factors and serum adiponectin concentrations in patients with metabolic syndrome. BMC Cardiovasc. Disord. 2018, 18, 68. [Google Scholar] [CrossRef]
- Cano-Ibáñez, N.; Martínez-Galiano, J.M.; Amezcua-Prieto, C.; Olmedo-Requena, R.; Bueno-Cavanillas, A.; Delgado-Rodríguez, M. Maternal dietary diversity and risk of small for gestational age newborn: Findings from a case—Control study. Clin. Nutr. 2019, 39, 1943–1950. [Google Scholar] [CrossRef]
- Moreiras, O.; Carbajal, A.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos/Guía de Prácticas, 16th ed.; Ediciones Pirámide Madrid: Madrid, Spain, 2013. [Google Scholar]
- Kant, A.K. Consumption of energy-dense, nutrient-poor foods by adult Americans: Nutritional and health implications. The third National Health and Nutrition Examination Survey, 1988–1994. Am. J. Clin. Nutr. 2000, 72, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, E. Dietary diversity, diet quality, and body weight regulation. Nutr. Rev. 2004, 62, S78–S81. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Dietary Reference Values and Dietary Guidelines. 2018. Available online: https://www.efsa.europa.eu/en/topics/topic/dietary-reference-values (accessed on 1 October 2018).
- Aranceta-Bartrina, J.; Serra-Majem, L.; Pérez-Rodrigo, C.; Llopis, J.; Mataix, J.; Ribas, L.; Tojo, R.; Tur, J.A. Vitamins in Spanish food patterns: The eVe study. Public Health Nutr. 2001, 4, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.I.; Egevad, L.; Amin, M.B.; Delahunt, B.; Srigley, J.R.; Humphrey, P.A. The 2014 international society of urological pathology (ISUP) consensus conference on gleason grading of prostatic carcinoma definition of grading patterns and proposal for a new grading system. Am. J. Surg. Pathol. 2016, 40, 1. [Google Scholar] [CrossRef]
- Neuzillet, Y.; Raynaud, J.-P.; Dreyfus, J.-F.; Radulescu, C.; Rouanne, M.; Schneider, M.; Krish, S.; Rouprêt, M.; Drouin, S.J.; Comperat, E.; et al. Aggressiveness of Localized Prostate Cancer: The Key Value of Testosterone Deficiency Evaluated by Both Total and Bioavailable Testosterone: AndroCan Study Results. Horm. Cancer 2019, 10, 36–44. [Google Scholar] [CrossRef]
- Messina, M. Legumes and soybeans: Overview of their nutritional profiles and health effects. Am. J. Clin. Nutr. 1999, 70 (Suppl. 3), 439S–450S. [Google Scholar] [CrossRef]
- Kolonel, L.N.; Hankin, J.H.; Whittemore, A.S.; Wu, A.H.; Gallagher, R.P.; Wilkens, L.R.; John, E.M.; Howe, G.R.; Dreon, D.M.; West, D.W.; et al. Vegetables, fruits, legumes and prostate cancer: A multiethnic case-control study. Cancer Epidemiol. Biomark. Prev. 2000, 9, 795–804. [Google Scholar]
- Giovannucci, E.; Rimm, E.B.; Liu, Y.; Stampfer, M.J.; Willett, W.C. A prospective study of tomato products, lycopene, and prostate cancer risk. J. Natl. Cancer Inst. 2002, 94, 391–398. [Google Scholar] [CrossRef]
- Giovannucci, E.; Rimm, E.B.; Colditz, G.A.; Stampfer, M.J.; Ascherio, A.; Chute, C.C.; Willett, W.C. A prospective study of dietary fat and risk of prostate cancer. J. Natl. Cancer Inst. 1993, 85, 1571–1579. [Google Scholar] [CrossRef]
- Tzonou, A.; Signorello, L.; Lagiou, P.; Wuu, J.; Trichopoulos, D.; Trichopoulou, A. Diet and cancer of the prostate: A case-control study in Greece. International. Int. J. Cancer 1999, 80, 704–708. [Google Scholar] [CrossRef]
- Cano-Ibáñez, N.; Gea, A.; Ruiz-Canela, M.; Corella, D.; Salas-Salvadó, J.; Schröder, H.; Navarrete-Muñoz, E.M.; Romaguera, D.; Martínez, J.A.; Barón-López, F.J.; et al. Diet quality and nutrient density in subjects with metabolic syndrome: Influence of socioeconomic status and lifestyle factors. A cross-sectional assessment in the PREDIMED-Plus study. Clin. Nutr. 2019, 39, 1161–1173. [Google Scholar] [CrossRef] [PubMed]
- Schröder, H.; for the REGICOR Investigators; Marrugat, J.; Covas, M.; Elosua, R.; Peña, A.; Weinbrenner, T.; Fitó, M.; Vidal, M.A.; Masia, R. Population dietary habits and physical activity modification with age. Eur. J. Clin. Nutr. 2004, 58, 302–311. [Google Scholar] [CrossRef]
- De La Fuente, C.; Ruiz, Z.V.; Bes-Rastrollo, M.; Sampson, L.; A Martínez-González, M. Reproducibility of an FFQ validated in Spain. Public Health Nutr. 2010, 13, 1364–1372. [Google Scholar] [CrossRef] [PubMed]
- Steck, S.E.; Murphy, E.A. Dietary patterns and cancer risk. Nat. Rev. Cancer 2020, 20, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Mondul, A.M.; Weinstein, S.J.; Albanes, D. Vitamins, metabolomics, and prostate cancer. World J. Urol. 2017, 35, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Sonkusre, P. Specificity of Biogenic Selenium Nanoparticles for Prostate Cancer Therapy with Reduced Risk of Toxicity: An in vitro and in vivo Study. Front. Oncol. 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Karunasinghe, N.; Ng, L.; Wang, A.; Vaidyanathan, V.; Zhu, S.; Ferguson, L.R. Selenium supplementation and prostate health in a New Zealand cohort. Nutrients 2020, 12, 2. [Google Scholar] [CrossRef]
- Duffield-Lillico, A.J.; Dalkin, B.L.; Reid, M.E.; Turnbull, B.W.; Slate, E.H.; Jacobs, E.T.; Marshall, J.R.; Clark, L.C. Nutritional Prevention of Cancer Study Group. Selenium supplementation, baseline plasma selenium status and incidence of prostate cancer: an analysis of the complete treatment period of the Nutritional Prevention of Cancer Trial. BJU. Int. 2003, 91, 608–612. [Google Scholar] [CrossRef]
- Lim, J.T.; Tan, Y.Q.; Valeri, L.; Lee, J.; Geok, P.P.; Chia, S.E.; Ong, C.N.; Seow, W.J. Association between serum heavy metals and prostate cancer risk—A multiple metal analysis. Environ. Int. 2019, 132, 105109. [Google Scholar] [CrossRef]
- Ramamoorthy, V.I.; Rubens, M.; Saxena, A.; Shehadeh, N. Selenium and vitamin E for prostate cancer—Justifications for the SELECT study. Asian Pac. J. Cancer Prev. 2015, 16, 2619–2627. [Google Scholar] [CrossRef]
- Ioannidis, J.P. Why most published research findings are false. PLoS Med. 2005, 2, e124. [Google Scholar] [CrossRef] [PubMed]
| Food Groups | Food Subgroups | Recommended Servings |
|---|---|---|
| Vegetables | (1) Green vegetables: spinach, cruciferous, lettuce, green beans, eggplant, peppers, asparagus (2) Tomatoes (3) Yellow vegetables: carrots, pumpkin (4) Mushrooms | 2 servings/day |
| Fruits | (1) Citrus fruits: orange (2) Tropical Fruits: banana, kiwi, grapes (3) Other seasonal fruits: Apple, peach, strawberries, watermelon, melon | 3 servings/day |
| Dairy Products | (1) Milk: low fat and high fat (2) Yogurt: low fat and high fat (3) Cheese: low fat and high fat | 2 servings/day |
| Cereals | (1) Potatoes (2) Grain: bread, pasta, rice, and whole breakfast cereals | 4 servings/day |
| Protein food groups | (1) Legumes: peas, beans, lentils, and chickpeas (2) White meats: poultry and rabbit (3) Fish: oily fish, white fish and other shellfish/seafood (4) Eggs (5) Nuts: almonds, pistachios and walnuts | 3 servings/week |
| Controls | PCa Cases | p Value | |||
|---|---|---|---|---|---|
| n = 302 | n = 402 | ||||
| Age (years), mean, (SD) | 65.3 | (8.2) | 67.8 | (7.4) | <0.001 |
| BMI, mean, (SD) | 28.7 | (4.2) | 28.5 | (4.1) | 0.533 |
| BMI, n, (%) | |||||
| Normal weight (<25 kg/m2) | 48 | (15.9) | 76 | (18.9) | 0.569 |
| Overweight (25–29.9 kg/m2) | 162 | (53.6) | 205 | (51.0) | |
| Obesity (≥30 kg/m2) | 92 | (30.5) | 121 | (30.1) | |
| Marital status, n, (%) | |||||
| Married | 254 | (84.1) | 338 | (84.1) | 0.938 |
| Singled, never married | 17 | (5.6) | 18 | (4.5) | |
| Separated/divorced | 20 | (6.6) | 27 | (6.7) | |
| Widowed | 11 | (3.6) | 19 | (4.7) | |
| Education, n, (%) | |||||
| Primary | 92 | (30.5) | 122 | (30.4) | 0.368 |
| Secondary | 147 | (48.6) | 212 | (52.7) | |
| University | 63 | (20.9) | 68 | (16.9) | |
| Physical Activity (PA), n, (%) | |||||
| Low PA | 113 | (37.7) | 157 | (39.2) | 0.350 |
| Moderate PA | 143 | (47.7) | 199 | (49.8) | |
| High PA | 46 | (14.6) | 46 | (11.0) | |
| Smoking status, n, (%) | |||||
| Never | 88 | (29.1) | 107 | (26.6) | 0.745 |
| Former | 157 | (52.0) | 214 | (53.2) | |
| Current | 57 | (18.9) | 81 | (20.2) | |
| Alcohol consumption (g), mean, (SD) | 11.6 | (0.9) | 14.1 | (0.9) | 0.046 |
| Aggressiveness, n, (%) | |||||
| Low aggressiveness (ISUP 1–2) | - | - | 309 | (76.9) | - |
| High aggressiveness (ISUP 3–5) | - | - | 93 | (23.1) | - |
| First-degree family history of PCa, yes ± n, (%) | 15 | (5.0) | 25 | (6.2) | 0.478 |
| Controls n = 302 | PCa Cases n = 402 | Low Aggressiveness n = 308 | High Aggressiveness n = 93 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | (SD) | Mean | (SD) | p Value 1 | Mean | (SD) | p Value 1 | Mean | (SD) | p Value 1 | |
| Total DDS 2 | 5.5 | (0.8) | 5.6 | (0.6) | 0.217 | 5.6 | (1.3) | 0.399 | 5.8 | (1.3) | 0.165 |
| Vegetables group 3 | 1.4 | (0.6) | 1.5 | (0.6) | 0.291 | 1.5 | (0.6) | 0.141 | 1.4 | (0.7) | 0.769 |
| Fruits group 3 | 1.0 | (0.6) | 1.0 | (0.6) | 0.349 | 1.0 | (0.6) | 0.271 | 1.0 | (0.6) | 0.880 |
| Cereals group 3 | 0.6 | (0.4) | 0.6 | (0.3) | 0.569 | 0.6 | (0.4) | 0.792 | 0.6 | (0.3) | 0.403 |
| Dairy group 3 | 1.0 | (0.6) | 1.0 | (0.6) | 0.283 | 1.0 | (0.6) | 0.568 | 1.1 | (0.6) | 0.099 |
| Protein group 3 | 1.6 | (0.4) | 1.6 | (0.4) | 0.396 | 1.6 | (0.4) | 0.662 | 1.7 | (0.4) | 0.282 |
| PCa Cases | |||||
|---|---|---|---|---|---|
| cOR | 95% CI | aOR a | 95% CI | p for Trend | |
| Total DDS | |||||
| Q1 b (≥0.9, <4.7 points) | 1 (ref.) | 1 (ref.) | 0.257 | ||
| Q2 (≥4.7, <5.7 points) | 1.07 | (0.69, 1.66) | 1.11 | (0.69, 1.77) | |
| Q3 (≥5.7, <6.6 points) | 1.61 | (1.06, 2.46) | 1.65 | (1.05, 2.60) | |
| Q4 (≥6.6, <8.5 points) | 1.18 | (0.77, 1.83) | 1.16 | (0.72, 1.88) | |
| Vegetable group | |||||
| C1 c (0 point) | 1 (ref.) | 1 (ref.) | 0.701 | ||
| C2 (>0, <0.5 points) | 1.29 | (0.50, 1.83) | 0.96 | (0.49, 1.88) | |
| C3 (≥0.5, <1 point) | 0.96 | (0.70, 2.00) | 0.97 | (0.49, 1.89) | |
| C4 (≥1 point) | 1.18 | (0.71, 2.07) | 1.08 | (0.63, 1.87) | |
| Fruit group | |||||
| C1 (0 point) | 1 (ref.) | 1 (ref.) | 0.159 | ||
| C2 (>0, <1 point) | 1.01 | (0.62, 1.65) | 0.90 | (0.54, 1.50) | |
| C3 (≥1, <1.5 points) | 0.64 | (0.36, 1.11) | 0.58 | (0.32, 1.05) | |
| C4 (≥1.5 points) | 0.91 | (0.49, 1.71) | 0.81 | (0.42, 1.59) | |
| Cereal group | |||||
| C1 (0 point) | 1 (ref.) | 1 (ref.) | 0.364 | ||
| C2 (>0, <0.5 points) | 1.38 | (0.86, 2.21) | 1.42 | (0.88, 2.32) | |
| C3 (≥0.5, <1 point) | 1.15 | (0.67, 1.97) | 1.18 | (0.68, 2.06) | |
| C4 (≥1 point) | 2.20 | (0.86, 5.61) | 2.31 | (0.88, 6.03) | |
| Protein group | |||||
| C1 (0 point) | 1 (ref.) | 1 (ref.) | 0.462 | ||
| C2 (>0, <1 point) | 1.26 | (0.75, 2.10) | 0.79 | (0.44, 1.41) | |
| C3 (≥1, <1.5 points) | 0.81 | (0.46, 1.41) | 0.82 | (0.61, 1.39) | |
| C4 (≥1.5 points) | 0.89 | (0.60, 1.33) | 0.92 | (0.57, 1.24) | |
| Dairy group d | |||||
| C1 (0 point) | 1 (ref.) | 1 (ref.) | 0.336 | ||
| C2 (>0, <1 point) | 0.95 | (0.59, 1.52) | 0.93 | (0.56, 1.52) | |
| C3 (≥1, <1.5 points) | 1.37 | (0.84, 2.24) | 1.36 | (0.81, 2.27) | |
| C4 (≥1.5 points) | 1.05 | (0.59, 1.88) | 1.00 | (0.55, 1.85) | |
| Controls | PCa Cases | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| n = 302 | n = 402 | ||||||||||
| Low Aggressiveness (n = 308) | High Aggressiveness (n = 93) | ||||||||||
| DI a | AI b | EI c | DI a | AI b | EI c | p Value d | DIa | AIb | EIc | p Value d | |
| Dietary Fiber (g/day) | 7.0 | 93.0 | - | 7.5 | 92.5 | - | 0.806 | 8.6 | 91.4 | - | 0.594 |
| Vitamin A (µg/day) | 2.0 | 58.6 | 39.7 | 1.0 | 55.2 | 43.8 | 0.481 | 1.1 | 55.9 | 43.0 | 0.804 |
| Vitamin B6 (mg/day) | 0 | 100.0 | 0 | 0.6 | 99.4 | 0 | 0.170 | 0 | 100.0 | 0 | - |
| Vitamin B9 (µg/day) | 2.0 | 92.4 | 6.0 | 1.0 | 92.9 | 6.2 | 0.758 | 0 | 92.5 | 7.5 | 0.403 |
| Vitamin B12 (µg/day) | 0.3 | 99.7 | 0 | 0.3 | 99.7 | 0 | 0.964 | 1.1 | 99.0 | 0 | 0.386 |
| Vitamin C (mg/day) | 1.0 | 99.0 | - | 0.6 | 99.4 | - | 0.602 | 2.1 | 97.9 | - | 0.397 |
| Vitamin D (µg/day) | 66.6 | 33.4 | 0 | 70.8 | 29.2 | 0 | 0.261 | 57.0 | 43.0 | 0 | 0.092 |
| Vitamin E (mg/day) | 24.2 | 75.8 | 0 | 19.8 | 80.2 | 0 | 0.193 | 22.6 | 77.4 | 0 | 0.753 |
| Calcium (mg/day) | 4.0 | 85.1 | 10.9 | 3.3 | 86.7 | 10.1 | 0.829 | 2.2 | 85.0 | 12.9 | 0.634 |
| Magnesium (mg/day) | 0 | 0 | 100.0 | 0 | 0 | 100.0 | - | 1.1 | 0 | 98.9 | 0.071 |
| Iodine (µg/day) | 7.8 | 73.8 | 18.9 | 4.2 | 76.6 | 19.2 | 0.265 | 5.4 | 73.1 | 21.5 | 0.727 |
| Potassium (mg/day) | 0.7 | 99.3 | 0 | 1.0 | 99.0 | 0 | 0.669 | 1.1 | 98.9 | 0 | 0.688 |
| Selenium (µg/day) | 4.0 | 93.7 | 2.3 | 2.0 | 97.8 | 0.3 | 0.030 | 1.1 | 98.9 | 0 | 0.124 |
| Nutrient | PCa Cases | ||||
|---|---|---|---|---|---|
| cOR | (95% CI) | aOR a | (95% CI) | p for Trend | |
| Dietary Fiber | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake b | 1.16 | (0.65, 2.05) | 1.34 | (0.73, 2.44) | 0.343 |
| Vitamin A | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 1.14 | (0.84, 1.54) | 1.07 | (0.78, 1.47) | 0.656 |
| Vitamin B9 | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 0.94 | (0.53, 1.67) | 0.96 | (0.53, 1.73) | 0.886 |
| Vitamin B12 | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 1.51 | (0.14, 16.7) | 1.68 | (0.15, 19.4) | 0.676 |
| Vitamin C | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 1.00 | (0.22, 4.51) | 0.91 | (0.20, 4.19) | 0.900 |
| Vitamin D | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 1.04 | (0.76, 1.43) | 1.11 | (0.80, 1.55) | 0.518 |
| Vitamin E | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 0.82 | (0.57, 1.17) | 0.86 | (0.59, 1.25) | 0.420 |
| Calcium | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 0.91 | (0.59, 1.39) | 0.95 | (0.61, 1.48) | 0.818 |
| Iodine | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 0.90 | (0.64, 1.27) | 0.91 | (0.64, 1.30) | 0.611 |
| Potassium | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 0.66 | (0.12, 3.65) | 0.67 | (0.12, 3.84) | 0.898 |
| Selenium | |||||
| Adequate intake | 1 (ref.) | 1 (ref.) | |||
| Inadequate intake | 0.30 | (0.13, 0.70) | 0.33 | (0.14, 0.79) | 0.013 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cano-Ibáñez, N.; Barrios-Rodríguez, R.; Lozano-Lorca, M.; Vázquez-Alonso, F.; Arrabal-Martín, M.; Triviño-Juárez, J.M.; Salcedo-Bellido, I.; Jiménez-Moleón, J.J.; Olmedo-Requena, R. Dietary Diversity and Prostate Cancer in a Spanish Adult Population: CAPLIFE Study. Nutrients 2020, 12, 1694. https://doi.org/10.3390/nu12061694
Cano-Ibáñez N, Barrios-Rodríguez R, Lozano-Lorca M, Vázquez-Alonso F, Arrabal-Martín M, Triviño-Juárez JM, Salcedo-Bellido I, Jiménez-Moleón JJ, Olmedo-Requena R. Dietary Diversity and Prostate Cancer in a Spanish Adult Population: CAPLIFE Study. Nutrients. 2020; 12(6):1694. https://doi.org/10.3390/nu12061694
Chicago/Turabian StyleCano-Ibáñez, Naomi, Rocío Barrios-Rodríguez, Macarena Lozano-Lorca, Fernando Vázquez-Alonso, Miguel Arrabal-Martín, José Matías Triviño-Juárez, Inmaculada Salcedo-Bellido, José Juan Jiménez-Moleón, and Rocío Olmedo-Requena. 2020. "Dietary Diversity and Prostate Cancer in a Spanish Adult Population: CAPLIFE Study" Nutrients 12, no. 6: 1694. https://doi.org/10.3390/nu12061694
APA StyleCano-Ibáñez, N., Barrios-Rodríguez, R., Lozano-Lorca, M., Vázquez-Alonso, F., Arrabal-Martín, M., Triviño-Juárez, J. M., Salcedo-Bellido, I., Jiménez-Moleón, J. J., & Olmedo-Requena, R. (2020). Dietary Diversity and Prostate Cancer in a Spanish Adult Population: CAPLIFE Study. Nutrients, 12(6), 1694. https://doi.org/10.3390/nu12061694







