Lactobacillus rhamnosus GG (ATCC 53103) for the Management of Infantile Colic: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Design
2.3. Sample Collection
2.3.1. DNA Extraction
2.3.2. Faecal Calprotectin Analysis
2.3.3. Real-Time PCR
2.4. Statistical Analysis
3. Results
3.1. Clinical Data: Crying and Fussing
3.2. Fecal Calprotectin
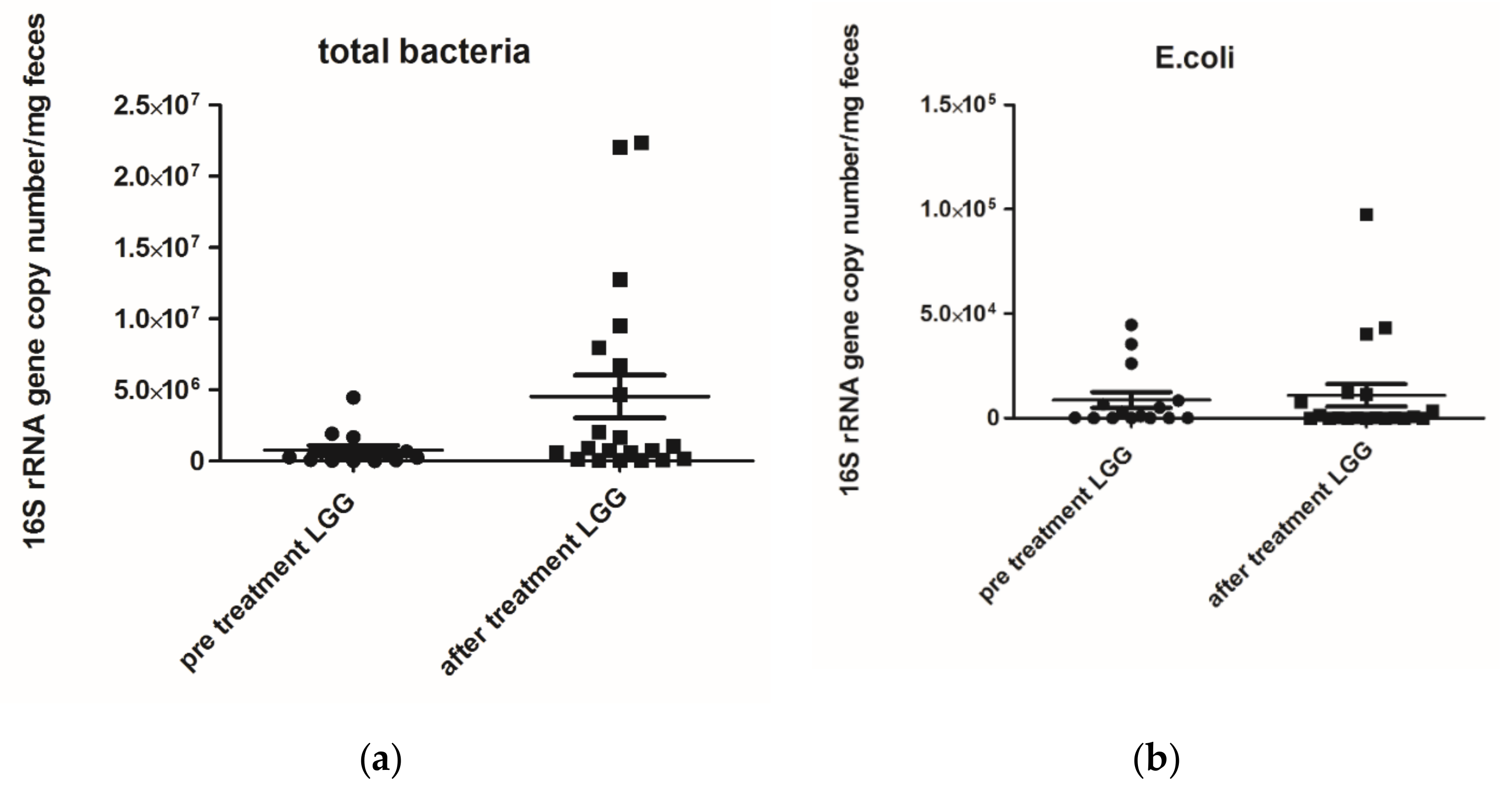
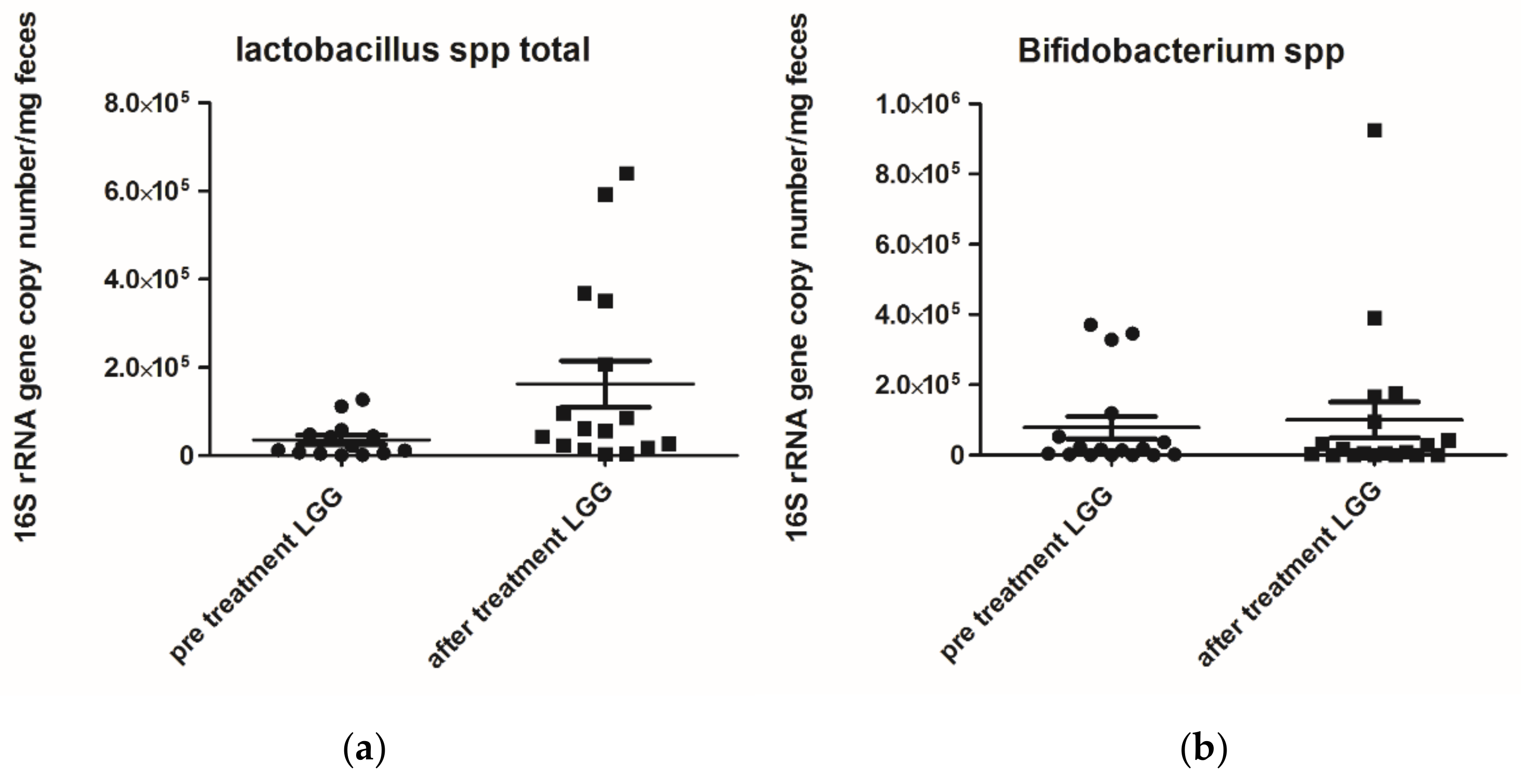
3.3. Microbiological Analysis
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| L. rhamnosus | Lactobacillus rhamnosus ATCC 53103 |
| SD | Standard deviation |
| IC | Infantile colic |
| IQR | Interquartile range |
| PCR | Polymerase Chain Reaction |
| g | grams |
| m | minutes |
| cfu | Colony-forming unit |
| TaqMan RT-PCR | TaqMan Real-Time reverse-transcription |
References
- Biedermann, L.; Rogler, G. The intestinal microbiota: Its role in health and disease. Eur. J. Pediatr. 2015, 174, 151–167. [Google Scholar] [CrossRef]
- Rhoads, J.M.; Collins, J.; Fatheree, N.Y.; Hashmi, S.S.; Taylor, C.M.; Luo, M.; Hoang, T.K.; Gleason, W.A.; Van Arsdall, M.R.; Navarro, F.; et al. Infant Colic Represents Gut Inflammation and Dysbiosis. J. Pediatr. 2018, 203, 55–61.e3. [Google Scholar] [CrossRef]
- Camilleri, M.; Park, S.Y.; Scarpato, E.; Staiano, A. Exploring hypotheses and rationale for causes of infantile colic. Neurogastroenterol. Motil. 2017, 29. [Google Scholar] [CrossRef]
- Pärtty, A.; Kalliomäki, M.; Endo, A.; Salminen, S.; Isolauri, E. Compositional development of Bifidobacterium and Lactobacillus microbiota is linked with crying and fussing in early infancy. PLoS ONE 2012, 7, e32495. [Google Scholar] [CrossRef] [PubMed]
- de Weerth, C.; Fuentes, S.; Puylaert, P.; de Vos, W.M. Intestinal microbiota of infants with colic: Development and specific signatures. Pediatrics 2013, 131, e550–e558. [Google Scholar] [CrossRef]
- Savino, F.; Cordisco, L.; Tarasco, V.; Calabrese, R.; Palumeri, E.; Matteuzzi, D. Molecular identification of coliform bacteria from colicky breastfed infants. Acta Paediatr. 2009, 98, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Pelle, E.; Palumeri, E.; Oggero, R.; Miniero, R. Lactobacillus reuteri (American Type Culture Collection Strain 55730) versus simethicone in the treatment of infantile colic: A prospective randomized study. Pediatrics 2007, 119, e124–e130. [Google Scholar] [CrossRef] [PubMed]
- Savino, F.; Cordisco, L.; Tarasco, V.; Palumeri, E.; Calabrese, R.; Oggero, R.; Matteuzzi, D. Lactobacillus reuteri DSM 17938 in infantile colic: A randomized, double-blind, placebo-controlled trial. Pediatrics 2010, 126, e526–e533. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H.; Gyrczuk, E.; Horvath, A. Lactobacillus reuteri DSM 17938 for the management of infantile colic in breastfed infants: A randomized, double-blind, placebo-controlled trial. J. Pediatr. 2013, 162, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Chau, K.; Lau, E.; Greenberg, S.; Jacobson, S.; Yazdani-Brojeni, P.; Verma, N.; Koren, G. Probiotics for Infantile Colic: A Randomized, Double-Blind, Placebo-Controlled Trial Investigating Lactobacillus reuteri DSM 17938. J. Pediatr. 2015, 166, 74–78.e1. [Google Scholar] [CrossRef]
- Pärtty, A.; Kalliomäki, M.; Salminen, S.; Isolauri, E. Infantile Colic Is Associated With Low-grade Systemic Inflammation. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 691–695. [Google Scholar] [CrossRef] [PubMed]
- Segers, M.E.; Lebeer, S. Towards a better understanding of Lactobacillus rhamnosus GG--host interactions. Microbe Cell Fact. 2014, 13 (Suppl. 1), S7. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.A.K.; Sarker, M.; Wan, D. Immunomodulatory Effects of Probiotics on Cytokine Profiles. BioMed Res. Int. 2018, 2018, 8063647. [Google Scholar] [CrossRef] [PubMed]
- Fujimura, K.E.; Sitarik, A.R.; Havstad, S.; Lin, D.L.; Levan, S.; Fadrosh, D.; Panzer, A.R.; LaMere, B.; Rackaityte, E.; Lukacs, N.W.; et al. Neonatal gut microbiota associates with childhood multisensitized atopy and T cell differentiation. Nat. Med. 2016, 22, 1187–1191. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.J.; Roy, N.; Heine, R.G.; Hosking, C.S.; Francis, D.E.; Brown, J.; Speirs, B.; Sadowsky, J.; Carlin, J.B. Effect of a low-allergen maternal diet on colic among breastfed infants: A randomized, controlled trial. Pediatrics 2005, 116, e709–e715. [Google Scholar] [CrossRef]
- Gordon, M.; Biagioli, E.; Sorrenti, M.; Lingua, C.; Moja, L.; Banks, S.S.; Ceratto, S.; Savino, F. Dietary modifications for infantile colic. Cochrane Database Syst. Rev. 2018, 10, CD011029. [Google Scholar] [CrossRef]
- Wessel, M.A.; Cobb, J.C.; Jackson, E.B.; Harris, G.S.; Detwiler, A.C. Paroxysmal fussing in infancy, sometimes called colic. Pediatrics 1954, 14, 421–435.1. [Google Scholar]
- Zeevenhooven, J.; Koppen, I.J.; Benninga, M.A. The New Rome IV Criteria for Functional Gastrointestinal Disorders in Infants and Toddlers. Pediatr. Gastroenterol. Hepatol. Nutr. 2017, 20, 1–13. [Google Scholar] [CrossRef]
- Barr, R.G.; Kramer, M.S.; Boisjoly, C.; McVey-White, L.; Pless, I.B. Parental diary of infant cry and fuss behaviour. Arch. Dis. Child. 1988, 63, 380–387. [Google Scholar] [CrossRef]
- Savino, F.; Garro, M.; Montanari, P.; Galliano, I.; Bergallo, M. Crying Time and RORγ/FOXP3 Expression in Lactobacillus reuteri DSM17938-Treated Infants with Colic: A Randomized Trial. J. Pediatr. 2018, 192, 171–177.e1. [Google Scholar] [CrossRef]
- St James-Roberts, I.; Alvarez, M.; Hovish, K. Emergence of a developmental explanation for prolonged crying in 1- to 4-month-old infants: Review of the evidence. J. Pediatr. Gastroenterol. Nutr. 2013, 57, S30–S35. [Google Scholar] [CrossRef]
- Buyuktiryaki, B.; Celik, I.K.; Erdem, S.B.; Capanoglu, M.; Civelek, E.; Guc, B.U.; Guvenir, H.; Cakir, M.; Misirlioglu, E.D.; Akcal, O.; et al. Risk Factors Influencing Tolerance and Clinical Features of Food Protein Induced Allergic Proctocolitis. J. Pediatr. Gastroenterol. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Durack, J.; Kimes, N.E.; Lin, D.L.; Rauch, M.; McKean, M.; McCauley, K.; Panzer, A.R.; Mar, J.S.; Cabana, M.D.; Lynch, S.V. Delayed gut microbiota development in high-risk for asthma infants is temporarily modifiable by Lactobacillus supplementation. Nat. Commun. 2018, 16, 707. [Google Scholar] [CrossRef] [PubMed]
- Tan-Lim, C.S.C.; Esteban-Ipac, N.A.R. Probiotics as treatment for food allergies among pediatric patients: A meta-analysis. World Allergy Organ J. 2018, 6, 25. [Google Scholar] [CrossRef]
- Kannampalli, P.; Pochiraju, S.; Chichlowski, M.; Berg, B.M.; Rudolph, C.; Bruckert, M.; Miranda, A.; Sengupta, J.N. Probiotic Lactobacillus rhamnosus GG (LGG) and prebiotic prevent neonatalinflammation-induced visceral hypersensitivity in adult rats. Neurogastroenterol. Motil. 2014, 26, 1694–1704. [Google Scholar] [CrossRef] [PubMed]
- Nocerino, R.; Pezzella, V.; Cosenza, L.; Amoroso, A.; Di Scala, C.; Amato, F.; Iacono, G.; Canani, R.B. The controversial role of food allergy in infantile colic: Evidence and clinical management. Nutrients 2015, 7, 2015–2025. [Google Scholar] [CrossRef]
- Lothe, L.; Lindberg, T. Cow’s milk whey protein elicits symptoms of infantile colic in colicky formula-fed infants: A double-blind crossover study. Pediatrics 1989, 83, 262–266. [Google Scholar]
- Heine, R.G. Allergic gastrointestinal motility disorders in infancy and early childhood. Pediatric Allergy Immunol. 2008, 19, 383–391. [Google Scholar] [CrossRef]
- Lundelin, K.; Poussa, T.; Salminen, S.; Isolauri, E. Long-term safety and efficacy of perinatal probiotic intervention: Evidence from a follow-up study of four randomized, double-blind, placebo-controlled trials. Pediatric Allergy Immunol. 2017, 28, 170–175. [Google Scholar] [CrossRef]
- Roseth, A.G. Determination of faecal calprotectin, a novel marker of organic gastrointestinal disorders. Dig. Liver Dis. 2003, 35, 607–609. [Google Scholar] [CrossRef]
- Ashorov, O.; Hamouda, D.; Dickman, R.; Perets, T.T. Clinical Accuracy of a New Rapid Assay for Fecal Calprotectin Measurement. Clin. Lab. 2020, 66. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.S.; Lee, H.S.; Kim, S.K.; Kim, E.Y.; Jang, B.I.; Kim, K.O.; Yang, C.H.; Lee, Y.J.; Crohn’s and Colitis Association in Daegu-Gyeongbuk (CCAiD). Fecal calprotectin is more accurate than fecal immunochemical test for predicting mucosal healing in quiescent ulcerative colitis: A prospective multicenter study. Scand. J. Gastroenterol. 2020, 55, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Orivuori, L.; Mustonen, K.; de Goffau, M.C.; Hakala, S.; Paasela, M.; Roduit, C.; Dalphin, J.C.; Genuneit, J.; Lauener, R.; Riedler, J.; et al. High level of fecal calprotectin at age 2 months as a marker of intestinal inflammation predicts atopic dermatitis and asthma by age 6. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 928–939. [Google Scholar] [CrossRef] [PubMed]
- Peura, S.; Fall, T.; Almqvist, C.; Andolf, E.; Hedman, A.; Pershagen, G.; Helmersson-Karlqvist, J.; Larsson, A. Normal values for calprotectin in stool samples of infants from the population-based longitudinal born into life study. Scand. J. Clin. Lab. Investig. 2018, 78, 120–124. [Google Scholar] [CrossRef]
- Sung, V.; D’Amico, F.; Cabana, M.D.; Chau, K.; Koren, G.; Savino, F.; zajewska, H.; Deshpande, G.; Dupont, C.; Indrio, F.; et al. Lactobacillus reuteri to Treat Infant Colic: A Meta-analysis. Pediatrics 2018, 141, e20171811. [Google Scholar] [CrossRef]
- Qi, S.R.; Cui, Y.J.; Liu, J.X.; Luo, X.; Wang, H.F. Lactobacillus rhamnosus GG components, SLP, gDNA and CpG, exert protective effects on mouse macrophages upon lipopolysaccharide challenge. Lett. Appl. Microbiol. 2020, 70, 118–127. [Google Scholar] [CrossRef]
- Pärtty, A.; Luoto, R.; Kalliomäki, M.; Salminen, S.; Isolauri, E. Effects of early prebiotic and probiotic supplementation on development of gut microbiota and fussing and crying in preterm infants: A randomized, double-blind, placebo-controlled trial. J Pediatr. 2013, 163, 1272–1277.e1-2. [Google Scholar] [CrossRef]
- Loughman, A.; Quinn, T.; Nation, M.L.; Reichelt, A.; Moore, R.J.; Van, T.T.H.; Sung, V.; Tang, M.L.K. Infant microbiota in colic: Predictive associations with problem crying and subsequent child behavior. J. Dev. Orig. Health Dis. 2020, 1–11. [Google Scholar] [CrossRef]
- Isenbarger, T.A.; Carr, C.E.; Johnson, S.S.; Finney, M.; Church, G.M.; Gilbert, W.; Zuber, M.T.; Ruvkun, G. The most conserved genome segments for life detection on earth and other planets. Orig. Life Evol. Biosph. 2008, 38, 517–533. [Google Scholar] [CrossRef]
| L. rhamnosus (n = 24) | Placebo (n = 21) | p Value | |
|---|---|---|---|
| Type of delivery: | |||
| vaginal/caesarean | 14/10 | 12/9 | p > 0.05 # |
| Age at enrolment (days ± s.d.) | 37.9 ± 15 | 41.8 ± 17 | p > 0.05 * |
| Gender | |||
| Female, n (%) | 10 (41.6) | 9 (42.8) | p > 0.05 # |
| Male, n (%) | 14 (58.4) | 12 (57.2) | p > 0.05 # |
| Birth weight (g ± s.d.) | 3520 ± 450 | 3290 ± 390 | p > 0.05 * |
| Weigth at day 0 age (g ± s.d.) | 4270 ± 420 | 4080 ± 405 | p > 0.05 * |
| Weight gain per day (g) | 34.3 IR = 16.1 | 29.9 IR = 19.4 | p > 0.05 * |
| Crying and time at day 0 (m) | 242 ± 39.4 | 247 ± 19.5 | p > 0.05 * |
| Group | Day 0 Mean | Day 28 Mean | Difference between Means | p Value |
|---|---|---|---|---|
| L. rhamnosus (n = 24) | 242.0 | 104.7 | −137.3 | 0.001 |
| Placebo (n = 21) | 247.9 | 239.6 | −8.3 | p > 0.05 |
| L. rhamnosus Day 28 Mean (n = 24) | Placebo Day 28 Mean (n = 21) | Difference | p Value |
|---|---|---|---|
| 104.7 | 239.6 | 134.9 | 0.001 |
| Fecal Calprotectin | Day 0 | Day 28 | Mean Difference | p Value |
|---|---|---|---|---|
| L. rhamnosus (n = 24) * | 255.4 (±131.58) | 135.86 (±108.22) | −119.53 | 0.026 |
| Placebo (n = 21) * | 204.91 (±143.49) | 194.50 (±176.98) | −10.41 | 0.821 |
| L. rhamnosus Group (n = 24) | p Value | Placebo Group (n = 21) | p Value | |
|---|---|---|---|---|
| Total Bacteria | ||||
| Day 0 | 293,576 (±1,433,471) | 0.040 # | 16,110 (±64,580) | p > 0.05 |
| Day 30 | 409,845 (±721,248) | 13,655 (±14,445) | ||
| E. Coli | ||||
| Day 0 | 318 (±34) | p > 0.05 | 156 (±1169) | |
| Day 28 | 3123 (±1864) | 131 (±4235) | p > 0.05 | |
| Lactobacillus | ||||
| Day 0 | 28,857 (1 ± 55,662) | 0.048 # | 3835 (±20,023) | p > 0.05 |
| Day 28 | 79,570 (±298,763) | 4625 (±28,606) | ||
| Bifidobacteria | ||||
| Day 0 | 20,350 (±432,389) | p > 0.05 | 10,884(3 ± 9032) | p > 0.05 |
| Day 28 | 66,043 (±503,326) | 10,127(±26,786) | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savino, F.; Montanari, P.; Galliano, I.; Daprà, V.; Bergallo, M. Lactobacillus rhamnosus GG (ATCC 53103) for the Management of Infantile Colic: A Randomized Controlled Trial. Nutrients 2020, 12, 1693. https://doi.org/10.3390/nu12061693
Savino F, Montanari P, Galliano I, Daprà V, Bergallo M. Lactobacillus rhamnosus GG (ATCC 53103) for the Management of Infantile Colic: A Randomized Controlled Trial. Nutrients. 2020; 12(6):1693. https://doi.org/10.3390/nu12061693
Chicago/Turabian StyleSavino, Francesco, Paola Montanari, Ilaria Galliano, Valentina Daprà, and Massimiliano Bergallo. 2020. "Lactobacillus rhamnosus GG (ATCC 53103) for the Management of Infantile Colic: A Randomized Controlled Trial" Nutrients 12, no. 6: 1693. https://doi.org/10.3390/nu12061693
APA StyleSavino, F., Montanari, P., Galliano, I., Daprà, V., & Bergallo, M. (2020). Lactobacillus rhamnosus GG (ATCC 53103) for the Management of Infantile Colic: A Randomized Controlled Trial. Nutrients, 12(6), 1693. https://doi.org/10.3390/nu12061693






