Effects of Olive Oil on Blood Pressure: Epidemiological, Clinical, and Mechanistic Evidence
Abstract
1. Introduction
2. Dietary Strategies in the Management of BP
3. Olive Oil: A Succinct History, Composition, Production, and Consumption
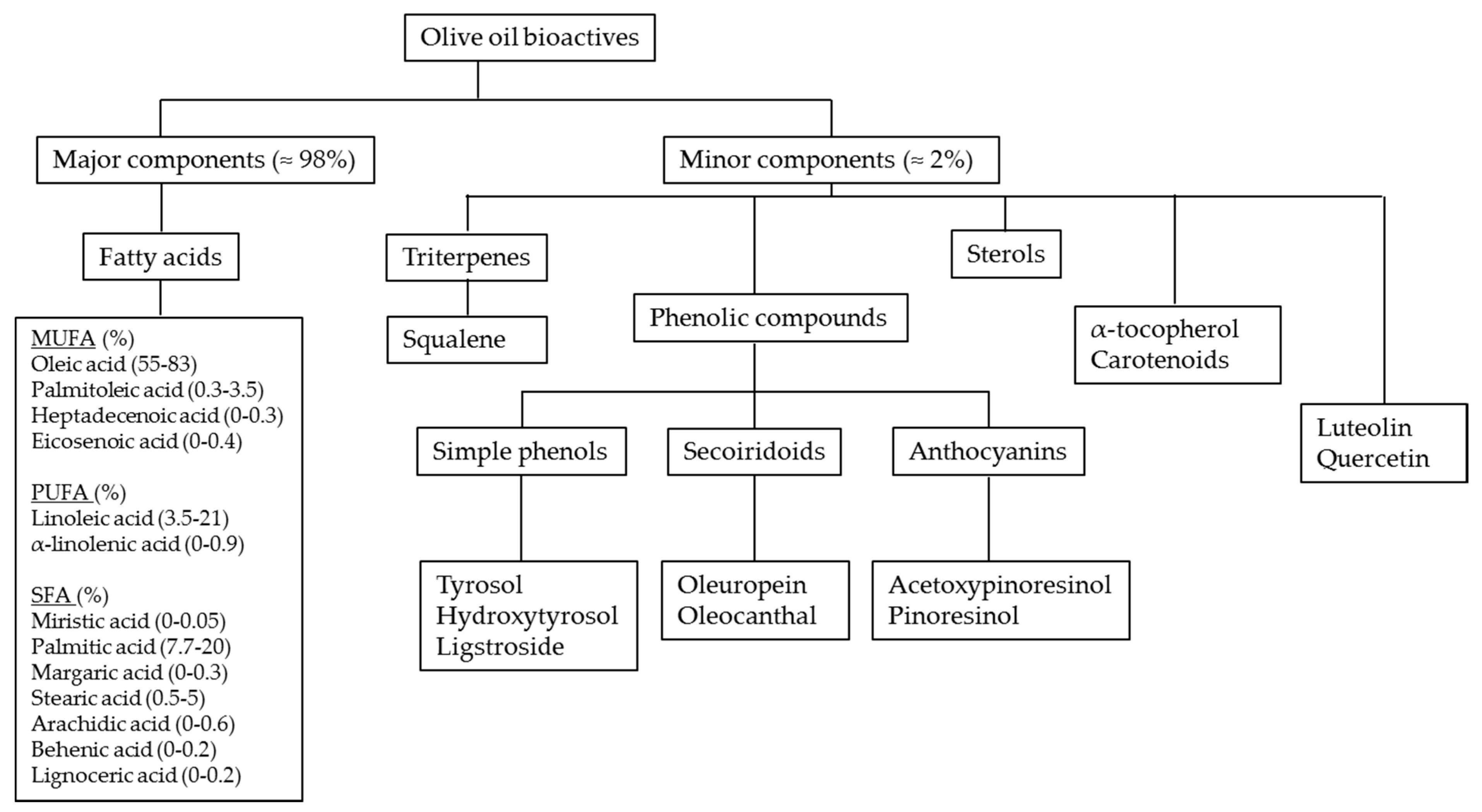
4. Olive Oil Composition
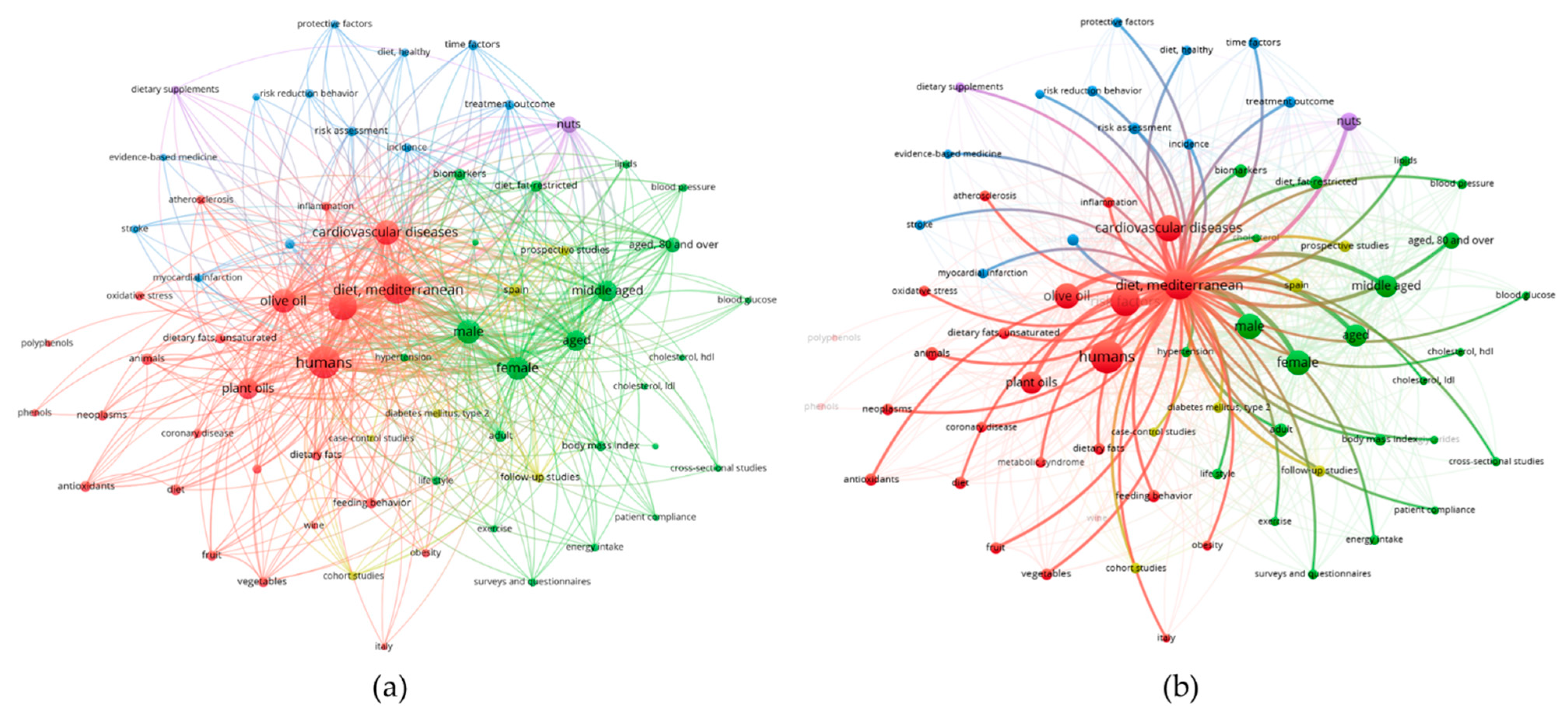
5. Olive Oil and Hypertension: Evidence from Epidemiological Studies
6. Olive Oil and Hypertension: Evidence from Clinical Trial. Search Strategy
7. Olive Oil and Hypertension: Clinical Trial Results
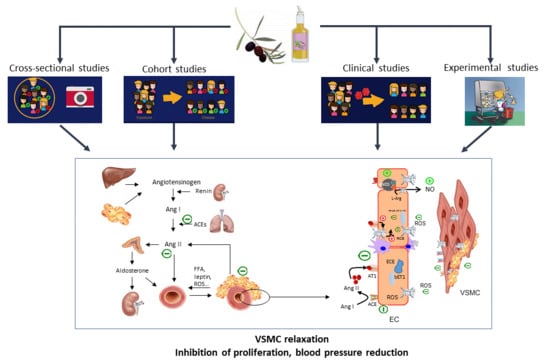
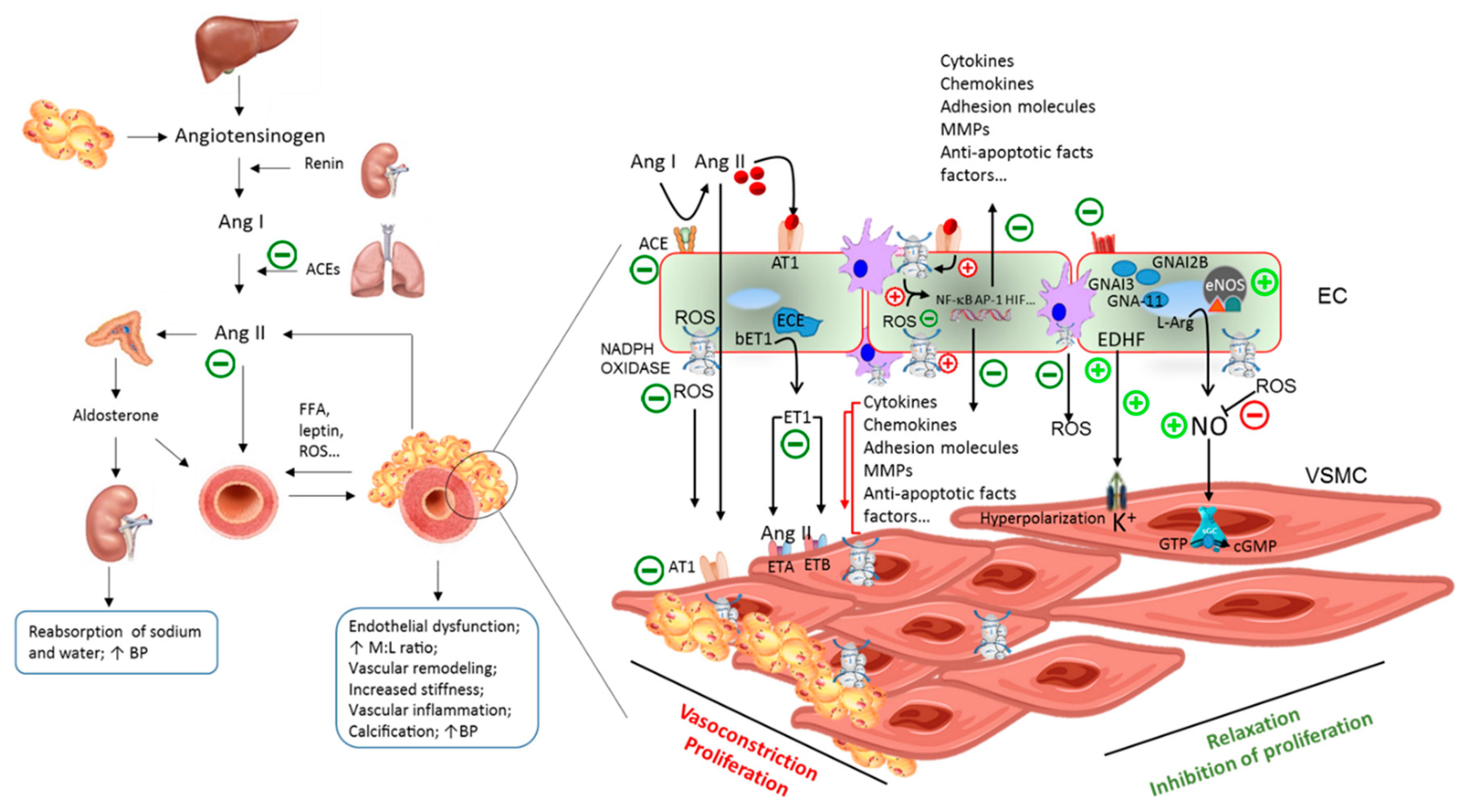
8. Pathogenesis of Hypertension
9. Olive Oil Anti-Hypertensive Effects: Mechanistic Evidence from Animal and Human Studies
10. Discussion
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oparil, S.; Acelajado, M.C.; Bakris, G.L.; Berlowitz, D.R.; Cifkova, R.; Dominiczak, A.F.; Grassi, G.; Jordan, J.; Poulter, N.R.; Rodgers, A.; et al. Hypertension. Nat. Rev. Dis. Primers 2018, 4, 18014. [Google Scholar] [CrossRef] [PubMed]
- Briasoulis, A.; Agarwal, V.; Tousoulis, D.; Stefanadis, C. Effects of antihypertensive treatment in patients over 65 years of age: A meta-analysis of randomised controlled studies. Heart 2014, 100, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Naghavi, M.; Wang, H.; Lozano, R.; Davis, A.; Liang, X.; Zhou, M.; Vollset, S.E.; Ozgoren, A.A.; Abdalla, S.; Abd-Allah, F.; et al. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [Google Scholar]
- WHO. A Global Brief on Hypertension; WHO: Geneva, Switzerland, 2013; pp. 1–40. [Google Scholar]
- Patel, R.S.; Masi, S.; Taddei, S. Understanding the role of genetics in hypertension. Eur. Heart J. 2017, 38, 2309–2312. [Google Scholar] [CrossRef]
- Singh, M.; Mensah, G.A.; Bakris, G. Pathogenesis and Clinical Physiology of Hypertension. Cardiol. Clin. 2010, 28, 545–559. [Google Scholar] [CrossRef]
- O’Shaughnessy, K.M. Role of diet in hypertension management. Curr. Hypertens. Rep. 2006, 8, 292–297. [Google Scholar] [CrossRef]
- Ndanuko, R.N.; Tapsell, L.C.; Charlton, K.E.; Neale, E.P.; Batterham, M.J. Dietary Patterns and Blood Pressure in Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2016, 7, 76–89. [Google Scholar] [CrossRef]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A Clinical Trial of the Effects of Dietary Patterns on Blood Pressure. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Martínez-González, M.A.; Tong, T.Y.N.; Forouhi, N.G.; Khandelwal, S.; Prabhakaran, D.; Mozaffarian, D.; de Lorgeril, M. Definitions and potential health benefits of the Mediterranean diet: Views from experts around the world. BMC Med. 2014, 12, 112. [Google Scholar] [CrossRef]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean Diet; a Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef]
- Aboul-Enein, B.H.; Puddy, W.C.; Bernstein, J. Ancel Benjamin Keys (1904–2004): His early works and the legacy of the modern Mediterranean diet. J. Med. Bioorg. 2017, 1, 0967772017727696. [Google Scholar] [CrossRef] [PubMed]
- Aparicio-Soto, M.; Sanchez-Hidalgo, M.; Rosillo, M.A.; Castejon, M.L.; Alarcon-de-la-Lastra, C. Extra virgin olive oil: A key functional food for prevention of immune-inflammatory diseases. Food Funct. 2016, 7, 4492–4505. [Google Scholar] [CrossRef] [PubMed]
- van Eck, N.J.; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010, 84, 523–538. [Google Scholar] [CrossRef] [PubMed]
- Clodoveo, M.L.; Camposeo, S.; De Gennaro, B.; Pascuzzi, S.; Roselli, L. In the ancient world, virgin olive oil was called “liquid gold” by Homer and “the great healer” by Hippocrates. Why has this mythic image been forgotten? Food Res. Int. 2014, 62, 1062–1068. [Google Scholar] [CrossRef]
- Berry, E.M.; Arnoni, Y.; Aviram, M. The Middle Eastern and biblical origins of the Mediterranean diet. Public Health Nutr. 2011, 14, 2288–2295. [Google Scholar] [CrossRef]
- Turkekul, B.; Gunden, C.; Abay, C.; Miran, B. A Market Share Analysis of Virgin Olive Oil Producer Countries with special respect to Competitiveness. In Proceedings of the 103rd Seminar, Barcelona, Spain, 23–25 April 2007. [Google Scholar]
- Visioli, F.; Poli, A.; Gall, C. Antioxidant and other biological activities of phenols from olives and olive oil. Med. Res. Rev. 2002, 22, 65–75. [Google Scholar] [CrossRef]
- Bedbabis, S.; Clodoveo, M.L.; Rouina, B.B.; Boukhris, M. Influence of irrigation with moderate saline water on “Chemlali” extra virgin olive oil composition and quality. J. Food Qual. 2010, 33, 228–247. [Google Scholar] [CrossRef]
- Boskou, D.; Blekas, G.; Tsimidou, M. 4—Olive Oil Composition. In Olive Oil, 2nd ed.; Boskou, D., Ed.; AOCS Press: Urbana, IL, USA, 2006; pp. 41–72. [Google Scholar] [CrossRef]
- Ortega, R. Importance of functional foods in the Mediterranean diet. Public Health Nutr. 2006, 9, 1136–1140. [Google Scholar] [CrossRef]
- Aparicio, R.; Ferreiro, L.; Alonso, V. Effect of climate on the chemical composition of virgin olive oil. Anal. Chim. Acta 1994, 292, 235–241. [Google Scholar] [CrossRef]
- Servili, M.; Selvaggini, R.; Esposto, S.; Taticchi, A.; Montedoro, G.; Morozzi, G. Health and sensory properties of virgin olive oil hydrophilic phenols: Agronomic and technological aspects of production that affect their occurrence in the oil. J. Chromatogr. A 2004, 1054, 113–127. [Google Scholar] [CrossRef]
- Bulotta, S.; Celano, M.; Lepore, S.M.; Montalcini, T.; Pujia, A.; Russo, D. Beneficial effects of the olive oil phenolic components oleuropein and hydroxytyrosol: Focus on protection against cardiovascular and metabolic diseases. J. Transl. Med. 2014, 12, 219. [Google Scholar] [CrossRef] [PubMed]
- Serreli, G.; Deiana, M. Biological Relevance of Extra Virgin Olive Oil Polyphenols Metabolites. Antioxidants 2018, 7, 170. [Google Scholar] [CrossRef] [PubMed]
- Bellumori, M.; Cecchi, L.; Innocenti, M.; Clodoveo, M.L.; Corbo, F.; Mulinacci, N. The EFSA Health Claim on Olive Oil Polyphenols: Acid Hydrolysis Validation and Total Hydroxytyrosol and Tyrosol Determination in Italian Virgin Olive Oils. Molecules 2019, 24, 2179. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.T.; Fortmann, S.P.; Terry, R.B.; Garay, S.C.; Vranizan, K.M.; Ellsworth, N.; Wood, P.D. Associations of dietary fat, regional adiposity, and blood pressure in men. JAMA 1987, 257, 3251–3256. [Google Scholar] [CrossRef]
- Stamler, J.; Caggiula, A.; Grandits, G.A.; Kjelsberg, M.; Cutler, J.A. Relationship to blood pressure of combinations of dietary macronutrients. Findings of the Multiple Risk Factor Intervention Trial (MRFIT). Circulation 1996, 94, 2417–2423. [Google Scholar] [CrossRef]
- Stamler, J.; Liu, K.; Ruth, K.J.; Pryer, J.; Greenland, P. Eight-year blood pressure change in middle-aged men: Relationship to multiple nutrients. Hypertension 2002, 39, 1000–1006. [Google Scholar] [CrossRef]
- Hajjar, I.; Kotchen, T. Regional variations of blood pressure in the United States are associated with regional variations in dietary intakes: The NHANES-III data. J. Nutr. 2003, 133, 211–214. [Google Scholar] [CrossRef]
- Trevisan, M.; Krogh, V.; Freudenheim, J.; Blake, A.; Muti, P.; Panico, S.; Farinaro, E.; Mancini, M.; Menotti, A.; Ricci, G. Consumption of olive oil, butter, and vegetable oils and coronary heart disease risk factors. The Research Group ATS-RF2 of the Italian National Research Council. JAMA 1990, 263, 688–692. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Psaltopoulou, T.; Naska, A.; Orfanos, P.; Trichopoulos, D.; Mountokalakis, T.; Trichopoulou, A. Olive oil, the Mediterranean diet, and arterial blood pressure: The Greek European Prospective Investigation into Cancer and Nutrition (EPIC) study. Am. J. Clin. Nutr. 2004, 80, 1012–1018. [Google Scholar] [CrossRef]
- Masala, G.; Bendinelli, B.; Versari, D.; Saieva, C.; Ceroti, M.; Santagiuliana, F.; Caini, S.; Salvini, S.; Sera, F.; Taddei, S.; et al. Anthropometric and dietary determinants of blood pressure in over 7000 Mediterranean women: The European Prospective Investigation into Cancer and Nutrition-Florence cohort. J. Hypertens. 2008, 26, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gonzalez, M.A.; Sanchez-Villegas, A.; De Irala, J.; Marti, A.; Martinez, J.A. Mediterranean diet and stroke: Objectives and design of the SUN project. Seguimiento Universidad de Navarra. Nutr. Neurosci. 2002, 5, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; de la Fuente, C.; Martin-Arnau, A.M.; de Irala, J.; Martinez, J.A.; Martinez-Gonzalez, M.A. Fruit and vegetable consumption is inversely associated with blood pressure in a Mediterranean population with a high vegetable-fat intake: The Seguimiento Universidad de Navarra (SUN) Study. Br. J. Nutr. 2004, 92, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; Martinez-Gonzalez, M.A. Olive oil consumption and reduced incidence of hypertension: The SUN study. Lipids 2004, 39, 1233–1238. [Google Scholar] [CrossRef]
- Miura, K.; Stamler, J.; Brown, I.J.; Ueshima, H.; Nakagawa, H.; Sakurai, M.; Chan, Q.; Appel, L.J.; Okayama, A.; Okuda, N.; et al. Relationship of dietary monounsaturated fatty acids to blood pressure: The International Study of Macro/Micronutrients and Blood Pressure. J. Hypertens. 2013, 31, 1144–1150. [Google Scholar] [CrossRef]
- Maki, K.C.; Slavin, J.L.; Rains, T.M.; Kris-Etherton, P.M. Limitations of observational evidence: Implications for evidence-based dietary recommendations. Adv. Nutr. 2014, 5, 7–15. [Google Scholar] [CrossRef]
- Pan, A.; Lin, X.; Hemler, E.; Hu, F.B. Diet and Cardiovascular Disease: Advances and Challenges in Population-Based Studies. Cell. Metab. 2018, 27, 489–496. [Google Scholar] [CrossRef]
- Ceriello, A.; Esposito, K.; La Sala, L.; Pujadas, G.; De Nigris, V.; Testa, R.; Bucciarelli, L.; Rondinelli, M.; Genovese, S. The protective effect of the Mediterranean diet on endothelial resistance to GLP-1 in type 2 diabetes: A preliminary report. Cardiovasc. Diabetol. 2014, 13, 140. [Google Scholar] [CrossRef]
- Toledo, E.; Hu, F.B.; Estruch, R.; Buil-Cosiales, P.; Corella, D.; Salas-Salvado, J.; Covas, M.I.; Aros, F.; Gomez-Gracia, E.; Fiol, M.; et al. Effect of the Mediterranean diet on blood pressure in the PREDIMED trial: Results from a randomized controlled trial. BMC Med. 2013, 11, 207. [Google Scholar] [CrossRef]
- Domenech, M.; Roman, P.; Lapetra, J.; Garcia de la Corte, F.J.; Sala-Vila, A.; de la Torre, R.; Corella, D.; Salas-Salvado, J.; Ruiz-Gutierrez, V.; Lamuela-Raventos, R.M.; et al. Mediterranean diet reduces 24-hour ambulatory blood pressure, blood glucose, and lipids: One-year randomized, clinical trial. Hypertension 2014, 64, 69–76. [Google Scholar] [CrossRef]
- Konstantinidou, V.; Covas, M.I.; Munoz-Aguayo, D.; Khymenets, O.; de la Torre, R.; Saez, G.; Tormos Mdel, C.; Toledo, E.; Marti, A.; Ruiz-Gutierrez, V.; et al. In vivo nutrigenomic effects of virgin olive oil polyphenols within the frame of the Mediterranean diet: A randomized controlled trial. FASEB J. 2010, 24, 2546–2557. [Google Scholar] [CrossRef] [PubMed]
- Fito, M.; Cladellas, M.; de la Torre, R.; Marti, J.; Alcantara, M.; Pujadas-Bastardes, M.; Marrugat, J.; Bruguera, J.; Lopez-Sabater, M.C.; Vila, J.; et al. Antioxidant effect of virgin olive oil in patients with stable coronary heart disease: A randomized, crossover, controlled, clinical trial. Atherosclerosis 2005, 181, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Luna, R.; Munoz-Hernandez, R.; Miranda, M.L.; Costa, A.F.; Jimenez-Jimenez, L.; Vallejo-Vaz, A.J.; Muriana, F.J.; Villar, J.; Stiefel, P. Olive oil polyphenols decrease blood pressure and improve endothelial function in young women with mild hypertension. Am. J. Hypertens. 2012, 25, 1299–1304. [Google Scholar] [CrossRef]
- Ruiz-Gutierrez, V.; Muriana, F.J.; Guerrero, A.; Cert, A.M.; Villar, J. Plasma lipids, erythrocyte membrane lipids and blood pressure of hypertensive women after ingestion of dietary oleic acid from two different sources. J. Hypertens. 1996, 14, 1483–1490. [Google Scholar] [CrossRef]
- Perona, J.S.; Canizares, J.; Montero, E.; Sanchez-Dominguez, J.M.; Catala, A.; Ruiz-Gutierrez, V. Virgin olive oil reduces blood pressure in hypertensive elderly subjects. Clin. Nutr. 2004, 23, 1113–1121. [Google Scholar] [CrossRef]
- Thomsen, C.; Rasmussen, O.W.; Hansen, K.W.; Vesterlund, M.; Hermansen, K. Comparison of the effects on the diurnal blood pressure, glucose, and lipid levels of a diet rich in monounsaturated fatty acids with a diet rich in polyunsaturated fatty acids in type 2 diabetic subjects. Diabet. Med. 1995, 12, 600–606. [Google Scholar] [CrossRef]
- Rasmussen, O.W.; Thomsen, C.; Hansen, K.W.; Vesterlund, M.; Winther, E.; Hermansen, K. Effects on blood pressure, glucose, and lipid levels of a high-monounsaturated fat diet compared with a high-carbohydrate diet in NIDDM subjects. Diabetes Care 1993, 16, 1565–1571. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, L.A.; Raimondi, A.S.; d’Episcopo, L.; Guida, L.; Dello Russo, A.; Marotta, T. Olive oil and reduced need for antihypertensive medications. Arch. Intern. Med. 2000, 160, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.S.; Williams, S.R.; Rhys, R.; James, P.; Frenneaux, M.P. Conjugated linoleic acid impairs endothelial function. Arter. Thromb Vasc. Biol. 2006, 26, 307–312. [Google Scholar] [CrossRef]
- Appel, L.J.; Sacks, F.M.; Carey, V.J.; Obarzanek, E.; Swain, J.F.; Miller, E.R., 3rd; Conlin, P.R.; Erlinger, T.P.; Rosner, B.A.; Laranjo, N.M.; et al. Effects of protein, monounsaturated fat, and carbohydrate intake on blood pressure and serum lipids: Results of the OmniHeart randomized trial. JAMA 2005, 294, 2455–2464. [Google Scholar] [CrossRef]
- Rozati, M.; Barnett, J.; Wu, D.; Handelman, G.; Saltzman, E.; Wilson, T.; Li, L.; Wang, J.; Marcos, A.; Ordovas, J.M.; et al. Cardio-metabolic and immunological impacts of extra virgin olive oil consumption in overweight and obese older adults: A randomized controlled trial. Nutr. Metab. 2015, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Venturini, D.; Simao, A.N.; Urbano, M.R.; Dichi, I. Effects of extra virgin olive oil and fish oil on lipid profile and oxidative stress in patients with metabolic syndrome. Nutrition 2015, 31, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Passfall, J.; Philipp, T.; Woermann, F.; Quass, P.; Thiede, M.; Haller, H. Different effects of eicosapentaenoic acid and olive oil on blood pressure, intracellular free platelet calcium, and plasma lipids in patients with essential hypertension. Clin. Investig. 1993, 71, 628–633. [Google Scholar] [CrossRef]
- Bondia-Pons, I.; Schroder, H.; Covas, M.I.; Castellote, A.I.; Kaikkonen, J.; Poulsen, H.E.; Gaddi, A.V.; Machowetz, A.; Kiesewetter, H.; Lopez-Sabater, M.C. Moderate consumption of olive oil by healthy European men reduces systolic blood pressure in non-Mediterranean participants. J. Nutr. 2007, 137, 84–87. [Google Scholar] [CrossRef]
- Rasmussen, B.M.; Vessby, B.; Uusitupa, M.; Berglund, L.; Pedersen, E.; Riccardi, G.; Rivellese, A.A.; Tapsell, L.; Hermansen, K.; Group, K.S. Effects of dietary saturated, monounsaturated, and n-3 fatty acids on blood pressure in healthy subjects. Am. J. Clin. Nutr. 2006, 83, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Mensink, R.P.; Janssen, M.C.; Katan, M.B. Effect on blood pressure of two diets differing in total fat but not in saturated and polyunsaturated fatty acids in healthy volunteers. Am. J. Clin. Nutr. 1988, 47, 976–980. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, E.; Parati, G.; Stergiou, G.; Asmar, R.; Beilin, L.; Bilo, G.; Clement, D.; de la Sierra, A.; de Leeuw, P.; Dolan, E.; et al. European Society of Hypertension position paper on ambulatory blood pressure monitoring. J. Hypertens. 2013, 31, 1731–1768. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef]
- Singer, P.; Jaeger, W.; Berger, I.; Barleben, H.; Wirth, M.; Richter-Heinrich, E.; Voigt, S.; Godicke, W. Effects of dietary oleic, linoleic and alpha-linolenic acids on blood pressure, serum lipids, lipoproteins and the formation of eicosanoid precursors in patients with mild essential hypertension. J. Hum. Hypertens. 1990, 4, 227–233. [Google Scholar]
- Martin-Pelaez, S.; Castaner, O.; Konstantinidou, V.; Subirana, I.; Munoz-Aguayo, D.; Blanchart, G.; Gaixas, S.; de la Torre, R.; Farre, M.; Saez, G.T.; et al. Effect of olive oil phenolic compounds on the expression of blood pressure-related genes in healthy individuals. Eur. J. Nutr. 2017, 56, 663–670. [Google Scholar] [CrossRef]
- Patel, S.; Rauf, A.; Khan, H.; Abu-Izneid, T. Renin-angiotensin-aldosterone (RAAS): The ubiquitous system for homeostasis and pathologies. Biomed. Pharmacother. Biomed. Pharmacother. 2017, 94, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Williams, B. Angiotensin II and the pathophysiology of cardiovascular remodeling. Am. J. Cardiol. 2001, 87, 10c–17c. [Google Scholar] [CrossRef]
- Williams, B. Drug discovery in renin-angiotensin system intervention: Past and future. Ther. Adv. Cardiovasc. Dis. 2016, 10, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Briet, M.; Schiffrin, E.L. Vascular Actions of Aldosterone. J. Vasc. Res. 2013, 50, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Leggio, M.; Lombardi, M.; Caldarone, E.; Severi, P.; D’Emidio, S.; Armeni, M.; Bravi, V.; Bendini, M.G.; Mazza, A. The relationship between obesity and hypertension: An updated comprehensive overview on vicious twins. Hypertens. Res. 2017, 40, 947–963. [Google Scholar] [CrossRef] [PubMed]
- Guzik, T.J.; Touyz, R.M. Oxidative Stress, Inflammation, and Vascular Aging in Hypertension. Hypertension 2017, 70, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Massaro, M.; Scoditti, E.; Carluccio, M.A.; De Caterina, R. Oxidative stress and vascular stiffness in hypertension: A renewed interest for antioxidant therapies? Vasc. Pharm. 2019, 116, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Montezano, A.C.; Touyz, R.M. Molecular mechanisms of hypertension—Reactive oxygen species and antioxidants: A basic science update for the clinician. Can. J. Cardiol. 2012, 28, 288–295. [Google Scholar] [CrossRef]
- Drew, B.; Leeuwenburgh, C. Aging and the role of reactive nitrogen species. Ann. N. Y. Acad. Sci. 2002, 959, 66–81. [Google Scholar] [CrossRef]
- Garcia-Redondo, A.B.; Aguado, A.; Briones, A.M.; Salaices, M. NADPH oxidases and vascular remodeling in cardiovascular diseases. Pharm. Res. 2016, 114, 110–120. [Google Scholar] [CrossRef]
- Duansak, N.; Schmid-Schonbein, G.W. The oxygen free radicals control MMP-9 and transcription factors expression in the spontaneously hypertensive rat. Microvasc. Res. 2013, 90, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Villarejo, A.B.; Ramirez-Sanchez, M.; Segarra, A.B.; Martinez-Canamero, M.; Prieto, I. Influence of extra virgin olive oil on blood pressure and kidney angiotensinase activities in spontaneously hypertensive rats. Planta. Med. 2015, 81, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Rodriguez, R.; Herrera, M.D.; de Sotomayor, M.A.; Ruiz-Gutierrez, V. Pomace olive oil improves endothelial function in spontaneously hypertensive rats by increasing endothelial nitric oxide synthase expression. Am. J. Hypertens. 2007, 20, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, A.; Sanchez-Rodriguez, E.; Vargas, F.; Montoro-Molina, S.; Romero, M.; Espejo-Calvo, J.A.; Vilchez, P.; Jaramillo, S.; Olmo-Garcia, L.; Carrasco-Pancorbo, A.; et al. Cardioprotective Effect of a Virgin Olive Oil Enriched with Bioactive Compounds in Spontaneously Hypertensive Rats. Nutrients 2019, 11, 1728. [Google Scholar] [CrossRef]
- Romero, M.; Toral, M.; Gomez-Guzman, M.; Jimenez, R.; Galindo, P.; Sanchez, M.; Olivares, M.; Galvez, J.; Duarte, J. Antihypertensive effects of oleuropein-enriched olive leaf extract in spontaneously hypertensive rats. Food Funct. 2016, 7, 584–593. [Google Scholar] [CrossRef]
- Calabriso, N.; Massaro, M.; Scoditti, E.; D’Amore, S.; Gnoni, A.; Pellegrino, M.; Storelli, C.; De Caterina, R.; Palasciano, G.; Carluccio, M.A. Extra virgin olive oil rich in polyphenols modulates VEGF-induced angiogenic responses by preventing NADPH oxidase activity and expression. J. Nutr. Biochem. 2016, 28, 19–29. [Google Scholar] [CrossRef]
- Calabriso, N.; Gnoni, A.; Stanca, E.; Cavallo, A.; Damiano, F.; Siculella, L.; Carluccio, M.A. Hydroxytyrosol Ameliorates Endothelial Function under Inflammatory Conditions by Preventing Mitochondrial Dysfunction. Oxid. Med. Cell. Longev. 2018, 2018, 9086947. [Google Scholar] [CrossRef]
- Scoditti, E.; Pellegrino, M.; Massaro, M.; Carluccio, M.A.; Calabriso, N.; Wabitsch, M.; Storelli, C.; De Caterina, R. Anti-inflammatory nutrigenomic effects of hydroxytyrosol in human adipocytes—Protective mechanisms of mediterranean diets in obesity-related inflammation. Cardiovasc. Res. 2016, 111, S40. [Google Scholar]
- Scoditti, E.; Carpi, S.; Massaro, M.; Pellegrino, M.; Polini, B.; Carluccio, M.A.; Wabitsch, M.; Verri, T.; Nieri, P.; De Caterina, R. Hydroxytyrosol Modulates Adipocyte Gene and miRNA Expression Under Inflammatory Condition. Nutrients 2019, 11, 2493. [Google Scholar] [CrossRef]
- Teres, S.; Barcelo-Coblijn, G.; Benet, M.; Alvarez, R.; Bressani, R.; Halver, J.E.; Escriba, P.V. Oleic acid content is responsible for the reduction in blood pressure induced by olive oil. Proc. Natl. Acad. Sci. USA 2008, 105, 13811–13816. [Google Scholar] [CrossRef]
- Yang, Q.; Alemany, R.; Casas, J.; Kitajka, K.; Lanier, S.M.; Escriba, P.V. Influence of the membrane lipid structure on signal processing via G protein-coupled receptors. Mol. Pharmacol. 2005, 68, 210–217. [Google Scholar] [CrossRef]
- Covas, M.I.; Nyyssonen, K.; Poulsen, H.E.; Kaikkonen, J.; Zunft, H.J.; Kiesewetter, H.; Gaddi, A.; de la Torre, R.; Mursu, J.; Baumler, H.; et al. The effect of polyphenols in olive oil on heart disease risk factors: A randomized trial. Ann. Intern. Med. 2006, 145, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Storniolo, C.E.; Casillas, R.; Bullo, M.; Castaner, O.; Ros, E.; Saez, G.T.; Toledo, E.; Estruch, R.; Ruiz-Gutierrez, V.; Fito, M.; et al. A Mediterranean diet supplemented with extra virgin olive oil or nuts improves endothelial markers involved in blood pressure control in hypertensive women. Eur. J. Nutr. 2017, 56, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Massaro, M.; Basta, G.; Lazzerini, G.; Carluccio, M.A.; Bosetti, F.; Solaini, G.; Visioli, F.; Paolicchi, A.; De Caterina, R. Quenching of intracellular ROS generation as a mechanism for oleate-induced reduction of endothelial activation and early atherogenesis. Thromb. Haemost. 2002, 88, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A.; Massaro, M.; Scoditti, E.; Storelli, C.; Visioli, F.; Distante, A.; De Caterina, R. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation—Antiatherogenic properties of Mediterranean diet phytochemicals. Arter. Throm. Vas. 2003, 23, 622–629. [Google Scholar] [CrossRef]
- Scoditti, E.; Massaro, M.; Carluccio, M.A.; Pellegrino, M.; Wabitsch, M.; Calabriso, N.; Storelli, C.; De Caterina, R. Additive regulation of adiponectin expression by the mediterranean diet olive oil components oleic Acid and hydroxytyrosol in human adipocytes. Cardiovasc. Diabetol. 2015, 10, e0128218. [Google Scholar] [CrossRef] [PubMed]
- Massaro, M.; Scoditti, E.; Carluccio, M.A.; De Caterina, R. Nutraceuticals and Prevention of Atherosclerosis: Focus on omega-3 Polyunsaturated Fatty Acids and Mediterranean Diet Polyphenols. Cardiovasc. Ther. 2010, 28, e13–e19. [Google Scholar] [CrossRef]
- Milenkovic, D.; Jude, B.; Morand, C. miRNA as molecular target of polyphenols underlying their biological effects. Free. Radic. Biol. Med. 2013, 64, 40–51. [Google Scholar] [CrossRef]
- Pacurari, M.; Tchounwou, P.B. Role of MicroRNAs in Renin-Angiotensin-Aldosterone System-Mediated Cardiovascular Inflammation and Remodeling. Int. J. Inflamm. 2015, 2015, 101527. [Google Scholar] [CrossRef]
- Filipek, A.; Czerwinska, M.E.; Kiss, A.K.; Polanski, J.A.; Naruszewicz, M. Oleacein may inhibit destabilization of carotid plaques from hypertensive patients. Impact on high mobility group protein-1. Phytomedicine. Int. J. Phytother. Phytopharm. 2017, 32, 68–73. [Google Scholar] [CrossRef]
- Carpi, S.; Scoditti, E.; Massaro, M.; Polini, B.; Manera, C.; Digiacomo, M.; Esposito Salsano, J.; Poli, G.; Tuccinardi, T.; Doccini, S.; et al. The Extra-Virgin Olive Oil Polyphenols Oleocanthal and Oleacein Counteract Inflammation-Related Gene and miRNA Expression in Adipocytes by Attenuating NF-kappaB Activation. Nutrients 2019, 11, 2855. [Google Scholar] [CrossRef] [PubMed]
- Trial, V.A. Effects of treatment on morbidity in hypertension. Results in patients with diastolic blood pressures averaging 115 through 129 mm Hg. JAMA 1967, 202, 1028–1034. [Google Scholar] [CrossRef]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E., Jr.; Collins, K.J.; Dennison Himmelfarb, C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 2018, 71, 1269–1324. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, J.; Heaney, R.P.; Huncharek, M.; Scholl, T.; Stampfer, M.; Vieth, R.; Weaver, C.M.; Zeisel, S.H. Evidence-based criteria in the nutritional context. Nutr. Rev. 2010, 68, 478–484. [Google Scholar] [CrossRef]
- Lennon, S.L.; DellaValle, D.M.; Rodder, S.G.; Prest, M.; Sinley, R.C.; Hoy, M.K.; Papoutsakis, C. 2015 Evidence Analysis Library Evidence-Based Nutrition Practice Guideline for the Management of Hypertension in Adults. J. Acad. Nutr. Diet. 2017, 117, 1445–1458. [Google Scholar] [CrossRef]
- World Health Organization, International Society of Hypertension Writing Group. 2003 World Health Organization (WHO)/International Society of Hypertension (ISH) statement on management of hypertension. J. Hypertens. 2003, 21, 1983–1992. [Google Scholar] [CrossRef]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The Mediterranean diet, its components, and cardiovascular disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef]
- Alonso, Á.; Ruiz-Gutierrez, V.; Martínez-González, M.Á. Monounsaturated fatty acids, olive oil and blood pressure: Epidemiological, clinical and experimental evidence. Public Health Nutr. 2007, 9, 251–257. [Google Scholar] [CrossRef]
- Guasch-Ferre, M.; Zong, G.; Willett, W.C.; Zock, P.L.; Wanders, A.J.; Hu, F.B.; Sun, Q. Associations of Monounsaturated Fatty Acids From Plant and Animal Sources With Total and Cause-Specific Mortality in Two US Prospective Cohort Studies. Circ. Res. 2019, 124, 1266–1275. [Google Scholar] [CrossRef]
- Stamler, R. Implications of the INTERSALT study. Hypertension 1991, 17, I16–I20. [Google Scholar] [CrossRef]
- Garaulet, M.; Lee, Y.C.; Shen, J.; Parnell, L.D.; Arnett, D.K.; Tsai, M.Y.; Lai, C.Q.; Ordovas, J.M. CLOCK genetic variation and metabolic syndrome risk: Modulation by monounsaturated fatty acids. Am. J. Clin. Nutr. 2009, 90, 1466–1475. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Asensio, E.M.; Coltell, O.; Sorli, J.V.; Estruch, R.; Martinez-Gonzalez, M.A.; Salas-Salvado, J.; Castaner, O.; Aros, F.; Lapetra, J.; et al. CLOCK gene variation is associated with incidence of type-2 diabetes and cardiovascular diseases in type-2 diabetic subjects: Dietary modulation in the PREDIMED randomized trial. Cardiovasc. Diabetol. 2016, 15, 4. [Google Scholar] [CrossRef] [PubMed]
- Nair, S.R. Personalized medicine: Striding from genes to medicines. Perspect. Clin. Res. 2010, 1, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Scoditti, E.; Capurso, C.; Capurso, A.; Massaro, M. Vascular effects of the Mediterranean diet-Part II: Role of omega-3 fatty acids and olive oil polyphenols. Vasc. Pharm. 2014, 63, 127–134. [Google Scholar] [CrossRef] [PubMed]

| First Author, Year [Ref] | Country | Design | Participants | Sex | Main Results |
|---|---|---|---|---|---|
| Williams, 1987, [1] | USA | Cross-sectional | 76 | male | MUFA ↓ SBP and DBP |
| Stamler, 1996 [2] | USA | Cohort | 11,342 | male | MUFA ↔ SBP and DBP |
| Stamler, 2002 [3] | USA | Cohort | 1714 | male | MUFA ↑ SBP |
| Hajjar, 2004, [4] | USA | Cross-sectional | 17,752 | male and female | MUFA ↑ SBP and DBP |
| Trevisan, 1990 [5] | Italy | Cross-sectional | 4903 | male and female | OO ↓ SBP |
| Alonso, 2004 [6] | Spain | Cohort | 6863 | male and female | OO ↓ hypertension risk in men |
| Psaltopoulou, 2004 [7] | Greece | Cross-sectional | 20,343 | male and female | OO ↓ SBP and DBP |
| Masala, 2008 [8] | Italy | Cross-sectional | 7601 | female | OO ↓ DBP |
| Miura, 2013 [9] | China, Japan, UK, USA | Cross-sectional | 4680 | male and female | MUFA ↓ DBP |
| First Author, Year [ref] | N, Sex, Age (yr), Weight (kg) (or BMI) | Health Status | Study Design and Country | Intervention | Measure | Administration | Δ SBP (mmHg) | Δ DBP (mmHg) | Main Results |
|---|---|---|---|---|---|---|---|---|---|
| Mensink, 1988 [59] | 47, male and female, 27 ± NR; 71 ± NR | healthy | RCT, parallel, Netherlands | 8-week OO-enriched diet vs. CHO diet | Office | liquid | −2.3 in CHO-group; −2.7 in OO-group | −4.7 in CHO group; −4.4 in OO-group | Both interventions decreased SBP and DBP significantly, but there were no differences between groups |
| Rasmussen, 1993 [50] | 15, male and female, 57 ± 2; 80.5 ± 3.8 | diabetics | RCT, cross-over, Denmark | 3-week intervention with 3-week wash-out period, EVOO-enriched diet vs. CHO diet | 24-AMBP | liquid, cold pressed olive oil | −4.0 in EVOO diet vs. CHO diet | −3.0 in EVOO diet vs. CHO diet | SBP and DBP significantly lower after MUFA diet than CHO diet |
| Rasmussen, 2006 [58] | 162, male and female, 48.5 ± 8.0, 26.5 ± 3.8 kg/m2 (BMI) | healthy | RCT, parallel, multicenter | 12-week intervention with MUFA diet or SFA diet. Each group was further randomly assigned to receive supplementation with fish oil or placebo | Office | margarine with a high proportion of oleic acid, derived from high-oleic acid sunflower oil | −2.2% | −3.8% | SBP and DBP decreased with the MUFA diet but did not change with the SFA diet |
| Thomsen, 1995 [49] | 16, male and female, 59 ± 7, 81.6 ± 15.1 | diabetics | RCT, cross-over, Denmark | 3-week intervention with 3-week wash-out period, MUFA diet (EVOO) vs. PUFA diet | 24-AMBP | liquid, cold pressed olive oil | −5.1 in EVOO diet vs. PUFA diet | −3.8 in EVOO diet vs. CHO diet | SBP and DBP were lower after EVOO diet than after PUFA diet |
| Ruiz-Gutierrez, 1996 [47] | 16, female, 56.2 ± 5.4, NR | NC and HC hypertensive women | RCT, parallel, Spain | 4-week intervention with 4-week wash-out period, EVOO vs. HOSO | Office | liquid, cold pressed OO or SO | −10 in NC women on EVOO; −7 in HC women on EVOO; −6 in NC women on HOSO; −2,26 in HC women on HOSO | −10 in NC women on EVOO; −6 in HC women on EVOO; −4 in NC women on HOSO; −5.92 in HC women on HOSO | Significant decrease in SBP and DBP after EVOO but not after HOSO |
| Ferrara, 2000 [51] | 23, male and female, age range 25–70, 70 ± 9 | hypertensive | RCT, cross-over, Italy | 24-week intervention, EVOO diet vs. SO diet | Office | liquid, cold pressed OO or SO | −7 in EVOO diet; +1 in SO diet | −6 in EVOO diet; no change in SO diet | SBP and DBP decresed after EVOO but not after SO. Reduced need for antihypertensive drugs after EVOO |
| Appel, 2005 [53] | 164, male and female, 53.6 ± 10.9; 87.3 ± 18.7 | pre-hypertensives and hypertensives | RCT, cross-over, USA | CHO-rich diet (similar to the DASH trial) vs. protein-rich diet vs. MUFA-rich diet-2-4-week wash-out period between each feeding period. | Office | liquid, olive, canola, and safflower oils beside to nuts and seeds | −9.3 in MUFA rich diet; −8.2 in CHO rich diet | −4.8 in MUFA rich diet; −4.1 in CHO rich diet | SBP and DBP were lower after MUFA-rich diet compared with CHO diet; no significant difference there were between protein and MUFA diets |
| Perona, 2004 [48] | 62, male and female, 84.0 ± 7.4; 28.8 ± 5.2 kg/m2 (BMI) | hypertensive and normotensive | RCT, cross-over, Spain | 4-week intervention with 4-week wash-out period, EVOO diet vs. SO diet | Office | liquid, cold pressed OO or SO | −12 in EVOO diet vs. SO diet in hypertensive | no difference between EVOO and SO | Normalization of SBP after EVOO in hypertensive individuals. No effect on DBP |
| Taylor, 2006 [52] | 40, men, 47 ± 8; 97 ± 13 | overweight | RCT, parallel, UK | OO, 6 g/day capsules; CLA, 4.5 g/day capsules | Office | capsules | +0.2 in OO group; −0.4 in CLA group | −0.8 in OO group; +0.1 in CLA group | With OO only DBP decrease (by trend); with conjugated linoleic acid only SBP decrease by trend |
| Konstantinidou, 2010 [44] | 90, male and female, 45 ± 10; 68 ± 15 | healthy | RCT, parallel, Spain | 12-week intervention with MD + OO (low polyphenol content) or MD +EVOO (high polyphenol content); habitual diet | Office | liquid | −1.63 in low polyphenol OO; −0.4 in EVOO; +1.4 in control diet | −0.8 in low polyphenol OO; +1.12 in EVOO +1.67 in control diet | With low polyphenols OO SBP and DBP decrease (by trend); with EVO only SBP decrease (by trend) |
| Fitò, 2005 [45] | 40, male, 68 ± 8; 27.5 ± 3 kg/m2 (BMI) | at CHD risk and hypertensive | RCT, cross-over, Spain | 3-week intervention with 2-week wash-out period, EVOO vs. ROO | Office | liquid | −2.53 in EVOO group vs. ROO group | +1.16 in EVOO group vs. ROO group | With EVOO SBP decreased in hypertensive patients. No changes were observed in DBP |
| Moreno Luna, 2012 [46] | 24, women, age range: 24–27 years, BMI range: 23.5–27.1 kg/m2 | hypertensive and normotensive women | RCT, cross-over, Spain | 8-week intervention with MD + OO (polyphenol free) or MD + EVOO (rich in polyphenols); 4-week wash-out period; | Office | liquid | −7.91 in EVOO group vs. baseline; −1.65 in OO polyphenol free group vs. baseline | −6.65 in EVOO group vs. baseline; −2.17 in OO polyphenol free group vs. baseline | Only polyphenol-rich OO decrease SBP and DBP |
| Bondia-Pons, 2008 [57] | 160, male, 33.3 ± 11.1; 75.8 ± 9.7 kg | healthy | RCT, cross-over multicenter | 3-week intervention with 2-week wash-out periods. OO with different polyphenol content (low, medium, high) | Office | liquid | −4.7 after consuming OO for 9 wk in Northern Europe subjects vs. baseline; −4.4 after consuming OO for 9 wk in Central Europe subjects vs. baseline | −2.2 after consuming OO for 9 wk in Northern Europe subjects vs. baseline; −3.1 after consuming OO for 9 wk in Central Europe subjects vs. baseline | Only SBP significantly decreased after 9 wk of OO |
| Rozati, 2015 [54] | 41; male and female; 72.0 ± 1; 80 ± 2 | overweight and obese | RCT, parallel, USA | 12-week intervention with American diet + EVOO or American diet + Control oil (corn oil and soybean oil) | Office | liquid | −6 in EVOO group vs. baseline; no change in control oil group vs. baseline | −3 in EVOO group vs. baseline; −3 in control oil group vs. baseline | Only SBP significantly decreased after 12 wk of EVOO |
| Venturini, 2015 [55] | 102; male and female; 51.4 ± 8.27; NR | metabolic syndrome | RCT, parallel, Brazil | (1) 12-week intervention with 3 g/d of fish oil; (2) 10 mL/d of EVOO at lunch and dinner; (3) fish oil and plus EVOO. (4) control group (usual diet); | Office | liquid | −5 in EVOO group vs. baseline; no change in control oil group vs. baseline | −5 in EVOO group vs. baseline; no change in control oil group vs. baseline | In the OO group both SBP and DBP decreased |
| Toledo, 2013 [42] | 7158, male and female, 66.1 ± 6.1, 30 ± 4 kg/m2 (BMI) | at CHD risk and hypertensive | RCT, parallel, multicenter, Spain | 4.8-year intervention with (1) MD supplemented with EEVOO, (2) MD supplemented with mixed nuts or (3) control diet (low-fat diet). | Office | liquid | no change in EVOO vs. control | −1.53 in EVOO vs. control; −0.65 in nut group vs. control | Only DBP significantly decreased after 4.5 yr of EVOO |
| Doménech, 2014 [43] | 235, male and female, 66.1 ± 6.1, 78 ± 11 | at CHD risk and hypertensive | RCT, parallel, multicenter, Spain | 4.8-year intervention with (1) MD supplemented with EVOO, (2) MD supplemented with mixed nuts or (3) control diet (low-fat diet). | 24-AMBP | liquid | −2.3 in EVOO vs. baseline; −2.6 in nut groups vs. baseline; +1.7 in the control group vs. baseline | −1.2 in EVOO vs. baseline; −1.2 in nut groups vs. baseline; +0.7 in the control group vs. baseline | SBP and DBP decreased with the MD enriched in EVOO or nut |
| Martin-Pelàez, 2015 [63] | 22, male, 36.0 ± 11.1, 78.5 ± 11.9 | healthy | RCT, cross-over, Spain | 3-week intervention with 2-week wash-out period, EVOO vs. ROO | Office | liquid | −4.22 in EVOO group vs. baseline | −2.11 in EVOO group vs. baseline | SBP and DBP were significantly reduced only in EVOO |
| Ceriello, 2104 [41] | 22, male and female, NR, 29.1 ± 1.2 | diabetics | RCT, parallel, Spain | 12-week intervention with MD + EVOO or a control low-fat diet | Office | liquid | no change in EVOO vs. baseline | no change in EVOO vs. baseline | No effect |
| Passfall, 1993 [56] | 10, male and female, age range 40–61, NR | hypertensive | RCT, cross-over, Germany | 6-week intervention with 4-week wash-out period, supplementation with OO (9 g) vs. fish oil (9 g) | Office | capsules | no change after OO vs. baseline | no change after OO vs. baseline | No effect |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Massaro, M.; Scoditti, E.; Carluccio, M.A.; Calabriso, N.; Santarpino, G.; Verri, T.; De Caterina, R. Effects of Olive Oil on Blood Pressure: Epidemiological, Clinical, and Mechanistic Evidence. Nutrients 2020, 12, 1548. https://doi.org/10.3390/nu12061548
Massaro M, Scoditti E, Carluccio MA, Calabriso N, Santarpino G, Verri T, De Caterina R. Effects of Olive Oil on Blood Pressure: Epidemiological, Clinical, and Mechanistic Evidence. Nutrients. 2020; 12(6):1548. https://doi.org/10.3390/nu12061548
Chicago/Turabian StyleMassaro, Marika, Egeria Scoditti, Maria Annunziata Carluccio, Nadia Calabriso, Giuseppe Santarpino, Tiziano Verri, and Raffaele De Caterina. 2020. "Effects of Olive Oil on Blood Pressure: Epidemiological, Clinical, and Mechanistic Evidence" Nutrients 12, no. 6: 1548. https://doi.org/10.3390/nu12061548
APA StyleMassaro, M., Scoditti, E., Carluccio, M. A., Calabriso, N., Santarpino, G., Verri, T., & De Caterina, R. (2020). Effects of Olive Oil on Blood Pressure: Epidemiological, Clinical, and Mechanistic Evidence. Nutrients, 12(6), 1548. https://doi.org/10.3390/nu12061548