Metabolic Impacts of Confinement during the COVID-19 Pandemic Due to Modified Diet and Physical Activity Habits
Abstract
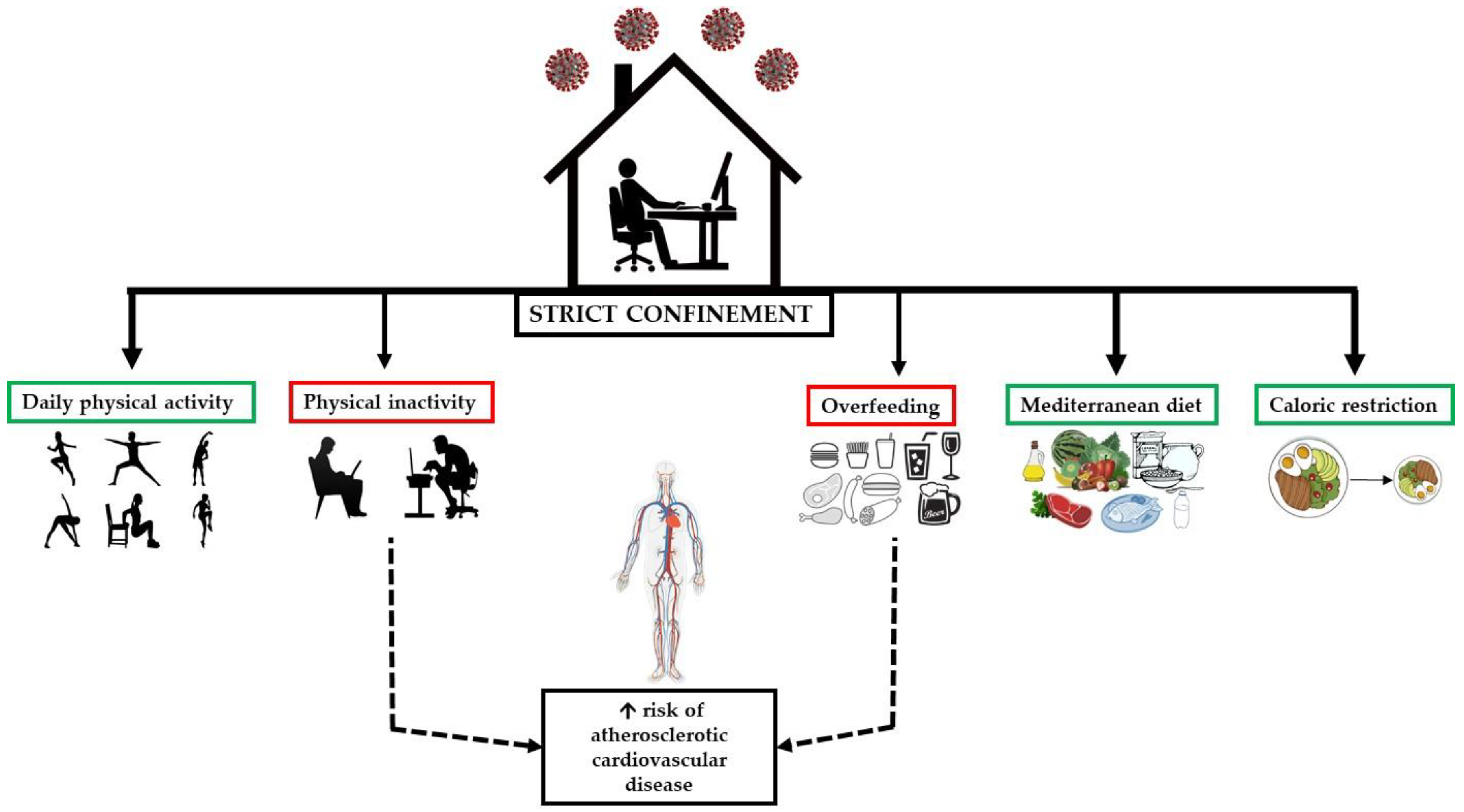
1. Introduction
2. Methods
3. Results and Discussion
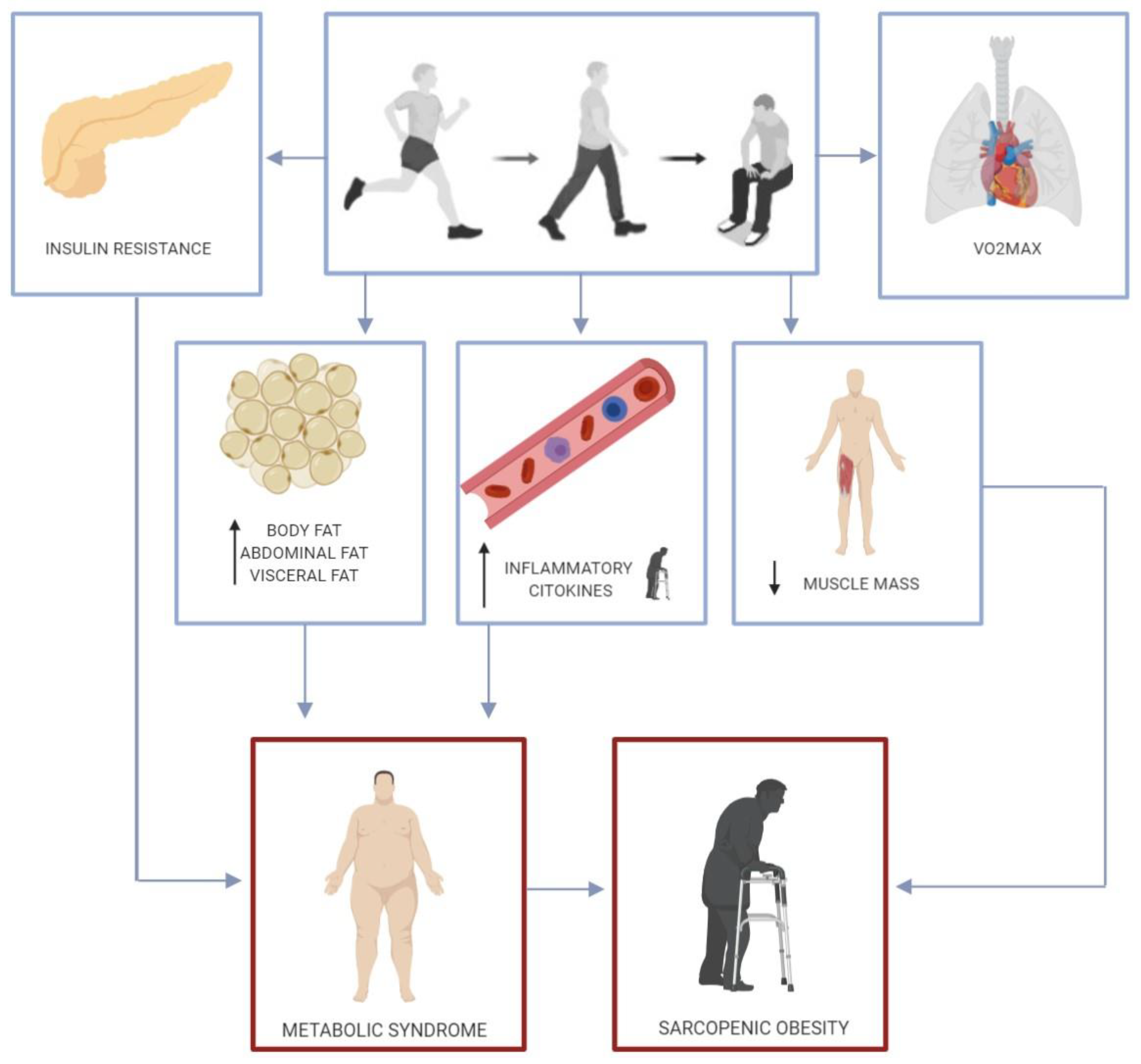
3.1. Metabolic Consequences of Acute Physical Inactivity in Healthy Adults
3.2. Metabolic Consequences of Acute Physical Inactivity in Overweight Adults
3.3. Detrimental Health Effects of an Acute Sedentary Lifestyle in the Elderly
3.4. Metabolic Effects of Acute Physical Inactivity plus Overfeeding
3.5. Manipulating Dietary Intake to Offset the Metabolic Impacts of Confinement
3.6. Physical Activity to Mitigate the Metabolic Impacts of Confinement
4. Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guthold, R.; Stevens, G.A.; Riley, L.M.; Bull, F.C. Worldwide trends in insufficient physical activity from 2001 to 2016: A pooled analysis of 358 population-based surveys with 1·9 million participants. Lancet Glob. Health 2018, 6, e1077–e1086. [Google Scholar] [CrossRef]
- Booth, F.W.; Roberts, C.K.; Laye, M.J. Lack of exercise is a major cause of chronic diseases. Compr. Physiol. 2012, 2, 1143–1211. [Google Scholar] [PubMed]
- Sanchis-Gomar, F.; Lucia, A.; Yvert, T.; Ruiz-Casado, A.; Pareja-Galeano, H.; Santos-Lozano, A.; Fiuza-Luces, C.; Garatachea, N.; Lippi, G.; Bouchard, C.; et al. Physical inactivity and low fitness deserve more attention to alter cancer risk and prognosis. Cancer Prev. Res. 2015, 8, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Biswas, A.; Oh, P.I.; Faulkner, G.E.; Bajaj, R.R.; Silver, M.A.; Mitchell, M.S.; Alter, D.A. Sedentary time and its association with risk for disease incidence, mortality, and hospitalization in adults a systematic review and meta-analysis. Ann. Intern. Med. 2015, 162, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Pareja-Galeano, H.; Sanchis-Gomar, F.; Santos-Lozano, A.; Fiuza-Luces, C.; Garatachea, N.; Ruiz-Casado, A.; Lucia, A. Regular physical activity: A little is good, but is it good enough? Am. J. Clin. Nutr. 2015, 101, 1099–1101. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pareja-Galeano, H.; Sanchis-Gomar, F.; García-Giménez, J.L. Physical exercise and epigenetic modulation: Elucidating intricate mechanisms. Sports Med. 2014, 44, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, P.L.; Morales, J.S.; Pareja-Galeano, H.; Izquierdo, M.; Emanuele, E.; de la Villa, P.; Lucia, A. Physical strategies to prevent disuse-induced functional decline in the elderly. Ageing Res. Rev. 2018, 47, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gomez, D.; Lavie, C.J.; Hamer, M.; Cabanas-Sanchez, V.; Garcia-Esquinas, E.; Pareja-Galeano, H.; Struijk, E.; Sadarangani, K.P.; Ortega, F.B.; Rodríguez-Artalejo, F. Physical activity without weight loss reduces the development of cardiovascular disease risk factors—A prospective cohort study of more than one hundred thousand adults. Prog. Cardiovasc. Dis. 2019, 62, 522–530. [Google Scholar] [CrossRef]
- Romagnoli, M.; Alis, R.; Aloe, R.; Salvagno, G.L.; Basterra, J.; Pareja-Galeano, H.; Sanchis-Gomar, F.; Lippi, G. Influence of training and a maximal exercise test in analytical variability of muscular, hepatic, and cardiovascular biochemical variables. Scand. J. Clin. Lab. Investig. 2014, 74, 192–198. [Google Scholar] [CrossRef]
- Garatachea, N.; Pareja-Galeano, H.; Sanchis-Gomar, F.; Santos-Lozano, A.; Fiuza-Luces, C.; Morán, M.; Emanuele, E.; Joyner, M.J.; Lucia, A. Exercise attenuates the major hallmarks of aging. Rejuvenation Res. 2015, 18, 57–89. [Google Scholar] [CrossRef]
- Inglés, M.; Serra-Añó, P.; Gambini, J.; Abu-Sharif, F.; Dromant, M.; Garcia-Valles, R.; Pareja-Galeano, H.; Garcia-Lucerga, C.; Gomez-Cabrera, M.C. Active paraplegics are protected against exercise-induced oxidative damage through the induction of antioxidant enzymes. Spinal Cord 2016, 54, 830–837. [Google Scholar] [CrossRef] [PubMed]
- Pareja-Galeano, H.; Brioche, T.; Sanchis-Gomar, F.; Montal, A.; Jovaní, C.; Martínez-Costa, C.; Gomez-Cabrera, M.C.; Viña, J. Impact of exercise training on neuroplasticity-related growth factors in adolescents. J. Musculoskelet. Neuronal Interact. 2013, 13, 368–371. [Google Scholar] [PubMed]
- Pareja-Galeano, H.; Garatachea, N.; Lucia, A. Exercise as a Polypill for Chronic Diseases, 1st ed.; Elsevier Inc.: Philadelphia, PA, USA, 2015; Volume 135. [Google Scholar]
- Pareja-Galeano, H.; Sanchis-Gomar, F.; Lucia, A. Physical activity and depression: Type of exercise matters. JAMA Pediatr. 2015, 169, 288–289. [Google Scholar] [CrossRef] [PubMed]
- Pareja-Galeano, H.; Mayero, S.; Perales, M.; Garatachea, N.; Santos-Lozano, A.; Fiuza-Luces, C.; Emanuele, E.; Gálvez, B.G.; Sanchis-Gomar, F.; Lucia, A. Biological Rationale for Regular Physical Exercise as an Effective Intervention for the Prevention and Treatment of Depressive Disorders. Curr. Pharm. Des. 2016, 22, 3764–3775. [Google Scholar] [CrossRef] [PubMed]
- Santos-Lozano, A.; Pareja-Galeano, H.; Sanchis-Gomar, F.; Quindós-Rubial, M.; Fiuza-Luces, C.; Cristi-Montero, C.; Emanuele, E.; Garatachea, N.; Lucia, A. Physical Activity and Alzheimer Disease: A Protective Association. Mayo Clin. Proc. 2016, 91, 999–1020. [Google Scholar] [CrossRef]
- Thyfault, J.P.; Krogh-Madsen, R. Metabolic disruptions induced by reduced ambulatory activity in free-living humans. J. Appl. Physiol. 2011, 111, 1218–1224. [Google Scholar] [CrossRef]
- López-Otín, C.; Galluzzi, L.; Freije, J.M.P.; Madeo, F.; Kroemer, G. Metabolic Control of Longevity. Cell 2016, 166, 802–821. [Google Scholar] [CrossRef]
- Olsen, R.H.; Krogh-Madsen, R.; Thomsen, C.; Booth, F.; Pedersen, B. Metabolic Responses to Reduced Daily Steps in Healthy Nonexercising Men. JAMA J. Am. Med. Assoc. 2008, 299, 1261–1263. [Google Scholar] [CrossRef]
- Krogh-Madsen, R.; Thyfault, J.P.; Broholm, C.; Mortensen, O.H.; Olsen, R.H.; Mounier, R.; Plomgaard, P.; Van Hall, G.; Booth, F.W.; Pedersen, B.K.; et al. A 2-wk reduction of ambulatory activity attenuates peripheral insulin sensitivity. J. Appl. Physiol. 2010, 108, 1034–1040. [Google Scholar] [CrossRef]
- Mikus, C.R.; Oberlin, D.J.; Libla, J.L.; Taylor, A.M.; Booth, F.W.; Thyfault, J.P. Lowering Physical Activity Impairs Glycemic Control in Healthy Volunteers. Med. Sci. Sports Exerc. 2017, 25, 1032–1057. [Google Scholar] [CrossRef]
- Knudsen, S.H.; Hansen, L.S.; Pedersen, M.; Dejgaard, T.; Hansen, J.; Van Hall, G.; Thomsen, C.; Solomon, T.P.J.; Pedersen, B.K.; Krogh-Madsen, R. Changes in insulin sensitivity precede changes in body composition during 14 days of step reduction combined with overfeeding in healthy young men. J. Appl. Physiol. 2012, 113, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Winn, N.C.; Pettit-Mee, R.; Walsh, L.K.; Restaino, R.M.; Ready, S.T.; Padilla, J.; Kanaley, J.A. Metabolic Implications of Diet and Energy Intake during Physical Inactivity. Med. Sci. Sports Exerc. 2019, 51, 995–1005. [Google Scholar] [CrossRef] [PubMed]
- Bowden Davies, K.A.; Sprung, V.S.; Norman, J.A.; Thompson, A.; Mitchell, K.L.; Halford, J.C.G.; Harrold, J.A.; Wilding, J.P.H.; Kemp, G.J.; Cuthbertson, D.J. Short-term decreased physical activity with increased sedentary behaviour causes metabolic derangements and altered body composition: Effects in individuals with and without a first-degree relative with type 2 diabetes. Diabetologia 2018, 61, 1282–1294. [Google Scholar] [CrossRef] [PubMed]
- Dixon, N.C.; Hurst, T.L.; Talbot, D.C.S.; Tyrrell, R.M.; Thompson, D. Effect of short-term reduced physical activity on cardiovascular risk factors in active lean and overweight middle-aged men. Metabolism 2013, 62, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Bowden Davies, K.A.; Sprung, V.S.; Norman, J.A.; Thompson, A.; Mitchell, K.L.; Harrold, J.A.; Finlayson, G.; Gibbons, C.; Wilding, J.P.H.; Kemp, G.J.; et al. Physical Activity and Sedentary Time: Association with Metabolic Health and Liver Fat. Med. Sci. Sports Exerc. 2019, 51, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Lavie, C.J.; Ozemek, C.; Carbone, S.; Katzmarzyk, P.T.; Blair, S.N. Sedentary Behavior, Exercise, and Cardiovascular Health. Circ. Res. 2019, 124, 799–815. [Google Scholar] [CrossRef]
- Marzetti, E.; Calvani, R.; Tosato, M.; Cesari, M.; Di Bari, M.; Cherubini, A.; Collamati, A.; D’Angelo, E.; Pahor, M.; Bernabei, R.; et al. Sarcopenia: An overview. Aging Clin. Exp. Res. 2017, 29, 11–17. [Google Scholar] [CrossRef]
- Donato, K.A. Executive summary of the clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults. Arch. Intern. Med. 1998, 158, 1855–1867. [Google Scholar]
- Breen, L.; Stokes, K.A.; Churchward-Venne, T.A.; Moore, D.R.; Baker, S.K.; Smith, K.; Atherton, P.J.; Phillips, S.M. Two weeks of reduced activity decreases leg lean mass and induces “anabolic resistance” of myofibrillar protein synthesis in healthy elderly. J. Clin. Endocrinol. Metab. 2013, 98, 2604–2612. [Google Scholar] [CrossRef]
- McGlory, C.; Von Allmen, M.T.; Stokes, T.; Morton, R.W.; Hector, A.J.; Lago, B.A.; Raphenya, A.R.; Smith, B.K.; McArthur, A.G.; Steinberg, G.R.; et al. Failed recovery of glycemic control and myofibrillar protein synthesis with 2 wk of physical inactivity in overweight, prediabetic older adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2018, 73, 1070–1077. [Google Scholar] [CrossRef]
- Walhin, J.P.; Richardson, J.D.; Betts, J.A.; Thompson, D. Exercise counteracts the effects of short-term overfeeding and reduced physical activity independent of energy imbalance in healthy young men. J. Physiol. 2013, 591, 6231–6243. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.O.; Wyatt, H.R.; Peters, J.C. The importance of energy balance. Eur. Endocrinol. 2013, 9, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Dorling, J.; Broom, D.R.; Burns, S.F.; Clayton, D.J.; Deighton, K.; James, L.J.; King, J.A.; Miyashita, M.; Thackray, A.E.; Batterham, R.L.; et al. Acute and chronic effects of exercise on appetite, energy intake, and appetite-related hormones: The modulating effect of adiposity, sex, and habitual physical activity. Nutrients 2018, 10, 1140. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, M.; Blundell, J.E. Energy balance, body composition, sedentariness and appetite regulation: Pathways to obesity. Clin. Sci. 2016, 130, 1615–1628. [Google Scholar] [CrossRef] [PubMed]
- Mayer, J.; Roy, P.; Mitra, K.P. Relation between caloric intake, body weight, and physical work: Studies in an industrial male population in West Bengal. Am. J. Clin. Nutr. 1956, 4, 169–175. [Google Scholar] [CrossRef]
- Stubbs, R.J.; Hughes, D.A.; Johnstone, A.M.; Horgan, G.W.; King, N.; Blundell, J.E. A decrease in physical activity affects appetite, energy, and nutrient balance in lean men feeding ad libitum. Am. J. Clin. Nutr. 2004, 79, 62–69. [Google Scholar] [CrossRef]
- Shook, R.P.; Hand, G.A.; Drenowatz, C.; Hebert, J.R.; Paluch, A.E.; Blundell, J.E.; Hill, J.O.; Katzmarzyk, P.T.; Church, T.S.; Blair, S.N. Low levels of physical activity are associated with dysregulation of energy intake and fat mass gain over 1 year. Am. J. Clin. Nutr. 2015, 102, 1332–1338. [Google Scholar] [CrossRef]
- Chaput, J.P.; Klingenberg, L.; Astrup, A.; Sjödin, A.M. Modern sedentary activities promote overconsumption of food in our current obesogenic environment. Obes. Rev. 2011, 12, 12–20. [Google Scholar] [CrossRef]
- Wei, M.; Brandhorst, S.; Shelehchi, M.; Mirzaei, H.; Cheng, C.W.; Budniak, J.; Groshen, S.; Mack, W.J.; Guen, E.; Di Biase, S.; et al. Fasting-mimicking diet and markers/risk factors for aging, diabetes, cancer, and cardiovascular disease. Sci. Transl. Med. 2017, 9, eaai8700. [Google Scholar] [CrossRef]
- Redman, L.M.; Smith, S.R.; Burton, J.H.; Martin, C.K.; Il’yasova, D.; Ravussin, E. Metabolic Slowing and Reduced Oxidative Damage with Sustained Caloric Restriction Support the Rate of Living and Oxidative Damage Theories of Aging. Cell Metab. 2018, 27, 805–815.e4. [Google Scholar] [CrossRef]
- Sala, V.P.; Martínez, F.D.; Biescas, A.P. Restricción calórica, un método eficaz, sencillo y saludable para perder peso. Nutr. Clin. Diet. Hosp. 2017, 37, 77–86. [Google Scholar]
- Wewege, M.A.; Thom, J.M.; Rye, K.A.; Parmenter, B.J. Aerobic, resistance or combined training: A systematic review and meta-analysis of exercise to reduce cardiovascular risk in adults with metabolic syndrome. Atherosclerosis 2018, 274, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Akins, J.D.; Crawford, C.K.; Burton, H.M.; Wolfe, A.S.; Vardarli, E.; Coyle, E.F. Inactivity induces resistance to the metabolic benefits following acute exercise. J. Appl. Physiol. 2019, 126, 1088–1094. [Google Scholar] [CrossRef] [PubMed]
- Saint-Maurice, P.F.; Troiano, R.P.; Bassett, D.R.; Graubard, B.I.; Carlson, S.A.; Shiroma, E.J.; Fulton, J.E.; Matthews, C.E. Association of Daily Step Count and Step Intensity With Mortality Among US Adults. JAMA 2020, 323, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Campbell, B.; Kreider, R.B.; Ziegenfuss, T.; La Bounty, P.; Roberts, M.; Burke, D.; Landis, J.; Lopez, H.; Antonio, J. International Society of Sports Nutrition position stand: Protein and exercise. J. Int. Soc. Sports Nutr. 2007, 4, 8. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Kerksick, C.M.; Campbell, B.I.; Cribb, P.J.; Wells, S.D.; Skwiat, T.M.; Purpura, M.; Ziegenfuss, T.N.; Ferrando, A.A.; Arent, S.M.; et al. International Society of Sports Nutrition Position Stand: Protein and exercise. J. Int. Soc. Sports Nutr. 2017, 14, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Witard, O.C.; Wardle, S.L.; Macnaughton, L.S.; Hodgson, A.B.; Tipton, K.D. Protein considerations for optimising skeletal muscle mass in healthy young and older adults. Nutrients 2016, 8, 181. [Google Scholar] [CrossRef]
- Jiménez-Pavón, D.; Carbonell-Baeza, A.; Lavie, C.J. Physical exercise as therapy to fight against the mental and physical consequences of COVID-19 quarantine: Special focus in older people. Progress in Cardiovascular Diseases. Prog. Cardiovasc. Dis. 2020, in press. [Google Scholar]
- Fallon, K. Exercise in the time of COVID-19. Aust. J. Gen. Pract. 2020, 49. [Google Scholar] [CrossRef]
- Kis, O.; Buch, A.; Stern, N.; Moran, D.S. Minimally supervised home-based resistance training and muscle function in older adults: A meta-analysis. Arch. Gerontol. Geriatr. 2019, 84, 103909. [Google Scholar] [CrossRef]
- Peterson, M.D.; Rhea, M.R.; Sen, A.; Gordon, P.M. Resistance exercise for muscular strength in older adults: A meta-analysis. Ageing Res. Rev. 2010, 9, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Fragala, M.S.; Cadore, E.L.; Dorgo, S.; Izquierdo, M.; Kraemer, W.J.; Peterson, M.D.; Ryan, E.D. Resistance Training for Older Adults: Position Statement From the National Strength and Conditioning Association. J. Strength Cond. Res. 2019, 33, 2019–2052. [Google Scholar] [CrossRef] [PubMed]
- Marques, E.A.; Mota, J.; Carvalho, J. Exercise effects on bone mineral density in older adults: A meta-analysis of randomized controlled trials. Age (Omaha) 2012, 34, 1493–1515. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J. A comprehensive review on metabolic syndrome. Cardiol. Res. Pract. 2014, 2014. [Google Scholar] [CrossRef]
- Zafar, U.; Khaliq, S.; Ahmad, H.U.; Manzoor, S.; Lone, K.P. Metabolic syndrome: An update on diagnostic criteria, pathogenesis, and genetic links. Hormones 2018, 17, 299–313. [Google Scholar] [CrossRef]
- Oikonomou, E.K.; Antoniades, C. The role of adipose tissue in cardiovascular health and disease. Nat. Rev. Cardiol. 2019, 16, 83–99. [Google Scholar] [CrossRef]
- Gustafson, B.; Hammarstedt, A.; Andersson, C.X.; Smith, U. Inflamed adipose tissue: A culprit underlying the metabolic syndrome and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 2276–2283. [Google Scholar] [CrossRef]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2005, 366, 1415–1428. [Google Scholar] [CrossRef]
- Rochlani, Y.; Naga, V.P.; Swathi, K.; Jawahar, L.M. Metabolic syndrome: Pathophysiology, management, and modulation by natural compounds. Ther. Adv. Cardiovasc. Dis. 2017, 11, 215–225. [Google Scholar] [CrossRef]
- Wannamethee, S.G.; Atkins, J.L. Muscle loss and obesity: The health implications of sarcopenia and sarcopenic obesity. Proc. Nutr. Soc. 2015, 74, 405–412. [Google Scholar] [CrossRef]
- Gupta, R.; Ghosh, A.; Singh, A.K.; Misra, A. Clinical considerations for patients with diabetes in times of COVID-19 epidemic. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 211–212. [Google Scholar] [CrossRef] [PubMed]
- Bornstein, S.R.; Dalan, R.; Hopkins, D.; Mingrone, G.; Boehm, B.O. Endocrine and metabolic link to coronavirus infection. Nat. Rev. Endocrinol. 2020, 16, 297–298. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Yang, J.; Zhao, F.; Zhi, L.; Wang, X.; Liu, L.; Bi, Z.; Zhao, Y. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin. Res. Cardiol. 2020, 109, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Dietz, W.; Santos-Burgoa, C. Obesity and its Implications for COVID-19 Mortality. Obesity 2020. [Google Scholar] [CrossRef]
- Ryan, D.H.; Ravussin, E.; Heymsfield, S. COVID 19 and the Patient with Obesity—The Editors Speak Out. Obesity 2020. [Google Scholar] [CrossRef]
| Study | Subjects | Intervention | Blood Parameters | Inflammatory Parameters | Lipid Profile | Body Composition | CRF |
|---|---|---|---|---|---|---|---|
| Olsen et al. 2014 [19] | 8 healthy men <2 h EX/week, >3500 steps/day 27.1 (5.7) years BMI 22.9 (4.0) kg/m2 | 3-weeks SR 6203 (5135–7271) to 1394 (1261–1528) steps/day Maintained dietary habits | OGTT ↑ insulin AUC | Not reported | Not reported | Not reported | Not reported |
| 10 healthy men <2 h EX/week, >3500 steps/day 23.8 (4.6) years BMI 22.1 (2.1) kg/m2 | 2-week SR 10,501 (8755–12,247) to 1344 (1272–1416) steps/day Maintained dietary habits | OGTT ↑ insulin AUC, C-peptide levels OFTT ↑ insulin, C-peptide, TG | Not reported | Not reported |  FM FM↑ intra-abdominal fat ↓ fat free mass | Not reported | |
| Krogh-Madsen et al. 2010 [20] | 10 healthy men <2 h EX/week, >3500 steps/day 23.8 (1.5) years BMI 22.1 (0.7) kg/m2 | 2-week SR 10,501 (808) to 1344 (33) Maintained dietary habits | ↓ GIR during the clamp ↓ peripheral insulin sensitivity (H-E) ↓ insulin provoked muscle Akt phosphorylation  hepatic glucose production, plasma glucose, insulin, C-peptide hepatic glucose production, plasma glucose, insulin, C-peptide |  TNF, IL-6, IL-15, adiponectin, leptin TNF, IL-6, IL-15, adiponectin, leptin |  TG, FFA TG, FFA |  FM FM↓ BM, leg lean mass  trunk and arm lean mass trunk and arm lean mass | ↓ VO2max |
| Mikus et al. 2012 [21] | 12 healthy participants (4 F, 8 M) >10000 steps/day 29 (1) years BMI 23.6 (0.9) kg/m2 | 3-day SR 12,956 (769) to 4319 (256) steps/day Maintained dietary habits | CGM ↑ post-prandial glucose, fasting insulin  pre-meal blood glucose, 24 h average glucose pre-meal blood glucose, 24 h average glucoseOGTT ↑ insulin AUC, C-peptide, HOMA-IR  glucose AUC glucose AUC↓ Matsuda | Not reported | Not reported | Not reported | Not reported |
| Dixon et al. 2013 [25] | 18 healthy men EX > 30 min/ 5 d/week 9 overweight: 49 (1.0) years BMI 29.3 (1.2) kg/m2 9 lean men: 51.5 (1.4) years BMI 29.3 (1.2) kg/m2 | 1-week SR <4000 steps/day Maintained dietary habits | OGTT ↑ insulin AUC, glucose AUC Overweight: >insulin AUC, glucose AUC |  CRP, IL-6, TNF-α, WBC, sICAM, ALT CRP, IL-6, TNF-α, WBC, sICAM, ALTOverweigh: >CRP, ALT | ↑ TG FFA, TC, HDL-c, LDL-c FFA, TC, HDL-c, LDL-cOverweight: >FFA, TG | Not reported | Not reported |
| Bowden-Davies et al. 2018 [24] | 45 healthy participants (28 F, 17 M) >10,000 steps/day, >2 h EX/week 16 FDR+ve: 40(14) yearsBMI 27 (5) kg/m2 29 FDR-ve: 33 (13) years BMI 24 (3) kg/m2 | 2-week SR <1500 steps/day (mean decreased: 10,285) Maintained dietary habits | OGTT ↑ insulin AUC, glucose AUC ↓ Matsuda, muscle insulin sensitivity  hepatic insulin resistance, NEFA AUC hepatic insulin resistance, NEFA AUCFDR+ve: <insulin sensitivity (after SR and resuming of PA) | Not reported | ↑ TC, LDL-c, TG | ↑ FM. liver fat ↓ total lean mass, lower limb lean mass  arm lean mass arm lean massFDR+ve: >increase of android fat | ↓ VO2max |
| 2-week resumed usual PA | |||||||
| Knudsen et al. 2012 [22] | 9 healthy men >10,000 steps/day 24.3 (3.3) years BMI 21.6 (2.5) kg/m2 | 2-week SR + overfeeding 10,278 (2399) to 1521 (488) steps/day 2762 (299) kcal to 4197 (290) kcal | OGTT ↑ insulin AUC (day 7, day 14), Matsuda (day 3, day 7)  glucose AUC glucose AUC↓ Peripheral insulin sensitivity (H-E)  plasma glucose, C-peptide, hepatic glucose production plasma glucose, C-peptide, hepatic glucose production |  TNF-α, IL-6 TNF-α, IL-6↑ leptin, adiponectin |  FFA, TG FFA, TG | ↑ BM, FM, android and visceral fat FFM FFM | ↓ VO2max |
| 16-day resumed usual PA | Returned to baseline | Remained elevated | Returned to baseline | ||||
| Winn et al. 2019 [23] | 10 healthy participants (4 F, 6 M) >90 min PA 3 days/week and >10,000 steps/day 24 (1) years BMI < 28 kg/m2 | 10-day SR (>10,000 to <50,000 step/day) + control diet (400 kcal/day deficit) 4-week washout 10-day SR + higher protein diet (400 kcal/day deficit) | OGTT Postprandial glucose, insulin, NEFA, 2-h glucose and 2-h insulin, C-peptide, hepatic insulin extraction, plasma glucose and insulin Postprandial glucose, insulin, NEFA, 2-h glucose and 2-h insulin, C-peptide, hepatic insulin extraction, plasma glucose and insulin | Not reported |  TG, LDL-c, oxidized LDL TG, LDL-c, oxidized LDL↓ HDL-C, TC | ↓ BM abdominal FM FFM, FM FFM, FM | ↓ VO2max |
| 10-day SR + overfeeding (880 kcal/d) | OGTT ↑ HOMA-IR, 2h-glucose, 2h-insulin  glucose AUC, NEFA glucose AUC, NEFA↑ plasma glucose, insulin, C-peptide  hepatic insulin extraction hepatic insulin extraction | Not reported |  TG, TC, HDL-c, LDL-c, oxidized LDL TG, TC, HDL-c, LDL-c, oxidized LDL | ↑ BM, FM FFM FFM | ↓ VO2max | ||
| Walhin et al. 2013 [26] | 26 healthy men Vigorous-intensity EX > 30 min/ 3 day/week 25 (7) years BMI 23.8 (2.5) | 1-week SR: 12,562 (3520) to 3762 (860) + overfeeding (+50% kcal) + 45 min/day treadmill running at 70% VO2max | OGTT Matsuda, B- cell function, insulin AUC Matsuda, B- cell function, insulin AUC |  ALT, CRP, IL-6, WBC ALT, CRP, IL-6, WBC |  HDL-C. LDL-C, NEFAs, TG, CT HDL-C. LDL-C, NEFAs, TG, CT | ↑ BM, waist/hip circumference, lean mass FM FM | Not reported |
| 1-week SR: 10,544 (2756) to 3690 (400) + overfeeding (+50% kcal) + not training | ↓ Matsuda, B- cell function ↑ insulin AUC OGTT |  ALT, CRP, IL-6 ALT, CRP, IL-6↑ adiponectin, WBC | ↑ CT HDL-C. LDL-C, NEFAs, TG HDL-C. LDL-C, NEFAs, TG | ↑ BM, waist/hip circumference, lean mass FM FM | Not reported | ||
| Breen et al. 2013 [27] | 10 healthy older adults (5 F, 5 M) >3500 steps/day 72.3 (1.0) years; BMI 29.0 (1.8) kg/m2 | 2-week SR: 5962 (695) to 1413 (110) steps/day Maintained dietary habits | OGTT ↑ HOMA-IR, glucose AUC and AUC ↓ Matsuda  C-peptide AUC C-peptide AUC | ↑ TNF-α, CRP IL-6 IL-6 | Not reported |  total FM, FFM total FM, FFM↓ leg FFM ↑ % FM ↓ MPS  isometric MVC, SPPB isometric MVC, SPPB | Not reported |
| McGlory et al. 2018 [28] | 22 healthy older adults >3500 steps/day 12 M: 69 (3) years; BMI 27.3 (4.6) 10 F: 70 (5]) years; BMI 27.7 (5.1) | 2-week SR: 7362 (3294) to 991 (97) steps/day Maintained dietary habits 2-week resumed usual PA | OGTT ↑ glucose and insulin AUC, HOMA-IR ↓ Matsuda | ↑ TNF-α, CRP, IL-6 | Not reported |  BMI, %total FM, lean mass BMI, %total FM, lean mass↓ MPS  isometric MVC isometric MVC | Not reported |
 no significant change, ↑ significant increase, ↓ significant decrease, > significantly higher, < significantly lower.
no significant change, ↑ significant increase, ↓ significant decrease, > significantly higher, < significantly lower.© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martinez-Ferran, M.; de la Guía-Galipienso, F.; Sanchis-Gomar, F.; Pareja-Galeano, H. Metabolic Impacts of Confinement during the COVID-19 Pandemic Due to Modified Diet and Physical Activity Habits. Nutrients 2020, 12, 1549. https://doi.org/10.3390/nu12061549
Martinez-Ferran M, de la Guía-Galipienso F, Sanchis-Gomar F, Pareja-Galeano H. Metabolic Impacts of Confinement during the COVID-19 Pandemic Due to Modified Diet and Physical Activity Habits. Nutrients. 2020; 12(6):1549. https://doi.org/10.3390/nu12061549
Chicago/Turabian StyleMartinez-Ferran, María, Fernando de la Guía-Galipienso, Fabián Sanchis-Gomar, and Helios Pareja-Galeano. 2020. "Metabolic Impacts of Confinement during the COVID-19 Pandemic Due to Modified Diet and Physical Activity Habits" Nutrients 12, no. 6: 1549. https://doi.org/10.3390/nu12061549
APA StyleMartinez-Ferran, M., de la Guía-Galipienso, F., Sanchis-Gomar, F., & Pareja-Galeano, H. (2020). Metabolic Impacts of Confinement during the COVID-19 Pandemic Due to Modified Diet and Physical Activity Habits. Nutrients, 12(6), 1549. https://doi.org/10.3390/nu12061549






