Effect of a Combination of Citrus Flavones and Flavanones and Olive Polyphenols for the Reduction of Cardiovascular Disease Risk: An Exploratory Randomized, Double-Blind, Placebo-Controlled Study in Healthy Subjects
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
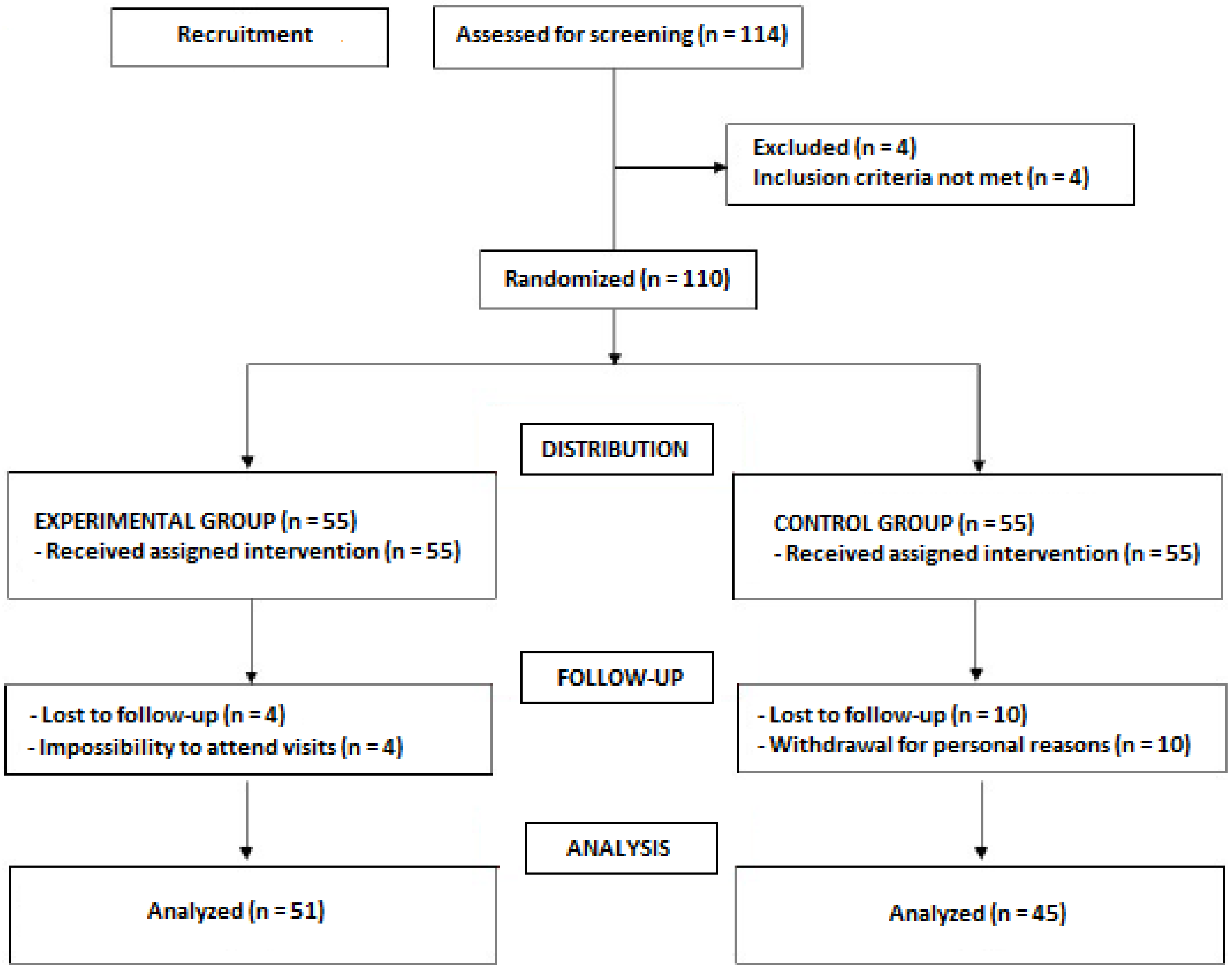
2.2. Participant Selection and Randomization
2.3. Intervention and Study Procedures
2.4. Study Variables
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart Disease and Stroke Statistics—2019 Update: A Report From the American Heart Association. Circulation 2019, 139, e56–e528. [Google Scholar] [CrossRef] [PubMed]
- European Heart Network. European Cardiovascular Disease Statistics 2017. Available online: http://www.ehnheart.org/cvd-statistics.html (accessed on 22 October 2019).
- Lopez, A.D.; Adair, T. Is the long-term decline in cardiovascular-disease mortality in high-income countries over? Evidence from national vital statistics. Int. J. Epidemiol. 2019, 48, 1815–1823. [Google Scholar] [CrossRef] [PubMed]
- Mc Namara, K.; Alzubaidi, H.; Jackson, J.K. Cardiovascular disease as a leading cause of death: How are pharmacists getting involved? Integr. Pharm. Res. Pract. 2019, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, T.; Davey, R.; Gidlow, C.J.; Iqbal, Z.; Kumar, J.; Mawby, Y.; Chambers, R. Contribution of Individual Risk Factor Changes to Reductions in Population Absolute Cardiovascular Risk. BioMed Res. Int. 2014, 2014, 626205. [Google Scholar] [CrossRef]
- Tzoulaki, I.; Elliott, P.; Kontis, V.; Ezzati, M. Mbbs Worldwide Exposures to Cardiovascular Risk Factors and Associated Health Effects. Circulation 2016, 133, 2314–2333. [Google Scholar] [CrossRef]
- Barnes, A.S. Emerging Modifiable Risk Factors for Cardiovascular Disease in Women. Tex. Heart Inst. J. 2013, 40, 293–295. [Google Scholar]
- Yusuf, S.; Joseph, P.; Rangarajan, S.; Islam, S.; Mente, A.; Hystad, P.; Brauer, M.; Kutty, V.R.; Gupta, R.; Wielgosz, A.; et al. Modifiable risk factors, cardiovascular disease, and mortality in 155 722 individuals from 21 high-income, middle-income, and low-income countries (PURE): A prospective cohort study. Lancet 2020, 395, 795–808. [Google Scholar] [CrossRef]
- He, F.J.; Nowson, C.; Lucas, M.; A MacGregor, G. Increased consumption of fruit and vegetables is related to a reduced risk of coronary heart disease: Meta-analysis of cohort studies. J. Hum. Hypertens. 2007, 21, 717–728. [Google Scholar] [CrossRef]
- He, F.J.; Nowson, C.; A MacGregor, G. Fruit and vegetable consumption and stroke: Meta-analysis of cohort studies. Lancet 2006, 367, 320–326. [Google Scholar] [CrossRef]
- Afshin, A.; Micha, R.; Khatibzadeh, S.; Mozaffarian, D. Consumption of nuts and legumes and risk of incident ischemic heart disease, stroke, and diabetes: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 278–288. [Google Scholar] [CrossRef]
- Mellen, P.B.; Walsh, T.F.; Herrington, D.M. Whole grain intake and cardiovascular disease: A meta-analysis. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Micha, R.; Wallace, S.K.; Mozaffarian, D. Red and processed meat consumption and risk of incident coronary heart disease, stroke, and diabetes mellitus: A systematic review and meta-analysis. Circulation 2010, 121, 2271–2283. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.J.; Mente, A.; Maroleanu, A.; I Cozma, A.; Ha, V.; Kishibe, T.; Uleryk, E.; Budylowski, P.; Schünemann, H.; Beyene, J.; et al. Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: Systematic review and meta-analysis of observational studies. BMJ 2015, 351, h3978. [Google Scholar] [CrossRef]
- Strazzullo, P.; D’Elia, L.; Kandala, N.-B.; Cappuccio, F.P. Salt intake, stroke, and cardiovascular disease: Meta-analysis of prospective studies. BMJ 2009, 339, b4567. [Google Scholar] [CrossRef] [PubMed]
- Eilat-Adar, S.; Sinai, T.; Yosefy, C.; Henkin, Y. Nutritional Recommendations for Cardiovascular Disease Prevention. Nutrients 2013, 5, 3646–3683. [Google Scholar] [CrossRef] [PubMed]
- Levy, L.B. Dietary strategies, policy and cardiovascular disease risk reduction in England. Proc. Nutr. Soc. 2013, 72, 386–389. [Google Scholar] [CrossRef]
- Calabrese, I.; Riccardi, G. Effectiveness of Changes in Diet Composition on Reducing the Incidence of Cardiovascular Disease. Curr. Cardiol. Rep. 2019, 21, 88. [Google Scholar] [CrossRef]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 140, e596–e646. [Google Scholar] [CrossRef]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA. Guideline on the management of blood cholesterol. Circulation 2019, 139, e1082–e1143. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Atherosclerosis 2019, 290, 140–205. [Google Scholar] [CrossRef]
- Vogiatzoglou, A.; Mulligan, A.A.; Lentjes, M.; Luben, R.; Spencer, J.P.E.; Schroeter, H.; Khaw, K.-T.; Kuhnle, G.G. Flavonoid Intake in European Adults (18 to 64 Years). PLoS ONE 2015, 10, e0128132. [Google Scholar] [CrossRef] [PubMed]
- Vogiatzoglou, A.; Heuer, T.; Mulligan, A.; Lentjes, M.; Luben, R.N.; Kuhnle, G.G. Estimated dietary intakes and sources of flavanols in the German population (German National Nutrition Survey II). Eur. J. Nutr. 2013, 53, 635–643. [Google Scholar] [CrossRef] [PubMed]
- Peyrol, J.; Riva, C.; Amiot, M.J. Hydroxytyrosol in the Prevention of the Metabolic Syndrome and Related Disorders. Nutrients 2017, 9, 306. [Google Scholar] [CrossRef] [PubMed]
- Ali, F.; Rahul; Naz, F.; Jyoti, S.; Siddique, Y.H. Health functionality of apigenin: A review. Int. J. Food Prop. 2016, 20, 1197–1238. [Google Scholar] [CrossRef]
- Salehi, B.; Venditti, A.; Sharifi-Rad, J.; Kregiel, D.; Sharifi-Rad, J.; Durazzo, A.; Lucarini, M.; Santini, A.; Souto, E.; Novellino, E.; et al. The Therapeutic Potential of Apigenin. Int. J. Mol. Sci. 2019, 20, 1305. [Google Scholar] [CrossRef] [PubMed]
- Benavente-García, O.; Castillo, J. Update on Uses and Properties of Citrus Flavonoids: New Findings in Anticancer, Cardiovascular, and Anti-inflammatory Activity. J. Agric. Food Chem. 2008, 56, 6185–6205. [Google Scholar] [CrossRef]
- Ortuño, A.; Benavente-García, O.; Castillo, J.; Alcaraz, M.; Vicente, V.; Del Rio, J. Beneficial Action of Citrus Flavonoids on Multiple Cancer-Related Biological Pathways. Curr. Cancer Drug Targets 2007, 7, 795–809. [Google Scholar] [CrossRef]
- Balez, R.; Steiner, N.; Engel, M.; Muñoz, S.S.; Lum, J.; Wu, Y.; Wang, D.; Vallotton, P.; Sachdev, P.S.; O’Connor, M.; et al. Neuroprotective effects of apigenin against inflammation, neuronal excitability and apoptosis in an induced pluripotent stem cell model of Alzheimer’s disease. Sci. Rep. 2016, 6, 31450. [Google Scholar] [CrossRef]
- Paredes, M.D.; Romecín, P.; Atucha, N.M.; O’Valle, F.; Castillo, J.; Ortiz-Ruiz, C.; Garcia-Estañ, J. Moderate Effect of Flavonoids on Vascular and Renal Function in Spontaneously Hypertensive Rats. Nutrients 2018, 10, 1107. [Google Scholar] [CrossRef]
- Paredes, M.D.; Romecín, P.; Atucha, N.M.; O’Valle, F.; Castillo, J.; Ortiz-Ruiz, C.; Garcia-Estañ, J. Beneficial Effects of Different Flavonoids on Vascular and Renal Function in L-NAME Hypertensive Rats. Nutrients 2018, 10, 484. [Google Scholar] [CrossRef]
- Merola, N.; Castillo, J.; Benavente-García, O.; Ros-Berruezo, G.; Nieto, G. The Effect of Consumption of Citrus Fruit and Olive Leaf Extract on Lipid Metabolism. Nutrients 2017, 9, 1062. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, J.A.; Lozano, M.L.; Castillo, J.; Benavente-García, O.; Vicente, V.; Rivera, J. Flavonoids inhibit platelet function through binding to the thromboxane A2 receptor. J. Thromb. Haemost. 2005, 3, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Benavente-García, O.; Castillo, J.; Marín, F.R.; Ortuño, A.; Del Río, J.A. Uses and Properties ofCitrusFlavonoids. J. Agric. Food Chem. 1997, 45, 4505–4515. [Google Scholar] [CrossRef]
- López-Carreras, N.; Castillo, J.; Mugureza, B.; Aleixandre, A. Endothelium-dependent vascular relaxing effects of different citrus and olive extracts in aorta rings from spontaneously hypertensive rats. Food Res. Int. 2015, 77, 484–490. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Lockyer, S.; Rowland, I.; Spencer, J.P.E.; Yaqoob, P.; Stonehouse, W. Impact of phenolic-rich olive leaf extract on blood pressure, plasma lipids and inflammatory markers: A randomized controlled trial. Eur. J. Nutr. 2017, 56, 1421–1432. [Google Scholar] [CrossRef]
- Hermans, N.; Van Der Auwera, A.; Breynaert, A.; Verlaet, A.; De Bruyne, T.; Van Gaal, L.; Pieters, L.; Verhoeven, V. A red yeast rice-olive extract supplement reduces biomarkers of oxidative stress, OxLDL and Lp-PLA2, in subjects with metabolic syndrome: A randomised, double-blind, placebo-controlled trial. Trials 2017, 18, 302. [Google Scholar] [CrossRef]
- Colica, C.; Di Renzo, L.; Trombetta, D.; Smeriglio, A.; Bernardini, S.; Cioccoloni, G.; De Miranda, R.C.; Gualtieri, P.; Salimei, P.S.; De Lorenzo, A. Antioxidant Effects of a Hydroxytyrosol-Based Pharmaceutical Formulation on Body Composition, Metabolic State, and Gene Expression: A Randomized Double-Blinded, Placebo-Controlled Crossover Trial. Oxid. Med. Cell. Longev. 2017, 2017, 2473495. [Google Scholar] [CrossRef]
- Ponzo, V.; Goitre, I.; Fadda, M.; Gambino, R.; De Francesco, A.; Soldati, L.; Gentile, L.; Magistroni, P.; Cassader, M.; Bo, S. Dietary flavonoid intake and cardiovascular risk: A population-based cohort study. J. Transl. Med. 2015, 13, 218. [Google Scholar] [CrossRef]
- Peterson, J.J.; Dwyer, J.; Jacques, P.F.; McCullough, M.L. Associations between flavonoids and cardiovascular disease incidence or mortality in European and US populations. Nutr. Rev. 2012, 70, 491–508. [Google Scholar] [CrossRef]
- Rees, A.; Dodd, G.F.; Spencer, J.P. The Effects of Flavonoids on Cardiovascular Health: A Review of Human Intervention Trials and Implications for Cerebrovascular Function. Nutrients 2018, 10, 1852. [Google Scholar] [CrossRef] [PubMed]
- Huffman, M.D.; Salam, A.; Patel, A. Implementation Strategies for Cardiovascular Polypills. JAMA 2019, 322, 2279. [Google Scholar] [CrossRef] [PubMed]
- Corretti, M.C.; Anderson, T.J.; Benjamin, E.J.; Celermajer, D.; Charbonneau, F.; A Creager, M.; Deanfield, J.; Drexler, H.; Gerhard-Herman, M.; Herrington, D.; et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: A report of the International Brachial Artery Reactivity Task Force. J. Am. Coll. Cardiol. 2002, 39, 257–265. [Google Scholar] [CrossRef]
- Celermajer, D.; Sorensen, K.; Gooch, V.; Spiegelhalter, D.; Miller, O.; Sullivan, I.; Lloyd, J.; E Deanfield, J. Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet 1992, 340, 1111–1115. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Rosei, E.A.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.F.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Alonso, J.; Regidor, E.; Barrio, G.; Prieto, L.; Rodríguez, C.; de la Fuente, L. Valores poblacionales de referencia de la versión española del Cuestionario de Salud SF-36. Med. Clin. 1998, 111, 410–416. [Google Scholar]
- World Health Organization. Global Physical Activity Questionnaire (GPAQ). Analysis Guide. Available online: https://www.who.int/ncds/surveillance/steps/resources/GPAQ_Analysis_Guide.pdf (accessed on 22 March 2020).
- Widmer, R.J.; Lerman, A. Endothelial dysfunction and cardiovascular disease. Glob. Cardiol. Sci. Pract. 2014, 2014, 43. [Google Scholar] [CrossRef]
- Vita, J.A.; Keaney, J.F., Jr. Endothelial function: A barometer for cardiovascular risk? Circulation 2002, 106, 640–642. [Google Scholar] [CrossRef] [PubMed]
- Reriani, M.K.; O Lerman, L.; Lerman, A. Endothelial function as a functional expression of cardiovascular risk factors. Biomark. Med. 2010, 4, 351–360. [Google Scholar] [CrossRef]
- Oyama, J.-I.; Maeda, T.; Kouzuma, K.; Ochiai, R.; Tokimitsu, I.; Higuchi, Y.; Sugano, M.; Makino, N. Green tea catechins improve human forearm endothelial dysfunction and have antiatherosclerotic effects in smokers. Circ. J. 2010, 74, 578–588. [Google Scholar] [CrossRef]
- Noad, R.L.; Rooney, C.; McCall, D.; Young, I.S.; McCance, D.; McKinley, M.C.; Woodside, J.; McKeown, P.P. Beneficial effect of a polyphenol-rich diet on cardiovascular risk: A randomised control trial. Heart 2016, 102, 1371–1379. [Google Scholar] [CrossRef]
- Greyling, A.; Wolters, T.L.C.; De Bresser, D.M.; Roerink, S.H.; Riksen, N.P.; Mulder, T.; Rowson, M.J.; Hopman, M.T.; Thijssen, D.H. The acute effect of black tea consumption on resistance artery endothelial function in healthy subjects. A randomized controlled trial. Clin. Nutr. ESPEN 2018, 23, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Kirch, N.; Berk, L.; Liegl, Y.; Adelsbach, M.; Zimmermann, B.F.; Stehle, P.; Stoffel-Wagner, B.; Ludwig, N.; Schieber, A.; Helfrich, H.-P.; et al. A nutritive dose of pure (–)-epicatechin does not beneficially affect increased cardiometabolic risk factors in overweight-to-obese adults—a randomized, placebo-controlled, double-blind crossover study. Am. J. Clin. Nutr. 2018, 107, 948–956. [Google Scholar] [CrossRef]
- Hollands, W.J.; Tapp, H.; Defernez, M.; Moral, N.P.; Winterbone, M.S.; Philo, M.; Lucey, A.; E Kiely, M.; Kroon, P.A. Lack of acute or chronic effects of epicatechin-rich and procyanidin-rich apple extracts on blood pressure and cardiometabolic biomarkers in adults with moderately elevated blood pressure: A randomized, placebo-controlled crossover trial. Am. J. Clin. Nutr. 2018, 108, 1006–1014. [Google Scholar] [CrossRef] [PubMed]
- George, E.; Marshall, S.; Mayr, H.L.; Trakman, G.L.; Tatucu, O.; Lassemillante, A.-C.M.; Bramley, A.; Reddy, A.J.; Forsyth, A.; Tierney, A.C.; et al. The effect of high-polyphenol extra virgin olive oil on cardiovascular risk factors: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 59, 2772–2795. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Krause, M.; Schmucker, C.; Hoffmann, G.; Rücker, G.; Meerpohl, J.J. Impact of different types of olive oil on cardiovascular risk factors: A systematic review and network meta-analysis. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1030–1039. [Google Scholar] [CrossRef]
- Rees, K.; Takeda, A.; Martin, N.; Ellis, L.; Wijesekara, D.; Vepa, A.; Das, A.; Hartley, L.; Stranges, S. Mediterranean-style diet for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2019, 3, CD009825. [Google Scholar] [CrossRef]
- Davinelli, S.; Bertoglio, J.C.; Zarrelli, A.; Pina, R.; Scapagnini, G. A Randomized Clinical Trial Evaluating the Efficacy of an Anthocyanin–Maqui Berry Extract (Delphinol®) on Oxidative Stress Biomarkers. J. Am. Coll. Nutr. 2015, 34, 28–33. [Google Scholar] [CrossRef]
- Valls, R.M.; Llauradó, E.; Fernández-Castillejo, S.; Puiggrós, F.; Solà, R.; Arola, L.; Pedret, A. Effects of low molecular weight procyanidin rich extract from french maritime pine bark on cardiovascular disease risk factors in stage-1 hypertensive subjects: Randomized, double-blind, crossover, placebo-controlled intervention trial. Phytomedicine 2016, 23, 1451–1461. [Google Scholar] [CrossRef]
- Verhoeven, V.; Van Der Auwera, A.; Van Gaal, L.; Remmen, R.; Apers, S.; Stalpaert, M.; Wens, J.; Hermans, N. Can red yeast rice and olive extract improve lipid profile and cardiovascular risk in metabolic syndrome? A double blind, placebo controlled randomized trial. BMC Complement Altern. Med. 2015, 15, 52. [Google Scholar] [CrossRef]
- Conterno, L.; Martinelli, F.; Tamburini, M.; Fava, F.; Mancini, A.; Sordo, M.; Pindo, M.; Martens, S.; Masuero, D.; Vrhovsek, U.; et al. Measuring the impact of olive pomace enriched biscuits on the gut microbiota and its metabolic activity in mildly hypercholesterolaemic subjects. Eur. J. Nutr. 2017, 58, 63–81. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.; Nallasamy, P.; Liu, D.; Shah, H.; Li, J.Z.; Chitrakar, R.; Si, H.; McCormick, J.; Zhu, H.; Zhen, W.; et al. Luteolin protects against vascular inflammation in mice and TNF-alpha-induced monocyte adhesion to endothelial cells via suppressing IΚBα/NF-κB signaling pathway. J. Nutr. Biochem. 2014, 26, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.-H.; Hung, L.-M.; Chen, Y.-H.; Kuan, Y.-H.; Zhang, F.B.-Y.; Lin, R.-H.; Shih, H.-C.; Tsai, S.-K.; Huang, S.S. Cardioprotective effects of luteolin during ischemia-reperfusion injury in rats. Circ. J. 2010, 75, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Shen, H.; Zang, L.; Su, Z.; Huang, J.; Sun, Y.; Wang, H. Protective effect of luteolin on skin ischemia-reperfusion injury through an AKT-dependent mechanism. Int. J. Mol. Med. 2018, 42, 3073–3082. [Google Scholar] [CrossRef] [PubMed]
- Atkin, M.; Laight, D.; Cummings, M.H. The effects of garlic extract upon endothelial function, vascular inflammation, oxidative stress and insulin resistance in adults with type 2 diabetes at high cardiovascular risk. A pilot double blind randomized placebo controlled trial. J. Diabetes Complicat. 2016, 30, 723–727. [Google Scholar] [CrossRef]
- Kar, P.; Laight, D.; Rooprai, H.K.; Shaw, K.M.; Cummings, M. Effects of grape seed extract in Type 2 diabetic subjects at high cardiovascular risk: A double blind randomized placebo controlled trial examining metabolic markers, vascular tone, inflammation, oxidative stress and insulin sensitivity. Diabet. Med. 2009, 26, 526–531. [Google Scholar] [CrossRef]
- Bacchiega, B.C.; Bacchiega, A.B.; Usnayo, M.J.G.; Bedirian, R.; Singh, G.; Pinheiro, G.D.R.C. Interleukin 6 Inhibition and Coronary Artery Disease in a High-Risk Population: A Prospective Community-Based Clinical Study. J. Am. Heart Assoc. 2017, 6, 005038. [Google Scholar] [CrossRef]
- IL6R Genetics Consortium Emerging Risk Factors Collaboration; Sarwar, N.; Butterworth, A.S.; Freitag, D.F.; Gregson, J.; Willeit, P.; Gorman, N.N.; Gao, P.; Saleheen, D.; Rendon, A. Interleukin-6 receptor pathways in coronary heart disease: A collaborative meta-analysis of 82 studies. Lancet 2012, 379, 1205–1213. [Google Scholar] [CrossRef]
- Cesari, M. Inflammatory Markers and Onset of Cardiovascular Events: Results from the Health ABC Study. Circulation 2003, 108, 2317–2322. [Google Scholar] [CrossRef]
- Ginwala, R.; Bhavsar, R.; Chigbu, D.G.I.; Khan, Z.K.; Khan, Z.K. Potential Role of Flavonoids in Treating Chronic Inflammatory Diseases with a Special Focus on the Anti-Inflammatory Activity of Apigenin. Antioxidants 2019, 8, 35. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, G.; Gurley, E.C.; Zhou, H. Flavonoid Apigenin Inhibits Lipopolysaccharide-Induced Inflammatory Response through Multiple Mechanisms in Macrophages. PLoS ONE 2014, 9, e107072. [Google Scholar] [CrossRef] [PubMed]
| Variables | Study Groups | Between-Group p-Value (F Snedecor) | |||||
|---|---|---|---|---|---|---|---|
| Active Group (n = 51) | Placebo Group (n = 45) | ||||||
| Baseline (1) ± SD | 8-Week (2) ± SD | Δ 2-1 (95% CI) p-Value | Baseline(1) ± SD | 8-Week (2) ± SD | Δ 2-1 (95% CI) p-Value | ||
| Flow mediated vasodilation (FMD), % | 6.73 ± 5.27 | 9.77 ± 5.56 | 3.04 (1.448 to 4.617) <0.001 | 6.85 ± 4.56 | 7.39 ± 5.81 | 0.530 (−1.237 to 2.297) 0.636 | 0.039 (F = 4.39) |
| Blood pressure, mm Hg | |||||||
| Systolic | 123.6 ± 13.8 | 118.7 ± 11.9 | −4.89 (−7.7 to −2.06) 0.001 | 122.8 ± 8.8 | 120.8 ± 11.6 | −1.98 (−5.04 to 1.09) 0.203 | 0.168 (F = 1.94) |
| Diastolic | 84.2 ± 9.2 | 81.7 ± 8.9 | −2.49 (−4.88 to −0.01) 0.041 | 79.7 ± 8.3 | 80.8 ± 7.8 | 1.12 (−1.46 to 3.70) 0.392 | 0.045 (F = 4.15) |
| Laboratory tests | |||||||
| Total cholesterol, mg/dL | 242.2 ± 31.9 | 228.6 ± 29.6 | −13.57 (−21.09 to −6.05) 0.001 | 235.7 ± 38.9 | 242.0 ± 35.6 | 6.38 (−1.63 to 14.38) 0.117 | 0.001 (F = 12.99) |
| LDL-C, mg/dL | 144.2 ± 28.1 | 135.4 ± 26.6 | −8.80 (−14.49 to −3.12) 0.003 | 138.8 ± 31.3 | 139.9 ± 28.0 | 1.11 (−4.94 to 7.16) 0.717 | 0.02 (F = 5.62) |
| HDL-C, mg/dL | 63.6 ± 12.8 | 65.3 ± 13.1 | 1.65 (−0.82 to 4.11) 0.188 | 63.2 ± 15.8 | 62.1 ± 16.4 | −1.09 (−3.71 to 1.54) 0.412 | 0.135 (F = 2.28) |
| Triglycerides, mg/dL | 115.1 ± 54.0 | 115.5 ± 67.1 | 0.471 (−14.73 to 15.67) 0.951 | 118.9 ± 55.1 | 125.0 ± 74.1 | 6.02 (−10.16 to 27.20) 0.462 | 0.621 (F = 0.25) |
| Von Willebrand factor, % | 120.9 ± 45.9 | 119.9 ± 36.7 | 0.833 −1.08 (−11.28 to 9.11) | 119.5 ± 50.4 | 124.4 ± 46.0 | 4.95 (−6.04 to 15.94) 0.374 | 0.426 (F = 0.64) |
| LDL-oxidase, pg/mL | 810.3 ± 555.4 | 467.5 ± 239.6 | −342.84 (−459.68 to −226.00) <0.001 | 656.6 ± 402.3 | 692.2 ± 363.9 | 35.66 (−88.72 to 160.05) 0.571 | <0.001 (F = 19.39) |
| Oxidized Glutathione (GSSH), nmol/mg protein | 0.47 ± 0.095 | 0.35 ± 1.59 | −0.12 (−0.19 to −0.06) <0.001 | 0.52 ± 0.092 | 0.48 ± 0.11 | -0.04(−0.11 to 0.04) 0.345 | 0.081 (F = 3.16) |
| Reduced Glutathione (GSH), nmol/mg protein | 22.6 ± 6.0 | 23.6 ± 3.0 | 0.99 (−1.08 to 3.06) 0.342 | 21.9 ± 3.1 | 22.1 ± 3.1 | 0.20 (−2.15 to 2.56) 0.864 | 0.617 (F = 0.25) |
| GSH/GSSH, ratio | 49.8 ± 15.4 | 81.9 ± 33.8 | 32.02 (19.90 to 44.15) <0.001 | 43.8 ± 11.9 | 47.6 ± 10.9 | 3.8 (−9.96 to 17.60) 0.580 | 0.003 (F = 9.50) |
| Protein carbonyl, nmol/ mg protein | 0.94 ± 0.48 | 0.65 ± 0.29 | −0.29 (−0.45 to −0.13)) 0.001 | 0.77 ± 0.54 | 0.79 ± 0.44 | 0.013 (−0.16 to 0.19) 0.881 | 0.012 (F = 6.52) |
| C-reactive protein, mg/L | 2.53 ± 3.07 | 2.35 ± 2.61 | −0.18 (−0.94 to 0.58) 0.641 | 2.06 ± 2.50 | 1.91 ± 1.87 | −0.15 (−0.97 to 0.67) 0.716 | 0.960 (F = 0.003) |
| Interleukin-6 (IL-6), pg/mL | 1.49 ± 0.96 | 0.91 ± 0.56 | −0.57 (−0.79 to −0.35) <0.001 | 1.38 ± 1.82 | 1.52 ± 2.24 | 0.14(−0.10 to 0.37) 0.251 | <0.001 (F = 18.78) |
| TNF-α, pg/mL | 6.82 ± 0.64 | 6.63 ± 1.04 | −0.19(−0.51 to 0.13) 0.250 | 6.96 ± 0.61 | 6.97 ± 0.81 | 0.01 (−0.34 to 0.35) 0.974 | 0.417 (F = 0.66) |
| Body weight, kg | 76.8 ± 13.9 | 76.7 ± 14.6 | −0.15 (−0.90 to 0.60) 0.695 | 76.7 ± 14.8 | 76.8 ± 14.9 | 0.06 (−0.74 to 0.86) 0.891 | 0.712 (F = 0.14) |
| Body mass index, kg/m2 | 27.2 ± 4.2 | 27.1 ± 4.2 | −0.08 (−0.24 to 0.07) 0.286 | 26.5 ± 3.9 | 26.5 ± 3.8 | 0.03 (−0.13 to 0.20) 0.711 | 0.317 (F = 1.01) |
| Fat mass, kg | 25.4 ± 11.7 | 25.1 ± 11.4 | −0.32 (−2.36 to 1.71) 0.752 | 21.8 ± 7.5 | 22.1 ± 8.0 | 0.31 (−1.82 to 2.43) 0.775 | 0.671 (F = 0.18) |
| SF-12 questionnaire | |||||||
| Physical health, score | 76.6 ± 17.3 | 79.0 ± 16.8 | 2.38 (−0.98 to 5.74) 0.163 | 82.2 ± 11.1 | 82.8 ± 11.9 | 0.66 (−3.00 to 4.32)) 0.721 | 0.493 (F = 0.47) |
| Mental health, score | 75.0 ± 17.1 | 77.7 ± 17.2 | 2.71 (−1.17 to 6.59) 0.168 | 78.5 ± 12.5 | 78.5 ± 13.8 | −0.05 (−4.27 to 4.17) 0.980 | 0.340 (F = 0.92) |
| Total score | 75.7 ± 15.7 | 77.9 ± 15.1 | 2.2 (−0.89 to 5.29) 0.161 | 80.7 ± 9.9 | 80.2 ± 10.5 | −0.5 (−3.87 to 2.87) 0.768 | 0.243 (F = 1.38) |
| Physical activity, MET-min/week | 3125.2 ± 3582.1 | 3004.3 ± 3254.1 | −120.9 (−738.1 to 496.4) 0.698 | 3260.6 ± 3261.0 | 3254.5 ± 3121.7 | −6.1 (−734.8 to 722.7) 0.987 | 0.811 (F = 0.06) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez Macarro, M.; Martínez Rodríguez, J.P.; Bernal Morell, E.; Pérez-Piñero, S.; Victoria-Montesinos, D.; García-Muñoz, A.M.; Cánovas García, F.; Castillo Sánchez, J.; López-Román, F.J. Effect of a Combination of Citrus Flavones and Flavanones and Olive Polyphenols for the Reduction of Cardiovascular Disease Risk: An Exploratory Randomized, Double-Blind, Placebo-Controlled Study in Healthy Subjects. Nutrients 2020, 12, 1475. https://doi.org/10.3390/nu12051475
Sánchez Macarro M, Martínez Rodríguez JP, Bernal Morell E, Pérez-Piñero S, Victoria-Montesinos D, García-Muñoz AM, Cánovas García F, Castillo Sánchez J, López-Román FJ. Effect of a Combination of Citrus Flavones and Flavanones and Olive Polyphenols for the Reduction of Cardiovascular Disease Risk: An Exploratory Randomized, Double-Blind, Placebo-Controlled Study in Healthy Subjects. Nutrients. 2020; 12(5):1475. https://doi.org/10.3390/nu12051475
Chicago/Turabian StyleSánchez Macarro, Maravillas, Juan Pablo Martínez Rodríguez, Enrique Bernal Morell, Silvia Pérez-Piñero, Desirée Victoria-Montesinos, Ana María García-Muñoz, Fernando Cánovas García, Julián Castillo Sánchez, and Francisco Javier López-Román. 2020. "Effect of a Combination of Citrus Flavones and Flavanones and Olive Polyphenols for the Reduction of Cardiovascular Disease Risk: An Exploratory Randomized, Double-Blind, Placebo-Controlled Study in Healthy Subjects" Nutrients 12, no. 5: 1475. https://doi.org/10.3390/nu12051475
APA StyleSánchez Macarro, M., Martínez Rodríguez, J. P., Bernal Morell, E., Pérez-Piñero, S., Victoria-Montesinos, D., García-Muñoz, A. M., Cánovas García, F., Castillo Sánchez, J., & López-Román, F. J. (2020). Effect of a Combination of Citrus Flavones and Flavanones and Olive Polyphenols for the Reduction of Cardiovascular Disease Risk: An Exploratory Randomized, Double-Blind, Placebo-Controlled Study in Healthy Subjects. Nutrients, 12(5), 1475. https://doi.org/10.3390/nu12051475







