Dietary Habits and Nutrition in Rheumatoid Arthritis: Can Diet Influence Disease Development and Clinical Manifestations?
Abstract
1. Introduction
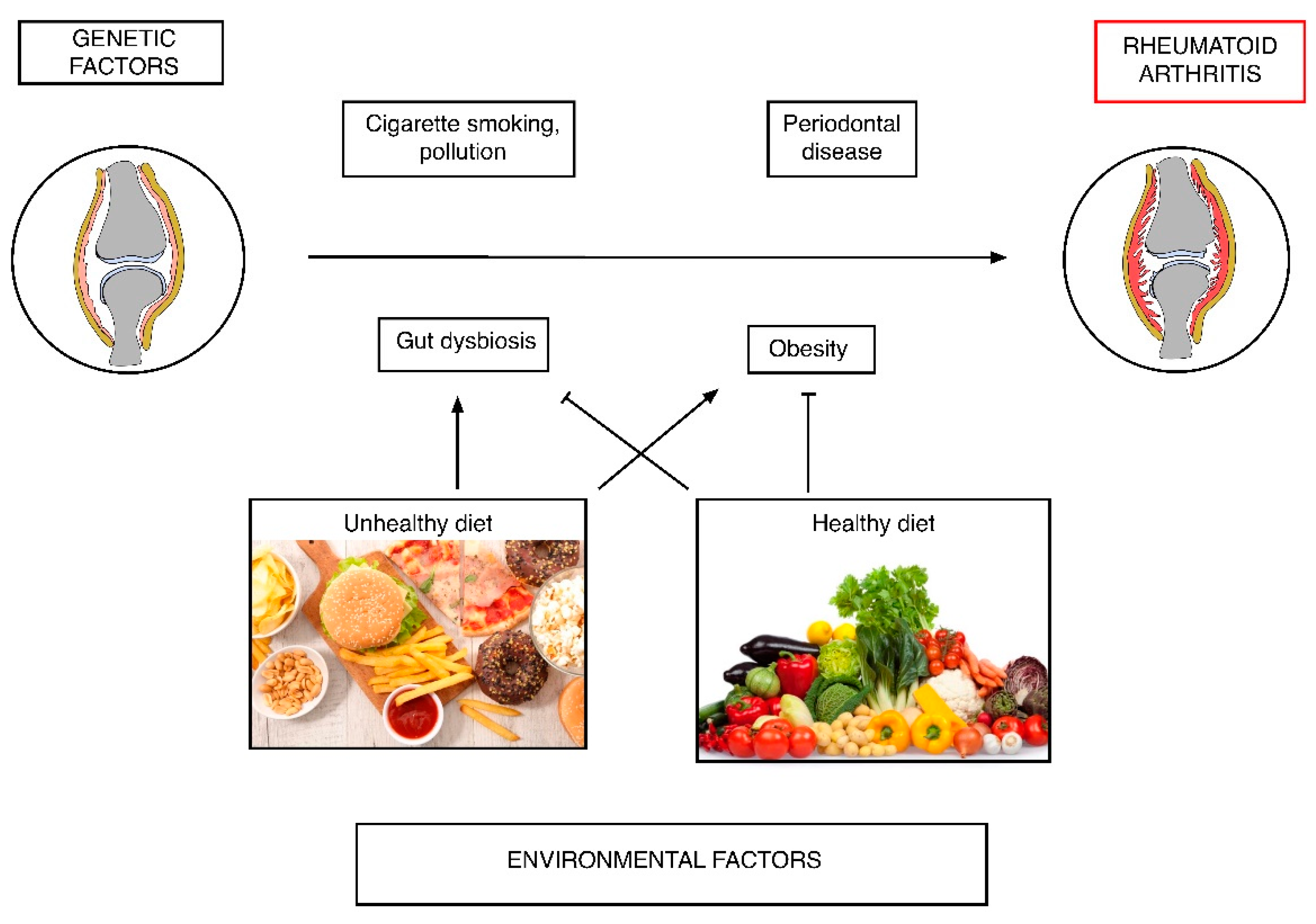
2. Rheumatoid Arthritis (RA) Pathogenesis: Diet as a Risk Factor
2.1. Diet and Inflammation
2.2. Dietary Habits and the Risk of RA Development
2.3. Changes in Body Composition and the Risk of RA
2.4. Diet, Microbiota and RA Risk
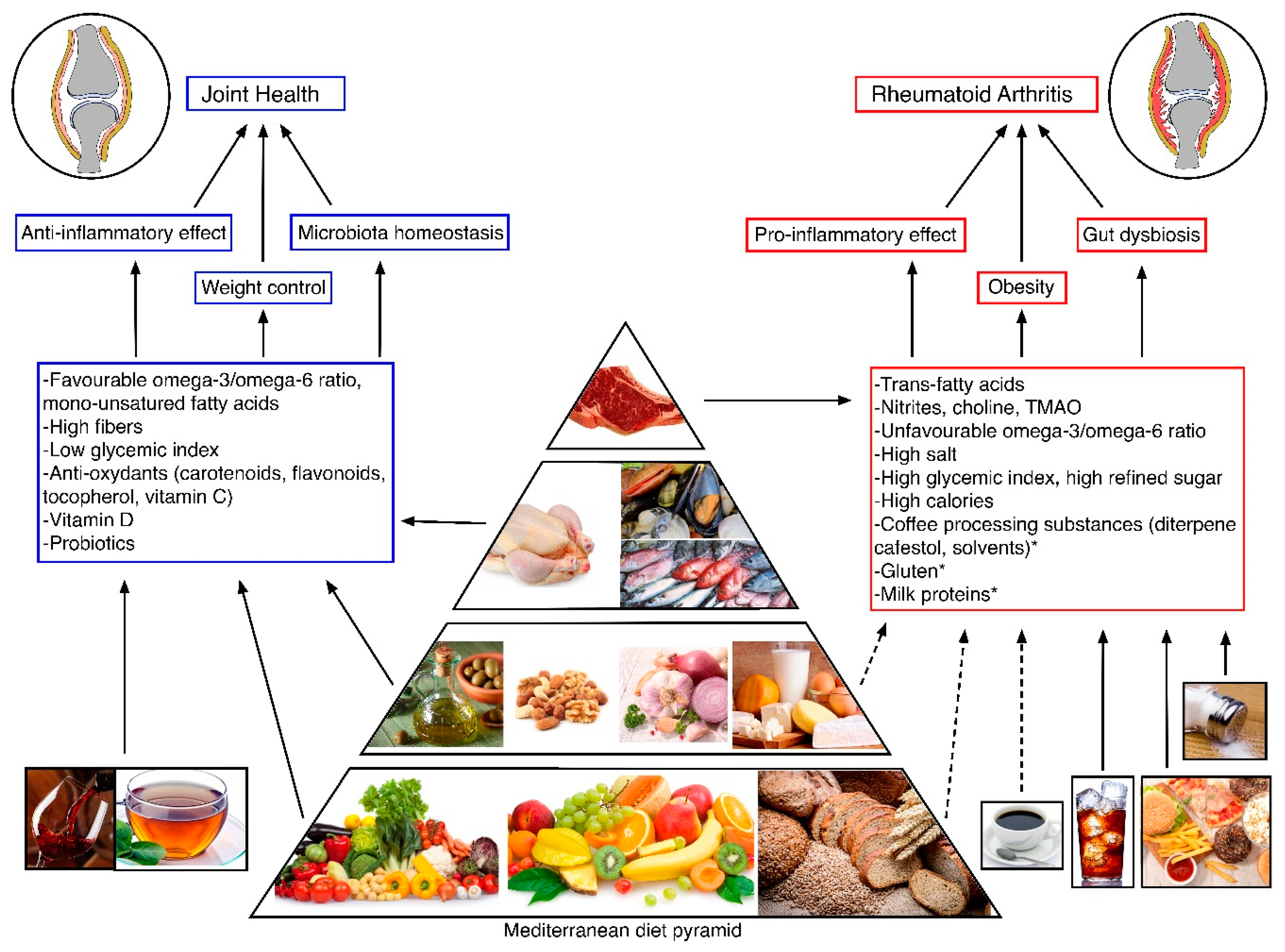
3. Protective Dietary Styles and Nutrients in Subjects at Risk for RA
3.1. Mediterranean Diet (MD)
3.2. Vitamin D
3.3. Alcohol and Red Wine
3.4. Green Tea
4. Effects of Diet and Nutrients on Disease Activity in Established RA
4.1. Omega 3 and Omega 6
4.2. Caloric Restriction
4.3. Antioxidant
4.4. Flavonoids
4.5. Gluten
4.6. Fasting
4.7. Vegan Diet
4.8. Vitamin D
4.9. Alcohol
4.10. Probiotics
5. Dietary Recommendation for RA Patients
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cross, M.; Smith, E.; Hoy, D.; Carmona, L.; Wolfe, F.; Vos, T.; Williams, B.; Gabriel, S.; Lassere, M.; Johns, N. The global burden of rheumatoid arthritis: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Alamanos, Y.; Drosos, A.A. Epidemiology of adult rheumatoid arthritis. Autoimmun. Rev. 2005, 4, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Balandraud, N.; Picard, C.; Reviron, D.; Landais, C.; Toussirot, E.; Lambert, N.; Telle, E.; Charpin, C.; Wendling, D.; Pardoux, E. HLA-DRB1 genotypes and the risk of developing anti citrullinated protein antibody (ACPA) positive rheumatoid arthritis. PLoS ONE 2013, 8, e64108. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Knevel, R.; Suwannalai, P.; van der Linden, M.P.; Janssen, G.M.C.; van Veelen, P.A.; Levarht, N.E.W.; van der Helm-van, A.H.M.; Cerami, A.; Huizinga, T.W.J. Autoantibodies recognizing carbamylated proteins are present in sera of patients with rheumatoid arthritis and predict joint damage. Proc. Natl. Acad. Sci. USA 2011, 108, 17372–17377. [Google Scholar] [CrossRef] [PubMed]
- Gerlag, D.M.; Raza, K.; van Baarsen, L.G.M.; Brouwer, E.; Buckley, C.D.; Burmester, G.R.; Gabay, C.; Catrina, A.I.; Cope, A.P.; Cornelis, F. EULAR recommendations for terminology and research in individuals at risk of rheumatoid arthritis: Report from the Study Group for Risk Factors for Rheumatoid Arthritis. Ann. Rheum. Dis. 2012, 71, 638–641. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, D.; Nishimura, K.; Tamaki, K.; Tsuji, G.; Nakazawa, T.; Morinobu, A.; Kumagai, S. Impact of smoking as a risk factor for developing rheumatoid arthritis: A meta-analysis of observational studies. Ann. Rheum. Dis. 2010, 69, 70–81. [Google Scholar] [CrossRef]
- Lucchino, B.; Spinelli, F.R.; Iannuccelli, C.; Guzzo, M.P.; Conti, F.; Franco, M.D. Mucosa–Environment interactions in the pathogenesis of rheumatoid arthritis. Cells 2019, 8, 700. [Google Scholar] [CrossRef]
- Pedersen, M.; Stripp, C.; Klarlund, M.; Olsen, S.F.; Tjønneland, A.M.; Frisch, M. Diet and risk of rheumatoid arthritis in a prospective cohort. J. Rheum. 2005, 32, 1249–1252. [Google Scholar]
- Cerhan, J.R.; Saag, K.G.; Merlino, L.A.; Mikuls, T.R.; Criswell, L.A. Antioxidant micronutrients and risk of rheumatoid arthritis in a cohort of older women. Am.J. Epidemiol. 2003, 157, 345–354. [Google Scholar] [CrossRef]
- Pattison, D.J.; Symmons, D.P.M.; Lunt, M.; Welch, A.; Luben, R.; Bingham, S.A.; Khaw, K.T.; Day, N.E.; Silman, A.J. Dietary risk factors for the development of inflammatory polyarthritis: Evidence for a role of high level of red meat consumption. Arthritis Rheum. 2004, 50, 3804–3812. [Google Scholar] [CrossRef]
- Pattison, D.J.; Silman, A.J.; Goodson, N.J.; Lunt, M.; Bunn, D.; Luben, R.; Welch, A.; Bingham, S.; Khaw, K.T.; Day, N. Vitamin C and the risk of developing inflammatory polyarthritis: Prospective nested case-control study. Ann. Rheum. Dis. 2004, 63, 843–847. [Google Scholar] [CrossRef] [PubMed]
- Benito-Garcia, E.; Feskanich, D.; Hu, F.B.; Mandl, L.A.; Karlson, E.W. Protein, iron, and meat consumption and risk for rheumatoid arthritis: A prospective cohort study. Arthritis Res. Ther. 2007, 9, R16. [Google Scholar] [CrossRef] [PubMed]
- Costenbader, K.H.; Kang, J.H.; Karlson, E.W. Antioxidant intake and risks of rheumatoid arthritis and systemic lupus erythematosus in women. Am.J. Epidemiol. 2010, 172, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B. The role of meat in the expression of rheumatoid arthritis. Br. J. Nutr. 2000, 84, 589–595. [Google Scholar] [CrossRef]
- Philippou, E.; Nikiphorou, E. Are we really what we eat? Nutrition and its role in the onset of rheumatoid arthritis. Autoimmun. Rev. 2018, 17, 1074–1077. [Google Scholar] [CrossRef]
- Oliviero, F.; Spinella, P.; Fiocco, U.; Ramonda, R.; Sfriso, P.; Punzi, L. How the Mediterranean diet and some of its components modulate inflammatory pathways in arthritis. Swiss Med. Wkly. 2015, 145, w14190. [Google Scholar] [CrossRef]
- Manzel, A.; Muller, D.N.; Hafler, D.A.; Erdman, S.E.; Linker, R.A.; Kleinewietfeld, M. Role of “Western diet” in inflammatory autoimmune diseases. Curr. Allergy Asthma Rep. 2014, 14, 404. [Google Scholar] [CrossRef]
- Rudan, I.; Sidhu, S.; Papana, A.; Meng, S.J.; Xin–Wei, Y.; Wang, W.; Campbell–Page, R.M.; Demaio, A.R.; Nair, H.; Sridhar, D. Prevalence of rheumatoid arthritis in low–and middle–income countries: A systematic review and analysis. J. Glob. Health 2015, 5, 010409. [Google Scholar]
- Minihane, A.M.; Vinoy, S.; Russell, W.R.; Baka, A.; Roche, H.M.; Tuohy, K.M.; Teeling, J.L.; Blaak, E.E.; Fenech, M.; Vauzour, D. Low-grade inflammation, diet composition and health: Current research evidence and its translation. Br. J. Nutr. 2015, 114, 999–1012. [Google Scholar] [CrossRef]
- Qin, B.; Yang, M.; Fu, H.; Ma, N.; Wei, T.; Tang, Q.; Hu, Z.; Liang, Y.; Yang, Z.; Zhong, R. Body mass index and the risk of rheumatoid arthritis: A systematic review and dose-response meta-analysis. Arthritis Res. Ther. 2015, 17, 86. [Google Scholar] [CrossRef]
- Galland, L. Diet and inflammation. Nutr. Clin. Pract. 2010, 25, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Ahluwalia, N.; Albers, R.; Bosco, N.; Bourdet-Sicard, R.; Haller, D.; Holgate, S.T.; Jönsson, L.S.; Latulippe, M.E.; Marcos, A. A consideration of biomarkers to be used for evaluation of inflammation in human nutritional studies. Br. J. Nutr. 2013, 109, S1–S34. [Google Scholar] [CrossRef]
- Ma, Y.; Hébert, J.R.; Li, W.; Bertone-Johnson, E.R.; Olendzki, B.; Pagoto, S.L.; Tinker, L.; Rosal, M.C.; Ockene, I.S.; Ockene, J.K. Association between dietary fiber and markers of systemic inflammation in the Women’s Health Initiative Observational Study. Nutrition 2008, 24, 941–949. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Cesari, F.; Abbate, R.; Gensini, G.F.; Casini, A. Adherence to Mediterranean diet and health status: Meta-analysis. BMJ 2008, 337, a1344. [Google Scholar] [CrossRef]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G. Mediterranean diet pyramid today. Science and cultural updates. Publ. Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef] [PubMed]
- Vallverdú-Queralt, A.; Regueiro, J.; Martínez-Huélamo, M.; Alvarenga, J.F.R.; Leal, L.N.; Lamuela-Raventos, R.M. A comprehensive study on the phenolic profile of widely used culinary herbs and spices: Rosemary, thyme, oregano, cinnamon, cumin and bay. Food Chem. 2014, 154, 299–307. [Google Scholar] [CrossRef] [PubMed]
- de Lorgeril, M.; Salen, P. The Mediterranean-style diet for the prevention of cardiovascular diseases. Publ. Health Nutr. 2006, 9, 118–123. [Google Scholar] [CrossRef] [PubMed]
- González-Gallego, J.; García-Mediavilla, M.V.; Sánchez-Campos, S.; Tuñón, M.J. Anti-inflammatory and immunomodulatory properties of dietary flavonoids. In Polyphenols in Human Health and Disease; Elsevier: Leon, Spain, 2014; pp. 435–452. [Google Scholar]
- Sesso, H.D.; Liu, S.; Gaziano, J.M.; Buring, J.E. Dietary lycopene, tomato-based food products and cardiovascular disease in women. J. Nutr. 2003, 133, 2336–2341. [Google Scholar] [CrossRef]
- Cheng, H.M.; Koutsidis, G.; Lodge, J.K.; Ashor, A.; Siervo, M.; Lara, J. Tomato and lycopene supplementation and cardiovascular risk factors: A systematic review and meta-analysis. Atherosclerosis 2017, 257, 100–108. [Google Scholar] [CrossRef]
- McGill, C.R.; Kurilich, A.C.; Davignon, J. The role of potatoes and potato components in cardiometabolic health: A review. Ann. Med. 2013, 45, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.J.; Blake, D.R.; Wainwright, A.C.; Steven, M.M. Relationship between iron deposits and tissue damage in the synovium: An ultrastructural study. Ann. Rheum. Dis. 1986, 45, 21–26. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Wang, Y.; Feng, M.; Zhang, X.; Jin, Y.-B.; Li, X.; Su, L.-C.; Liu, S.; Wang, A.-X.; Chen, X.-M. Dietary intake and risk of rheumatoid arthritis—A cross section multicenter study. Clin. Rheum. 2016, 35, 2901–2908. [Google Scholar] [CrossRef] [PubMed]
- Salgado, E.; Bes-Rastrollo, M.; de Irala, J.; Carmona, L.; Gomez-Reino, J.J. High sodium intake is associated with self-reported rheumatoid arthritis: A cross sectional and case control analysis within the SUN cohort. Medicine 2015, 94. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Yosef, N.; Thalhamer, T.; Zhu, C.; Xiao, S.; Kishi, Y.; Regev, A.; Kuchroo, V.K. Induction of pathogenic T H 17 cells by inducible salt-sensing kinase SGK1. Nature 2013, 496, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Sundström, B.; Alfredsson, L.; Klareskog, L.; Rantapää-Dahlqvist, S.; Bengtsson, C. High sodium chloride consumption enhances the effects of smoking but does not interact with SGK1 polymorphisms in the development of ACPA-positive status in patients with RA. Ann. Rheum. Dis. 2016, 75, 943–946. [Google Scholar] [CrossRef]
- Merlino, L.A.; Curtis, J.; Mikuls, T.R.; Cerhan, J.R.; Criswell, L.A.; Saag, K.G. Vitamin D intake is inversely associated with rheumatoid arthritis: Results from the Iowa Women’s Health Study. Arthritis Rheum. 2004, 50, 72–77. [Google Scholar] [CrossRef]
- Sundström, B.; Ljung, L.; Di Giuseppe, D. Consumption of meat and dairy products is not associated with the risk for rheumatoid arthritis among women: A population-based cohort study. Nutrients 2019, 11, 2825. [Google Scholar] [CrossRef]
- Panush, R.S.; Stroud, R.M.; Webster, E.M. Food—Induced (allergic) arthritis. Inflammatory arthritis exacerbated by milk. Arthritis Rheum. 1986, 29, 220–226. [Google Scholar] [CrossRef]
- Li, J.; Yan, H.; Chen, H.; Ji, Q.; Huang, S.; Yang, P.; Liu, Z.; Yang, B. The pathogenesis of rheumatoid arthritis is associated with milk or egg allergy. North Am. J. Med. Sci. 2016, 8, 40. [Google Scholar]
- Lidén, M.; Kristjánsson, G.; Valtysdottir, S.; Venge, P.; Hällgren, R. Self-reported food intolerance and mucosal reactivity after rectal food protein challenge in patients with rheumatoid arthritis. Scand. J. Rheumatol. 2010, 39, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nature Med. 2013, 19, 576. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; DuGar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.-M. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Coras, R.; D Murillo-Saich, J.; Guma, M. Circulating pro-and anti-inflammatory metabolites and its potential role in rheumatoid arthritis pathogenesis. Cells 2020, 9, 827. [Google Scholar] [CrossRef] [PubMed]
- Saidane, O.; Semerano, L.; Sellam, J. Could omega-3 fatty acids prevent rheumatoid arthritis? Joint Bone Spine 2018, 86, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Di Giuseppe, D.; Wallin, A.; Bottai, M.; Askling, J.; Wolk, A. Long-term intake of dietary long-chain n-3 polyunsaturated fatty acids and risk of rheumatoid arthritis: A prospective cohort study of women. Ann. Rheum. Dis. 2014, 73, 1949–1953. [Google Scholar] [CrossRef]
- Rosell, M.; Wesley, A.-M.; Rydin, K.; Klareskog, L.; Alfredsson, L.; Group, E.S. Dietary fish and fish oil and the risk of rheumatoid arthritis. Epidemiology 2009, 896–901. [Google Scholar] [CrossRef]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef]
- Linos, A.; Kaklamani, V.G.; Kaklamani, E.; Koumantaki, Y.; Giziaki, E.; Papazoglou, S.; Mantzoros, C.S. Dietary factors in relation to rheumatoid arthritis: A role for olive oil and cooked vegetables? Am. J. Clin. Nutr. 1999, 70, 1077–1082. [Google Scholar] [CrossRef]
- Serra-Majem, L.; De la Cruz, J.N.; Ribas, L.; Tur, J.A. Olive oil and the Mediterranean diet: Beyond the rhetoric. Eur. J. Clin. Nutr. 2003, 57, S2–S7. [Google Scholar] [CrossRef]
- Bustamante, M.F.; Agustín-Perez, M.; Cedola, F.; Coras, R.; Narasimhan, R.; Golshan, S.; Guma, M. Design of an anti-inflammatory diet (ITIS diet) for patients with rheumatoid arthritis. Contemp. Clin. Trials Commun. 2020, 17, 100524. [Google Scholar] [CrossRef]
- Hu, Y.; Costenbader, K.H.; Gao, X.; Al-Daabil, M.; Sparks, J.A.; Solomon, D.H.; Hu, F.B.; Karlson, E.W.; Lu, B. Sugar-sweetened soda consumption and risk of developing rheumatoid arthritis in women. Am. J. Clin. Nutr. 2014, 100, 959–967. [Google Scholar] [CrossRef] [PubMed]
- DeChristopher, L.R.; Uribarri, J.; Tucker, K.L. Intake of high-fructose corn syrup sweetened soft drinks, fruit drinks and apple juice is associated with prevalent arthritis in US adults, aged 20–30 years. Nutr. Diabetes 2016, 6, e199. [Google Scholar] [CrossRef] [PubMed]
- Mikuls, T.R.; Cerhan, J.R.; Criswell, L.A.; Merlino, L.; Mudano, A.S.; Burma, M.; Folsom, A.R.; Saag, K.G. Coffee, tea, and caffeine consumption and risk of rheumatoid arthritis: Results from the Iowa Women’s Health Study. Arthritis Rheum. 2002, 46, 83–91. [Google Scholar] [CrossRef]
- Heliövaara, M.; Aho, K.; Knekt, P.; Impivaara, O.; Reunanen, A.; Aromaa, A. Coffee consumption, rheumatoid factor, and the risk of rheumatoid arthritis. Ann. Rheum. Dis. 2000, 59, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, G.B.; Robins, M.; Maffeo, C.E.; Masters, M.N.; Bond, A.P.; Morganstein, D. Confounders contributing to the reported associations of coffee or caffeine with disease. Prev. Med. 1988, 17, 295–309. [Google Scholar] [CrossRef]
- Lee, Y.H.; Bae, S.-C.; Song, G.G. Coffee or tea consumption and the risk of rheumatoid arthritis: A meta-analysis. Clin. Rheum. 2014, 33, 1575–1583. [Google Scholar] [CrossRef]
- Heliövaara, M.; Aho, K.; Knekt, P.; Reunamen, A.; Aromaa, A. Serum cholesterol and risk of rheumatoid arthritis in a cohort of 52 800 men and women. Rheumatology 1996, 35, 255–257. [Google Scholar] [CrossRef][Green Version]
- Garabrant, D.H.; Dumas, C. Epidemiology of organic solvents and connective tissue disease. Arthritis Res. Ther. 1999, 2, 5. [Google Scholar] [CrossRef][Green Version]
- Hu, Y.; Sparks, J.A.; Malspeis, S.; Costenbader, K.H.; Hu, F.B.; Karlson, E.W.; Lu, B. Long-term dietary quality and risk of developing rheumatoid arthritis in women. Ann. Rheum. Dis. 2017, 76, 1357–1364. [Google Scholar] [CrossRef]
- Ljung, L.; Rantapää-Dahlqvist, S. Abdominal obesity, gender and the risk of rheumatoid arthritis—A nested case-control study. Arthritis Res. Ther. 2016, 18, 277. [Google Scholar] [CrossRef] [PubMed]
- de Hair, M.J.H.; Landewé, R.B.M.; van de Sande, M.G.H.; van Schaardenburg, D.; van Baarsen, L.G.M.; Gerlag, D.M.; Tak, P.P. Smoking and overweight determine the likelihood of developing rheumatoid arthritis. Ann. Rheum. Dis. 2013, 72, 1654–1658. [Google Scholar] [CrossRef] [PubMed]
- Ouchi, N.; Parker, J.L.; Lugus, J.J.; Walsh, K. Adipokines in inflammation and metabolic disease. Nat. Rev. Immunol. 2011, 11, 85. [Google Scholar] [CrossRef] [PubMed]
- Naylor, C.; Petri, W.A., Jr. Leptin regulation of immune responses. Trends Mol. Med. 2016, 22, 88–98. [Google Scholar] [CrossRef]
- Lam, Q.L.K.; Wang, S.; Ko, O.K.H.; Kincade, P.W.; Lu, L. Leptin signaling maintains B-cell homeostasis via induction of Bcl-2 and Cyclin D1. Proc. Natl. Acad. Sci. USA 2010, 107, 13812–13817. [Google Scholar] [CrossRef]
- Ehling, A.; Schäffler, A.; Herfarth, H.; Tarner, I.H.; Anders, S.; Distler, O.; Paul, G.; Distler, J.; Gay, S.; Schölmerich, J. The potential of adiponectin in driving arthritis. J. Immunol. 2006, 176, 4468–4478. [Google Scholar] [CrossRef]
- Luo, X.H.; Guo, L.J.; Xie, H.; Yuan, L.Q.; Wu, X.P.; Zhou, H.D.; Liao, E.Y. Adiponectin stimulates RANKL and inhibits OPG expression in human osteoblasts through the MAPK signaling pathway. J. Bone Mineral. Res. 2006, 21, 1648–1656. [Google Scholar] [CrossRef]
- Tang, C.-H.; Chiu, Y.-C.; Tan, T.-W.; Yang, R.-S.; Fu, W.-M. Adiponectin enhances IL-6 production in human synovial fibroblast via an AdipoR1 receptor, AMPK, p38, and NF-κB pathway. J. Immunol. 2007, 179, 5483–5492. [Google Scholar] [CrossRef]
- Chung, C.P.; Oeser, A.; Solus, J.F.; Avalos, I.; Gebretsadik, T.; Shintani, A.; Raggi, P.; Sokka, T.; Pincus, T.; Stein, C.M. Prevalence of the metabolic syndrome is increased in rheumatoid arthritis and is associated with coronary atherosclerosis. Atherosclerosis 2008, 196, 756–763. [Google Scholar] [CrossRef]
- Ferguson, L.D.; Brown, R.; Celis-Morales, C.; Welsh, P.; Lyall, D.M.; Pell, J.P.; McInnes, I.B.; Siebert, S.; Sattar, N. Association of central adiposity with psoriasis, psoriatic arthritis and rheumatoid arthritis: A cross-sectional study of the UK Biobank. Rheumatology 2019, 58, 2137–2142. [Google Scholar] [CrossRef]
- Ferguson, L.D.; Siebert, S.; McInnes, I.B.; Sattar, N. Cardiometabolic comorbidities in RA and PsA: Lessons learned and future directions. Nat. Rev. Rheumatol. 2019, 15, 461–474. [Google Scholar] [CrossRef] [PubMed]
- Engvall, I.L.; Elkan, A.C.; Tengstrand, B.; Cederholm, T.; Brismar, K.; Hafström, I. Cachexia in rheumatoid arthritis is associated with inflammatory activity, physical disability, and low bioavailable insulin—Like growth factor. Scand. J. Rheumatol. 2008, 37, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Dent, E.; Morley, J.E.; Cruz-Jentoft, A.J.; Arai, H.; Kritchevsky, S.B.; Guralnik, J.; Bauer, J.M.; Pahor, M.; Clark, B.C.; Cesari, M. International clinical practice guidelines for sarcopenia (ICFSR): Screening, diagnosis and management. J. Nutr. Health Aging 2018, 22, 1148–1161. [Google Scholar] [CrossRef]
- Beaudart, C.; Dawson, A.; Shaw, S.C.; Harvey, N.C.; Kanis, J.A.; Binkley, N.; Reginster, J.Y.; Chapurlat, R.; Chan, D.C.; Bruyère, O. Nutrition and physical activity in the prevention and treatment of sarcopenia: Systematic review. Osteoporos. Int. 2017, 28, 1817–1833. [Google Scholar] [CrossRef] [PubMed]
- Dao, H.-H.; Do, Q.-T.; Sakamoto, J. Abnormal body composition phenotypes in Vietnamese women with early rheumatoid arthritis. Rheumatology 2011, 50, 1250–1258. [Google Scholar] [CrossRef] [PubMed]
- Book, C.; Karlsson, M.K.; Åkesson, K.; Jacobsson, L.T.H. Early rheumatoid arthritis and body composition. Rheumatology 2009, 48, 1128–1132. [Google Scholar] [CrossRef] [PubMed]
- Turk, S.A.; van Schaardenburg, D.; Boers, M.; de Boer, S.; Fokker, C.; Lems, W.F.; Nurmohamed, M.T. An unfavorable body composition is common in early arthritis patients: A case control study. PLoS ONE 2018, 13, e0193377. [Google Scholar] [CrossRef]
- Linauskas, A.; Overvad, K.; Symmons, D.; Johansen, M.B.; Stengaard-Pedersen, K.; de Thurah, A. Body fat percentage, waist circumference, and obesity as risk factors for rheumatoid arthritis: A Danish cohort study. Arthritis Care Res. 2019, 71, 777–786. [Google Scholar] [CrossRef]
- Zhong, D.; Wu, C.; Zeng, X.; Wang, Q. The role of gut microbiota in the pathogenesis of rheumatic diseases. Clin. Rheum. 2018, 37, 25–34. [Google Scholar] [CrossRef]
- Mu, Q.; Kirby, J.; Reilly, C.M.; Luo, X.M. Leaky gut as a danger signal for autoimmune diseases. Front. Immunol. 2017, 8, 598. [Google Scholar] [CrossRef]
- Thorburn, A.N.; Macia, L.; Mackay, C.R. Diet, metabolites, and “western-lifestyle” inflammatory diseases. Immunity 2014, 40, 833–842. [Google Scholar] [CrossRef] [PubMed]
- van der Meulen, T.A.; Harmsen, H.J.M.; Bootsma, H.; Spijkervet, F.K.L.; Kroese, F.G.M.; Vissink, A. The microbiome–systemic diseases connection. Oral Dis. 2016, 22, 719–734. [Google Scholar] [CrossRef] [PubMed]
- Trompette, A.; Gollwitzer, E.S.; Yadava, K.; Sichelstiel, A.K.; Sprenger, N.; Ngom-Bru, C.; Blanchard, C.; Junt, T.; Nicod, L.P.; Harris, N.L. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nutr. Med. 2014, 20, 159. [Google Scholar] [CrossRef] [PubMed]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; van der Veeken, J.; Deroos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef]
- Duerkop, B.A.; Vaishnava, S.; Hooper, L.V. Immune responses to the microbiota at the intestinal mucosal surface. Immunity 2009, 31, 368–376. [Google Scholar] [CrossRef]
- Macpherson, A.J.; Harris, N.L. Interactions between commensal intestinal bacteria and the immune system. Nat. Rev. Immunol. 2004, 4, 478–485. [Google Scholar] [CrossRef]
- Mazmanian, S.K.; Kasper, D.L. The love–hate relationship between bacterial polysaccharides and the host immune system. Nat. Rev. Immunol. 2006, 6, 849–858. [Google Scholar] [CrossRef]
- Yeoh, N.; Burton, J.P.; Suppiah, P.; Reid, G.; Stebbings, S. The role of the microbiome in rheumatic diseases. Curr. Rheumatol. Rep. 2013, 15, 314. [Google Scholar] [CrossRef]
- Wu, H.-J.; Ivanov, I.I.; Darce, J.; Hattori, K.; Shima, T.; Umesaki, Y.; Littman, D.R.; Benoist, C.; Mathis, D. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 2010, 32, 815–827. [Google Scholar] [CrossRef]
- Scher, J.U.; Sczesnak, A.; Longman, R.S.; Segata, N.; Ubeda, C.; Bielski, C.; Rostron, T.; Cerundolo, V.; Pamer, E.G.; Abramson, S.B. Expansion of intestinal Prevotella copri correlates with enhanced susceptibility to arthritis. Elife 2013, 2, e01202. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, D.; Jia, H.; Feng, Q.; Wang, D.; Liang, D.; Wu, X.; Li, J.; Tang, L.; Li, Y. The oral and gut microbiomes are perturbed in rheumatoid arthritis and partly normalized after treatment. Nat. Med. 2015, 21, 895. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wright, K.; Davis, J.M.; Jeraldo, P.; Marietta, E.V.; Murray, J.; Nelson, H.; Matteson, E.L.; Taneja, V. An expansion of rare lineage intestinal microbes characterizes rheumatoid arthritis. Genome Med. 2016, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Horta-Baas, G.; Romero-Figueroa, M.d.S.; Montiel-Jarquín, A.J.; Pizano-Zárate, M.L.; García-Mena, J.; Ramírez-Durán, N. Intestinal dysbiosis and rheumatoid arthritis: A link between gut microbiota and the pathogenesis of rheumatoid arthritis. J. Immunol. Res. 2017, 2017, 4835189. [Google Scholar] [CrossRef] [PubMed]
- De Santis, S.; Cavalcanti, E.; Mastronardi, M.; Jirillo, E.; Chieppa, M. Nutritional keys for intestinal barrier modulation. Front. Immunol. 2015, 6, 612. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Yoo, S.-A.; Kim, W.-U. Gut microbiota in autoimmunity: Potential for clinical applications. Arch. Pharm. Res. 2016, 39, 1565–1576. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Kurakawa, T.; Umemoto, E.; Motooka, D.; Ito, Y.; Gotoh, K.; Hirota, K.; Matsushita, M.; Furuta, Y.; Narazaki, M. Dysbiosis contributes to arthritis development via activation of autoreactive T cells in the intestine. Arthritis Rheumatol. 2016, 68, 2646–2661. [Google Scholar] [CrossRef]
- Bernard, N.J. Prevotella copri associated with new-onset untreated RA. Nat. Rev. Rheumatol 2014, 10, 2–3. [Google Scholar] [CrossRef]
- Vaahtovuo, J.; Munukka, E.; KorkeamÄKi, M.; Luukkainen, R.; Toivanen, P. Fecal microbiota in early rheumatoid arthritis. J. Rheum. 2008, 35, 1500–1505. [Google Scholar]
- Marietta, E.V.; Murray, J.A.; Luckey, D.H.; Jeraldo, P.R.; Lamba, A.; Patel, R.; Luthra, H.S.; Mangalam, A.; Taneja, V. Human gut-derived Prevotella histicola suppresses inflammatory arthritis in humanized mice. Arthritis Rheumatol. (Hoboken, NJ) 2016, 68, 2878. [Google Scholar] [CrossRef]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.-Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef]
- Gorvitovskaia, A.; Holmes, S.P.; Huse, S.M. Interpreting Prevotella and Bacteroides as biomarkers of diet and lifestyle. Microbiome 2016, 4, 15. [Google Scholar] [CrossRef] [PubMed]
- Valdes, A.M.; Walter, J.; Segal, E.; Spector, T.D. Role of the gut microbiota in nutrition and health. BMJ 2018, 361, k2179. [Google Scholar] [CrossRef] [PubMed]
- Mitsou, E.K.; Kakali, A.; Antonopoulou, S.; Mountzouris, K.C.; Yannakoulia, M.; Panagiotakos, D.B.; Kyriacou, A. Adherence to the Mediterranean diet is associated with the gut microbiota pattern and gastrointestinal characteristics in an adult population. Br. J. Nutr. 2017, 117, 1645–1655. [Google Scholar] [CrossRef]
- Lerner, A.; Patricia, J.; Matthias, T. Nutrients, bugs and us: The short-chain fatty acids story in celiac disease. Int. J. Celiac. Dis. 2016, 4, 92–94. [Google Scholar]
- Michalsen, A.; Riegert, M.; Lüdtke, R.; Bäcker, M.; Langhorst, J.; Schwickert, M.; Dobos, G.J. Mediterranean diet or extended fasting’s influence on changing the intestinal microflora, immunoglobulin a secretion and clinical outcome in patients with rheumatoid arthritis and fibromyalgia: An observational study. BMC Complement. Altern. Med. 2005, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Pagliai, G.; Russo, E.; Niccolai, E.; Dinu, M.; Di Pilato, V.; Magrini, A.; Bartolucci, G.; Baldi, S.; Menicatti, M.; Giusti, B. Influence of a 3-month low-calorie Mediterranean diet compared to the vegetarian diet on human gut microbiota and SCFA: The CARDIVEG Study. Eur. J. Nutr. 2019, 1–14. [Google Scholar] [CrossRef]
- Dinu, M.; Pagliai, G.; Casini, A.; Sofi, F. Mediterranean diet and multiple health outcomes: An umbrella review of meta-analyses of observational studies and randomised trials. Eur. J. Clin. Nutr. 2018, 72, 30–43. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Sugioka, Y.; Tada, M.; Okano, T.; Mamoto, K.; Inui, K.; Habu, D.; Koike, T. Monounsaturated fatty acids might be key factors in the Mediterranean diet that suppress rheumatoid arthritis disease activity: The TOMORROW study. Clin. Nutr. 2018, 37, 675–680. [Google Scholar] [CrossRef]
- Philippou, E. The Glycemic Index: Applications in Practice; CRC Press: Nicosia, Cyprus; London, UK, 2016. [Google Scholar]
- Hu, Y.; Costenbader, K.H.; Gao, X.; Hu, F.B.; Karlson, E.W.; Lu, B. Mediterranean diet and incidence of rheumatoid arthritis in women. Arthritis Care Res. 2015, 67, 597–606. [Google Scholar] [CrossRef]
- Sundström, B.; Johansson, I.; Rantapää-Dahlqvist, S. Diet and alcohol as risk factors for rheumatoid arthritis: A nested case–control study. Rheumatol. Int. 2015, 35, 533–539. [Google Scholar] [CrossRef]
- Forsyth, C.; Kouvari, M.; D’Cunha, N.M.; Georgousopoulou, E.N.; Panagiotakos, D.B.; Mellor, D.D.; Kellett, J.; Naumovski, N. The effects of the Mediterranean diet on rheumatoid arthritis prevention and treatment: A systematic review of human prospective studies. Rheumatol. Int. 2018, 38, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Raza, K.; Hewison, M. Vitamin D in rheumatoid arthritis—Towards clinical application. Nat. Rev. Rheumatol. 2016, 12, 201. [Google Scholar] [CrossRef] [PubMed]
- Haussler, M.R.; Haussler, C.A.; Whitfield, G.K.; Hsieh, J.-C.; Thompson, P.D.; Barthel, T.K.; Bartik, L.; Egan, J.B.; Wu, Y.; Kubicek, J.L. The nuclear vitamin D receptor controls the expression of genes encoding factors which feed the “Fountain of Youth” to mediate healthful aging. J. Steroid Biochem. Mol. Biol. 2010, 121, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.R.; Li, D.; Jeffery, L.E.; Raza, K.; Hewison, M. Vitamin D, autoimmune disease and rheumatoid arthritis. Calcif. Tissue Int. 2019, 1–18. [Google Scholar] [CrossRef]
- Zwerina, K.; Baum, W.; Axmann, R.; Heiland, G.R.; Distler, J.H.; Smolen, J.; Hayer, S.; Zwerina, J.; Schett, G. Vitamin D receptor regulates TNF-mediated arthritis. Ann. Rheum. Dis. 2011, 70, 1122–1129. [Google Scholar] [CrossRef]
- Nielen, M.M.J.; van Schaardenburg, D.; Lems, W.F.; van de Stadt, R.J.; de Koning, M.H.M.T.; Reesink, H.W.; Habibuw, M.R.; van der Horst-Bruinsma, I.E.; Twisk, J.W.R.; Dijkmans, B.A.C. Vitamin D deficiency does not increase the risk of rheumatoid arthritis: Comment on the article by Merlino et al. Arthritis Rheum. 2006, 54, 3719–3720. [Google Scholar] [CrossRef]
- Feser, M.; Derber, L.A.; Deane, K.D.; Lezotte, D.C.; Weisman, M.H.; Buckner, J.H.; Mikuls, T.; O’Dell, J.; Gregersen, P.K.; Holers, V.M. Plasma 25, OH vitamin D concentrations are not associated with rheumatoid arthritis (RA)-related autoantibodies in individuals at elevated risk for RA. J. Rheum. 2009, 36, 943–946. [Google Scholar] [CrossRef]
- Racovan, M.; Walitt, B.; Collins, C.E.; Pettinger, M.; Parks, C.G.; Shikany, J.M.; Wactawski-Wende, J.; Manson, J.E.; Moreland, L.; Wright, N. Calcium and vitamin D supplementation and incident rheumatoid arthritis: The Women’s Health Initiative Calcium plus Vitamin D trial. Rheumatol. Int. 2012, 32, 3823–3830. [Google Scholar] [CrossRef]
- Song, G.G.; Bae, S.-C.; Lee, Y.H. Association between vitamin D intake and the risk of rheumatoid arthritis: A meta-analysis. Clin. Rheum. 2012, 31, 1733–1739. [Google Scholar] [CrossRef]
- Dennis, G.; Holweg, C.T.J.; Kummerfeld, S.K.; Choy, D.F.; Setiadi, A.F.; Hackney, J.A.; Haverty, P.M.; Gilbert, H.; Lin, W.Y.; Diehl, L. Synovial phenotypes in rheumatoid arthritis correlate with response to biologic therapeutics. Arthritis Res. Ther. 2014, 16, R90. [Google Scholar] [CrossRef]
- Hiraki, L.T.; Arkema, E.V.; Cui, J.; Malspeis, S.; Costenbader, K.H.; Karlson, E.W. Circulating 25-hydroxyvitamin D level and risk of developing rheumatoid arthritis. Rheumatology 2014, 53, 2243–2248. [Google Scholar] [CrossRef] [PubMed]
- Wood, C.L.; Cheetham, T.D. Vitamin D: Increasing supplement use among at-risk groups (NICE guideline PH56). Arch. Dis. Child. Educ. Pract. 2016, 101, 43–45. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metabol. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Jeffery, L.E.; Jenkinson, C.; Harrison, S.R.; Chun, R.F.; Adams, J.S.; Raza, K.; Hewison, M. Serum and synovial fluid vitamin D metabolites and rheumatoid arthritis. J. Steroid Biochem. Mol. Biol. 2019, 187, 1–8. [Google Scholar] [CrossRef]
- Lu, B.; Solomon, D.H.; Costenbader, K.H.; Karlson, E.W. Alcohol consumption and risk of incident rheumatoid arthritis in women: A prospective study. Arthritis Rheumatol. 2014, 66, 1998–2005. [Google Scholar] [CrossRef]
- Cerhan, J.R.; Saag, K.G.; Criswell, L.A.; Merlino, L.A.; Mikuls, T.R. Blood transfusion, alcohol use, and anthropometric risk factors for rheumatoid arthritis in older women. J. Rheum. 2002, 29, 246–254. [Google Scholar]
- Fekjær, H.O. Alcohol—A universal preventive agent? A critical analysis. Addiction 2013, 108, 2051–2057. [Google Scholar] [CrossRef]
- Fekjær, H.O. Response to commentaries. Addiction 2013, 108, 2060–2061. [Google Scholar] [CrossRef][Green Version]
- Hansel, B.; Kontush, A.; Bruckert, E. Is a cardioprotective action of alcohol a myth? Curr. Opin. Cardiol. 2012, 27, 550–555. [Google Scholar] [CrossRef]
- Snopek, L.; Mlcek, J.; Sochorova, L.; Baron, M.; Hlavacova, I.; Jurikova, T.; Kizek, R.; Sedlackova, E.; Sochor, J. Contribution of red wine consumption to human health protection. Molecules 2018, 23, 1684. [Google Scholar] [CrossRef]
- Khan, N.; Afaq, F.; Saleem, M.; Ahmad, N.; Mukhtar, H. Targeting multiple signaling pathways by green tea polyphenol (−)-epigallocatechin-3-gallate. Cancer Res. 2006, 66, 2500–2505. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Silverman, M.D.; Marotte, H.; Kwan, K.; Matuszczak, N.; Koch, A.E. Down-regulation of myeloid cell leukemia 1 by epigallocatechin-3-gallate sensitizes rheumatoid arthritis synovial fibroblasts to tumor necrosis factor α-induced apoptosis. Arthritis Rheum. 2009, 60, 1282–1293. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Pakozdi, A.; Koch, A.E. Regulation of interleukin-1β–induced chemokine production and matrix metalloproteinase 2 activation by epigallocatechin-3-gallate in rheumatoid arthritis synovial fibroblasts. Arthritis Rheum. 2006, 54, 2393–2401. [Google Scholar] [CrossRef] [PubMed]
- Min, S.-Y.; Yan, M.; Kim, S.B.; Ravikumar, S.; Kwon, S.-R.; Vanarsa, K.; Kim, H.-Y.; Davis, L.S.; Mohan, C. Green tea epigallocatechin-3-gallate suppresses autoimmune arthritis through indoleamine-2, 3-dioxygenase expressing dendritic cells and the nuclear factor, erythroid 2-like 2 antioxidant pathway. J. Inflamm. 2015, 12, 53. [Google Scholar] [CrossRef]
- Bykerk, V.P.; Shadick, N.; Frits, M.; Bingham, C.O.; Jeffery, I.; Iannaccone, C.; Weinblatt, M.; Solomon, D.H. Flares in rheumatoid arthritis: Frequency and management. A report from the BRASS registry. J. Rheum. 2014, 41, 227–234. [Google Scholar] [CrossRef]
- Smedslund, G.; Byfuglien, M.G.; Olsen, S.U.; Hagen, K.B. Effectiveness and safety of dietary interventions for rheumatoid arthritis: A systematic review of randomized controlled trials. J. Am. Diet. Assoc. 2010, 110, 727–735. [Google Scholar] [CrossRef]
- Norris, P.C.; Dennis, E.A. Omega-3 fatty acids cause dramatic changes in TLR4 and purinergic eicosanoid signaling. Proc. Natl. Acad. Sci. USA 2012, 109, 8517–8522. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, J.H.; Allayee, H.; Dwyer, K.M.; Fan, J.; Wu, H.; Mar, R.; Lusis, A.J.; Mehrabian, M. Arachidonate 5-lipoxygenase promoter genotype, dietary arachidonic acid, and atherosclerosis. N. Engl. J. Med. 2004, 350, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Raper, N.R.; Cronin, F.J.; Exler, J. Omega-3 fatty acid content of the US food supply. J. Am. Coll. Nutr. 1992, 11, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Whelan, J. Antagonistic effects of dietary arachidonic acid and n-3 polyunsaturated fatty acids. J. Nutr. 1996, 126, 1086S–1091S. [Google Scholar] [CrossRef] [PubMed]
- Malawista, S.E.; de Boisfleury Chevance, A.; van Damme, J.; Serhan, C.N. Tonic inhibition of chemotaxis in human plasma. Proc. Natl. Acad. Sci. USA 2008, 105, 17949–17954. [Google Scholar] [CrossRef] [PubMed]
- Lämmermann, T.; Afonso, P.V.; Angermann, B.R.; Wang, J.M.; Kastenmüller, W.; Parent, C.A.; Germain, R.N. Neutrophil swarms require LTB4 and integrins at sites of cell death in vivo. Nature 2013, 498, 371–375. [Google Scholar] [CrossRef] [PubMed]
- Wall, R.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Fatty acids from fish: The anti-inflammatory potential of long-chain omega-3 fatty acids. Nutr. Rev. 2010, 68, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.; Wall, R.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. Health implications of high dietary omega-6 polyunsaturated fatty acids. J. Nutr. Metabol. 2012, 2012, 539426. [Google Scholar] [CrossRef]
- Anderson, B.M.; Ma, D.W.L. Are all n-3 polyunsaturated fatty acids created equal? Lipids Health Dis. 2009, 8, 33. [Google Scholar] [CrossRef]
- Lee, T.H.; Hoover, R.L.; Williams, J.D.; Sperling, R.I.; Ravalese Iii, J.; Spur, B.W.; Robinson, D.R.; Corey, E.J.; Lewis, R.A.; Austen, K.F. Effect of dietary enrichment with eicosapentaenoic and docosahexaenoic acids on in vitro neutrophil and monocyte leukotriene generation and neutrophil function. N. Engl. J. Med. 1985, 312, 1217–1224. [Google Scholar] [CrossRef]
- L’Homme, L.; Esser, N.; Riva, L.; Scheen, A.; Paquot, N.; Piette, J.; Legrand-Poels, S. Unsaturated fatty acids prevent activation of NLRP3 inflammasome in human monocytes/macrophages. J. Lipid Res. 2013, 54, 2998–3008. [Google Scholar] [CrossRef]
- Sweeney, S.E.; Firestein, G.S. Rheumatoid arthritis: Regulation of synovial inflammation. Int. J. Biochem. Cell Biol. 2004, 36, 372–378. [Google Scholar] [CrossRef]
- Dell’Agli, M.; Fagnani, R.; Mitro, N.; Scurati, S.; Masciadri, M.; Mussoni, L.; Galli, G.V.; Bosisio, E.; Crestani, M.; De Fabiani, E. Minor components of olive oil modulate proatherogenic adhesion molecules involved in endothelial activation. J. Agric. Food Chem. 2006, 54, 3259–3264. [Google Scholar] [CrossRef]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A.; Massaro, M.; Scoditti, E.; Storelli, C.; Visioli, F.; Distante, A.; De Caterina, R. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: Antiatherogenic properties of Mediterranean diet phytochemicals. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 622–629. [Google Scholar] [CrossRef]
- Serhan, C.N.; Chiang, N.; Dalli, J. The Resolution Code of Acute Inflammation: Novel Pro-Resolving Lipid Mediators in Resolution; Academic Press: Boston, MA, USA, 2015; pp. 200–215. [Google Scholar]
- Volker, D.; Fitzgerald, P.; Major, G.; Garg, M. Efficacy of fish oil concentrate in the treatment of rheumatoid arthritis. J. Rheum. 2000, 27, 2343–2346. [Google Scholar] [PubMed]
- Adam, O.; Beringer, C.; Kless, T.; Lemmen, C.; Adam, A.; Wiseman, M.; Adam, P.; Klimmek, R.; Forth, W. Anti-inflammatory effects of a low arachidonic acid diet and fish oil in patients with rheumatoid arthritis. Rheumatol. Int. 2003, 23, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Berbert, A.A.; Kondo, C.R.M.; Almendra, C.L.; Matsuo, T.; Dichi, I. Supplementation of fish oil and olive oil in patients with rheumatoid arthritis. Nutrition 2005, 21, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Proudman, S.M.; James, M.J.; Spargo, L.D.; Metcalf, R.G.; Sullivan, T.R.; Rischmueller, M.; Flabouris, K.; Wechalekar, M.D.; Lee, A.T.; Cleland, L.G. Fish oil in recent onset rheumatoid arthritis: A randomised, double-blind controlled trial within algorithm-based drug use. Ann. Rheum. Dis. 2015, 74, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Kremer, J.M.; Lawrence, D.A.; Petrillo, G.F.; Litts, L.L.; Mullaly, P.M.; Rynes, R.I.; Stocker, R.P.; Parhami, N.; Greenstein, N.S.; Fuchs, B.R. Effects of high-dose fish oil on rheumatoid arthritis after stopping nonsteroidal antiinflammatory drugs clinical and immune correlates. Arthritis Rheum. 1995, 38, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Miles, E.A.; Calder, P.C. Influence of marine n-3 polyunsaturated fatty acids on immune function and a systematic review of their effects on clinical outcomes in rheumatoid arthritis. Br. J. Nutr. 2012, 107, S171–S184. [Google Scholar] [CrossRef]
- Goldberg, R.J.; Katz, J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain 2007, 129, 210–223. [Google Scholar] [CrossRef]
- Park, Y.; Lee, A.; Shim, S.-C.; Lee, J.H.; Choe, J.-Y.; Ahn, H.; Choi, C.B.; Sung, Y.K.; Bae, S.C. Effect of n-3 polyunsaturated fatty acid supplementation in patients with rheumatoid arthritis: A 16-week randomized, double-blind, placebo-controlled, parallel-design multicenter study in Korea. J. Nutr.Biochem. 2013, 24, 1367–1372. [Google Scholar] [CrossRef]
- Kjeldsen-Kragh, J.; Borchgrevink, C.F.; Laerum, E.; Haugen, M.; Eek, M.; Førre, O.; Mowinkel, P.; Hovi, K. Controlled trial of fasting and one-year vegetarian diet in rheumatoid arthritis. Lancet 1991, 338, 899–902. [Google Scholar] [CrossRef]
- Sköldstam, L.; Larsson, L.; Lindström, F.D. Effects of fasting and lactovegetarian diet on rheumatoid arthritis. Scand. J. Rheumatol. 1979, 8, 249–255. [Google Scholar] [CrossRef]
- Müller, H.; de Toledo, F.W.; Resch, K.L. Fasting followed by vegetarian diet in patients with rheumatoid arthritis: A systematic review. Scand. J. Rheumatol. 2001, 30, 1–10. [Google Scholar] [PubMed]
- Youm, Y.-H.; Nguyen, K.Y.; Grant, R.W.; Goldberg, E.L.; Bodogai, M.; Kim, D.; D’Agostino, D.; Planavsky, N.; Lupfer, C.; Kanneganti, T.D. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome–mediated inflammatory disease. Nat. Med. 2015, 21, 263. [Google Scholar] [CrossRef] [PubMed]
- Vandanmagsar, B.; Youm, Y.-H.; Ravussin, A.; Galgani, J.E.; Stadler, K.; Mynatt, R.L.; Ravussin, E.; Stephens, J.M.; Dixit, V.D. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 2011, 17, 179. [Google Scholar] [CrossRef] [PubMed]
- Ravussin, E.; Redman, L.M.; Rochon, J.; Das, S.K.; Fontana, L.; Kraus, W.E.; Romashkan, S.; Williamson, D.A.; Meydani, S.N.; Villareal, D.T. A 2-year randomized controlled trial of human caloric restriction: Feasibility and effects on predictors of health span and longevity. J. Gerontol. Ser. A 2015, 70, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, K.; Aggarwal, B.B.; Singh, R.B.; Buttar, H.S.; Wilson, D.; De Meester, F. Food antioxidants and their anti-inflammatory properties: A potential role in cardiovascular diseases and cancer prevention. Diseases 2016, 4, 28. [Google Scholar] [CrossRef] [PubMed]
- Reis, J.F.; Monteiro, V.V.S.; de Souza Gomes, R.; do Carmo, M.M.; da Costa, G.V.; Ribera, P.C.; Monteiro, M.C. Action mechanism and cardiovascular effect of anthocyanins: A systematic review of animal and human studies. J. Transl. Med. 2016, 14, 315. [Google Scholar] [CrossRef]
- De Lorenzo, A.; Bernardini, S.; Gualtieri, P.; Cabibbo, A.; Perrone, M.A.; Giambini, I.; Di Renzo, L. Mediterranean meal versus Western meal effects on postprandial ox-LDL, oxidative and inflammatory gene expression in healthy subjects: A randomized controlled trial for nutrigenomic approach in cardiometabolic risk. Acta Diabetol. 2017, 54, 141–149. [Google Scholar] [CrossRef]
- Mateen, S.; Moin, S.; Shahzad, S.; Khan, A.Q. Level of inflammatory cytokines in rheumatoid arthritis patients: Correlation with 25-hydroxy vitamin D and reactive oxygen species. PLoS ONE 2017, 12, e0178879. [Google Scholar] [CrossRef]
- Jalili, M.; Kolahi, S.; Aref-Hosseini, S.-R.; Mamegani, M.E.; Hekmatdoost, A. Beneficial role of antioxidants on clinical outcomes and erythrocyte antioxidant parameters in rheumatoid arthritis patients. Int. J. Prev. Med. 2014, 5, 835. [Google Scholar]
- Keysser, G. Are there effective dietary recommendations for patients with rheumatoid arthritis? Z. Rheumatol. 2001, 60, 17–27. [Google Scholar]
- Sahebari, M.; Ayati, R.; Mirzaei, H.; Sahebkar, A.; Hejazi, S.; Saghafi, M.; Saadati, N.; Ferns, G.A.; Ghayour-Mobarhan, M. Serum trace element concentrations in rheumatoid arthritis. Biol. Trace Elem. Res. 2016, 171, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Prescha, A.; Zabłocka-Słowińska, K.; Płaczkowska, S.; Gorczyca, D.; Łuczak, A.; Grajeta, H. Silicon intake and plasma level and their relationships with systemic redox and inflammatory markers in rheumatoid arthritis patients. Adv. Clin. Exp. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Aryaeian, N.; Shahram, F.; Mahmoudi, M.; Tavakoli, H.; Yousefi, B.; Arablou, T.; Karegar, S.J. The effect of ginger supplementation on some immunity and inflammation intermediate genes expression in patients with active Rheumatoid Arthritis. Gene 2019, 698, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Gang, D.; Yu, X.; Hu, Y.; Yue, Y.; Cheng, W.; Pan, X.; Zhang, P. Genistein: The potential for efficacy in rheumatoid arthritis. Clin. Rheum. 2013, 32, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Dong, J.; He, P.; Li, W.; Zhang, Q.; Li, N.; Sun, T. Genistein inhibit cytokines or growth factor-induced proliferation and transformation phenotype in fibroblast-like synoviocytes of rheumatoid arthritis. Inflammation 2012, 35, 377–387. [Google Scholar] [CrossRef]
- Warjri, S.B.; Ete, T.; Beyong, T.; Barman, B.; Lynrah, K.G.; Nobin, H.; Perme, O. Coeliac disease with rheumatoid arthritis: An unusual association. Gastroenterol. Res. 2015, 8, 167. [Google Scholar] [CrossRef][Green Version]
- El-Chammas, K.; Danner, E. Gluten-free diet in nonceliac disease. Nutr. Clin. Pract. 2011, 26, 294–299. [Google Scholar] [CrossRef]
- Elkan, A.-C.; Sjöberg, B.; Kolsrud, B.; Ringertz, B.; Hafström, I.; Frostegård, J. Gluten-free vegan diet induces decreased LDL and oxidized LDL levels and raised atheroprotective natural antibodies against phosphorylcholine in patients with rheumatoid arthritis: A randomized study. Arthritis Res. Ther. 2008, 10, R34. [Google Scholar] [CrossRef]
- Tripathy, A.; Khanna, S.; Padhan, P.; Smita, S.; Raghav, S.; Gupta, B. Direct recognition of LPS drive TLR4 expressing CD8+ T cell activation in patients with rheumatoid arthritis. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Longo, V.D.; Mattson, M.P. Fasting: Molecular mechanisms and clinical applications. Cell Metabol. 2014, 19, 181–192. [Google Scholar] [CrossRef]
- Toivanen, P.; Eerola, E. A vegan diet changes the intestinal flora. Rheumatology 2002, 41, 950–951. [Google Scholar] [CrossRef] [PubMed]
- McDougall, J.; Bruce, B.; Spiller, G.; Westerdahl, J.; McDougall, M. Effects of a very low-fat, vegan diet in subjects with rheumatoid arthritis. J. Altern. Complement. Med. 2002, 8, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Liu, J.; Davies, M.L.; Chen, W. Serum vitamin D level and rheumatoid arthritis disease activity: Review and meta-analysis. PLoS ONE 2016, 11, e0146351. [Google Scholar] [CrossRef] [PubMed]
- Di Franco, M.; Barchetta, I.; Iannuccelli, C.; Gerardi, M.C.; Frisenda, S.; Ceccarelli, F.; Valesini, G.; Cavallo, M.G. Hypovitaminosis D in recent onset rheumatoid arthritis is predictive of reduced response to treatment and increased disease activity: A 12 month follow-up study. BMC Musculoskelet. Disord. 2015, 16, 53. [Google Scholar] [CrossRef]
- Franco, A.S.; Freitas, T.Q.; Bernardo, W.M.; Pereira, R.M.R. Vitamin D supplementation and disease activity in patients with immune-mediated rheumatic diseases: A systematic review and meta-analysis. Medicine 2017, 96, e7024. [Google Scholar] [CrossRef]
- Allan, G.M.; Cranston, L.; Lindblad, A.; McCormack, J.; Kolber, M.R.; Garrison, S.; Korownyk, C. Vitamin D: A narrative review examining the evidence for ten beliefs. J.Gen. Intern. Med. 2016, 31, 780–791. [Google Scholar] [CrossRef][Green Version]
- Imhof, A.; Froehlich, M.; Brenner, H.; Boeing, H.; Pepys, M.B.; Koenig, W. Effect of alcohol consumption on systemic markers of inflammation. Lancet 2001, 357, 763–767. [Google Scholar] [CrossRef]
- Sierksma, A.; Patel, H.; Ouchi, N.; Kihara, S.; Funahashi, T.; Heine, R.J.; Grobbee, D.E.; Kluft, C.; Hendriks, H.F.J. Effect of moderate alcohol consumption on adiponectin, tumor necrosis factor-α, and insulin sensitivity. Diabetes Care 2004, 27, 184–189. [Google Scholar] [CrossRef]
- Brien, S.E.; Ronksley, P.E.; Turner, B.J.; Mukamal, K.J.; Ghali, W.A. Effect of alcohol consumption on biological markers associated with risk of coronary heart disease: Systematic review and meta-analysis of interventional studies. BMJ 2011, 342, d636. [Google Scholar] [CrossRef]
- Lu, B.; Rho, Y.H.; Cui, J.; Iannaccone, C.K.; Frits, M.L.; Karlson, E.W.; Shadick, N.A. Associations of smoking and alcohol consumption with disease activity and functional status in rheumatoid arthritis. J. Rheum. 2014, 41, 24–30. [Google Scholar] [CrossRef]
- Bergman, S.; Symeonidou, S.; Andersson, M.L.; Söderlin, M.K.; BARFOT Study Group. Alcohol consumption is associated with lower self-reported disease activity and better health-related quality of life in female rheumatoid arthritis patients in Sweden: Data from BARFOT, a multicenter study on early RA. BMC Musculoskelet. Disord. 2013, 14, 218. [Google Scholar] [CrossRef]
- Sanders, M.E. Probiotics: Considerations for human health. Nutr. Rev. 2003, 61, 91–99. [Google Scholar] [CrossRef]
- Kullisaar, T.; Songisepp, E.; Zilmer, M. Oxidative stress-Environmental induction and dietary antioxidants In Probiotics and Oxidative Stress; Lushchak, V., Ed.; INTECH Open Acces Publisher: Tartu, Estonia, 2012; pp. 203–222. [Google Scholar]
- Mazloom, Z.; Yousefinejad, A.; Dabbaghmanesh, M.H. Effect of probiotics on lipid profile, glycemic control, insulin action, oxidative stress, and inflammatory markers in patients with type 2 diabetes: A clinical trial. Iran. J. Med. Sci. 2013, 38, 38. [Google Scholar]
- Mohammed, A.T.; Khattab, M.; Ahmed, A.M.; Turk, T.; Sakr, N.; Khalil, A.M.; Abdelhalim, M.; Sawaf, B.; Hirayama, K.; Huy, N.T. The therapeutic effect of probiotics on rheumatoid arthritis: A systematic review and meta-analysis of randomized control trials. Clin. Rheum. 2017, 36, 2697–2707. [Google Scholar] [CrossRef]
- Alipour, B.; Homayouni-Rad, A.; Vaghef-Mehrabany, E.; Sharif, S.K.; Vaghef-Mehrabany, L.; Asghari-Jafarabadi, M.; Nakhjavani, M.R.; Mohtadi-Nia, J. Effects of L actobacillus casei supplementation on disease activity and inflammatory cytokines in rheumatoid arthritis patients: A randomized double-blind clinical trial. Int. J. Rheum. Dis. 2014, 17, 519–527. [Google Scholar]
- Vaghef-Mehrabany, E.; Vaghef-Mehrabany, L.; Asghari-Jafarabadi, M.; Homayouni-Rad, A.; Issazadeh, K.; Alipour, B. Effects of probiotic supplementation on lipid profile of women with rheumatoid arthritis: A randomized placebo-controlled clinical trial. Health Promot. Perspect. 2017, 7, 95. [Google Scholar] [CrossRef]
- Vaghef-Mehrabany, E.; Homayouni-Rad, A.; Alipour, B.; Sharif, S.-K.; Vaghef-Mehrabany, L.; Alipour-Ajiry, S. Effects of probiotic supplementation on oxidative stress indices in women with rheumatoid arthritis: A randomized double-blind clinical trial. J. Am. Coll. Nutr. 2016, 35, 291–299. [Google Scholar] [CrossRef]
- Vadell, A.K.E.; Bärebring, L.; Hulander, E.; Gjertsson, I.; Lindqvist, H.M.; Winkvist, A. Anti-inflammatory Diet In Rheumatoid Arthritis (ADIRA)—A randomized, controlled crossover trial indicating effects on disease activity. Am. J. Clin. Nutr. 2020. [Google Scholar] [CrossRef]
- Di Carlo, M.; Salaffi, F.; Gremese, E.; Iannone, F.; Lapadula, G.; Ferraccioli, G.; Group, G.S. Body mass index as a driver of selection of biologic therapy in rheumatoid arthritis. Results from the US-CLARA study. Eur. J. Int. Med. 2019, 66, 57–61. [Google Scholar] [CrossRef]
- Liu, Y.; Hazlewood, G.S.; Kaplan, G.G.; Eksteen, B.; Barnabe, C. Impact of obesity on remission and disease activity in rheumatoid arthritis: A systematic review and meta-analysis. Arthritis Care Res. 2017, 69, 157–165. [Google Scholar] [CrossRef]
| Study | Duration | Population | Evaluation Method | Nutrients/Foods | Outcomes (Effect on RA Risk) |
|---|---|---|---|---|---|
| Pattison et al. (2004) [10] | 9 years (1993–2002) | 25630 subjects (EPIC-Norfolk) (88 with IP and 1:2 ratio with healthy controls) | Prospective 7-day food diary | 1. Red meat 2. Protein intake 3. Vitamin C | 1. Harmful effect 2. Harmful effect 3. Protective effect |
| He J et al. (2016) [34] | 1 year (2012–2013) | 968 RA patients and 1037 healthy controls | Self-administrated weakly retrospective FFQ (over the last 5 years before RA onset) | 1. Potatoes 2. Fruits (no citrus fruits) 3. Citrus fruits 4. Mushrooms 5. Dairy products 6. Red meats 7. Vegetables | 1. Harmful effect 2. No effects 3. Protective effect 4. Protective effect 5. Protective effect 6. No effects 7. No effects |
| Benito-Garcia et al. (2007) [12] | 22 years (1980–2002) | 82,064 women in NHS (546 RA) | Prospective semi-quantitative FFQ at baseline and every 2 years in follow-up | 1. Proteins 2. Iron 3. Red meats 4. Fish | 1. No effects 2. No effects 3. No effects 4. No effects |
| Study | Duration | Type | Dose gr/daily | Therapy | N Patients | Clinical Efficacy |
|---|---|---|---|---|---|---|
| Volker et al. (2000) [154] | 15 weeks | Double-blind randomized trial | >2 gr | DMARDs | * | Improvement HAQ, MS |
| Adam et al. (2003) [155] | 12 weeks | Double-blind crossover study | 30 mg/kg | DMARDs | 60 | TJ, SJ |
| Berbert et al. (2005) [156] | 12/24 weeks | Parallel randomized study | >3 gr, 9.6 gr olive oil | DMARDs | 43 | TJ, MS |
| Proudman et al. (2015) [157] | 12 months | Double-blind placebo-controlled trial | 5.5 gr | Triple therapy | 140 | Reduced failure of triple therapy |
| Kremer et al. (1995) [158] | 48 weeks | Double-blind randomized trial | > 9 gr | NSAIDs | 66 | TJ, VAS physician |
| Miles et al. (2012) [159] | * | Systematic review (23 studies) | * | NSAID, DMARDs | * | TJ, SJ, MS, joint pain |
| Goldbeg et al. (2007) [160] | 3–4 months | Meta-analysis (17 RCT) | * | NSAID, DMARDs | * | TJ, SJ, MS, joint pain, NSAID use |
| Park et al. (2013) [161] | 16 weeks | Double-blind placebo controlled randomized trial | 2.1 gr EPA + 1.2 gr DHA | NSAID, DMARDs | 109 | Decreased use of NSAIDs |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gioia, C.; Lucchino, B.; Tarsitano, M.G.; Iannuccelli, C.; Di Franco, M. Dietary Habits and Nutrition in Rheumatoid Arthritis: Can Diet Influence Disease Development and Clinical Manifestations? Nutrients 2020, 12, 1456. https://doi.org/10.3390/nu12051456
Gioia C, Lucchino B, Tarsitano MG, Iannuccelli C, Di Franco M. Dietary Habits and Nutrition in Rheumatoid Arthritis: Can Diet Influence Disease Development and Clinical Manifestations? Nutrients. 2020; 12(5):1456. https://doi.org/10.3390/nu12051456
Chicago/Turabian StyleGioia, Chiara, Bruno Lucchino, Maria Grazia Tarsitano, Cristina Iannuccelli, and Manuela Di Franco. 2020. "Dietary Habits and Nutrition in Rheumatoid Arthritis: Can Diet Influence Disease Development and Clinical Manifestations?" Nutrients 12, no. 5: 1456. https://doi.org/10.3390/nu12051456
APA StyleGioia, C., Lucchino, B., Tarsitano, M. G., Iannuccelli, C., & Di Franco, M. (2020). Dietary Habits and Nutrition in Rheumatoid Arthritis: Can Diet Influence Disease Development and Clinical Manifestations? Nutrients, 12(5), 1456. https://doi.org/10.3390/nu12051456






