Influence of Hesperidin on Systemic Immunity of Rats Following an Intensive Training and Exhausting Exercise
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Diet and Hesperidin Supplementation
2.3. Exercise Training Program and Samples Collection
2.4. Lymphocyte Composition in Blood, Thymus and Spleen
2.5. Phagocytic Activity
2.6. Macrophage Cytokine Production
2.7. Natural Killer (NK) Cell Cytotoxic Activity
2.8. Spleen Lymphocyte Proliferation
2.9. Plasma Cortisol Concentration
2.10. Statistical Analysis
3. Results
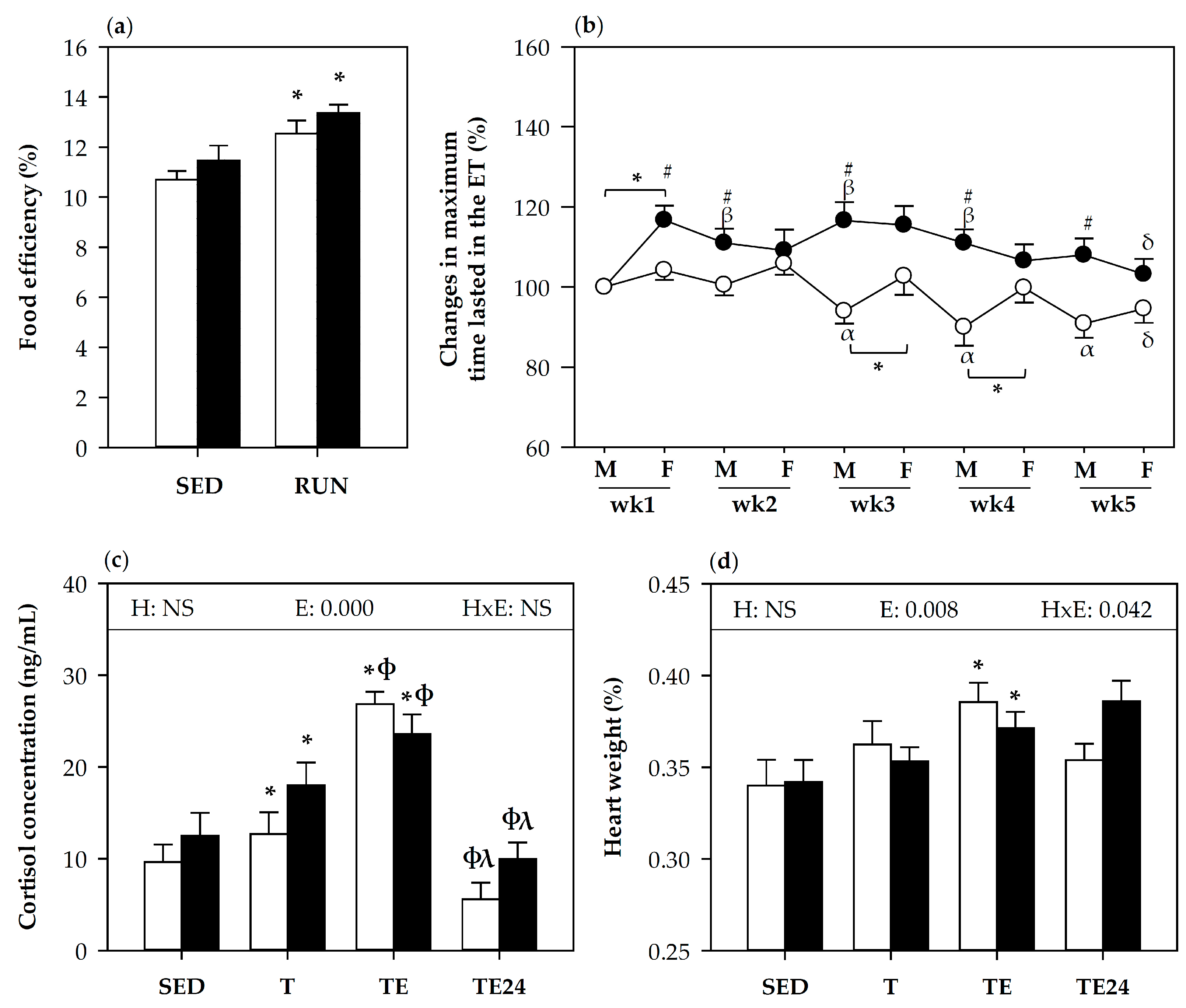
3.1. Effect of Hesperidin Supplementation on Food Efficiency and Training Performance
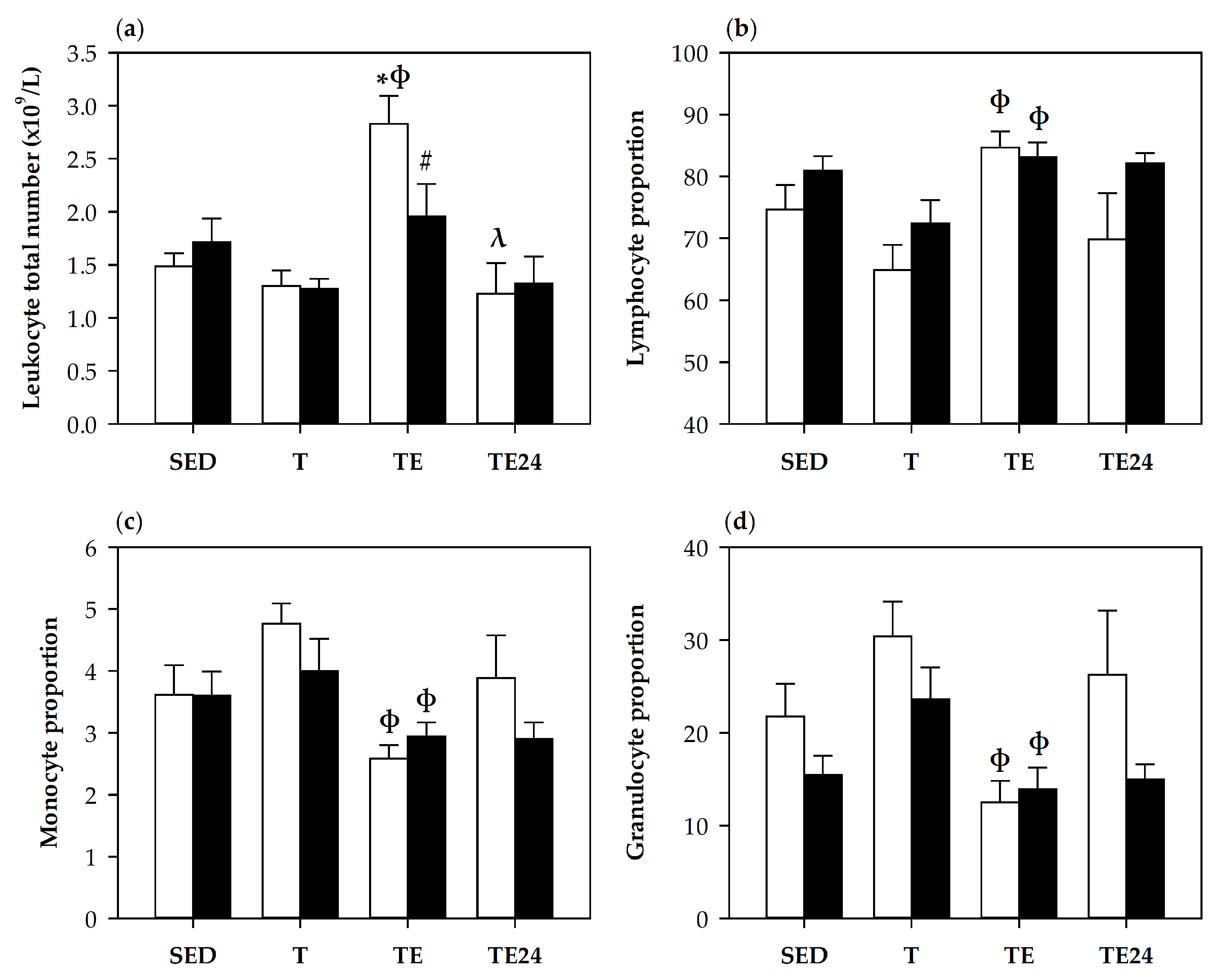
3.2. Blood Leukocytes Count and Lymphocyte Proportions
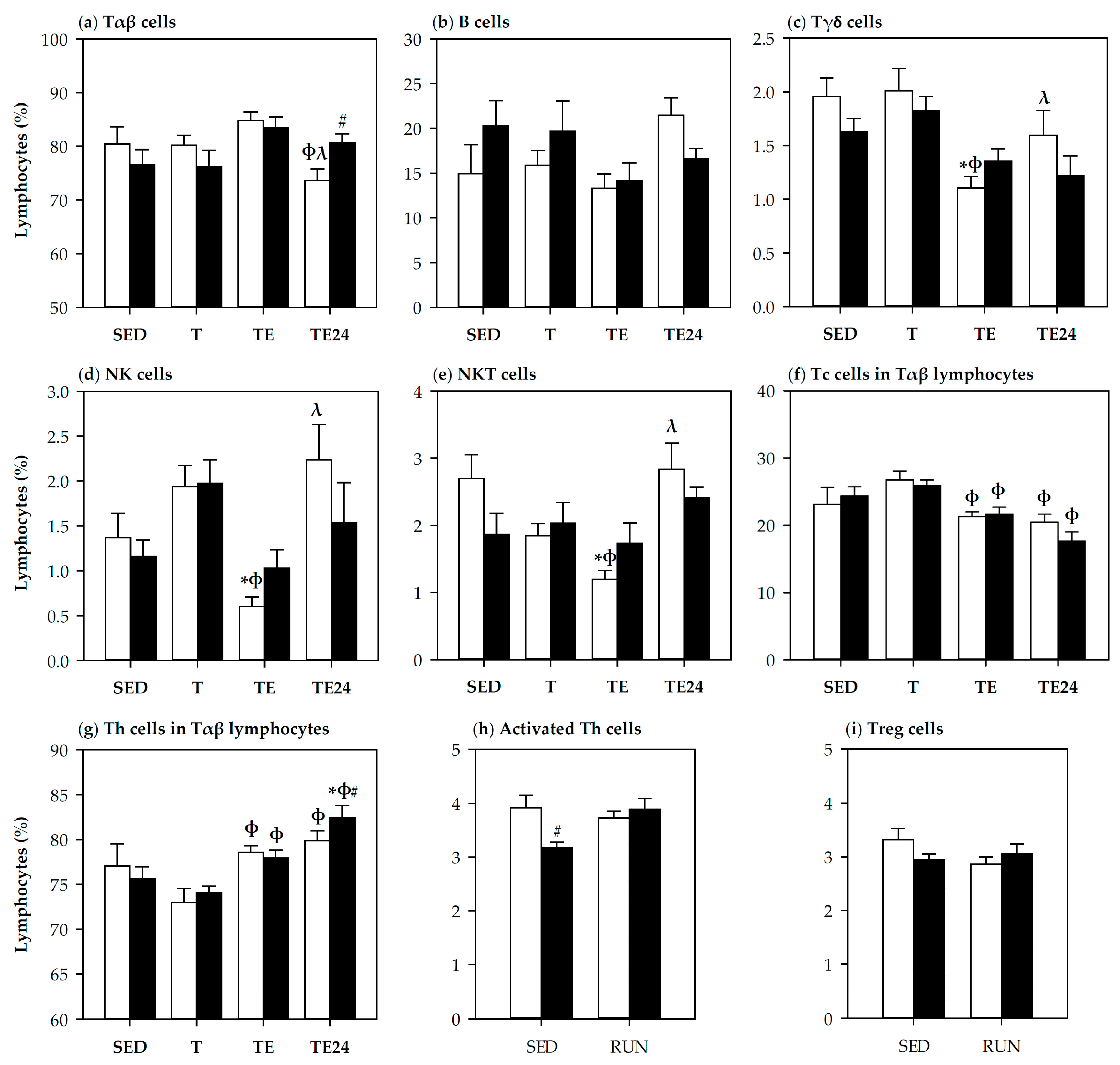
3.3. Lymphocyte Composition in Thymus and Spleen
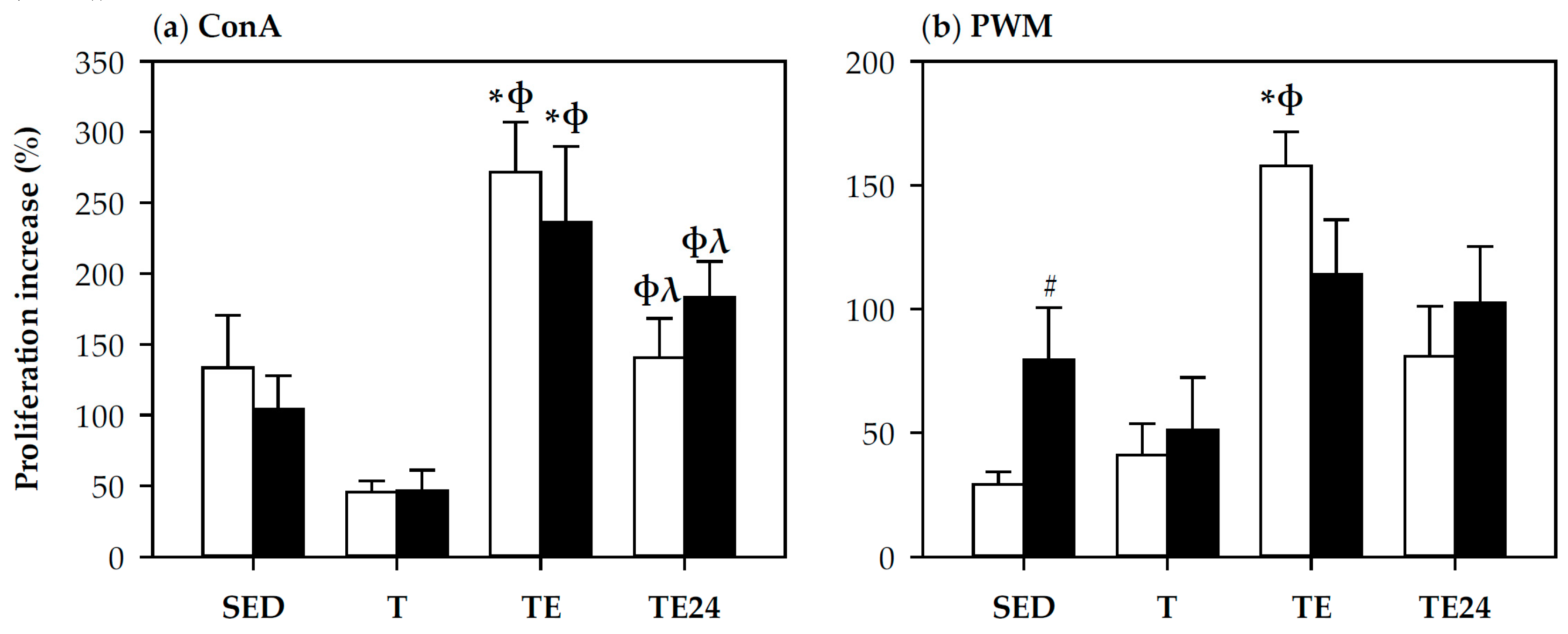
3.4. Spleen Lymphocyte Proliferation
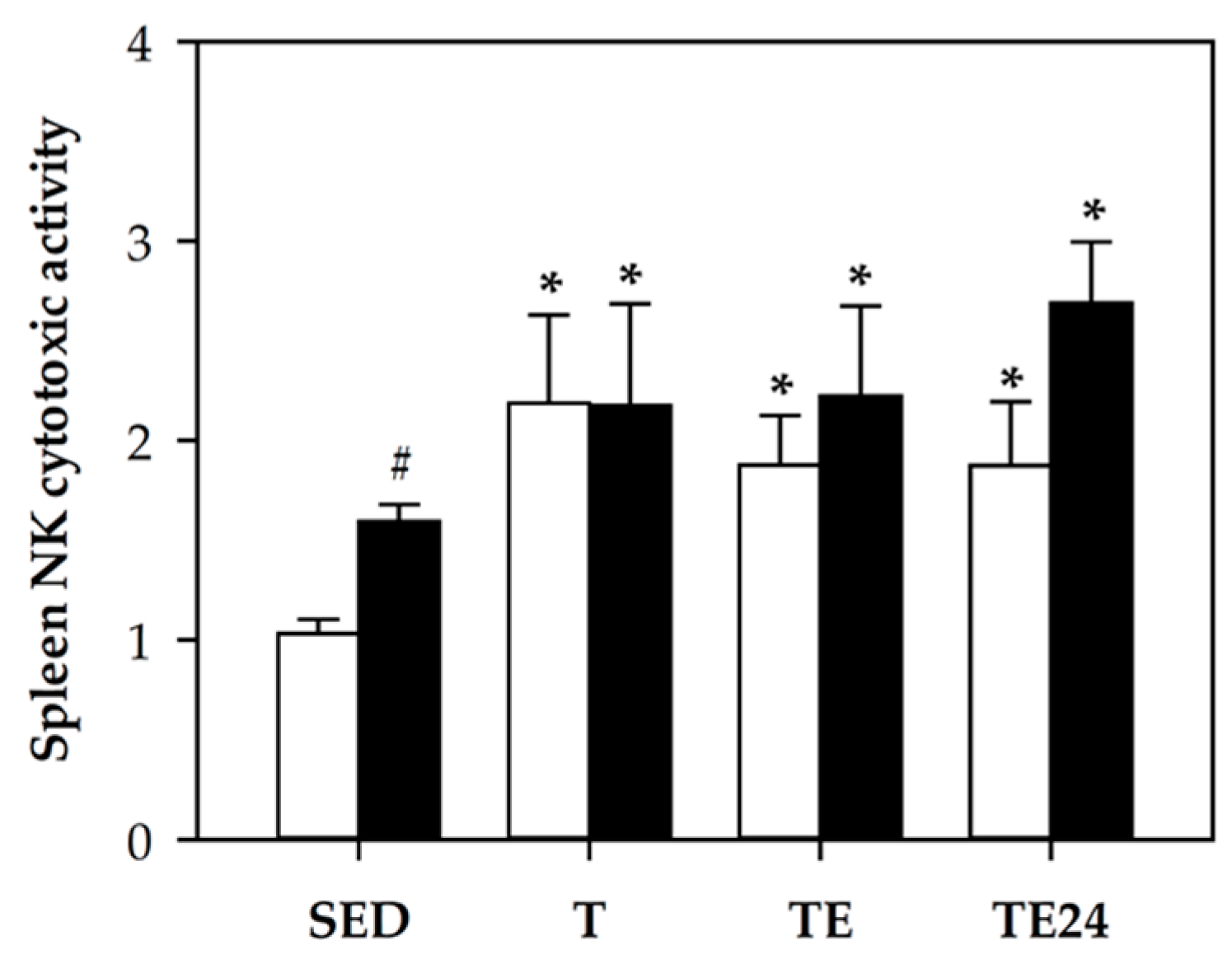
3.5. NK Cell Cytotoxic Activity
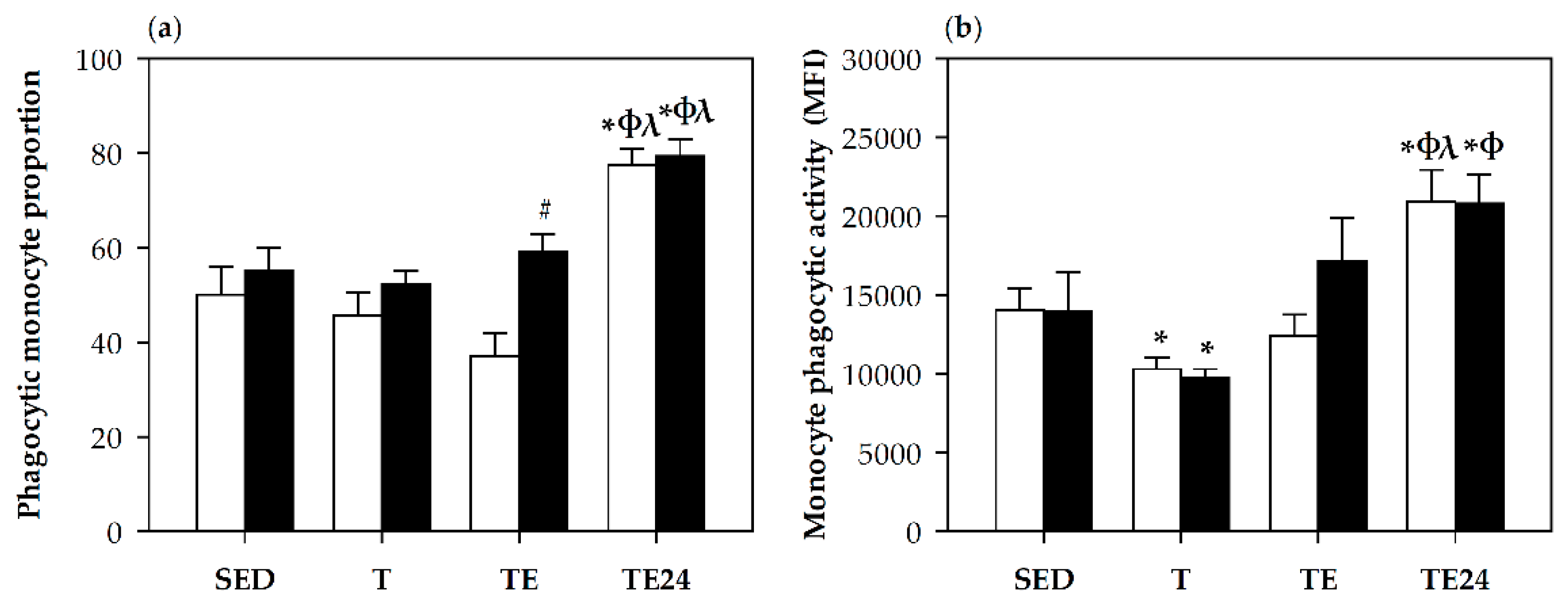
3.6. Phagocytic Activity
3.7. Macrophage Cytokine Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Warburton, D.E.R.; Nicol, C.W.; Bredin, S.S.D. Health benefits of physical activity: The evidence. Can. Med. Assoc. J. 2006, 174, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Miles, L. Physical activity and health. Br. Nutr. Found. 2007, 32, 314–363. [Google Scholar] [CrossRef]
- Pape, K.; Ryttergaard, L.; Rotevatn, T.A.; Nielsen, B.J.; Torp-Pedersen, C.; Overgaard, C.; BØggild, H. Leisure-time physical activity and the risk of suspected bacterial infections. Med. Sci. Sports Exerc. 2016, 48, 1737–1744. [Google Scholar] [CrossRef] [PubMed]
- Walsh, N.P.; Gleeson, M.; Shephard, R.J.; Gleeson, M.; Woods, J.A.; Bishop, N.C.; Fleshner, M.; Green, C.; Pedersen, B.K.; Hoffman-Goetz, L.; et al. Position statement part one: Immune function and exercise. Exerc. Immunol. Rev. 2011, 17, 6–63. [Google Scholar]
- Walsh, N.P.; Gleeson, M.; Pyne, D.B.; Nieman, D.C.; Dhabhar, S.; Shephard, R.J.; Oliver, S.J.; Bermon, S.; Kajeniene, A. Position statement part two: Maintaining immune health. Exerc. Immunol. Rev. 2011, 17, 64–103. [Google Scholar]
- Estruel-Amades, S.; Camps-Bossacoma, M.; Massot-Cladera, M.; Pérez-Cano, F.J.; Castell, M. Alterations in the innate immune system due to exhausting exercise in intensively trained rats. Sci. Rep. 2020, 10, 967. [Google Scholar] [CrossRef]
- Estruel-Amades, S.; Ruiz-Iglesias, P.; Périz, M.; Franch, À.; Pérez-Cano, F.J.; Camps-Bossacoma, M.; Castell, M. Changes in lymphocyte composition and functionality after intensive training and exhausting exercise in rats. Front. Physiol. 2019, 10, 1491. [Google Scholar] [CrossRef]
- Schwellnus, M.; Soligard, T.; Alonso, J.-M.; Bahr, R.; Clarsen, B.; Dijkstra, H.P.; Gabbett, T.J.; Gleeson, M.; Hägglund, M.; Hutchinson, M.R.; et al. How much is too much? (Part 2) International olympic committee consensus statement on load in sport and risk of illness. Br. J. Sports Med. 2016, 50, 1043–1052. [Google Scholar] [CrossRef]
- Cantó, E.; Roca, E.; Perea, L.; Rodrigo-Troyano, A.; Suarez-Cuartin, G.; Giner, J.; Feliu, A.; Soria, J.M.; Nescolarde, L.; Vidal, S.; et al. Salivary immunity and lower respiratory tract infections in non-elite marathon runners. PLoS ONE 2018, 13, e0206059. [Google Scholar] [CrossRef]
- Campbell, J.P.; Turner, J.E. Debunking the myth of exercise-induced immune suppression: Redefining the impact of exercise on immunological health across the lifespan. Front. Immunol. 2018, 9, 648. [Google Scholar] [CrossRef]
- Kawamura, T.; Muraoka, I. Exercise-induced oxidative stress and the effects of antioxidant intake from a physiological viewpoint. Antioxidants 2018, 7, 119. [Google Scholar] [CrossRef] [PubMed]
- Nieman, D.; Mitmesser, S. Potential impact of nutrition on immune system recovery from heavy exertion: A metabolomics perspective. Nutrients 2017, 9, 513. [Google Scholar] [CrossRef] [PubMed]
- De Ferrars, R.M.; Czank, C.; Zhang, Q.; Botting, N.P.; Kroon, P.A.; Cassidy, A.; Kay, C.D. The pharmacokinetics of anthocyanins and their metabolites in humans. Br. J. Pharmacol. 2014, 171, 3268–3282. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.M.; Carlstedt, C.J.; Chen, S.; Carmichael, M.D.; Murphy, E.A. The dietary flavonoid quercetin increases VO(2max) and endurance capacity. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 56–62. [Google Scholar] [CrossRef]
- Nieman, D.C.; Williams, A.S.; Shanely, R.A.; Jin, F.; McAnulty, S.R.; Triplett, N.T.; Austin, M.D.; Henson, D.A. Quercetin’s influence on exercise performance and muscle mitochondrial biogenesis. Med. Sci. Sports Exerc. 2010, 42, 338–345. [Google Scholar] [CrossRef]
- Bowtell, J.L.; Sumners, D.P.; Dyer, A.; Fox, P.; Mileva, K.N. Montmorency cherry juice reduces muscle damage caused by intensive strength exercise. Med. Sci. Sports Exerc. 2011, 43, 1544–1551. [Google Scholar] [CrossRef]
- Parhiz, H.; Roohbakhsh, A.; Soltani, F.; Rezaee, R.; Iranshahi, M. Antioxidant and anti-inflammatory properties of the citrus flavonoids hesperidin and hesperetin: An updated review of their molecular mechanisms and experimental models. Phyther. Res. 2015, 29, 323–331. [Google Scholar] [CrossRef]
- Camps-Bossacoma, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Influence of hesperidin on the systemic and intestinal rat immune response. Nutrients 2017, 9, 580. [Google Scholar] [CrossRef]
- Estruel-Amades, S.; Massot-Cladera, M.; Pérez-Cano, F.J.; Franch, À.; Castell, M.; Camps-Bossacoma, M. Hesperidin effects on gut microbiota and gut-associated lymphoid tissue in healthy rats. Nutrients 2019, 11, 324. [Google Scholar] [CrossRef]
- Overdevest, E.; Wouters, J.A.; Wolfs, K.H.M.; van Leeuwen, J.J.M.; Possemiers, S. Citrus flavonoid supplementation improves exercise performance in trained athletes. J. Sports Sci. Med. 2018, 17, 24–30. [Google Scholar]
- De Oliveira, D.M.; Dourado, G.K.Z.S.; Cesar, T.B. Hesperidin associated with continuous and interval swimming improved biochemical and oxidative biomarkers in rats. J. Int. Soc. Sports Nutr. 2013, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Estruel-Amades, S.; Massot-Cladera, M.; Garcia-Cerdà, P.; Pérez-Cano, F.J.; Franch, Á.; Castell, M.; Camps-Bossacoma, M. Protective effect of hesperidin on the oxidative stress induced by an exhausting exercise in intensively trained rats. Nutrients 2019, 11, 783. [Google Scholar] [CrossRef] [PubMed]
- Camps-Bossacoma, M.; Abril-Gil, M.; Saldaña-Ruiz, S.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Cocoa diet prevents antibody synthesis and modifies lymph node composition and functionality in a rat oral sensitization model. Nutrients 2016, 8, 242. [Google Scholar] [CrossRef] [PubMed]
- Grases-Pintó, B.; Abril-Gil, M.; Rodríguez-Lagunas, M.J.; Castell, M.; Pérez-Cano, F.J.; Franch, À. Leptin and adiponectin supplementation modifies mesenteric lymph node lymphocyte composition and functionality in suckling rats. Br. J. Nutr. 2018, 119, 486–495. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Noguera, F.J.; Marín-Pagán, C.; Carlos-Vivas, J.; Rubio-Arias, J.A.; Alcaraz, P.E. Acute effects of hesperidin in oxidant/antioxidant state markers and performance in amateur cyclists. Nutrients 2019, 11, 1898. [Google Scholar] [CrossRef] [PubMed]
- Margaritelis, N.V.; Theodorou, A.A.; Paschalis, V.; Veskoukis, A.S.; Dipla, K.; Zafeiridis, A.; Panayiotou, G.; Vrabas, I.S.; Kyparos, A.; Nikolaidis, M.G. Adaptations to endurance training depend on exercise-induced oxidative stress: Exploiting redox interindividual variability. Acta Physiol. 2018, 222, e12898. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, A.; Saidullah, B.; Sultana, S. Anti-carcinogenic effect of hesperidin against renal cell carcinoma by targeting COX-2/PGE2 pathway in Wistar rats. Environ. Toxicol. 2018, 33, 1069–1077. [Google Scholar] [CrossRef]
- Fu, Z.; Chen, Z.; Xie, Q.; Lei, H.; Xiang, S. Hesperidin protects against IL-1β-induced inflammation in human osteoarthritis chondrocytes. Exp. Ther. Med. 2018, 16, 3721–3727. [Google Scholar] [CrossRef]
- Markworth, J.F.; Vella, L.D.; Figueiredo, V.C.; Cameron-Smith, D. Ibuprofen treatment blunts early translational signaling responses in human skeletal muscle following resistance exercise. J. Appl. Physiol. 2014, 117, 20–28. [Google Scholar] [CrossRef]
- Lilja, M.; Mandić, M.; Apró, W.; Melin, M.; Olsson, K.; Rosenborg, S.; Gustafsson, T.; Lundberg, T.R. High doses of anti-inflammatory drugs compromise muscle strength and hypertrophic adaptations to resistance training in young adults. Acta Physiol. 2018, 222, e12948. [Google Scholar] [CrossRef]
- Blank, S.E.; Johansson, J.O.; Pfister, L.J.; Gallucci, R.M.; Lee, E.G.; Meadows, G.G. Mechanistic differences in NK cell cytolytic activity in treadmill-trained and chronic ethanol-consuming mice. J. Appl Physiol. 1994, 76, 2031–2036. [Google Scholar] [CrossRef] [PubMed]
- Sassi, A.; Mokdad Bzéouich, I.; Mustapha, N.; Maatouk, M.; Ghedira, K.; Chekir-Ghedira, L. Immunomodulatory potential of hesperetin and chrysin through the cellular and humoral response. Eur. J. Pharmacol. 2017, 812, 91–96. [Google Scholar] [CrossRef] [PubMed]
- McAnulty, L.S.; Nieman, D.C.; Dumke, C.L.; Shooter, L.A.; Henson, D.A.; Utter, A.C.; Milne, G.; McAnulty, S.R. Effect of blueberry ingestion on natural killer cell counts, oxidative stress, and inflammation prior to and after 2.5 h of running. Appl. Physiol. Nutr. Metab. 2011, 36, 976–984. [Google Scholar] [CrossRef]
- Burkard, M.; Leischner, C.; Lauer, U.M.; Busch, C.; Venturelli, S.; Frank, J. Dietary flavonoids and modulation of natural killer cells: Implications in malignant and viral diseases. J. Nutr. Biochem. 2017, 46, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, C.; Bobrowska-Hägerstrand, M.; Mrówczyńska, L.; Engblom, C.; Hägerstrand, H. Potentiation of natural killer cell activity with Myricetin. Anticancer Res. 2014, 34, 3975–3980. [Google Scholar]
- Leischner, C.; Burkard, M.; Pfeiffer, M.M.; Lauer, U.M.; Busch, C.; Venturelli, S. Nutritional immunology: Function of natural killer cells and their modulation by resveratrol for cancer prevention and treatment. Nutr. J. 2016, 15, 47. [Google Scholar] [CrossRef]
- Perche, O.; Vergnaud-Gauduchon, J.; Morand, C.; Dubray, C.; Mazur, A.; Vasson, M.P. Orange juice and its major polyphenol hesperidin consumption do not induce immunomodulation in healthy well-nourished humans. Clin. Nutr. 2014, 33, 130–135. [Google Scholar] [CrossRef]
- Iranshahi, M.; Rezaee, R.; Parhiz, H.; Roohbakhsh, A.; Soltani, F. Protective effects of flavonoids against microbes and toxins: The cases of hesperidin and hesperetin. Life Sci. 2015, 137, 125–132. [Google Scholar] [CrossRef]
- Su, S.; Chen, H.; Jen, C.J. Severe exercise enhances phagocytosis by murine bronchoalveolar macrophages. J. Leukoc. Biol. 2001, 69, 75–80. [Google Scholar]
- Hassouna, I.; Ibrahim, H.; Abdel Gaffar, F.; El-Elaimy, I.; Abdel Latif, H. Simultaneous administration of hesperidin or garlic oil modulates diazinon-induced hemato- and immunotoxicity in rats. Immunopharmacol. Immunotoxicol. 2015, 37, 442–449. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Steensberg, A.; Schjerling, P. Muscle-derived interleukin-6: Possible biological effects. J. Physiol. 2001, 536, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Sahl, R.E.; Andersen, P.R.; Gronbaek, K.; Morville, T.H.; Rosenkilde, M.; Rasmusen, H.K.; Poulsen, S.S.; Prats, C.; Dela, F.; Helge, J.W. Repeated excessive exercise attenuates the anti-inflammatory effects of exercise in older men. Front. Physiol. 2017, 8, 407. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Steensberg, A.; Schjerling, P. Exercise and interleukin-6. Curr. Opin. Hematol. 2001, 8, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Akhtar, F.; Rizvi, S.I. Hesperidin attenuates altered redox homeostasis in an experimental hyperlipidaemic model of rat. Clin. Exp. Pharmacol. Physiol. 2020, 47, 571–582. [Google Scholar] [CrossRef]
- Shen, C.Y.; Jiang, J.G.; Huang, C.L.; Zhu, W.; Zheng, C.Y. Polyphenols from blossoms of Citrus aurantium L. var. amara Engl. show significant anti-complement and anti-inflammatory effects. J. Agric. Food Chem. 2017, 65, 9061–9068. [Google Scholar] [CrossRef]
- Xiao, S.; Liu, W.; Bi, J.; Liu, S.; Zhao, H.; Gong, N.; Xing, D.; Gao, H.; Gong, M. Anti-inflammatory effect of hesperidin enhances chondrogenesis of human mesenchymal stem cells for cartilage tissue repair. J. Inflamm. 2018, 15, 14. [Google Scholar] [CrossRef]
- Bai, X.; Yang, P.; Zhou, Q.; Cai, B.; Buist-Homan, M.; Cheng, H.; Jiang, J.; Shen, D.; Li, L.; Luo, X.; et al. The protective effect of the natural compound hesperetin against fulminant hepatitis in vivo and in vitro. Br. J. Pharmacol. 2017, 174, 41–56. [Google Scholar] [CrossRef]
- Da Cunha, L.R.; Muniz-Junqueira, M.I.; Dos Santos Borges, T.K. Impact of polyphenols in phagocyte functions. J. Inflamm. Res. 2019, 12, 205–217. [Google Scholar] [CrossRef]
- Dourado, G.K.Z.S.; Ribeiro, L.C.D.A.; Carlos, I.Z.; César, T.B. Orange juice and hesperidin promote differential innate immune response in macrophages ex vivo. Int. J. Vitam. Nutr. Res. 2014, 83, 162–167. [Google Scholar] [CrossRef]
- Kong, L.N.; Lin, X.; Huang, C.; Ma, T.T.; Meng, X.M.; Hu, C.J.; Wang, Q.Q.; Liu, Y.H.; Shi, Q.P.; Li, J. Hesperetin derivative-12 (HDND-12) regulates macrophage polarization by modulating JAK2/STAT3 signaling pathway. Chin. J. Nat. Med. 2019, 17, 122–130. [Google Scholar] [CrossRef]
- Shephard, R.J. Adhesion molecules, catecholamines and leucocyte redistribution during and following exercise. Sports Med. 2003, 33, 261–284. [Google Scholar] [CrossRef] [PubMed]
- Krüger, K.; Mooren, F.C. T cell homing and exercise. Exerc. Immunol. Rev. 2007, 13, 37–54. [Google Scholar] [PubMed]
- Jaganathan, S.K.; Mandal, M. Antiproliferative effects of honey and of its polyphenols: A review. J. Biomed. Biotechnol. 2009, 2009, 830616. [Google Scholar] [CrossRef] [PubMed]
| Thymus Lymphocyte Composition | |||||
|---|---|---|---|---|---|
| Hesperidin | SED | T | TE | TE24 | |
| % in total lymphocytes: | |||||
| TCRαβ+ | - | 12.35 ± 1.27 | 14.94 ± 1.34 | 10.19 ± 0.74 ϕ | 15.76 ± 1.13 λ |
| + | 15.11 ± 1.42 | 12.72 ± 0.64 | 9.23 ± 0.98 | 17.31 ± 1.42 λ | |
| DN | - | 3.62 ± 0.69 | 3.15 ± 0.45 | 2.82 ± 0.11 | 4.48 ± 0.43 λ |
| + | 3.90 ± 0.52 | 3.15 ± 0.35 | 2.24 ± 0.31 | 4.44 ± 0.38 λ | |
| DP | - | 81.85 ± 2.59 | 81.71 ± 1.88 | 84.11 ± 0.76 | 80.46 ± 1.48 |
| + | 81.71 ± 1.06 | 84.51 ± 1.06 | 86.73 ± 1.59 | 76.5 ± 2.21 | |
| TCD4+ | - | 8.90 ± 1.47 | 8.93 ± 0.91 | 6.62 ± 0.37 | 8.21 ± 0.81 |
| + | 10.36 ± 0.82 | 7.99 ± 0.71 | 5.93 ± 1.34 | 12.57 ± 0.98 | |
| TCD8+ | - | 5.62 ± 0.51 | 4.61 ± 0.61 | 6.25 ± 0.29 | 5.86 ± 0.39 |
| + | 4.53 ± 0.41 | 4.33 ± 0.32 | 4.11 ± 0.19 # | 6.16 ± 0.37 | |
| Spleen Lymphocyte Composition | |||||
|---|---|---|---|---|---|
| Hesperidin | SED | T | TE | TE24 | |
| % in total lymphocytes: | |||||
| T | - | 41.68 ± 1.72 | 40.41 ± 5.49 | 43.71 ± 2.68 | 43.24 ± 1.84 |
| + | 47.07 ± 2.13 | 45.47 ± 1.89 | 43.51 ± 2.64 | 50.91 ± 1.16 # | |
| B | - | 36.83 ± 1.63 | 32.06 ± 2.06 | 38.10 ± 2.20 | 37.24 ± 1.97 |
| + | 33.35 ± 1.96 | 34.46 ± 2.18 | 37.60 ± 1.62 | 32.41 ± 1.21 | |
| Tγδ | - | 3.95 ± 0.22 | 3.32 ± 0.48 | 3.54 ± 0.26 | 2.89 ± 0.20 * |
| + | 3.64 ± 0.24 | 3.24 ± 0.24 | 3.44 ± 0.12 | 2.78 ± 0.22 * | |
| NK | - | 7.92 ± 1.05 | 6.21 ± 0.90 | 5.28 ± 0.51 | 5.01 ± 0.80 ϕ |
| + | 6.43 ± 0.41 | 6.51± 1.13 | 5.70 ± 0.84 | 4.10 ± 0.41 ϕ | |
| NKT | - | 4.71 ± 0.80 | 3.29 ± 0.49 | 3.48 ± 0.22 | 3.21 ± 0.29 |
| + | 3.50 ± 0.17 | 4.24 ± 0.67 | 3.10 ± 0.23 | 3.33 ± 0.30 | |
| % in T lymphocytes: | |||||
| Th | - | 74.88 ± 0.57 | 72.19 ± 1.09 | 74.09 ± 0.74 | 74.01 ± 0.92 |
| + | 72.59 ± 1.66 | 70.00 ± 3.26 | 74.43 ± 0.38 | 75.34 ± 0.46 | |
| Tc | - | 27.87 ± 1.27 | 28.57 ± 0.70 | 27.23 ± 0.85 | 26.60 ± 0.74 |
| + | 28.21 ± 1.95 | 30.36 ± 2.73 | 27.71 ± 0.47 | 26.95 ± 1.90 | |
| Peritoneal Macrophage Cytokine Secretion | |||||
|---|---|---|---|---|---|
| Hesperidin | SED | T | TE | TE24 | |
| IFN-γ | - | 28.14 ± 10.33 | 161.36 ± 34.75 * | 46.01 ± 8.64 ϕ | 34.92 ± 15.00 ϕ |
| + | 63.12 ± 11.19 # | 46.92 ± 19.32 # | 33.48 ± 10.40 | 124.6 ± 55.87 | |
| IL-1β | - | 131.7 ± 23.01 | 102.7 ± 23.15 | 165.3 ± 12.05 | 141.7 ± 31.66 |
| + | 127.6 ± 18.21 | 90.29 ± 16.41 | 147.1 ± 37.84 | 168.8 ± 21.69 | |
| IL-6 | - | 710.6 ± 167.3 | 1282.5 ± 155.4 | 1635.0 ± 224.6 * | 1080.9 ± 316.5 |
| + | 1054.0 ± 192.2 | 1032.2 ± 222.5 | 1358.7 ± 334.9 | 1737.2 ± 268.3 | |
| TNF-α | - | 304.6 ± 76.60 | 146.8 ± 38.30 | 408.5 ± 63.30 ϕ | 359.2 ± 86.57 ϕ |
| + | 350.0 ± 54.97 | 238.9 ± 56.38 | 353.9 ± 73.59 | 419.0 ± 51.32 | |
| IL-10 | - | 83.41 ± 19.73 | 97.22 ± 16.57 | 58.02 ± 28.21 | 87.03 ± 17.78 |
| + | 118.7 ± 27.28 | 47.40 ± 8.74 # | 54.48 ± 15.14 | 76.70 ± 27.53 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Iglesias, P.; Estruel-Amades, S.; Camps-Bossacoma, M.; Massot-Cladera, M.; Franch, À.; Pérez-Cano, F.J.; Castell, M. Influence of Hesperidin on Systemic Immunity of Rats Following an Intensive Training and Exhausting Exercise. Nutrients 2020, 12, 1291. https://doi.org/10.3390/nu12051291
Ruiz-Iglesias P, Estruel-Amades S, Camps-Bossacoma M, Massot-Cladera M, Franch À, Pérez-Cano FJ, Castell M. Influence of Hesperidin on Systemic Immunity of Rats Following an Intensive Training and Exhausting Exercise. Nutrients. 2020; 12(5):1291. https://doi.org/10.3390/nu12051291
Chicago/Turabian StyleRuiz-Iglesias, Patricia, Sheila Estruel-Amades, Mariona Camps-Bossacoma, Malén Massot-Cladera, Àngels Franch, Francisco J. Pérez-Cano, and Margarida Castell. 2020. "Influence of Hesperidin on Systemic Immunity of Rats Following an Intensive Training and Exhausting Exercise" Nutrients 12, no. 5: 1291. https://doi.org/10.3390/nu12051291
APA StyleRuiz-Iglesias, P., Estruel-Amades, S., Camps-Bossacoma, M., Massot-Cladera, M., Franch, À., Pérez-Cano, F. J., & Castell, M. (2020). Influence of Hesperidin on Systemic Immunity of Rats Following an Intensive Training and Exhausting Exercise. Nutrients, 12(5), 1291. https://doi.org/10.3390/nu12051291









