Effects of Virgin Olive Oil on Bone Health in Ovariectomized Rats
Abstract
1. Introduction
2. Material and Methods
2.1. Animals
2.2. Bone Mineral Density
2.3. Trabecular Microarchitecture Analysis of Femur
2.4. Biomechanical Analysis
2.5. Biochemical Markers of Bone Turnover
2.6. Weight of the Rats
2.7. Statistical Analysis
3. Results
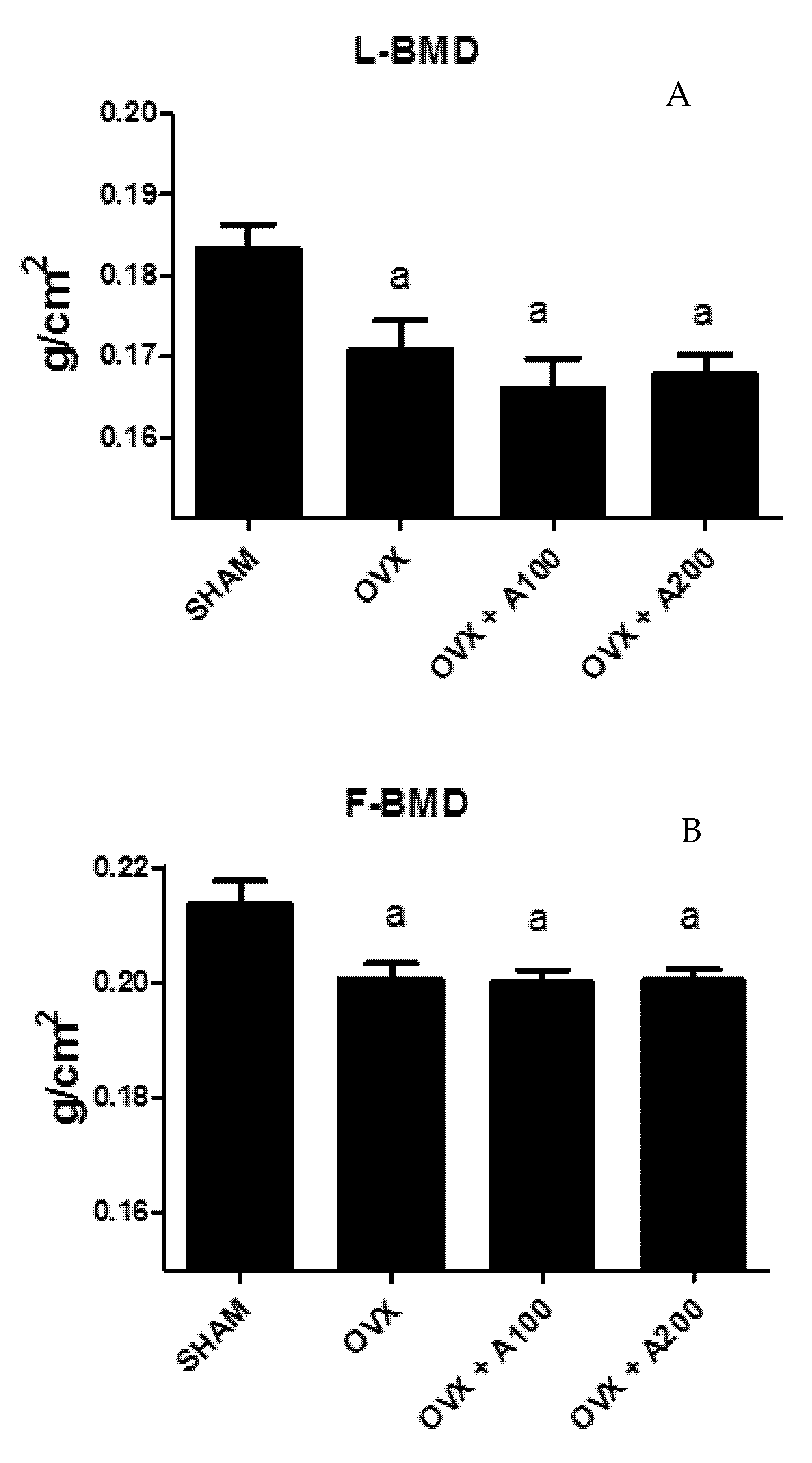
3.1. Effect of VOO on FBMD and LBMD
3.2. Effect of VOO on Microtomographic Parameters
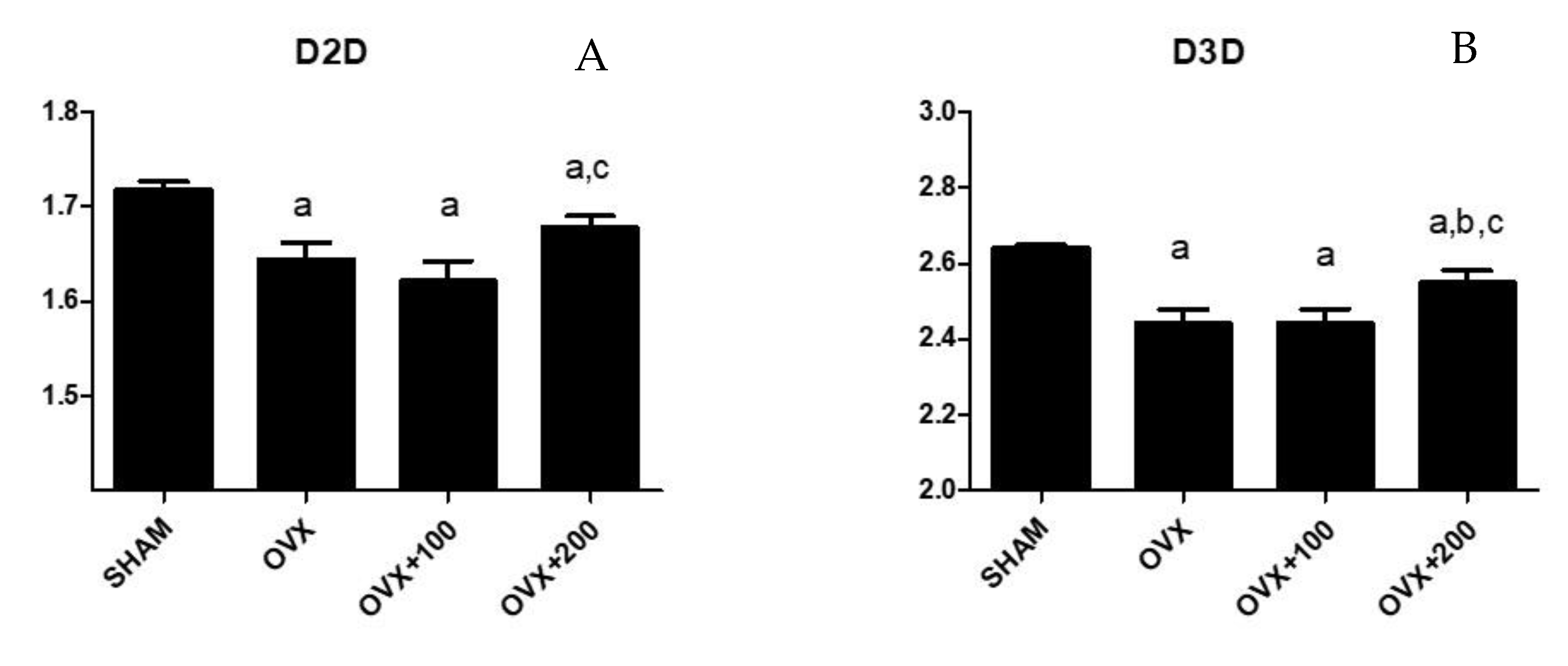
3.3. Effect of VOO on Fractal Dimension
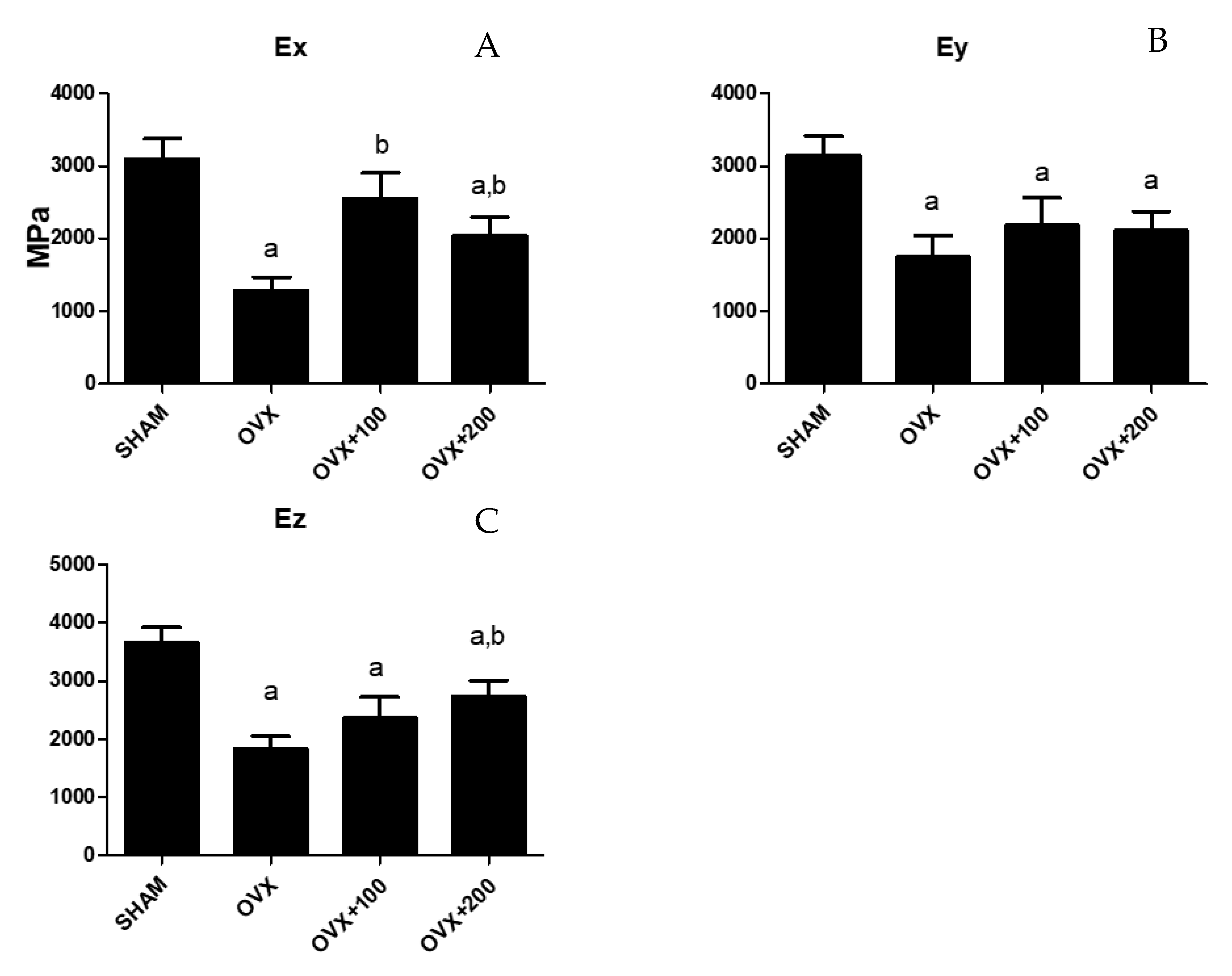
3.4. Effect of VOO on Young’s Modulus
3.5. Effect of VOO on Biochemical Markers of Bone Turnover
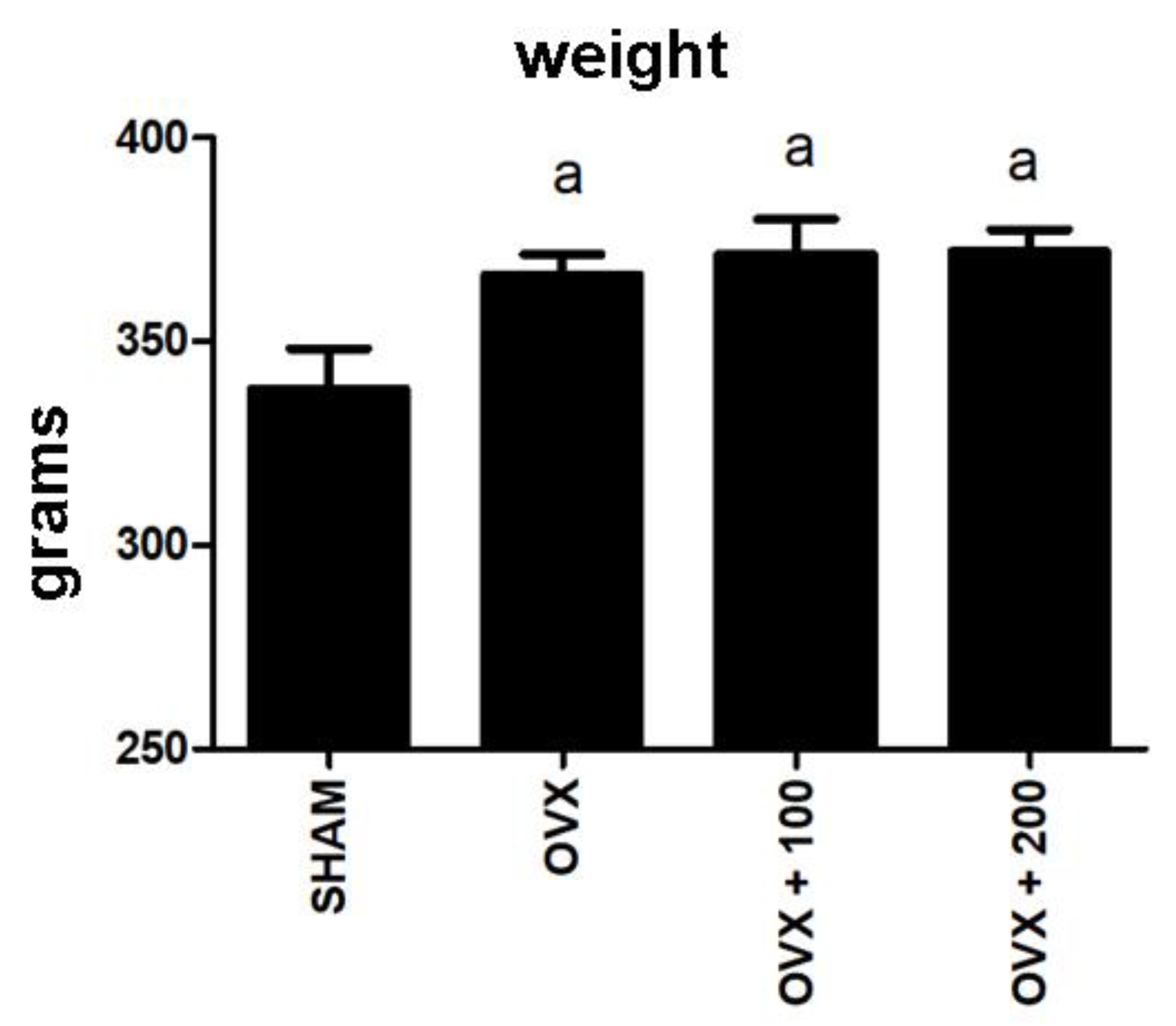
3.6. Effect of VOO on Final Weight of the Rats
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- NIH. Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA 2001, 85, 785–795. [Google Scholar]
- Bonjour, J.P. Protein intake and bone health. Int. J. Vitam. Nutr. Res. 2011, 81, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Pedrera-Zamorano, J.D.; Calderón-García, J.F.; Roncero-Martín, R.; Mañas-Núñez, P.; Morán, J.M.; Lavado-García, J.M. The protective efecto of calcium on bone mass in postmenopausal women with high selenium intake. J. Nutr. Health Aging 2012, 16, 743–748. [Google Scholar] [CrossRef] [PubMed]
- Díaz Curiel, M.; Gil, A.; Mataix, J. Nutrición y salud ósea; Sociedad Española de Investigaciones Óseas y Metabolismo Mineral. Instituto Omega 3 y Fundación Hispana de Osteoporosis y Enfermedades Metabólicas Óseas: Madrid, España, 2004. [Google Scholar]
- Keiler, A.M.; Zierau, O.; Bernhardt, R.; Scharnweber, D.; Lemonakis, N.; Termetzi, A.; Skaltsounis, L.; Vollmer, G.; Halabalaki, M. Impact of a functionalized olive oil extract on the uterus and the bone in a model of postmenopausal osteoporosis. Eur. J. Nutr. 2014, 53, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Pelucci, C.; Bosetti, C.; Negri, E.; Lipworth, L.; La Vecchia, C. Olive oil and cáncer risk: An update of epidemiological findings through. Curr. Pharm. Des. 2010, 17, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Kontigianni, M.D.; Melistas, L.; Yannakoulia, M.; Malagaris, I.; Panagiotakos, D.B.; Yiannakouris, N. Association between dietary patterns and indices of bone mass in a sample of Mediterranean women. Nutrition 2009, 25, 165–171. [Google Scholar] [CrossRef]
- Rivas, A.; Romero, A.; Mariscal-Arcas, M.; Monteagudo, C.; Feriche, B.; Lorenzo, M.L.; Olea, F. Mediterranean diet and bone mineral density in two age groups of women. Int. J. Food. Sci. Nutr. 2013, 64, 155–161. [Google Scholar] [CrossRef]
- Savanelli, M.C.; Barrea, L.; Macchia, P.E.; Savastano, S.; Falco, A.; Renzullo, A.; Scarano, E.; Nettore, I.C.; Colao, A.; Di Somma, C. Preliminary results demonstrating the impact of Mediterranean diet on bone health. J. Transl. Med. 2017, 15, 81. [Google Scholar] [CrossRef]
- Silva, T.D.R.; Martins, C.C.; Ferreira, L.L.; Spritzer, P.M. Mediterranean diet is associated with bone mineral density and muscle mass in postmenopausal women. Climateric 2019, 22, 162–168. [Google Scholar] [CrossRef]
- Garcia-Martinez, O.; Rivas, A.; Ramos-Torrecilla, J.; De Luna-Bertos, E.; Ruiz, C. The effect of olive oil on osteoporosis prevention. Int. J. Food Sci. Nutr. 2014, 65, 834–840. [Google Scholar] [CrossRef]
- Servili, M.; Esposto, S.; Fabiani, R.; Urbani, S.; Taticchi, A.; Mariucci, F.; Selvaggini, R.; Montedoro, G.F. Phenolic compounds in olive oil: Antioxidant, health and organoleptic activities according to their chemical structure. Inflammopharmacology 2009, 17, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Kanakis, P.; Termentzi, A.; Michel, T.; Gikas, E.; Halabaki, M.; Skaltsounis, A.L. From olive drupes to olive oil. An HPLC-orbitrap-based qualitativeand quantitative exploration of olve key metabolites. Planta Med. 2013, 79, 1576–1587. [Google Scholar] [PubMed]
- Kim, T.H.; Jung, J.W.; Ha, B.G.; Hong, J.M.; Park, E.K.; Kim, H.J.; Kim, S.Y. The effects of luteolin on osteoclast differentiation function in vitro and ovariectomy-inducedbone loss. J. Nutr. Biochem. 2011, 22, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Tagliaferri, C.; Davicco, M.J.; Lebecque, P.; Georgé, S.; Amiot, M.J.; Mercier, S.; Dhaussy, A.; Huertas, A.; Walrand, S.; Wittrant, Y.; et al. Olive oil and vitamin D synergistically prevent bone loss in mice. PLoS ONE 2014, 9, e115817. [Google Scholar] [CrossRef]
- Borchers, R.E.; Gibson, L.J.; Burchardt, H.; Hayes, W.C. Effects of selected thermal variables on the mechanical properties of trabecular bone. Biomaterials 1995, 16, 545–551. [Google Scholar] [CrossRef]
- Baum, T.; Carballido-Gamio, J.; Huber, M.B.; Müller, D.; Monetti, R.; Räth, C.; Eckstein, F.; Lochmüller, E.M.; Majumdar, S.; Rummeny, E.J.; et al. Automated 3D trabecular bone structure analysis of the proximal femur—Prediction of biomechanical strength by CT and DXA. Osteoporos. Int. 2010, 21, 1553–1564. [Google Scholar] [CrossRef]
- Alberich-Bayarri, A.; Martí-Bonmatí, L.; Sanz-Requena, R.; Sánchez-González, J.; Briz, V.H.; García-Martí, G.; Pérez, M.Á. Reproducibility and accuracy in the morphometric and mechanical quantification of trabecular bone from 3 Tesla Magnetic resonance images. Radiologia 2014, 56, 27–34. [Google Scholar] [CrossRef]
- Alberich-Bayarri, A.; Marti-Bonmati, L.; Pérez, M.A.; Sanz-Requena, R.; Lerma-Garrido, J.J.; García-Martí, G.; Moratal, D. Assesment of 2D and 3D fractal dimension measurements of trabecular bone from high-spatial resolution magnetic resonance images in 3T. Med. Phys. 2010, 37, 4930–4937. [Google Scholar] [CrossRef]
- Alberich-Bayarri, A.; Moratal, D.; Ivirico, J.L.E.; Hernández, J.C.R.; Vallés-Lluch, A.; Martí-Bonmatí, L.; Estellés, J.M.; Mano, J.F.; Pradas, M.M.; Ribelles, J.L.G.; et al. MIcrocomputed tomography and microfinite element modeling for evaluating polymer scaffolds architecture and their mechanical properties. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 91, 191–202. [Google Scholar] [CrossRef]
- Roncero-Martín, R.; Aliaga Vera, I.; Moreno-Corral, L.J.; Moran, J.M.; Lavado-Garcia, J.M.; Pedrera-Zamorano, J.D.; Pedrera-Canal, M. Olive oil consumption and bone microarchitecture in Spanish Women. Nutrients 2018, 10, 968. [Google Scholar] [CrossRef]
- Rivas, A.; Romero, A.; Mariscal, M.; Monteagudo, C.; Hernandez, J.; Olea-Serrano, F. Validation of questionnaries for the study of food habits and bone mass. Nutr. Hosp. 2009, 24, 521–528. [Google Scholar] [PubMed]
- Ostrowska, E.; Gabler, N.K.; Ridley, D.; Suster, D.; Eagling, D.R.; Dunshea, F.R. Extra-virgin and refined olive oil decrease plasma triglyceride, moderately affect lipoprotein oxidation susceptibility and increase bone density in growing pigs. J. Sci. Food Agric. 2006, 86, 1955–1963. [Google Scholar] [CrossRef]
- Saleh, N.K.; Saleh, H.A. Olive oil effectively mitigates ovariectomy-induced osteoporosis in rats. BMC Complement. Altern. Med. 2011, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Mora, R.; Casado-Díaz, A.; De Castro, M.D.; Quesada-Gómez, J.M. Oleuropein enhances osteoblastogenesis and inhibits adipogenesis: The effect on differentiation in stem cells derived from bone marrow. Osteoporosis. Int. 2011, 22, 675–684. [Google Scholar] [CrossRef]
- Puel, C.; Quintin, A.; Agalias, A.; Mathey, J.; Obled, C.; Mazur, A.; Davicco, M.J.; Lebecque, P.; Skaltsounis, A.L.; Coxam, V. Olive oil and its main phenolic micronutrient (oleuropein) prevent inflammation-induced bone loss in the ovariectomosed rat. Br. J. Ntr. 2004, 92, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Hagiwara, K.; Goto, T.; Araki, M.; Miyazaki, H.; Hagiwara, H. Olive polyphenol hydroxytyrosol prevents bone loss. Eur. J. Pharmacol. 2011, 662, 78–84. [Google Scholar] [CrossRef]
- Melguizo-Rodriguez, L.; Manzano Moreno, F.J.; De Luna-Bertos, E.; Rivas, A.; Ramos-Torrecillas, J.; Ruiz, C.; García-Martínez, O. Effect of olive phenolic compounds on osteoblast differentiation. Eur. J. Clin. Investig. 2018, 48. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díaz-Curiel, M.; Torrubia, B.; Martín-Fernández, M.; Rubert, M.; Piedra, C.D.l. Effects of Virgin Olive Oil on Bone Health in Ovariectomized Rats. Nutrients 2020, 12, 1270. https://doi.org/10.3390/nu12051270
Díaz-Curiel M, Torrubia B, Martín-Fernández M, Rubert M, Piedra CDl. Effects of Virgin Olive Oil on Bone Health in Ovariectomized Rats. Nutrients. 2020; 12(5):1270. https://doi.org/10.3390/nu12051270
Chicago/Turabian StyleDíaz-Curiel, Manuel, Blanca Torrubia, Marta Martín-Fernández, Mercedes Rubert, and Concepción De la Piedra. 2020. "Effects of Virgin Olive Oil on Bone Health in Ovariectomized Rats" Nutrients 12, no. 5: 1270. https://doi.org/10.3390/nu12051270
APA StyleDíaz-Curiel, M., Torrubia, B., Martín-Fernández, M., Rubert, M., & Piedra, C. D. l. (2020). Effects of Virgin Olive Oil on Bone Health in Ovariectomized Rats. Nutrients, 12(5), 1270. https://doi.org/10.3390/nu12051270




