Vitamin D and Immunity in Infants and Children
Abstract
1. Introduction
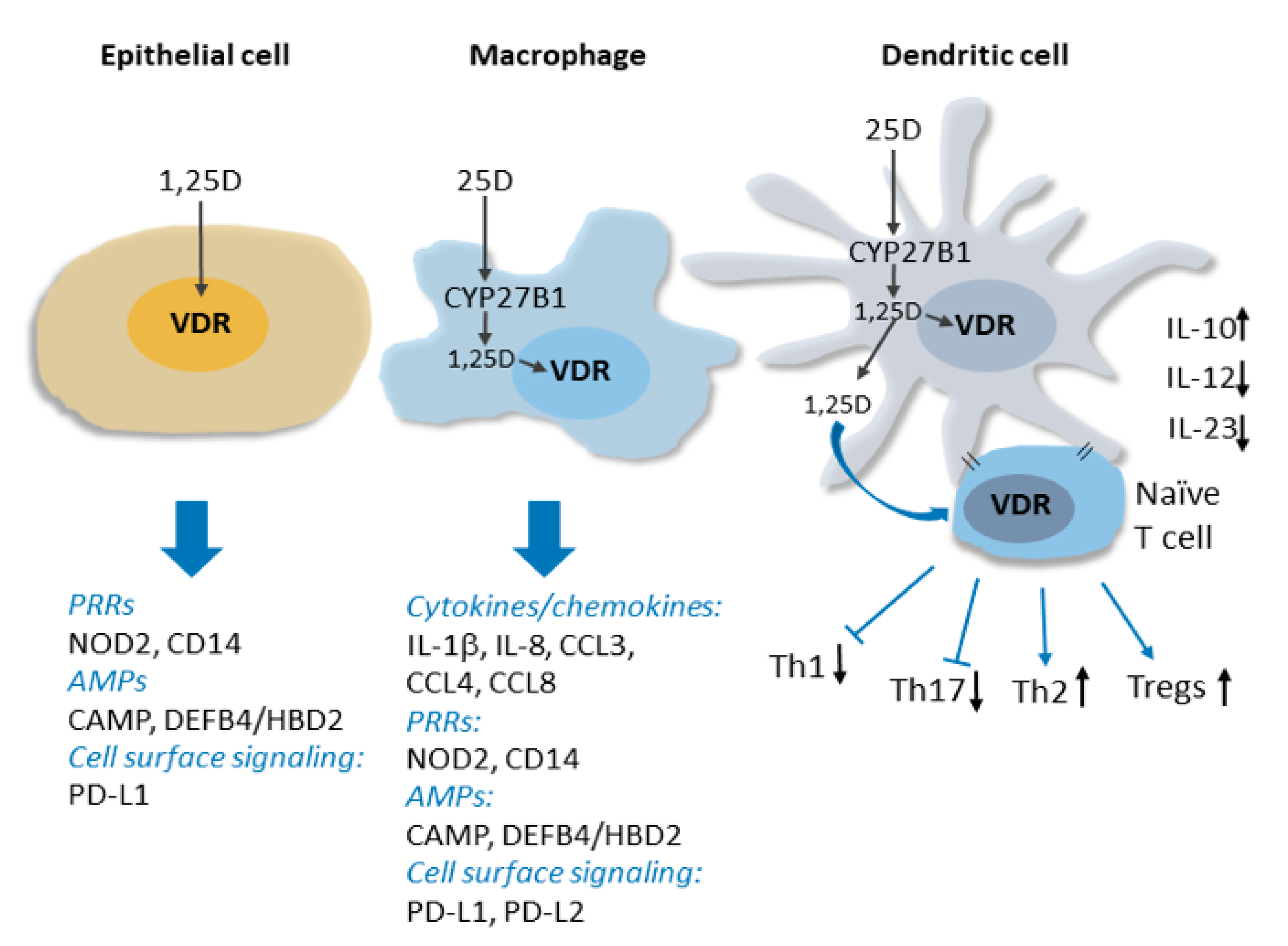
2. Overview of the Molecular Basis for the Non-Classical Actions of Vitamin D in the Immune System
3. Vitamin D and Infectious Diseases in Pediatric Populations
4. Crohn’s Disease
5. Vitamin D Deficiency and Dental Caries
6. Links between Vitamin D Deficiency and Autoimmune Conditions in Pediatric Populations
6.1. Juvenile-Onset Type 1 Diabetes
6.2. Multiple Sclerosis
6.3. Other Autoimmune Conditions
7. Allergic Conditions
7.1. Asthma
7.2. Other Allergic Conditions
7.2.1. Atopic Dermatitis
7.2.2. Food Allergies
7.2.3. Allergic Rhinitis and Aeroallergen Sensitization
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tavera-Mendoza, L.; White, J.H. Cell defenses and the sunshine vitamin. Sci. Am. 2007, 297, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Arabi, A.; El Rassi, R.; El-Hajj Fuleihan, G. Hypovitaminosis D in developing countries-prevalence, risk factors and outcomes. Nat. Rev. Endocrinol. 2010, 66, 550–561. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Antonio, L. Nutritional rickets: Historic overview and plan for worldwide eradication. J. Steroid Biochem. Mol. Biol. 2020, 198, 105563. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Marcocci, C.; Carmeliet, G.; Bikle, D.; White, J.H.; Dawson-Hughes, B.; Lips, P.; Munns, C.F.; Lazaretti-Castro, M.; Giustina, A.; et al. Skeletal and Extraskeletal Actions of Vitamin D: Current Evidence and Outstanding Questions. Endocr. Rev. 2018, 40, 1109–1151. [Google Scholar] [CrossRef]
- Pettifor, J.M.; Thandrayen, K.; Thacher, T.D. Chapter 67—Vitamin D Deficiency and Nutritional Rickets in Children. In Vitamin D, 4th ed.; Feldman, D., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 179–201. [Google Scholar]
- Guy, R.A. The history of cod liver oil as a remedy. Am. J. Dis. Child. 1923, 26, 112–116. [Google Scholar] [CrossRef]
- Aul, A.J.; Fischer, P.R.; O’Grady, J.S.; Mara, K.C.; Maxson, J.A.; Meek, A.M.; Petterson, T.M.; Thacher, T.D. Population-Based Incidence of Potentially Life-Threatening Complications of Hypocalcemia and the Role of Vitamin D Deficiency. J. Pediatrics 2019, 211, e104. [Google Scholar] [CrossRef]
- Wagner, C.L. Seeing Beyond Our Expectations: The Case of Pediatric Hypocalcemia. J. Pediatrics 2019, 211, 9–12. [Google Scholar] [CrossRef]
- Thacher, T.D.; Clarke, B.L. Vitamin D Insufficiency. Mayo Clin. Proc. 2011, 86, 50–60. [Google Scholar] [CrossRef]
- Borzutzky, A.; Grob, F.; Camargo, C.A., Jr.; Martinez-Aguayo, A. Vitamin D deficiency rickets in an adolescent with severe atopic dermatitis. Pediatrics 2014, 133, e451–e454. [Google Scholar] [CrossRef]
- Hewison, M.; Burke, F.; Evans, K.N.; Lammas, D.A.; Sansom, D.M.; Liu, P.; Modlin, R.L.; Adams, J.S. Extra-renal 25-hydroxyvitamin D-3-1 alpha-hydroxylase in human health and disease. J. Steroid Biochem. Mol. Biol. 2007, 103, 316–321. [Google Scholar] [CrossRef]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-Like Receptor Triggering of a Vitamin D-Mediated Human Antimicrobial Response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef] [PubMed]
- Iannuzzi, M.C.; Rybicki, B.A.; Teirstein, A.S. Medical progress: Sarcoidosis. New Engl. J. Med. 2007, 357, 2153–2165. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M.; Barker, S.; Brennan, A.; Katz, D.R.; O’Riordan, J.L.H. Modulation of myelomonocytic U937 cells by vitamin D metabolites. Bone Miner. 1989, 55, 323–333. [Google Scholar] [CrossRef]
- Wang, T.-T.; Dabbas, B.; Laperriere, D.; Bitton, A.J.; Soualhine, H.; Tavera-Mendoza, L.E.; Dionne, S.; Servant, M.J.; Bitton, A.; Seidman, E.G.; et al. Direct and Indirect Induction by 1, 25-Dihydroxyvitamin D3 of the NOD2/CARD15-Defensin β2 Innate Immune Pathway Defective in Crohn Disease. J. Biol. Chem. 2010, 285, 2227–2231. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.-T.; Nestel, F.P.; Bourdeau, V.; Nagai, Y.; Wang, Q.; Liao, J.; Tavera-Mendoza, L.; Lin, R.; Hanrahan, J.H.; Mader, S.; et al. Cutting Edge: 1, 25-Dihydroxyvitamin D3 Is a Direct Inducer of Antimicrobial Peptide Gene Expression. J. Immunol. 2004, 173, 2909–2912. [Google Scholar] [CrossRef] [PubMed]
- Gombart, A.F.; Borregaard, N.; Koeffler, H.P. Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1, 25-dihydroxyvitamin D3. FASEB J. 2005, 19, 1067–1077. [Google Scholar] [CrossRef]
- Dimitrov, V.; White, J.H. Species-specific regulation of innate immunity by vitamin D signaling. J. Steroid Biochem. Mol. Biol. 2016, 164, 246–253. [Google Scholar] [CrossRef]
- Vargas Buonfiglio, L.G.; Cano, M.; Pezzulo, A.A.; Vanegas Calderon, O.G.; Zabner, J.; Gerke, A.K.; Comellas, A.P. Effect of vitamin D3 on the antimicrobial activity of human airway surface liquid: Preliminary results of a randomised placebo-controlled double-blind trial. BMJ Open Respir. Res. 2017, 44, e00021. [Google Scholar]
- Verway, M.; Bouttier, M.; Wang, T.-T.; Carrier, M.; Calderon, M.; An, B.-S.; Devemy, E.; McIntosh, F.; Divangahi, M.; Behr, M.A.; et al. Vitamin D Induces Interleukin-1 beta Expression: Paracrine Macrophage Epithelial Signaling Controls M-tuberculosis Infection. PLoS Pathog. 2013, 9, 6. [Google Scholar] [CrossRef]
- Penna, G.; Adorini, L. 1α,25-dihydroxyvitamin D3 inhibits differentiation, maturation, activation, and survival of dendritic cells leading to impaired alloreactive T cell activation. J. Immunol. 2000, 164, 2405–2411. [Google Scholar] [CrossRef]
- Adorini, L. Intervention in autoimmunity: The potential of vitamin D receptor agonists. Cell Immunol. 2005, 233, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Kamen, D.L.; Tangpricha, V. Vitamin D and molecular actions on the immune system: Modulation of innate and autoimmunity. J. Mol. Med. 2010, 88, 441–450. [Google Scholar] [CrossRef]
- Baeke, F.; Takiishi, T.; Korf, H.; Gysemans, C.; Mathieu, C. Vitamin D: Modulator of the immune system. Curr. Opin. Pharmacol. 2010, 10, 482–496. [Google Scholar] [CrossRef] [PubMed]
- Urry, Z.; Chambers, E.S.; Xystrakis, E.; Dimeloe, S.; Richards, D.F.; Gabrysova, L.; Christensen, J.; Gupta, A.; Saglani, S.; Bush, A.; et al. The role of 1, 25-dihydroxyvitamin D3 and cytokines in the promotion of distinct Foxp3+ and IL-10+CD4+ T cells. Eur. J. Immunol. 2012, 42, 2697–2708. [Google Scholar] [CrossRef] [PubMed]
- Chun, R.F.; Liu, P.T.; Modlin, R.L.; Adams, J.S.; Hewison, M. Impact of vitamin D on immune function: Lessons learned from genome-wide analysis. Front. Physiol. 2014, 5, 151. [Google Scholar] [CrossRef]
- Watkins, J. Preventing a covid-19 pandemic. BMJ 2020, 368, m810. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Shao, J.; Guo, Y.; Peng, X.; Li, Z.; Hu, D. Clinical and CT features in pediatric patients with COVID-19 infection: Different points from adults. Pediatric Pulmonol. 2020, 55, 1169–1174. [Google Scholar] [CrossRef]
- Barlow, P.G.; Svoboda, P.; Mackellar, A.; Nash, A.A.; York, I.A.; Pohl, J.; Davidson, D.J.; Donis, R.O. Antiviral Activity and Increased Host Defense against Influenza Infection Elicited by the Human Cathelicidin LL-37. PLoS ONE 2011, 6, e25333. [Google Scholar] [CrossRef]
- Telcian, A.G.; Zdrenghea, M.T.; Edwards, M.R.; Laza-Stanca, V.; Mallia, P.; Johnston, S.L.; Stanciu, L.A. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antivir. Res. 2017, 137, 93–101. [Google Scholar] [CrossRef]
- Li, Y.C.; Qiao, G.; Uskokovic, M.; Xiang, W.; Zheng, W.; Kong, J. Vitamin D: A negative endocrine regulator of the renin–angiotensin system and blood pressure. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 387–392. [Google Scholar] [CrossRef]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444. [Google Scholar] [CrossRef] [PubMed]
- Riera, M.; Anguiano, L.; Clotet, S.; Roca-Ho, H.; Rebull, M.; Pascual, J.; Soler, M.J. Paricalcitol modulates ACE2 shedding and renal ADAM17 in NOD mice beyond proteinuria. Am. J. Physiol. Ren. Physiol. 2015, 310, F534–F546. [Google Scholar] [CrossRef]
- Fang, L.; Karakiulakis, G.; Roth, M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir. Med. 2020, 8, e21. [Google Scholar] [CrossRef]
- Gonzalez-Gross, M.; Valtuena, J.; Breidenassel, C.; Moreno, L.A.; Ferrari, M.; Kersting, M.; De Henauw, S.; Gottrand, F.; Azzini, E.; Widhalm, K.; et al. Vitamin D status among adolescents in Europe: The Healthy Lifestyle in Europe by Nutrition in Adolescence study. Br. J. Nutr. 2012, 107, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Rabufetti, A.; Milani, G.P.; Lava, S.A.G.; Edefonti, V.; Bianchetti, M.G.; Stettbacher, A.; Muggli, F.; Simonetti, G. Vitamin D Status Among Male Late Adolescents Living in Southern Switzerland: Role of Body Composition and Lifestyle. Nutrients 2019, 11, 2727. [Google Scholar] [CrossRef] [PubMed]
- Diethelm, K.; Huybrechts, I.; Moreno, L.; De Henauw, S.; Manios, Y.; Beghin, L.; Gonzalez-Gross, M.; Le Donne, C.; Cuenca-Garcia, M.; Castillo, M.J.; et al. Nutrient intake of European adolescents: Results of the HELENA (Healthy Lifestyle in Europe by Nutrition in Adolescence) Study. Public Health Nutr. 2014, 17, 486–497. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Skrabakova, Z.; Gonzalez-Gross, M.; Valtuena, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Molgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef]
- Lips, P.; Cashman, K.D.; Lamberg-Allardt, C.; Bischoff-Ferrari, H.A.; Obermayer-Pietsch, B.; Bianchi, M.L.; Stepan, J.; Fuleihan, G.E.; Bouillon, R. European Calcified Tissue S., Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: A position statement of the European Calcified Tissue Society. Eur. J. Endocrinol. 2019, 180, P23–P54. [Google Scholar] [CrossRef]
- Kamboj, P.; Dwivedi, S.; Toteja, G.S. Prevalence of hypovitaminosis D in India & way forward. Indian J. Med. Res. 2018, 148, 548–556. [Google Scholar]
- Chowdhury, R.; Taneja, S.; Bhandari, N.; Sinha, B.; Upadhyay, R.P.; Bhan, M.K.; Strand, T.A. Vitamin-D deficiency predicts infections in young north Indian children: A secondary data analysis. PLoS ONE 2017, 12, 12. [Google Scholar] [CrossRef]
- Banajeh, S.M. Nutritional rickets and vitamin D deficiency—Association with the outcomes of childhood very severe pneumonia: A prospective cohort study. Pediatric Pulmonol. 2009, 44, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. Br. Med. J. [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; Goodall, E.C.; et al. Vitamin D supplementation to prevent acute respiratory infections: Individual participant data meta-analysis. Health Technol. Assess. 2019, 23, 1–4. [Google Scholar] [CrossRef]
- Urashima, M.; Segawa, T.; Okazaki, M.; Kurihara, M.; Wada, Y.; Ida, H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am. J. Clin. Nutr. 2010, 91, 1255–1260. [Google Scholar] [CrossRef]
- Camargo, C.A.; Ganmaa, D.; Frazier, A.L.; Kirchberg, F.F.; Stuart, J.J.; Kleinman, K.; Sumberzul, N.; Rich-Edwards, J.W. Randomized Trial of Vitamin D Supplementation and Risk of Acute Respiratory Infection in Mongolia. Pediatrics 2012, 130, E561–E567. [Google Scholar] [CrossRef]
- Marchisio, P.; Consonni, D.; Baggi, E.; Zampiero, A.; Bianchini, S.; Terranova, L.; Tirelli, S.; Esposito, S.; Principi, N. Vitamin D Supplementation Reduces the Risk of Acute Otitis Media in Otitis-prone Children. Pediatric Infect. Dis. J. 2013, 32, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Manaseki-Holland, S.; Maroof, Z.; Bruce, J.; Mughal, M.Z.; Masher, M.I.; Bhutta, Z.A.; Walraven, G.; Chandramohan, D. Effect on the incidence of pneumonia of vitamin D supplementation by quarterly bolus dose to infants in Kabul: A randomised controlled superiority trial. Lancet 2012, 379, 1419–1427. [Google Scholar] [CrossRef]
- Manaseki-Holland, S.; Qader, G.; Masher, M.; Bruce, J.; Mughal, M.; Chandramohan, D.; Walraven, G. Effects of vitamin D supplementation to children diagnosed with pneumonia in Kabul: A randomised controlled trial. Trop. Med. Int. Health 2010, 15, 1148–1155. [Google Scholar] [CrossRef]
- Singh, N.; Kamble, D.; Mahantshetti, N.S. Effect of Vitamin D Supplementation in the Prevention of Recurrent Pneumonia in Under-Five Children. Indian J. Pediatrics 2019, 86, 1105–1111. [Google Scholar] [CrossRef]
- Cariolou, M.; Cupp, M.A.; Evangelou, E.; Tzoulaki, I.; Berlanga-Taylor, A.J. Importance of vitamin D in acute and critically ill children with subgroup analyses of sepsis and respiratory tract infections: A systematic review and meta-analysis. BMJ Open 2019, 9, e027666. [Google Scholar] [CrossRef]
- McNally, J.D.; Leis, K.; Matheson, L.A.; Karuananyake, C.; Sankaran, K.; Rosenberg, A.M. Vitamin D deficiency in young children with severe acute lower respiratory infection. Pediatric Pulmonol. 2009, 44, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.C.; Shi, H.Y.; Hamidi, N.; Underwood, F.E.; Tang, W.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Wu, J.C.Y.; Chan, F.K.L.; et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: A systematic review of population-based studies. Lancet 2017, 390, 2769–2778. [Google Scholar] [CrossRef]
- McGovern, D.P.B.; Kugathasan, S.; Cho, J.H. Genetics of Inflammatory Bowel Diseases. Gastroenterology 2015, 149, 1163–1176.e2. [Google Scholar] [CrossRef] [PubMed]
- Jantchou, P.; Clavel-Chapelon, F.; Racine, A.; Kvaskoff, M.; Carbonnel, F.; Boutron-Ruault, M.-C. High Residential Sun Exposure Is Associated With a Low Risk of Incident Crohn’s Disease in the Prospective E3N Cohort. Inflamm. Bowel Dis. 2014, 20, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.A.; Harris, R.M.R.; Lucas, R.M. Low Sun Exposure and Vitamin D Deficiency as Risk Factors for Inflammatory Bowel Disease, With a Focus on Childhood Onset. Photochem. Photobiol. 2019, 95, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Vernier–Massouille, G.; Balde, M.; Salleron, J.; Turck, D.; Dupas, J.L.; Mouterde, O.; Merle, V.; Salomez, J.L.; Branche, J.; Marti, R.; et al. Natural History of Pediatric Crohn’s Disease: A Population-Based Cohort Study. Gastroenterology 2008, 135, 1106–1113. [Google Scholar] [CrossRef]
- White, J.H. Vitamin D deficiency and the pathogenesis of Crohn’s disease. J. Steroid Biochem. Mol. Biol. 2018, 175, 23–28. [Google Scholar] [CrossRef]
- Hugot, J.-P.; Chamaillard, M.; Zouali, H.; Lesage, S.; Cezard, J.-P.; Belaiche, J.; Almer, S.; Tysk, C.; O’Morain, C.A.; Gassull, M.; et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature 2001, 411, 599–603. [Google Scholar] [CrossRef]
- Dimitrov, V.; Bouttier, M.; Boukhaled, G.; Salehi-Tabar, R.; Avramescu, R.G.; Memari, B.; Hasaj, B.; Lukacs, G.L.; Krawczyk, C.M.; White, J.H. Hormonal vitamin D up-regulates tissue-specific PD-L1 and PD-L2 surface glycoprotein expression in humans but not mice. J. Biol. Chem. 2017, 292, 20657–20668. [Google Scholar] [CrossRef]
- Scandiuzzi, L.; Ghosh, K.; Hofmeyer, K.A.; Abadi, Y.M.; Lazar-Molnar, E.; Lin, E.Y.; Liu, Q.; Jeon, H.; Almo, S.C.; Chen, L.; et al. Tissue-Expressed B7-H1 Critically Controls Intestinal Inflammation. Cell Rep. 2014, 6, 625–632. [Google Scholar] [CrossRef]
- Pappa, H.M.; Mitchell, P.D.; Jiang, H.; Kassiff, S.; Filip-Dhima, R.; DiFabio, D.; Quinn, N.; Lawton, R.C.; Bronzwaer, M.E.S.; Koenen, M.; et al. Maintenance of optimal vitamin D status in children and adolescents with inflammatory bowel disease: A randomized clinical trial comparing two regimens. J. Clin. Endocrinol. Metab. 2014, 99, 3408–3417. [Google Scholar] [CrossRef]
- Li, J.; Chen, N.; Wang, D.; Zhang, J.; Gong, X. Efficacy of vitamin D in treatment of inflammatory bowel disease: A meta-analysis. Medicine 2018, 97, e12662. [Google Scholar] [CrossRef]
- Gubatan, J.; Chou, N.D.; Nielsen, O.H.; Moss, A.C. Systematic review with meta-analysis: Association of vitamin D status with clinical outcomes in adult patients with inflammatory bowel disease. Aliment. Pharmacol. Ther. 2019, 50, 1146–1158. [Google Scholar] [CrossRef]
- McCullough, P.; Heaney, R. Correction of vitamin D deficiency using sublingually administered vitamin D2 in a Crohn’s disease patient with mal-absorption and a new ileostomy. J. Steroid Biochem. Mol. Biol. 2017, 173, 211–214. [Google Scholar] [CrossRef] [PubMed]
- Koutkia, P.; Lu, Z.; Chen, T.C.; Holick, M.F. Treatment of vitamin d deficiency due to crohn’s disease with tanning bed ultraviolet b radiation. Gastroenterology 2001, 121, 1485–1488. [Google Scholar] [CrossRef] [PubMed]
- Pierce, A.; Singh, S.; Lee, J.; Grant, C.; de Jesus, V.C.; Schroth, R.J. The Burden of Early Childhood Caries in Canadian Children and Associated Risk Factors. Front. Public Health 2019, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Singleton, R.; Day, G.; Thomas, T.; Schroth, R.; Klejka, J.; Lenaker, D.; Berner, J. Association of Maternal Vitamin D Deficiency with Early Childhood Caries. J. Dent. Res. 2019, 98, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K.; Hitsumoto, S.; Miyake, Y.; Okubo, H.; Sasaki, S.; Miyatake, N.; Arakawa, M. Higher vitamin D intake during pregnancy is associated with reduced risk of dental caries in young Japanese children. Ann. Epidemiol. 2015, 25, 620–625. [Google Scholar] [CrossRef]
- Hujoel, P.P. Vitamin D and dental caries in controlled clinical trials: Systematic review and meta-analysis. Nutr. Rev. 2013, 71, 88–97. [Google Scholar] [CrossRef]
- Souza, A.P.; Kobayashi, T.Y.; Neto, N.L.; Silva, S.M.B.; Machado, M.; Oliveira, T.M. Dental manifestations of patient with Vitamin D-resistant rickets. J. Appl. Oral Sci. 2013, 21, 601–606. [Google Scholar] [CrossRef]
- Norrisgaard, P.E.; Haubek, D.; Kuhnisch, J.; Chawes, B.L.; Stokholm, J.; Bonnelykke, K.; Bisgaard, H. Association of High-Dose Vitamin D Supplementation During Pregnancy With the Risk of Enamel Defects in Offspring: A 6-Year Follow-up of a Randomized Clinical Trial. JAMA Pediatr. 2019, 173, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Kuhnisch, J.; Thiering, E.; Kratzsch, J.; Heinrich-Weltzien, R.; Hickel, R.; Heinrich, J.; Grp, G.I.S.; Grp, L.I.S. Elevated Serum 25(OH)-Vitamin D Levels Are Negatively Correlated with Molar-Incisor Hypomineralization. J. Dent. Res. 2015, 94, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, T.; Nunn, M.; Dawson-Hughes, B.; Bischoff-Ferrari, H.A. Association between serum concentrations of 25-hydroxyvitamin D and gingival inflammation. Am. J. Clin. Nutr. 2005, 82, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Gyll, J.; Ridell, K.; Ohlund, I.; Akeson, P.K.; Johansson, I.; Holgerson, P.L. Vitamin D status and dental caries in healthy Swedish children. Nutr. J. 2018, 17, 10. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Suzuki, S.; Kawada-Matsuo, M.; Nakanishi, J.; Hirata-Tsuchiya, S.; Komatsuzawa, H.; Yamada, S.; Shiba, H. Heparin-LL37 complexes are less cytotoxic for human dental pulp cells and have undiminished antimicrobial and LPS-neutralizing abilities. Int. Endod. J. 2019, 52, 1327–1343. [Google Scholar] [CrossRef]
- Bhalla, A.; Amento, E.; Clemens, T.; Holick, M.; Krane, S. Specific high-affinity receptors for 1, 25-dihydroxyvitamin D3 in human peripheral blood mononuclear cells: Presence in monocytes and induction in T lymphocytes following activation. J. Clin. Endocrinol. Metab. 1983, 57, 1308–1310. [Google Scholar] [CrossRef]
- Bhalla, A.; Amento, E.; Serog, B.; Glimcher, L. 1, 25-Dihydroxyvitamin D3 inhibits antigen-induced T cell activation. J. Immunol. 1984, 133, 1748–1754. [Google Scholar]
- Adorini, L.; Penna, G. Dendritic cell tolerogenicity: A key mechanism in immunomodulation by vitamin D receptor agonists. Hum. Immunol. 2009, 70, 345–352. [Google Scholar] [CrossRef]
- Ferreira, G.B.; Vanherwegen, A.-S.; Eelen, G.; Gutierrez, A.C.F.; Van Lommel, L.; Marchal, K.; Verlinden, L.; Verstuyf, A.; Nogueira, T.; Georgiadou, M.; et al. Vitamin D3 Induces Tolerance in Human Dendritic Cells by Activation of Intracellular Metabolic Pathways. Cell Rep. 2015, 10, 711–725. [Google Scholar] [CrossRef]
- Dankers, W.; Colin, E.M.; van Hamburg, J.P.; Lubberts, E. Vitamin D in Autoimmunity: Molecular Mechanisms and Therapeutic Potential. Front. Immunol 2017. [Google Scholar] [CrossRef]
- Mason, A.R.; Mason, J.; Cork, M.; Dooley, G.; Hancock, H. Topical treatments for chronic plaque psoriasis. Cochrane Database Syst. Rev. [CrossRef]
- Infante, M.; Ricordi, C.; Sanchez, J.; Clare-Salzler, M.J.; Padilla, N.; Fuenmayor, V.; Chavez, C.; Alvarez, A.; Baidal, D.; Alejandro, R.; et al. Influence of Vitamin D on Islet Autoimmunity and Beta-Cell Function in Type 1 Diabetes. Nutrients 2019, 11, 2185. [Google Scholar] [CrossRef] [PubMed]
- Kachapati, K.; Adams, D.; Bednar, K.; Ridgway, W.M. The Non-Obese Diabetic (NOD) Mouse as a Model of Human Type 1 Diabetes. In Animal Models in Diabetes Research; Joost, H.-G., Al-Hasani, H., Schürmann, A., Eds.; Humana Press: Totowa, NJ, USA, 2012; pp. 3–16. [Google Scholar]
- Giulietti, A.; Gysemans, C.; Stoffels, K.; van Etten, E.; Decallonne, B.; Overbergh, L.; Bouillon, R.; Mathieu, C. Vitamin D deficiency in early life accelerates Type 1 diabetes in non-obese diabetic mice. Diabetologia 2004, 47, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, C.; Laureys, J.; Sobis, H.; Vandeputte, M.; Waer, M.; Bouillon, R. 1, 25-Dihydroxyvitamin D3 Prevents Insulitis in NOD Mice. Diabetes 1992, 41, 1491. [Google Scholar]
- Mathieu, C.; Waer, M.; Laureys, J.; Rutgeerts, O.; Bouillon, R. Prevention of autoimmune diabetes in NOD mice by 1, 25 dihydroxyvitamin D3. Diabetologia 1994, 37, 552–558. [Google Scholar] [CrossRef]
- Mathieu, C.; Waer, M.; Casteels, K.; Laureys, J.; Bouillon, R. Prevention of type I diabetes in NOD mice by nonhypercalcemic doses of a new structural analog of 1, 25-dihydroxyvitamin D3, KH1060. Endocrinology 1995, 136, 866–872. [Google Scholar] [CrossRef]
- Overbergh, L.; Decallonne, B.; Valckx, D.; Verstuyf, A.; Depovere, J.; Laureys, J.; Rutgeerts, O.; Saint-Arnaud, R.; Bouillon, R.; Mathieu, C. Identification and immune regulation of 25-hydroxyvitamin D-1-α-hydroxylase in murine macrophages. Clin. Exp. Immunol. 2000, 120, 139–146. [Google Scholar] [CrossRef]
- Li, X.; Liao, L.; Yan, X.; Huang, G.; Lin, J.; Lei, M.; Wang, X.; Zhou, Z. Protective effects of 1-α-hydroxyvitamin D3 on residual β-cell function in patients with adult-onset latent autoimmune diabetes (LADA). Diabetes Metab. Res. Rev. 2009, 25, 411–416. [Google Scholar] [CrossRef]
- Karvonen, M.; Jantti, V.; Muntoni, S.; Stabilini, M.; Stabilini, L.; Muntoni, S.; Tuomilehto, J. Comparison of the seasonal pattern in the clinical onset of IDDM in Finland and Sardinia. Diabetes Care 1998, 21, 1101–1109. [Google Scholar] [CrossRef]
- Moltchanova, E.V.; Schreier, N.; Lammi, N.; Karvonen, M. Seasonal variation of diagnosis of Type 1 diabetes mellitus in children worldwide. Diabetic Med. 2009, 26, 673–678. [Google Scholar] [CrossRef]
- Makinen, M.; Mykkanen, J.; Koskinen, M.; Simell, V.; Veijola, R.; Hyoty, H.; Ilonen, J.; Knip, M.; Simell, O.; Toppari, J. Serum 25-Hydroxyvitamin D Concentrations in Children Progressing to Autoimmunity and Clinical Type 1 Diabetes. J. Clin. Endocrinol. Metab. 2016, 101, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Cadario, F.; Savastio, S.; Pagliardini, V.; Bagnati, M.; Vidali, M.; Cerutti, F.; Rabbone, I.; Fontana, F.; Lera, R.; De Donno, V.; et al. Vitamin D levels at birth and risk of type 1 diabetes in childhood: A case-control study. Acta Diabetol. 2015, 52, 1077–1081. [Google Scholar] [CrossRef] [PubMed]
- Zipitis, C.S.; Akobeng, A.K. Vitamin D supplementation in early childhood and risk of type 1 diabetes: A systematic review and meta-analysis. Arch. Dis. Child. 2008, 93, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.-Y.; Zhang, W.; Chen, J.; Zhang, Z.-L.; Han, S.-F.; Qin, L.-Q. Vitamin D Intake and Risk of Type 1 Diabetes: A Meta-Analysis of Observational Studies. Nutrients 2013, 5, 3551–3562. [Google Scholar] [CrossRef] [PubMed]
- Dahlquist, G.; Patterson, C.; Soltesz, G.; Schober, E.; Schneider, U.; Goranova, S.; Stovanova, D.; Ivanova, M.; Urbonaite, B.; Pundziute-Lycka, A.; et al. Vitamin D supplement in early childhood and risk for Type I (insulin-dependent) diabetes mellitus. Diabetologia 1999, 42, 51–54. [Google Scholar]
- Hypponen, E.; Laara, E.; Reunanene, A.; Jarvelin, M.R.; Virtanen, S.M. Intake of vitamin D and risk of type 1 diabetes: A birth-cohort study. Lancet 2001, 358, 1500–1503. [Google Scholar] [CrossRef]
- Stene, L.C.; Ulriksen, J.; Magnus, P.; Joner, G. Use of cod liver oil during pregnancy associated with lower risk of Type I diabetes in the offspring. Diabetologia 2000, 43, 1093–1098. [Google Scholar] [CrossRef]
- Reinehr, T.; Schnabel, D.; Wabitsch, M.; Bechtold-Dalla Pozza, S.; Bührer, C.; Heidtmann, B.; Jochum, F.; Kauth, T.; Körner, A.; Mihatsch, W.; et al. Vitamin D supplementation after the second year of life: Joint position of the Committee on Nutrition, German Society for Pediatric and Adolescent Medicine (DGKJ e.V.), and the German Society for Pediatric Endocrinology and Diabetology (DGKED e.V.). Mol. Cell. Pediatrics 2019, 6, 3. [Google Scholar] [CrossRef]
- van der Mei, I.A.F.; Ponsonby, A.L.; Dwyer, T.; Blizzard, L.; Simmons, R.; Taylor, B.V.; Butzkueven, H.; Kilpatrick, T. Past exposure to sun, skin phenotype, and risk of multiple sclerosis: Case-control study. Br. Med. J. 2003, 327, 316–320. [Google Scholar] [CrossRef]
- Simpson, S.; Blizzard, L.; Otahal, P.; Van der Mei, I.; Taylor, B. Latitude is significantly associated with the prevalence of multiple sclerosis: A meta-analysis. J. Neurol. Neurosurg. Psychiatry 2011, 82, 1132–1141. [Google Scholar] [CrossRef]
- Alonso, A.; Hernan, M.A. Temporal trends in the incidence of multiple sclerosis. Neurology 2008, 71, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Rosati, G. The prevalence of multiple sclerosis in the world: An update. Neurol. Sci. 2001, 22, 117–139. [Google Scholar] [CrossRef] [PubMed]
- Waldman, A.; Ghezzi, A.; Bar-Or, A.; Mikaeloff, Y.; Tardieu, M.; Banwell, B. Multiple sclerosis in children: An update on clinical diagnosis, therapeutic strategies, and research. Lancet Neurol. 2014, 13, 936–948. [Google Scholar] [CrossRef]
- Mowry, E.M.; Krupp, L.B.; Milazzo, M.; Chabas, D.; Strober, J.B.; Belman, A.L.; McDonald, J.C.; Oksenberg, J.R.; Bacchetti, P.; Waubant, E. Vitamin D Status Is Associated with Relapse Rate in Pediatric-Onset Multiple Sclerosis. Ann. Neurol. 2010, 67, 618–624. [Google Scholar] [PubMed]
- Hedstrom, A.K.; Bomfim, I.L.; Barcellos, L.; Gianfrancesco, M.; Schaefer, C.; Kockum, I.; Olsson, T.; Alfredsson, L. Interaction between adolescent obesity and HLA risk genes in the etiology of multiple sclerosis. Neurology 2014, 82, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Mokry, L.E.; Ross, S.; Ahmad, O.S.; Forgetta, V.; Smith, G.D.; Leong, A.; Greenwood, C.M.T.; Thanassoulis, G.; Richards, J.B. Vitamin D and Risk of Multiple Sclerosis: A Mendelian Randomization Study. PLoS Med. 2015, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Gianfrancesco, M.A.; Stridh, P.; Rhead, B.; Shao, X.R.; Xu, E.; Graves, J.S.; Chitnis, T.; Waldman, A.; Lotze, T.; Schreiner, T.; et al. Evidence for a causal relationship between low vitamin D, high BMI, and pediatric-onset MS. Neurology 2017, 88, 1623–1629. [Google Scholar] [CrossRef]
- Marshak-Rothstein, A.; Rifkin, I.R. Immunologically Active Autoantigens: The Role of Toll-Like Receptors in the Development of Chronic Inflammatory Disease. Annu. Rev. Immunol. 2007, 25, 419–441. [Google Scholar] [CrossRef]
- Lima, G.L.; Paupitz, J.; Aikawa, N.E.; Takayama, L.; Bonfa, E.; Pereira, R.M.R. Vitamin D Supplementation in Adolescents and Young Adults With Juvenile Systemic Lupus Erythematosus for Improvement in Disease Activity and Fatigue Scores: A Randomized, Double-Blind, Placebo-Controlled Trial. Arthritis Care Res. 2016, 68, 91–98. [Google Scholar] [CrossRef]
- Ben-Zvi, I.; Aranow, C.; Mackay, M.; Stanevsky, A.; Kamen, D.L.; Marinescu, L.M.; Collins, C.E.; Gilkeson, G.S.; Diamond, B.; Hardin, J.A. The Impact of Vitamin D on Dendritic Cell Function in Patients with Systemic Lupus Erythematosus. PLoS ONE 2010, 5, 8. [Google Scholar] [CrossRef]
- Abou-Raya, A.; Abou-Raya, S.; Helmii, M. The Effect of Vitamin D Supplementation on Inflammatory and Hemostatic Markers and Disease Activity in Patients with Systemic Lupus Erythematosus: A Randomized Placebo-controlled Trial. J. Rheumatol. 2013, 40, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Aranow, C.; Kamen, D.L.; Dall’Era, M.; Massarotti, E.M.; Mackay, M.C.; Koumpouras, F.; Coca, A.; Chatham, W.W.; Clowse, M.E.B.; Criscione-Schreiber, L.G.; et al. Randomized, Double-Blind, Placebo-Controlled Trial of the Effect of Vitamin D3 on the Interferon Signature in Patients With Systemic Lupus Erythematosus. Arthritis Rheumatol. 2015, 67, 1848–1857. [Google Scholar] [CrossRef] [PubMed]
- Ravelli, A.; Martini, A. Juvenile idiopathic arthritis. Lancet 2007, 369, 767–778. [Google Scholar] [CrossRef]
- Ellis, J.A.; Munro, J.E.; Ponsonby, A.-L. Possible environmental determinants of juvenile idiopathic arthritis. Rheumatology 2009, 49, 411–425. [Google Scholar] [CrossRef]
- Chiaroni-Clarke, R.C.; Munro, J.E.; Pezic, A.; Cobb, J.E.; Akikusa, J.D.; Allen, R.C.; Dwyer, T.; Ponsonby, A.L.; Ellis, J.A. Association of Increased Sun Exposure Over the Life-course with a Reduced Risk of Juvenile Idiopathic Arthritis. Photochem. Photobiol. 2019, 95, 867–873. [Google Scholar] [CrossRef]
- Nisar, M.K.; Masood, F.; Cookson, P.; Sansome, A.; Ostor, A.J.K. What do we know about juvenile idiopathic arthritis and vitamin D? A systematic literature review and meta-analysis of current evidence. Clin. Rheumatol. 2013, 32, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Pelajo, C.F.; Lopez-Benitez, J.M.; Kent, D.M.; Price, L.L.; Miller, L.C.; Dawson-Hughes, B. 25-Hydroxyvitamin D levels and juvenile idiopathic arthritis: Is there an association with disease activity? Rheumatol. Int. 2012, 32, 3923–3929. [Google Scholar] [CrossRef]
- Çomak, E.; Doğan, Ç.S.; Uslu-Gökçeoğlu, A.; Akbaş, H.; Özdem, S.; Koyun, M.; Akman, S. Association between vitamin D deficiency and disease activity in juvenile idiopathic arthritis. Turk. J. Pediatrics 2014, 56, 626–631. [Google Scholar]
- Sengler, C.; Zink, J.; Klotsche, J.; Niewerth, M.; Liedmann, I.; Horneff, G.; Kessel, C.; Ganser, G.; Thon, A.; Haas, J.P.; et al. Vitamin D deficiency is associated with higher disease activity and the risk for uveitis in juvenile idiopathic arthritis - data from a German inception cohort. Arthritis Res. Ther. 2018, 20, 276. [Google Scholar] [CrossRef]
- Tang, T.; Zhang, Y.; Luo, C.; Liu, M.Y.; Xu, L.; Tang, X.M. Adjunctive vitamin D for the treatment of active juvenile idiopathic arthritis: An open-label, prospective, randomized controlled trial. Exp. Ther. Med. 2019, 18, 4921–4926. [Google Scholar] [CrossRef]
- Vassallo, M.F.; Banerji, A.; Rudders, S.A.; Clark, S.; Mullins, R.J.; Camargo, C.A., Jr. Season of birth and food allergy in children. Ann. Allergy Asthma Immunol. 2010, 104, 307–313. [Google Scholar] [CrossRef]
- Vuillermin, P.J.; Ponsonby, A.L.; Kemp, A.S.; Allen, K.J. Potential links between the emerging risk factors for food allergy and vitamin D status. Clin. Exp. Allergy 2013, 43, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Mullins, R.J.; Clark, S.; Katelaris, C.; Smith, V.; Solley, G.; Camargo, C.A., Jr. Season of birth and childhood food allergy in Australia. Pediatric Allergy Immunol. 2011, 22, 583–589. [Google Scholar] [CrossRef]
- Wjst, M. Introduction of oral vitamin D supplementation and the rise of the allergy pandemic. Allergy Asthma Clin. Immunol. 2009, 5, 8. [Google Scholar] [CrossRef] [PubMed]
- Kull, I.; Bergstrom, A.; Melen, E.; Lilja, G.; van Hage, M.; Pershagen, G.; Wickman, M. Early-life supplementation of vitamins A and D, in water-soluble form or in peanut oil, and allergic diseases during childhood. J. Allergy Clin. Immunol. 2006, 118, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Hypponen, E.; Sovio, U.; Wjst, M.; Patel, S.; Pekkanen, J.; Hartikainen, A.L.; Jarvelinb, M.R. Infant vitamin d supplementation and allergic conditions in adulthood: Northern Finland birth cohort 1966. Ann. NY Acad. Sci. 2004, 1037, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Robinson, S.M.; Harvey, N.C.; Javaid, M.K.; Jiang, B.; Martyn, C.N.; Godfrey, K.M.; Cooper, C. Maternal vitamin D status during pregnancy and child outcomes. Eur. J. Clin. Nutr. 2008, 62, 68–77. [Google Scholar] [CrossRef]
- Rothers, J.; Wright, A.L.; Stern, D.A.; Halonen, M.; Camargo, C.A., Jr. Cord blood 25-hydroxyvitamin D levels are associated with aeroallergen sensitization in children from Tucson, Arizona. J. Allergy Clin. Immunol. 2011, 128, 1093–1099 e1091–1095. [Google Scholar] [CrossRef]
- Hypponen, E.; Berry, D.J.; Wjst, M.; Power, C. Serum 25-hydroxyvitamin D and IgE—A significant but nonlinear relationship. Allergy 2009, 64, 613–620. [Google Scholar] [CrossRef]
- Wenzel, S.E. Asthma phenotypes: The evolution from clinical to molecular approaches. Nat. Med. 2012, 18, 716–725. [Google Scholar] [CrossRef]
- Zosky, G.R.; Berry, L.J.; Elliot, J.G.; James, A.L.; Gorman, S.; Hart, P.H. Vitamin D deficiency causes deficits in lung function and alters lung structure. Am. J. Respir. Crit. Care Med. 2011, 183, 1336–1343. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, P.E.; Hawrylowicz, C.M. Vitamin D in Asthma: Mechanisms of Action and Considerations for Clinical Trials. Chest 2018, 153, 1229–1239. [Google Scholar] [CrossRef]
- Saadoon, A.; Ambalavanan, N.; Zinn, K.; Ashraf, A.P.; MacEwen, M.; Nicola, T.; Fanucchi, M.V.; Harris, W.T. Effect of Prenatal versus Postnatal Vitamin D Deficiency on Pulmonary Structure and Function in Mice. Am. J. Respir. Cell Mol. Biol. 2017, 56, 383–392. [Google Scholar] [CrossRef]
- Song, H.; Yang, L.; Jia, C. Maternal vitamin D status during pregnancy and risk of childhood asthma: A meta-analysis of prospective studies. Mol. Nutr. Food Res. 2017, 61, 1600657. [Google Scholar] [CrossRef]
- Shen, S.Y.; Xiao, W.Q.; Lu, J.H.; Yuan, M.Y.; He, J.R.; Xia, H.M.; Qiu, X.; Cheng, K.K.; Lam, K.B.H. Early life vitamin D status and asthma and wheeze: A systematic review and meta-analysis. BMC Pulm. Med. 2018, 18, 120. [Google Scholar] [CrossRef] [PubMed]
- Thorsteinsdottir, F.; Cardoso, I.; Keller, A.; Stougaard, M.; Frederiksen, P.; Cohen, A.S.; Maslova, E.; Jacobsen, R.; Backer, V.; Heitmann, B.L. Neonatal Vitamin D Status and Risk of Asthma in Childhood: Results from the D-Tect Study. Nutrients 2020, 12, 842. [Google Scholar] [CrossRef] [PubMed]
- Litonjua, A.A.; Carey, V.J.; Laranjo, N.; Harshfield, B.J.; McElrath, T.F.; O’Connor, G.T.; Sandel, M.; Iverson, R.E., Jr.; Lee-Paritz, A.; Strunk, R.C.; et al. Effect of Prenatal Supplementation With Vitamin D on Asthma or Recurrent Wheezing in Offspring by Age 3 Years: The VDAART Randomized Clinical Trial. JAMA 2016, 315, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Litonjua, A.A.; Lange, N.E.; Carey, V.J.; Brown, S.; Laranjo, N.; Harshfield, B.J.; O’Connor, G.T.; Sandel, M.; Strunk, R.C.; Bacharier, L.B.; et al. The Vitamin D Antenatal Asthma Reduction Trial (VDAART): Rationale, design, and methods of a randomized, controlled trial of vitamin D supplementation in pregnancy for the primary prevention of asthma and allergies in children. Contemp. Clin. Trials. 2014, 38, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Chawes, B.L.; Bonnelykke, K.; Stokholm, J.; Vissing, N.H.; Bjarnadottir, E.; Schoos, A.M.; Wolsk, H.M.; Pedersen, T.M.; Vinding, R.K.; Thorsteinsdottir, S.; et al. Effect of Vitamin D3 Supplementation During Pregnancy on Risk of Persistent Wheeze in the Offspring: A Randomized Clinical Trial. JAMA 2016, 315, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Wolsk, H.M.; Chawes, B.L.; Litonjua, A.A.; Hollis, B.W.; Waage, J.; Stokholm, J.; Bonnelykke, K.; Bisgaard, H.; Weiss, S.T. Prenatal vitamin D supplementation reduces risk of asthma/recurrent wheeze in early childhood: A combined analysis of two randomized controlled trials. PLoS ONE 2017, 12, e0186657. [Google Scholar] [CrossRef]
- Litonjua, A.A.; Carey, V.J.; Laranjo, N.; Stubbs, B.J.; Mirzakhani, H.; O’Connor, G.T.; Sandel, M.; Beigelman, A.; Bacharier, L.B.; Zeiger, R.S.; et al. Six-Year Follow-up of a Trial of Antenatal Vitamin D for Asthma Reduction. N. Engl. J. Med. 2020, 382, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Brustad, N.; Eliasen, A.U.; Stokholm, J.; Bonnelykke, K.; Bisgaard, H.; Chawes, B.L. High-Dose Vitamin D Supplementation During Pregnancy and Asthma in Offspring at the Age of 6 Years. JAMA 2019, 321, 1003–1005. [Google Scholar] [CrossRef] [PubMed]
- Hornsby, E.; Pfeffer, P.E.; Laranjo, N.; Cruikshank, W.; Tuzova, M.; Litonjua, A.A.; Weiss, S.T.; Carey, V.J.; O’Connor, G.; Hawrylowicz, C. Vitamin D supplementation during pregnancy: Effect on the neonatal immune system in a randomized controlled trial. J. Allergy Clin. Immunol. 2018, 141, 269–278 e2. [Google Scholar] [CrossRef] [PubMed]
- Hamzaoui, A.; Berraies, A.; Hamdi, B.; Kaabachi, W.; Ammar, J.; Hamzaoui, K. Vitamin D reduces the differentiation and expansion of Th17 cells in young asthmatic children. Immunobiology 2014, 219, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Jeffery, L.E.; Burke, F.; Mura, M.; Zheng, Y.; Qureshi, O.S.; Hewison, M.; Walker, L.S.; Lammas, D.A.; Raza, K.; Sansom, D.M. 1, 25-Dihydroxyvitamin D3 and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing CTLA-4 and FoxP3. J. Immunol. 2009, 183, 5458–5467. [Google Scholar] [CrossRef] [PubMed]
- Nanzer, A.M.; Chambers, E.S.; Ryanna, K.; Richards, D.F.; Black, C.; Timms, P.M.; Martineau, A.R.; Griffiths, C.J.; Corrigan, C.J.; Hawrylowicz, C.M. Enhanced production of IL-17A in patients with severe asthma is inhibited by 1alpha,25-dihydroxyvitamin D3 in a glucocorticoid-independent fashion. J. Allergy Clin. Immunol. 2013, 132, 297–304 e3. [Google Scholar] [CrossRef]
- Hibbs, A.M.; Ross, K.; Kerns, L.A.; Wagner, C.; Fuloria, M.; Groh-Wargo, S.; Zimmerman, T.; Minich, N.; Tatsuoka, C. Effect of Vitamin D Supplementation on Recurrent Wheezing in Black Infants Who Were Born Preterm: The D-Wheeze Randomized Clinical Trial. JAMA 2018, 319, 2086–2094. [Google Scholar] [CrossRef]
- Ducharme, F.M.; Jensen, M.; Mailhot, G.; Alos, N.; White, J.; Rousseau, E.; Tse, S.M.; Khamessan, A.; Vinet, B. Impact of two oral doses of 100,000 IU of vitamin D3 in preschoolers with viral-induced asthma: A pilot randomised controlled trial. Trials 2019, 20, 138. [Google Scholar] [CrossRef]
- Jensen, M.E.; Mailhot, G.; Alos, N.; Rousseau, E.; White, J.H.; Khamessan, A.; Ducharme, F.M. Vitamin D intervention in preschoolers with viral-induced asthma (DIVA): A pilot randomised controlled trial. Trials 2016, 17, 353. [Google Scholar] [CrossRef]
- Freishtat, R.J.; Iqbal, S.F.; Pillai, D.K.; Klein, C.J.; Ryan, L.M.; Benton, A.S.; Teach, S.J. High prevalence of vitamin D deficiency among inner-city African American youth with asthma in Washington, DC. J. Pediatr. 2010, 156, 948–952. [Google Scholar] [CrossRef]
- Gupta, A.; Sjoukes, A.; Richards, D.; Banya, W.; Hawrylowicz, C.; Bush, A.; Saglani, S. Relationship between serum vitamin D, disease severity, and airway remodeling in children with asthma. Am. J. Respir. Crit. Care Med. 2011, 184, 1342–1349. [Google Scholar] [CrossRef] [PubMed]
- Beigelman, A.; Zeiger, R.S.; Mauger, D.; Strunk, R.C.; Jackson, D.J.; Martinez, F.D.; Morgan, W.J.; Covar, R.; Szefler, S.J.; Taussig, L.M.; et al. The association between vitamin D status and the rate of exacerbations requiring oral corticosteroids in preschool children with recurrent wheezing. J. Allergy Clin. Immunol. 2014, 133, 1489–1492. [Google Scholar] [CrossRef] [PubMed]
- Searing, D.A.; Zhang, Y.; Murphy, J.R.; Hauk, P.J.; Goleva, E.; Leung, D.Y. Decreased serum vitamin D levels in children with asthma are associated with increased corticosteroid use. J. Allergy Clin. Immunol. 2010, 125, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Stenberg Hammar, K.; Hedlin, G.; Konradsen, J.R.; Nordlund, B.; Kull, I.; Giske, C.G.; Pedroletti, C.; Soderhall, C.; Melen, E. Subnormal levels of vitamin D are associated with acute wheeze in young children. Acta Paediatr 2014, 103, 856–861. [Google Scholar] [CrossRef]
- Turkeli, A.; Ayaz, O.; Uncu, A.; Ozhan, B.; Bas, V.N.; Tufan, A.K.; Yilmaz, O.; Yuksel, H. Effects of vitamin D levels on asthma control and severity in pre-school children. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 26–36. [Google Scholar]
- Uysalol, M.; Uysalol, E.P.; Yilmaz, Y.; Parlakgul, G.; Ozden, T.A.; Ertem, H.V.; Omer, B.; Uzel, N. Serum level of vitamin D and trace elements in children with recurrent wheezing: A cross-sectional study. BMC Pediatr. 2014, 14, 270. [Google Scholar] [CrossRef]
- Brehm, J.M.; Acosta-Perez, E.; Klei, L.; Roeder, K.; Barmada, M.; Boutaoui, N.; Forno, E.; Kelly, R.; Paul, K.; Sylvia, J.; et al. Vitamin D insufficiency and severe asthma exacerbations in Puerto Rican children. Am. J. Respir. Crit. Care Med. 2012, 186, 140–146. [Google Scholar] [CrossRef]
- Brehm, J.M.; Celedon, J.C.; Soto-Quiros, M.E.; Avila, L.; Hunninghake, G.M.; Forno, E.; Laskey, D.; Sylvia, J.S.; Hollis, B.W.; Weiss, S.T.; et al. Serum vitamin D levels and markers of severity of childhood asthma in Costa Rica. Am. J. Respir. Crit. Care Med. 2009, 179, 765–771. [Google Scholar] [CrossRef]
- Brehm, J.M.; Schuemann, B.; Fuhlbrigge, A.L.; Hollis, B.W.; Strunk, R.C.; Zeiger, R.S.; Weiss, S.T.; Litonjua, A.A.; Childhood Asthma Management Program Research Group. Serum vitamin D levels and severe asthma exacerbations in the Childhood Asthma Management Program study. J. Allergy Clin. Immunol. 2010, 126, 52–58.e5. [Google Scholar] [CrossRef]
- Chinellato, I.; Piazza, M.; Sandri, M.; Peroni, D.; Piacentini, G.; Boner, A.L. Vitamin D serum levels and markers of asthma control in Italian children. J. Pediatr. 2011, 158, 437–441. [Google Scholar] [CrossRef]
- Korn, S.; Hubner, M.; Jung, M.; Blettner, M.; Buhl, R. Severe and uncontrolled adult asthma is associated with vitamin D insufficiency and deficiency. Respir. Res. 2013, 14, 25. [Google Scholar] [CrossRef] [PubMed]
- Samrah, S.; Khatib, I.; Omari, M.; Khassawneh, B.; Momany, S.; Daoud, A.; Malkawi, M.; Khader, Y. Vitamin D deficiency and level of asthma control in women from North of Jordan: A case-control study. J. Asthma. 2014, 51, 832–838. [Google Scholar] [CrossRef]
- Wu, A.C.; Tantisira, K.; Li, L.; Fuhlbrigge, A.L.; Weiss, S.T.; Litonjua, A.; Childhood Asthma Management Program Research Group. Effect of vitamin D and inhaled corticosteroid treatment on lung function in children. Am. J. Respir. Crit. Care Med. 2012, 186, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Hysinger, E.B.; Roizen, J.D.; Mentch, F.D.; Vazquez, L.; Connolly, J.J.; Bradfield, J.P.; Almoguera, B.; Sleiman, P.M.; Allen, J.L.; Levine, M.A.; et al. Mendelian randomization analysis demonstrates that low vitamin D is unlikely causative for pediatric asthma. J. Allergy Clin. Immunol. 2016, 138, 1747–1749.e4. [Google Scholar] [CrossRef] [PubMed]
- Manousaki, D.; Paternoster, L.; Standl, M.; Moffatt, M.F.; Farrall, M.; Bouzigon, E.; Strachan, D.P.; Demenais, F.; Lathrop, M.; Cookson, W.; et al. Vitamin D levels and susceptibility to asthma, elevated immunoglobulin E levels, and atopic dermatitis: A Mendelian randomization study. PLoS Med. 2017, 14, e1002294. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Dozor, A.J. Do you believe milk makes mucus? Arch. Pediatr. Adolesc. Med. 2004, 158, 601–603. [Google Scholar] [CrossRef]
- Haas, F.; Bishop, M.C.; Salazar-Schicchi, J.; Axen, K.V.; Lieberman, D.; Axen, K. Effect of milk ingestion on pulmonary function in healthy and asthmatic subjects. J. Asthma. 1991, 28, 349–355. [Google Scholar] [CrossRef]
- Nguyen, M.T. Effect of cow milk on pulmonary function in atopic asthmatic patients. Ann. Allergy Asthma. Immunol. 1997, 79, 62–64. [Google Scholar] [CrossRef]
- Waldron, J.L.; Ashby, H.L.; Cornes, M.P.; Bechervaise, J.; Razavi, C.; Thomas, O.L.; Chugh, S.; Deshpande, S.; Ford, C.; Gama, R. Vitamin D: A negative acute phase reactant. J. Clin. Pathol. 2013, 66, 620–622. [Google Scholar] [CrossRef]
- Jolliffe, D.A.; Stefanidis, C.; Wang, Z.; Kermani, N.Z.; Dimitrov, V.; White, J.H.; McDonough, J.E.; Janssens, W.; Pfeffer, P.; Griffiths, C.J.; et al. Vitamin D Metabolism is Dysregulated in Asthma and Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2020. [Google Scholar] [CrossRef]
- Martineau, A.R.; Cates, C.J.; Urashima, M.; Jensen, M.; Griffiths, A.P.; Nurmatov, U.; Sheikh, A.; Griffiths, C.J. Vitamin D for the management of asthma. Cochrane Database Syst. Rev. 2016, 9, CD011511. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, D.A.; Greenberg, L.; Hooper, R.L.; Griffiths, C.J.; Camargo, C.A., Jr.; Kerley, C.P.; Jensen, M.E.; Mauger, D.; Stelmach, I.; Urashima, M.; et al. Vitamin D supplementation to prevent asthma exacerbations: A systematic review and meta-analysis of individual participant data. Lancet Respir. Med. 2017, 5, 881–890. [Google Scholar] [CrossRef]
- Hansdottir, S.; Monick, M.M.; Hinde, S.L.; Lovan, N.; Look, D.C.; Hunninghake, G.W. Respiratory epithelial cells convert inactive vitamin D to its active form: Potential effects on host defense. J. Immunol. 2008, 181, 7090–7099. [Google Scholar] [CrossRef] [PubMed]
- Greiller, C.L.; Suri, R.; Jolliffe, D.A.; Kebadze, T.; Hirsman, A.G.; Griffiths, C.J.; Johnston, S.L.; Martineau, A.R. Vitamin D attenuates rhinovirus-induced expression of intercellular adhesion molecule-1 (ICAM-1) and platelet-activating factor receptor (PAFR) in respiratory epithelial cells. J. Steroid Biochem. Mol. Biol. 2019, 187, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Hansdottir, S.; Monick, M.M.; Lovan, N.; Powers, L.; Gerke, A.; Hunninghake, G.W. Vitamin D decreases respiratory syncytial virus induction of NF-kappaB-linked chemokines and cytokines in airway epithelium while maintaining the antiviral state. J. Immunol. 2010, 184, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Pfeffer, P.E.; Chen, Y.H.; Woszczek, G.; Matthews, N.C.; Chevretton, E.; Gupta, A.; Saglani, S.; Bush, A.; Corrigan, C.; Cousins, D.J.; et al. Vitamin D enhances production of soluble ST2, inhibiting the action of IL-33. J. Allergy Clin. Immunol. 2015, 135, 824–827.e3. [Google Scholar]
- Ducharme, F.M.; Dell, S.D.; Radhakrishnan, D.; Grad, R.M.; Watson, W.T.; Yang, C.L.; Zelman, M. Diagnosis and management of asthma in preschoolers: A Canadian Thoracic Society and Canadian Paediatric Society position paper. Can. Respir. J. 2015, 22, 135–143. [Google Scholar] [CrossRef]
- Pruteanu, A.I.; Chauhan, B.F.; Zhang, L.; Prietsch, S.O.; Ducharme, F.M. Inhaled corticosteroids in children with persistent asthma: Dose-response effects on growth. Cochrane Database Syst. Rev. 2014, 7, CD009878. [Google Scholar] [CrossRef]
- Ducharme, F.M.; Zemek, R.; Chauhan, B.F.; Gravel, J.; Chalut, D.; Poonai, N.; Guertin, M.C.; Quach, C.; Blondeau, L.; Laberge, S.; et al. Factors associated with failure of emergency department management in children with acute moderate or severe asthma: A prospective, multicentre, cohort study. Lancet Respir. Med. 2016, 4, 990–998. [Google Scholar] [CrossRef]
- Goleva, E.; Searing, D.A.; Jackson, L.P.; Richers, B.N.; Leung, D.Y. Steroid requirements and immune associations with vitamin D are stronger in children than adults with asthma. J. Allergy Clin. Immunol. 2012, 129, 1243–1251. [Google Scholar] [CrossRef][Green Version]
- Zhang, Y.; Leung, D.Y.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.W.; Goleva, E. Vitamin D inhibits monocyte/macrophage proinflammatory cytokine production by targeting MAPK phosphatase-1. J. Immunol. 2012, 188, 2127–2135. [Google Scholar] [CrossRef]
- Ferreira, G.B.; Kleijwegt, F.S.; Waelkens, E.; Lage, K.; Nikolic, T.; Hansen, D.A.; Workman, C.T.; Roep, B.O.; Overbergh, L.; Mathieu, C. Differential protein pathways in 1, 25-dihydroxyvitamin D3 and dexamethasone modulated tolerogenic human dendritic cells. J. Proteome Res. 2012, 11, 941–971. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.S.; Nanzer, A.M.; Pfeffer, P.E.; Richards, D.F.; Timms, P.M.; Martineau, A.R.; Griffiths, C.J.; Corrigan, C.J.; Hawrylowicz, C.M. Distinct endotypes of steroid-resistant asthma characterized by IL-17A(high) and IFN-gamma(high) immunophenotypes: Potential benefits of calcitriol. J. Allergy Clin. Immunol. 2015, 136, 628–637.e4. [Google Scholar] [CrossRef] [PubMed]
- Nanzer, A.M.; Chambers, E.S.; Ryanna, K.; Freeman, A.T.; Colligan, G.; Richards, D.F.; Timms, P.M.; Martineau, A.R.; Griffiths, C.J.; Corrigan, C.J.; et al. The effects of calcitriol treatment in glucocorticoid-resistant asthma. J. Allergy Clin. Immunol. 2014, 133, 1755–1757 e1754. [Google Scholar] [CrossRef] [PubMed]
- Lyons, J.J.; Milner, J.D.; Stone, K.D. Atopic dermatitis in children: Clinical features, pathophysiology, and treatment. Immunol. Allergy Clin. North. Am. 2015, 35, 161–183. [Google Scholar] [CrossRef]
- Ong, P.Y.; Ohtake, T.; Brandt, C.; Strickland, I.; Boguniewicz, M.; Ganz, T.; Gallo, R.L.; Leung, D.Y. Endogenous antimicrobial peptides and skin infections in atopic dermatitis. N. Engl. J. Med. 2002, 347, 1151–1160. [Google Scholar] [CrossRef]
- Mallbris, L.; Carlen, L.; Wei, T.; Heilborn, J.; Nilsson, M.F.; Granath, F.; Stahle, M. Injury downregulates the expression of the human cathelicidin protein hCAP18/LL-37 in atopic dermatitis. Exp. Dermatol. 2010, 19, 442–449. [Google Scholar] [CrossRef]
- Schauber, J.; Dorschner, R.A.; Coda, A.B.; Buchau, A.S.; Liu, P.T.; Kiken, D.; Helfrich, Y.R.; Kang, S.; Elalieh, H.Z.; Steinmeyer, A.; et al. Injury enhances TLR2 function and antimicrobial peptide expression through a vitamin D-dependent mechanism. J. Clin. Invest. 2007, 117, 803–811. [Google Scholar] [CrossRef]
- Hata, T.R.; Kotol, P.; Jackson, M.; Nguyen, M.; Paik, A.; Udall, D.; Kanada, K.; Yamasaki, K.; Alexandrescu, D.; Gallo, R.L. Administration of oral vitamin D induces cathelicidin production in atopic individuals. J. Allergy Clin. Immunol. 2008, 122, 829–831. [Google Scholar] [CrossRef]
- Albenali, L.H.; Danby, S.; Moustafa, M.; Brown, K.; Chittock, J.; Shackley, F.; Cork, M.J. Vitamin D and antimicrobial peptide levels in patients with atopic dermatitis and atopic dermatitis complicated by eczema herpeticum: A pilot study. J. Allergy Clin. Immunol. 2016, 138, 1715–1719.e4. [Google Scholar] [CrossRef]
- Hattangdi-Haridas, S.R.; Lanham-New, S.A.; Wong, W.H.S.; Ho, M.H.K.; Darling, A.L. Vitamin D Deficiency and Effects of Vitamin D Supplementation on Disease Severity in Patients with Atopic Dermatitis: A Systematic Review and Meta-Analysis in Adults and Children. Nutrients 2019, 11, 1854. [Google Scholar] [CrossRef] [PubMed]
- Schram, M.E.; Spuls, P.I.; Leeflang, M.M.; Lindeboom, R.; Bos, J.D.; Schmitt, J. EASI, (objective) SCORAD and POEM for atopic eczema: Responsiveness and minimal clinically important difference. Allergy 2012, 67, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Heine, G.; Anton, K.; Henz, B.M.; Worm, M. 1alpha, 25-dihydroxyvitamin D3 inhibits anti-CD40 plus IL-4-mediated IgE production in vitro. Eur. J. Immunol. 2002, 32, 3395–3404. [Google Scholar] [PubMed]
- Matheu, V.; Back, O.; Mondoc, E.; Issazadeh-Navikas, S. Dual effects of vitamin D-induced alteration of TH1/TH2 cytokine expression: Enhancing IgE production and decreasing airway eosinophilia in murine allergic airway disease. J. Allergy Clin. Immunol. 2003, 112, 585–592. [Google Scholar] [CrossRef]
- Hartmann, B.; Heine, G.; Babina, M.; Steinmeyer, A.; Zugel, U.; Radbruch, A.; Worm, M. Targeting the vitamin D receptor inhibits the B cell-dependent allergic immune response. Allergy 2011, 66, 540–548. [Google Scholar] [CrossRef]
- Fujimura, K.E.; Lynch, S.V. Microbiota in allergy and asthma and the emerging relationship with the gut microbiome. Cell Host Microbe. 2015, 17, 592–602. [Google Scholar] [CrossRef]
- Sordillo, J.E.; Zhou, Y.; McGeachie, M.J.; Ziniti, J.; Lange, N.; Laranjo, N.; Savage, J.R.; Carey, V.; O’Connor, G.; Sandel, M.; et al. Factors influencing the infant gut microbiome at age 3–6 months: Findings from the ethnically diverse Vitamin D Antenatal Asthma Reduction Trial (VDAART). J. Allergy Clin. Immunol. 2017, 139, 482–491.e4. [Google Scholar] [CrossRef]
- Sharief, S.; Jariwala, S.; Kumar, J.; Muntner, P.; Melamed, M.L. Vitamin D levels and food and environmental allergies in the United States: Results from the National Health and Nutrition Examination Survey 2005–2006. J. Allergy Clin. Immunol. 2011, 127, 1195–1202. [Google Scholar] [CrossRef]
- Allen, K.J.; Koplin, J.J.; Ponsonby, A.L.; Gurrin, L.C.; Wake, M.; Vuillermin, P.; Martin, P.; Matheson, M.; Lowe, A.; Robinson, M.; et al. Vitamin D insufficiency is associated with challenge-proven food allergy in infants. J. Allergy Clin. Immunol. 2013, 131, 1109–1116. [Google Scholar] [CrossRef]
- Liu, X.; Arguelles, L.; Zhou, Y.; Wang, G.; Chen, Q.; Tsai, H.J.; Hong, X.; Liu, R.; Price, H.E.; Pearson, C.; et al. Longitudinal trajectory of vitamin D status from birth to early childhood in the development of food sensitization. Pediatr. Res. 2013, 74, 321–326. [Google Scholar] [CrossRef]
- Liu, X.; Wang, G.; Hong, X.; Wang, D.; Tsai, H.J.; Zhang, S.; Arguelles, L.; Kumar, R.; Wang, H.; Liu, R.; et al. Gene-vitamin D interactions on food sensitization: A prospective birth cohort study. Allergy 2011, 66, 1442–1448. [Google Scholar] [CrossRef]
- Persson, K.; Ohlund, I.; Nordstrom, L.; Winberg, A.; Ronmark, E.; West, C.E. Vitamin D deficiency at the Arctic Circle - a study in food-allergic adolescents and controls. Acta Paediatr. 2013, 102, 644–649. [Google Scholar] [CrossRef] [PubMed]
- Junge, K.M.; Bauer, T.; Geissler, S.; Hirche, F.; Thurmann, L.; Bauer, M.; Trump, S.; Bieg, M.; Weichenhan, D.; Gu, L.; et al. Increased vitamin D levels at birth and in early infancy increase offspring allergy risk-evidence for involvement of epigenetic mechanisms. J. Allergy Clin. Immunol. 2016, 137, 610–613. [Google Scholar] [CrossRef] [PubMed]
- Weisse, K.; Winkler, S.; Hirche, F.; Herberth, G.; Hinz, D.; Bauer, M.; Roder, S.; Rolle-Kampczyk, U.; von Bergen, M.; Olek, S.; et al. Maternal and newborn vitamin D status and its impact on food allergy development in the German LINA cohort study. Allergy 2013, 68, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Allen, K.J.; Panjari, M.; Koplin, J.J.; Ponsonby, A.L.; Vuillermin, P.; Gurrin, L.C.; Greaves, R.; Carvalho, N.; Dalziel, K.; Tang, M.L.; et al. VITALITY trial: Protocol for a randomised controlled trial to establish the role of postnatal vitamin D supplementation in infant immune health. BMJ Open 2015, 5, e009377. [Google Scholar] [CrossRef]
- Aryan, Z.; Rezaei, N.; Camargo, C.A., Jr. Vitamin D status, aeroallergen sensitization, and allergic rhinitis: A systematic review and meta-analysis. Int. Rev. Immunol. 2017, 36, 41–53. [Google Scholar] [CrossRef]
- Grant, C.C.; Crane, J.; Mitchell, E.A.; Sinclair, J.; Stewart, A.; Milne, T.; Knight, J.; Gilchrist, C.; Camargo, C.A., Jr. Vitamin D supplementation during pregnancy and infancy reduces aeroallergen sensitization: A randomized controlled trial. Allergy 2016, 71, 1325–1334. [Google Scholar] [CrossRef]
| Clinicaltrials.gov Identifier | Setting | n * | Population | Interventions | Primary Outcomes | Expected Completion Year |
|---|---|---|---|---|---|---|
| Crohn’s Disease | ||||||
| NCT03999580 (ViDiPeC-2) | Canada | 316 | Pediatric Crohn’s disease patients (4–18 years) | 3000 or 4000 IU/day, acc. to body weight for 4 weeks, 2000 IU/day for 48 weeks. Control: 600 IU/day induction (4 weeks) and maintenance (48 weeks). | Number of relapses and quality of life, incl. levels of physical activity | 2024 |
| Asthma | ||||||
| NCT03365687 (DIVA) | Canada | 864 | Preschoolers (1–5 years) with viral infection-triggered wheezing/asthma | Two oral boluses of 100,000 IU vitamin D3 3.5 months apart with daily 400 IU vitamin D3 for 7 months. Placebo boluses, daily placebo | Number of asthma exacerbations treated with rescue OCS | 2023 |
| NCT02687815 (Vit-D-Kids Asthma) | USA | 400 | Schoolchildren (6–16 years) Treated with ICS Vitamin D insufficient (25OHD < 75 nM) | Daily 4000 IU vitamin D3 or placebo for 48 weeks | Severe asthma exacerbations requiring systemic CS or an increase in stable maintenance dose for at least 3 days Asthma-related hospitalization or emergency room visit requiring OCS | Completed, unpublished |
| Food allergies | ||||||
| NCT02112734 (Vitality) | Australia | 3555 | Healthy term infants (6 to 12 weeks) Predominantly breastfed | Daily 400 IU vitamin D3 or placebo for 12 months | Prevalence of challenge-proven food allergy at 1 year of age | 2022 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mailhot, G.; White, J.H. Vitamin D and Immunity in Infants and Children. Nutrients 2020, 12, 1233. https://doi.org/10.3390/nu12051233
Mailhot G, White JH. Vitamin D and Immunity in Infants and Children. Nutrients. 2020; 12(5):1233. https://doi.org/10.3390/nu12051233
Chicago/Turabian StyleMailhot, Geneviève, and John H. White. 2020. "Vitamin D and Immunity in Infants and Children" Nutrients 12, no. 5: 1233. https://doi.org/10.3390/nu12051233
APA StyleMailhot, G., & White, J. H. (2020). Vitamin D and Immunity in Infants and Children. Nutrients, 12(5), 1233. https://doi.org/10.3390/nu12051233






