Role of Vitamin D in Preventing and Treating Selected Extraskeletal Diseases—An Umbrella Review
Abstract
1. Introduction
2. Methods
2.1. Protocol, Registration, and Study Design
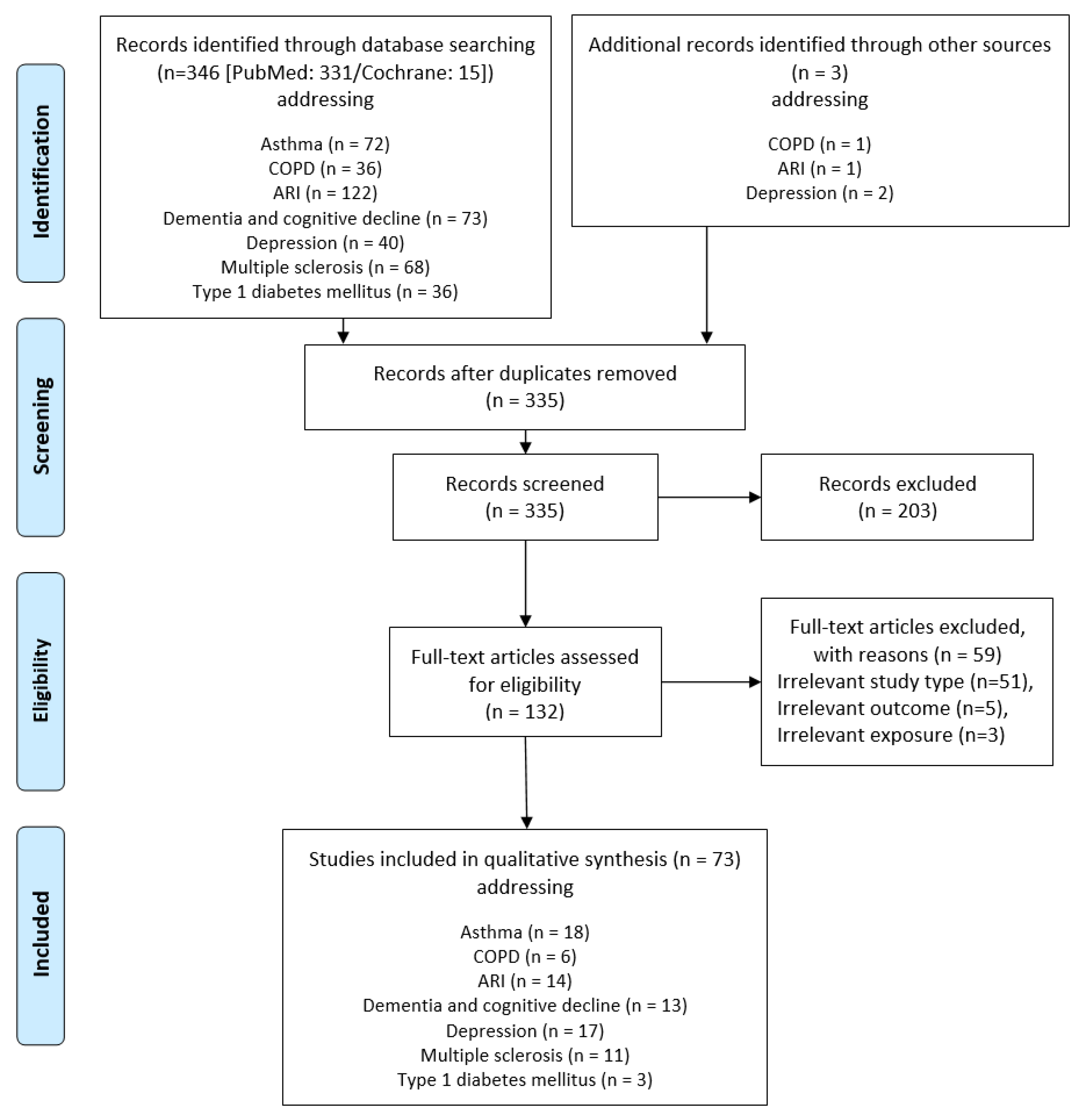
2.2. Search Strategy and Eligibility Criteria
2.3. Data Extraction
2.4. Assessment of Methodological Quality
2.5. Summarisation of Data Results and Conclusion Drawing
3. Results
3.1. Respiratory Tract Diseases
3.1.1. Asthma
3.1.2. Chronic Obstructive Pulmonary Disease
3.1.3. Acute Respiratory Tract Infections
3.2. Neurodegenerative and Mental Diseases
3.2.1. Dementia and Cognitive Decline
3.2.2. Depression
3.3. Autoimmune Diseases
3.3.1. Multiple Sclerosis
3.3.2. Type 1 Diabetes Mellitus
4. Overall Discussion and Conclusion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zittermann, A.; Pilz, S. Vitamin D in Klinik und Praxis. Dtsch. Med. Wochenschr. 2017, 142, 601–616. [Google Scholar]
- Institute of Medicine. Dietary Reference Intakes: Calcium and Vitamin D; The National Academic Press: Washington, DC, USA, 2011. [Google Scholar]
- Nordic Council of Ministers. Nordic Nutrition Recommendations 2012. Integrating Nutrition and Physical Activity, 5th ed.; Nordic Council of Ministers: Copenhagen, Denmark, 2014; pp. 349–384. [Google Scholar]
- German Nutrition Society; Austrian Nutrition Society; Swiss Nutrition Society. Referenzwerte für die Nährstoffzufuhr, 2nd ed.; DGE: Bonn, Germany, 2015. [Google Scholar]
- EFSA (European Food Safety Authority). Dietary reference values for vitamin D: EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). EFSA J. 2016, 14, e04547. [Google Scholar] [CrossRef]
- Seckmeyer, G.; Schrempf, M.; Wieczorek, A.; Riechelmann, S.; Graw, K.; Seckmeyer, S.; Zankl, M. A novel method to calculate solar UV exposure relevant to vitamin D production in humans. Photochem. Photobiol. 2013, 89, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am. J. Clin. Nutr. 2004, 80, 1678–1688. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Heaney, R.P.; Holick, M.F.; Lips, P.; Meunier, P.J.; Vieth, R. Estimates of optimal vitamin D status. Osteoporos. Int. 2005, 16, 713–716. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D status: Measurement, interpretation, and clinical application. Ann. Epidemiol. 2009, 19, 73–78. [Google Scholar] [CrossRef]
- Linseisen, J.; Bechthold, A.; Bischoff-Ferrari, H.A.; Hintzpeter, B.; Leschik-Bonnet, E.; Reichrath, J.; Stehle, P.; Volkert, D.; Wolfram, G.; Zittermann, A. DGE-Stellungnahme: Vitamin D und Prävention Ausgewählter Chronischer Krankheiten. Available online: http://www.dge.de/pdf/ws/DGE-Stellungnahme-VitD-111220.pdf (accessed on 26 June 2013).
- Pilz, S.; Zittermann, A.; Trummer, C.; Theiler-Schwetz, V.; Lerchbaum, E.; Keppel, M.H.; Grübler, M.R.; März, W.; Pandis, M. Vitamin D testing and treatment: A narrative review of current evidence. Endocr. Connect. 2019, 8, R27–R43. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; de Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef]
- Van Schoor, N.M.; Lips, P. Worldwide vitamin D status. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 671–680. [Google Scholar] [CrossRef]
- Lips, P. Vitamin D status and nutrition in Europe and Asia. J. Steroid. Biochem. Mol. Biol. 2007, 103, 620–625. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Altieri, B.; Annweiler, C.; Balercia, G.; Pal, H.B.; Boucher, B.J.; Cannell, J.J.; Foresta, C.; Grübler, M.R.; Kotsa, K.; et al. Vitamin D and chronic diseases: The current state of the art. Arch. Toxicol. 2017, 91, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, S.; Na, S.; Rathnachalam, R. Noncalcemic actions of vitamin D receptor ligands. Endocr. Rev. 2005, 26, 662–687. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.S.; Hewison, M. Extrarenal expression of the 25-hydroxyvitamin D-1-hydroxylase. Arch. Biochem. Biophys. 2012, 523, 95–102. [Google Scholar] [CrossRef]
- Barnard, K.; Colón-Emeric, C. Extraskeletal effects of vitamin D in older adults: Cardiovascular disease, mortality, mood, and cognition. Am. J. Geriatr. Pharmacother. 2010, 8, 4–33. [Google Scholar] [CrossRef]
- Elamin, M.B.; Abu Elnour, N.O.; Elamin, K.B.; Fatourechi, M.M.; Alkatib, A.A.; Almandoz, J.P.; Liu, H.; Lane, M.A.; Mullan, R.J.; Hazem, A.; et al. Vitamin D and cardiovascular outcomes: A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2011, 96, 1931–1942. [Google Scholar] [CrossRef]
- Martins, D.; Wolf, M.; Pan, D.; Zadshir, A.; Tareen, N.; Thadhani, R.; Felsenfeld, A.; Levine, B.; Mehrotra, R.; Norris, K. Prevalence of cardiovascular risk factors and the serum levels of 25-hydroxyvitamin D in the United States: Data from the Third National Health and Nutrition Examination Survey. Arch. Intern. Med. 2007, 167, 1159–1165. [Google Scholar] [CrossRef]
- Gorham, E.D.; Garland, C.F.; Garland, F.C.; Grant, W.B.; Mohr, S.B.; Lipkin, M.; Newmark, H.L.; Giovannucci, E.; Wei, M.; Holick, M.F. Optimal vitamin D status for colorectal cancer prevention: A quantitative meta analysis. Am. J. Prev. Med. 2007, 32, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Grandi, N.; Raum, E.; Haug, U.; Arndt, V.; Brenner, H. Meta-analysis: Longitudinal studies of serum vitamin D and colorectal cancer risk. Aliment. Pharmacol. Ther. 2009, 30, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Gandini, S.; Boniol, M.; Haukka, J.; Byrnes, G.; Cox, B.; Sneyd, M.J.; Mullie, P.; Autier, P. Meta-analysis of observational studies of serum 25-hydroxyvitamin D levels and colorectal, breast and prostate cancer and colorectal adenoma. Int. J. Cancer 2011, 128, 1414–1424. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Lau, J.; Hu, F.B.; Dawson-Hughes, B. The role of vitamin D and calcium in type 2 diabetes. A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2007, 92, 2017–2029. [Google Scholar] [CrossRef]
- Mitri, J.; Muraru, M.D.; Pittas, A.G. Vitamin D and type 2 diabetes: A systematic review. Eur. J. Clin. Nutr. 2011, 65, 1005–1015. [Google Scholar] [CrossRef] [PubMed]
- Witham, M.D.; Nadir, M.A.; Struthers, A.D. Effect of vitamin D on blood pressure: A systematic review and meta-analysis. J. Hypertens. 2009, 27, 1948–1954. [Google Scholar] [CrossRef] [PubMed]
- Golzarand, M.; Shab-Bidar, S.; Koochakpoor, G.; Speakman J, R.; Djafarian, K. Effect of vitamin D3 supplementation on blood pressure in adults: An updated meta-analysis. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Keum, N.; Lee, D.H.; Greenwood, D.C.; Manson, J.E.; Giovannucci, E. Vitamin D supplementation and total cancer incidence and mortality: A meta-analysis of randomized controlled trials. Ann. Oncol. 2019, 30, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Caraba, A.; Crisan, V.; Romosan, I.; Mozos, I.; Murariu, M. Vitamin D status, disease activity, and endothelial dysfunction in early rheumatoid arthritis patients. Dis. Markers 2017, 2017, 5241012. [Google Scholar] [CrossRef]
- Rejnmark, L.; Bislev, L.S.; Cashman, K.D.; Eiríksdottir, G.; Gaksch, M.; Grübler, M.; Grimnes, G.; Gudnason, V.; Lips, P.; Pilz, S.; et al. Non-skeletal health effects of vitamin D supplementation: A systematic review on findings from meta-analyses summarizing trial data. PLoS ONE 2017, 12, e0180512. [Google Scholar] [CrossRef]
- Autier, P.; Mullie, P.; Macacu, A.; Dragomir, M.; Boniol, M.; Coppens, K.; Pizot, C.; Boniol, M. Effect of vitamin D supplementation on non-skeletal disorders: A systematic review of meta-analyses and randomised trials. Lancet Diabetes Endocrinol. 2017, 5, 986–1004. [Google Scholar] [CrossRef]
- Murdaca, G.; Tonacci, A.; Negrini, S.; Greco, M.; Borro, M.; Puppo, F.; Gangemi, S. Emerging role of vitamin D in autoimmune diseases: An update on evidence and therapeutic implications. Autoimmun. Rev. 2019, 18, 102350. [Google Scholar] [CrossRef]
- Hejazi, M.E.; Modarresi-Ghazani, F.; Entezari-Maleki, T. A review of Vitamin D effects on common respiratory diseases: Asthma, chronic obstructive pulmonary disease, and tuberculosis. J. Res. Pharm. Pract. 2016, 5, 7–15. [Google Scholar]
- Rooney, M.R.; Harnack, L.; Michos, E.D.; Ogilvie, R.P.; Sempos, C.T.; Lutsey, P.L. Trends in use of high-dose vitamin D supplements exceeding 1000 or 4000 international units daily, 1999-2014. JAMA 2017, 317, 2448–2450. [Google Scholar] [CrossRef]
- Manousaki, D.; Paternoster, L.; Standl, M.; Moffatt, M.F.; Farrall, M.; Bouzigon, E.; Strachan, D.P.; Demenais, F.; Lathrop, M.; Cookson, W.O.C.M.; et al. Vitamin D levels and susceptibility to asthma, elevated immunoglobulin E levels, and atopic dermatitis: A Mendelian randomization study. PLoS Med. 2017, 14, e1002294. [Google Scholar] [CrossRef] [PubMed]
- Vimaleswaran, K.S.; Berry, D.J.; Lu, C.; Tikkanen, E.; Pilz, S.; Hiraki, L.T.; Cooper, J.D.; Dastani, Z.; Li, R.; Houston, D.K.; et al. Causal relationship between obesity and vitamin D status: Bi-directional mendelian randomization analysis of multiple cohorts. PLoS Med. 2013, 10, e1001383. [Google Scholar] [CrossRef] [PubMed]
- Shea, B.J.; Reeves, B.C.; Wells, G.; Thuku, M.; Hamel, C.; Moran, J.; Moher, D.; Tugwell, P.; Welch, V.; Kristjansson, E.; et al. AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017, 358, j4008. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Liu, D.; Liu, C.-T. Can vitamin D supplementation in addition to asthma controllers improve clinical outcomes in patients with asthma? A meta-analysis. Medicine (Baltimore) 2015, 94, e2185. [Google Scholar] [CrossRef] [PubMed]
- Global Asthma Network. The Global Asthma Report 2018. Available online: http://www.globalasthmareport.org/Global%20Asthma%20Report%202018.pdf (accessed on 26 June 2019).
- Mukherjee, S.; Lindell, D.M.; Berlin, A.A.; Morris, S.B.; Shanley, T.P.; Hershenson, M.B.; Lukacs, N.W. IL-17-induced pulmonary pathogenesis during respiratory viral infection and exacerbation of allergic disease. Am. J. Pathol. 2011, 179, 248–258. [Google Scholar] [CrossRef]
- Nanzer, A.M.; Chambers, E.S.; Ryanna, K.; Richards, D.F.; Black, C.; Timms, P.M.; Martineau, A.R.; Griffiths, C.J.; Corrigan, C.J.; Hawrylowicz, C.M. Enhanced production of IL-17A in patients with severe asthma is inhibited by 1α,25-dihydroxyvitamin D3 in a glucocorticoid-independent fashion. J. Allergy Clin. Immunol. 2013, 133, 297–304. [Google Scholar] [CrossRef]
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R. The global burden of asthma: Executive summary of the GINA Dissemination Committee report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef]
- Jackson, D.J.; Johnston, S.L. The role of viruses in acute exacerbations of asthma. J. Allergy Clin. Immunol. 2010, 125, 1178–1187. [Google Scholar] [CrossRef]
- Wang, H.; Naghavi, M.; Allen, C.; Barber, R.M.; Bhutta, Z.A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; Coates, M.M.; et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the Study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- Eder, W.; Ege, M.J.; von Mutius, E. The asthma epidemic. N. Engl. J. Med. 2006, 355, 2226–2235. [Google Scholar] [CrossRef]
- Björkstén, B.; Clayton, T.; Ellwood, P.; Stewart, A.; Strachan, D. Worldwide time trends for symptoms of rhinitis and conjunctivitis: Phase III of the International Study of Asthma and Allergies in Childhood. Pediatr. Allergy Immunol. 2008, 19, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Flohr, C. Recent perspectives on the global epidemiology of childhood eczema. Allergol. Immunopathol. 2011, 39, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Asthma’s Effect on the Nation. Available online: https://www.cdc.gov/asthma/asthmadata.htm (accessed on 26 June 2019).
- Global Initiative for Asthma (GINA). The Global Asthma Report; GINA: Paris, France, 2011. [Google Scholar]
- Zasloff, M. Fighting infections with vitamin D. Nat. Med. 2006, 12, 388–390. [Google Scholar] [CrossRef] [PubMed]
- Van Oeffelen, A.A.M.; Bekkers, M.B.M.; Smit, H.A.; Kerkhof, M.; Koppelman, G.H.; Haveman-Nies, A.; van der A, D.L.; Jansen, E.H.J.M.; Wijga, A.H. Serum micronutrient concentrations and childhood asthma: The PIAMA birth cohort study. Pediatr. Allergy Immunol. 2011, 22, 784–793. [Google Scholar] [CrossRef]
- Chinellato, I.; Piazza, M.; Sandri, M.; Paiola, G.; Tezza, G.; Boner, A.L. Correlation between vitamin D serum levels and passive smoking exposure in children with asthma. Allergy Asthma Proc. 2018, 39, 8–14. [Google Scholar] [CrossRef]
- Cantorna, M.T.; Mahon, B.D. D-hormone and the immune system. J. Rheumatol. Suppl. 2005, 76, 11–20. [Google Scholar]
- To, T.; Stanojevic, S.; Moores, G.; Gershon, A.S.; Bateman, E.D.; Cruz, A.A.; Boulet, L.-P. Global asthma prevalence in adults: Findings from the cross-sectional world health survey. BMC Public Health 2012, 12, 204. [Google Scholar] [CrossRef]
- Hewison, M. Vitamin D and immune function: Autocrine, paracrine or endocrine? Scand. J. Clin. Lab. Investig. Suppl. 2012, 243, 92–102. [Google Scholar]
- Hansdottir, S.; Monick, M.M.; Hinde, S.L.; Lovan, N.; Look, D.C.; Hunninghake, G.W. Respiratory epithelial cells convert inactive vitamin D to its active form: Potential effects on host defense. J. Immunol. 2008, 181, 7090–7099. [Google Scholar] [CrossRef]
- Brehm, J.M.; Schuemann, B.; Fuhlbrigge, A.L.; Hollis, B.W.; Strunk, R.C.; Zeiger, R.S.; Weiss, S.T.; Litonjua, A.A. Serum vitamin D levels and severe asthma exacerbations in the Childhood Asthma Management Program study. J. Allergy Clin. Immunol. 2010, 126, 52–58. [Google Scholar] [CrossRef]
- Hansdottir, S.; Monick, M.M. Vitamin D effects on lung immunity and respiratory diseases. Vitam. Horm. 2011, 86, 217–237. [Google Scholar] [PubMed]
- Hansdottir, S.; Monick, M.M.; Lovan, N.; Powers, L.; Gerke, A.; Hunninghake, G.W. Vitamin D decreases respiratory syncytial virus induction of NF-kappaB-linked chemokines and cytokines in airway epithelium while maintaining the antiviral state. J. Immunol. 2010, 184, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Litonjua, A.A.; Weiss, S.T. Is vitamin D deficiency to blame for the asthma epidemic? J. Allergy Clin. Immunol. 2007, 120, 1031–1035. [Google Scholar] [CrossRef]
- Adams, J.S.; Hewison, M. Unexpected actions of vitamin D: New perspectives on the regulation of innate and adaptive immunity. Nat. Clin. Pract. Endocrinol. Metab. 2008, 4, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A.J.; Ingham, T.; Wickens, K.; Thadhani, R.; Silvers, K.M.; Epton, M.J.; Town, G.I.; Pattemore, P.K.; Espinola, J.A.; Crane, J. Cord-blood 25-hydroxyvitamin D levels and risk of respiratory infection, wheezing, and asthma. Pediatrics 2011, 127, e180–e187. [Google Scholar] [CrossRef]
- Brown, S.D.; Calvert, H.H.; Fitzpatrick, A.M. Vitamin D and asthma. Dermatoendocrinology 2012, 4, 137–145. [Google Scholar] [CrossRef]
- Finklea, J.D.; Grossmann, R.E.; Tangpricha, V. Vitamin D and chronic lung disease: A review of molecular mechanisms and clinical studies. Adv. Nutr. 2011, 2, 244–253. [Google Scholar] [CrossRef]
- Herr, C.; Greulich, T.; Koczulla, R.A.; Meyer, S.; Zakharkina, T.; Branscheidt, M.; Eschmann, R.; Bals, R. The role of vitamin D in pulmonary disease: COPD, asthma, infection, and cancer. Respir. Res. 2011, 12, 31. [Google Scholar] [CrossRef]
- Paul, G.; Brehm, J.M.; Alcorn, J.F.; Holguín, F.; Aujla, S.J.; Celedón, J.C. Vitamin D and asthma. Am. J. Respir. Crit. Care Med. 2012, 185, 124–132. [Google Scholar] [CrossRef]
- Rajabbik, M.H.; Lotfi, T.; Alkhaled, L.; Fares, M.; El-Hajj Fuleihan, G.; Mroueh, S.; Akl, E.A. Association between low vitamin D levels and the diagnosis of asthma in children: A systematic review of cohort studies. Allergy Asthma Clin. Immunol. 2014, 10, 31. [Google Scholar] [CrossRef]
- Cassim, R.; Russell, M.A.; Lodge, C.J.; Lowe, A.J.; Koplin, J.J.; Dharmage, S.C. The role of circulating 25 hydroxyvitamin D in asthma: A systematic review. Allergy 2015, 70, 339–354. [Google Scholar] [CrossRef] [PubMed]
- Man, L.; Zhang, Z.; Zhang, M.; Zhang, Y.; Li, J.; Zheng, N.; Cao, Y.; Chi, M.; Chao, Y.; Huang, Q.; et al. Association between vitamin D deficiency and insufficiency and the risk of childhood asthma: Evidence from a meta-analysis. Int. J. Clin. Exp. Med. 2015, 8, 5699–5706. [Google Scholar] [PubMed]
- Wei, Z.; Zhang, J.; Yu, X. Maternal vitamin D status and childhood asthma, wheeze, and eczema: A systematic review and meta-analysis. Pediatr. Allergy Immunol. 2016, 27, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Xun, P.; Pike, K.; Wills, A.K.; Chawes, B.L.; Bisgaard, H.; Cai, W.; Wan, Y.; He, K. In utero exposure to 25-hydroxyvitamin D and risk of childhood asthma, wheeze, and respiratory tract infections: A meta-analysis of birth cohort studies. J. Allergy Clin. Immunol. 2017, 139, 1508–1517. [Google Scholar] [CrossRef]
- Jat, K.R.; Khairwa, A. Vitamin D and asthma in children: A systematic review and meta-analysis of observational studies. Lung India 2017, 34, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Yang, L.; Jia, C. Maternal vitamin D status during pregnancy and risk of childhood asthma: A meta-analysis of prospective studies. Mol. Nutr. Food Res. 2017, 61, 1600657. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-González, R.M.; García-Marcos, L.; Morales, E. Prenatal vitamin D status and respiratory and allergic outcomes in childhood: A meta-analysis of observational studies. Pediatr. Allergy Immunol. 2018, 29, 243–253. [Google Scholar] [CrossRef]
- Shen, S.-Y.; Xiao, W.-Q.; Lu, J.-H.; Yuan, M.-Y.; He, J.-R.; Xia, H.-M.; Qiu, X.; Cheng, K.K.; Lam, K.B.H. Early life vitamin D status and asthma and wheeze: A systematic review and meta-analysis. BMC Pulm. Med. 2018, 18, 120. [Google Scholar] [CrossRef]
- Harvey, N.C.; Holroyd, C.; Ntani, G.; Javaid, K.; Cooper, P.; Moon, R.; Cole, Z.; Tinati, T.; Godfrey, K.; Dennison, E.; et al. Vitamin D supplementation in pregnancy: A systematic review. Health Technol. Assess. 2014, 18, 1–190. [Google Scholar] [CrossRef]
- Xiao, L.; Xing, C.; Yang, Z.; Xu, S.; Wang, M.; Du, H.; Liu, K.; Huang, Z. Vitamin D supplementation for the prevention of childhood acute respiratory infections: A systematic review of randomised controlled trials. Br. J. Nutr. 2015, 114, 1026–1034. [Google Scholar] [CrossRef]
- Pojsupap, S.; Iliriani, K.; Sampaio, T.Z.A.L.; O’Hearn, K.; Kovesi, T.; Menon, K.; McNally, J.D. Efficacy of high-dose vitamin D in pediatric asthma: A systematic review and meta-analysis. J. Asthma 2015, 52, 382–390. [Google Scholar] [CrossRef]
- Riverin, B.D.; Maguire, J.L.; Li, P. Vitamin D supplementation for childhood asthma: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0136841. [Google Scholar] [CrossRef]
- Fares, M.M.; Alkhaled, L.H.; Mroueh, S.M.; Akl, E.A. Vitamin D supplementation in children with asthma: A systematic review and meta-analysis. BMC Res. Notes 2015, 8, 23. [Google Scholar] [CrossRef]
- Vahdaninia, M.; Mackenzie, H.; Helps, S.; Dean, T. Prenatal intake of vitamins and allergic outcomes in the offspring: A systematic review and meta-analysis. J. Allergy Clin. Immunol. Pract. 2017, 5, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Cates, C.J.; Urashima, M.; Jensen, M.; Griffiths, A.P.; Nurmatov, U.; Sheikh, A.; Griffiths, C.J. Vitamin D for the management of asthma. Cochrane Database Syst. Rev. 2016, 9, CD011511. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, D.A.; Greenberg, L.; Hooper, R.L.; Griffiths, C.J.; Camargo, C.A.; Kerley, C.P.; Jensen, M.E.; Mauger, D.; Stelmach, I.; Urashima, M.; et al. Vitamin D supplementation to prevent asthma exacerbations: A systematic review and meta-analysis of individual participant data. Lancet Respir. Med. 2017, 5, 881–890. [Google Scholar] [CrossRef]
- Davies, N.M.; Holmes, M.V.; Davey Smith, G. Reading Mendelian randomization studies: A guide, glossary, and checklist for clinicians. BMJ 2018, 362, k601. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J.; Celli, B.R. Systemic manifestations and comorbidities of COPD. Eur. Respir. J. 2009, 33, 1165–1185. [Google Scholar] [CrossRef]
- Wedzicha, J.A.; Seemungal, T.A.R. COPD exacerbations: Defining their cause and prevention. Lancet 2007, 370, 786–796. [Google Scholar] [CrossRef]
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management and Prevention of COPD. Available online: https://goldcopd.org/gold-2017-global-strategy-diagnosis-management-prevention-copd/ (accessed on 27 March 2019).
- GBD 2015 Chronic Respiratory Disease Collaborators. Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990–2015. Lancet Respir. Med. 2017, 5, 691–706. [Google Scholar] [CrossRef]
- Lopez, A.D.; Shibuya, K.; Rao, C.; Mathers, C.D.; Hansell, A.L.; Held, L.S.; Schmid, V.; Buist, S. Chronic obstructive pulmonary disease: Current burden and future projections. Eur. Respir. J. 2006, 27, 397–412. [Google Scholar] [CrossRef] [PubMed]
- Faner, R.; Gonzalez, N.; Cruz, T.; Kalko, S.G.; Agustí, A. Systemic inflammatory response to smoking in chronic obstructive pulmonary disease: Evidence of a gender effect. PLoS ONE 2014, 9, e97491. [Google Scholar] [CrossRef] [PubMed]
- Smolonska, J.; Wijmenga, C.; Postma, D.S.; Boezen, H.M. Meta-analyses on suspected chronic obstructive pulmonary disease genes: A summary of 20 years’ research. Am. J. Respir. Crit. Care Med. 2009, 180, 618–631. [Google Scholar] [CrossRef] [PubMed]
- Kokturk, N.; Baha, A.; Oh, Y.-M.; Young Ju, J.; Jones, P.W. Vitamin D deficiency: What does it mean for chronic obstructive pulmonary disease (COPD)? a compherensive review for pulmonologists. Clin. Respir. J. 2018, 12, 382–397. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Tenderich, G.; Koerfer, R. Vitamin D and the adaptive immune system with special emphasis to allergic reactions and allograft rejection. Inflamm. Allergy Drug Targets 2009, 8, 161–168. [Google Scholar] [CrossRef]
- Hewison, M. Vitamin D and the immune system: New perspectives on an old theme. Endocrinol. Metab. Clin. North Am. 2010, 39, 365–379. [Google Scholar] [CrossRef]
- Szymczak, I.; Pawliczak, R. The active metabolite of vitamin D3 as a potential immunomodulator. Scand. J. Immunol. 2016, 83, 83–91. [Google Scholar] [CrossRef]
- Black, P.N.; Scragg, R. Relationship between serum 25-hydroxyvitamin d and pulmonary function in the third national health and nutrition examination survey. Chest 2005, 128, 3792–3798. [Google Scholar] [CrossRef]
- Choi, C.-J.; Seo, M.; Choi, W.-S.; Kim, K.-S.; Youn, S.-A.; Lindsey, T.; Choi, Y.-J.; Kim, C.-M. Relationship between serum 25-hydroxyvitamin D and lung function among Korean adults in Korea National Health and Nutrition Examination Survey (KNHANES), 2008-2010. J. Clin. Endocrinol. Metab. 2013, 98, 1703–1710. [Google Scholar] [CrossRef]
- Manavi, K.R.; Alston-Mills, B.P.; Thompson, M.P. History of tobacco, vitamin D and women. Int J Vitam. Nutr. Res. 2020. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematicS review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Zhu, B.; Zhu, B.; Xiao, C.; Zheng, Z. Vitamin D deficiency is associated with the severity of COPD: A systematic review and meta-analysis. Int. J. Chron. Obstruct. Pulmon. Dis. 2015, 10, 1907–1916. [Google Scholar] [PubMed]
- Zhu, M.; Wang, T.; Wang, C.; Ji, Y. The association between vitamin D and COPD risk, severity, and exacerbation: An updated systematic review and meta-analysis. Int. J. Chron. Obstruct. Pulmon. Dis. 2016, 11, 2597–2607. [Google Scholar] [CrossRef]
- Ferrari, R.; Caram, L.M.O.; Tanni, S.E.; Godoy, I.; Rupp de Paiva, S.A. The relationship between Vitamin D status and exacerbation in COPD patients- a literature review. Respir. Med. 2018, 139, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Jolliffe, D.A.; Greenberg, L.; Hooper, R.L.; Mathyssen, C.; Rafiq, R.; de Jongh, R.T.; Camargo, C.A.; Griffiths, C.J.; Janssens, W.; Martineau, A.R. Vitamin D to prevent exacerbations of COPD: Systematic review and meta-analysis of individual participant data from randomised controlled trials. Thorax 2019, 74, 337–345. [Google Scholar] [CrossRef]
- Bjerk, S.M.; Edgington, B.D.; Rector, T.S.; Kunisaki, K.M. Supplemental vitamin D and physical performance in COPD: A pilot randomized trial. Int. J. Chron. Obstruct. Pulmon. Dis. 2013, 8, 97–104. [Google Scholar]
- Zendedel, A.; Gholami, M.; Anbari, K.; Ghanadi, K.; Bachari, E.C.; Azargon, A. Effects of vitamin D intake on FEV1 and COPD exacerbation: A randomized clinical trial study. Glob. J. Health Sci. 2015, 7, 243–248. [Google Scholar] [CrossRef]
- Rafiq, R.; Prins, H.J.; Boersma, W.G.; Daniels, J.M.; den Heijer, M.; Lips, P.; de Jongh, R.T. Effects of daily vitamin D supplementation on respiratory muscle strength and physical performance in vitamin D-deficient COPD patients: A pilot trial. Int. J. Chron. Obstruct. Pulmon. Dis. 2017, 12, 2583–2592. [Google Scholar] [CrossRef]
- Lehouck, A.; Mathieu, C.; Carremans, C.; Baeke, F.; Verhaegen, J.; van Eldere, J.; Decallonne, B.; Bouillon, R.; Decramer, M.; Janssens, W. High doses of vitamin D to reduce exacerbations in chronic obstructive pulmonary disease: A randomized trial. Ann. Intern. Med. 2012, 156, 105–114. [Google Scholar] [CrossRef]
- Martineau, A.R.; James, W.Y.; Hooper, R.L.; Barnes, N.C.; Jolliffe, D.A.; Greiller, C.L.; Islam, K.; McLaughlin, D.; Bhowmik, A.; Timms, P.M.; et al. Vitamin D3 supplementation in patients with chronic obstructive pulmonary disease (ViDiCO): A multicentre, double-blind, randomised controlled trial. Lancet Respir. Med. 2015, 3, 120–130. [Google Scholar] [CrossRef]
- Keaney, J.F.; Rosen, C.J. VITAL signs for dietary supplementation to prevent cancer and heart disease. N. Engl. J. Med. 2019, 380, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Ernst, J.B.; Gummert, J.F.; Börgermann, J. Vitamin D supplementation, body weight and human serum 25-hydroxyvitamin D response: A systematic review. Eur. J. Nutr. 2014, 53, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Forum of International Respiratory Societies. The Global Impact of Respiratory Disease, 2nd ed.; European Respiratory Society: Sheffield, UK, 2017. [Google Scholar]
- GBD 2015 LRI Collaborators. Estimates of the global, regional, and national morbidity, mortality, and etiologies of lower respiratory tract infections in 195 countries: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 2017, 17, 1133–1161. [Google Scholar] [CrossRef]
- Cilloniz, C.; Martin-Loeches, I.; Garcia-Vidal, C.; San Jose, A.; Torres, A. Microbial etiology of pneumonia: Epidemiology, diagnosis and resistance patterns. Int. J. Mol. Sci. 2016, 17, 2120. [Google Scholar] [CrossRef]
- Ochoa-Gondar, O.; Vila-Córcoles, A.; de Diego, C.; Arija, V.; Maxenchs, M.; Grive, M.; Martin, E.; Pinyol, J.L. The burden of community-acquired pneumonia in the elderly: The Spanish EVAN-65 study. BMC Public Health 2008, 8, 222. [Google Scholar] [CrossRef]
- Vila-Corcoles, A.; Ochoa-Gondar, O.; Rodriguez-Blanco, T.; Raga-Luria, X.; Gomez-Bertomeu, F. Epidemiology of community-acquired pneumonia in older adults: A population-based study. Respir. Med. 2009, 103, 309–316. [Google Scholar] [CrossRef]
- Ewig, S.; Birkner, N.; Strauss, R.; Schaefer, E.; Pauletzki, J.; Bischoff, H.; Schraeder, P.; Welte, T.; Hoeffken, G. New perspectives on community-acquired pneumonia in 388 406 patients. Results from a nationwide mandatory performance measurement programme in healthcare quality. Thorax 2009, 64, 1062–1069. [Google Scholar] [CrossRef]
- Scott, J.A.G.; Wonodi, C.; Moïsi, J.C.; Deloria-Knoll, M.; DeLuca, A.N.; Karron, R.A.; Bhat, N.; Murdoch, D.R.; Crawley, J.; Levine, O.S.; et al. The definition of pneumonia, the assessment of severity, and clinical standardization in the Pneumonia Etiology Research for Child Health study. Clin. Infect. Dis. 2012, 54, S109–S116. [Google Scholar] [CrossRef]
- Harris, A.M.; Hicks, L.A.; Qaseem, A. Appropriate antibiotic use for acute respiratory tract infection in adults: Advice for high-value care from the American College of Physicians and the Centers for Disease Control and Prevention. Ann. Intern. Med. 2016, 164, 425–434. [Google Scholar] [CrossRef]
- Baeke, F.; Takiishi, T.; Korf, H.; Gysemans, C.; Mathieu, C. Vitamin D: Modulator of the immune system. Curr. Opin. Pharmacol. 2010, 10, 482–496. [Google Scholar] [CrossRef] [PubMed]
- Tamblyn, J.A.; Hewison, M.; Wagner, C.L.; Bulmer, J.N.; Kilby, M.D. Immunological role of vitamin D at the maternal-fetal interface. J. Endocrinol. 2015, 224, R107–R121. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef] [PubMed]
- Wayse, V.; Yousafzai, A.; Mogale, K.; Filteau, S. Association of subclinical vitamin D deficiency with severe acute lower respiratory infection in Indian children under 5 y. Eur. J. Clin. Nutr. 2004, 58, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Najada, A.S.; Habashneh, M.S.; Khader, M. The frequency of nutritional rickets among hospitalized infants and its relation to respiratory diseases. J. Trop. Pediatr. 2004, 50, 364–368. [Google Scholar] [CrossRef]
- Williams, B.; Williams, A.J.; Anderson, S.T. Vitamin D deficiency and insufficiency in children with tuberculosis. Pediatr. Infect. Dis. J. 2008, 27, 941–942. [Google Scholar] [CrossRef]
- Jolliffe, D.A.; Griffiths, C.J.; Martineau, A.R. Vitamin D in the prevention of acute respiratory infection: Systematic review of clinical studies. J. Steroid. Biochem. Mol. Biol. 2013, 136, 321–329. [Google Scholar] [CrossRef]
- Das, R.R.; Singh, M.; Panigrahi, I.; Naik, S.S. Vitamin D supplementation for the treatment of acute childhood pneumonia: A systematic review. ISRN Pediatr 2013, 19, 459160. [Google Scholar] [CrossRef]
- Fried, D.A.; Rhyu, J.; Odato, K.; Blunt, H.; Karagas, M.R.; Gilbert-Diamond, D. Maternal and cord blood vitamin D status and childhood infection and allergic disease: A systematic review. Nutr. Rev. 2016, 74, 387–410. [Google Scholar] [CrossRef]
- Das, R.R.; Singh, M.; Naik, S.S. Vitamin D as an adjunct to antibiotics for the treatment of acute childhood pneumonia. Cochrane Database Syst. Rev. 2018, 7, CD011597. [Google Scholar] [CrossRef]
- Charan, J.; Goyal, J.P.; Saxena, D.; Yadav, P. Vitamin D for prevention of respiratory tract infections: A systematic review and meta-analysis. J. Pharmacol. Pharmacother. 2012, 3, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.; Lindh, A.U.; Björkhem-Bergman, L.; Lindh, J.D. Vitamin D and respiratory tract infections: A systematic review and meta-analysis of randomized controlled trials. PLoS ONE 2013, 8, e65835. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Huang, S. Vitamin D supplementation and risk of respiratory tract infections: A meta-analysis of randomized controlled trials. Scand. J. Infect. Dis. 2013, 45, 696–702. [Google Scholar] [CrossRef]
- Vuichard Gysin, D.; Dao, D.; Gysin, C.M.; Lytvyn, L.; Loeb, M. Effect of vitamin D3 supplementation on respiratory tract infections in healthy individuals: A systematic review and meta-analysis of randomized controlled trials. PLoS ONE 2016, 11, e0162996. [Google Scholar] [CrossRef]
- Yakoob, M.Y.; Salam, R.A.; Khan, F.R.; Bhutta, Z.A. Vitamin D supplementation for preventing infections in children under five years of age. Cochrane Database Syst. Rev. 2016, 11, CD008824. [Google Scholar] [CrossRef] [PubMed]
- Martineau, A.R.; Jolliffe, D.A.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; Goodall, E.C.; et al. Vitamin D supplementation to prevent acute respiratory infections: Individual participant data meta-analysis. Health Technol. Assess 2019, 23, 1–44. [Google Scholar] [CrossRef]
- Camargo, C.A.; Sluyter, J.; Stewart, A.W.; Khaw, K.-T.; Lawes, C.M.M.; Toop, L.; Waayer, D.; Scragg, R. Effect of monthly high-dose vitamin D supplementation on acute respiratory infections in older adults: A randomized controlled trial. Clin Infect Dis 2019. [Google Scholar] [CrossRef]
- Feldman, H.H.; Jacova, C.; Robillard, A.; Garcia, A.; Chow, T.; Borrie, M.; Schipper, H.M.; Blair, M.; Kertesz, A.; Chertkow, H. Diagnosis and treatment of dementia: 2. Diagnosis. CMAJ 2008, 178, 825–836. [Google Scholar] [CrossRef]
- WHO. Facts in Pictures: Dementia. Available online: http://www.who.int/news-room/facts-in-pictures/detail/dementia (accessed on 6 January 2020).
- Deutsche Alzheimer Gesellschaft e.V. Informationsblatt 1. Die Häufigkeit von Demenzerkrankungen. Available online: https://www.deutsche-alzheimer.de/fileadmin/alz/pdf/factsheets/infoblatt1_haeufigkeit_demenzerkrankungen_dalzg.pdf (accessed on 6 January 2020).
- Annweiler, C.; Llewellyn, D.J.; Beauchet, O. Low serum vitamin D concentrations in Alzheimer’s disease: A systematic review and meta-analysis. J. Alzheimers. Dis. 2013, 33, 659–674. [Google Scholar] [CrossRef]
- Aspell, N.; Lawlor, B.; O’Sullivan, M. Is there a role for vitamin D in supporting cognitive function as we age? Proc. Nutr. Soc. 2018, 77, 124–134. [Google Scholar] [CrossRef]
- Buell, J.S.; Dawson-Hughes, B. Vitamin D and neurocognitive dysfunction: Preventing “D”ecline? Mol. Aspects Med. 2008, 29, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Kesby, J.P.; Eyles, D.W.; Burne, T.H.J.; McGrath, J.J. The effects of vitamin D on brain development and adult brain function. Mol Cell Endocrinol. 2011, 347, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Mozos, I.; Stoian, D.; Luca, C.T. Crosstalk between Vitamins A, B12, D, K, C, and E status and arterial stiffness. Dis. Markers 2017, 2017, 8784971. [Google Scholar] [CrossRef] [PubMed]
- Etgen, T.; Sander, D.; Huntgeburth, U.; Poppert, H.; Forstl, H.; Bickel, H. Physical activity and incident cognitive impairment in elderly persons: The INVADE study. Arch. Intern. Med. 2010, 170, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Balion, C.; Griffith, L.E.; Strifler, L.; Henderson, M.; Patterson, C.; Heckman, G.; Llewellyn, D.J.; Raina, P. Vitamin D, cognition, and dementia: A systematic review and meta-analysis. Neurology 2012, 79, 1397–1405. [Google Scholar] [CrossRef]
- Annweiler, C.; Montero-Odasso, M.; Llewellyn, D.J.; Richard-Devantoy, S.; Duque, G.; Beauchet, O. Meta-analysis of memory and executive dysfunctions in relation to vitamin D. J. Alzheimers. Dis. 2013, 37, 147–171. [Google Scholar] [CrossRef]
- Shen, L.; Ji, H.-F. Vitamin D deficiency is associated with increased risk of Alzheimer’s disease and dementia: Evidence from meta-analysis. Nutr. J. 2015, 14, 76. [Google Scholar] [CrossRef]
- Cao, L.; Tan, L.; Wang, H.-F.; Jiang, T.; Zhu, X.-C.; Lu, H.; Tan, M.-S.; Yu, J.-T. Dietary patterns and risk of dementia: A systematic review and meta-analysis of cohort studies. Mol. Neurobiol. 2016, 53, 6144–6154. [Google Scholar] [CrossRef]
- Sommer, I.; Griebler, U.; Kien, C.; Auer, S.; Klerings, I.; Hammer, R.; Holzer, P.; Gartlehner, G. Vitamin D deficiency as a risk factor for dementia: A systematic review and meta-analysis. BMC Geriatr. 2017, 17, 16. [Google Scholar] [CrossRef] [PubMed]
- Goodwill, A.M.; Szoeke, C. A systematic review and meta-analysis of the effect of low vitamin D on cognition. J. Am. Geriatr. Soc. 2017, 65, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- Jayedi, A.; Rashidy-Pour, A.; Shab-Bidar, S. Vitamin D status and risk of dementia and Alzheimer’s disease: A meta-analysis of dose-response. Nutr. Neurosci. 2018, 15, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xue, W.; Li, J.; Fu, K.; Shi, H.; Zhang, B.; Teng, W.; Tian, L. 25-hydroxyvitamin D levels and the risk of dementia and Alzheimer’s disease: A dose-response meta-analysis. Front. Aging Neurosci. 2018, 10, 368. [Google Scholar] [CrossRef]
- Van der Schaft, J.; Koek, H.L.; Dijkstra, E.; Verhaar, H.J.J.; van der Schouw, Y.T.; Emmelot-Vonk, M.H. The association between vitamin D and cognition: A systematic review. Aging Res. Rev. 2013, 12, 1013–1023. [Google Scholar] [CrossRef] [PubMed]
- Killin, L.O.J.; Starr, J.M.; Shiue, I.J.; Russ, T.C. Environmental risk factors for dementia: A systematic review. BMC Geriatr. 2016, 16, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Lerner, P.P.; Sharony, L.; Miodownik, C. Association between mental disorders, cognitive disturbances and vitamin D serum level: Current state. Clin. Nutr. ESPEN 2018, 23, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Alfred, T.; Ben-Shlomo, Y.; Cooper, R.; Hardy, R.; Deary, I.J.; Elliott, J.; Harris, S.E.; Hyppönen, E.; Kivimaki, M.; Kumari, M.; et al. Genetic variants influencing biomarkers of nutrition are not associated with cognitive capability in middle-aged and older adults. J. Nutr. 2013, 143, 606–612. [Google Scholar]
- Mokry, L.E.; Ross, S.; Morris, J.A.; Manousaki, D.; Forgetta, V.; Richards, J.B. Genetically decreased vitamin D and risk of Alzheimer disease. Neurology 2016, 87, 2567–2574. [Google Scholar] [CrossRef]
- Maddock, J.; Zhou, A.; Cavadino, A.; Kuźma, E.; Bao, Y.; Smart, M.C.; Saum, K.-U.; Schöttker, B.; Engmann, J.; Kjærgaard, M.; et al. Vitamin D and cognitive function: A Medelian randomization study. Sci. Rep. 2017, 7, 13230. [Google Scholar] [CrossRef]
- Kuźma, E.; Hannon, E.; Zhou, A.; Lourida, I.; Bethel, A.; Levine, D.A.; Lunnon, K.; Thompson-Coon, J.; Hyppönen, E.; Llewellyn, D.J. Which risk factors causally influence dementia? A systematic review of mendelian randomization studies. J. Alzheimers. Dis. 2018, 64, 181–193. [Google Scholar] [CrossRef]
- Rossom, R.C.; Espeland, M.A.; Manson, J.E.; Dysken, M.W.; Johnson, K.C.; Lane, D.S.; Leblanc, E.S.; Lederle, F.A.; Masaki, K.H.; Margolis, K.L. Calcium and vitamin D supplementation and cognitive impairment in the women’s health initiative. J. Am. Geriatr. Soc. 2012, 60, 2197–2205. [Google Scholar] [CrossRef]
- Schietzel, S.; Fischer, K.; Brugger, P.; Orav, E.J.; Renerts, K.; Gagesch, M.; Freystaetter, G.; Stähelin, H.B.; Egli, A.; Bischoff-Ferrari, H.A. Effect of 2000 IU compared with 800 IU vitamin D on cognitive performance among adults age 60 years and older: A randomized controlled trial. Am. J. Clin. Nutr. 2019, 110, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Gugger, A.; Marzel, A.; Orav, E.J.; Willett, W.C.; Dawson-Hughes, B.; Theiler, R.; Freystätter, G.; Egli, A.; Bischoff-Ferrari, H.A. Effect of monthly high-dose vitamin D on mental health in older adults: Secondary analysis of a RCT. J. Am. Geriatr. Soc. 2019, 67, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organization). WHO Fact Sheet on Depression. Available online: https://www.who.int/news-room/fact-sheets/detail/depression (accessed on 10 June 2019).
- Gezen-Ak, D.; Yılmazer, S.; Dursun, E. Why vitamin D in Alzheimer’s disease? The hypothesis. J. Alzheimers. Dis. 2014, 40, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Landel, V.; Annweiler, C.; Millet, P.; Morello, M.; Féron, F. Vitamin D, cognition and Alzheimer’s disease: The therapeutic benefit is in the D-Tails. J. Alzheimers. Dis. 2016, 53, 419–444. [Google Scholar] [CrossRef] [PubMed]
- McCann, J.C.; Ames, B.N. Is there convincing biological or behavioral evidence linking vitamin D deficiency to brain dysfunction? FASEB J. 2008, 22, 982–1001. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Pelekanos, M.; Liu, P.-Y.; Burne, T.H.J.; McGrath, J.J.; Eyles, D.W. The vitamin D receptor in dopamine neurons; its presence in human substantia nigra and its ontogenesis in rat midbrain. Neuroscience 2013, 236, 77–87. [Google Scholar] [CrossRef]
- Eyles, D.W.; Liu, P.Y.; Josh, P.; Cui, X. Intracellular distribution of the vitamin D receptor in the brain: Comparison with classic target tissues and redistribution with development. Neuroscience 2014, 268, 1–9. [Google Scholar] [CrossRef]
- Brouwer-Brolsma, E.M.; de Groot, L.C.P.G.M. Vitamin D and cognition in older adults: An update of recent findings. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 11–16. [Google Scholar] [CrossRef]
- Obradovic, D.; Gronemeyer, H.; Lutz, B.; Rein, T. Cross-talk of vitamin D and glucocorticoids in hippocampal cells. J. Neurochem. 2006, 96, 500–509. [Google Scholar] [CrossRef]
- Anglin, R.E.S.; Samaan, Z.; Walter, S.D.; McDonald, S.D. Vitamin D deficiency and depression in adults: Systematic review and meta-analysis. Br. J. Psychiatry 2013, 202, 100–107. [Google Scholar] [CrossRef]
- Ju, S.-Y.; Lee, Y.-J.; Jeong, S.-N. Serum 25-hydroxyvitamin D levels and the risk of depression: A systematic review and meta-analysis. J. Nutr. Health Aging 2013, 17, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J.; Murphy, J.; Mischoulon, D.; Papakostas, G.I.; Fava, M.; Berk, M.; Ng, C.H. Adjunctive nutraceuticals for depression: A systematic review and meta-analyses. Am. J. Psychiatry 2016, 173, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Föcker, M.; Antel, J.; Ring, S.; Hahn, D.; Kanal, Ö.; Öztürk, D.; Hebebrand, J.; Libuda, L. Vitamin D and mental health in children and adolescents. Eur. Child Adolesc. Psychiatry 2017, 26, 1043–1066. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Mbuagbaw, L.; Samaan, Z.; Falavigna, M.; Zhang, S.; Adachi, J.D.; Cheng, J.; Papaioannou, A.; Thabane, L. Efficacy of vitamin D supplementation in depression in adults: A systematic review. J. Clin. Endocrinol. Metab. 2014, 99, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, J.A.; Edmondson, D.; Wasson, L.T.; Falzon, L.; Homma, K.; Ezeokoli, N.; Li, P.; Davidson, K.W. Vitamin D supplementation for depressive symptoms: A systematic review and meta-analysis of randomized controlled trials. Psychosom. Med. 2014, 76, 190–196. [Google Scholar] [CrossRef]
- Spedding, S. Vitamin D and depression: A systematic review and meta-analysis comparing studies with and without biological flaws. Nutrients 2014, 6, 1501–1518. [Google Scholar] [CrossRef]
- Gowda, U.; Mutowo, M.P.; Smith, B.J.; Wluka, A.E.; Renzaho, A.M.N. Vitamin D supplementation to reduce depression in adults: Meta-analysis of randomized controlled trials. Nutrition 2015, 31, 421–429. [Google Scholar] [CrossRef]
- Vellekkatt, F.; Menon, V. Efficacy of vitamin D supplementation in major depression: A meta-analysis of randomized controlled trials. J. Postgrad. Med. 2018, 65, 74. [Google Scholar]
- Sparling, T.M.; Nesbitt, R.C.; Henschke, N.; Gabrysch, S. Nutrients and perinatal depression: A systematic review. J. Nutr. Sci. 2017, 6, e61. [Google Scholar] [CrossRef]
- Aghajafari, F.; Letourneau, N.; Mahinpey, N.; Cosic, N.; Giesbrecht, G. Vitamin D deficiency and antenatal and postpartum depression: A systematic review. Nutrients 2018, 10, 478. [Google Scholar] [CrossRef]
- Amini, S.; Jafarirad, S.; Amani, R. Postpartum depression and vitamin D: A systematic review. Crit. Rev. Food Sci. Nutr. 2018, 59, 1514–1520. [Google Scholar] [CrossRef] [PubMed]
- Trujillo, J.; Vieira, M.C.; Lepsch, J.; Rebelo, F.; Poston, L.; Pasupathy, D.; Kac, G. A systematic review of the associations between maternal nutritional biomarkers and depression and/or anxiety during pregnancy and postpartum. J. Affect. Disord. 2018, 232, 185–203. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, N.; Sun, W.; Chen, D.; Zhao, J.; Zhang, W. Association between vitamin D deficiency and antepartum and postpartum depression: A systematic review and meta-analysis of longitudinal studies. Arch. Gynecol. Obstet. 2018, 298, 1045–1059. [Google Scholar] [CrossRef] [PubMed]
- Michaëlsson, K.; Melhus, H.; Larsson, S.C. Serum 25-hydroxyvitamin D concentration and major depression: A Mendelian randomization study. Nutrients 2018, 10, 1987. [Google Scholar] [CrossRef]
- Jiang, X.; O’Reilly, P.F.; Aschard, H.; Hsu, Y.-H.; Richards, J.B.; Dupuis, J.; Ingelsson, E.; Karasik, D.; Pilz, S.; Berry, D.; et al. Genome-wide association study in 79,366 European-ancestry individuals informs the genetic architecture of 25-hydroxyvitamin D levels. Nat. Commun. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Li, H.; Sun, D.; Wang, A.; Pan, H.; Feng, W.; Ng, C.H.; Ungvari, G.S.; Tao, L.; Li, X.; Wang, W.; et al. Serum 25-hydroxyvitamin D levels and depression in older adults: A dose-response meta-analysis of prospective cohort studies. Am. J. Geriatr. Psychiatry 2019, 27, 1192–1202. [Google Scholar] [CrossRef]
- Libuda, L.; Laabs, B.-H.; Ludwig, C.; Bühlmeier, J.; Antel, J.; Hinney, A.; Naaresh, R.; Föcker, M.; Hebebrand, J.; König, I.R.; et al. Vitamin D and the risk of depression: A causal relationship? Findings from a Mendelian randomization study. Nutrients 2019, 11, 1085. [Google Scholar] [CrossRef]
- Meng, X.; Li, X.; Timofeeva, M.N.; He, Y.; Spiliopoulou, A.; Wei, W.-Q.; Gifford, A.; Wu, H.; Varley, T.; Joshi, P.; et al. Phenome-wide Mendelian-randomization study of genetically determined vitamin D on multiple health outcomes using the UK Biobank study. Int. J. Epidemiol. 2019, 48, 1425–1434. [Google Scholar] [CrossRef]
- Alavi, N.M.; Khademalhoseini, S.; Vakili, Z.; Assarian, F. Effect of vitamin D supplementation on depression in elderly patients: A randomized clinical trial. Clin. Nutr. 2019, 38, 2065–2070. [Google Scholar] [CrossRef]
- Vaughn, C.B.; Jakimovski, D.; Kavak, K.S.; Ramanathan, M.; Benedict, R.H.B.; Zivadinov, R.; Weinstock-Guttman, B. Epidemiology and treatment of multiple sclerosis in elderly populations. Nat. Rev. Neurol. 2019, 15, 329–342. [Google Scholar] [CrossRef]
- Kister, I.; Bacon, T.E.; Chamot, E.; Salter, A.R.; Cutter, G.R.; Kalina, J.T.; Herbert, J. Natural history of multiple sclerosis symptoms. Int. J. MS Care 2013, 15, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: A expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef] [PubMed]
- Kaunzner, U.W.; Gauthier, S.A. MRI in the assessment and monitoring of multiple sclerosis: An update on best practice. Ther. Adv. Neurol. Disord. 2017, 10, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Milo, R.; Kahana, E. Multiple sclerosis: Geoepidemiology, genetics and the environment. Autoimmun. Rev. 2010, 9, A387–A394. [Google Scholar] [CrossRef]
- Thompson, A.J.; Baneke, P. Atlas of MS. Mapping Multiple Sclerosis around the World. Available online: https://www.msif.org/wp-content/uploads/2014/09/Atlas-of-MS.pdf (accessed on 25 August 2019).
- Scalfari, A.; Knappertz, V.; Cutter, G.; Goodin, D.S.; Ashton, R.; Ebers, G.C. Mortality in patients with multiple sclerosis. Neurology 2013, 81, 184–192. [Google Scholar] [CrossRef]
- Olsson, T.; Barcellos, L.F.; Alfredsson, L. Interactions between genetic, lifestyle and environmental risk factors for multiple sclerosis. Nat. Rev. Neurol. 2017, 13, 25–36. [Google Scholar] [CrossRef]
- Simpson, S.; Blizzard, L.; Otahal, P.; van der Mei, I.; Taylor, B. Latitude is significantly associated with the prevalence of multiple sclerosis: A meta-analysis. J. Neurol. Neurosurg. Psychiatr. 2011, 82, 1132–1141. [Google Scholar] [CrossRef]
- Agliardi, C.; Guerini, F.R.; Saresella, M.; Caputo, D.; Leone, M.A.; Zanzottera, M.; Bolognesi, E.; Marventano, I.; Barizzone, N.; Fasano, M.E.; et al. Vitamin D receptor (VDR) gene SNPs influence VDR expression and modulate protection from multiple sclerosis in HLA-DRB1*15-positive individuals. Brain Behav. Immun. 2011, 25, 1460–1467. [Google Scholar] [CrossRef]
- Sawcer, S.; Hellenthal, G.; Pirinen, M.; Spencer, C.C.A.; Patsopoulos, N.A.; Moutsianas, L.; Dilthey, A.; Su, Z.; Freeman, C.; Hunt, S.E.; et al. Genetic risk and a primary role for cell-mediated immune mechanisms in multiple sclerosis. Nature 2011, 476, 214–219. [Google Scholar]
- Ascherio, A.; Munger, K.L.; Simon, K.C. Vitamin D and multiple sclerosis. Lancet Neurol. 2010, 9, 599–612. [Google Scholar] [CrossRef]
- Ganesh, A.; Apel, S.; Metz, L.; Patten, S. The case for vitamin D supplementation in multiple sclerosis. Mult. Scler. Relat. Disord. 2013, 2, 281–306. [Google Scholar] [CrossRef] [PubMed]
- Iacopetta, K.; Collins-Praino, L.E.; Buisman-Pijlman, F.T.A.; Liu, J.; Hutchinson, A.D.; Hutchinson, M.R. Are the protective benefits of vitamin D in neurodegenerative disease dependent on route of administration? A systematic review. Nutr. Neurosci. 2018, 23, 251–280. [Google Scholar] [CrossRef] [PubMed]
- James, E.; Dobson, R.; Kuhle, J.; Baker, D.; Giovannoni, G.; Ramagopalan, S.V. The effect of vitamin D-related interventions on multiple sclerosis relapses: A meta-analysis. Mult. Scler. 2013, 19, 1571–1579. [Google Scholar] [CrossRef]
- Pozuelo-Moyano, B.; Benito-León, J.; Mitchell, A.J.; Hernández-Gallego, J. A systematic review of randomized, double-blind, placebo-controlled trials examining the clinical efficacy of vitamin D in multiple sclerosis. Neuroepidemiology 2013, 40, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Bagur, M.J.; Murcia, M.A.; Jiménez-Monreal, A.M.; Tur, J.A.; Bibiloni, M.M.; Alonso, G.L.; Martínez-Tomé, M. Influence of diet in multiple sclerosis: A systematic review. Adv. Nutr. 2017, 8, 463–472. [Google Scholar] [CrossRef]
- Hempel, S.; Graham, G.D.; Fu, N.; Estrada, E.; Chen, A.Y.; Miake-Lye, I.; Miles, J.N.V.; Shanman, R.; Shekelle, P.G.; Beroes, J.M.; et al. A systematic review of modifiable risk factors in the progression of multiple sclerosis. Mult. Scler. 2017, 23, 525–533. [Google Scholar] [CrossRef]
- Jagannath, V.A.; Filippini, G.; Di Pietrantonj, C.; Asokan, G.V.; Robak, E.W.; Whamond, L.; Robinson, S.A. Vitamin D for the management of multiple sclerosis. Cochrane Database Syst. Rev. 2018, 9, CD008422. [Google Scholar] [CrossRef]
- McLaughlin, L.; Clarke, L.; Khalilidehkordi, E.; Butzkueven, H.; Taylor, B.; Broadley, S.A. Vitamin D for the treatment of multiple sclerosis: A meta-analysis. J. Neurol. 2018, 265, 2893–2905. [Google Scholar] [CrossRef]
- Zheng, C.; He, L.; Liu, L.; Zhu, J.; Jin, T. The efficacy of vitamin D in multiple sclerosis: A meta-analysis. Mult. Scler. Relat. Disord. 2018, 23, 56–61. [Google Scholar] [CrossRef]
- Berezowska, M.; Coe, S.; Dawes, H. Effectiveness of vitamin D supplementation in the management of multiple sclerosis: A systematic review. Int. J. Mol. Sci. 2019, 20, 1301. [Google Scholar] [CrossRef]
- Mokry, L.E.; Ross, S.; Ahmad, O.S.; Forgetta, V.; Smith, G.D.; Goltzman, D.; Leong, A.; Greenwood, C.M.T.; Thanassoulis, G.; Richards, J.B. Vitamin D and risk of multiple sclerosis: A Mendelian randomization study. PLoS Med. 2015, 12, e1001866. [Google Scholar] [CrossRef] [PubMed]
- Rhead, B.; Bäärnhielm, M.; Gianfrancesco, M.; Mok, A.; Shao, X.; Quach, H.; Shen, L.; Schaefer, C.; Link, J.; Gyllenberg, A.; et al. Mendelian randomization shows a causal effect of low vitamin D on multiple sclerosis risk. Neurol. Genet. 2016, 2, e97. [Google Scholar] [CrossRef] [PubMed]
- Gianfrancesco, M.A.; Stridh, P.; Rhead, B.; Shao, X.; Xu, E.; Graves, J.S.; Chitnis, T.; Waldman, A.; Lotze, T.; Schreiner, T.; et al. Evidence for a causal relationship between low vitamin D, high BMI, and pediatric-onset MS. Neurology 2017, 88, 1623–1629. [Google Scholar] [CrossRef] [PubMed]
- Munger, K.L.; Zhang, S.M.; O’Reilly, E.; Hernán, M.A.; Olek, M.J.; Willett, W.C.; Ascherio, A. Vitamin D intake and incidence of multiple sclerosis. Neurology 2004, 62, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Munger, K.L.; Levin, L.I.; Hollis, B.W.; Howard, N.S.; Ascherio, A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 2006, 296, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Ismailova, K.; Poudel, P.; Parlesak, A.; Frederiksen, P.; Heitmann, B.L. Vitamin D in early life and later risk of multiple sclerosis-A systematic review, meta-analysis. PLoS ONE 2019, 14, e0221645. [Google Scholar] [CrossRef]
- Multiple Sclerosis Society of Canada. MS Society of Canada Launches Vitamin D Recommendations for MS. Available online: https://mssociety.ca/research-news/article/ms-society-of-canada-launches-vitamin-d-recommendations-for-ms (accessed on 25 August 2019).
- Atkinson, M.A.; Eisenbarth, G.S.; Michels, A.W. Type 1 diabetes. Lancet 2014, 383, 69–82. [Google Scholar] [CrossRef]
- International Diabetes Federation (IDF). IDF Diabetes Atlas. Available online: https://www.idf.org/e-library/epidemiology-research/diabetes-atlas/134-idf-diabetes-atlas-8th-edition.html (accessed on 3 June 2019).
- Fullerton, B.; Jeitler, K.; Seitz, M.; Horvath, K.; Berghold, A.; Siebenhofer, A. Intensive glucose control versus conventional glucose control for type 1 diabetes mellitus. Cochrane Database Syst. Rev. 2014, CD009122. [Google Scholar] [CrossRef]
- Daneman, D. Type 1 diabetes. Lancet 2006, 367, 847–858. [Google Scholar] [CrossRef]
- Rak, K.; Bronkowska, M. Immunomodulatory effect of vitamin D and its potential role in the prevention and treatment of type 1 diabetes mellitus—A narrative review. Molecules 2018, 24, 53. [Google Scholar] [CrossRef]
- Antico, A.; Tampoia, M.; Tozzoli, R.; Bizzaro, N. Can supplementation with vitamin D reduce the risk or modify the course of autoimmune diseases? A systematic review of the literature. Autoimmun. Rev. 2012, 12, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.-Y.; Zhang, W.-G.; Chen, J.J.; Zhang, Z.-L.; Han, S.-F.; Qin, L.-Q. Vitamin D intake and risk of type 1 diabetes: A meta-analysis of observational studies. Nutrients 2013, 5, 3551–3562. [Google Scholar] [CrossRef] [PubMed]
- Gregoriou, E.; Mamais, I.; Tzanetakou, I.; Lavranos, G.; Chrysostomou, S. The effects of vitamin D supplementation in newly diagnosed type 1 diabetes patients: Systematic review of randomized controlled trials. Rev. Diabet. Stud. 2017, 14, 260–268. [Google Scholar] [CrossRef]
- Zipitis, C.S.; Akobeng, A.K. Vitamin D supplementation in early childhood and risk of type 1 diabetes: A systematic review and meta-analysis. Arch. Dis. Child 2008, 93, 512–517. [Google Scholar] [CrossRef]
- Rewers, M.; Ludvigsson, J. Environmental risk factors for type 1 diabetes. Lancet 2016, 387, 2340–2348. [Google Scholar] [CrossRef]
- Hyppönen, E. Vitamin D and increasing incidence of type 1 diabetes-evidence for an association? Diabetes Obes. Metab. 2010, 12, 737–743. [Google Scholar] [CrossRef]
- Guo, S.-W.; Magnuson, V.L.; Schiller, J.J.; Wang, X.; Wu, Y.; Ghosh, S. Meta-analysis of vitamin D receptor polymorphisms and type 1 diabetes: A HuGE review of genetic association studies. Am. J. Epidemiol. 2006, 164, 711–724. [Google Scholar] [CrossRef] [PubMed]
- Sahin, O.A.; Goksen, D.; Ozpinar, A.; Serdar, M.; Onay, H. Association of vitamin D receptor polymorphisms and type 1 diabetes susceptibility in children: A meta-analysis. Endocr. Connect. 2017, 6, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Al Anouti, F.; Moukayed, M. Targeted 25-hydroxyvitamin D concentration measurements and vitamin D3 supplementation can have important patient and public health benefits. Eur. J. Clin. Nutr. 2020, 74, 366–376. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.-M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2018, 380, 33–44. [Google Scholar] [CrossRef]
- Jorde, R.; Grimnes, G. Serum cholecalciferol may be a better marker of vitamin D status than 25-hydroxyvitamin D. Med. Hypotheses 2018, 111, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Hahn, A.; Schön, C.; Wilhelm, M.; Obeid, R. Effect of two different multimicronutrient supplements on vitamin D status in women of childbearing age: A randomized trial. Nutrients 2017, 9, 30. [Google Scholar] [CrossRef] [PubMed]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
|
|
|
|
|
|
|
|
|
|
|
|
|
| SRs of | Effects of Vitamin D in Primary Prevention | Effects of Vitamin D in Patients | |
|---|---|---|---|
| Asthma | Observational studies | - | no data |
| RCTs | - | +1,2 | |
| COPD | Observational studies | no data | - |
| RCTs | no data | +1 | |
| ARI | Observational studies | +3 | no data |
| RCTs | +1 | o | |
| Dementia and cognitive decline | Observational studies | +1,4 | no data |
| RCTs | - | no data | |
| Depression | Observational studies | +1,4 | no data |
| RCTs | o (general)– (postpartum) | (major depression) | |
| MS | Observational studies | - | no data |
| RCTs | no data | o | |
| T1DM | Observational studies | - | no data |
| RCTs | no data | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maretzke, F.; Bechthold, A.; Egert, S.; Ernst, J.B.; Melo van Lent, D.; Pilz, S.; Reichrath, J.; Stangl, G.I.; Stehle, P.; Volkert, D.; et al. Role of Vitamin D in Preventing and Treating Selected Extraskeletal Diseases—An Umbrella Review. Nutrients 2020, 12, 969. https://doi.org/10.3390/nu12040969
Maretzke F, Bechthold A, Egert S, Ernst JB, Melo van Lent D, Pilz S, Reichrath J, Stangl GI, Stehle P, Volkert D, et al. Role of Vitamin D in Preventing and Treating Selected Extraskeletal Diseases—An Umbrella Review. Nutrients. 2020; 12(4):969. https://doi.org/10.3390/nu12040969
Chicago/Turabian StyleMaretzke, Friederike, Angela Bechthold, Sarah Egert, Jana B. Ernst, Debora Melo van Lent, Stefan Pilz, Jörg Reichrath, Gabriele I. Stangl, Peter Stehle, Dorothee Volkert, and et al. 2020. "Role of Vitamin D in Preventing and Treating Selected Extraskeletal Diseases—An Umbrella Review" Nutrients 12, no. 4: 969. https://doi.org/10.3390/nu12040969
APA StyleMaretzke, F., Bechthold, A., Egert, S., Ernst, J. B., Melo van Lent, D., Pilz, S., Reichrath, J., Stangl, G. I., Stehle, P., Volkert, D., Wagner, M., Waizenegger, J., Zittermann, A., & Linseisen, J., on behalf of the German Nutrition Society (DGE). (2020). Role of Vitamin D in Preventing and Treating Selected Extraskeletal Diseases—An Umbrella Review. Nutrients, 12(4), 969. https://doi.org/10.3390/nu12040969








