African Sorghum-Based Fermented Foods: Past, Current and Future Prospects
Abstract
1. Introduction
2. Overview on Sorghum
2.1. Nutitional Composition of Sorghum
2.2. Bioactive Constituents of Sorghum
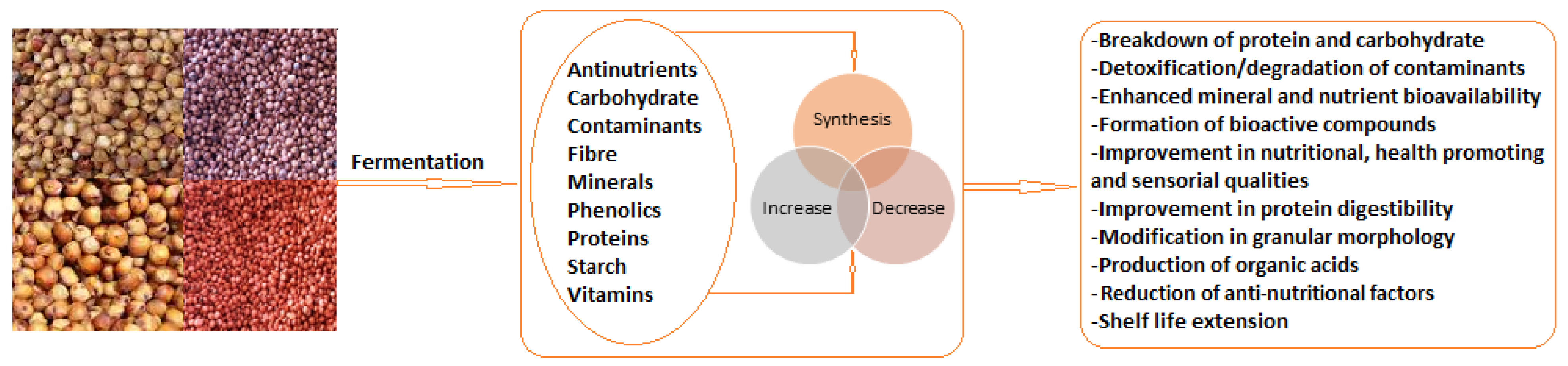
3. Sorghum Fermentation
4. African Sorghum Fermented Food Products: Traditional and Value Added Products
5. Safety of African Sorghum Fermented Foods
6. Future Projections
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT Food and Agriculture Organization Statistics. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 31 March 2020).
- Mabhaudhi, T.; O’Reilly, P.; Walker, S.; Mwale, S. Opportunities for underutilised crops in Southern Africa’s post-2015 development agenda. Sustainability 2016, 8, 302. [Google Scholar] [CrossRef]
- Sobowale, S.S.; Adebo, O.A.; Mulaba-Bafubiandi, A.F. Production of extrudate pasta from optimal-sorghum-peanut flour blend and influence of composite flours on some quality characteristics and sorption isotherms. Trans. Royal Soc. S. Afr. 2019, 74, 268–275. [Google Scholar] [CrossRef]
- Adebo, O.A.; Njobeh, P.B.; Mulaba-Bafubiandi, A.F.; Adebiyi, J.A.; Desobgo, S.C.Z.; Kayitesi, E. Optimization of fermentation conditions for ting production using response surface methodology. J. Food Process Preserv. 2018, 42, e13381. [Google Scholar] [CrossRef]
- Schober, T.J.; Bean, S.R. Sorghum and maize. In Gluten-Free Cereal Products and Beverages; Arendt, E.K., Bello, F.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 101–118. [Google Scholar] [CrossRef]
- Taylor, J.R.N.; Duodu, K.G. Sorghum and millets: Grain quality characteristics and management of quality requirements. In Cereal Grains; Wrigley, C., Batey, I., Miskelly, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 317–351. [Google Scholar] [CrossRef]
- Odunmbaku, L.A.; Sobowale, S.S.; Adenekan, M.K.; Oloyede, T.; Adebiyi, J.A.; Adebo, O.A. Influence of steeping duration, drying temperature, and duration on the chemical composition of sorghum starch. Food Sci. Nutr. 2018, 6, 348–355. [Google Scholar] [CrossRef]
- Adebo, O.A.; Njobeh, P.B.; Adebiyi, J.A.; Gbashi, S.; Kayitesi, E. Food metabolomics (Foodomics), a new frontier in food analysis and its potential in understanding fermented foods. In Functional Food–Improve Health through Adequate Food; Hueda, M.C., Ed.; InTech: Rijeka, Croatia, 2017; pp. 211–234. [Google Scholar] [CrossRef]
- Adebo, O.A.; Njobeh, P.B.; Adebiyi, J.A.; Gbashi, S.; Phoku, J.Z.; Kayitesi, E. Fermented pulse-based foods in developing nations as sources of functional foods. In Functional Food–Improve Health through Adequate Food; Hueda, M.C., Ed.; InTech: Rijeka, Croatia, 2017; pp. 77–109. [Google Scholar] [CrossRef]
- Rosales, E.; Pasoz, M.; Sanroman, M.A. Solid-state fermentation for food applications. In Current Developments in Biotechnology and Bioengineering; Pandey, A., Larroche, C., Soccol, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 319–355. [Google Scholar] [CrossRef]
- Adebo, O.A.; Medina-Meza, I.E. Impact of fermentation on the phenolic compounds and antioxidant activity of whole cereal grains: A mini review. Molecules 2020, 25, 927. [Google Scholar] [CrossRef]
- Xiang, H.; Sun-Waterhuse, D.; Waterhouse, G.I.N.; Cui, C.; Ruan, Z. Fermentation-enabled wellness foods: A fresh perspective. Food Sci. Hum. Well 2019, 8, 203–243. [Google Scholar] [CrossRef]
- Galati, A.; Oguntoyinbo, F.A.; Moschetti, G.; Crescimanno, M.; Settanni, L. The cereal market and the role of fermentation in cereal-based food production in Africa. Food Rev. Int. 2014, 30, 317–337. [Google Scholar] [CrossRef]
- Adebiyi, J.A.; Obadina, A.O.; Adebo, O.A.; Kayitesi, E. Fermented and malted millet products in Africa: Expedition from traditional/ethnic foods to industrial value added products. Crit. Rev. Food Sci. Nutr. 2018, 58, 463–474. [Google Scholar] [CrossRef]
- Tamang, J.P. Fermented foods for human life. In Microbes for Human Life; Chauhan, A.K., Verma, A., Kharakwal, H., Eds.; I.K International Publishing House Pvt: New Delhi, India, 2007; pp. 73–87. [Google Scholar] [CrossRef]
- Farhad, M.; Kailasapathy, K.; Tamang, J.P. Health aspects of fermented food. In Fermented Foods and Beverages of the World; Tamang, J.P., Kailasapathy, K., Eds.; CRC Press: London, UK, 2010; pp. 391–414. [Google Scholar] [CrossRef]
- Taylor, J.R.N.; Duodu, K.G. Effects of processing sorghum and millets on their phenolic phytochemicals and the implications of this to the health-enhancing properties of sorghum and millet food and beverage products. J. Sci. Food Agric. 2015, 95, 225–237. [Google Scholar] [CrossRef]
- Alavi, S.; Mazumdar, S.D.; Taylor, J.R.N. Modern convenient sorghum and millet food, beverage and animal feed products, and their technologies. In Sorghum and Millets: Chemistry, Technology and Nutritional Attributes, 2nd ed.; Taylor, J.R.N., Duodu, K.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 293–329. [Google Scholar] [CrossRef]
- Visarada, K.B.R.S.; Aruna, C. Sorghum: A Bundle of Opportunities in the 21st Century. In Breeding Sorghum for Diverse End Uses; Aruna, C., Visarada, K.B.R.S., Bhat, B.V., Tonapi, V.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–14. [Google Scholar] [CrossRef]
- Iqbal, M.A.; Iqbal, A. Overview on sorghum for food, feed, forage and fodder: Opportunities and problems in Pakistan’s perspectives. Am.-Eurasian J. Agric. Environ. Sci. 2015, 15, 1818–1826. [Google Scholar] [CrossRef]
- Wrigley, C. The cereal grains: Providing our food, feed and fuel needs. In Cereal Grains; Wrigley, C., Batey, I., Miskelly, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 27–40. [Google Scholar] [CrossRef]
- Rooney, L.W.; Serna-Sladiver, S.O. Sorghum. In Encyclopedia of Food Science and Nutrition; Caballero, B., Ed.; Elsevier: Amsterdam, The Netherlands, 2003; pp. 5370–5375. [Google Scholar] [CrossRef]
- Grayboasch, R.A. The grain crops: An overview. In Encyclopedia of Food Grains, 2nd ed.; Wrigley, C., Corke, H., Seetharaman, K., Faubion, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 16–21. [Google Scholar] [CrossRef]
- Linder, H.P.; Lehmann, C.E.R.; Archibald, S.; Osborne, C.P.; Richardson, D.M. Global grass (Poaceae) success underpinned by traits facilitating colonization, persistence and habitat transformation. Biol. Rev. 2017, 93, 1125–1144. [Google Scholar] [CrossRef] [PubMed]
- Ratnavathi, C.V.; Komala, V.V. Sorghum grain quality. In Sorghum Biochemistry: An Industrial Perspective; Ratnavathi, C.V., Patil, J.V., Chavan, U.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–61. [Google Scholar] [CrossRef]
- Eckhoff, S.R.; Watson, S.A. Corn and sorghum starch production. In Starch: Chemistry and Technology, 3rd ed.; BeMiller, J., Whistler, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 373–439. [Google Scholar] [CrossRef]
- Awika, J.M. Sorghum: Its unique nutritional and health-promoting attributes. In Gluten-Free Ancient Grains; Taylor, J.R.N., Awika, J.M., Eds.; Woodhead Publishing: Cambridge, UK, 2017; pp. 21–54. [Google Scholar] [CrossRef]
- Pontieri, P.; Del Giudice, L. Sorghum: A novel and healthy food. Encycl. Food Health 2016, 33–43. [Google Scholar] [CrossRef]
- Batey, I. The diversity of uses for cereal grains. In Cereal Grains; Wrigley, C., Batey, I., Miskelly, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 41–53. [Google Scholar] [CrossRef]
- Taylor, J.R.N.; Emmambux, M.N. Gluten-free cereal products and beverages. In Gluten-Free Foods and Beverages from Millets; Arendt, E.K., Bello, F.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 119–148. [Google Scholar] [CrossRef]
- Beta, T.; Corke, H.; Taylor, J.R.N. Starch properties of Barnard red, a South African red sorghum variety of significance in traditional African brewing. Starch 2000, 52, 467–470. [Google Scholar] [CrossRef]
- Topping, D.L. Soluble fiber polysaccharides: Effects on plasma cholesterol and colonic fermentation. Nutr. Rev. 1991, 49, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Warrand, J. Healthy polysaccharides the next chapter in food products. Food Technol. Biotechnol. 2006, 44, 355–370. [Google Scholar]
- Jones, R.W.; Beckwith, A.C. Proximate composition and proteins of three grain sorghum hybrids and their dry-mill fractions. J. Agric. Food Chem. 1970, 18, 33–36. [Google Scholar] [CrossRef]
- Okoh, P.N.; Obilana, A.T.; Njoku, P.C.; Aduku, A.O. Proximate analysis, amino acid composition and tannin content of improved Nigerian sorghum varieties and their potential in poultry feeds. Anim. Feed Sci. Technol. 1982, 7, 359–364. [Google Scholar] [CrossRef]
- Adebiyi, A.O.; Adebiyi, A.P.; Olaniyi, E.O. Nutritional composition of Sorghum bicolor starch hydrolyzed with amylase from Rhizopus sp. Afr. J. Biotechnol. 2005, 4, 1089–1094. [Google Scholar] [CrossRef]
- Shawrang, P.; Sadeghi, A.A.; Behgar, M.; Zareshahi, H.; Shahhoseini, G. Study of chemical compositions, anti-nutritional contents and digestibility of electron beam irradiated sorghum grains. Food Chem. 2011, 125, 376–379. [Google Scholar] [CrossRef]
- Shargie, N. Physico-Chemical Characteristics and Nutritional Value of Sorghum Grain. Available online: http://www.grainsa.co.za/physio-chemical-characteristics-and-nutritional-value-of-sorghum-grain (accessed on 31 March 2020).
- Udachan, I.S.; Sahoo, A.K.; Hend, G.M. Extraction and characterization of sorghum (Sorghum bicolor L. Moench) starch. Int. Food Res. J. 2012, 19, 315–319. [Google Scholar]
- Awadelkareem, A.M.; Hassan, E.G.; Fageer, A.S.M.; Sulieman, A.M.; Mustafa, A.M.I. The nutritive value of two sorghum cultivar. Int. J. Food Nutr. Sci. 2015, 4, 1–7. [Google Scholar]
- Ndimba, R.; Grootboom, A.W.; Mehlo, L.; Mkhonza, N.L.; Kossmann, J.; Barnabas, A.D.; Mtshali, C.; Pineda-Vargas, C. Detecting changes in the nutritional value and elemental composition of transgenic sorghum grain. Nuclear Instr. Methods Phys. Res. B 2015, 363, 183–187. [Google Scholar] [CrossRef]
- Singh, E.; Jain, P.K.; Sharma, S. Effect of different household processing on nutritional and anti-nutritional factors in Vigna aconitifolia and Sorghum bicolour (L.) Moench seeds and their product development. J. Med. Nutr. Nutraceut. 2015, 4, 95–100. [Google Scholar] [CrossRef]
- Ape, D.I.; Nwogu, N.A.; Uwakwe, E.I.; Ikedinobi, C.S. Comparative proximate analysis of maize and sorghum bought from Ogbete main market of Enugu state, Nigeria. Greener J. Agric. Sci. 2016, 6, 272–275. [Google Scholar] [CrossRef]
- Jimoh, W.L.O.; Abdullahi, M.S. Proximate analysis of selected sorghum cultivars. Bayero. J. Pure Appl. Sci. 2017, 10, 285–288. [Google Scholar] [CrossRef]
- Mohapatra, D.; Patel, A.S.; Kar, A.; Deshpande, S.S.; Tripathi, M.K. Effect of different processing conditions on proximate composition, antioxidants, anti-nutrients and amino acid profile of grain sorghum. Food Chem. 2019, 271, 129–135. [Google Scholar] [CrossRef]
- Kamath, V.; Niketh, S.; Chandrashekar, A.; Rajini, P.S. Chymotryptic hydrolysates of α-kafirin, the storage protein of sorghum (Sorghum bicolor) exhibited angiotensin converting enzyme inhibitory activity. Food Chem. 2007, 100, 306–311. [Google Scholar] [CrossRef]
- Camargo-Filho, I.; Cortez, D.A.G.; Ueda-Nakamura, T.; Nakamura, C.V.; Dias-Filho, B.P. Antiviral activity and mode of action of a peptide isolated from Sorghum bicolor. Phytomedicine 2008, 15, 202–208. [Google Scholar] [CrossRef]
- Lin, P.; Wong, J.H.; Ng, T.B.; Ho, V.S.; Xia, L. A sorghum xylanase inhibitor-like protein with highly potent antifungal, antitumor and HIV-1 reverse transcriptase inhibitory activities. Food Chem. 2013, 141, 2916–2922. [Google Scholar] [CrossRef]
- Cruz, R.A.O.; López, J.L.C.; Aguilar, G.A.G.; García, H.A.; Gorinstein, S.; Romero, R.C.; Sánchez, M.R. Influence of sorghum karifin on serum lipid profile and antioxidant activity in hyperlipidemic rats (in vitro and in vivo studies). BioMed Res. Int. 2015, 164725. [Google Scholar] [CrossRef]
- Khalil, J.K.; Sawaya, W.N.; Safi, W.F.; Al-Mohammad, H.M. Chemical composition and nutritional quality of sorghum flour and bread. Plant Food Hum. Nutr. 1984, 34, 141–150. [Google Scholar] [CrossRef]
- Hulse, J.H.; Laing, E.M.; Pearson, O.E. Sorghum and Millets: Their Chemical Composition and Nutritive Value; Academic Press: New York, NY, USA, 1980. [Google Scholar]
- Salunkhe, D.K.; Kadam, S.S.; Chavan, J.K. Nutritional quality of proteins in grain sorghum. Plant Food Hum. Nutr. 1977, 2, 187–205. [Google Scholar] [CrossRef]
- Afify, A.E.M.R.; El-Beltagi, H.S.; Abd El-Salam, S.M.; Omran, A.A. Protein solubility, digestibility and fractionation after germination of sorghum varieties. PLoS ONE 2012, 7, e31154. [Google Scholar] [CrossRef] [PubMed]
- Mabelele, M.; Siwela, M.; Gous, R.M.; Iji, A. Chemical composition and nutritive value of South African sorghum varieties as feed for broiler chickens. S. Afr. J. Anim. Sci. 2015, 45, 206–213. [Google Scholar] [CrossRef]
- Shegro, A.; Shargie, N.G.; van Biljon, A.; Labuschagne, M.T. Diversity in starch, protein and mineral composition of sorghum landrace accessions from Ethiopia. J. Crop. Sci. Biotechnol. 2012, 15, 275–280. [Google Scholar] [CrossRef]
- Pontieri, P.; Troisi, J.; Di Fiore, R.; Di Maro, A.; Bean, S.R.; Tuinstra, M.R.; Roemer, E.; Boffa, A.; Del Giddice, A.; Pizzolante, G.; et al. Mineral contents in grains of seven food-grade sorghum hybrids grown in a Mediterranean environment. Austr. J. Crop. Sci. 2014, 8, 1550–1559. [Google Scholar]
- Gerrano, A.S.; Labuschagne, M.T.; van Biljon, A.; Shargie, N.G. Quantification of mineral composition and total protein content in sorghum [Sorghum bicolor (L.) Moench] genotypes. Cereal Res. Comm. 2016, 44, 272–285. [Google Scholar] [CrossRef]
- Serna-Saldivar, S.O.; Espinosa-Ramirez, J. Grain structure and grain chemical composition. In Sorghum and millets: Chemistry, Technology and Nutritional Attributes, 2nd ed.; Taylor, J.R.N., Duodu, K.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 85–130. [Google Scholar] [CrossRef]
- Kulamarva, A.G.; Sosle, V.R.; Raghavan, G.S.V. Nutritional and rheological properties of sorghum. Int. J. Food Prop. 2009, 12, 55–69. [Google Scholar] [CrossRef]
- Ochanda, S.O.; Akoth, O.C.; Mwasaru, A.M.; Kagwiria, O.J.; Mutiso, M.F. Effects of malting and fermentation treatments on group B-vitamins of red sorghum, white sorghum and pearl millets in Kenya. J. Appl. Biosci. 2010, 34, 2128–2134. [Google Scholar]
- Saleh, A.S.M.; Zhang, Q.; Chen, J.; Shen, Q. Millet grains: Nutritional quality, processing, and potential health benefits. Compr. Rev. Food Sci. Food Saf. 2013, 12, 281–295. [Google Scholar] [CrossRef]
- Leguizamón, C.; Weller, C.L.; Schlegel, V.L.; Carr, T.P. Plant sterol and policosanol characterization of hexane extracts from grain sorghum, corn and their DDGS. J. Amer. Oil Chem. Soc. 2009, 86, 707–716. [Google Scholar] [CrossRef]
- Lee, B.H.; Carr, T.P.; Weller, C.L.; Cuppett, S.; Dweikat, I.M.; Schlegel, V. Grain sorghum whole kernel oil lowers plasma and liver cholesterol in male hamsters with minimal wax involvement. J. Funct. Foods 2014, 7, 709–718. [Google Scholar] [CrossRef][Green Version]
- Adebo, O.A.; Kayitesi, E.; Tugizimana, F.; Njobeh, P.B. Differential metabolic signatures in naturally and lactic acid bacteria (LAB) fermented ting (a Southern African food) with different tannin content, as revealed by gas chromatography mass spectrometry (GC–MS)-based metabolomics. Food Res. Int. 2019, 121, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Aruna, C.; Visarada, K.B.R.S. Other industrial uses of sorghum. In Breeding Sorghum for Diverse End Uses; Aruna, C., Visarada, K.B.R.S., Bhat, B.V., Tonapi, V.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 271–292. [Google Scholar] [CrossRef]
- Khan, I.; Yousif, A.; Johnson, S.K.; Gamlath, S. Effect of sorghum flour addition on resistant starch content, phenolic profile and antioxidant capacity of durum wheat pasta. Food Res. Int. 2013, 54, 578–586. [Google Scholar] [CrossRef]
- Licata, R.; Chu, J.; Wang, S.; Coorey, R.; James, A.; Zhao, Y.; Johnson, S. Determination of formulation and processing factors affecting slowly digestible starch, protein digestibility and antioxidant capacity of extruded sorghum-maize composite flour. Int. J. Food Sci. Technol. 2014, 49, 1408–1419. [Google Scholar] [CrossRef]
- Kaur, K.D.; Jha, A.; Sabikhi, L.; Singh, A.K. Significance of coarse cereals in health and nutrition: A review. J. Food Sci. Technol. 2014, 51, 1429–1441. [Google Scholar] [CrossRef]
- Rao, B.D.; Bharti, N.; Srinivas, K. Reinventing the commercialization of sorghum as health and convenient foods: Issues and challenges. Indian J. Econ. Dev. 2017, 13, 1–10. [Google Scholar] [CrossRef]
- Girard, A.L.; Awika, J.M. Sorghum polyphenols and other bioactive components as functional and health promoting food ingredients. J. Cereal. Sci. 2018, 84, 112–124. [Google Scholar] [CrossRef]
- Etuk, E.B.; Okuedo, N.J.; Esonu, B.O.; Udedibie, A.B.I. Antinutritional factors in sorghum: Chemistry, mode of action and effects on livestock and poultry. Online J. Anim. Feed Res. 2012, 2, 113–119. [Google Scholar]
- Faquinello, P.; Murakami, A.E.; Cella, P.S.; Franco, J.R.G.; Sakamoto, M.I.; Bruno, L.D.G. High tannin sorghum in diets of Japanese quails (Coturnix coturnix japonica). Revista. Brasileira de Ciencia Avícola 2004, 6, 81–86. [Google Scholar] [CrossRef]
- Goel, G.; Puniya, A.K.; Aguliar, C.N.; Singh, K. Interaction of gut microflora with tannins in feeds. Naturwissenschaften 2005, 92, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Aronson, J.K. Plant poisons and traditional medicines. In Manson’s Tropical Infectious Diseases, 23rd ed.; Cereal grains Farrar, J., Hotez, P.J., Junghanss, T., Kang, G., Lalloo, D., White, N.J., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 1128–1150. [Google Scholar] [CrossRef]
- Proietti, I.; Frazzoli, C.; Mantovani, A. Exploiting nutritional value of staple foods in the world’s semi-arid areas: Risks, benefits, challenges and opportunities of sorghum. Healthcare 2015, 3, 172–193. [Google Scholar] [CrossRef] [PubMed]
- Obizoba, C.I.; Atii, J.V. Effect of soaking, sprouting, fermentation and cooking on nutrient composition and some anti-nutritional factors of sorghum (Guinesia) seeds. Plant Foods Hum. Nutr. 1991, 45, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Mahgoub, S.E.O.; Elhag, S.A. Effect of milling, soaking, malting, heat-treatment and fermentation on phytate level of four Sudanese sorghum cultivars. Food Chem. 1998, 61, 77–80. [Google Scholar] [CrossRef]
- Prasad, S.; Dhanya, M.S. Determination and detoxification of cyanide content in sorghum for ethanol production using Saccharomyces cerevisiae strain. J. Metab. Syst. Biol. 2011, 2, 10–14. [Google Scholar]
- Ojha, P.; Adhikari, R.; Karki, R.; Mishra, A.; Subedi, U.; Karki, B.K. Malting and fermentation effects on antinutritional components and functional characteristics of sorghum flour. Food Sci. Nutr. 2018, 6, 47–53. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W. Phenolic compounds in cereal grains and their health benefits. Cereal. Foods World 2007, 52, 105–111. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W.; Waniska, R.D.; Rooney, W.L. Phenolic compounds and antioxidant activity of sorghum grains of varying genotypes. J. Agric. Food Chem. 2005, 53, 6813–6818. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W. Sorghum and millet phenols and antioxidants. J. Cereal. Sci. 2006, 44, 236–251. [Google Scholar] [CrossRef]
- Van Rensburg, S.J. Epidemiological and dietary evidence for a specific nutritional disposition to esophageal cancer. J. Natl. Cancer Inst. 1981, 67, 243–251. [Google Scholar]
- Ly, D.; Mayrhofer, S.; Dimig, K.J. Significance of traditional fermented foods in the lower Mekong subregion: A focus on lactic acid bacteria. Food Biosci. 2018, 26, 113–125. [Google Scholar] [CrossRef]
- Shih, C.H.; Siu, S.O.; Ng, R.; Wong, E.; Chiu, L.C.M.; Chu, I.K.; Lo, C. Quantitative analysis of anticancer 3-deoxyanthocyanidins in infected sorghum seedlings. J. Agric. Food Chem. 2007, 55, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Burdette, A.; Garner, P.L.; Mayer, E.P.; Hargrove, J.L.; Hartle, D.K.; Greenspan, P. Anti-inflammatory activity of select sorghum (Sorghum bicolor) brans. J. Med. Foods 2010, 13, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Oboh, G.; Akomolafe, T.L.; Adetuyi, A.O. Inhibition of cyclophosphamide-induced oxidative stress in brain by dietary inclusion of red dye extracts from sorghum (Sorghum bicolor) stem. J. Med. Foods 2010, 13, 1075–1080. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Park, Y. Anti-diabetic effect of sorghum extract on hepatic gluconeogenesis of streptozotocin-induced diabetic rats. Nutr. Metab. 2012, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.R.N.; Belton, P.S.; Beta, T.; Duodu, K.G. Increasing the utilization of sorghum, millets and pseudocereals: Developments in the science of their phenolic phytochemicals, biofortification and protein functionality. J. Cereal. Sci. 2014, 59, 257–275. [Google Scholar] [CrossRef]
- Capozzi, V.; Fragassa, M.; Romaniello, R.; Berbegal, C.; Russo, P.; Spano, G. Spontaneous food fermentations and potential risks for human health. Fermentation 2017, 3, 49. [Google Scholar] [CrossRef]
- Adebo, O.A.; Njobeh, P.B.; Adeboye, A.S.; Adebiyi, J.A.; Sobowale, S.S.; Ogundele, O.M.; Kayitesi, E. Advances in fermentation technology for novel food products. In Innovations in Technologies for Fermented Food and Beverage Industries; Panda, S., Shetty, P., Eds.; Springer: Cham, Switzerland, 2018; pp. 71–87. [Google Scholar] [CrossRef]
- Masebe, K.M.; Adebo, O.A. Production and quality characteristics of a probiotic beverage from watermelon (Citrullus lanatus). In Engineering, Technology and Waste Management (SETWM-19), Proceedings of the 17th Johannesburg International Conference on Science, Johannesburg, South Africa, 18–19 November 2019; Fosso-Kankeu, E., Waanders, F., Bulsara, H.K.P., Eds.; Eminent Association of Pioneers and North-West University: Johannesburg, South Africa, 2019; pp. 42–49. [Google Scholar] [CrossRef]
- Brandt, M.J. Starter cultures for cereal based foods. Food Microbiol. 2014, 37, 41–43. [Google Scholar] [CrossRef]
- Elkhalifa, A.E.O.; Ali, A.M.; El Tinay, A.H. Fermented sorghum foods of Sudan-a review. J. Food Sci. Technol. 2007, 44, 343–349. [Google Scholar]
- Adebo, O.A.; Njobeh, P.B.; Adebiyi, J.A.; Kayitesi, E. Co-influence of fermentation time and temperature on physicochemical properties, bioactive components and microstructure of ting (a Southern African food) from whole grain sorghum. Food Biosci. 2018, 25, 118–127. [Google Scholar] [CrossRef]
- Teusink, B.; Molenaar, D. Systems biology of lactic acid bacteria: For food and thought. Curr. Opin. Syst. Biol. 2017, 6, 7–13. [Google Scholar] [CrossRef]
- Gänzle, M.G. Lactic metabolism revisited: Metabolism of lactic acid bacteria in food fermentations and food spoilage. Curr. Opin. Food Sci. 2015, 2, 106–117. [Google Scholar] [CrossRef]
- Nyanzi, R.; Jooste, P.J. Cereal-based functional foods. In Probiotics; Rigobelo, E., Ed.; InTech: Rijeka, Croatia, 2012; pp. 161–196. [Google Scholar] [CrossRef]
- Eggum, B.O.; Monowar, L.; Back Knudsen, K.E.; Munck, L.; Axtell, J. Nutritional quality of sorghum and sorghum foods from Sudan. J. Cereal. Sci. 1983, 1, 127–137. [Google Scholar] [CrossRef]
- Franz, C.M.A.P.; Holzapfel, W.H. Examples of lactic-fermented foods of the African continent. In Lactic Acid Bacteria: Microbiological and Functional Aspects; Vinderola, G., Ouwehand, A., Salminen, S., von Wright, A., Eds.; CRC Press: Boca Raton, FL, USA, 2019; pp. 235–254. [Google Scholar] [CrossRef]
- Kolawole, O.M.; Kayode, R.M.O.; Akindayo, B. Proximate and microbial analyses of burukutu and pito produced in Ilorin. Nigeria. Afr. J. Biotechnol. 2007, 6, 587–590. [Google Scholar]
- Eze, V.C.; Eleke, O.I.; Omeh, Y.S. Microbiological and nutritional qualities of burukutu sold in mammy market Abakpa, Enugu State, Nigeria. Am. J. Food Nutr. 2011, 1, 141–146. [Google Scholar] [CrossRef]
- Alo, M.N.; Eze, U.A.; Eda, N.E. Microbiological qualities of burukutu produced from a mixture of sorghum and millet. Am. J. Food Nutr. 2012, 2, 96–102. [Google Scholar] [CrossRef]
- Blandino, A.; Al-Aseeri, M.E.; Pandiella, S.S.; Cantero, D.; Webb, C. Cereal-based fermented foods and beverages. Food Res. Int. 2003, 36, 527–543. [Google Scholar] [CrossRef]
- Marsh, A.J.; Hill, C.; Ross, R.P.; Cotter, P.D. Fermented beverages with health-promoting potential: Past and future perspectives. Trends Food Sci. Technol. 2014, 38, 113–124. [Google Scholar] [CrossRef]
- Mwale, M.M. Microbiological quality and safety of the Zambian fermented cereal beverage: Chibwantu. PhD Thesis, University of Free State, Bloemfontein, South Africa, 2014. [Google Scholar]
- Togo, A.H.; Ferusa, B.S.; Mutukumira, N.A. Identification of lactic acid bacteria isolated from opaque beer (chibuku) for potential use as a starter culture. J. Food Technol. Afr. 2002, 7, 93–97. [Google Scholar] [CrossRef][Green Version]
- Gadaga, T.H.; Mutakumira, A.N.; Narvhus, J.A.; Ferusu, S.B. A review of traditional fermented foods and beverages in Zimbabwe. Int. J. Food Microbiol. 1999, 53, 1–11. [Google Scholar] [CrossRef]
- Van der Aa Kühle, A.; Jespersen, L.; Glover, R.L.; Diawara, B.; Jakobsen, M. Identification and characterization of Saccharomyces cerevisiae strains isolated from West African sorghum beer. Yeast 2001, 18, 1069–1079. [Google Scholar] [CrossRef] [PubMed]
- Sawadogo-Lingani, H.; Lei, V.; Diawara, B.; Nielsen, D.S.; Moller, P.L.; Traore, A.S.; Jakobsen, M. The biodiversity of predominant lactic acid bacteria in dolo and pito wort, for production of sorghum beer. J. Appl. Microbiol. 2007, 103, 765–777. [Google Scholar] [CrossRef] [PubMed]
- Mukisa, I.M.; Ntaate, D.; Byakika, S. Application of starter cultures in the production of Enturire–A traditional sorghum-based alcoholic beverage. Food Sci. Nutr. 2017, 5, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Adinsi, L.; Vieira-Dalode, G.; Akissoe, N.H.; Anihouvi, V.; Mestres, C.; Jacobs, A.; Dlamini, N.; Pallet, D.; Hounhouigan, J.D. Processing and quality attributes of gowe: A malted and fermented cereal-based beverage from Benin. Food Chain 2014, 4, 171–183. [Google Scholar] [CrossRef]
- Vieira-Dalodé, G.; Jespersen, L.; Hounhouigan, J.; Moller, P.L.; Nago, C.M.; Jakobsen, M. Lactic acid bacteria and yeasts associated with gowé production from sorghum in Bénin. J. Appl. Microbiol. 2007, 103, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.R. Fermented weaning foods. In Microbiology of Fermented Foods; Wood, B.J.B., Ed.; Thomson Science: London, UK, 1998; pp. 790–811. [Google Scholar] [CrossRef]
- Yousif, N.M.K.; Huch, M.; Schuster, T.; Cho, G.S.; Dirar, H.A.; Holzapfel, W.H.; Franz, C.M.A.P. Diversity of lactic acid bacteria from Hussuwa, a traditional African fermented sorghum food. Food Microbiol. 2010, 6, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Lyumugabe, F.; Kamaliza, G.; Bajyana, E.; Thonart, P. Microbiological and physico-chemical characteristic of Rwandese traditional beer “Ikigage”. Afr. J. Biotechnol. 2010, 9, 4241–4246. [Google Scholar]
- Dandessa, C. Review on Ethiopian traditional fermented foods, its microbial ecology and nutritional value. Int. J. Curr. Res. Acad. Rev. 2019, 7, 13–27. [Google Scholar] [CrossRef]
- Mohammed, S.I.; Steenson, L.R.; Kirleis, A.W. Isolation and characterization of microorganisms associated with the traditional sorghum fermentation for production of Sudanese Kisra. Appl. Environ. Microbiol. 1991, 57, 2529–2533. [Google Scholar] [CrossRef]
- Hamad, S.H.; Dieng, M.C.; Ehrmann, M.A.; Vogel, R.F. Characterization of the bacterial flora of Sudanese sorghum, flour and sorghum sourdough. J. Appl. Microbiol. 1997, 28, 764–770. [Google Scholar] [CrossRef]
- Ali, A.A.; Mustafa, M.M. Isolation, characterization and identification of lactic acid bacteria from fermented sorghum dough used in Sudanese kisra preparation. Pakistan J. Nutri. 2009, 8, 1814–1818. [Google Scholar] [CrossRef][Green Version]
- Gassem, M.A.A. Study of micro-organisms associated with the fermented bread (khamir) produced from sorghum in Gizan region, Saudi Arabia. J. Appl. Microbiol. 1999, 86, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Hesseltine, C.W. Some important fermented foods in mid-Asia, the Middle East and Africa. J. Amer. Oil Chem. Soc. 1979, 56, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Kayitesi, E.; Behera, S.K.; Panda, S.K.; Dlamini, B.C.; Mulaba-Bafubiandi, A.F. Amasi and mageu: Expedition from ethnic Southern African foods to cosmopolitan markets. In Fermented Food-Part II: Technological Interventions; Ray, R.C., Montet, D., Eds.; CRC Press: Boca Raton, FL, USA, 2017; pp. 384–399. [Google Scholar]
- Odunfa, S.A.; Oyewole, O.B. African Fermented Foods; Blackie Academic and Professional: London, UK, 1997. [Google Scholar] [CrossRef]
- Dirar, H.A. The Indigenous Fermented Foods of the Sudan; C.A.B. International: Wallingford, UK, 1993. [Google Scholar]
- Dirar, H.A. The fermented foods of the Sudan. Ecol. Food Nutr. 1994, 32, 207–218. [Google Scholar] [CrossRef]
- Graham, G.G.; MacLean, W.C.; Morales, E.; Hamaker, B.R.; Kirleis, A.W.; Mertz, E.T.; Axtell, J.D. Digestibility and utilization of protein and energy from Nasha, a traditional Sudanese fermented sorghum weaning food. J. Nutr. 1986, 116, 978–984. [Google Scholar] [CrossRef] [PubMed]
- Omemu, A.M.; Bankole, M.O. Consumer’s knowledge, attitude, usage and storage pattern of ogi–a fermented cereal gruel in South West, Nigeria. Food Public Health 2015, 5, 77–83. [Google Scholar] [CrossRef]
- Adebayo-Tayo, B.C.; Needum, G.E. Microbiological, physicochemical and sensory evaluation of “ori-ese” produced from fortified sorghum. Afr. J. Food Agric. Nutr. Dev. 2011, 11, 4785–4799. [Google Scholar] [CrossRef]
- Shayo, N.B.; Kamala, A.; Gidamis, A.B.; Nnko, S.A. Aspects of manufacture, composition and safety of orubisi: A traditional alcoholic beverage in the north-western region of Tanzania. Int. J. Food Sci. Nutr. 2000, 51, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Oriola, O.B.; Boboye, B.E.; Adetuyi, F.C. Bacterial and fungal communities associated with the production of a Nigerian fermented beverage, “otika”. Jordan J. Biolog. Sci. 2017, 10, 127–133. [Google Scholar]
- Sawadogo-Lingani, H.; Diawara, B.; Traore, A.S.; Jakobsen, M. Technological properties of Lactobacillus fermentum in the processing of dolo and pito, West African sorghum beers, for selection of starter cultures. J. Appl. Microbiol. 2008, 104, 873–882. [Google Scholar] [CrossRef] [PubMed]
- Ajiboye, T.O.; Iliasu, G.A.; Adeleye, A.O.; Abdussalam, F.A.; Akinpelu, S.A.; Ogunbode, S.M.; Jimoh, S.O.; Oloyede, O.B. Nutritional and antioxidant dispositions of sorghum/millet beverages indigenous to Nigeria. Food Sci. Nutr. 2014, 2, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Lemi, B.W. Microbiology of Ethiopian traditionally fermented beverages and condiments. Int. J. Microbiol. 2020, 1478536. [Google Scholar] [CrossRef]
- Djè, K.M.; Aka, S.; Zinzendorf, N.Y.; Yao, K.C.; Loukou, Y.G. Predominant lactic acid bacteria involved in the spontaneous fermentation step of tchapalo process, a traditional sorghum beer of Côte d’Ivoire. Res. J. Biol. Sci. 2009, 4, 789–795. [Google Scholar]
- N’guessan, K.F.; Brou, K.; Jacques, N.; Casaregola, S.; Djè, K.M. Identification of yeasts during alcoholic fermentation of tchapalo, a traditional sorghum beer from Côte d’Ivoire. Antonie Van Leeuwenhoek 2011, 99, 855–864. [Google Scholar] [CrossRef] [PubMed]
- Kayodé, A.P.P.; Adegbidi, A.; Hounhouigan, J.D.; Linnemann, A.R.; Nout, M.J.R. Quality of farmer’s varieties of sorghum and derived foods as perceived by consumers in Benin. Ecol. Food Nutr. 2005, 44, 271–294. [Google Scholar] [CrossRef]
- Kayodé, A.P.P.; Hounhouigana, J.D.; Nout, M.J.R. Impact of brewing process operations on phyta, phenolic compounds and in vitro solubility of iron and zinc in opaque sorghum beer. LWT 2007, 40, 834–841. [Google Scholar] [CrossRef]
- Madoroba, E.; Steenkamp, E.T.; Theron, J.; Huys, G.; Scheirlinck, I.; Cloete, T.E. Polyphasic taxonomic characterization of lactic acid bacteria isolated from spontaneous sorghum fermentations used to produce ting, a traditional South African food. Afr. J. Biotechnol. 2009, 8, 458–463. [Google Scholar]
- Madoroba, E.; Steenkamp, E.T.; Theron, J.; Scheirlinck, I.; Cloete, T.E.; Huys, G. Diversity and dynamics of bacterial populations during spontaneous sorghum fermentations used to produce ting, a South African food. Syst. Appl. Microbiol. 2011, 34, 227–234. [Google Scholar] [CrossRef]
- Sekwati-Monang, B.; Gänzle, M.G. Microbiological and chemical characterization of ting, a sorghum-based sourdough product from Botswana. Int. J. Food Microbiol. 2011, 150, 115–121. [Google Scholar] [CrossRef]
- Matumba, L.; Monjerezi, M.; Khonga, E.B.; Lakudzala, D.D. Aflatoxins in sorghum, sorghum malt and traditional opaque beer in southern Malawi. Food Cont. 2011, 22, 266–268. [Google Scholar] [CrossRef]
- Katongole, J.N. The microbial succession in indigenous fermented maize products. Master’s Thesis, University of the Free State, Bloemfontein, South Africa, 2008. [Google Scholar]
- Wakil, S.M.; Kazeem, M.O. Quality assessment of weaning food produced from fermented cereal-legume blends using starters. Int. Food Res. J. 2012, 19, 1679–1685. [Google Scholar]
- Osman, M.A. Changes in sorghum enzyme inhibitors, phytic acid, tannins and in vitro protein digestibility occurring during khamir (local bread) fermentation. Food Chem. 2004, 88, 129–134. [Google Scholar] [CrossRef]
- Kohajdova, Z.; Karovicova, J. Fermentation of cereals for specific purpose. J. Food Nutr. Res. 2007, 46, 51–57. [Google Scholar]
- Kaushik, G.; Satya, S.; Naik, S.N. Food processing a tool to pesticide residue dissipation–A review. Food Rev. Int. 2009, 42, 26–40. [Google Scholar] [CrossRef]
- Ezekiel, C.N.; Abia, W.A.; Ogara, I.M.; Sulyok, M.; Warth, B.; Krska, R. Fate of mycotoxins in two popular traditional cereal-based beverages (kunu-zaki and pito) from rural Nigeria. LWT-Food Sci. Technol. 2015, 60, 137–141. [Google Scholar] [CrossRef]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef]
- Adebiyi, J.A.; Kayitesi, E.; Adebo, O.A.; Changwa, R.; Njobeh, P.B. Food fermentation and mycotoxin detoxification: An African perspective. Food Cont. 2019, 106, 106731. [Google Scholar] [CrossRef]
- Adebo, O.A.; Njobeh, P.B.; Gbashi, S.; Nwinyi, O.C.; Mavumengwana, V. Review on microbial degradation of aflatoxins. Crit. Rev. Food Sci. Nutr. 2017, 57, 3208–3217. [Google Scholar] [CrossRef]
- Adebo, O.A. Metabolomics, Physicochemical properties and mycotoxin reduction of whole grain ting (a Southern African fermented food) produced via natural and lactic acid bacteria (LAB) fermentation. Ph.D. Thesis, University of Johannesburg, Johannesburg, South Africa, October 2018. [Google Scholar]
- Adebo, O.A.; Kayitesi, E.; Njobeh, P.B. Reduction of mycotoxins during the fermentation of whole grain sorghum to whole grain ting (a Southern African food). Toxins 2019, 11, 180. [Google Scholar] [CrossRef]
- Prakash, J. Safety of fermented cereals and legumes. In Regulating Safety of Traditional and Ethnic Foods; Prakash, V., Martin-Belloso, O., Keener, L., Astley, S., Braun, S., McMahon, H., Lelieveld, H., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 283–310. [Google Scholar] [CrossRef]
- Correia, I.; Nunes, A.; Guedes, S.; Barros, A.S.; Delgadillo, I. Screening of lactic acid bacteria potentially useful for sorghum fermentation. J. Cereal. Sci. 2010, 52, 9–15. [Google Scholar] [CrossRef]
- Towo, E.; Matuschek, E.; Svanberg, U. Fermentation and enzyme treatment of tannin sorghum gruels: Effects on phenolic compounds, phytate and in vitro accessible iron. Food Chem. 2006, 94, 369–376. [Google Scholar] [CrossRef]
- Dlamini, N.R.; Taylor, J.R.N.; Rooney, L.W. The effect of sorghum type and processing on the antioxidant properties of African sorghum-based foods. Food Chem. 2007, 105, 1412–1419. [Google Scholar] [CrossRef]
- Curiel, J.A.; Rodríguez, H.; Acebrón, I.; Mancheño, J.M.; de las Rivas, B.; Muñoz, R. Production and physicochemical properties of recombinant Lactobacillus plantarum tannase. J. Agric. Food Chem. 2009, 57, 6224–6230. [Google Scholar] [CrossRef] [PubMed]
- Svensson, L.; Sekwati-Monang, B.; Lutz, D.L.; Schieber, A.; Gänzle, M.G. Phenolic acids and flavonoids in nonfermented and fermented red sorghum (Sorghum bicolor (L.) Moench). J. Agric. Food Chem. 2010, 58, 9214–9220. [Google Scholar] [CrossRef]
- Adebo, O.A.; Njobeh, P.B.; Kayitesi, E. Fermentation by Lactobacillus fermentum strains (singly and in combination) enhances the properties of ting from two whole grain sorghum types. J. Cereal. Sci. 2018, 82, 49–56. [Google Scholar] [CrossRef]
- Odunfa, S.A.; Oyewole, O.B. African fermented foods. In Microbiology of Fermented Foods; Wood, B.J.B., Ed.; Springer: Boston, MA, USA, 1998; pp. 713–752. [Google Scholar] [CrossRef]
- Anagnostopoulos, D.A.; Tsaltas, D. Fermented foods and beverages. In Innovations in Traditional Foods; Galanakis, C., Ed.; Woodhead Publishing: Cambridge, UK, 2019; pp. 257–291. [Google Scholar] [CrossRef]
- Rosentrater, K.A.; Evers, A.D. Other food products. In Kent’s Technology of Cereals, 5th ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 699–727. [Google Scholar] [CrossRef]
- Kunene, N.F.; Hastings, J.W.; von Holy, A. Bacterial populations associated with a sorghum-based fermented weaning cereal. Int. J. Food Microbiol. 1999, 49, 75–83. [Google Scholar] [CrossRef]
- Yousif, N.M.K.; Dawyndt, P.; Abriouel, H.; Wijaya, A.; Schillinger, U.; Vancanneyt, M.; Swings, J.; Dirar, H.A.; Holzapfel, W.H.; Franz, C.M.A.P. Molecular characterization, technological properties and safety aspects of enterococci from ‘Hussuwa’, an African fermented sorghum product. J. Appl. Microbiol. 2005, 98, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Adinsi, L.; Mestres, C.; Akisoé, N.; Vieira-Dalodé, G.; Anihouvi, V.; Durand, N.; Hounhouigan, D.J. Comprehensive quality and potential hazards of gowe, a malted and fermented cereal beverage from West Africa. A diagnostic for a future re-engineering. Food Cont. 2017, 82, 18–25. [Google Scholar] [CrossRef]
- Simango, C.; Rukure, G. Survival of Campylobacter jejuni and pathogenic Escherichia coli in mahewu, a fermented cereal gruel. Trans. R. Soc. Trop. Med. Hyg. 1991, 85, 399–400. [Google Scholar] [CrossRef]
- Nyatoti, V.N.; Mtero, S.; Rukure, G. Pathogenic Escherichia coli in traditional African weaning foods. Food Cont. 1997, 8, 51–54. [Google Scholar] [CrossRef]
- Byakika, S.; Mukisa, I.V.; Byaruhanga, Y.B.; Male, D.; Muyanja, C. Influence of food safety knowledge, attitudes and practices of processors on microbiological quality of commercially produced traditional fermented cereal beverages, a case of Obushera in Kampala. Food Cont. 2019, 100, 212–219. [Google Scholar] [CrossRef]
- Adekoya, I.; Njobeh, P.; Obadina, O.; Chilaka, C.; Okoth, S.; De Boevre, M.; De Saeger, S. Awareness and prevalence of mycotoxin contamination in selected Nigerian fermented foods. Toxins 2017, 9, 363. [Google Scholar] [CrossRef] [PubMed]
- Chilaka, C.A.; De Boevre, M.; Atanda, O.O.; De Saeger, S. Quantification of Fusarium mycotoxins in Nigerian traditional beers and spices using a multi-mycotoxin LC-MS/MS method. Food Cont. 2018, 87, 203–210. [Google Scholar] [CrossRef]
- Turbic, A.; Ahokas, J.T.; Haskard, C.A. Selective in vitro binding of dietary mutagens, individually or in combination, by lactic acid bacteria. Food Addit. Contam. 2002, 19, 144–152. [Google Scholar] [CrossRef]
- Zhou, X.W.; Liu, H.F.; Zhao, X.H. The potencies of three microorganisms to dissipate four organophosphorus pesticides in three food materials during traditional fermentation. J. Food Sci. Technol. 2015, 52, 7353–7360. [Google Scholar] [CrossRef]
- Trinder, M.; McDowell, T.W.; Daisley, B.A.; Ali, S.N.; Leong, H.S.; Sumarah, M.W.; Reid, G. Probiotic Lactobacillus rhamnosus reduces organophosphate pesticide absorption and toxicity to Drosophila melanogaster. Appl. Environ. Microbiol. 2016, 82, 6204–6213. [Google Scholar] [CrossRef]
- Pinto, G.D.A.; Castro, I.M.; Miguel, M.A.L.; Koblitz, M.G.B. Lactic acid bacteria - Promising technology for organophosphate degradation in food: A pilot study. LWT-Food Sci. Technol. 2019, 353–359. [Google Scholar] [CrossRef]
- Zhai, Q.; Guo, Y.; Tang, X.; Tian, F.; Zhao, J.; Zhang, H. Removal of cadmium from rice by Lactobacillus plantarum fermentation. Food Cont. 2019, 96, 357–364. [Google Scholar] [CrossRef]
- Shukla, S.; Park, H.-K.; Lee, J.-S.; Kim, J.-K.; Kim, M. Reduction of biogenic amines and aflatoxins in doenjang samples fermented with various meju as starter cultures. Food Cont. 2014, 42, 181–187. [Google Scholar] [CrossRef]
- De Angelis, M.; Campanella, D.; Cosmai, L.; Summo, C.; Rizzello, C.G.; Caponio, F. Microbiota and metabolome of un-started and started Greek-type fermentation of Bella di Cerignola table olives. Food Microbiol. 2015, 52, 18–30. [Google Scholar] [CrossRef]
- Sanchart, C.; Rattanaporn, O.; Haltrich, D.; Phukpattaranont, P.; Maneerat, S. Enhancement of gamma-aminobutyric acid (GABA) levels using an autochthonous Lactobacillus futsaii CS3 as starter culture in Thai fermented shrimp (Kung-Som). World J. Microbiol. Biotechnol. 2017, 33, 152. [Google Scholar] [CrossRef] [PubMed]
- Oria, M.P.; Hamaker, B.R.; Axtell, J.D.; Huang, C.-P. A highly digestible sorghum mutant cultivar exhibits unique folded structure of endosperm protein bodies. Proc. Natl. Acad. Sci. USA 2000, 97, 5065–5070. [Google Scholar] [CrossRef] [PubMed]
- Elkonin, L.; Italyanskaya, J.; Panin, V. Genetic modification of sorghum for improved nutritional value: State of the problem and current approaches. J. Investig. Genom. 2018, 5, 39–48. [Google Scholar] [CrossRef]
- Latham, M.C. Human Nutrition in the Developing World; FAO Food and Nutrition Series; FAO: Rome, Italy, 1997. [Google Scholar]
- Temba, M.C.; Njobeh, P.B.; Adebo, O.A.; Olugbile, A.O.; Kayitesi, E. The role of compositing cereals with legumes to alleviate protein energy malnutrition in Africa. Int. J. Food Sci. Technol. 2016, 51, 543–554. [Google Scholar] [CrossRef]
- Kashiri, M.; Garmakhany, A.D.; Dehghani, A.A. Modelling of sorghum soaking using artificial neural networks (MLP). Qual. Assur. Saf. Crops. 2012, 4, 179–184. [Google Scholar] [CrossRef]
- Kaliba, A.R.; Mushi, R.J.; Gongwe, A.G.; Mazvimavi, K. A typology of adopters and nonadopters of improved sorghum seeds in Tanzania: A deep learning neural network approach. World Dev. 2020, 127, 104839. [Google Scholar] [CrossRef]
- Sebayang, A.H.; Masjuki, H.H.; Ong, H.C.; Dharma, S.; Silitonga, A.S.; Kusumo, F.; Milano, J. Optimization of bioethanol production from sorghum grains using artificial neural networks integrated with ant colony. Ind. Crop. Prod. 2017, 97, 146–155. [Google Scholar] [CrossRef]
- Zhou, L.; Zhang, C.; Liu, F.; Qiu, Z.; He, Y. Application of deep learning in food: A review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 1793–1811. [Google Scholar] [CrossRef]
- Yordi, E.G.; Koelig, R.; Matos, M.J.; Martinez, A.P.; Caballero, Y.; Santana, L.; Quintana, M.P.; Molina, E.; Uriarte, E. Artificial intelligence applied to flavonoid data in food matrices. Foods 2019, 8, 573. [Google Scholar] [CrossRef]
- Huang, Y.; Kangas, L.J.; Rasco, B.A. Applications of artificial neural networks (ANNs) in food science. Crit. Rev. Food Sci. Nutr. 2007, 47, 113–126. [Google Scholar] [CrossRef]
- Goel, L.; Raman, S.; Dora, S.S.; Bhutani, A.; Aditya, A.S.; Mehta, A. Hybrid computational intelligence algorithms and their applications to detect food quality. Artif. Intell. Rev. 2020, 53, 1415–1440. [Google Scholar] [CrossRef]
- Marwala, T.; Hurwitz, E. Introduction to man and machines. In Artificial Intelligence and Economic Theory: Skynet in the Market; Marwala, T., Hurwitz, E., Eds.; Springer: Cham, Switzerland, 2017; pp. 1–13. [Google Scholar] [CrossRef]
- IFT (Institute of Food Technologists). Getting smart about artificial intelligence. Food Technol. Mag. 2018, 72, 27. [Google Scholar]
- Zagorec, M.; Chaillou, S.; Champomier-Vergès, M.C.; Coq, A.M.C.L. Role of bacterial ‘omics’ in food fermentation. In Molecular Techniques in the Microbial Ecology of Fermented Foods; Cocolin, L., Ercolini, D., Eds.; Springer: New York, NY, USA, 2008; pp. 255–273. [Google Scholar] [CrossRef]
- Alkema, W.; Boekhorst, J.; Wels, M.; van Hijum, S.A.F.T. Microbial bioinformatics for food safety and production. Brief Bioinform. 2016, 17, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Chen, C.; Lei, Z. Meta-omics insights in the microbial community profiling and functional characterization of fermented foods. Trends Food Sci. Technol. 2017, 65, 23–31. [Google Scholar] [CrossRef]
- Weckx, S.; Van Kerrebroeck, S.; De Vuyst, L. Omics approaches to understand sourdough fermentation processes. Int. J. Food Microbiol. 2018, 302, 90–102. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, F.; Parente, E.; Ercolini, D. Metagenomics insights into food fermentations. Microb Biotechnol. 2017, 10, 91–102. [Google Scholar] [CrossRef]
- McRae, M.P. Health benefits of dietary whole grains: An umbrella review of meta-analyses. J. Chiropr. Med. 2017, 16, 10–18. [Google Scholar] [CrossRef]
- Schaffer-Lequart, C.; Lehmann, U.; Ross, A.B.; Roger, O.; Eldridge, A.L.; Ananta, E.; Bietry, M.F.; King, L.R.; Moroni, A.V.; Srichuwong, S.; et al. Whole grain in manufactured foods: Current use, challenges and the way forward. Crit. Rev. Food Sci. Nutr. 2017, 57, 1562–1568. [Google Scholar] [CrossRef] [PubMed]
- Links, M.R.; Taylor, J.; Kruger, M.C.; Taylor, J.R. Sorghum condensed tannins encapsulated in kafirin microparticles as a nutraceutical for inhibition of amylases during digestion to attenuate hyperglycaemia. J. Funct. Foods 2015, 12, 55–63. [Google Scholar] [CrossRef]
- García-Gurrola, A.; Rincón, S.; Escobar-Puentes, A.A.; Zepeda, A.; Martínez-Bustos, F. Microencapsulation of red sorghum phenolic compounds with esterified sorghum starch as encapsulant materials by spray drying. Food Technol. Biotechnol. 2019, 57, 341–349. [Google Scholar] [CrossRef]
- Khan, M.A.; Fang, Z.; Wusigale; Cheng, H.; Gao, Y.; Deng, Z.; Liang, L. Encapsulation and protection of resveratrol in kafirin and milk protein nanoparticles. Int. J. Food Sci. Technol. 2019, 54, 2998–3007. [Google Scholar] [CrossRef]
- Vallons, K.J.R.; Ryan, L.A.M.; Koehler, P.; Arendt, E.K. High pressure–treated sorghum flour as a functional ingredient in the production of sorghum bread. Euro. Food Res. Technol. 2010, 231, 711–717. [Google Scholar] [CrossRef]
- Correia, I.; Nunes, A.; Saraiva, J.A.; Barros, A.S.; Delgadillo, I. High pressure treatments largely avoid/revert decrease of cooked sorghum protein digestibility when applied before/after cooking. LWT-Food Sci. Technol. 2011, 4, 1245–1249. [Google Scholar] [CrossRef]
- Noreña, C.P.Z.; Lescano-Anadón, C.E. Dielectric properties of importance in operations of post-harvest of sorghum. Int. J. Food Eng. 2017, 13, 20160369. [Google Scholar] [CrossRef]
- Audu, J.; Ijabo, O.J.; Awulu, J.O. Influence of moisture and current frequency on electrical potential of sorghum grains (Sorghum bicolour (l.) Moench). Hungarian. Agric. Eng. 2018, 33, 24–29. [Google Scholar] [CrossRef]
- Lohani, U.C.; Muthukumarappan, K. Application of the pulsed electric field to release bound phenolics in sorghum flour and apple pomace. Innov. Food Sci. Emerg. Technol. 2016, 35, 29–35. [Google Scholar] [CrossRef]
- Corey, M. Developing functional food products through novel processing, ingredient, and shelf stability evaluation. Ph.D. Thesis, University of Georgia, Athens, GA, USA, 2009. [Google Scholar]
- Khan, M.K.; Ahmad, K.; Hassan, S.; Imran, M.; Ahmad, N.; Xu, C. Effect of novel technologies on polyphenols during food processing. Innov. Food Sci. Emerg. Technol. 2018, 45, 361–381. [Google Scholar] [CrossRef]

| Proximate composition (%) | Jones and Beckwith [34] | Okoh et al. [35] | Adebiyi et al. [36] | Shawrang et al. [37] | Shargie [38] | Udachan et al. [39] | Awadelkareem et al. [40] | Ndimba et al. [41] | Singh et al. [42] | Ape et al. [43] | Jimoh and Abdullahi [44] | Mohapatra et al. [45] |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ash | 1.2–1.3 | 0.90–1.52 | 1.98 | 4.20 | 1.44 | 0.92–1.75 | 1.28–1.78 | 1.61–2.03 | 1.90 | 2.07 | 1.12–1.68 | 3.17 |
| CHO | NR | 71.80–85.20 | 72.41 | NR | NR | 70.65–76.20 | 72.44–77.28 | NR | NR | 76.51 | 65.15–76.28 | 71.95 |
| Fat | 3.1–3.4 | 1.38–4.50 | 3.35 | 6.9 | 3.32 | 2.30–2.80 | 2.84–3.02 | 2.37–2.75 | 3.30 | 3.10 | 5.12–10.54 | 4.70 |
| Fiber | 1.8–1.9 | 1.47–2.45 | 2.25 | 19.5 | 1.83 | 1.40–2.70 | 1.72–2.02 | NR | 1.7 | 2.86 | 1.65–7.94 | 2.76 |
| Moisture | NR | NR | 10.66 | 8.1–8.5 | NR | 8.10–9.99 | 6.67–7.29 | 8.95–11.16 | 9.80 | 6.36 | 1.39–19.02 | 6.07 |
| Protein | 11.5–11.7 | 9.28–14.86 | 9.35 | 11.80 | 9.95 | 8.90–11.02 | 10.21–13.45 | 11.90–12.82 | 12.5 | 9.10 | 6.23–13.81 | 11.36 |
| Reference | Ala | Arg | Asp | Cys | Glu | Gly | His | Ile | Leu | Lys | Met | Phe | Pro | Ser | Thr | Trp | Tyr | Val |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Awadelkareem et al. [40] | 8.69–8.70 | 2.79–3.61 | 4.83–5.28 | NR | 17.50–19.57 | 2.36–3.08 | 1.75–1.77 | 3.61–3.72 | 12.48–13.48 | 1.57–2.31 | 1.55–1.88 | 4.73–5.10 | 7.70–8.16 | 3.77–3.85 | 2.48–2.64 | NR | 3.72–3.75 | 4.65–4.74 |
| Khalil et al. [50] | 9.0–9.2 | 3.3 | 6.5–6.8 | 1.2 | 21.6–22 | 3.0 | 1.7–1.8 | 4.0–4.1 | 13.6–13.8 | 2.5–2.6 | 1.3–1.4 | 5.1–5.2 | 9.4–10.7 | 4.3–4.4 | 3.2 | 0.9 | 2.9–3.0 | 4.8–4.9 |
| Salunkhe et al. [52] | 7.34–9.62 | 3.20–4.68 | 4.98–6.66 | 1.94–3.06 | 23.42–28.12 | 3.12–4.12 | 1.46–2.46 | 3.92–4.86 | 12.02–14.48 | 1.42–2.72 | 1.36–2.34 | 4.03–5.62 | 8.92–12.34 | 3.92–5.66 | 1.92–2.42 | 0.49–1.16 | 2.12–3.62 | 5.12–6.86 |
| Afify et al. [53] | 7.43–8.83 | 3.58–4.01 | 6.24–7.06 | 1.69–2.11 | 18.45–20.63 | 2.84–3.05 | 1.93–2.17 | 3.49–3.85 | 11.74–13.56 | 2.11–2.26 | 2.73–2.94 | 4.40–4.98 | 6.66–8.99 | 3.49–4.17 | 2.75–3.21 | NR | 4.22–4.33 | 4.22–4.82 |
| Reference | Ca | Cd | Co | Cr | Cu | Fe | I | K | Mg | Mn | Na | Ni | P | Pb | Se | Zn |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mabelele et al. [54] | 100.1–121.3 | NR | NR | NR | 1.9–2.4 | 24.3–40.3 | ND | 2751–3524 | 1130–1440 | 14.2–20.2 | 20–40 | NR | 2210–3327 | NR | NR | 16.2–24.2 |
| Shegro et al. [55] | 204.50–447.50 | NR | NR | NR | NR | 41.17–127.50 | NR | 1150–2568.75 | NR | 9.5–23.83 | 11.5–54.38 | NR | 1498–3787.25 | NR | NR | 13.5–34.67 |
| Pontieri et al. [56] | 233.84–411.83 | 9.92–60.54 | 7.12–15.24 | 121.59–254.18 | NR | 39.36–77.03 | 14.81–212.70 | 3434.46–6957.67 | 1454.92–2862.00 | 8.93–19.44 | 455.09–840.69 | 0.46–1.27 | 2148.60–2963.40 | 92.62–303.89 | 2.98–14.13 | 21.10–47.05 |
| Gerrano et al. [57] | 44.57–477.04 | NR | NR | NR | NR | 13.50–55.13 | NR | 900–3366 | 854–1631.17 | 11.17–20.17 | 12.50–62.03 | NR | 2042.19–3775 | NR | NR | 12–44.83 |
| Vitamins | Khalil et al. [50] | Serna-Saldivar and Espinosa-Ramirez [58] | Kulamarva et al. [59] | Ochanda et al. [60] | Saleh et al. [61] |
|---|---|---|---|---|---|
| B1 | 0.69–0.73 | 0.45 | NR | 0.34–0.35 | 0.38 |
| B2 | 0.12–0.14 | 0.16 | 0.13 | 0.15–0.16 | 0.15 |
| B3 | 2.99–3.01 | 4.88 | 4.5 | 4.20–4.55 | 4.3 |
| B5 | 1.55–1.63 | NR | NR | NR | NR |
| B6 | 0.40–0.43 | 0.59 | 0.47 | 0.17–0.35 | NR |
| B9 | 0.02 | 0.02 | NR | 0.02 | NR |
| Product Name | Country/Region | Product Use | Microorganism Identified | Reference |
|---|---|---|---|---|
| Aceda | Sudan | Porridge | Unknown | Eggum et al. [99]; Franz & Holzapfel [100] |
| Burukutu | West Africa | Alcoholic beverage | Acetobacter spp., Candida spp., Enterobacter spp., Lactobacillus spp., Saccharomyces cerevisiae, S. chavelieri, Leuconostoc mesenteroides | Kolawole et al. [101]; Eze et al. [102]; Alo et al. [103]; Blandino et al. [104] |
| Bushera | Uganda | Beverage | Lactobacillus brevis, L. delbrueckii, L. paracasei, L. plantarum | Marsh et al. [105]; Mwale [106] |
| Chibuku | Zimbabwe | Alcoholic beverage | Lactobacillus spp. | Togo et al. [107]; Gadaga et al. [108] |
| Dolo | Burkina Faso/Togo | Alcoholic beverage | L. delbrueckii, L. fermentum, L. lactis, Pediococcus acidilactici, S. cerevisae | Van der Aa Kühle et al. [109]; Sawadogo-Lingani et al. [110] |
| Enturire | Uganda | Alcoholic beverage | L. plantarum, S. cerevisae, Weissela confusa | Mukisa et al. [111] |
| Gowe | Benin | Porridge | L. fermentum, L. mucosae | Adinsi et al. [112]; Vieira-Dalodé et al. [113] |
| Humulur | Sudan | Gruel | Bacillus spp., Lactobacillus spp., yeasts | Adams [114] |
| Hussuwa | Sudan | Porridge | A. xylinum, Gluconobacter oxydans, L. fermentum, L. saccharolyticum, Pediococcus acidilactici, S. cerevisiae | Mwale [106]; Yousif et al. [115] |
| Ikigage | Rwanda | Alcoholic beverage | Issatchenkia orientalis, L. buchneri, L. fermentum, Lactobacillus spp., S. cerevisiae | Lyumugabe et al. [116] |
| Injera | Ethiopia | Sourdough/bread | C. guillermondii, Lactobacillus spp, yeasts | Dandessa et al. [117] |
| Kisra | Sudan | Pancake, flat bread, sourdough | C. intermedia, C. krusei, Debrayomyces hansenii, Enterococcus faecium, L. amylovorus, L. brevis, L. confusus, L. fermentum, Pichia kudriavzevii | Mohammed et al. [118]; Hamad et al. [119]; Ali & Mustafa [120] |
| Khamir | Sudan | Bread | L. brevis, L. cellobiosus | Gassem [121] |
| Kunun-zaki | Nigeria | Beverage, breakfast meal | Ent. faecalis, Lactobacillus spp., P. pentosaceus, W. confusa | Franz & Holzapfel [100] |
| Mahewu | South Africa | Porridge gruel | L. brevis, L. bulgaricus, L. delbruckii, Leuconostoc spp., Streptococcus lactis | Franz & Holzapfel [100]; Hesseltine [122]; Kayitesi et al. [123] |
| Mbege | Tanzania | Beverage | L. plantarum, Leuc. mesenteroides, S. cerevisiae, Schizosaccharomyces pombe | Odunfa & Oyewole [124] |
| Merissa | Sudan | Alcoholic drink | Saccharomyces spp. | Dirar [125,126] |
| Nasha | Sudan | Infant food | Candida spp., Lactobacillus spp., S. cerevisiae, Strep. spp. | Graham et al. [127] |
| Ogi | West Africa | Gruel | L. acidophilus, L. agilis, L. cellobiosus, L. confusus, L. murinus, L. plantarum | Graham et al. [127]; Omemu & Bankole [128] |
| Ori-ese | Nigeria | Porridge | Bacillus subtilis, C. tropicalis, L. acidophilus, L. fermentum, L. plantarum, Mucor spp., Pediococcus spp., Penicillium spp., S. pombe | Adebayo-Tayo & Needum [129] |
| Orubisi | Tanzania | Alcoholic beverage | LABs, yeasts | Shayo et al. [130] |
| Otika | Nigeria | Alcoholic beverage | B. cereus, B. subtilis, C. krusei, C. tropicalis, Enterobacter clocae, L. brevis,L. fermentum, L. plantarum, Leuconostoc mesenteroides, S. cerevisae | Oriola et al. [131] |
| Pito | Nigeria | Alcoholic beverage | B. subtillis, Candida spp., Geotrichum candidum, L. delbrueckii, L. fermentum | Kolawole et al. [101], Sawadogo-Lingani et al. [132]; Ajiboye et al. [133] |
| Tella | Ethiopia | Beverage | L. pastorianumi, S. cerevisae | Lemi [134] |
| Tchapalo | Ivory Coast | Alcoholic beverage | L. brevis, L. cellobiosus, L. coprophilus, L. fermentum, L. hilgardii, L. plantarum | Djè et al. [135]; N’guessan et al. [136] |
| Tchoukoutou | Benin | Alcoholic beverage | L. divergens, L. fermentum, L. fructivorans, S. cerevisae, S. pastorianus, Torulasposa delbrueckii | Kayodé et al. [137,138] |
| Ting | Botswana, South Africa | Porridge | L. casei, L. coryniformis, L. curvatus, L. fermentum, L. harbinensis, L. parabuchneri, L. plantarum, L. reuteri, L. rhamnosus | Madoroba et al. [139,140]; Sekwati-Monang & Gänzle [141] |
| Thobwa | Malawi | Alcoholic beverage | Unknown | Nyanzi & Jooste [98]; Matumba et al. [142] |
| Uji | East Africa | Porridge | L. cellobiosus, L. fermentum, L. plantarum, Ped. acidilactici, Ped. pentosaceus | Blandino et al. [104] |
| Umqombothi | Southern Africa | Beverage | Lactobacillus spp. | Katongole [143] |
| Weaning food | Nigeria | Weaning food | L. plantarum, Ped. acidilactici, S. cerevisae | Wakil & Kazeem [144] |
| Food | Safety risk | Probable Source | Reference |
|---|---|---|---|
| Fermented sorghum meal | Food pathogens | B. cereus, Clostridium perfringes, Escherichia coli, Listeria monocytogenes | Kunene et al. [164] |
| Hussuwa | Hygienic risk, antimicrobial resistances, biogenic amines, presence of virulence determinants | Enterococci | Yousif et al. [165] |
| Gowe | Cyanogenic compounds, food pathogens, mycotoxins | E. coli, Enterobacteriacae, mycotoxigenic fungi | Adinsi et al. [166] |
| Ikigage | Food pathogens | E. coli, Streptococci | Lyumugabe et al. [116] |
| Mahewu | Food pathogens | E. coli | Simango and Rukure [167]; Nyatoti et al. [168] |
| Obushera | Food pathogens | E. coli, Staphylococcus | Byakika et al. [169] |
| Ogi | Mycotoxins | Mycotoxigenic fungi | Adekoya et al. [170] |
| Pito | Mycotoxins | Mycotoxigenic fungi | Ezekiel et al. [148]; Chilaka et al. [171] |
| Thobwa | Mycotoxins | Mycotoxigenic fungi | Matumba et al. [142] |
| Ting | Mycotoxins | Mycotoxigenic fungi | Adebo et al. [153] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adebo, O.A. African Sorghum-Based Fermented Foods: Past, Current and Future Prospects. Nutrients 2020, 12, 1111. https://doi.org/10.3390/nu12041111
Adebo OA. African Sorghum-Based Fermented Foods: Past, Current and Future Prospects. Nutrients. 2020; 12(4):1111. https://doi.org/10.3390/nu12041111
Chicago/Turabian StyleAdebo, Oluwafemi Ayodeji. 2020. "African Sorghum-Based Fermented Foods: Past, Current and Future Prospects" Nutrients 12, no. 4: 1111. https://doi.org/10.3390/nu12041111
APA StyleAdebo, O. A. (2020). African Sorghum-Based Fermented Foods: Past, Current and Future Prospects. Nutrients, 12(4), 1111. https://doi.org/10.3390/nu12041111





