Dietary Patterns Based on Estimated Glomerular Filtration Rate and Kidney Function Decline in the General Population: The Lifelines Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Dietary Intake Assessment
2.3. Other Variables
2.4. Prospective Outcomes
2.5. Dietary Pattern Analysis
2.6. Statistical Analysis
3. Results
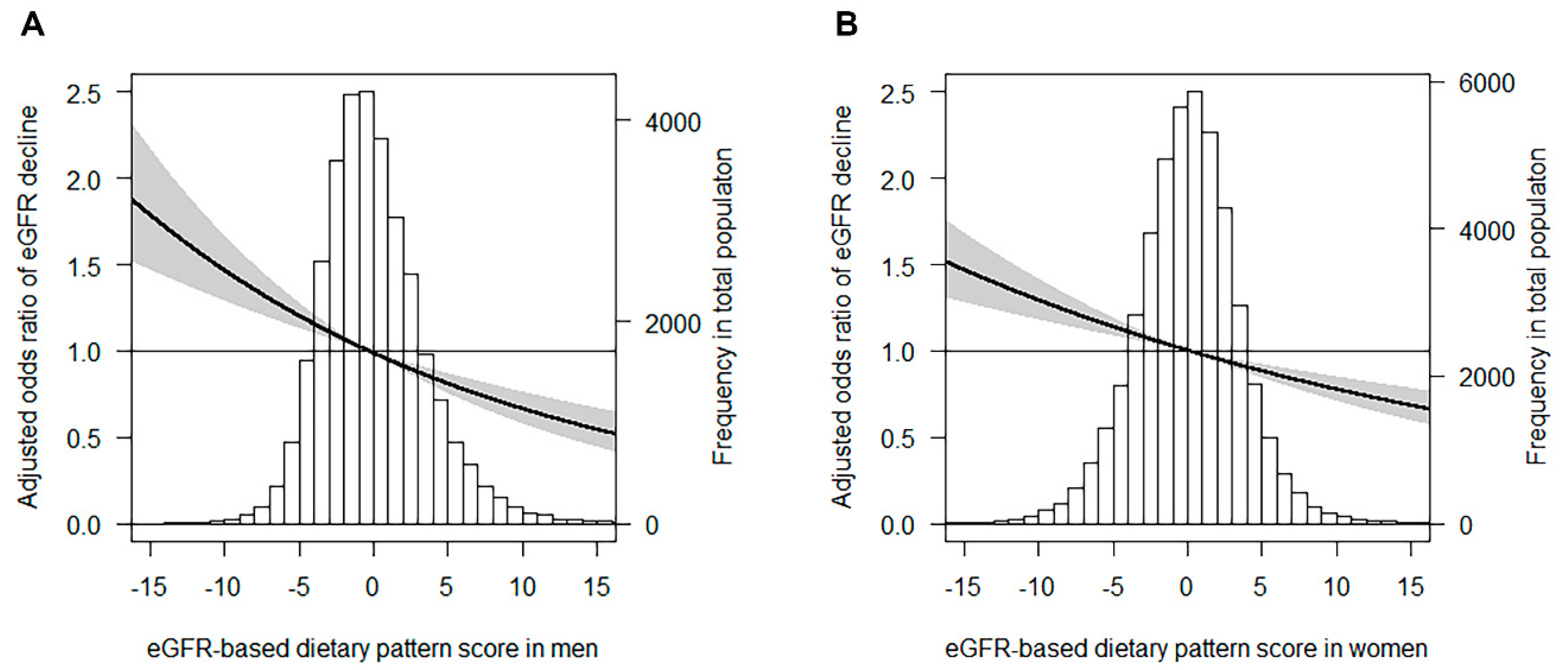
3.1. eGFR-Based Dietary Pattern (eGFR-DP)
3.2. Baseline Characteristics across the Quartiles of eGFR-DP Score
3.3. eGFR-DP Scores and Renal Outcomes
3.4. MDS and Renal Outcomes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kalantar-Zadeh, K.; Fouque, D. Nutritional Management of Chronic Kidney Disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef]
- Lin, J.; Hu, F.B.; Curhan, G.C. Associations of diet with albuminuria and kidney function decline. Clin. J. Am. Soc. Nephrol. 2010, 5, 836–843. [Google Scholar] [CrossRef]
- Yuzbashian, E.; Asghari, G.; Mirmiran, P.; Hosseini, F.-S.; Azizi, F. Associations of dietary macronutrients with glomerular filtration rate and kidney dysfunction: Tehran lipid and glucose study. J. Nephrol. 2014, 28, 173–180. [Google Scholar] [CrossRef]
- Herber-Gast, G.-C.M.; Boersma, M.; Verschuren, W.M.M.; Stehouwer, C.D.A.; Gansevoort, R.T.; Bakker, S.J.L.; Spijkerman, A.M.W. Consumption of whole grains, fruit and vegetables is not associated with indices of renal function in the population-based longitudinal Doetinchem study. Br. J. Nutr. 2017, 118, 375–382. [Google Scholar] [CrossRef]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Fung, T.T.; Hu, F.B.; Curhan, G.C. Association of Dietary Patterns with Albuminuria and Kidney Function Decline in Older White Women: A Subgroup Analysis from the Nurses’ Health Study. Am. J. Kidney Dis. 2011, 57, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Khatri, M.; Moon, Y.P.; Scarmeas, N.; Gu, Y.; Gardener, H.; Cheung, K.; Wright, C.B.; Sacco, R.L.; Nickolas, T.L.; Elkind, M.S. The Association between a Mediterranean-Style Diet and Kidney Function in the Northern Manhattan Study Cohort. Clin. J. Am. Soc. Nephrol. 2014, 9, 1868–1875. [Google Scholar] [CrossRef] [PubMed]
- Bach, K.E.; Kelly, J.T.; Palmer, S.C.; Khalesi, S.; Strippoli, G.F.M.; Campbell, K.L. Healthy Dietary Patterns and Incidence of CKD: A Meta-Analysis of Cohort Studies. Clin. J. Am. Soc. Nephrol. 2019, 14, 1441–1449. [Google Scholar] [CrossRef] [PubMed]
- Weikert, C.; Schulze, M.B. Evaluating dietary patterns. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, K.; Schulze, M.B.; Schienkiewitz, A.; Nöthlings, U.; Boeing, H. Application of a new statistical method to derive dietary patterns in nutritional epidemiology. Am. J. Epidemiol. 2004, 159, 935–944. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, S.A.; Mishra, G.D.; Brunner, E.J. Dietary Patterns, Insulin Resistance, and Incidence of Type 2 Diabetes in the Whitehall II Study. Diabetes Care 2008, 31, 1343–1348. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.D.; Weis, K.E.; Schulz, M.; Tooze, J.A. Food intake patterns associated with incident type 2 diabetes: The insulin resistance atherosclerosis study. Diabetes Care 2009, 32, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, K.; Zyriax, B.-C.; Boeing, H.; Windler, E. A dietary pattern derived to explain biomarker variation is strongly associated with the risk of coronary artery disease. Am. J. Clin. Nutr. 2004, 80, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Huybrechts, I.; Lioret, S.; Mouratidou, T.; Gunter, M.J.; Manios, Y.; Kersting, M.; Gottrand, F.; Kafatos, A.; De Henauw, S.; Cuenca-García, M.; et al. Using reduced rank regression methods to identify dietary patterns associated with obesity: A cross-country study among European and Australian adolescents. Br. J. Nutr. 2017, 117, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Scholtens, S.; Smidt, N.; Swertz, M.A.; Bakker, S.J.; Dotinga, A.; Vonk, J.M.; Van Dijk, F.; Van Zon, S.K.; Wijmenga, C.; Wolffenbuttel, B.H.R.; et al. Cohort Profile: LifeLines, a three-generation cohort study and biobank. Int. J. Epidemiol. 2014, 44, 1172–1180. [Google Scholar] [CrossRef] [PubMed]
- Siebelink, E.; Geelen, A.; De Vries, J.H.M. Self-reported energy intake by FFQ compared with actual energy intake to maintain body weight in 516 adults. Br. J. Nutr. 2011, 106, 274–281. [Google Scholar] [CrossRef]
- Steppel, M.T.; De Vries, J.H.; Meijboom, S.; Beekman, M.; De Craen, A.; Slagboom, P.E.; Feskens, E.J. Relative validity of the food frequency questionnaire used to assess dietary intake in the Leiden Longevity Study. Nutr. J. 2013, 12, 75. [Google Scholar] [CrossRef]
- Goldberg, G.R.; Black, A.E.; Jebb, S.A.; Cole, T.J.; Murgatroyd, P.R.; Coward, W.A.; Prentice, A.M. Critical evaluation of energy intake data using fundamental principles of energy physiology: 1. Derivation of cut-off limits to identify under-recording. Eur. J. Clin. Nutr. 1991, 45, 569–581. [Google Scholar]
- Black, A. Critical evaluation of energy intake using the Goldberg cut-off for energy intake:basal metabolic rate. A practical guide to its calculation, use and limitations. Int. J. Obes. 2000, 24, 1119–1130. [Google Scholar] [CrossRef]
- Slagter, S.N.; Byambasukh, O.; Van Der Klauw, M.M.; Sijtsma, A.; Swart-Busscher, L.G.; Perenboom, C.W.M.; De Vries, J.H.; Feskens, E.J.; Wolffenbuttel, B.H.R.; Kromhout, D.; et al. Dietary patterns and physical activity in the metabolically (un)healthy obese: The Dutch Lifelines cohort study. Nutr. J. 2018, 17, 18. [Google Scholar] [CrossRef]
- Dekker, L.; Rijnks, R.H.; Strijker, D.; Navis, G. A spatial analysis of dietary patterns in a large representative population in the north of The Netherlands—The Lifelines cohort study. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 166. [Google Scholar] [CrossRef]
- Slagter, S.N.; Van Vliet-Ostaptchouk, J.V.; Vonk, J.M.; Boezen, H.M.; Dullaart, R.P.F.; Kobold, A.C.M.; Feskens, E.J.; Van Beek, A.P.; Van Der Klauw, M.M.; Wolffenbuttel, B.H.R. Combined Effects of Smoking and Alcohol on Metabolic Syndrome: The LifeLines Cohort Study. PLoS ONE 2014, 9, e96406. [Google Scholar] [CrossRef]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A New Equation to Estimate Glomerular Filtration Rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Schulze, M.B.; Hoffmann, K.; Kroke, A.; Boeing, H. An approach to construct simplified measures of dietary patterns from exploratory factor analysis. Br. J. Nutr. 2003, 89, 409–418. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean Diet and Survival in a Greek Population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Rebholz, C.M.; Coresh, J.; Grams, M.E.; Steffen, L.M.; Anderson, C.A.; Appel, L.J.; Crews, D.C. Dietary Acid Load and Incident Chronic Kidney Disease: Results from the ARIC Study. Am. J. Nephrol. 2016, 42, 427–435. [Google Scholar] [CrossRef]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A Clinical Trial of the Effects of Dietary Patterns on Blood Pressure. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef]
- Lentine, K.L.; Wrone, E.M. New insights into protein intake and progression of renal disease. Curr. Opin. Nephrol. Hypertens. 2004, 13, 333–336. [Google Scholar] [CrossRef]
- Knight, E.L.; Stampfer, M.J.; Hankinson, S.E.; Spiegelman, N.; Curhan, G.C. The impact of protein intake on renal function decline in women with normal renal function or mild renal insufficiency. Ann. Intern. Med. 2003, 138, 460–467. [Google Scholar] [CrossRef]
- Poortmans, J.R.; Dellalieux, O. Do Regular High Protein Diets Have Potential Health Risks on Kidney Function in Athletes? Int. J. Sport Nutr. Exerc. Metab. 2000, 10, 28–38. [Google Scholar] [CrossRef]
- Martin, W.F.; Armstrong, L.E.; Rodriguez, N.R. Dietary protein intake and renal function. Nutr. Metab. 2005, 2, 25. [Google Scholar] [CrossRef] [PubMed]
- Said, M.Y.; Deetman, P.E.; De Vries, A.; Zelle, D.M.; Gans, R.O.B.; Navis, G.; Joosten, M.; Bakker, S.J.L. Causal path analyses of the association of protein intake with risk of mortality and graft failure in renal transplant recipients. Clin. Transplant. 2015, 29, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Lew, Q.-L.J.; Jafar, T.H.; Koh, H.W.L.; Jin, A.; Chow, K.Y.; Yuan, J.-M.; Koh, W.-P. Red Meat Intake and Risk of ESRD. J. Am. Soc. Nephrol. 2016, 28, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Azadbakht, L.; Atabak, S.; Esmaillzadeh, A. Soy Protein Intake, Cardiorenal Indices, and C-Reactive Protein in Type 2 Diabetes With Nephropathy: A longitudinal randomized clinical trial. Diabetes Care 2008, 31, 648–654. [Google Scholar] [CrossRef]
- Adeva-Andany, M.M.; Souto, G. Diet-induced metabolic acidosis. Clin. Nutr. 2011, 30, 416–421. [Google Scholar] [CrossRef]
- Metges, C.C.; Barth, C.A. Metabolic consequences of a high dietary-protein intake in adulthood: Assessment of the available evidence. J. Nutr. 2000, 130, 886–889. [Google Scholar] [CrossRef]
- Epstein, F.H.; Brenner, B.M.; Meyer, T.W.; Hostetter, T.H. Dietary Protein Intake and the Progressive Nature of Kidney Disease. N. Engl. J. Med. 1982, 307, 652–659. [Google Scholar] [CrossRef]
| Quartiles of Dietary Pattern Score in Women 2 (n = 45,746) (g/Day) | ||||||
|---|---|---|---|---|---|---|
| Food Groups | Factor Loading 1 | 1 | 2 | 3 | 4 | P-Trend |
| High intake | ||||||
| Eggs | 0.42 | 7.2 (4.5–14.3) | 7.2 (4.5–17.9) | 7.2 (4.5–17.9) | 17.9 (7.2–32.2) | <0.001 |
| Low-fat cheese | 0.23 | 0 (0–4.6) | 0.4 (0–8.2) | 2.0 (0–11.8) | 5.1 (0–17.9) | <0.001 |
| High-fat cheese | 0.23 | 11.9 (4.6–22.4) | 14.3 (5.9–26.1) | 16.3 (6.5–30.5) | 22.5 (8.5–42.8) | <0.001 |
| Legumes | 0.20 | 0 (0–6.6) | 4.4 (0–11.0) | 4.4 (0–11.0) | 11.0 (0–17.6) | <0.001 |
| Low intake | ||||||
| Sweetened dairy drinks | −0.27 | 101.6 (53.6–174.6) | 80.3 (40.4–139.4) | 58.8 (23.4–104.8) | 40.1 (8.9–83.9) | <0.001 |
| Desserts | −0.26 | 13.4 (3.4–39.9) | 8.3 (0–21.0) | 3.4 (0–8.3) | 3.4 (0–8.3) | <0.001 |
| Cakes and cookies | −0.24 | 46.3 (30.4–66.4) | 35.3 (23.1–49.9) | 27.8 (17.4–40.8) | 20.4 (11.6–33.5) | <0.001 |
| Sweet sandwich toppings | −0.22 | 19.3 (9.7–28.1) | 10.8 (2.7–19.4) | 5.4 (1.3–14.0) | 2.2 (0–9.7) | <0.001 |
| White meat | −0.22 | 12.4 (8.4–19.4) | 10.8 (6.8–15.6) | 9.4 (5.4–13.5) | 7.4 (2.7–11.2) | <0.001 |
| Commercially prepared dishes | −0.21 | 33.3 (14.4–53.8) | 31.3 (11.8–48.6) | 21.2 (5.9–35.6) | 13.2 (0–32.4) | <0.001 |
| Quartiles of Dietary Pattern Score in Men 2 (n = 32,589) (g/Day) | ||||||
|---|---|---|---|---|---|---|
| Food Groups | Factor Loading 1 | 1 | 2 | 3 | 4 | P–Trend |
| High intake | ||||||
| High–fat cheese | 0.38 | 11.6 (3.6–22.9) | 16.6 (6.3–30.6) | 21.2 (7.8–39.7) | 28.3 (10.2–55.5) | <0.001 |
| Bread | 0.34 | 129.9 (90.2–165.6) | 150.1 (115.0–197.0) | 169.9 (132.8–211.1) | 198.0 (148.0–257.7) | <0.001 |
| Full–fat milk | 0.23 | 0 (0–11.9) | 0 (0–34.6) | 5.4 (0–71.8) | 38.3 (0–139.4) | <0.001 |
| Fruits | 0.23 | 42.3 (16.9–110.1) | 84.6 (42.3–152.4) | 110.1 (52.7–220.2) | 220.2 (84.6–228.6) | <0.001 |
| Vegetables | 0.21 | 74.3 (41.6–110.5) | 81.8 (62.1–113.1) | 110.2 (63.5–149.1) | 113.1 (76.3–162.5) | <0.001 |
| Beer | 0.21 | 43.0 (0–107.4) | 57.3 (11.9–142.8) | 71.4 (18.9–171.9) | 73.9 (19.1–214.2) | <0.001 |
| Low–fat cheese | 0.20 | 0 (0–3.1) | 0 (0–6.9) | 0 (0–10.3) | 0 (0–17.9) | <0.001 |
| Legumes | 0.20 | 0 (0–11.0) | 5.5 (0–16.4) | 8.9 (0–17.6) | 16.4 (4.4–27.4) | <0.001 |
| Low intake | ||||||
| White meat | −0.33 | 13.2 (9.5–19.4) | 9.6 (6.7–13.9) | 8.4 (5.3–12.4) | 7.5 (2.3–11.2) | <0.001 |
| Red meat | −0.22 | 29.4 (20.4–39.5) | 24.5 (15.1–32.1) | 22.3 (12.9–30.5) | 18.5 (9.3–28.2) | <0.001 |
| Women | Quartiles of Dietary Pattern Score OR (95% CI) | Continuous Dietary Pattern Score | |||||
| 1 | 2 | 3 | 4 | P for Trend | OR (95% CI) | P | |
| Cases/population | 1316/11,438 | 1286/11,436 | 1215/11,436 | 1155/11,436 | 4972/45,746 | ||
| eGFR decline ≥20% (%) | 11.5 | 11.2 | 10.6 | 10.1 | <0.001 | 10.9 | |
| Model 1 | 1.00 | 0.97 (0.90–1.06) | 0.91 (0.84–0.99) | 0.86 (0.79–0.94) | <0.001 | 0.93 (0.90–0.97) | <0.001 |
| Model 2 | 1.00 | 0.97 (0.90–1.06) | 0.91 (0.84–0.99) | 0.86 (0.79–0.94) | <0.001 | 0.93 (0.90–0.97) | <0.001 |
| Model 3 | 1.00 | 0.95 (0.88–1.04) | 0.88 (0.81–0.96) | 0.83 (0.76–0.91) | <0.001 | 0.92 (0.88–0.95) | <0.001 |
| Model 4 | 1.00 | 0.95 (0.88–1.03) | 0.88 (0.81–0.96) | 0.83 (0.76–0.91) | <0.001 | 0.92 (0.88–0.95) | <0.001 |
| Model 5 | 1.00 | 0.93 (0.86–1.02) | 0.86 (0.79–0.94) | 0.79 (0.73–0.87) | <0.001 | 0.90 (0.86–0.93) | <0.001 |
| Men | Quartiles of Dietary Pattern Score OR (95% CI) | Continuous Dietary Pattern Score | |||||
| 1 | 2 | 3 | 4 | P for Trend | OR (95% CI) | P | |
| Cases/population | 756/8147 | 660/8148 | 648/8147 | 574/8147 | 2638/32,589 | ||
| eGFR decline ≥20% (%) | 9.3 | 8.1 | 8.0 | 7.0 | <0.001 | 8.1 | |
| Model 1 | 1.00 | 0.85 (0.76–0.94) | 0.82 (0.73–0.92) | 0.71 (0.64–0.80) | <0.001 | 0.87 (0.83–0.92) | <0.001 |
| Model 2 | 1.00 | 0.85 (0.76–0.95) | 0.81 (0.73–0.91) | 0.70 (0.63–0.79) | <0.001 | 0.86 (0.82–0.91) | <0.001 |
| Model 3 | 1.00 | 0.84 (0.75–0.94) | 0.80 (0.72–0.90) | 0.68 (0.60–0.77) | <0.001 | 0.85 (0.80–0.90) | <0.001 |
| Model 4 | 1.00 | 0.84 (0.76–0.94) | 0.80 (0.72–0.90) | 0.68 (0.60–0.77) | <0.001 | 0.85 (0.80–0.90) | <0.001 |
| Model 5 | 1.00 | 0.84 (0.75–0.94) | 0.80 (0.71–0.89) | 0.67 (0.59–0.76) | <0.001 | 0.85 (0.80–0.90) | <0.001 |
| Women | Quartiles of Dietary Pattern Score | Continuous Dietary Pattern Score | |||||
| 1 | 2 | 3 | 4 | P for Trend | OR (95% CI) | P | |
| Cases/population | 255/11,438 | 332/11,436 | 331/11,436 | 344/11,436 | 1262/45,746 | ||
| CKD incidence (%) | 2.2 | 2.9 | 2.9 | 3.0 | 0.001 | 2.8 | |
| Model 1 | 1.00 | 0.94 (0.79–1.12) | 0.78 (0.66–0.93) | 0.67 (0.57–0.80) | <0.001 | 0.80 (0.74–0.86) | <0.001 |
| Model 2 | 1.00 | 0.94 (0.79–1.11) | 0.77 (0.65–0.92) | 0.67 (0.56–0.79) | <0.001 | 0.80 (0.74–0.86) | <0.001 |
| Model 3 | 1.00 | 0.92 (0.77–1.09) | 0.74 (0.62–0.89) | 0.64 (0.54–0.77) | <0.001 | 0.78 (0.72–0.85) | <0.001 |
| Model 4 | 1.00 | 0.92 (0.77–1.09) | 0.74 (0.62–0.89) | 0.64 (0.54–0.77) | <0.001 | 0.78 (0.72–0.85) | <0.001 |
| Model 5 | 1.00 | 1.04 (0.85–1.27) | 0.88 (0.72–1.07)) | 0.88 (0.72–1.08) | 0.079 | 0.93 (0.85–1.01) | 0.095 |
| Men | Quartiles of Dietary Pattern Score | Continuous Dietary Pattern Score | |||||
| 1 | 2 | 3 | 4 | P for Trend | OR (95% CI) | P | |
| Cases/population | 216/8147 | 216/8148 | 195/8147 | 183/8147 | 810/32,589 | ||
| CKD incidence (%) | 2.7 | 2.7 | 2.4 | 2.2 | 0.056 | 2.5 | |
| Model 1 | 1.00 | 0.77 (0.62–0.94) | 0.57 (0.46–0.70) | 0.50 (0.40–0.61) | <0.001 | 0.70 (0.64–0.78) | <0.001 |
| Model 2 | 1.00 | 0.78 (0.64–0.96) | 0.58 (0.47–0.71) | 0.51 (0.41–0.63) | <0.001 | 0.71 (0.64–0.79) | <0.001 |
| Model 3 | 1.00 | 0.80 (0.65–0.98) | 0.60 (0.49–0.75) | 0.55 (0.44–0.69) | <0.001 | 0.73 (0.66–0.82) | <0.001 |
| Model 4 | 1.00 | 0.80 (0.65–0.98) | 0.60 (0.49–0.75) | 0.54 (0.43–0.68) | <0.001 | 0.73 (0.66–0.82) | <0.001 |
| Model 5 | 1.00 | 0.90 (0.71–1.14) | 0.76 (0.59–0.97) | 0.95 (0.73–1.23) | 0.372 | 0.96 (0.85–1.09) | 0.578 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, Q.; Dekker, L.H.; Bakker, S.J.L.; de Borst, M.H.; Navis, G.J. Dietary Patterns Based on Estimated Glomerular Filtration Rate and Kidney Function Decline in the General Population: The Lifelines Cohort Study. Nutrients 2020, 12, 1099. https://doi.org/10.3390/nu12041099
Cai Q, Dekker LH, Bakker SJL, de Borst MH, Navis GJ. Dietary Patterns Based on Estimated Glomerular Filtration Rate and Kidney Function Decline in the General Population: The Lifelines Cohort Study. Nutrients. 2020; 12(4):1099. https://doi.org/10.3390/nu12041099
Chicago/Turabian StyleCai, Qingqing, Louise H. Dekker, Stephan J. L. Bakker, Martin H. de Borst, and Gerjan J. Navis. 2020. "Dietary Patterns Based on Estimated Glomerular Filtration Rate and Kidney Function Decline in the General Population: The Lifelines Cohort Study" Nutrients 12, no. 4: 1099. https://doi.org/10.3390/nu12041099
APA StyleCai, Q., Dekker, L. H., Bakker, S. J. L., de Borst, M. H., & Navis, G. J. (2020). Dietary Patterns Based on Estimated Glomerular Filtration Rate and Kidney Function Decline in the General Population: The Lifelines Cohort Study. Nutrients, 12(4), 1099. https://doi.org/10.3390/nu12041099





