Mediterranean Diet Nutrients to Turn the Tide against Insulin Resistance and Related Diseases
Abstract
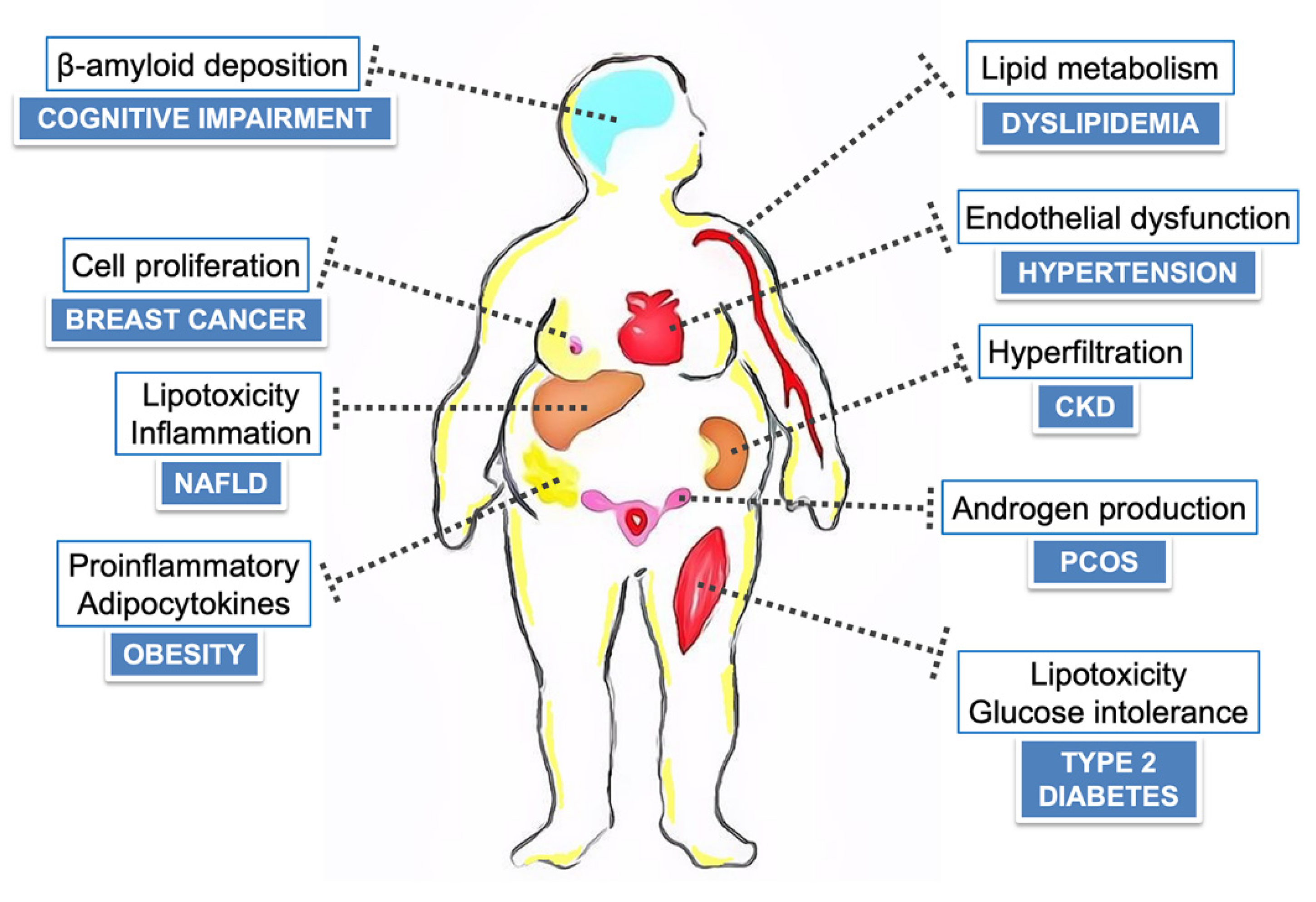
1. Introduction
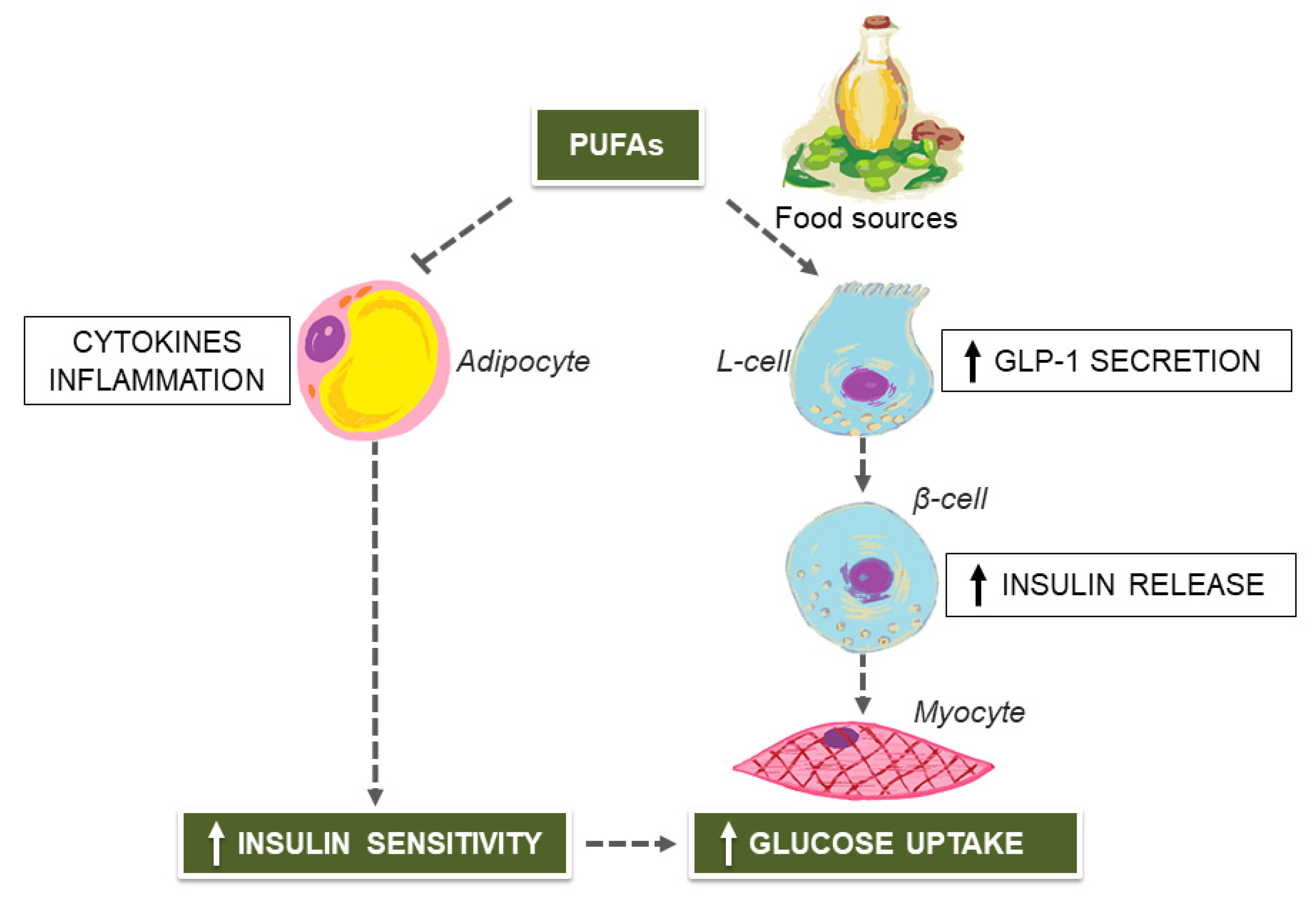
2. Mediterranean Diet (MedDiet) and Unsaturated Fatty Acids for Managing Type 2 Diabetes (T2D)
3. MedDiet Flavonoids for Preventing T2D
4. MedDiet and Extra-Virgin Olive Oil (EVOO)-Derived Secoiridoids for Treating Non-Alcoholic Fatty Liver Disease (NAFLD)
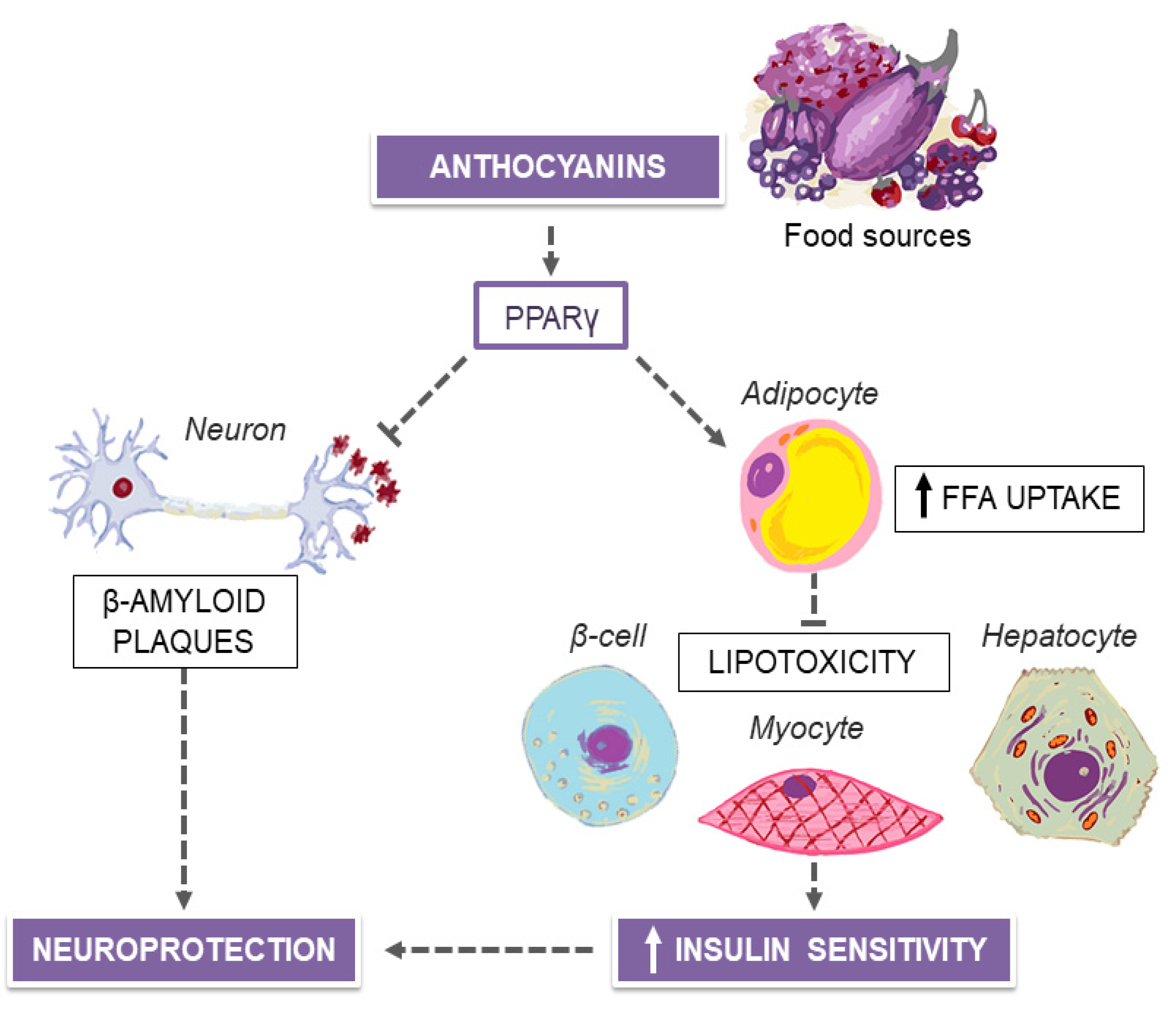
5. MedDiet and Purple Plant-Derived Anthocyanin Extracts for Neuroprotection
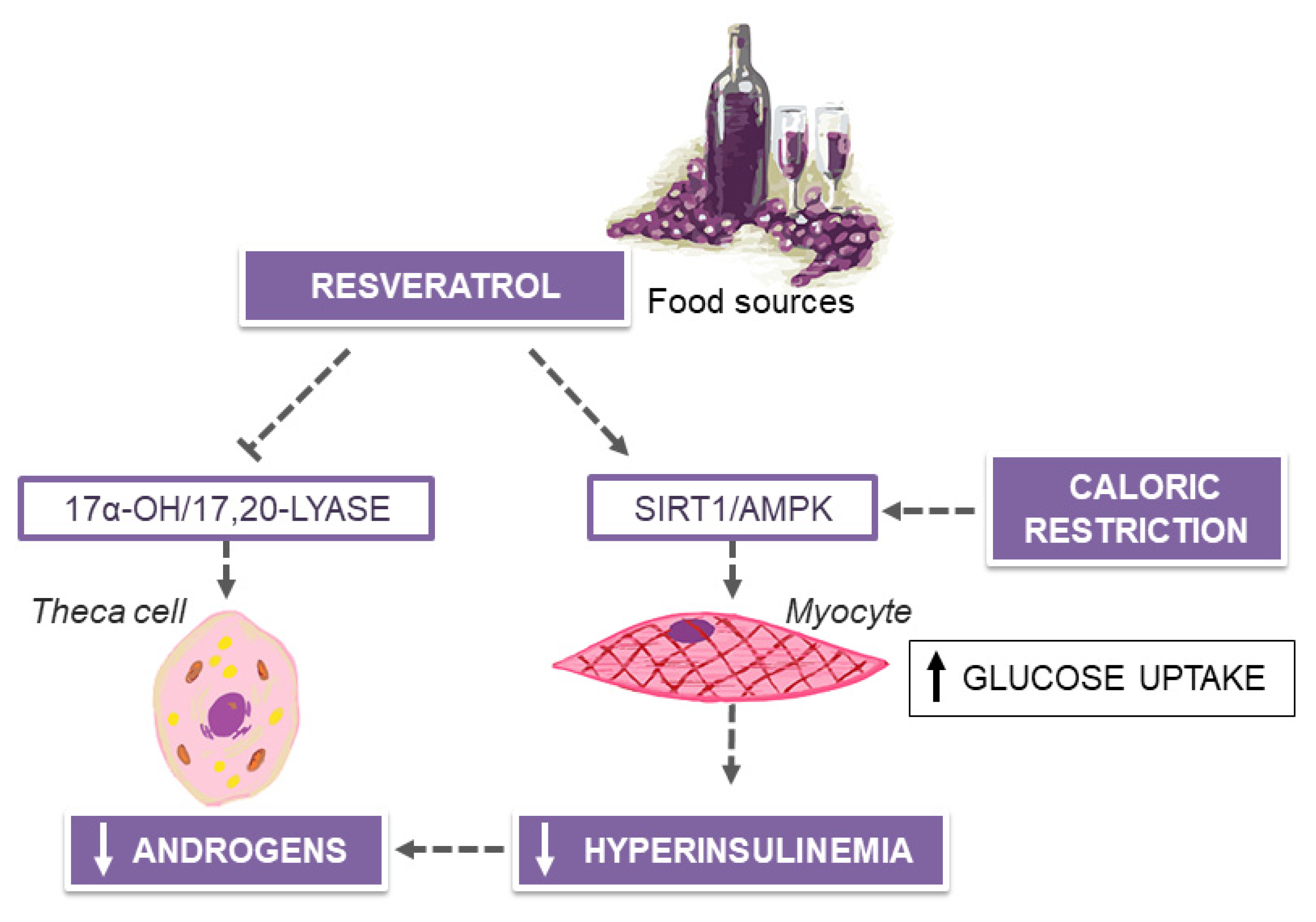
6. MedDiet and Resveratrol in Polycystic Ovary Syndrome (PCOS)
7. MedDiet and Nutrient–Gene Interactions in the Modulation of Breast Cancer Risk
8. Influence of Polyphenols on Endothelial Dysfunction and Atherosclerosis
9. MedDiet, EVOO and EVOO-Derived Polyphenols on Hypertension
10. MedDiet, EVOO and EVOO-Derived Polyphenols on Lipid Abnormalities
11. MedDiet and Management of Chronic Kidney Disease (CKD)
12. Limitations and Future Research Perspectives
13. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Brunetti, A.; Chiefari, E.; Foti, D. Recent advances in the molecular genetics of type 2 diabetes mellitus. World J. Diabetes 2014, 5, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Kelly, G.S. Insulin resistance: Lifestyle and nutritional interventions. Altern Med. Rev. 2000, 5, 109–132. [Google Scholar] [PubMed]
- Czech, M.P. Insulin action and resistance in obesity and type 2 diabetes. Nat. Med. 2017, 23, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, A.; Foti, D.; Goldfine, I.D. Identification of unique nuclear regulatory proteins for the insulin receptor gene, which appear during myocyte and adipocyte differentiation. J. Clin. Invest. 1993, 92, 1288–1295. [Google Scholar] [CrossRef]
- Hong, S.H.; Choi, K.M. Sarcopenic Obesity, Insulin Resistance, and Their Implications in Cardiovascular and Metabolic Consequences. Int. J. Mol. Sci. 2020, 21, 494. [Google Scholar] [CrossRef]
- De Rosa, S.; Arcidiacono, B.; Chiefari, E.; Brunetti, A.; Indolfi, C.; Foti, D.P. Type 2 diabetes mellitus and cardiovascular disease: Genetic and epigenetic links. Front. Endocrinol. 2018, 9, 2. [Google Scholar] [CrossRef]
- Arcidiacono, B.; Iiritano, S.; Nocera, A.; Possidente, K.; Nevolo, M.T.; Ventura, V.; Foti, D.; Chiefari, E.; Brunetti, A. Insulin resistance and cancer risk: An overview of the pathogenetic mechanisms. Exp. Diabetes Res. 2012, 2012, 789174. [Google Scholar] [CrossRef]
- Torchen, L.C. Cardiometabolic Risk in PCOS: More than a Reproductive Disorder. Curr. Diab. Rep. 2017, 17, 137. [Google Scholar] [CrossRef]
- Della Pepa, G.; Vetrani, C.; Lombardi, G.; Bozzetto, L.; Annuzzi, G.; Rivellese, A.A. Isocaloric Dietary Changes and Non-Alcoholic Fatty Liver Disease in High Cardiometabolic Risk Individuals. Nutrients 2017, 9, 1065. [Google Scholar] [CrossRef]
- Chiefari, E.; Tanyolaç, S.; Iiritano, S.; Sciacqua, A.; Capula, C.; Arcidiacono, B.; Nocera, A.; Possidente, K.; Baudi, F.; Ventura, V.; et al. A polymorphism of HMGA1 is associated with increased risk of metabolic syndrome and related components. Sci. Rep. 2013, 3, 1491. [Google Scholar] [CrossRef]
- Arnold, S.E.; Arvanitakis, Z.; Macauley-Rambach, S.L.; Koenig, A.M.; Wang, H.Y.; Ahima, R.S.; Craft, S.; Gandy, S.; Buettner, C.; Stoeckel, L.E.; et al. Brain insulin resistance in type 2 diabetes and Alzheimer disease: Concepts and conundrums. Nat. Rev. Neurol. 2018, 14, 168–181. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, G.S.; Smastuen, M.C.; Sandbu, R.; Nordstrand, N.; Hofsø, D.; Lindberg, M.; Hertel, J.K.; Hjelmesæth, J. Association of Bariatric Surgery vs Medical Obesity Treatment with Long-term Medical Complications and Obesity-Related Comorbidities. JAMA 2018, 319, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Rueda-Clausen, C.F.; Ogunleye, A.A.; Sharma, A.M. Health Benefits of Long-Term Weight-Loss Maintenance. Annu. Rev. Nutr. 2015, 35, 475–516. [Google Scholar] [CrossRef]
- Heggen, E.; Klemsdal, T.O.; Haugen, F.; Holme, I.; Tonstad, S. Effect of a low-fat versus a low-gycemic-load diet on inflammatory biomarker and adipokine concentrations. Metab. Syndr. Relat. Disord. 2012, 10, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Chen, R.; Wang, H.; Liang, F. Mechanisms Linking Inflammation to Insulin Resistance. Int. J. Endocrinol. 2015, 2015, 508409. [Google Scholar] [CrossRef]
- Accattato, F.; Greco, M.; Pullano, S.A.; Carè, I.; Fiorillo, A.S.; Pujia, A.; Montalcini, T.; Foti, D.P.; Brunetti, A.; Gulletta, E. Effects of acute physical exercise on oxidative stress and inflammatory status in young, sedentary obese subjects. PLoS ONE 2017, 12, e0178900. [Google Scholar] [CrossRef]
- Shai, I.; Schwarzfuchs, D.; Henkin, Y.; Shahar, D.R.; Witkow, S.; Greenberg, I.; Golan, R.; Fraser, D.; Bolotin, A.; Vardi, H.; et al. Dietary Intervention Randomized Controlled Trial (DIRECT) Group. Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. N. Engl. J. Med. 2008, 359, 229–241. [Google Scholar] [CrossRef]
- Greco, M.; Chiefari, E.; Montalcini, T.; Accattato, F.; Costanzo, F.S.; Pujia, A.; Foti, D.; Brunetti, A.; Gulletta, E. Early effects of a hypocaloric, Mediterranean diet on laboratory parameters in obese individuals. Mediat. Inflamm. 2014, 2014, 750860. [Google Scholar] [CrossRef]
- Menotti, A.; Puddu, P.E. How the Seven Countries Study contributed to the definition and development of the Mediterranean diet concept: A 50-year journey. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 245–252. [Google Scholar] [CrossRef]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean Diet; a Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef]
- Diolintzi, A.; Panagiotakos, D.; Sidossis, L. From Mediterranean diet to Mediterranean lifestyle: A narrative review. Public Health Nutr. 2019, 22, 2703–2713. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for us? Oxid. Med. Cell Longev. 2016, 2016, 7432797. [Google Scholar] [CrossRef] [PubMed]
- Galan-Lopez, P.; Sanchez-Oliver, A.J.; Pihu, M.; Gisladottir, T.; Dominguez, R.; Ries, F. Association between Adherence to the Mediterranean Diet and Physical Fitness with Body Composition Parameters in 1717 European Adolescents: The AdolesHealth Study. Nutrients 2019, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Archero, F.; Ricotti, R.; Solito, A.; Carrera, D.; Civello, F.; Di Bella, R.; Bellone, S.; Prodam, F. Adherence to the Mediterranean Diet among School Children and Adolescents Living in Northern Italy and Unhealthy Food Behaviors Associated to Overweight. Nutrients 2018, 10, 1322. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, K.E.; Florence, C.S.; Howard, D.H.; Joski, P. The impact of obesity on rising medical spending. Health Aff. (Millwood) 2004. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef]
- American Diabetes Association. Economic Costs of Diabetes in the U.S. in 2017. Diabetes Care 2018, 41, 917–928. [Google Scholar] [CrossRef]
- Ye, J. Mechanisms of insulin resistance in obesity. Front. Med. 2013, 7, 14–24. [Google Scholar] [CrossRef]
- Leahy, J.L. Pathogenesis of type 2 diabetes mellitus. Arch. Med. Res. 2005, 36, 197–209. [Google Scholar] [CrossRef]
- Arcidiacono, B.; Iiritano, S.; Chiefari, E.; Brunetti, F.S.; Gu, G.; Foti, D.P.; Brunetti, A. Cooperation between HMGA1, PDX-1, and MafA is essential for glucose- induced insulin transcription in pancreatic beta cells. Front. Endocrinol. 2015, 5, 237. [Google Scholar] [CrossRef] [PubMed]
- Knowler, W.C.; Barrett-Connor, E.; Fowler, S.E.; Hamman, R.F.; Lachin, J.M.; Walker, E.A.; Nathan, D.M.; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N. Engl. J. Med. 2002, 346, 393–403. [Google Scholar] [PubMed]
- American Diabetes Association. 5. Lifestyle Management: Standards of Medical Care in Diabetes-2019. Diabetes Care 2019, 42 (Suppl. 1), S46–S60. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Maiorino, M.I.; Ciotola, M.; Di Palo, C.; Scognamiglio, P.; Gicchino, M.; Petrizzo, M.; Saccomanno, F.; Beneduce, F.; Ceriello, A.; et al. Effects of a Mediterranean-style diet on the need for antihyperglycemic drug therapy in patients with newly diagnosed type 2 diabetes: A randomized trial. Ann. Intern. Med. 2009, 151, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Boucher, J.L. Mediterranean eating pattern. Diabetes Spectr. 2017, 30, 72–76. [Google Scholar] [CrossRef]
- Rinaldi, S.; Campbell, E.E.; Fournier, J.; O’Connor, C.; Madill, J. A comprehensive review of the literature supporting recommendations from the Canadian Diabetes Association for the use of a plant-based diet for management of type 2 diabetes. Can. J. Diabetes 2016, 40, 471–477. [Google Scholar] [CrossRef]
- Pawlak, R. Vegetarian diets in the prevention and management of diabetes and its complications. Diabetes Spectr. 2017, 30, 82–88. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Bamia, C.; Trichopoulos, D. Mediterranean diet and survival among patients with coronary heart disease in Greece. Arch. Intern. Med. 2005, 165, 929–935. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Jennings, A.; Berendsen, A.M.; de Groot, L.C.P.G.M.; Feskens, E.J.M.; Brzozowska, A.; Sicinska, E.; Pietruszka, B.; Meunier, N.; Caumon, E.; Malpuech-Brugère, C.; et al. Mediterranean-Style Diet Improves Systolic Blood Pressure and Arterial Stiffness in Older Adults. Hypertension 2019, 73, 578–586. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Li, J.; Hu, F.B.; Salas-Salvadó, J.; Tobias, D.K. Effects of walnut consumption on blood lipids and other cardiovascular risk factors: An updated meta-analysis and systematic review of controlled trials. Am. J. Clin. Nutr. 2018, 108, 174–187. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Caulfield, L.E.; Garcia-Larsen, V.; Steffen, L.M.; Coresh, J.; Rebholz, C.M. Plant-Based Diets Are Associated with a Lower Risk of Incident Cardiovascular Disease, Cardiovascular Disease Mortality, and All-Cause Mortality in a General Population of Middle-Aged Adults. J. Am. Heart Assoc. 2019, 8, e012865. [Google Scholar] [CrossRef]
- Qian, F.; Korat, A.A.; Malik, V.; Hu, F.B. Metabolic Effects of Monounsaturated Fatty Acid-Enriched Diets Compared with Carbohydrate or Polyunsaturated Fatty Acid-Enriched Diets in Patients with Type 2 Diabetes: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Diabetes Care 2016, 39, 1448–1457. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; de la Fuente-Arrillaga, C.; Nunez-Cordoba, J.M.; Basterra-Gortari, F.J.; Beunza, J.J.; Vazquez, Z.; Benito, S.; Tortosa, A.; Bes-Rastrollo, M. Adherence to Mediterranean diet and risk of developing diabetes: Prospective cohort study. BMJ 2008, 336, 1348–1351. [Google Scholar] [CrossRef] [PubMed]
- Salas-Salvadó, J.; Bulló, M.; Babio, N.; Martínez-González, M.Á.; Ibarrola-Jurado, N.; Basora, J.; Estruch, R.; Covas, M.I.; Corella, D.; Arós, F.; et al. PREDIMED Study Investigators. Reduction in the incidence of type 2 diabetes with the Mediterranean diet: Results of the PREDIMED-Reus nutrition intervention randomized trial. Diabetes Care 2011, 34, 14–19. [Google Scholar] [CrossRef]
- Lamping, K.G.; Nuno, D.W.; Coppey, L.J.; Holmes, A.J.; Hu, S.; Oltman, C.L.; Norris, A.W.; Yorek, M.A. Modification of high saturated fat diet with n-3 polyunsaturated fat improves glucose intolerance and vascular dysfunction. Diabetes Obes. Metab. 2013, 15, 144–152. [Google Scholar] [CrossRef]
- Matravadia, S.; Herbst, E.A.; Jain, S.S.; Mutch, D.M.; Holloway, G.P. Both linoleic and α-linolenic acid prevent insulin resistance but have divergent impacts on skeletal muscle mitochondrial bioenergetics in obese Zucker rats. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E102–E114. [Google Scholar] [CrossRef]
- Nardi, F.; Lipina, C.; Magill, D.; Hage Hassan, R.; Hajduch, E.; Gray, A.; Hundal, H.S. Enhanced Insulin Sensitivity Associated with Provision of Mono and Polyunsaturated Fatty Acids in Skeletal Muscle Cells Involves Counter Modulation of PP2A. PLoS ONE 2014, 9, e92255. [Google Scholar] [CrossRef]
- Berger, M.M.; Delodder, F.; Liaudet, L.; Tozzi, P.; Schlaepfer, J.; Chiolero, R.L.; Tappy, L. Three short perioperative infusions of n-3 PUFAs reduce systemic inflammation induced by cardiopulmonary bypass surgery: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 246–254. [Google Scholar] [CrossRef]
- Sundström, L.; Myhre, S.; Sundqvist, M.; Ahnmark, A.; McCoull, W.; Raubo, P.; Groombridge, S.D.; Polla, M.; Nyström, A.C.; Kristensson, L.; et al. The acute glucose lowering effect of specific GPR120 activation in mice is mainly driven by glucagon-like peptide 1. PLoS ONE 2017, 12, e0189060. [Google Scholar] [CrossRef]
- Holst, J.J. The physiology of glucagon-like peptide 1. Physiol. Rev. 2007, 87, 1409–1439. [Google Scholar] [CrossRef]
- Færch, K.; Torekov, S.S.; Vistisen, D.; Johansen, N.B.; Witte, D.R.; Jonsson, A.; Pedersen, O.; Hansen, T.; Lauritzen, T.; Sandbæk, A.; et al. GLP-1 Response to Oral Glucose Is Reduced in Prediabetes, Screen-Detected Type 2 Diabetes, and Obesity and Influenced by Sex: The ADDITION-PRO Study. Diabetes 2015, 64, 2513–2525. [Google Scholar] [CrossRef] [PubMed]
- Mirabelli, M.; Chiefari, E.; Caroleo, P.; Arcidiacono, B.; Corigliano, D.M.; Giuliano, S.; Brunetti, F.S.; Tanyolaç, S.; Foti, D.P.; Puccio, L.; et al. Long-Term Effectiveness of Liraglutide for Weight Management and Glycemic Control in Type 2 Diabetes. Int. J. Environ. Res. Public Health 2020, 17, 207. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.J.; Brainard, J.; Song, F.; Wang, X.; Abdelhamid, A.; Hooper, L.; PUFAH Group. Omega-3, omega-6, and total dietary polyunsaturated fat for prevention and treatment of type 2 diabetes mellitus: Systematic review and meta-analysis of randomised controlled trials. BMJ 2019, 21, 4697. [Google Scholar] [CrossRef] [PubMed]
- Vessby, B.; Uusitupa, M.; Hermansen, K.; Riccardi, G.; Rivellese, A.A.; Tapsell, L.C.; Nälsén, C.; Berglund, L.; Louheranta, A.; Rasmussen, B.M.; et al. Substituting dietary saturated for monounsaturated fat impairs insulin sensitivity in healthy men and women: The KANWU Study. Diabetologia 2001, 44, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Alkhatib, A.; Tsang, C.; Tiss, A.; Bahorun, T.; Arefanian, H.; Barake, R.; Khadir, A.; Tuomilehto, J. Functional Foods and Lifestyle Approaches for Diabetes Prevention and Management. Nutrients 2017, 1, 1310. [Google Scholar] [CrossRef]
- Al-Ishaq, R.K.; Abotaleb, M.; Kubatka, P.; Kajo, K.; Büsselberg, D. Flavonoids and Their Anti-Diabetic Effects: Cellular Mechanisms and Effects to Improve Blood Sugar Levels. Biomolecules 2019, 9, 430. [Google Scholar] [CrossRef]
- Xu, H.; Luo, J.; Huang, J.; Wen, Q. Flavonoids intake and risk of type 2 diabetes mellitus: A meta-analysis of prospective cohort studies. Medicine (Baltimore) 2018, 97, e0686. [Google Scholar] [CrossRef]
- Eid, H.M.; Martineau, L.C.; Saleem, A.; Muhammad, A.; Vallerand, D.; Benhaddou-Andaloussi, A.; Nistor, L.; Afshar, A.; Arnason, J.T.; Haddad, P.S. Stimulation of AMP-activated protein kinase and enhancement of basal glucose uptake in muscle cells by quercetin and quercetin glycosides, active principles of the antidiabetic medicinal plant Vaccinium vitis-idaea. Mol. Nutr. Food Res. 2010, 54, 991–1003. [Google Scholar] [CrossRef]
- Dhanya, R.; Arya, A.D.; Nisha, P.; Jayamurthy, P. Quercetin, a Lead Compound against Type 2 Diabetes Ameliorates Glucose Uptake via AMPK Pathway in Skeletal Muscle Cell Line. Front. Pharm. 2017, 8, 336. [Google Scholar] [CrossRef]
- Zhang, A.J.; Rimando, A.M.; Mizuno, C.S.; Mathews, S.T. α-Glucosidase inhibitory effect of resveratrol and piceatannol. J. Nutr. Biochem. 2017, 47, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Proença, C.; Freitas, M.; Ribeiro, D.; Oliveira, E.F.T.; Sousa, J.L.C.; Tomé, S.M.; Ramos, M.J.; Silva, A.M.S.; Fernandes, P.A.; Fernandes, E. α-Glucosidase inhibition by flavonoids: An in vitro and in silico structure–activity relationship study. J. Enzyme. Inhib. Med. Chem. 2017, 32, 1216–1228. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-metaanalytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Portillo-Sanchez, P.; Bril, F.; Maximos, M.; Lomonaco, R.; Biernacki, D.; Orsak, B.; Subbarayan, S.; Webb, A.; Hecht, J.; Cusi, K. High prevalence of nonalcoholic fatty liver disease in patients with type 2 diabetes mellitus and normal plasma aminotransferase levels. J. Clin. Endocrinol. Metab. 2015, 100, 2231–2238. [Google Scholar] [CrossRef] [PubMed]
- Rinella, M.E. Nonalcoholic fatty liver disease: A systematic review. JAMA 2015, 313, 2263–2273. [Google Scholar] [CrossRef] [PubMed]
- Vilar-Gomez, E.; Calzadilla-Bertot, L.; Wai-Sun Wong, V.; Castellanos, M.; Aller-de la Fuente, R.; Metwally, M.; Eslam, M.; Gonzalez-Fabian, L.; Alvarez-Quiñones Sanz, M.; Conde-Martin, A.F.; et al. Fibrosis Severity as a Determinant of Cause-Specific Mortality in Patients with Advanced Nonalcoholic Fatty Liver Disease: A Multi-National Cohort Study. Gastroenterology 2018, 155, 443–457.e17. [Google Scholar] [CrossRef]
- Angulo, P.; Kleiner, D.E.; Dam-Larsen, S.; Adams, L.A.; Bjornsson, E.S.; Charatcharoenwitthaya, P.; Mills, P.R.; Keach, J.C.; Lafferty, H.D.; Stahler, A.; et al. Liver Fibrosis, but No Other Histologic Features, Is Associated with Long-term Outcomes of Patients with Nonalcoholic Fatty Liver Disease. Gastroenterology 2015, 149, 389–397.e10. [Google Scholar] [CrossRef]
- Adams, L.A.; Anstee, Q.M.; Tilg, H.; Targher, G. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut 2017, 66, 1138–1153. [Google Scholar] [CrossRef]
- Patel, N.S.; Doycheva, I.; Peterson, M.R.; Hooker, J.; Kisselva, T.; Schnabl, B.; Seki, E.; Sirlin, C.B.; Loomba, R. Effect of weight loss on magnetic resonance imaging estimation of liver fat and volume in patients with nonalcoholic steatohepatitis. Clin. Gastroenterol. Hepatol. 2015, 13, 561–568.e1. [Google Scholar] [CrossRef]
- Promrat, K.; Kleiner, D.E.; Niemeier, H.M.; Jackvony, E.; Kearns, M.; Wands, J.R.; Fava, J.L.; Wing, R.R. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology 2010, 51, 121–129. [Google Scholar] [CrossRef]
- Ganguli, S.; DeLeeuw, P.; Satapathy, S.K. A Review of Current and Upcoming Treatment Modalities in Non-Alcoholic Fatty Liver Disease And Non-Alcoholic Steatohepatitis. Hepat. Med. 2019, 15, 159–178. [Google Scholar] [CrossRef]
- Asrih, M.; Jornayvaz, F.R. Diets and nonalcoholic fatty liver disease: The good and the bad. Clin. Nutr. 2014, 33, 186–190. [Google Scholar] [CrossRef]
- Temmerman, J.C.; Friedman, A.N. Very low calorie ketogenic weight reduction diet in patients with cirrhosis: A case series. Nutr. Diabetes 2013, 3, e95. [Google Scholar] [CrossRef] [PubMed]
- George, E.S.; Forsyth, A.; Itsiopoulos, C.; Nicoll, A.J.; Ryan, M.; Sood, S.; Roberts, S.K.; Tierney, A.C. Practical Dietary Recommendations for the Prevention and Management of Nonalcoholic Fatty Liver Disease in Adults. Adv. Nutr. 2018, 9, 30–40. [Google Scholar] [CrossRef]
- Aller, R.; Izaola, O.; de la Fuente, B.; De Luis Román, D.A. Mediterranean diet is associated with liver histology in patients with non-alcoholic fatty liver disease. Nutr. Hosp. 2015, 32, 2518–2524. [Google Scholar] [CrossRef] [PubMed]
- Kontogianni, M.D.; Tileli, N.; Margariti, A.; Georgoulis, M.; Deutsch, M.; Tiniakos, D.; Fragopoulou, E.; Zafiropoulou, R.; Manios, Y.; Papatheodoridis, G. Adherence to the Mediterranean diet is associated with the severity of non-alcoholic fatty liver disease. Clin. Nutr. 2014, 33, 678–683. [Google Scholar] [CrossRef]
- Ryan, M.C.; Itsiopoulos, C.; Thodis, T.; Ward, G.; Trost, N.; Hofferberth, S.; O’Dea, K.; Desmond, P.V.; Johnson, N.A.; Wilson, A.M. The Mediterranean diet improves hepatic steatosis and insulin sensitivity in individuals with non-alcoholic fatty liver disease. J. Hepatol. 2013, 59, 138–143. [Google Scholar] [CrossRef]
- Misciagna, G.; Del Pilar Díaz, M.; Caramia, D.V.; Bonfiglio, C.; Franco, I.; Noviello, M.R.; Chiloiro, M.; Abbrescia, D.I.; Mirizzi, A.; Tanzi, M.; et al. Effect of a Low Glycemic Index Mediterranean Diet on Non-Alcoholic Fatty Liver Disease. A Randomized Controlled Clinici Trial. J. Nutr. Health Aging 2017, 21, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Kaliora, A.C.; Gioxari, A.; Kalafati, I.P.; Diolintzi, A.; Kokkinos, A.; Dedoussis, G.V. The Effectiveness of Mediterranean Diet in Nonalcoholic Fatty Liver Disease Clinical Course: An Intervention Study. J. Med. Food 2019, 22, 729–740. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASDEASO Clinical Practice Guidelines for the management of nonalcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef] [PubMed]
- Cicerale, S.; Lucas, L.; Keast, R. Biological activities of phenolic compounds present in virgin olive oil. Int. J. Mol. Sci. 2010, 11, 458–479. [Google Scholar] [CrossRef] [PubMed]
- Bulotta, S.; Celano, M.; Lepore, S.M.; Montalcini, T.; Pujia, A.; Russo, D. Beneficial effects of the olive oil phenolic components oleuropein and hydroxytyrosol: Focus on protection against cardiovascular and metabolic diseases. J. Transl. Med. 2014, 12, 219. [Google Scholar] [CrossRef] [PubMed]
- Barbaro, B.; Toietta, G.; Maggio, R.; Arciello, M.; Tarocchi, M.; Galli, A.; Balsano, C. Effects of the olive-derived polyphenol oleuropein on human health. Int. J. Mol. Sci. 2014, 15, 18508–18524. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, G.E.; Arcidiacono, B.; De Rose, R.F.; Lepore, S.M.; Costa, N.; Montalcini, T.; Brunetti, A.; Russo, D.; De Sarro, G.; Celano, M. Normocaloric diet restores weight gain and insulin sensitivity in obese mice. Front. Endocrinol. 2016, 7, 49. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, G.E.; Lepore, S.M.; Morittu, V.M.; Arcidiacono, B.; Colica, C.; Procopio, A.; Maggisano, V.; Bulotta, S.; Costa, N.; Mignogna, C.; et al. Effects of Oleacein on High-Fat Diet-Dependent Steatosis, Weight Gain, and Insulin Resistance in Mice. Front. Endocrinol. (Lausanne) 2018, 9, 116. [Google Scholar] [CrossRef]
- Costanzo, P.; Bonacci, S.; Cariati, L.; Nardi, M.; Oliverio, M.; Procopio, A. Simple and efficient sustainable semi-synthesis of oleacein [2-(3,4-hydroxyphenyl) ethyl (3S,4E)-4-formyl-3-(2-oxoethyl)hex-4-enoate] as potential additive for edible oils. Food Chem. 2018, 245, 410–414. [Google Scholar] [CrossRef]
- Lepore, S.M.; Maggisano, V.; Bulotta, S.; Mignogna, C.; Arcidiacono, B.; Procopio, A.; Brunetti, A.; Russo, D.; Celano, M. Oleacein Prevents High Fat Diet-Induced Adiposity and Ameliorates Some Biochemical Parameters of Insulin Sensitivity in Mice. Nutrients 2019, 11, 1829. [Google Scholar] [CrossRef]
- Ueyama, A.; Ban, N.; Fukazawa, M.; Hirayama, T.; Takeda, M.; Yata, T.; Muramatsu, H.; Hoshino, M.; Yamamoto, M.; Matsuo, M.; et al. Inhibition of MEK1 signaling pathway in the liver ameliorates insulin resistance. J. Diabetes Res. 2016, 2016, 8264830. [Google Scholar] [CrossRef]
- Willette, A.A.; Kapogiannis, D. Does the brain shrink as the waist expands? Ageing Res. Rev. 2015, 20, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Caunca, M.R.; Gardener, H.; Simonetto, M.; Cheung, Y.K.; Alperin, N.; Yoshita, M.; DeCarli, C.; Elkind, M.S.V.; Sacco, R.L.; Wright, C.B.; et al. Measures of obesity are associated with MRI markers of brain aging: The Northern Manhattan Study. Neurology 2019, 9, e791–e803. [Google Scholar] [CrossRef] [PubMed]
- Arnemann, K.L.; Stöber, F.; Narayan, S.; Rabinovici, G.D.; Jagust, W.J. Metabolic brain networks in aging and preclinical Alzheimer’s disease. Neuroimage Clin. 2017, 17, 987–999. [Google Scholar] [CrossRef] [PubMed]
- Adams, O.P. The impact of brief high-intensity exercise on blood glucose levels. Diabetes Metab. Syndr. Obes. 2013, 6, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Olloquequi, J.; Ettcheto, M.; Busquets, O.; Sánchez-López, E.; Cano, A.; Espinosa-Jiménez, T.; García, M.L.; Beas-Zarate, C.; Casadesús, G.; et al. The Involvement of Peripheral and Brain Insulin Resistance in Late Onset Alzheimer’s Dementia. Front. Aging Neurosci. 2019, 6, 236. [Google Scholar] [CrossRef] [PubMed]
- Kanoski, S.E.; Zhang, Y.; Zheng, W.; Davidson, T.L. The effects of a high-energy diet on hippocampal function and blood-brain barrier integrity in the rat. J. Alzheimers Dis. 2010, 21, 207–219. [Google Scholar] [CrossRef]
- Berti, V.; Walters, M.; Sterling, J.; Quinn, C.G.; Logue, M.; Andrews, R.; Matthews, D.C.; Osorio, R.S.; Pupi, A.; Vallabhajosula, S.; et al. Mediterranean diet and 3-year Alzheimer brain biomarker changes in middle-aged adults. Neurology 2018, 90, e1789–e1798. [Google Scholar] [CrossRef]
- van Harten, B.; de Leeuw, F.E.; Weinstein, H.C.; Scheltens, P.; Biessels, G.J. Brain imaging in patients with diabetes: A systematic review. Diabetes Care 2006, 29, 2539–2548. [Google Scholar] [CrossRef]
- Croll, P.H.; Voortman, T.; Ikram, M.A.; Franco, O.H.; Schoufour, J.D.; Bos, D.; Vernooij, M.W. Better diet quality relates to larger brain tissue volumes: The Rotterdam Study. Neurology 2018, 90, e2166–e2173. [Google Scholar] [CrossRef]
- Gu, Y.; Brickman, A.M.; Stern, Y.; Habeck, C.G.; Razlighi, Q.R.; Luchsinger, J.A.; Manly, J.J.; Schupf, N.; Mayeux, R.; Scarmeas, N. Mediterranean diet and brain structure in a multiethnic elderly cohort. Neurology 2015, 85, 1744–1751. [Google Scholar] [CrossRef]
- Pelletier, A.; Barul, C.; Féart, C.; Helmer, C.; Bernard, C.; Periot, O.; Dilharreguy, B.; Dartigues, J.F.; Allard, M.; Barberger-Gateau, P.; et al. Mediterranean diet and preserved brain structural connectivity in older subjects. Alzheimers Dement. 2015, 11, 1023–1031. [Google Scholar] [CrossRef]
- Gardener, H.; Scarmeas, N.; Gu, Y.; Boden-Albala, B.; Elkind, M.S.; Sacco, R.L.; DeCarli, C.; Wright, C.B. Mediterranean diet and white matter hyperintensity volume in the Northern Manhattan Study. Arch. Neurol. 2012, 69, 251–256. [Google Scholar] [CrossRef]
- Staubo, S.C.; Aakre, J.A.; Vemuri, P.; Syrjanen, J.A.; Mielke, M.M.; Geda, Y.E.; Kremers, W.K.; Machulda, M.M.; Knopman, D.S.; Petersen, R.C.; et al. Mediterranean diet, micronutrients and macronutrients, and MRI measures of cortical thickness. Alzheimers Dement. 2017, 13, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Afzal, M.; Redha, A.; AlHasan, R. Anthocyanins Potentially Contribute to Defense against Alzheimer’s Disease. Molecules 2019, 24, 4255. [Google Scholar] [CrossRef] [PubMed]
- Passamonti, S.; Vrhovsek, U.; Vanzo, A.; Mattivi, F. Fast access of some grape pigments to the brain. J. Agric. Food Chem. 2005, 53, 7029–7034. [Google Scholar] [CrossRef] [PubMed]
- Talavera, S.; Felgines, C.; Texier, O.; Besson, C.; Gil-Izquierdo, A.; Lamaison, J.L.; Remesy, C. Anthocyanin metabolism in rats and their distribution to digestive area, kidney, and brain. J. Agric. Food Chem. 2005, 53, 3902–3908. [Google Scholar] [CrossRef] [PubMed]
- Kalt, W.; Blumberg, J.B.; McDonald, J.E.; Vinqvist-Tymchuk, M.R.; Fillmore, S.A.; Graf, B.A.; O’Leary, J.M.; Milbury, P.E. Identification of anthocyanins in the liver, eye, and brain of blueberry-fed pigs. J. Agric. Food Chem. 2008, 56, 705–712. [Google Scholar] [CrossRef]
- Vepsäläinen, S.; Koivisto, H.; Pekkarinen, E.; Mäkinen, P.; Dobson, G.; McDougall, G.J.; Stewart, D.; Haapasalo, A.; Karjalainen, R.O.; Tanila, H.; et al. Anthocyanin-enriched bilberry and blackcurrant extracts modulate amyloid precursor protein processing and alleviate behavioral abnormalities in the APP/PS1 mouse model of Alzheimer’s disease. J. Nutr. Biochem. 2013, 24, 360–370. [Google Scholar] [CrossRef]
- Song, N.; Zhang, L.; Chen, W.; Zhu, H.; Deng, W.; Han, Y.; Guo, J.; Qin, C. Cyanidin 3-O-beta-glucopyranoside activates peroxisome proliferator-activated receptor-gamma and alleviates cognitive impairment in the APP(swe)/PS1(DeltaE9) mouse model. Biochim. Biophys. Acta 2016, 1862, 1786–1800. [Google Scholar] [CrossRef]
- Bianconcini, A.; Lupo, A.; Capone, S.; Quadro, L.; Monti, M.; Zurlo, D.; Fucci, A.; Sabatino, L.; Brunetti, A.; Chiefari, E.; et al. Transcriptional activity of the murine retinol-binding protein gene is regulated by a multiprotein complex containing HMGA1, p54nrb/NonO, protein-associated splicing factor (PSF) and steroidogenic factor 1 (SF1)/liver receptor homologue 1 (LRH-1). Int. J. Biochem. Cell Biol. 2009, 41, 2189–2203. [Google Scholar] [CrossRef][Green Version]
- Corigliano, D.M.; Syed, R.; Messineo, S.; Lupia, A.; Patel, R.; Reddy, C.V.R.; Dubey, P.K.; Colica, C.; Amato, R.; De Sarro, G.; et al. Indole and 2,4-Thiazolidinedione conjugates as potential anticancer modulators. PeerJ 2018, 8, e5386. [Google Scholar] [CrossRef]
- Villapol, S. Roles of Peroxisome Proliferator-Activated Receptor Gamma on Brain and Peripheral Inflammation. Cell Mol. Neurobiol. 2018, 38, 121–132. [Google Scholar] [CrossRef]
- Thangavel, N.; Al Bratty, M.; Akhtar Javed, S.; Ahsan, W.; Alhazmi, H.A. Targeting Peroxisome Proliferator-Activated Receptors Using Thiazolidinediones: Strategy for Design of Novel Antidiabetic Drugs. Int. J. Med. Chem. 2017, 2017, 1069718. [Google Scholar] [CrossRef] [PubMed]
- Costa, V.; Foti, D.; Paonessa, F.; Chiefari, E.; Palaia, L.; Brunetti, G.; Gulletta, E.; Fusco, A.; Brunetti, A. The insulin receptor: A new anticancer target for peroxisome proliferator-activated receptor-g (PPARg) and thiazolidinedione-PPARg agonists. Endocr. Relat. Cancer 2008, 15, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Nanjan, M.J.; Mohammed, M.; Prashantha Kumar, B.R.; Chandrasekar, M.J.N. Thiazolidinediones as antidiabetic agents: A critical review. Bioorg. Chem. 2018, 77, 548–567. [Google Scholar] [CrossRef]
- Stumvoll, M.; Goldstein, B.J.; van Haeften, T.W. Type 2 diabetes: Principles of pathogenesis and therapy. Lancet 2005, 365, 1333–1346. [Google Scholar] [CrossRef]
- Rhea, E.M.; Banks, W.A. Role of the Blood-Brain Barrier in Central Nervous System Insulin Resistance. Front. Neurosci. 2019, 4, 521. [Google Scholar] [CrossRef]
- Frolich, L.; Blum-Degen, D.; Riederer, P.; Hoyer, S. A disturbance in the neuronal insulin receptor signal transduction in sporadic Alzheimer’s disease. Ann. N. Y. Acad. Sci. 1999, 893, 290–293. [Google Scholar] [CrossRef]
- Pearson-Leary, J.; Jahagirdar, V.; Sage, J.; McNay, E.C. Insulin modulates hippocampally-mediated spatial working memory via glucose transporter-4. Behav. Brain Res. 2018, 15, 32–39. [Google Scholar] [CrossRef]
- Jia, Y.; Kim, J.Y.; Jun, H.J.; Kim, S.J.; Lee, J.H.; Hoang, M.H.; Kim, H.S.; Chang, H.I.; Hwang, K.Y.; Um, S.J.; et al. Cyanidin is an agonistic ligand for peroxisome proliferator-activated receptor-alpha reducing hepatic lipid. Biochim. Biophys. Acta 2013, 1831, 698–708. [Google Scholar] [CrossRef]
- Kent, K.; Charlton, K.; Roodenrys, S.; Batterham, M.; Potter, J.; Traynor, V.; Gilbert, H.; Morgan, O.; Richards, R. Consumption of anthocyanin-rich cherry juice for 12 weeks improves memory and cognition in older adults with mild-to-moderate dementia. Eur. J. Nutr. 2017, 56, 333–341. [Google Scholar] [CrossRef]
- Krikorian, R.; Nash, T.A.; Shidler, M.D.; Shukitt-Hale, B.; Joseph, J.A. Concord grape juice supplementation improves memory function in older adults with mild cognitive impairment. Br. J. Nutr. 2010, 103, 730–734. [Google Scholar] [CrossRef]
- Krikorian, R.; Shidler, M.D.; Nash, T.A.; Kalt, W.; Vinqvist-Tymchuk, M.R.; Shukitt-Hale, B.; Joseph, J.A. Blueberry supplementation improves memory in older adults. J. Agric. Food Chem. 2010, 58, 3996–4000. [Google Scholar] [CrossRef]
- Shenoy, S.F.; Kazaks, A.G.; Holt, R.R.; Chen, H.J.; Winters, B.L.; Khoo, C.S.; Poston, W.S.; Haddock, C.K.; Reeves, R.S.; Foreyt, J.P.; et al. The use of a commercial vegetable juice as a practical means to increase vegetable intake: A randomized controlled trial. Nutr. J. 2010, 9, 38. [Google Scholar] [CrossRef]
- de la Rubia Orti, J.E.; Garcia-Pardo, M.P.; Drehmer, E.; Sancho Cantus, D.; Julian Rochina, M.; Aguilar, M.A.; Hu Yang, I. Improvement of Main Cognitive Functions in Patients with Alzheimer’s Disease after Treatment with Coconut Oil Enriched Mediterranean Diet: A Pilot Study. J. Alzheimers Dis. 2018, 65, 577–587. [Google Scholar] [CrossRef]
- Cassar, S.; Misso, M.L.; Hopkins, W.G.; Shaw, C.S.; Teede, H.J.; Stepto, N.K. Insulin resistance in polycystic ovary syndrome: A systematic review and meta-analysis of euglycaemic-hyperinsulinaemic clamp studies. Hum. Reprod. 2016, 31, 2619–2631. [Google Scholar] [CrossRef]
- Dunaif, A. Insulin resistance and the polycystic ovary syndrome: Mechanism and implications for pathogenesis. Endocr. Rev. 1997, 18, 774–800. [Google Scholar] [CrossRef]
- Roberts, J.S.; Perets, R.A.; Sarfert, K.S.; Bowman, J.J.; Ozark, P.A.; Whitworth, G.B.; Blythe, S.N.; Toporikova, N. High-fat high-sugar diet induces polycystic ovary syndrome in a rodent model. Biol. Reprod. 2017. [Google Scholar] [CrossRef]
- González, F.; Sia, C.L.; Shepard, M.K.; Rote, N.S.; Minium, J. The altered mononuclear cell-derived cytokine response to glucose ingestion is not regulated by excess adiposity in polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2014, 99, E2244–E2251. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.; Davidson, C.R.; Billings, D.L. Dietary intake, eating behaviors, and quality of life in women with polycystic ovary syndrome who are trying to conceive. Hum. Fertil. (Camb.) 2015, 18, 16–21. [Google Scholar] [CrossRef]
- Caroleo, M.; Carbone, E.A.; Greco, M.; Corigliano, D.M.; Arcidiacono, B.; Fazia, G.; Rania, M.; Aloi, M.; Gallelli, L.; Segura-Garcia, C.; et al. Brain-behavior-immune interaction: Serum cytokines and growth factors in patients with eating disorders at extremes of the body mass index (BMI) spectrum. Nutrients 2019, 23, 1995. [Google Scholar] [CrossRef]
- Escobar-Morreale, H.F.; Botella-Carretero, J.I.; Alvarez-Blasco, F.; Sancho, J.; San Millán, J.L. The polycystic ovary syndrome associated with morbid obesity may resolve after weight loss induced by bariatric surgery. J. Clin. Endocrinol. Metab. 2005, 90, 6364–6369. [Google Scholar] [CrossRef]
- Douglas, C.C.; Gower, B.A.; Darnell, B.E.; Ovalle, F.; Oster, R.A.; Azziz, R. Role of diet in the treatment of polycystic ovary syndrome. Fertil. Steril. 2006, 85, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Marsh, K.A.; Steinbeck, K.S.; Atkinson, F.S.; Petocz, P.; Brand-Miller, J.C. Effect of a low glycemic index compared with a conventional healthy diet on polycystic ovary syndrome. Am. J. Clin. Nutr. 2010, 92, 83–92. [Google Scholar] [CrossRef]
- Mavropoulos, J.C.; Yancy, W.S.; Hepburn, J.; Westman, E.C. The effects of a low-carbohydrate, ketogenic diet on the polycystic ovary syndrome: A pilot study. Nutr. Metab. (Lond.) 2005, 16, 35. [Google Scholar] [CrossRef]
- McGrice, M.; Porter, J. The Effect of Low Carbohydrate Diets on Fertility Hormones and Outcomes in Overweight and Obese Women: A Systematic Review. Nutrients 2017, 9, 204. [Google Scholar] [CrossRef] [PubMed]
- Barrea, L.; Arnone, A.; Annunziata, G.; Muscogiuri, G.; Laudisio, D.; Salzano, C.; Pugliese, G.; Colao, A.; Savastano, S. Adherence to the Mediterranean Diet, Dietary Patterns and Body Composition in Women with Polycystic Ovary Syndrome (PCOS). Nutrients 2019, 11, 2278. [Google Scholar] [CrossRef] [PubMed]
- Orio, F.; Muscogiuri, G.; Palomba, S. Could the Mediterranean diet be effective in women with polycystic ovary syndrome? A proof of concept. Eur. J. Clin. Nutr. 2015, 69, 974. [Google Scholar] [CrossRef]
- Gaskins, A.J.; Rovner, A.J.; Mumford, S.L.; Yeung, E.; Browne, R.W.; Trevisan, M.; Perkins, N.J.; Wactawski-Wende, J.; Schisterman, E.F.; BioCycle Study Group. Adherence to a Mediterranean diet and plasma concentrations of lipid peroxidation in premenopausal women. Am. J. Clin. Nutr. 2010, 92, 1461–1467. [Google Scholar] [CrossRef]
- Banaszewska, B.; Wrotyńska-Barczyńska, J.; Spaczynski, R.Z.; Pawelczyk, L.; Duleba, A.J. Effects of Resveratrol on Polycystic Ovary Syndrome: A Double-blind, Randomized, Placebo-controlled Trial. J. Clin. Endocrinol. Metab. 2016, 101, 4322–4328. [Google Scholar] [CrossRef]
- Wong, D.H.; Villanueva, J.A.; Cress, A.B.; Duleba, A.J. Effects of resveratrol on proliferation and apoptosis in rat ovarian theca-interstitial cells. Mol. Hum. Reprod. 2010, 16, 251–259. [Google Scholar] [CrossRef]
- Ortega, I.; Villanueva, J.A.; Wong, D.H.; Cress, A.B.; Sokalska, A.; Stanley, S.D.; Duleba, A.J. Resveratrol reduces steroidogenesis in rat ovarian theca-interstitial cells: The role of inhibition of Akt/PKB signaling pathway. Endocrinology 2012, 153, 4019–4029. [Google Scholar] [CrossRef]
- Crandall, J.; Oram, V.; Trandafirescu, G.; Reid, M.; Kishore, P.; Hawkins, M.; Cohen, H.W.; Barzilai, N. Pilot study of resveratrol in older adults with impaired glucose tolerance. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 1307–1312. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, X.; Ran, L.; Wan, J.; Wang, X.; Qin, Y.; Shu, F.; Gao, Y.; Yuan, L.; Zhang, Q.; et al. Resveratrol improves insulin resistance, glucose and lipid metabolism in patients with non-alcoholic fatty liver disease: A randomized controlled trial. Dig. Liver Dis. 2015, 47, 226–232. [Google Scholar] [CrossRef]
- Faghihzadeh, F.; Adibi, P.; Hekmatdoost, A. The effects of resveratrol supplementation on cardiovascular risk factors in patients with non-alcoholic fatty liver disease: A randomised, double-blind, placebo-controlled study. Br. J. Nutr. 2015, 114, 796–803. [Google Scholar] [CrossRef]
- Heebøll, S.; Kreuzfeldt, M.; Hamilton-Dutoit, S.; Kjær Poulsen, M.; Stødkilde-Jørgensen, H.; Møller, H.; Jessen, N.; Thorsen, K.; Kristina Hellberg, Y.; Bønløkke Pedersen, S.; et al. Placebo-controlled, randomised clinical trial: High-dose resveratrol treatment for non-alcoholic fatty liver disease. Scand. J. Gastroenterol. 2016, 51, 456–464. [Google Scholar] [CrossRef]
- Kurzthaler, D.; Hadziomerovic-Pekic, D.; Wildt, L.; Seeber, B.E. Metformin induces a prompt decrease in LH-stimulated testosterone response in women with PCOS independent of its insulin-sensitizing effects. Reprod. Biol. Endocrinol. 2014, 12, 98. [Google Scholar] [CrossRef]
- Hirsch, A.; Hahn, D.; Kempná, P.; Hofer, G.; Nuoffer, J.M.; Mullis, P.E.; Flück, C.E. Metformin inhibits human androgen production by regulating steroidogenic enzymes HSD3B2 and CYP17A1 and complex I activity of the respiratory chain. Endocrinology 2012, 153, 4354–4366. [Google Scholar] [CrossRef]
- Fullerton, M.D.; Steinberg, G.R. SIRT1 takes a backseat to AMPK in the regulation of insulin sensitivity by resveratrol. Diabetes 2010, 59, 551–553. [Google Scholar] [CrossRef]
- Lan, F.; Weikel, K.A.; Cacicedo, J.M.; Ido, Y. Resveratrol-Induced AMP-Activated Protein Kinase Activation Is Cell-Type Dependent: Lessons from Basic Research for Clinical Application. Nutrients 2017, 9, 751. [Google Scholar] [CrossRef]
- Baur, J.A.; Pearson, K.J.; Price, N.L.; Jamieson, H.A.; Lerin, C.; Kalra, A.; Prabhu, V.V.; Allard, J.S.; Lopez-Lluch, G.; Lewis, K.; et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006, 444, 337–342. [Google Scholar] [CrossRef]
- Goh, K.P.; Lee, H.Y.; Lau, D.P.; Supaat, W.; Chan, Y.H.; Koh, A.F. Effects of resveratrol in patients with type 2 diabetes mellitus on skeletal muscle SIRT1 expression and energy expenditure. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 2–13. [Google Scholar] [CrossRef]
- Zhu, X.; Wu, C.; Qiu, S.; Yuan, X.; Li, L. Effects of resveratrol on glucose control and insulin sensitivity in subjects with type 2 diabetes: Systematic review and meta-analysis. Nutr. Metab. (Lond.) 2017, 14, 60. [Google Scholar] [CrossRef]
- Chiba, T.; Tsuchiya, T.; Komatsu, T.; Mori, R.; Hayashi, H.; Shimokawa, I. Development of calorie restriction mimetics as therapeutics for obesity, diabetes, inflammatory and neurodegenerative diseases. Curr. Genom. 2010, 11, 562–567. [Google Scholar] [CrossRef]
- Djiogue, S.; Nwabo Kamdje, A.H.; Vecchio, L.; Kipanyula, M.J.; Farahna, M.; Aldebasi, Y.; Seke Etet, P.F. Insulin resistance and cancer: The role of insulin and IGFs. Endocr. Relat. Cancer 2013, 20, R1–R17. [Google Scholar] [CrossRef]
- Sun, W.; Lu, J.; Wu, S.; Bi, Y.; Mu, Y.; Zhao, J.; Liu, C.; Chen, L.; Shi, L.; Li, Q.; et al. Association of insulin resistance with breast, ovarian, endometrial and cervical cancers in non-diabetic women. Am. J. Cancer Res. 2016, 6, 2334–2344. [Google Scholar] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Perrotta, I.; Bruno, L.; Maltese, L.; Russo, E.; Donato, A.; Donato, G. Immunohistochemical analysis of the ubiquitin-conjugating enzyme UbcH10 in lung cancer: A useful tool for diagnosis and therapy. J. Histochem. Cytochem. 2012, 60, 359–365. [Google Scholar] [CrossRef]
- Brown, K.F.; Rumgay, H.; Dunlop, C.; Ryan, M.; Quartly, F.; Cox, A.; Deas, A.; Elliss-Brookes, L.; Gavin, A.; Hounsome, L.; et al. The fraction of cancer attributable to modifiable risk factors in England, Wales, Scotland, Northern Ireland, and the United Kingdom in 2015. Br. J. Cancer 2018, 118, 1130–1141. [Google Scholar] [CrossRef]
- Castelló, A.; Pollán, M.; Buijsse, B.; Ruiz, A.; Casas, A.M.; Baena-Cañada, J.M.; Lope, V.; Antolín, S.; Ramos, M.; Muñoz, M.; et al. Spanish Mediterranean diet and other dietary patterns and breast cancer risk: Case-control EpiGEICAM study. Br. J. Cancer 2014, 111, 1454–1462. [Google Scholar] [CrossRef]
- Toledo, E.; Salas-Salvadó, J.; Donat-Vargas, C.; Buil-Cosiales, P.; Estruch, R.; Ros, E.; Corella, D.; Fitó, M.; Hu, F.B.; Arós, F.; et al. Mediterranean Diet and Invasive Breast Cancer Risk Among Women at High Cardiovascular Risk in the PREDIMED Trial: A Randomized Clinical Trial. JAMA Intern. Med. 2015, 175, 1752–1760. [Google Scholar] [CrossRef]
- Koene, R.J.; Prizment, A.E.; Blaes, A.; Konety, S.H. Shared Risk Factors in Cardiovascular Disease and Cancer. Circulation 2016, 133, 1104–1114. [Google Scholar] [CrossRef]
- Masoudkabir, F.; Sarrafzadegan, N.; Gotay, C.; Ignaszewski, A.; Krahn, A.D.; Davis, M.K.; Franco, C.; Mani, A. Cardiovascular disease and cancer: Evidence for shared disease pathways and pharmacologic prevention. Atherosclerosis 2017, 263, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Freisling, H.; Viallon, V.; Lennon, H.; Bagnardi, V.; Ricci, C.; Butterworth, A.S.; Sweeting, M.; Muller, D.; Romieu, I.; Bazelle, P.; et al. Lifestyle factors and risk of multimorbidity of cancer and cardiometabolic diseases: A multinational cohort study. BMC Med. 2020, 18, 5. [Google Scholar] [CrossRef] [PubMed]
- Fortin, M.; Haggerty, J.; Almirall, J.; Bouhali, T.; Sasseville, M.; Lemieux, M. Lifestyle factors and multimorbidity: A cross sectional study. BMC Public Health 2014, 14, 686. [Google Scholar] [CrossRef]
- Dhalwani, N.N.; Zaccardi, F.; O’Donovan, G.; Carter, P.; Hamer, M.; Yates, T.; Davies, M.; Khunti, K. Association Between Lifestyle Factors and the Incidence of Multimorbidity in an Older English Population. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 528–534. [Google Scholar] [CrossRef]
- World Health Organization. Global Action Plan. for the Prevention and Control. of Noncommunicable Diseases 2013–2020; World Health Organization: Geneva, Switzerland, 2013; Available online: https://apps.who.int/iris/handle/10665/94384. (accessed on 24 February 2020).
- Xiao, Y.; Xia, J.; Li, L.; Ke, Y.; Cheng, J.; Xie, Y.; Chu, W.; Cheung, P.; Kim, J.H.; Colditz, G.A.; et al. Associations between dietary patterns and the risk of breast cancer: A systematic review and meta-analysis of observational studies. Breast Cancer Res. 2019, 21, 16. [Google Scholar] [CrossRef]
- Key, T.J. Endogenous oestrogens and breast cancer risk in premenopausal and postmenopausal women. Steroids 2011, 76, 812–815. [Google Scholar] [CrossRef]
- Gérard, C.; Brown, K.A. Obesity and breast cancer - Role of estrogens and the molecular underpinnings of aromatase regulation in breast adipose tissue. Mol. Cell Endocrinol. 2018, 466, 15–30. [Google Scholar] [CrossRef]
- Paonessa, F.; Foti, D.; Costa, V.; Chiefari, E.; Brunetti, G.; Leone, F.; Luciano, F.; Wu, F.; Lee, A.S.; Gulletta, E.; et al. Activator protein-2 overexpression accounts for increased insulin receptor expression in human breast cancer. Cancer Res. 2006, 66, 5085–5093. [Google Scholar] [CrossRef][Green Version]
- Sgarra, R.; Pegoraro, S.; Ros, G.; Penzo, C.; Chiefari, E.; Foti, D.; Brunetti, A.; Manfioletti, G. High Mobility Group A (HMGA) proteins: Molecular instigators of breast cancer onset and progression. Biochim. Biophys. Acta Rev. Cancer 2018, 1869, 216–229. [Google Scholar] [CrossRef]
- Laria, A.E.; Messineo, S.; Arcidiacono, B.; Varano, M.; Chiefari, E.; Semple, R.K.; Rocha, N.; Russo, D.; Cuda, G.; Gaspari, M.; et al. Secretome Analysis of Hypoxia-Induced 3T3-L1 Adipocytes Uncovers Novel Proteins Potentially Involved in Obesity. Proteomics 2018, 18, e1700260. [Google Scholar] [CrossRef]
- Chiefari, E.; Foti, D.P.; Sgarra, R.; Pegoraro, S.; Arcidiacono, B.; Brunetti, F.S.; Greco, M.; Manfioletti, G.; Brunetti, A. Transcriptional Regulation of Glucose Metabolism: The Emerging Role of the HMGA1 Chromatin Factor. Front. Endocrinol. (Lausanne) 2018, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Capurso, A.; Crepaldi, G.; Capurso, C. Epigenetics/Epigenomics of Olive Oil and the Mediterranean Diet. In Benefits of the Mediterranean Diet in the Elderly Patient; Practical Issues in Geriatrics; Springer: Cham, Switzerland, 2018; pp. 115–138. [Google Scholar] [CrossRef]
- Menendez, J.A.; Lupu, R. Mediterranean dietary traditions for the molecular treatment of human cancer: Anti-oncogenic actions of the main olive oil’s monounsaturated fatty acid oleic acid (18:1n-9). Curr. Pharm. Biotechnol. 2006, 7, 495–502. [Google Scholar] [CrossRef] [PubMed]
- D’Amore, S.; Vacca, M.; Cariello, M.; Graziano, G.; D’Orazio, A.; Salvia, R.; Sasso, R.C.; Sabbà, C.; Palasciano, G.; Moschetta, A. Genes and miRNA expression signatures in peripheral blood mononuclear cells in healthy subjects and patients with metabolic syndrome after acute intake of extra virgin olive oil. Biochim. Biophys. Acta 2016, 1861, 1671–1680. [Google Scholar] [CrossRef] [PubMed]
- Khymenets, O.; Fitó, M.; Covas, M.I.; Farré, M.; Pujadas, M.A.; Muñoz, D.; Konstantinidou, V.; de la Torre, R. Mononuclear cell transcriptome response after sustained virgin olive oil consumption in humans: An exploratory nutrigenomics study. OMICS 2009, 13, 7–19. [Google Scholar] [CrossRef]
- Chiefari, E.; Arcidiacono, B.; Palmieri, C.; Corigliano, D.M.; Morittu, V.M.; Britti, D.; Armoni, M.; Foti, D.P.; Brunetti, A. Cross-talk among HMGA1 and FoxO1 in control of nuclear insulin signaling. Sci. Rep. 2018, 8, 8540. [Google Scholar] [CrossRef]
- Penzo, C.; Arnoldo, L.; Pegoraro, S.; Petrosino, S.; Ros, G.; Zanin, R.; Wiśniewski, J.R.; Manfioletti, G.; Sgarra, R. HMGA1 Modulates Gene Transcription Sustaining a Tumor Signalling Pathway Acting on the Epigenetic Status of Triple-Negative Breast Cancer Cells. Cancers 2019, 11, 1105. [Google Scholar] [CrossRef]
- Balletshofer, B.M.; Rittig, K.; Stock, J.; Lehn-Stefan, A.; Overkamp, D.; Dietz, K.; Häring, H.U. Insulin resistant young subjects at risk of accelerated atherosclerosis exhibit a marked reduction in peripheral endothelial function early in life but not differences in intima-media thickness. Atherosclerosis 2003, 17, 303–309. [Google Scholar] [CrossRef]
- Mäkimattila, S.; Liu, M.L.; Vakkilainen, J.; Schlenzka, A.; Lahdenperä, S.; Syvänne, M.; Mäntysaari, M.; Summanen, P.; Bergholm, R.; Taskinen, M.R.; et al. Impaired endothelium-dependent vasodilation in type 2 diabetes. Relation to LDL size, oxidized LDL, and antioxidants. Diabetes Care 1999, 22, 973–981. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Larson, M.G.; Keyes, M.J.; Mitchell, G.F.; Vasan, R.S.; Keaney, J.F., Jr.; Lehman, B.T.; Fan, S.; Osypiuk, E.; Vita, J.A. Clinical correlates and heritability of flow-mediated dilation in the community: The Framingham Heart Study. Circulation 2004, 109, 613–619. [Google Scholar] [CrossRef]
- Lteif, A.A.; Han, K.; Mather, K.J. Obesity, insulin resistance, and the metabolic syndrome: Determinants of endothelial dysfunction in whites and blacks. Circulation 2005, 112, 32–38. [Google Scholar] [CrossRef]
- Balletshofer, B.M.; Rittig, K.; Enderle, M.D.; Volk, A.; Maerker, E.; Jacob, S.; Matthaei, S.; Rett, K.; Häring, H.U. Endothelial dysfunction is detectable in young normotensive first-degree relatives of subjects with type 2 diabetes in association with insulin resistance. Circulation 2000, 101, 1780–1784. [Google Scholar] [CrossRef] [PubMed]
- Kolluru, G.; Bir, S.; Kevil, C. Endothelial dysfunction and diabetes: Effects on angiogenesis, vascular remodeling, and wound healing. Int. J. Vasc. Med. 2012, 2012, 918267. [Google Scholar] [CrossRef] [PubMed]
- Palella, E.; Cimino, R.; Pullano, S.A.; Fiorillo, A.S.; Gulletta, E.; Brunetti, A.; Foti, D.P.; Greco, M. Laboratory parameters of hemostasis, adhesion molecules, and inflammation in type 2 diabetes mellitus: Correlation with glycemic control. Int. J. Environ. Res. Public Health 2020, 17, 300. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Christoph, M.; Hoffmann, G. Effects of olive oil on markers of inflammation and endothelial function—A systematic review and metaanalysis. Nutrients 2015, 7, 7651–7675. [Google Scholar] [CrossRef]
- Summerhill, V.; Karagodin, V.; Grechko, A.; Myasoedova, V.; Orekhov, A. Vasculoprotective Role of Olive Oil Compounds via Modulation of Oxidative Stress in Atherosclerosis. Front. Cardiovasc. Med. 2018, 5, 188. [Google Scholar] [CrossRef]
- Li, M.; Qian, M.; Kyler, K.; Xu, J. Endothelial-Vascular Smooth Muscle Cells Interactions in Atherosclerosis. Front. Cardiovasc. Med. 2018, 5, 151. [Google Scholar] [CrossRef]
- Wang, G.; Jacquet, L.; Karamariti, E.; Xu, Q. Origin and differentiation of vascular smooth muscle cells. J. Physiol. 2015, 593, 3013–3030. [Google Scholar] [CrossRef]
- Schlueter, C.; Hauke, S.; Loeschke, S.; Wenk, H.H.; Bullerdiek, J. HMGA1 proteins in human atherosclerotic plaques. Pathol. Res. Pract. 2005, 201, 101–107. [Google Scholar] [CrossRef]
- Iwai, K.; Morita, T.; Iritani, O.; Morimoto, S.; Nakamura, Y.; Ishigaki, Y. Abstract 11211: CTCF and HMGA1 Opposite Directionally Regulate the Gene Expression of microRNAs at Scaffold/ Matrix-attachment Regions in the Arterial Smooth Muscle Cell. Circulation 2013, 128, A11211. [Google Scholar]
- Gogoi, B.; Chatterjee, P.; Mukherjee, S.; Buragohain, A.K.; Bhattacharya, S.; Dasgupta, S. A polyphenol rescues lipid induced insulin resistance in skeletal muscle cells and adipocytes. Biochem. Biophys. Res. Commun. 2014, 452, 382–388. [Google Scholar] [CrossRef]
- Whelton, P.K.; Carey, R.M.; Aronow, W.S.; Casey, D.E., Jr.; Collins, K.J.; Dennison Himmelfarb, C.; DePalma, S.M.; Gidding, S.; Jamerson, K.A.; Jones, D.W.; et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension 2018, 71, 1269–1324. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J. Lifestyle modification as a means to prevent and treat high blood pressure. J. Am. Soc. Nephrol. 2003, 14, S99–S102. [Google Scholar] [CrossRef]
- Bendinelli, B.; Masala, G.; Bruno, R.M.; Caini, S.; Saieva, C.; Boninsegni, A.; Ungar, A.; Ghiadoni, L.; Palli, D. A priori dietary patterns and blood pressure in the EPIC Florence cohort: A cross-sectional study. Eur. J. Nutr. 2019, 58, 455–466. [Google Scholar] [CrossRef] [PubMed]
- Toledo, E.; Hu, F.; Estruch, R.; Buil-Cosiales, P.; Corella, D.; Salas-Salvadó, J.; Covas, M.I.; Arós, F.; Gómez-Gracia, E.; Fiol, M.; et al. Effect of the Mediterranean diet on blood pressure in the PREDIMED trial: Results from a randomized controlled trial. BMC Med. 2013, 11, 207. [Google Scholar] [CrossRef]
- Campesi, I.; Marino, M.; Cipolletti, M.; Romani, A.; Franconi, F. Put “gender glasses” on the effects of phenolic compounds on cardiovascular function and diseases. Eur. J. Nutr. 2018, 57, 2677–2691. [Google Scholar] [CrossRef] [PubMed]
- Galvão Cândido, F.; Xavier Valente, F.; da Silva, L.E.; Gonçalves Leão Coelho, O.; Gouveia Peluzio, M.D.C.; Gonçalves Alfenas, R.C. Consumption of extra virgin olive oil improves body composition and blood pressure in women with excess body fat: A randomized, double-blinded, placebo-controlled clinical trial. Eur. J. Nutr. 2018, 57, 2445–2455. [Google Scholar] [CrossRef] [PubMed]
- Storniolo, C.E.; Roselló-Catafau, J.; Pintó, X.; Mitjavila, M.T.; Moreno, J.J. Polyphenol fraction of extra virgin olive oil protects against endothelial dysfunction induced by high glucose and free fatty acids through modulation of nitric oxide and endothelin-1. Redox Biol. 2014, 2, 971–977. [Google Scholar] [CrossRef]
- Jiménez-Morales, A.I.; Ruano, J.; Delgado-Lista, J.; Fernandez, J.M.; Camargo, A.; López-Segura, F.; Villarraso, J.C.; Fuentes-Jiménez, F.; López-Miranda, J.; Pérez-Jiménez, F. NOS3 Glu298Asp polymorphism interacts with virgin olive oil phenols to determine the postprandial endothelial function in patients with the metabolic syndrome. J. Clin. Endocrinol. Metab. 2011, 96, E1694–E1702. [Google Scholar] [CrossRef]
- Imrie, H.; Abbas, A.; Kearney, M. Insulin resistance, lipotoxicity and endothelial dysfunction. Biochim. Biophys. Acta 2010, 1801, 320–326. [Google Scholar] [CrossRef]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2019, 140, e596–e646. [Google Scholar] [CrossRef]
- Bjornstad, P.; Eckel, R.H. Pathogenesis of Lipid Disorders in Insulin Resistance: A Brief Review. Curr. Diab. Rep. 2018, 18, 127. [Google Scholar] [CrossRef] [PubMed]
- Hernáez, Á.; Castañer, O.; Goday, A.; Ros, E.; Pintó, X.; Estruch, R.; Salas-Salvadó, J.; Corella, D.; Arós, F.; Serra-Majem, L.; et al. The Mediterranean Diet decreases LDL atherogenicity in high cardiovascular risk individuals: A randomized controlled trial. Mol. Nutr. Food Res. 2017, 61, 9. [Google Scholar] [CrossRef] [PubMed]
- Meisinger, C.; Baumert, J.; Khuseyinova, N.; Loewel, H.; Koenig, W. Plasma Oxidized Low-Density Lipoprotein, a Strong Predictor for Acute Coronary Heart Disease Events in Apparently Healthy, Middle-AgedMen from the General Population. Circulation 2005, 112, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Galeano, N.F.; Al-Haideri, M.; Keyserman, F.; Rumsey, S.C.; Deckelbaum, R.J. Small dense low density lipoprotein has increased affinity for LDL receptor-independent cell surface binding sites: A potential mechanism for increased atherogenicity. J. Lipid Res. 1998, 39, 1263–1273. [Google Scholar]
- Hoogeveen, R.C.; Gaubatz, J.W.; Sun, W.; Dodge, R.C.; Crosby, J.R.; Jiang, J.; Couper, D.; Virani, S.S.; Kathiresan, S.; Boerwinkle, E.; et al. Small dense low-density lipoprotein-cholesterol concentrations predict risk for coronary heart disease: The Atherosclerosis Risk in Communities (ARIC) study. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1069–1077. [Google Scholar] [CrossRef]
- Gaforio, J.J.; Visioli, F.; Alarcón-de-la-Lastra, C.; Castañer, O.; Delgado-Rodríguez, M.; Fitó, M.; Hernández, A.F.; Huertas, J.R.; Martínez-González, M.A.; Menendez, J.A.; et al. Virgin Olive Oil and Health: Summary of the III International Conference on Virgin Olive Oil and Health Consensus Report, JAEN (Spain) 2018. Nutrients 2019, 11, 2039. [Google Scholar] [CrossRef]
- Hernáez, Á.; Fernández-Castillejo, S.; Farràs, M.; Catalán, Ú.; Subirana, I.; Montes, R.; Solà, R.; Muñoz-Aguayo, D.; Gelabert-Gorgues, A.; Díaz-Gil, Ó.; et al. Olive Oil Polyphenols Enhance High-Density Lipoprotein Function in Humans: A Randomized Controlled Trial. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 2115–2119. [Google Scholar] [CrossRef]
- Fitó, M.; Cladellas, M.; de la Torre, R.; Martí, J.; Alcántara, M.; Pujadas-Bastardes, M.; Marrugat, J.; Bruguera, J.; López-Sabater, M.C.; Vila, J.; et al. Antioxidant effect of virgin olive oil in patients with stable coronary heart disease: A randomized, crossover, controlled, clinical trial. Atherosclerosis 2005, 181, 149–158. [Google Scholar] [CrossRef]
- Weinbrenner, T.; Fitó, M.; de la Torre, R.; Saez, G.T.; Rijken, P.; Tormos, C.; Coolen, S.; Albaladejo, M.F.; Abanades, S.; Schroder, H.; et al. Olive oils high in phenolic compounds modulate oxidative/antioxidative status in men. J. Nutr. 2004, 134, 2314–2321. [Google Scholar] [CrossRef]
- Marrugat, J.; Covas, M.I.; Fitó, M.; Schröder, H.; Miró-Casas, E.; Gimeno, E.; López-Sabater, M.C.; de la Torre, R.; Farré, M. SOLOS Investigators. Effects of differing phenolic content in dietary olive oils on lipids and LDL oxidation—A randomized controlled trial. Eur. J. Nutr. 2004, 43, 140–147. [Google Scholar] [CrossRef]
- Rader, D.J. Mediterranean Approach to Improving High-Density Lipoprotein Function. Circulation 2017, 135, 644–647. [Google Scholar] [CrossRef] [PubMed]
- Hernáez, Á.; Castañer, O.; Elosua, R.; Pintó, X.; Estruch, R.; Salas-Salvadó, J.; Corella, D.; Arós, F.; Serra-Majem, L.; Fiol, M.; et al. Mediterranean Diet Improves High-Density Lipoprotein Function in High-Cardiovascular-Risk Individuals: A Randomized Controlled Trial. Circulation 2017, 135, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Castro-Barquero, S.; Tresserra-Rimbau, A.; Vitelli-Storelli, F.; Doménech, M.; Salas-Salvadó, J.; Martín-Sánchez, V.; Rubín-García, M.; Buil-Cosiales, P.; Corella, D.; Fitó, M.; et al. Dietary Polyphenol Intake is Associated with HDL-Cholesterol and A Better Profile of other Components of the Metabolic Syndrome: A PREDIMED-Plus Sub-Study. Nutrients 2020, 12, 689. [Google Scholar] [CrossRef]
- Vitale, M.; Masulli, M.; Rivellese, A.A.; Bonora, E.; Cappellini, F.; Nicolucci, A.; Squatrito, S.; Antenucci, D.; Barrea, A.; Bianchi, C.; et al. Dietary intake and major food sources of polyphenols in people with type 2 diabetes: The TOSCA.IT Study. Eur. J. Nutr. 2018, 57, 679–688. [Google Scholar] [CrossRef]
- PREDIMED study investigators. Intake of Total Polyphenols and Some Classes of Polyphenols Is Inversely Associated with Diabetes in Elderly People at High Cardiovascular Disease Risk. J. Nutr. 2015, 146, 767–777. [Google Scholar] [CrossRef]
- Gansevoort, R.T.; Correa-Rotter, R.; Hemmelgarn, B.R.; Jafar, T.H.; Heerspink, H.J.; Mann, J.F.; Matsushita, K.; Wen, C.P. Chronic kidney disease and cardiovascular risk: Epidemiology, mechanisms, and prevention. Lancet 2013, 382, 339–352. [Google Scholar] [CrossRef]
- Kobayashi, S.; Maesato, K.; Moriya, H.; Ohtake, T.; Ikeda, T. Insulin resistance in patients with chronic kidney disease. Am. J. Kidney Dis. 2005, 45, 275–280. [Google Scholar] [CrossRef]
- Shinohara, K.; Shoji, T.; Emoto, M.; Tahara, H.; Koyama, H.; Ishimura, E.; Miki, T.; Tabata, T.; Nishizawa, Y. Insulin resistance as an independent predictor of cardiovascular mortality in patients with end-stage renal disease. J. Am. Soc. Nephrol. 2002, 13, 1894–1900. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Alvestrand, A.; Smith, D.; Hendler, R.; Hendler, E.; Wahren, J. Insulin resistance in uremia. J. Clin. Invest. 1981, 67, 563–568. [Google Scholar] [CrossRef]
- Gluba, A.; Mikhailidis, D.P.; Lip, G.Y.; Hannam, S.; Rysz, J.; Banach, M. Metabolic syndrome and renal disease. Int. J. Cardiol. 2013, 164, 141–150. [Google Scholar] [CrossRef]
- Teta, D. Insulin resistance as a therapeutic target for chronic kidney disease. J. Ren. Nutr. 2015, 25, 226–229. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Carrero, J.J. Insulin resistance in chronic kidney disease. Nephrology (Carlton) 2017, 22 (Suppl. 4), 31–34. [Google Scholar] [CrossRef] [PubMed]
- Schrauben, S.J.; Jepson, C.; Hsu, J.; Wilson, F.P.; Zhang, X.; Lash, J.P.; Robinson, B.M.; Townsend, R.R.; Chen, J.; Fogelfeld, L.; et al. Insulin resistance and chronic kidney disease progression, cardiovascular events, and death: Findings from the chronic renal insufficiency cohort study. BMC Nephrol. 2019, 20, 60. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.T.; Huang, J.W.; Chiang, C.K.; Yen, C.J.; Hung, K.Y.; Wu, K.D. Metabolic syndrome and insulin resistance as risk factors for development of chronic kidney disease and rapid decline in renal function in elderly. J. Clin. Endocrinol. Metab. 2012, 97, 1268–1276. [Google Scholar] [CrossRef]
- Kobayashi, H.; Tokudome, G.; Hara, Y.; Sugano, N.; Endo, S.; Suetsugu, Y.; Kuriyama, S.; Hosoya, T. Insulin resistance is a risk factor for the progression of chronic kidney disease. Clin. Nephrol. 2009, 71, 643–651. [Google Scholar] [CrossRef]
- Kelly, J.T.; Palmer, S.C.; Wai, S.N.; Ruospo, M.; Carrero, J.J.; Campbell, K.L.; Strippoli, G.F. Healthy dietary patterns and risk of mortality and ESRD in CKD: A meta-analysis of cohort studies. Clin. J. Am. Soc. Nephrol. 2017, 12, 272–279. [Google Scholar] [CrossRef]
- Chauveau, P.; Aparicio, M.; Bellizzi, V.; Campbell, K.; Hong, X.; Johansson, L.; Kolko, A.; Molina, P.; Sezer, S.; Wanner, C.; et al. Mediterranean diet as the diet of choice for patients with chronic kidney disease. Nephrol. Dial. Transpl. 2018, 33, 725–735. [Google Scholar] [CrossRef]
- Kushi, L.H.; Lenart, E.B.; Willett, W.C. Health implications of Mediterranean diets in light of contemporary knowledge. 2. Meat, wine, fats, and oils. Am. J. Clin. Nutr. 1995, 61 (Suppl. 6), 1416S–1427S. [Google Scholar] [CrossRef]
- Krishnamurthy, V.M.; Wei, G.; Baird, B.C.; Murtaugh, M.; Chonchol, M.B.; Raphael, K.L.; Greene, T.; Beddhu, S. High dietary fiber intake is associated with decreased inflammation and all-cause mortality in patients with chronic kidney disease. Kidney Int. 2012, 81, 300–306. [Google Scholar] [CrossRef]
- Williamson, G. The role of polyphenols in modern nutrition. Nutr. Bull. 2017, 42, 226–235. [Google Scholar] [CrossRef]
- Maruca, A.; Ambrosio, F.A.; Lupia, A.; Romeo, I.; Rocca, R.; Moraca, F.; Talarico, C.; Bagetta, D.; Catalano, R.; Costa, G.; et al. Computer-based techniques for lead identification and optimization I: Basics. Phys. Sci. Rev. 2019, 4, 6. [Google Scholar] [CrossRef]
- Garcia-Cortes, M.; Robles-Diaz, M.; Ortega-Alonso, A.; Medina-Caliz, I.; Andrade, R.J. Hepatotoxicity by dietary supplements: A tabular listing and clinical characteristics. Int. J. Mol. Sci. 2016, 17, 537. [Google Scholar] [CrossRef] [PubMed]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 87. [Google Scholar] [CrossRef] [PubMed]
- Bode, A.M.; Dong, Z. Toxic phytochemicals and their potential risks for human cancer. Cancer Prev. Res. (Phila) 2015, 8, 1–8. [Google Scholar] [CrossRef]
- Azqueta, A.; Collins, A. Polyphenols and DNA Damage: A Mixed Blessing. Nutrients 2016, 8, 785. [Google Scholar] [CrossRef]

| Design | Subject | Diet/Supplement | Main Findings | Ref. |
|---|---|---|---|---|
| Prospective cohort study | 1302 patients with CVD | MedDiet | Higher adherence to the MedDiet was associated with a lower all-cause and CVD related-mortality rate (−27% and −31%, respectively) over 3.78 years of follow up. | [37] |
| Prospective cohort study | 22,043 adults free of cancer, T2D or CVD | MedDiet | Higher adherence to the MedDiet was associated with a lower all-cause, cancer and CVD related-mortality rate (−25%, −24% and −33%, respectively) over 44 months of follow up. | [38] |
| RCT | 1294 elderly adults | MedDiet adapted for the adults ≥65 years of age | After 1-year follow up, adherence to the MedDiet was associated with decreased levels of SPB (−4.7 mmHg), whereas in control group participants, requested to continue with their usual eating habits, SBP increased by 0.9 mmHg. Differences in SBP between groups were significant for males, but not for females. | [39] |
| Prospective cohort study | 12,168 middle-aged adults without CVD | Plant-based diets | Higher adherence to an overall plant-based diet, or a provegetarian diet, was associated with a lower risk of CVD-related and all-cause mortality (−19% and −11%, respectively) over 19 years of follow up. | [42] |
| Metanalysis of RCTs | 1460 patients with T2D | MUFA-enriched diets vs. CHO-enriched diets | High-MUFA diets were associated with significant reductions in fasting plasma glucose (WMD −0.57 mmol/L), triglycerides (−0.31 mmol/L) body weight (−1.56 Kg), and SBP (−2.31 mmHg) along with significant increases in HDL cholesterol (0.06 mmol/L) when compared to high-CHO diets. | [43] |
| Prospective cohort study | 13,380 adults without T2D | MedDiet | Higher adherence to the MedDiet was associated with lower incidence of T2D over 4.4 years of follow up. A two-point increase in the adherence score was associated with a 35% relative risk reduction of developing T2D. | [44] |
| RCT | 418 middle-aged and elderly adults without T2D | EVOO-enriched (1 L/week) MedDiet vs. nut-enriched (30 g/day) MedDiet vs. low-fat diet | Adherence to both EVOO-enriched and nut-enriched MedDiets were associated with lower incidence of T2D over a median 4-year follow up (HR of T2D 0.49 and 0.48, respectively) when compared to low-fat diet. | [45] |
| Metanalysis of RCTs | 121,070 patients with T2D or at risk for T2D | Individual or total PUFA supplements | PUFA supplements had little or no effect on likelihood of T2D diagnosis (RR 1.00) or measures of glucose metabolism and IR (mean differences in HbA1c −0.02%; plasma glucose 0.04 mmol/L; fasting insulin 1.02; HOMA-IR 0.06). | [54] |
| Metanalysis of prospective cohort studies | 312,015 adults without T2D | High (33.2 to 1452.3 mg/day) vs. low (8.9 to 501.8 mg/day) total flavonoid intake | High total flavonoid intake was associated with decreased risk (−11%) of developing T2D during 4–28 years of follow-up, when compared to a low intake. Each 300 mg/day increment in flavonoids consumption was associated with 5% reduction in T2D risk. The protective effect was significant for anthocyanidins, flavan-3-ols, flavonols, and isoflavones. | [58] |
| Cross-sectional | 58 patients with NAFLD | MedDiet | Higher adherence to the MedDiet was negatively correlated to serum liver enzymes, fasting insulin, HOMA-IR and NAFLD severity, and positively correlated to serum adiponectin levels. Patients with NASH exhibited lower adherence to the MedDiet compared to those with simple steatosis. | [76] |
| Crossover RCT | 12 patients with NAFLD | MedDiet vs. low-fat high CHO diet | A 6-week MedDiet intervention enhanced the relative reduction in hepatic steatosis (−39 ± 4%) when compared to a low-fat high CHO diet (−7 ± 3%). Insulin sensitivity, assessed by hyperinsulinemic clamp, improved significantly with the MedDiet intervention, but not with the control diet. | [77] |
| RCT | 98 patients with moderate-severe NAFLD | Low-glycemic index MedDiet | Adherence to a low-glycemic index MedDiet significantly reduced the NAFLD score (−4.14) within 6 months. | [78] |
| Prospective intervention study | 44 patients with liver steatosis | MedDiet | Adherence to the MedDiet was associated with significant amelioration of clinical, biochemical, and inflammatory biomarkers of NAFLD after 24 weeks. STAT3 rs2293152 G-carriers experienced more beneficial changes at the end of the dietary intervention. | [79] |
| Prospective cohort study | 70 cognitively normal middle-aged adults | MedDiet | Lower adherence to the MedDiet was associated with progressive AD biomarkers abnormalities, including lower FDG-PET glucose metabolism, and higher β amyloid load in AD-affected brain regions. | [96] |
| Cross-sectional | 4447 adults without dementia or cerebrovascular disease | High quality vs. low quality diets | High quality diets, with high consumption of vegetables, fruit, whole grains, nuts, dairy, and fish and low intake of sugar-containing beverages, were related to larger brain volumes, gray matter volumes, white matter volumes, and hippocampal volumes. | [97] |
| RCT | 49 elderly patients with mild to moderate AD | Anthocyanin-rich cherry juice (200 mL/day) | Twelve-week intervention with cherry juice was associated with significant improvements in verbal fluency, short-term memory, long-term memory and SBP levels when compared to the control group. Markers of inflammation (CRP and IL-6) were unchanged. | [119] |
| RCT | 12 elderly patients with mild cognitive impairment | Flavonoids-rich concord grape juice (6–9 mL/Kg/day) | Twelve-week intervention with flavonoids-rich concord grape juice was associated with improvement in verbal learning and non-significant enhancement of verbal and spatial recall. A small increase in fasting insulin was also observed. | [120] |
| RCT | 44 elderly patients with AD | Coconut oil-enriched (40 mL/day) MedDiet | After 21 days of intervention with coconut oil-enriched MedDiet, improvements in episodic, temporal orientation, and semantic memory were observed, and these were more pronounced in women with mild-moderate disease. | [123] |
| Cross-sectional | 112 treatment-naïve women with PCOS | MedDiet | PCOS women showed higher testosterone levels, Ferriman–Gallwey/hirsutism score, fasting insulin, fasting glucose levels and HOMA-IR when compared with control healthy women. Despite no differences in energy intake, PCOS women consumed less EVOO, legumes, fish/seafood, and nuts with a higher quantity of simple carbohydrate, total fat, SFA when compared to controls. In PCOS women, adherence to the MedDiet was negatively associated with testosterone levels. | [135] |
| Prospective cohort study | 259 healthy premenopausal women | MedDiet | Adherence to the MedDiet was associated with decreased plasma biomarkers of lipoperoxidation and oxidative stress, such as 8-iso-PGF2α and 9-HODE, and increased levels of ascorbic acid. | [137] |
| RCT | 34 women with PCOS | 1500 mg/day of oral micronized trans-resveratrol | After 3 months, resveratrol treatment led to a significant decrease of total testosterone, dehydroepiandrosterone sulfate and fasting insulin (−23.1%, −22.2%, and −31.8%, respectively), along with an increase of the Insulin Sensitivity Index (66.3%), when compared to placebo. | [138] |
| RCT | 50 patients with NAFLD | 500 mg/day of resveratrol | After 12 weeks, resveratrol supplementation reduced liver enzymes and steatosis significantly more than placebo. BP, IR markers or body weight remained unchanged. | [143] |
| Metanalysis of RCTs | 283 patients with T2D | 8–3000 mg/day of resveratrol | Treatment with resveratrol significantly improved fasting plasma glucose (−0.29 mmol), insulin levels (−0.64 U/mL), HOMA-IR (−0.52), SBP and DBP (−0.58 and −0.43 mmHg, respectively) when compared to placebo. Subgroup analysis revealed that resveratrol supplementation doses ≥ 100 mg/day were associated with more favorable results. | [151] |
| Case-control | 1017 women newly diagnosed with breast cancer, and 1017 healthy matched controls | MedDiet | Compared with controls, women with incident breast cancer were more likely to adhere to a high-energy Western diet. Adherence to the MedDiet was associated with decreased risk of breast cancer (OR 0.56), that was more pronounced for triple-negative tumors (OR 0.32), whereas adherence to the Western diet was related to a higher risk (OR 1.46), especially in premenopausal women. | [158] |
| RCT | 4152 women at high CVD risk without breast cancer | EVOO-enriched (1 L/week) MedDiet vs. nut-enriched (30 g/day) MedDiet vs. low-fat diet | After a median follow up of 4.8 years, breast cancer rates (per 1000 person-years) were 1.1 for the EVOO-enriched MedDiet, 1.8 for the nut-enriched MedDiet, and 2.9 for the control low-fat diet, with adjusted HR for the EVOO-enriched and nut-enriched MediDiets of 0.32 and 0.59, respectively. | [159] |
| Prospective cohort study | 291,778 middle-aged adults free of cancer, T2D, CVD | MedDiet | After a median cohort follow up of 10.7 years, 22,185 primary cancers, 9016 CVD events and 10,295 new cases of T2D were identified. Adherence to the MedDiet significantly reduced the risk of developing CVD and T2D multimorbidity among cancer patients, with a HR of 0.89. | [162] |
| Metanalysis of observational studies | 43,285 women with breast cancer | Prudent diet vs. western diet | Adherence to a prudent diet was associated with 23% reduced risk of breast cancer in premenopausal women, irrespective of the hormonal receptor status. Adherence to the Western diet resulted in 20% increased risk of ER+ and/or PR+ breast cancer in post-menopausal women. | [166] |
| RCT | 7447 middle-aged and elderly patients at high CVD risk | EVOO-enriched (1 L/week) MedDiet vs. nut-enriched (30 g/day) MedDiet vs. low-fat diet | After a median follow up of 4 years, the percentage of participants with controlled BP increased in all groups. However, adherence to EVOO-enriched and nut-enriched MedDiets was associated with significantly lower DBP values (–1.53 and –0.65 mmHg, respectively) when compared to the control low-fat diet. | [196] |
| RCT | 41 overweight young adult women | EVOO-enriched (25 mL/day) vs. soybean oil-enriched (25 mL/day) energy restricted normal fat diet | After 9 weeks, adherence to the EVOO-enriched diet was associated with higher fat loss (−2.4 vs. −1.3 Kg) and reduced DBP levels (−5.1 vs. 0.3 mmHg), than the control diet. There was also a trend of reduction for the proinflammatory IL-1β with EVOO. | [198] |
| RCT | 210 patients at high CVD risk | EVOO-enriched (1 L/week) MedDiet vs. nut-enriched (30 g/day) MedDiet vs. low-fat diet | After 1 year of follow up, adherence to the EVOO-enriched MedDiet significantly increased LDL resistance against oxidation (+6.46%) and estimated LDL particle size (+3.06%), with respect to the low-fat-diet. Adherence to the nut-enriched MedDiet was not associated with changes in LDL traits. | [204] |
| RCT | 296 patients at high CVD risk | EVOO-enriched (1 L/week) MedDiet vs. nut-enriched (30 g/day) MedDiet vs. low-fat diet | After 1 year follow up, adherence to both the EVOO-enriched and nut-enriched MedDiets significantly increased CEC relative to baseline, by improving HDL oxidative status and composition. | [214] |
| Metanalysis of prospective cohort studies | 15,285 patients with CKD | Plant-based healthy diets | Adherence to plant-based healthy diets, including MedDiet, were consistently associated with lower all-cause mortality (−27%) when compared to other dietary patterns. There was no statistically significant association between adherence to healthy diets and risk of ESRD. | [228] |
| Design | Subject | Nutrient (Dose) | Main Findings | Ref. |
|---|---|---|---|---|
| In vivo | HFD-fed mice | PUFA-enriched oil (30% of total energy) | Restoration of HFD-induced glucose intolerance, vascular dysfunction and hypercholesterolemia, via enhanced Akt/PKB phosphorylation. | [46] |
| In vivo | Obese Zucker rats | PUFA-enriched oil (10% of total energy) | Restoration of obesity-induced glucose intolerance via enhanced Akt/PKB phosphorylation and mitochondria bioenergetics. | [47] |
| In vitro | L6 rat skeletal muscle cells | MUFAs and PUFAs (200–700 μM) | Dose-dependent enhancement and prolongation of insulin-induced Akt/PKB and ERK1/2 phosphorylation, via repression of PP2A activity. | [48] |
| In vitro | C2C12 mouse skeletal muscle cells; L6 rat skeletal muscle cells | Quercetin and quercetin 3-O-glycosides (25–100 μM) | Enhanced muscular glucose uptake (38%–59%) in the absence of insulin via activation of the AMPK pathway and translocation of GLUT4. | [59,60] |
| In vitro | Yeast α-glucosidase | Flavonoids (0–200 μM) | Strong inhibition of α-glucosidase activity (IC50 ≤ 200 μM). | [62] |
| In vivo | HFD-fed mice | Oleacein (20 mg/Kg/day) | Prevention of HFD-induced adiposity, hyperglycemia, hyperinsulinemia, hyperlipidemia and liver pathology via reduced FAS, SREBP-1, ERK, and p-ERK liver protein levels. No signs of oleacein-induced organ toxicity. | [85] |
| In vivo; in vitro | HFD-fed mice; 3T3-L1 mouse preadipocytes | Oleacein (20 mg/Kg/day; 0–100 µM) | In vivo prevention of HFD-induced increase of adipocyte size, adipose tissue inflammation and fibrosis, via reduction of PPARγ and SREBP-1 protein expression; enhancement of adiponectin production in adipose tissue and increased expression of GLUT4 in skeletal muscle cells. In vitro dose-dependent prevention of lipid droplets accumulation during adipocyte differentiation via inhibition of FAS and PPARγ. | [87] |
| In vivo; in vitro | APP/PS1 mice; SH-SY5Y human neuroblastoma cells | Cyanidin-3-O-glucopyranoside (5 mg/Kg/day; 25 μM) | In vivo amelioration of object recognition, spatial memory, behavioral abnormalities and glucose intolerance. In vitro cytoprotective effects against amyloid β-induced toxicity via PPARγ upregulation. | [107] |
| In vitro | Rat ovarian theca-interstitial cells | Resveratrol (30–100 µM) | Dose-dependent inhibition of cell growth and cell viability; counteraction of insulin-induced pro-proliferative and anti-apoptotic effects. | [139] |
| In vitro | Rat ovarian theca-interstitial cells | Resveratrol (1–10 µM) | Inhibition of androstenedione and androsterone production (−78% and −74%, respectively), via Akt/PKB signaling pathway. | [140] |
| In vitro | Human embryonic kidney 293T cells; Human epithelial HeLa cells; HUVEC human endothelial cells; 3T3-L1 mouse preadipocytes; CHO hamster ovarian cells | Resveratrol (50–100 μM) | Cell-specific activation of AMPK, that can be linked to ATP synthase inhibition (energy restriction-sensitive), SIRT1-LKB1 stimulation, or both mechanisms. | [148] |
| In vivo | HFD-fed mice | Resveratrol (5.2–22.4 mg/Kg/day) | Longer lifespan, increased insulin sensitivity, reduced IGF-1 levels, increased liver AMPK activity, decreased liver, heart and aorta pathology, increased mitochondrial number, and improved motor function under chronic high fat hypernutrition. | [149] |
| In vitro | HFD-fed Rats; L6 rat skeletal muscle cells | Ferulic acid (0.6 mg/Kg/day; 2–20 µg/mL) | In vivo prevention of IR in adipose tissue, stimulated by SFA oversupply, via fetuin-A downregulation. In vitro dose-dependent improvement of SFA-induced muscular IR via inhibition of PKCε phosphorylation and restoration of HMGA1-mediated transcription of the INSR gene. | [192] |
| In vitro | ECV304 human endothelial cells | Hydroxytyrosol and EVOO total polyphenol extract (10 µM) | Prevention of NO reduction and ET-1 synthesis, induced by elevated glucose and FFA concentrations, via PI3K/Akt modulation. | [199] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mirabelli, M.; Chiefari, E.; Arcidiacono, B.; Corigliano, D.M.; Brunetti, F.S.; Maggisano, V.; Russo, D.; Foti, D.P.; Brunetti, A. Mediterranean Diet Nutrients to Turn the Tide against Insulin Resistance and Related Diseases. Nutrients 2020, 12, 1066. https://doi.org/10.3390/nu12041066
Mirabelli M, Chiefari E, Arcidiacono B, Corigliano DM, Brunetti FS, Maggisano V, Russo D, Foti DP, Brunetti A. Mediterranean Diet Nutrients to Turn the Tide against Insulin Resistance and Related Diseases. Nutrients. 2020; 12(4):1066. https://doi.org/10.3390/nu12041066
Chicago/Turabian StyleMirabelli, Maria, Eusebio Chiefari, Biagio Arcidiacono, Domenica Maria Corigliano, Francesco Saverio Brunetti, Valentina Maggisano, Diego Russo, Daniela Patrizia Foti, and Antonio Brunetti. 2020. "Mediterranean Diet Nutrients to Turn the Tide against Insulin Resistance and Related Diseases" Nutrients 12, no. 4: 1066. https://doi.org/10.3390/nu12041066
APA StyleMirabelli, M., Chiefari, E., Arcidiacono, B., Corigliano, D. M., Brunetti, F. S., Maggisano, V., Russo, D., Foti, D. P., & Brunetti, A. (2020). Mediterranean Diet Nutrients to Turn the Tide against Insulin Resistance and Related Diseases. Nutrients, 12(4), 1066. https://doi.org/10.3390/nu12041066











