Effect of β-caryophyllene from Cloves Extract on Helicobacter pylori Eradication in Mouse Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Ethics
2.3. In Vitro Quantification of Colony Forming Units (CFUs)
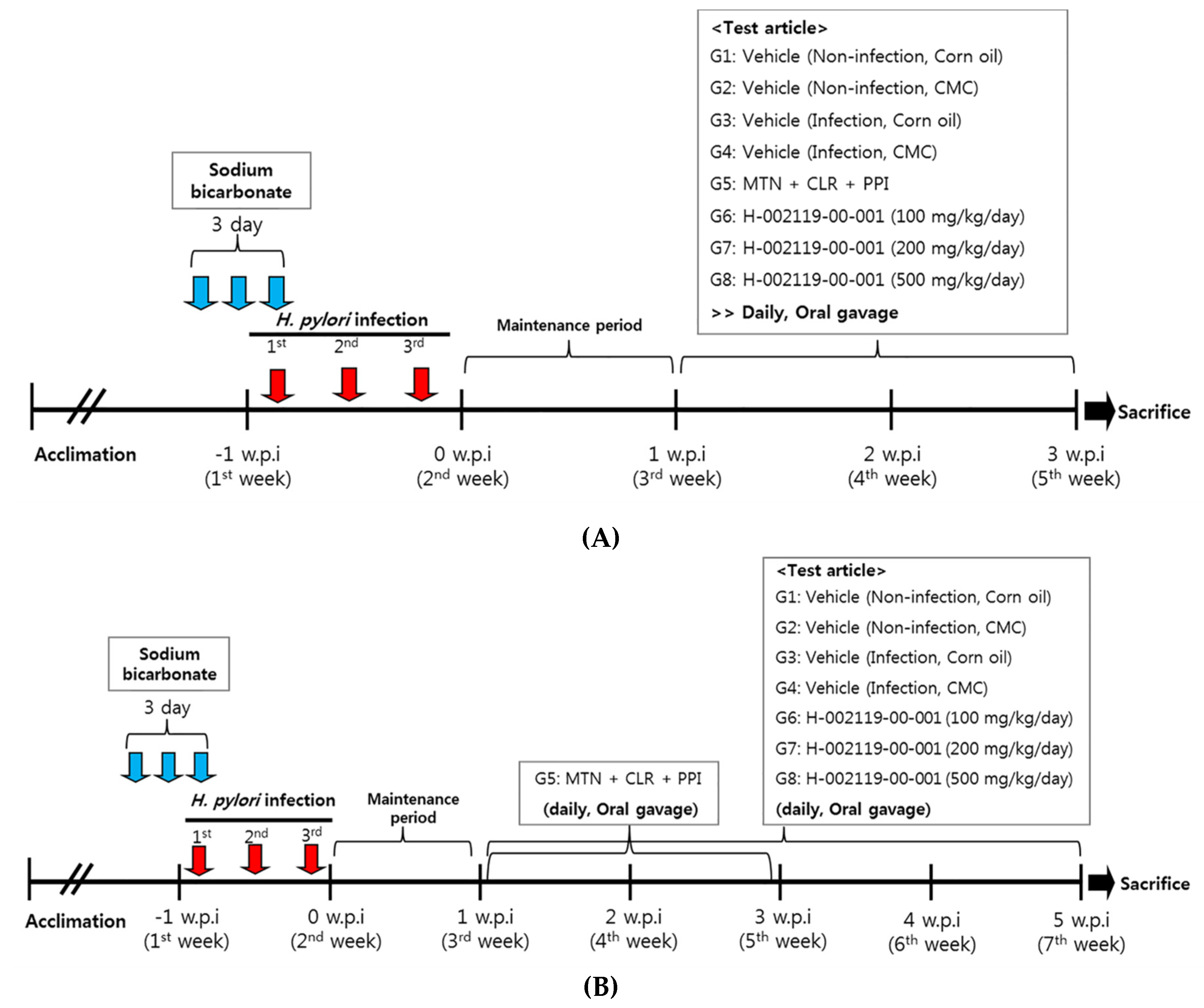
2.4. Inoculation of Experimental Animals
2.5. Distribution of Animals
2.6. Bacterial Identification
2.7. CLO Test
2.8. Real-Time Polymerase Chain Reaction (qpcr) for Identifying H. Pylori in Gastric Mucosa
2.9. Histopathological Assessment
2.9.1. Histological Evaluation
2.9.2. Immunohistochemistry
2.10. Statistical Analysis
3. Results
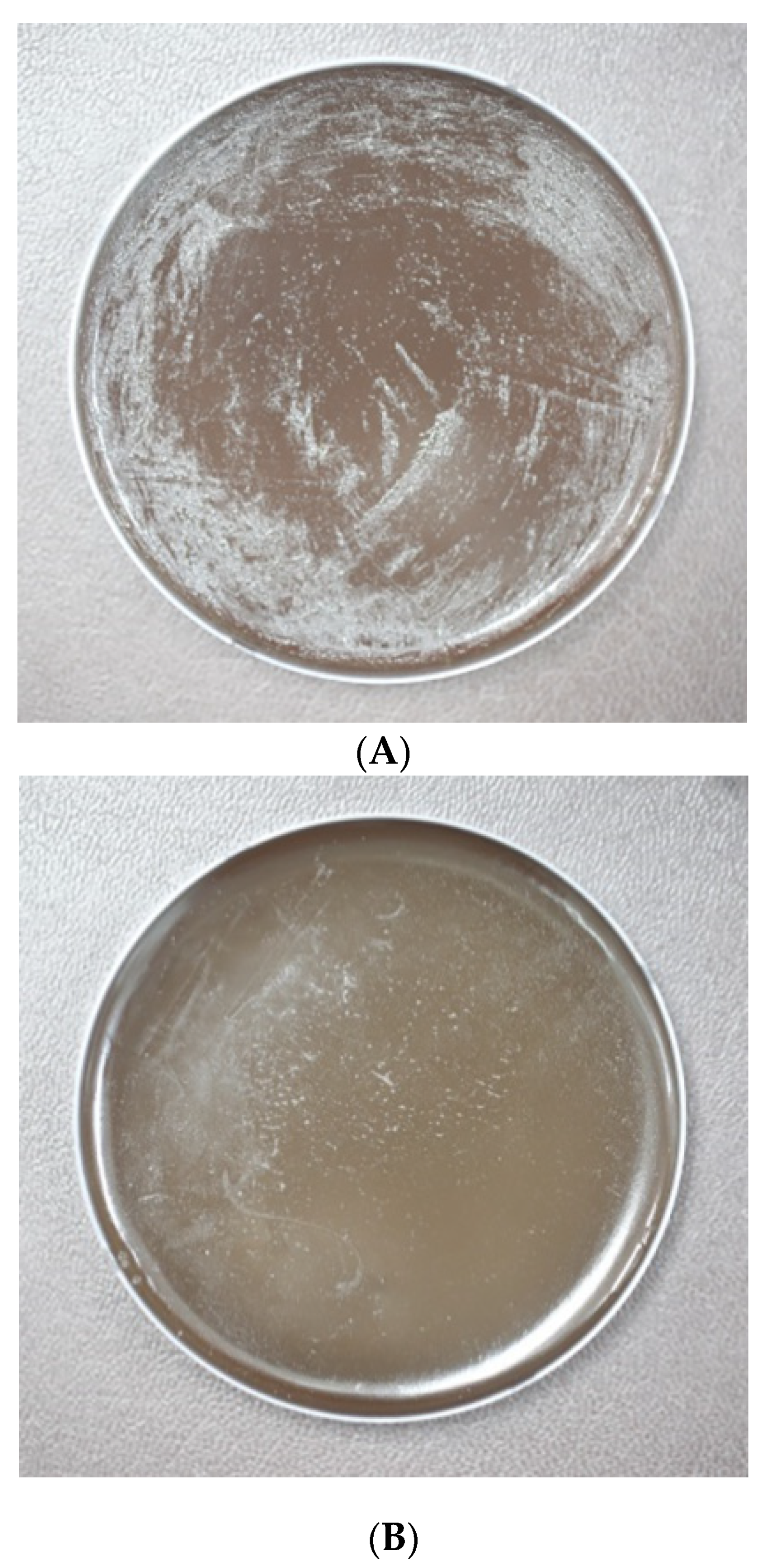
3.1. In Vitro Effect of H-002119-00-001 on Bacterial Colonization
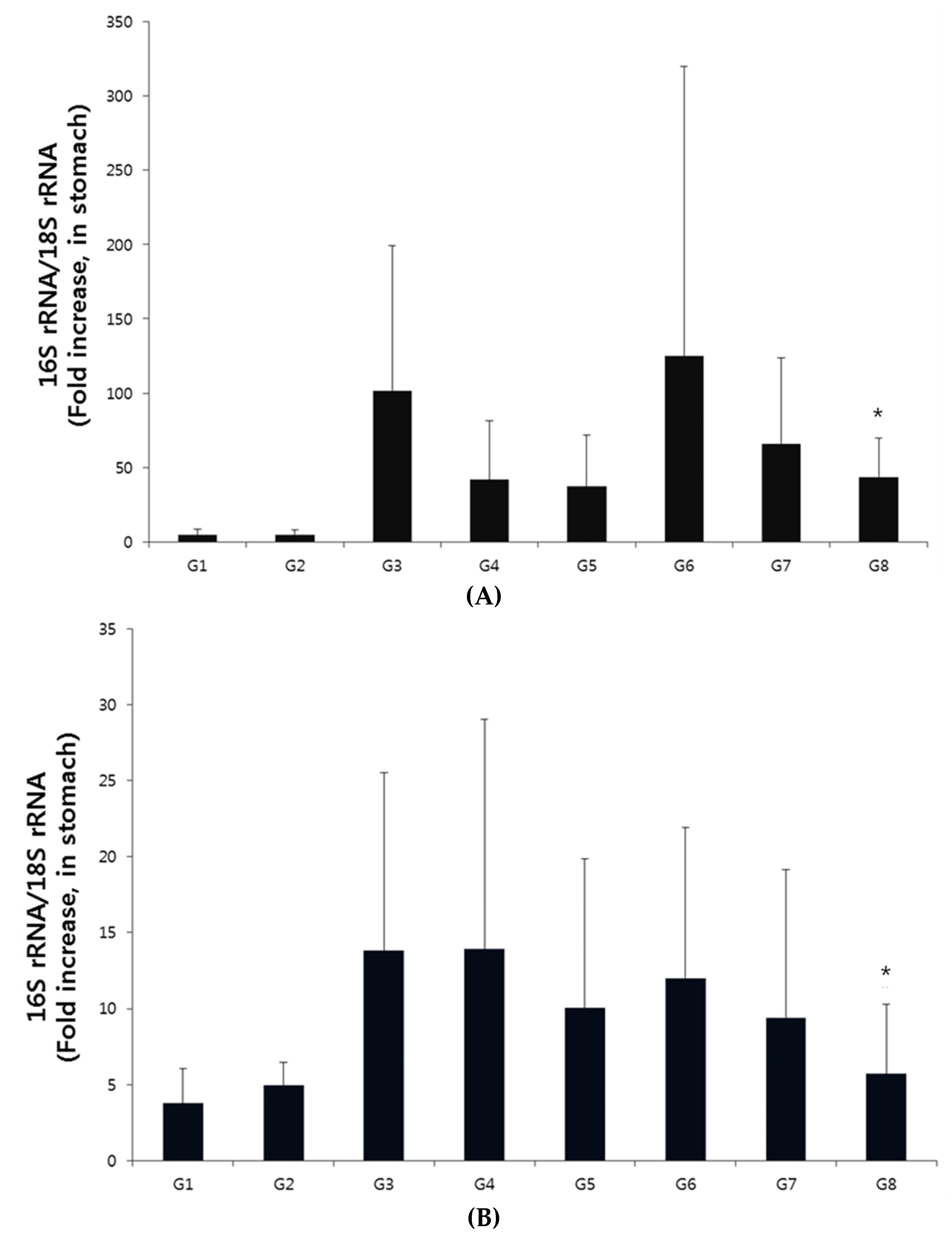
3.2. CLO Test and qPCR for H. Pylori in the Gastric Mucosa
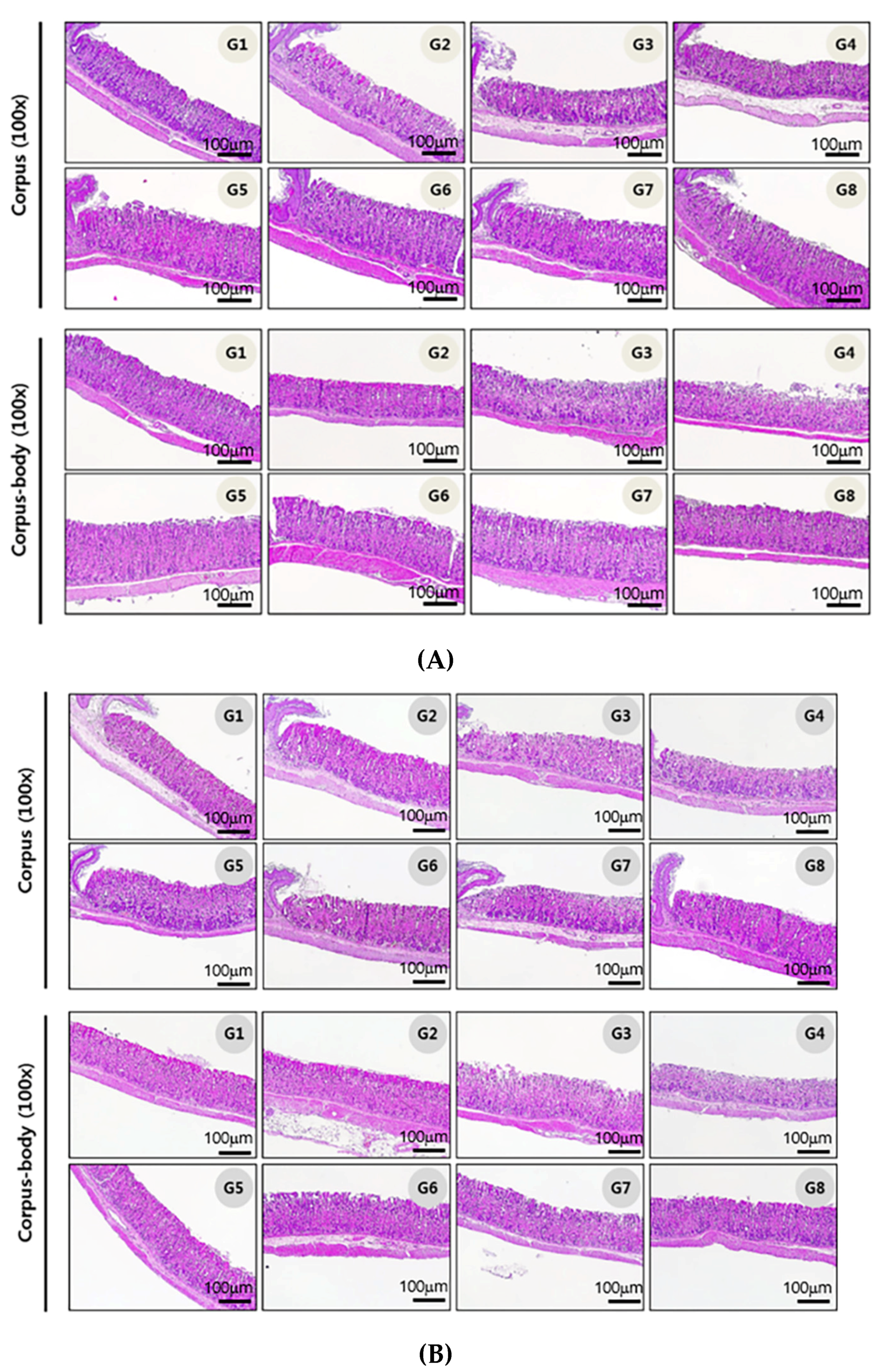
3.3. Gastric Histopathological Analysis
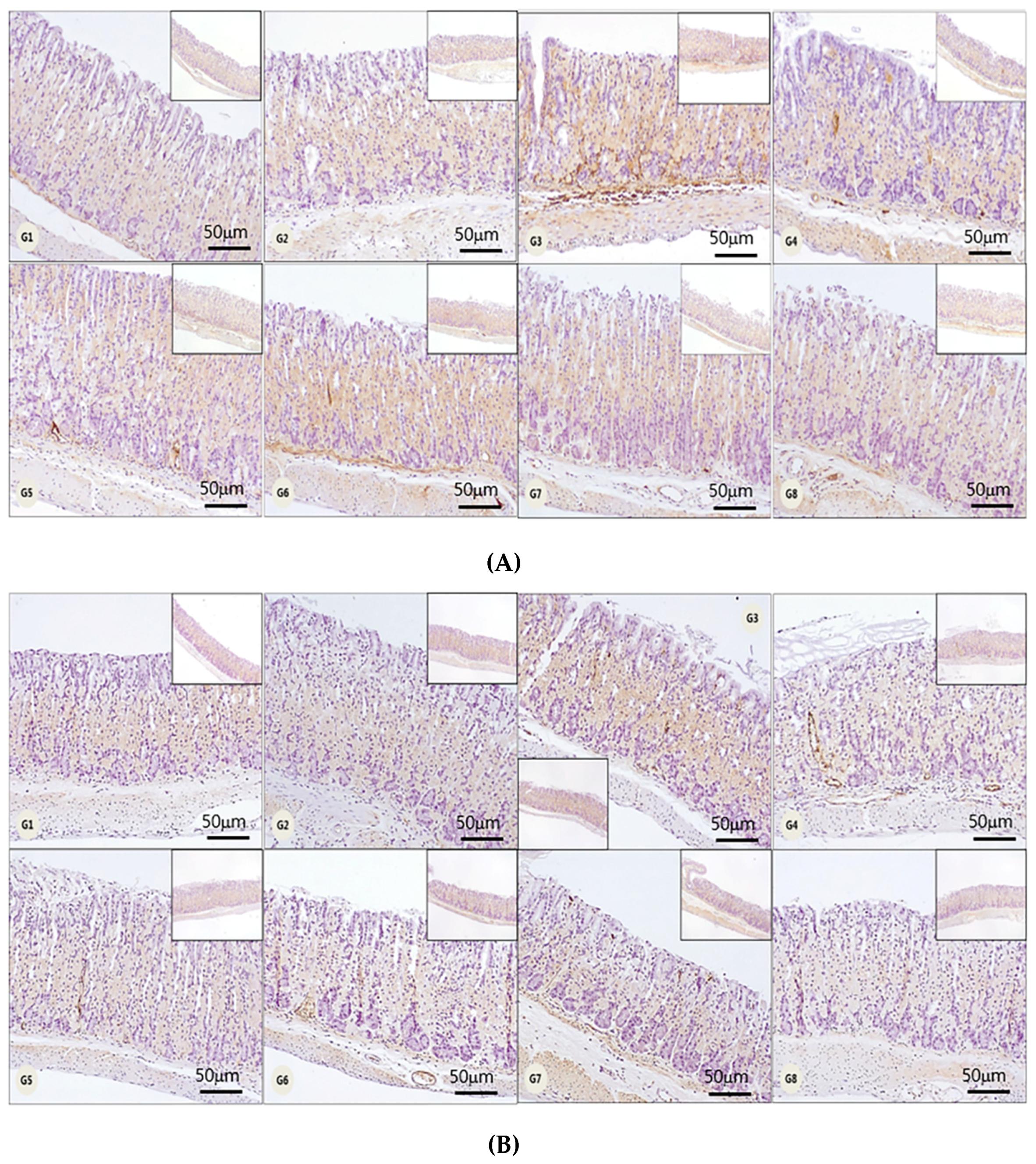
3.4. Gastric Tissue Immunostaining Assay
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Hooi, J.K.Y.; Lai, W.Y.; Ng, W.K.; Suen, M.M.Y.; Underwood, F.E.; Tanyingoh, D.; Malfertheiner, P.; Graham, D.Y.; Wong, V.W.S.; Wu, J.C.Y.; et al. Global Prevalence of Helicobacter pylori Infection: Systematic Review and Meta-Analysis. Gastroenterology 2017, 153, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Kusters, J.G.; van Vliet, A.H.M.; Kuipers, E.J. Pathogenesis of Helicobacter pylori infection. Clin. Microbiol. Rev. 2006, 19, 449–490. [Google Scholar] [CrossRef] [PubMed]
- Yucel, O. Prevention of Helicobacter pylori infection in childhood. World J. Gastroenterol. 2014, 20, 10348–10354. [Google Scholar] [CrossRef] [PubMed]
- Blanchard, T.G.; Czinn, S.J. Helicobacter pylori acquisition and transmission: Where does it all begin? Gastroenterology 2001, 121, 483–485. [Google Scholar] [CrossRef] [PubMed]
- Blaser, M.J.; Atherton, J.C. Helicobacter pylori persistence: Biology and disease. J. Clin. Invest. 2004, 113, 321–333. [Google Scholar] [CrossRef]
- Testerman, T.L.; Morris, J. Beyond the stomach: An updated view of Helicobacter pylori pathogenesis, diagnosis, and treatment. World J. Gastroenterol. 2014, 20, 12781–12808. [Google Scholar] [CrossRef]
- Marcus, E.A.; Sachs, G.; Scott, D.R. Eradication of Helicobacter pylori Infection. Curr. Gastroenterol. Rep. 2016, 18, 33. [Google Scholar] [CrossRef]
- Roesler, B.M.; Costa, S.C.B.; Zeitune, J.M.R. Eradication Treatment of Helicobacter pylori Infection: Its Importance and Possible Relationship in Preventing the Development of Gastric Cancer. ISRN Gastroenterol. 2012, 2012, 935410. [Google Scholar] [CrossRef][Green Version]
- Beales, I.L.P. Efficacy of Helicobacter pylori eradication therapies: A single centre observational study. BMC Gastroenterol. 2001, 1, 7. [Google Scholar] [CrossRef][Green Version]
- Yeo, Y.H.; Shiu, S.-I.; Ho, H.J.; Zou, B.; Lin, J.-T.; Wu, M.-S.; Liou, J.-M.; Wu, C.-Y. First-line Helicobacter pylori eradication therapies in countries with high and low clarithromycin resistance: A systematic review and network meta-analysis. Gut 2018, 67, 20. [Google Scholar] [CrossRef]
- De, R.; Kundu, P.; Swarnakar, S.; Ramamurthy, T.; Chowdhury, A.; Nair, G.B.; Mukhopadhyay, A.K. Antimicrobial activity of curcumin against Helicobacter pylori isolates from India and during infections in mice. Antimicrob. Agents Chemother. 2009, 53, 1592–1597. [Google Scholar] [CrossRef] [PubMed]
- Vítor, J.M.B.; Vale, F.F. Alternative therapies for Helicobacter pylori: Probiotics and phytomedicine. Pathog. Dis. 2011, 63, 153–164. [Google Scholar] [CrossRef]
- Goderska, K.; Agudo Pena, S.; Alarcon, T. Helicobacter pylori treatment: Antibiotics or probiotics. Appl. Microbiol. Biotechnol. 2018, 102, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tombola, F.; Campello, S.; De Luca, L.; Ruggiero, P.; Del Giudice, G.; Papini, E.; Zoratti, M. Plant polyphenols inhibit VacA, a toxin secreted by the gastric pathogen Helicobacter pylori. FEBS Lett. 2003, 543, 184–189. [Google Scholar] [CrossRef]
- Wang, Y.-C. Medicinal plant activity on Helicobacter pylori related diseases. World J. Gastroenterol. 2014, 20, 10368–10382. [Google Scholar] [CrossRef]
- Ruggiero, P.; Tombola, F.; Rossi, G.; Pancotto, L.; Lauretti, L.; Del Giudice, G.; Zoratti, M. Polyphenols reduce gastritis induced by Helicobacter pylori infection or VacA toxin administration in mice. Antimicrob. Agents Chemother. 2006, 50, 2550–2552. [Google Scholar] [CrossRef][Green Version]
- Opdyke, D.L. Monographs on fragrance raw materials. Food Cosmet. Toxicol. 1973, 11, 1011–1081. [Google Scholar] [CrossRef]
- Prashar, A.; Locke, I.C.; Evans, C.S. Cytotoxicity of clove (Syzygium aromaticum) oil and its major components to human skin cells. Cell Prolif. 2006, 39, 241–248. [Google Scholar] [CrossRef]
- Demirci, B.; Baser, K.H.; Demirci, F.; Hamann, M.T. New caryophyllene derivatives from Betula litwinowii. J. Nat. Prod. 2000, 63, 902–904. [Google Scholar] [CrossRef]
- Agarwal, R.B.; Rangari, V.D. Phytochemical investigation and evaluation of anti-inflammatory and anti-arthritic activities of essential oil of Strobilanthus ixiocephala Benth. Indian J. Exp. Biol. 2003, 41, 890–894. [Google Scholar]
- Cho, J.Y.; Chang, H.J.; Lee, S.K.; Kim, H.J.; Hwang, J.K.; Chun, H.S. Amelioration of dextran sulfate sodium-induced colitis in mice by oral administration of beta-caryophyllene, a sesquiterpene. Life Sci. 2007, 80, 932–939. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Marimuthu, P.; De Heluani, C.S.; Catalan, C.A.N. Antioxidant and biocidal activities of Carum nigrum (seed) essential oil, oleoresin, and their selected components. J. Agric. Food Chem. 2006, 54, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Kubo, I.; Chaudhuri, S.K.; Kubo, Y.; Sanchez, Y.; Ogura, T.; Saito, T.; Ishikawa, H.; Haraguchi, H. Cytotoxic and antioxidative sesquiterpenoids from Heterotheca inuloides. Planta Med. 1996, 62, 427–430. [Google Scholar] [CrossRef]
- Pichette, A.; Larouche, P.L.; Lebrun, M.; Legault, J. Composition and antibacterial activity of Abies balsamea essential oil. Phytother. Res. 2006, 20, 371–373. [Google Scholar] [CrossRef] [PubMed]
- Sipponen, P.; Price, A.B. The Sydney System for classification of gastritis 20 years ago. J. Gastroenterol. Hepatol. 2011, 26, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Sidahmed, H.M.A.; Hashim, N.M.; Mohan, S.; Abdelwahab, S.I.; Taha, M.M.E.; Dehghan, F.; Yahayu, M.; Ee, G.C.L.; Loke, M.F.; Vadivelu, J. Evidence of the gastroprotective and anti- Helicobacter pylori activities of β-mangostin isolated from Cratoxylum arborescens (vahl) blume. Drug Des. Dev. Ther. 2016, 10, 297–313. [Google Scholar] [CrossRef]
- Hitkova, I.; Yuan, G.; Anderl, F.; Gerhard, M.; Kirchner, T.; Reu, S.; Röcken, C.; Schäfer, C.; Schmid, R.M.; Vogelmann, R.; et al. Caveolin-1 protects B6129 mice against Helicobacter pylori gastritis. PLoS Pathog. 2013, 9, e1003251. [Google Scholar] [CrossRef] [PubMed]
- Ghelardini, C.; Galeotti, N.; Di Cesare Mannelli, L.; Mazzanti, G.; Bartolini, A. Local anaesthetic activity of beta-caryophyllene. Farmaco 2001, 56, 387–389. [Google Scholar] [CrossRef]
- Ormeno, E.; Baldy, V.; Ballini, C.; Fernandez, C. Production and diversity of volatile terpenes from plants on calcareous and siliceous soils: Effect of soil nutrients. J. Chem. Ecol. 2008, 34, 1219–1229. [Google Scholar] [CrossRef]
- Imai, H.; Osawa, K.; Yasuda, H.; Hamashima, H.; Arai, T.; Sasatsu, M. Inhibition by the essential oils of peppermint and spearmint of the growth of pathogenic bacteria. Microbios 2001, 106 (Suppl. 1), 31–39. [Google Scholar]
- Kalpoutzakis, E.; Aligiannis, N.; Mentis, A.; Mitaku, S.; Charvala, C. Composition of the essential oil of two Nepeta species and in vitro evaluation of their activity against Helicobacter pylori. Planta Med. 2001, 67, 880–883. [Google Scholar] [CrossRef]
- Ohno, T.; Kita, M.; Yamaoka, Y.; Imamura, S.; Yamamoto, T.; Mitsufuji, S.; Kodama, T.; Kashima, K.; Imanishi, J. Antimicrobial activity of essential oils against Helicobacter pylori. Helicobacter 2003, 8, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Klauke, A.L.; Racz, I.; Pradier, B.; Markert, A.; Zimmer, A.M.; Gertsch, J.; Zimmer, A. The cannabinoid CB(2) receptor-selective phytocannabinoid beta-caryophyllene exerts analgesic effects in mouse models of inflammatory and neuropathic pain. Eur. Neuropsychopharmacol. 2014, 24, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Bahi, A.; Al Mansouri, S.; Al Memari, E.; Al Ameri, M.; Nurulain, S.M.; Ojha, S. beta-Caryophyllene, a CB2 receptor agonist produces multiple behavioral changes relevant to anxiety and depression in mice. Physiol. Behav. 2014, 135, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.J.; Kim, J.M.; Lee, J.C.; Kim, W.K.; Chun, H.S. Protective effect of beta-caryophyllene, a natural bicyclic sesquiterpene, against cerebral ischemic injury. J. Med. Food 2013, 16, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Dong, Z.; Liu, S. beta-Caryophyllene ameliorates the Alzheimer-like phenotype in APP/PS1 Mice through CB2 receptor activation and the PPARgamma pathway. Pharmacology 2014, 94, 1–12. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, C.C.; de Oliveira, C.V.; Grigoletto, J.; Ribeiro, L.R.; Funck, V.R.; Grauncke, A.C.; de Souza, T.L.; Souto, N.S.; Furian, A.F.; Menezes, I.R.; et al. Anticonvulsant activity of beta-caryophyllene against pentylenetetrazol-induced seizures. Epilepsy Behav. 2016, 56, 26–31. [Google Scholar] [CrossRef]
- Sain, S.; Naoghare, P.K.; Devi, S.S.; Daiwile, A.; Krishnamurthi, K.; Arrigo, P.; Chakrabarti, T. Beta caryophyllene and caryophyllene oxide, isolated from Aegle marmelos, as the potent anti-inflammatory agents against lymphoma and neuroblastoma cells. Antiinflamm. Antiallergy Agents Med. Chem. 2014, 13, 45–55. [Google Scholar] [CrossRef]
- Suarez, G.; Reyes, V.E.; Beswick, E.J. Immune response to H. pylori. World J. Gastroenterol. 2006, 12, 5593–5598. [Google Scholar] [CrossRef]
| Group | H. pylori Infection | Treatment | 2-Week Treatment | 4-Week Treatment | ||||
|---|---|---|---|---|---|---|---|---|
| n | Percentage of Animals Negative by CLO Test (%) | CLO Score | n | Percentage of Animals Negative by CLO Test (%) | CLO Score | |||
| I | no | corn oil | 10 | 100 | 0.00 ± 0.00 | 10 | 100 | 0.00 ± 0.00 |
| II | no | 0.5% CMC | 9 | 100 | 0.00 ± 0.00 | 9 | 100 | 0.00 ± 0.00 |
| III | yes | corn oil | 10 | 0 | 3.00 ± 0.00 | 10 | 30 | 2.10 ± 1.45 |
| IV | yes | 0.5% CMC | 9 | 0 | 2.78 ± 0.44 | 10 | 40 | 1.70 ± 1.49 |
| V | yes | MTN + CLR + PPI | 10 | 80 | 0.20 ± 0.42 | 10 | 70 | 0.90 ± 1.45 |
| VI | yes | 100 mg/kg H-002119-00-001 | 10 | 40 | 1.30 ± 1.25 * | 10 | 60 | 1.20 ± 1.55 |
| VII | yes | 200 mg/kg H-002119-00-001 | 10 | 60 | 0.50 ± 0.71 * | 9 | 56 | 1.11 ± 1.45 |
| VIII | yes | 500 mg/kg H-002119-00-001 | 10 | 70 | 0.50 ± 0.85 * | 10 | 80 | 0.50 ± 1.08 * |
| Group | 2-Week Treatment | 4-Week Treatment | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | Damage of the Surface Epithelium | Inflammatory Cell Infiltration | Submucosal Edema | Total Score | n | Damage of the Surface Epithelium | Inflammatory Cell Infiltration | Submucosal Edema | Total Score | |
| I | 10 | 0.25 ± 0.35 | 0.25 ± 0.26 | 0.10 ± 0.21 | 0.60 ± 0.61 | 10 | 0.05 ± 0.16 | 0.15 ± 0.24 | 0.15 ± 0.24 | 0.35 ± 0.47 |
| II | 9 | 0.28 ± 0.26 | 0.28 ± 0.26 | 0.11 ± 0.22 | 0.67 ± 0.50 | 9 | 0.11 ± 0.22 | 0.22 ± 0.26 | 0.17 ± 0.25 | 0.50 ± 0.43 |
| III | 10 | 0.65 ± 0.34 | 0.50 ± 0.33 | 0.55 ± 0.37 | 1.70 ± 0.67 | 10 | 0.65 ± 0.24 | 0.45 ± 0.37 | 0.45 ± 0.28 | 1.55 ± 0.55 |
| IV | 9 | 0.61 ± 0.33 | 0.56 ± 0.30 | 0.44 ± 0.46 | 1.61 ± 0.74 | 10 | 0.60 ± 0.32 | 0.55 ± 0.37 | 0.50 ± 0.41 | 1.65 ± 0.88 |
| V | 10 | 0.30 ± 0.42 | 0.55 ± 0.44 | 0.15 ± 0.24 | 1.00 ± 0.78 | 10 | 0.35 ± 0.34 | 0.40 ± 0.39 | 0.35 ± 0.34 | 1.10 ± 0.77 |
| VI | 10 | 0.30 ± 0.35 * | 0.40 ± 0.32 | 0.25 ± 0.35 * | 0.95 ± 0.60 * | 10 | 0.40 ± 0.21 * | 0.40 ± 0.32 | 0.35 ± 0.34 | 1.15 ± 0.67 |
| VII | 10 | 0.35 ± 0.34 * | 0.25 ± 0.35 | 0.25 ± 0.35 * | 0.85 ± 0.88 * | 9 | 0.39 ± 0.42 | 0.44 ± 0.39 | 0.39 ± 0.33 | 1.22 ± 1.00 |
| VIII | 10 | 0.25 ± 0.26 * | 0.25 ± 0.26 * | 0.15 ± 0.34 * | 0.65 ± 0.63 * | 10 | 0.25 ± 0.26 * | 0.35 ± 0.24 | 0.25 ± 0.42 | 0.85 ± 0.53 * |
| Group | 2-Week Treatment | 4-Week Treatment | ||
|---|---|---|---|---|
| n | F4/80 Score | n | F4/80 Score | |
| I | 10 | 0.10 ± 0.21 | 10 | 0.10 ± 0.21 |
| II | 9 | 0.06 ± 0.17 | 9 | 0.15 ± 0.24 |
| III | 10 | 0.65 ± 0.34 | 10 | 0.58 ± 0.20 |
| IV | 9 | 0.44 ± 0.30 | 10 | 0.35 ± 0.34 |
| V | 10 | 0.55 ± 0.44 | 10 | 0.25 ± 0.35 |
| VI | 10 | 0.35 ± 0.24 * | 10 | 0.35 ± 0.41 |
| VII | 10 | 0.20 ± 0.26 * | 9 | 0.22 ± 0.26 |
| VIII | 10 | 0.25 ± 0.26 * | 10 | 0.20 ± 0.26 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, D.H.; Park, M.H.; Kim, C.J.; Lee, J.Y.; Keum, C.Y.; Kim, I.S.; Yun, C.-H.; Kim, S.-k.; Kim, W.H.; Lee, Y.C. Effect of β-caryophyllene from Cloves Extract on Helicobacter pylori Eradication in Mouse Model. Nutrients 2020, 12, 1000. https://doi.org/10.3390/nu12041000
Jung DH, Park MH, Kim CJ, Lee JY, Keum CY, Kim IS, Yun C-H, Kim S-k, Kim WH, Lee YC. Effect of β-caryophyllene from Cloves Extract on Helicobacter pylori Eradication in Mouse Model. Nutrients. 2020; 12(4):1000. https://doi.org/10.3390/nu12041000
Chicago/Turabian StyleJung, Da Hyun, Mi Hee Park, Chul Jin Kim, Jin Young Lee, Chang Yeop Keum, In Seon Kim, Chang-Hyun Yun, Sung-kyu Kim, Won Ho Kim, and Yong Chan Lee. 2020. "Effect of β-caryophyllene from Cloves Extract on Helicobacter pylori Eradication in Mouse Model" Nutrients 12, no. 4: 1000. https://doi.org/10.3390/nu12041000
APA StyleJung, D. H., Park, M. H., Kim, C. J., Lee, J. Y., Keum, C. Y., Kim, I. S., Yun, C.-H., Kim, S.-k., Kim, W. H., & Lee, Y. C. (2020). Effect of β-caryophyllene from Cloves Extract on Helicobacter pylori Eradication in Mouse Model. Nutrients, 12(4), 1000. https://doi.org/10.3390/nu12041000




