Low Energy Availability in Athletes 2020: An Updated Narrative Review of Prevalence, Risk, Within-Day Energy Balance, Knowledge, and Impact on Sports Performance
Abstract
1. Introduction
2. Methodology
3. Results
3.1. Low Energy Availability
3.2. Low Energy Availability Risk
3.2.1. Eating Disorders and Exercise Addiction
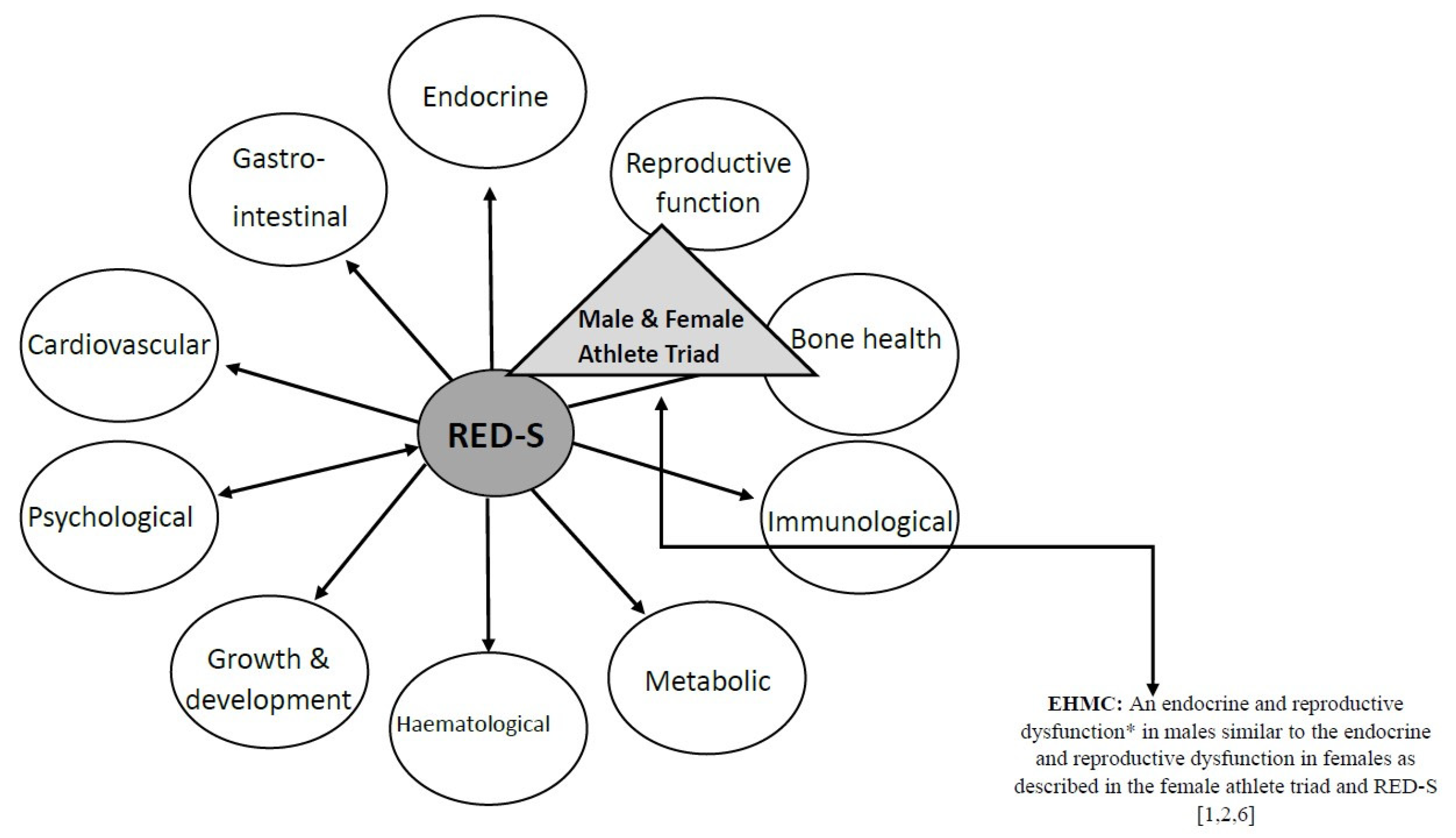
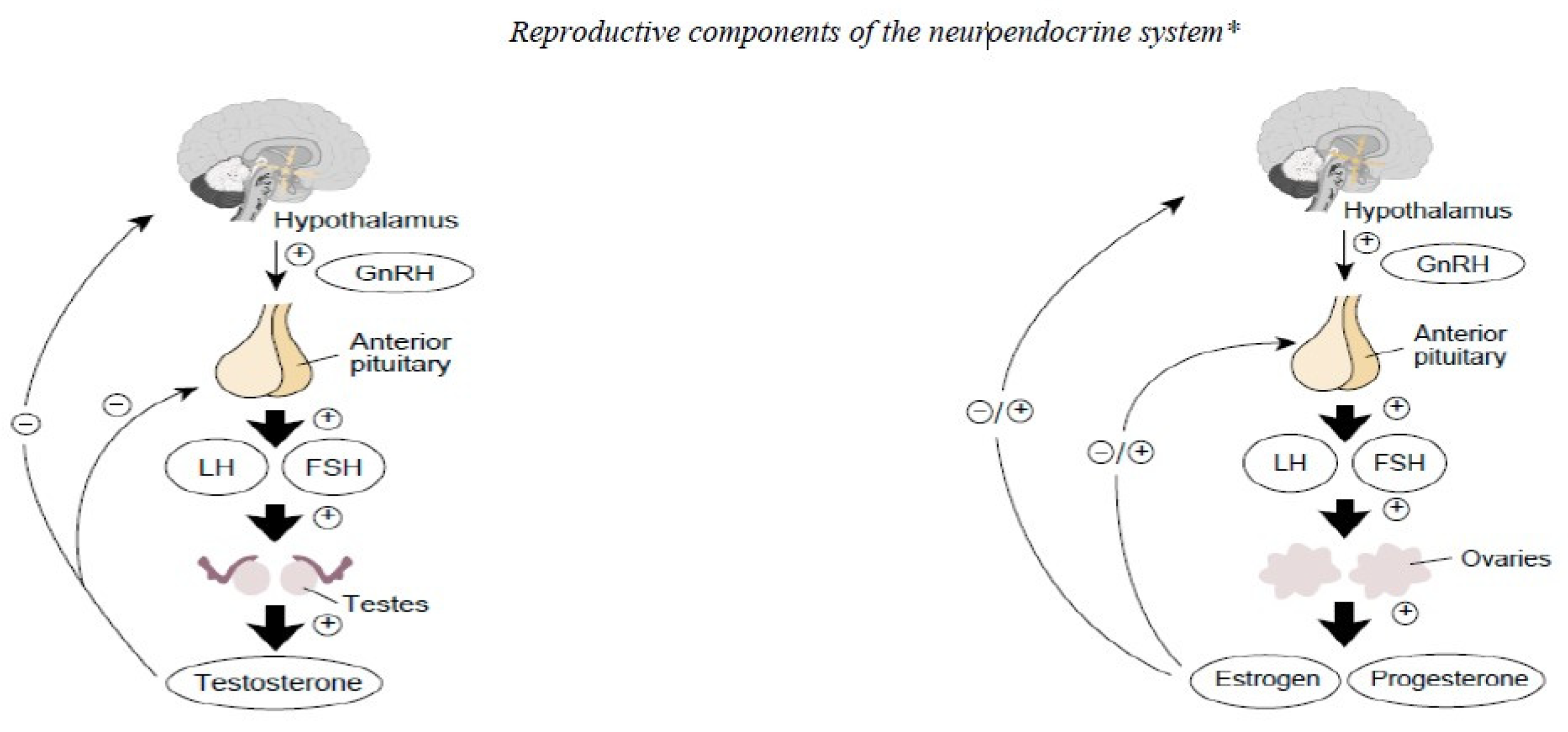
3.2.2. Exercise Hypogonadal Male Condition
4. Within-Day Energy Deficiency in Athletes
5. Low Energy Availability and Sports Performance
6. Knowledge of Low Energy Availability and Relative Energy Deficiency in Sport
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mountjoy, M.; Sundgot-Borgen, J.; Burke, L.; Carter, S.; Constantini, N.; Lebrun, C.; Meyer, N.; Sherman, R.; Steffen, K.; Budgett, R.; et al. The IOC consensus statement: Beyond the female athlete triad—relative energy deficiency in sport (RED-S). Br. J. Sports Med. 2014, 48, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Mountjoy, M.; Sundgot-Borgen, J.K.; Burke, L.M.; Ackerman, K.E.; Blauwet, C.; Constantini, N.; Lebrun, C.; Lundy, B.; Melin, A.K.; Meyer, N.L.; et al. IOC consensus statement on relative energy deficiency in sport (RED-S): 2018 update. Br. J. Sports Med. 2018, 52, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Loucks, A.B.; Kiens, B.; Wright, H.H. Energy availability in athletes. J. Sports Sci. 2011, 29 (Suppl. 1), S7–S15. [Google Scholar] [CrossRef] [PubMed]
- Raysmith, B.P.; Drew, M.K. Performance success or failure is influenced by weeks lost to injury and illness in elite Australian track and field athletes: A 5-year prospective study. J. Sci. Med. Sport 2016, 19, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Ackerman, K.E.; Holtzman, B.; Cooper, K.M.; Flynn, E.F.; Bruinvels, G.; Tenforde, A.S.; Popp, K.L.; Popp, K.L.; Simpkin, A.J.; Parziale, A.L. Low energy availability surrogates correlate with health and performance consequences of Relative Energy Deficiency in Sport. Br. J. Sports Med. 2019, 53, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Hackney, A.C.; Lane, A.R.; Register-Mihalik, J.; O’Leary, B.C. Endurance exercise training and male sexual libido. Med. Sci. Sports Exerc. 2017, 49, 1383–1388. [Google Scholar] [CrossRef] [PubMed]
- Logue, D.; Madigan, S.M.; Delahunt, E.; Heinen, M.; McDonnell, S.J.; Corish, C.A. Low energy availability in athletes: A review of prevalence, dietary patterns, physiological health, and sports performance. Sports Med. 2018, 48, 73–96. [Google Scholar] [CrossRef]
- Condo, D.; Lohman, R.; Kelly, M.; Carr, A. Nutritional intake, sports nutrition knowledge and energy availability in female australian rules football players. Nutrients 2019, 11, 971. [Google Scholar] [CrossRef]
- Keay, N.; Francis, G.; Entwistle, I.; Hind, K. Clinical evaluation of education relating to nutrition and skeletal loading in competitive male road cyclists at risk of relative energy deficiency in sports (RED-S): 6-month randomised controlled trial. BMJ Open Sport Exerc. Med. 2019, 5, e000523. [Google Scholar] [CrossRef]
- Kroshus, E.; DeFreese, J.D.; Kerr, Z.Y. Collegiate athletic trainers’ knowledge of the female athlete triad and relative energy deficiency in sport. J. Athl Train. 2018, 53, 51–59. [Google Scholar] [CrossRef]
- Krick, R.L.; Brown, A.F.; Brown, K.N. Increased female athlete triad knowledge following a brief video educational intervention. J. Nutr. Educ. Behav. 2019, 51, 1126–1129. [Google Scholar] [CrossRef] [PubMed]
- Harbour, R.; Miller, J. A new system for grading recommendations in evidence based guidelines. BMJ 2001, 323, 334–336. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.G.; Silva, H.H.; Paiva, T. Sleep duration, body composition, dietary profile and eating behaviours among children and adolescents: A comparison between Portuguese acrobatic gymnasts. Eur. J. Pediatr. 2018, 177, 815–825. [Google Scholar] [CrossRef] [PubMed]
- Civil, R.; Lamb, A.; Loosmore, D.; Ross, L.; Livingstone, K.; Strachan, F.; Dick, J.R.; Stevenson, E.J.; Brown, M.A.; Witard, O.C. Assessment of dietary intake, energy status, and factors associated with RED-S in vocational female ballet students. Front Nutr. 2018, 5, 136. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.A.; Howatson, G.; Quin, E.; Redding, E.; Stevenson, E.J. Energy intake and energy expenditure of pre-professional female contemporary dancers. PLoS ONE 2017, 12, e0171998. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.B.; Richmond, S.R.; Smith, C.R.; Currier, B.; Stecker, R.A.; Gieske, B.T.; Kemp, K.; Witherbee, K.E.; Kerksick, C.M. Physiologic, metabolic, and nutritional attributes of collegiate synchronized swimmers. Int. J. Sports Physiol. Perform. 2019, 14, 658–664. [Google Scholar] [CrossRef]
- Zanders, B.R.; Currier, B.S.; Harty, P.S.; Zabriskie, H.A.; Smith, C.R.; Stecker, R.A.; Richmond, S.R.; Jagim, A.R.; Kerksick, C.M. Changes in energy expenditure, dietary intake, and energy availability across an entire collegiate women’s basketball season. J. Strength Cond. Res. 2018, 17. [Google Scholar] [CrossRef]
- Ong, J.L.; Brownlee, I.A. Energy expenditure, availability, and dietary intake assessment in competitive female dragon boat athletes. Sports 2017, 5, 45. [Google Scholar] [CrossRef]
- Zabriskie, H.A.; Currier, B.S.; Harty, P.S.; Stecker, R.A.; Jagim, A.R.; Kerksick, C.M. Energy status and body composition across a collegiate women’s lacrosse season. Nutrients 2019, 11, 470. [Google Scholar] [CrossRef]
- Cherian, K.S.; Sainoji, A.; Nagalla, B.; Yagnambhatt, V.R. Energy balance coexists With disproportionate macronutrient consumption across pretraining, during training, and posttraining among indian junior soccer players. Pediatr. Exerc. Sci. 2018, 30, 506–515. [Google Scholar] [CrossRef]
- Braun, H.; von Andrian-Werburg, J.; Schänzer, W.; Thevis, M. Nutrition status of young elite female german football players. Pediatr. Exerc. Sci. 2017, 30, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.G.; Silva, H.H. Comparison of body composition and nutrients’ deficiencies between Portuguese rink-hockey players. Eur. J. Pediatr. 2017, 176, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Heikura, I.A.; Burke, L.M.; Bergland, D.; Uusitalo, A.L.T.; Mero, A.A.; Stellingwerff, T. Impact of energy availability, health, and sex on hemoglobin-mass responses following live-high-train-high altitude training in elite female and male distance athletes. Int. J. Sports Physiol. Perform. 2018, 13, 1090–1096. [Google Scholar] [CrossRef] [PubMed]
- Heikura, I.A.; Uusitalo, A.L.T.; Stellingwerff, T.; Bergland, D.; Mero, A.A.; Burke, L.M. Low energy availability is difficult to assess but outcomes have large impact on bone injury rates in elite distance athletes. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 403–411. [Google Scholar] [CrossRef] [PubMed]
- McCormack, W.P.; Shoepe, T.C.; LaBrie, J.; Almstedt, H.C. Bone mineral density, energy availability, and dietary restraint in collegiate cross-country runners and non-running controls. Eur. J. Appl. Physiol. 2019, 119, 1747–1756. [Google Scholar] [CrossRef] [PubMed]
- Koehler, K.; Achtzehn, S.; Braun, H.; Mester, J.; Schaenzer, W. Comparison of self-reported energy availability and metabolic hormones to assess adequacy of dietary energy intake in young elite athletes. Appl. Physiol. Nutr. Metab. 2013, 38, 725–733. [Google Scholar] [CrossRef]
- Melin, A.; Tornberg, Å.B.; Skouby, S.; Møller, S.S.; Sundgot-Borgen, J.; Faber, J.; Sidelmann, J.J.; Aziz, M.; Sjödin, A. Energy availability and the female athlete triad in elite endurance athletes. Scand. J. Med. Sci. Sports 2015, 25, 610–622. [Google Scholar] [CrossRef]
- Williams, N.I.; Leidy, H.J.; Hill, B.R.; Lieberman, J.L.; Legro, R.S.; De Souza, M.J. Magnitude of daily energy deficit predicts frequency but not severity of menstrual disturbances associated with exercise and caloric restriction. Am. J. Physiol. Endocrinol. Metab. 2015, 308, E29–E39. [Google Scholar] [CrossRef]
- Reed, J.L.; De Souza, M.J.; Mallinson, R.J.; Scheid, J.L.; Williams, N.I. Energy availability discriminates clinical menstrual status in exercising women. J. Int. Soc. Sports Nutr. 2015, 12, 11. [Google Scholar] [CrossRef]
- Melin, A.; Tornberg, A.B.; Skouby, S.; Faber, J.; Ritz, C.; Sjödin, A.; Sundgot-Borgen, J. The LEAF questionnaire: A screening tool for the identification of female athletes at risk for the female athlete triad. Br. J. Sports Med. 2014, 48, 540–545. [Google Scholar] [CrossRef]
- Joy, E.; De Souza, M.J.; Nattiv, A.; Misra, M.; Williams, N.I.; Mallinson, R.J.; Gibbs, J.C.; Olmsted, M.; Goolsby, M.; Matheson, G. 2014 female athlete triad coalition consensus statement on treatment and return to play of the female athlete triad. Curr. Sports Med. Rep. 2014, 13, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Mountjoy, M.; Sundgot-Borgen, J.; Burke, L.; Carter, S.; Constantini, N.; Lebrun, C.; Meyer, N.; Sherman, R.; Steffen, K.; Budgett, R.; et al. RED-S CAT. Relative Energy Deficiency in Sport (RED-S) Clinical Assessment Tool (CAT). Br. J. Sports Med. 2015. [Google Scholar] [CrossRef] [PubMed]
- Black, K.; Slater, J.; Brown, R.C.; Cooke, R. Low energy availability, plasma lipids, and hormonal profiles of recreational athletes. J. Strength Cond. Res. 2018, 32, 2816–2824. [Google Scholar] [CrossRef] [PubMed]
- Brook, E.M.; Tenforde, A.S.; Broad, E.M.; Matzkin, E.G.; Yang, H.Y.; Collins, J.E.; Blauwet, C.A. Low energy availability, menstrual dysfunction, and impaired bone health: A survey of elite para athletes. Scand. J. Med. Sci. Sports 2019, 29, 678–685. [Google Scholar] [CrossRef]
- Holtzman, B.; Tenforde, A.S.; Parziale, A.L.; Ackerman, K.E. Characterization of risk quantification differences using female athlete triad cumulative risk assessment and relative energy deficiency in sport clinical assessment tool. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 569–575. [Google Scholar] [CrossRef]
- Nose-Ogura, S.; Yoshino, O.; Dohi, M.; Kigawa, M.; Harada, M.; Hiraike, O.; Onda, T.; Osuga, Y.; Fujii, T.; Saito, S. Risk factors of stress fractures due to the female athlete triad: Differences in teens and twenties. Scand. J. Med. Sci. Sports 2019, 29, 1501–1510. [Google Scholar] [CrossRef]
- Logue, D.M.; Madigan, S.M.; Heinen, M.; McDonnell, S.J.; Delahunt, E.; Corish, C.A. Screening for risk of low energy availability in athletic and recreationally active females in Ireland. Eur. J. Sport Sci. 2018, 19, 112–122. [Google Scholar] [CrossRef]
- Staal, S.; Sjödin, A.; Fahrenholtz, I.; Bonnesen, K.; Melin, A.K. Low RMRratio as a surrogate marker for energy deficiency, the choice of predictive equation vital for correctly identifying male and female ballet dancers at risk. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 412–418. [Google Scholar] [CrossRef]
- Wilson, G.; Martin, D.; Morton, J.P.; Close, G.L. Male flat jockeys do not display deteriorations in bone density or resting metabolic rate in accordance with race riding experience: Implications for RED-S. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 434–439. [Google Scholar] [CrossRef]
- Drew, M.K.; Vlahovich, N.; Hughes, D.; Appaneal, R.; Peterson, K.; Burke, L.; Lundy, B.; Toomey, M.; Watts, D.; Lovell, G.; et al. A multifactorial evaluation of illness risk factors in athletes preparing for the Summer Olympic Games. J. Sci. Med. Sport 2017, 20, 745–750. [Google Scholar] [CrossRef]
- Sygo, J.; Coates, A.M.; Sesbreno, E.; Mountjoy, M.L.; Burr, J.F. Prevalence of indicators of low energy availability in elite female sprinters. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Schofield, K.L.; Thorpe, H.; Sims, S.T. Resting metabolic rate prediction equations and the validity to assess energy deficiency in the athlete population. Exp. Physiol. 2019, 104, 469–475. [Google Scholar] [CrossRef] [PubMed]
- Bone, J.L.; Burke, L.M. No difference in young adult athletes’ resting energy expenditure when measured under inpatient or outpatient conditions. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 464–467. [Google Scholar] [CrossRef] [PubMed]
- Mountjoy, M.L.; Burke, L.M.; Stellingwerff, T.; Sundgot-Borgen, J. Relative energy deficiency in sport: The tip of an iceberg. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 313–315. [Google Scholar] [CrossRef] [PubMed]
- Burke, L.M.; Lundy, B.; Fahrenholtz, I.L.; Melin, A.K. Pitfalls of conducting and interpreting estimates of energy availability in free-living athletes. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 350–363. [Google Scholar] [CrossRef] [PubMed]
- Sundgot-Borgen, J.; Torstveit, M.K. Prevalence of eating disorders in elite athletes is higher than in the general population. Clin. J. Sport Med. 2004, 14, 25–32. [Google Scholar] [CrossRef]
- Lichtenstein, M.B.; Hinze, C.J.; Emborg, B.; Thomsen, F.; Hemmingsen, S.D. Compulsive exercise: Links, risks and challenges faced. Psychol. Res. Behav. Manag. 2017, 10, 85–95. [Google Scholar] [CrossRef]
- Turton, R.; Goodwin, H.; Meyer, C. Athletic identity, compulsive exercise and eating psychopathology in long-distance runners. Eat Behav. 2017, 26, 129–132. [Google Scholar] [CrossRef]
- Torstveit, M.K.; Fahrenholtz, I.L.; Lichtenstein, M.B.; Stenqvist, T.B.; Melin, A.K. Exercise dependence, eating disorder symptoms and biomarkers of Relative Energy Deficiency in Sports (RED-S) among male endurance athletes. BMJ Open Sport Exerc. Med. 2019, 5, e000439. [Google Scholar] [CrossRef]
- Nattiv, A.; Loucks, A.B.; Manore, M.M.; Sanborn, C.F.; Sundgot-Borgen, J.; Warren, M.P. American college of sports medicine position stand. The female athlete triad. Med. Sci. Sports Exerc. 2007, 39, 1867–1882. [Google Scholar]
- Loucks, A.B.; Verdun, M.; Heath, E.M. Low energy availability, not stress of exercise, alters LH pulsatility in exercising women. J. Appl. Physiol. 1998, 84, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Slater, J.; Brown, R.; McLay-Cooke, R.; Black, K. Low energy availability in exercising women: Historical perspectives and future directions. Sports Med. 2017, 47, 207–220. [Google Scholar] [CrossRef] [PubMed]
- Hilton, L.K.; Loucks, A.B. Low energy availability, not exercise stress, suppresses the diurnal rhythm of leptin in healthy young women. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E43–E49. [Google Scholar] [CrossRef] [PubMed]
- Hackney, A.C.; Fahrner, C.L.; Stupnicki, R. Reproductive hormonal responses to maximal exercise in endurance-trained men with low resting testosterone levels. Exp. Clin. Endocrinol. Diabetes 1997, 105, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Fahrenholtz, I.L.; Sjödin, A.; Benardot, D.; Tornberg, Å.B.; Skouby, S.; Faber, J.; Sundgot-Borgen, J.K.; Melin, A.K. Within-day energy deficiency and reproductive function in female endurance athletes. Scand. J. Med. Sci. Sports 2018, 28, 1139–1146. [Google Scholar] [CrossRef]
- Torstveit, M.K.; Fahrenholtz, I.; Stenqvist, T.B.; Sylta, Ø.; Melin, A. Within-day energy deficiency and metabolic perturbation in male endurance athletes. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 419–427. [Google Scholar] [CrossRef]
- Hawley, J.A.; Burke, L.M. Effect of meal frequency and timing on physical performance. Br. J. Nutr. 1997, 77 (Suppl. 1), S91–S103. [Google Scholar] [CrossRef]
- Melin, A.; Tornberg, Å.B.; Skouby, S.; Møller, S.S.; Faber, J.; Sundgot-Borgen, J.; Sjödin, A. Low-energy density and high fiber intake are dietary concerns in female endurance athletes. Scand. J. Med. Sci. Sports 2016, 26, 1060–1071. [Google Scholar] [CrossRef]
- Gordon, C.M.; Ackerman, K.E.; Berga, S.L.; Kaplan, J.R.; Mastorakos, G.; Misra, M.; Murad, M.H.; Santoro, N.F.; Warren, M.P. Functional hypothalamic amenorrhea: An endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 2017, 102, 1413–1439. [Google Scholar] [CrossRef]
- Enns, D.L.; Tiidus, P.M. The influence of estrogen on skeletal muscle: Sex matters. Sports Med. 2010, 40, 41–58. [Google Scholar] [CrossRef]
- Elliott-Sale, K.J.; Tenforde, A.S.; Parziale, A.L.; Holtzman, B.; Ackerman, K.E. Endocrine effects of relative energy deficiency in sport. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Tornberg, Å.B.; Melin, A.; Koivula, F.M.; Johansson, A.; Skouby, S.; Faber, J.; Sjödin, A. Reduced neuromuscular performance in amenorrheic elite endurance athletes. Med. Sci. Sports Exerc. 2017, 49, 2478–2485. [Google Scholar] [CrossRef] [PubMed]
- Vanheest, J.L.; Rodgers, C.D.; Mahoney, C.E.; De Souza, M.J. Ovarian suppression impairs sport performance in junior elite female swimmers. Med. Sci. Sports Exerc. 2014, 46, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Harber, V.J.; Petersen, S.R.; Chilibeck, P.D. Thyroid hormone concentrations and muscle metabolism in amenorrheic and eumenorrheic athletes. Can. J. Appl. Physiol. 1998, 23, 293–306. [Google Scholar] [CrossRef]
- Zinn, C.; Schofield, G.; Wall, C. Development of a psychometrically valid and reliable sports nutrition knowledge questionnaire. J. Sci. Med. Sport 2005, 8, 346–351. [Google Scholar] [CrossRef]
| Year | Author | Sex | Sample Size | Athletes | Mean Age (y) | Mean ± SD EA | Subjects with | Comments | |
|---|---|---|---|---|---|---|---|---|---|
| (kcal/kg FFM/Day)* | Low EA* (%) | ||||||||
| Observational Studies | |||||||||
| 2019 | Civil et al. [14] | F | 20 | Ballet dancers | 18 | N/A | 22 | 44% had EA 30–45 kcal/kg FFM/day 22% had EA <30 kcal/kg FFM/day. 40% MD and 65% at risk of LEA. No associations between MD and EA (p = 0.17) or LEAF-Q score and EA (p = 0.11). | |
| 2019 | McCormack et al. [25] | M/F | 107 27 33 23 24 | M F M Ctrl F Ctrl | Cross-country skiers and Ctrl group | 20 | M:36 ± 16 F:37 ± 21 M Ctrl: 42 ± 15 F Ctrl: 40 ± 21 | N/A | F athletes whole-body BMD higher vs. F Ctrl. Higher dietary restraint score in athletes vs. Ctrl. Higher eating concern score in M athletes vs. M Ctrl. Higher shape concern score in F athletes vs. M athletes. |
| 2019 | Zabriskie et | F | 20 | NCAA Division II | 20 | Off season I: 30 ± 11 | N/A | Post hoc comparisons showed that ‘Pre- | |
| al. [19] | lacrosse athletes | Off season II: 26 ± 11 | season’ trended toward a lower EA than in | ||||||
| Pre-season: 23 ± 9 | ‘off season I’ (p = 0.058) and ‘in season II’ | ||||||||
| In season I: 29 ± 10 | (p = 0.057). | ||||||||
| In season II: 29 ± 9 | |||||||||
| 2018 | Braun et al. | F | 56 | Elite soccer players | 15 | N/A | 53 | Caloric deficit, low carbohydrate and fluid | |
| [21] | intakes were observed. | ||||||||
| 2018 | Cherian et | M/F | 40 | Junior national- | 12 | N/A | M: 24 | 4 of 5 M and 7 of 11 F with low EA were | |
| al. [20] | 21 | M | level soccer players | F: 58 | <16 years of age. | ||||
| 19 | F | ||||||||
| 2018 | Costa et al. | F | 21 | Collegiate | 20 | 26 ± 13 to 30 ± 13 | N/A | Estimated EA was associated with | |
| [16] | synchronized | measured RMR. No association between | |||||||
| swimmers | EA and RMR ratio independent of the | ||||||||
| prediction equation used** | |||||||||
| 2018 | Heikura et | M/F | 48 | Elite distance | M: 27 | M:36 ± 6 | N/A | No associations between EA and the | |
| al. [23] | 21 | M | athletes | F: 26 | F:33 ± 7 | magnitude of relative change in serum Hb mass. | |||
| 27 | F | ||||||||
| 2018 | Heikura et | M/F | 59 | Elite distance | M: 27 | N/A | M: 25 | Lower oestradiol, total testosterone, T3 and | |
| al. [24] | 24 | M | athletes | F: 26 | F: 31 | BMD in MD (37%) and low testosterone | |||
| 35 | F | (40%) athletes. | |||||||
| Bone injuries: ∼4.5 times more prevalent | |||||||||
| in MD and low testosterone athletes. | |||||||||
| 2018 | Silva et al. [13] | M/F | 82 21 M | 61 F | Children and adolesc. acrobatic gymnasts | M/F children: 11 M/F adolesc.: 16 | CM:54 ± 9 CF:46 ± 9 AM:45 ± 15 AF:33 ± 9 | N/A | Lower EA in M and F athletes vs. M and F Ctrl. Most participants did not eat or drink during or immediately after training. |
| 2018 | Zanders et | F | 13 | Collegiate | 20 | 0 | N/A | EA did not change across the season. | |
| al. [17] | basketball players | ||||||||
| 2017 | Brown et al. | F | 25 | Pre-professional | 21 | 7-day EA: 26 ± 13 | N/A | ||
| [15] | contemporary | Week EA: 24 ± 10 | |||||||
| dancers | Weekend EA: 36 ± 21 | ||||||||
| 2017 | Ong et al. | F | 9 | Dragon boat | 23 | 23.7 ± 13 | N/A | Eight of 9 subjects had EA < 45 kcal/kg | |
| [18] | athletes | FFM/day, with 6 < 30 kcal/kg FFM/day. | |||||||
| 2017 | Silva et al. | M | 151 | Rink-hockey*** | Children: 10 | Children: 48 ± 89 | N/A | Lower EI and higher EEE in athletes vs. | |
| [22] | 38 | Children | players and Ctrl | Adolesc.: 14 | Adolesc.: 50 ± 11 | Ctrl; resulting in some cases of LEA in | |||
| 34 | Adolesc. | group | Children Ctrl: 54 ± 9 | Athletes. | |||||
| 43 | Children Ctrl | Adolesc. Ctrl: 55 ± 18 | |||||||
| 36 | Adolesc. Ctrl | ||||||||
| Year | Author | Participants | Energy | Exercise | DE | Reproductive | BMD | Body | Biochemical | Other Parameters |
|---|---|---|---|---|---|---|---|---|---|---|
| (n) | Intake | Energy | Health | Composition | Parameters | Assessed | ||||
| Expenditure | Assessed | |||||||||
| Methods Used | ||||||||||
| Cross-Sectional and Longitudinal Studies | ||||||||||
| 2019 | Civil et al. | 20 ballet | Prospective | Accelometer | TFE-Q | Menstrual | DXA | DXA | Vitamin D | Healthier dance |
| [14] | dancers | weighed | history | practice national | ||||||
| dietary | questionnaire | survey | ||||||||
| record | and LEAF-Q | |||||||||
| 2019 | McCormack | 107 | FFQ | Activity log | EDE-Q | N/A | DXA | DXA | N/A | N/A |
| et al. [25] | 27 M runners | |||||||||
| 33 F runners | ||||||||||
| 23 M controls | ||||||||||
| 24 F controls | ||||||||||
| 2018 | Black et al. | 38 | Prospective | Activity log | N/A | Menstrual | N/A | Bio- | Serum | N/A |
| [33] | recreational | weighed | function | impedance | cholesterols, | |||||
| athletes | dietary | questions in | cortisol, | |||||||
| record | the LEAF-Q | progesterone | ||||||||
| and T3. | ||||||||||
| Salivary | ||||||||||
| testosterone | ||||||||||
| 2018 | Braun et al. | 56 F soccer | Prospective | Activity log | N/A | N/A | N/A | Bio- | Serum iron and | N/A |
| [21] | players | weighed | impedance | ferritin | ||||||
| dietary | ||||||||||
| record | ||||||||||
| 2018 | Cherian et al. [20] | 40 soccer players 21 M 19 F | Prospectiveweighed dietary record | HR monitors | N/A | N/A | N/A | 4-site skinfold measurements | N/A | N/A |
| 2018 | Costa et al. [16] | 21 F collegiate synchronized swimmers | Prospective dietary record | Activity log | N/A | N/A | DXA | 4- and 7-site | N/A | RMR using indirect |
| skinfold | calorimetry | |||||||||
| measurements | ||||||||||
| and DXA | ||||||||||
| 2019 | Zabriskie et | 20 NCAA | My Fitness | Accelometer | N/A | N/A | DXA | DXA | N/A | RMR using indirect |
| al. [19] | division II | Pal | calorimetry. | |||||||
| lacrosse | Application | Questionnaire to | ||||||||
| athletes | assess perceived | |||||||||
| rest, soreness and | ||||||||||
| training satisfaction | ||||||||||
| 2018 | Heikura et | 48 elite | Prospective | Activity log | N/A | N/A | N/A | DXA | Serum iron, | Total HB mass |
| al. [23] | distance | dietary | ferritin, | |||||||
| athletes | record | testosterone and | ||||||||
| 21 M | oestradiol | |||||||||
| 27 F | ||||||||||
| 2018 | Heikura et | 59 elite | Prospective | Activity log | N/A | Metabolic and | DXA | DXA | Oestradiol, | Informal |
| al. [24] | distance | dietary | reproductive | ferritin, IGF-1, | questionnaire of | |||||
| athletes | record | blood | testosterone and | injury and illness | ||||||
| 24 M | hormone | T3 | history | |||||||
| 35 F | concentrations | |||||||||
| and LEAF-Q | ||||||||||
| 2018 | Silva et al. | 82 children | Prospective | Activity log | N/A | Menstrual | N/A | 3-site | N/A | Sleep duration |
| [13] | and adolesc. | dietary | history | skinfold | ||||||
| acrobatic | record | questionnaire | measurements | |||||||
| gymnasts | ||||||||||
| 21 M | ||||||||||
| 61 F | ||||||||||
| 2018 | Zanders et | 13 F | Prospective | HR monitor | N/A | N/A | DXA | DXA | N/A | RMR using the |
| al. [17] | collegiate | dietary | and | Schofield equation, | ||||||
| basketball | record | accelometer | sleep and recovery | |||||||
| players | questionnaires | |||||||||
| 2017 | Brown et al. | 25 F Pre- | Prospective | Accelometer | TFE-Q | Menstrual | N/A | 7-site | N/A | Healthier dance |
| [15] | professional | weighed | history | skinfold | practice national | |||||
| contemporary | dietary | questionnaire | measurements | survey | ||||||
| dancers | record and | |||||||||
| 24-h | ||||||||||
| recall | ||||||||||
| 2017 | Ong et al. | 9 F Dragon | Prospective | Accelometer | N/A | N/A | N/A | Bio- | N/A | N/A |
| [18] | boat athletes | dietary | impedance | |||||||
| record | ||||||||||
| 2017 | Silva et al. [22] | 72 children and adolesc. M rink- hockey players and 79 M ctrl | Prospective dietary record | Activity log | N/A | N/A | N/A | 2-site skinfold measurements | N/A | N/A |
| Year | Author | Sex | Sample Size | Athletes | Mean | % at Risk of Low | % Reporting Health | % Reporting | Comments |
|---|---|---|---|---|---|---|---|---|---|
| age | EA/Triad/RED- | Outcomes of RED- | Performance | ||||||
| (y) | S a | S/Triad | Outcomes of RED-S | ||||||
| 2019 | Brook et | M/F | 260 | Elite para | 32 | N/A | Prior ED: 3.1 | N/A | Most athletes (95 M, 65 F) were |
| al. [34] | 150 M | athletes | Elevated EDE-Q scores: | attempting to change body | |||||
| 110 F | 32.4 | composition/weight to improve | |||||||
| MD: 44 | performance. Athletes with BSI, | ||||||||
| BSI: 9.2 | 54.5% had low BMD. <10% reported | ||||||||
| awareness of the Triad/RED-S | |||||||||
| 2019 | Condo et al. [8] | F | 30 | Australian rules | 24 | 30 | N/A | N/A | No differences in carbohydrate, protein, fat and energy intakes between those at risk and not at risk of LEA |
| football players | |||||||||
| 1000 | |||||||||
| 2019 | Holtzman et al. [35] | F | Adolesc/youngadult athletes | 19 | Triad risk: 54.7% moderate, 7.9% high; RED-S risk: 63.2% moderate, 33.0% high b | N/A | N/A | The tools agreed on risk for 55.5% of athletes. Agreement ↑ to 64.3% when only athletes with BMD measurements were considered. | |
| 2019 | Nose- Ogura et al. [36] | F | 390 | Adolesc/youngadult athletes | 21 | 14 c | MD: 39 Low BMD: 22.7 BSI (last 3 months): 9.2 | N/A | Higher BSI risk due to the Triad inteenage athletes vs. athletes in their 20s. |
| 2018 | Ackerman et al. [5] | F | 1000 | Adolesc/young adult athletes | 20 | 47.3 d | MD: 47.9 Impaired bone health: 26.9 Abnormal endocrine function: 3.4 Abnormal metabolic health: 4.4 Impaired haematologicalhealth: 32.1 Impaired growth and development: 14.16 Impaired psychological health: 50.1 Increased cardiovascular risk: 9.5 Impaired GI function: 55.8 Impaired immunological health: 37.5 | ↓endurance performance: 31.3 ↑injury risk: 38.5 ↓training response: 23.7 Impaired judgement: 8.5 ↓coordination: 20.5 ↓concentration: 14.2 Irritability: 30.7 Depression: 20.7 | Increased risk of MD, poor bonehealth, metabolic, haematological and cardiovascular impairment, GI dysfunction, psychological disorders (depression), reduced training response, judgement, coordination, concentration and endurance performance in those at risk vs. not at risk of low EA |
| 2018 | Black et | F | 38 | Recreational | 23 | 63.2 | TC > 5.0 mmol/L: 21 | N/A | Lower EA, ↓ T3, low energy and |
| al. [33] | LDL > 3.0 mmol/L: 25 | calcium intake in those at risk of low | |||||||
| EA | |||||||||
| 2018 | Keay et | M | 50 | Road cyclists | 36 | 28% e | Lower lumbar spine | N/A | Lack of load-bearing sport associated |
| al. [9] | BMD: 44 | with low BMD in cyclists with low | |||||||
| EA. The 10 with low EA had lower | |||||||||
| testosterone levels than those | |||||||||
| maintain adequate EA. Low EA | |||||||||
| associated with reduced body fat percentage. | |||||||||
| 2018 | Logue et | F | 833 | Elite, sub-elite | N/A | 40 | ≥22 days absence from | N/A | 1.7- and 1.8-times increased risk in |
| al. [37] | and recreational | training due to illness: | international and provincial/inter- | ||||||
| 24.2 | county athletes compared to | ||||||||
| recreationally active individuals | |||||||||
| 2018 | Staal et | M/F | 40 | Elite ballet | 25 | F: 40 | Low C-RMR: 100 F, 80 | N/A | Large variability in suppressed RMR |
| al. [38] | 20 M | dancers | M | using predictive RMR equations (M: | |||||
| 20 F | Low HB-RMR ratio: 45 | 25–80%; F: 35–100%). Cunningham | |||||||
| F,25M | equation showed highest sensitivity | ||||||||
| Low DXA-RMR ratio: 35 | for detecting both genders at risk for | ||||||||
| F,55M | energy deficiency. | ||||||||
| 2018 | Wilson et | M | 21 | Flat jockeys | A: 19 | N/A | N/A | N/A | No difference in RMR or hip and |
| al. [39] | 17 A | S: 32 | lumber spine BMD between groups. | ||||||
| 14 S | Measured RMR did not differ from | ||||||||
| predicted RMR in either group. | |||||||||
| 2017 | Drew et | M/F | 132 | Elite Olympic | M: 26 | 40 | N/A | N/A | Higher odds of reporting URTI |
| al. [40] | 47 M | athletes | F: 24 | (OR = 3.8), bodily aches (OR = 5.8), GI | |||||
| 85 F | disturbances (OR = 3.8) and head | ||||||||
| symptoms (OR = 4.4) in those at risk | |||||||||
| of low EA. | |||||||||
| 2017 | Sygo et | F | 13 | Elite sprinters | 21 | Pre-training | Pre-training season: | N/A | Primary low EA indicators: a LEAF- |
| al. [41] | season: 23 Post- | BMD: 8 RMR: 15 FSH: | Q score >8; RMR < 29 kcal/kg FFM, | ||||||
| training season: | 15 | low oestradiol, FSH or LH or a BMD | |||||||
| 39 | Post-training season: | of <1.09 g/cm2 | |||||||
| BMD: 15 RMR: 8 | |||||||||
| oestradiol: 31 LH: 23 | |||||||||
| FSH: 15 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Logue, D.M.; Madigan, S.M.; Melin, A.; Delahunt, E.; Heinen, M.; Donnell, S.-J.M.; Corish, C.A. Low Energy Availability in Athletes 2020: An Updated Narrative Review of Prevalence, Risk, Within-Day Energy Balance, Knowledge, and Impact on Sports Performance. Nutrients 2020, 12, 835. https://doi.org/10.3390/nu12030835
Logue DM, Madigan SM, Melin A, Delahunt E, Heinen M, Donnell S-JM, Corish CA. Low Energy Availability in Athletes 2020: An Updated Narrative Review of Prevalence, Risk, Within-Day Energy Balance, Knowledge, and Impact on Sports Performance. Nutrients. 2020; 12(3):835. https://doi.org/10.3390/nu12030835
Chicago/Turabian StyleLogue, Danielle M., Sharon M. Madigan, Anna Melin, Eamonn Delahunt, Mirjam Heinen, Sarah-Jane Mc Donnell, and Clare A. Corish. 2020. "Low Energy Availability in Athletes 2020: An Updated Narrative Review of Prevalence, Risk, Within-Day Energy Balance, Knowledge, and Impact on Sports Performance" Nutrients 12, no. 3: 835. https://doi.org/10.3390/nu12030835
APA StyleLogue, D. M., Madigan, S. M., Melin, A., Delahunt, E., Heinen, M., Donnell, S.-J. M., & Corish, C. A. (2020). Low Energy Availability in Athletes 2020: An Updated Narrative Review of Prevalence, Risk, Within-Day Energy Balance, Knowledge, and Impact on Sports Performance. Nutrients, 12(3), 835. https://doi.org/10.3390/nu12030835






