The Neuropathology of Gluten-Related Neurological Disorders: A Systematic Review
Abstract
1. Introduction
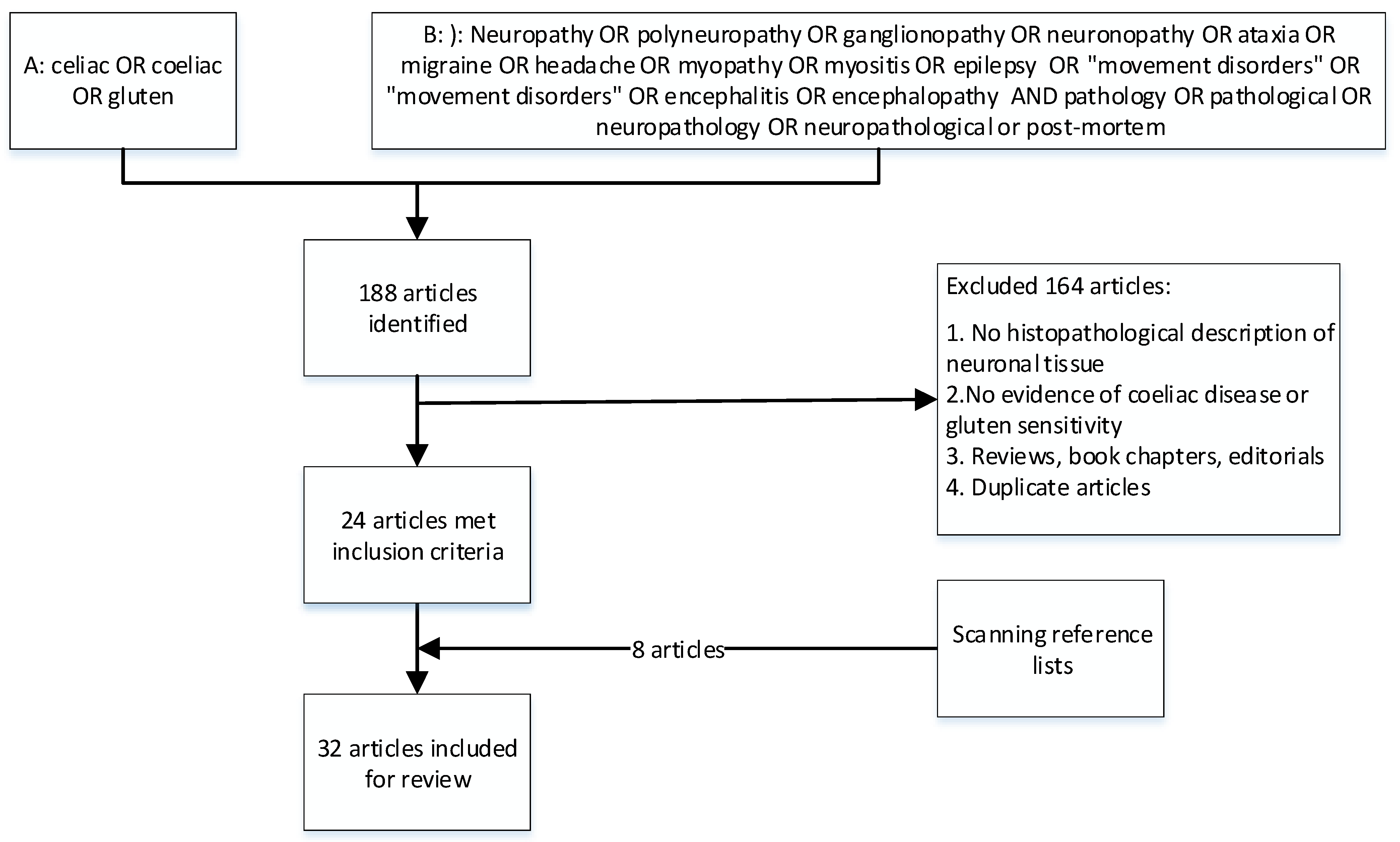
2. Materials and Methods
2.1. Literature and Search Strategy
- Original clinical studies
- Human subject study
- Pathological studies in patients with coeliac disease or gluten sensitivity, suffering from neurological illness
- If there was no histopathological description of neuronal/brain tissue or muscle
- No evidence of coeliac disease or gluten sensitivity
- Reviews, book chapters, editorials
- Duplicate articles
2.2. Ethical Considerations
3. Results
3.1. Study Characteristics
3.2. Ataxia and Gluten
3.3. Large Fibre Neuropathy and Gluten Sensitivity
3.4. Small-Fibre Neuropathy and Gluten Sensitivity
3.5. Myopathy and Gluten Sensitivity
3.6. Encephalopathy and Gluten Sensitivity
3.7. Epilepsy and Gluten Sensitivity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kelly, C.P.; Bai, J.C.; Liu, E.; Leffler, D.A. Advances in diagnosis and management of celiac disease. Gastroenterology 2015, 148, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Reilly, N.R.; Green, P.H. Epidemiology and clinical presentations of celiac disease. Semin. Immunopathol. 2012, 34, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Van Kalleveen, M.W.; de Meij, T.; Plotz, F.B. Clinical spectrum of paediatric coeliac disease: A 10-year single-centre experience. Eur. J. Pediatr. 2018, 177, 593–602. [Google Scholar] [CrossRef] [PubMed]
- Lindfors, K.; Ciacci, C.; Kurppa, K.; Lundin, K.E.; Makharia, G.K.; Mearin, M.L.; Murray, J.A.; Verdu, E.F.; Kaukinen, K. Coeliac disease. Nat. Rev. Dis. Primers 2019, 5, 3. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A.; Murray, J.A. Classification and management of refractory coeliac disease. Gut 2010, 59, 547–557. [Google Scholar] [CrossRef]
- Cicarelli, G.; Della Rocca, G.; Amboni, M.; Ciacci, C.; Mazzacca, G.; Filla, A.; Barone, P. Clinical and neurological abnormalities in adult celiac disease. Neurol. Sci. 2003, 24, 311–317. [Google Scholar] [CrossRef]
- Nikpour, S. Neurological manifestations, diagnosis, and treatment of celiac disease: A comprehensive review. Iran. J. Neurol. 2012, 11, 59–64. [Google Scholar]
- Hadjivassiliou, M.; Rao, D.G.; Grìnewald, R.A.; Aeschlimann, D.P.; Sarrigiannis, P.G.; Hoggard, N.; Aeschlimann, P.; Mooney, P.D.; Sanders, D.S. Neurological Dysfunction in Coeliac Disease and Non-Coeliac Gluten Sensitivity. Am. J. Gastroenterol. 2016, 111, 561–567. [Google Scholar] [CrossRef]
- Cooke, W.T.; Smith, W.T. Neurological disorders associated with adult coeliac disease. Brain 1966, 89, 683–722. [Google Scholar] [CrossRef]
- Tuzun, E.; Gurses, C.; Baykan, B.; Buyukbabani, N.; Ozturk, A.S.; Gokyigit, A. Lafora body-like inclusions in a case of progressive myoclonic ataxia associated with coeliac disease. Eur. Neurol. 2001, 46, 157–158. [Google Scholar] [CrossRef]
- Bhatia, K.P.; Brown, P.; Gregory, R.; Lennox, G.G.; Manji, H.; Thompson, P.D.; Ellison, D.W.; Marsden, C.D. Progressive myoclonic ataxia associated with coeliac disease. The myoclonus is of cortical origin, but the pathology is in the cerebellum. Brain 1995, 118 (Pt 5), 1087–1093. [Google Scholar] [CrossRef] [PubMed]
- Finelli, P.F.; McEntee, W.J.; Ambler, M.; Kestenbaum, D. Adult celiac disease presenting as cerebellar syndrome. Neurology 1980, 30, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Kinney, H.C.; Burger, P.C.; Hurwitz, B.J.; Hijmans, J.C.; Grant, J.P. Degeneration of the central nervous system associated with celiac disease. J. Neurol. Sci. 1982, 53, 9–22. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Grünewald, R.A.; Chattopadhyay, A.K.; Davies-Jones, G.A.; Gibson, A.; Jarratt, J.A.; Kandler, R.H.; Lobo, A.; Powell, T.; Smith, C.M. Clinical, radiological, neurophysiological, and neuropathological characteristics of gluten ataxia. Lancet 1998, 352, 1582–1585. [Google Scholar] [CrossRef]
- Mittelbronn, M.; Schittenhelm, J.; Bakos, G.; De Vos, R.A.; Wehrmann, M.; Meyermann, R.; Bürk, K. CD8(+)/perforin/granzyme B(+) effector cells infiltrating cerebellum and inferior olives in gluten ataxia. Neuropathology 2010, 30, 92–96. [Google Scholar] [CrossRef]
- Nanri, K.; Shibuya, M.; Taguchi, T.; Hasegawa, A.; Tanaka, N. Selective loss of Purkinje cells in a patient with anti-gliadin-antibody-positive autoimmune cerebellar ataxia. Diagn. Pathol. 2011, 6, 14. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Grünewald, R.A.; Kandler, R.H.; Chattopadhyay, A.K.; Jarratt, J.A.; Sanders, D.S.; Sharrack, B.; Wharton, S.B.; Davies-Jones, G.A. Neuropathy associated with gluten sensitivity. J. Neurol. Neurosurg. Psychiatry 2006, 77, 1262–1266. [Google Scholar] [CrossRef]
- Cooke, W.T.; Johnson, A.G.; Woolf, A.L. Vital staining and electron microscopy of the intramuscular nerve endings in the neuropathy of adult coeliac disease. Brain 1966, 89, 663–682. [Google Scholar] [CrossRef]
- Chin, R.L.; Sander, H.W.; Brannagan, T.H.; Green, P.H.; Hays, A.P.; Alaedini, A.; Latov, N. Celiac neuropathy. Neurology 2003, 60, 1581–1585. [Google Scholar] [CrossRef]
- Squintani, G.; Ferrari, S.; Caramaschi, P.; Cavallaro, T.; Refatti, N.; Rizzuto, N.; Tonin, P. Multineuropathy in a patient with HBV infection, polyarteritis nodosa and celiac disease. Rheumatol. Int. 2009, 29, 579–581. [Google Scholar] [CrossRef]
- Simonati, A.; Battistella, P.A.; Guariso, G.; Clementi, M.; Rizzuto, N. Coeliac disease associated with peripheral neuropathy in a child: A case report. Neuropediatrics 1998, 29, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Souayah, N.; Chin, R.L.; Brannagan, T.H.; Latov, N.; Green, P.H.; Kokoszka, A.; Sander, H.W. Effect of intravenous immunoglobulin on cerebellar ataxia and neuropathic pain associated with celiac disease. Eur. J. Neurol. 2008, 15, 1300–1303. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, E.A.; Hays, A.P.; Chin, R.L.; Sander, H.W.; Brannagan, T.H. Characteristics of patients with sensory neuropathy diagnosed with abnormal small nerve fibres on skin biopsy. J. Neurol. Neurosurg. Psychiatry 2006, 77, 983–985. [Google Scholar] [CrossRef] [PubMed]
- Brannagan, T.H.; Hays, A.P.; Chin, S.S.; Sander, H.W.; Chin, R.L.; Magda, P.; Green, P.H.; Latov, N. Small-fiber neuropathy/neuronopathy associated with celiac disease: Skin biopsy findings. Arch. Neurol. 2005, 62, 1574–1578. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Rao, D.G.; Wharton, S.B.; Sanders, D.S.; Grunewald, R.A.; Davies-Jones, A.G. Sensory ganglionopathy due to gluten sensitivity. Neurology 2010, 75, 1003–1008. [Google Scholar] [CrossRef]
- Danielsson, O.; Lindvall, B.; Hallert, C.; Vrethem, M.; Dahle, C. Increased prevalence of celiac disease in idiopathic inflammatory myopathies. Brain Behav. 2017, 7, e00803. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Chattopadhyay, A.K.; Davies-Jones, G.A.; Gibson, A.; Grunewald, R.A.; Lobo, A.J. Neuromuscular disorder as a presenting feature of coeliac disease. J. Neurol. Neurosurg. Psychiatry 1997, 63, 770–775. [Google Scholar] [CrossRef]
- Williams, S.F.; Mincey, B.A.; Calamia, K.T. Inclusion body myositis associated with celiac sprue and idiopathic thrombocytopenic purpura. South Med. J. 2003, 96, 721–723. [Google Scholar] [CrossRef]
- Henriksson, K.G.; Hallert, C.; Norrby, K.; Walan, A. Polymyositis and adult coeliac disease. Acta Neurol. Scand. 1982, 65, 301–319. [Google Scholar] [CrossRef]
- Kleopa, K.A.; Kyriacou, K.; Zamba-Papanicolaou, E.; Kyriakides, T. Reversible inflammatory and vacuolar myopathy with vitamin E deficiency in celiac disease. Muscle Nerve 2005, 31, 260–265. [Google Scholar] [CrossRef]
- Alawneh, K.; Ashley, C.; Carlson, J.A. Neutrophilic myositis as a manifestation of celiac disease: A case report. Clin. Rheumatol. 2008, 27, 11–13. [Google Scholar] [CrossRef] [PubMed]
- Hadjivassiliou, M.; Chattopadhyay, A.K.; Grünewald, R.A.; Jarratt, J.A.; Kandler, R.H.; Rao, D.G.; Sanders, D.S.; Wharton, S.B.; Davies-Jones, G.A. Myopathy associated with gluten sensitivity. Muscle Nerve 2007, 35, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Brücke, T.; Kollegger, H.; Schmidbauer, M.; Müller, C.; Podreka, I.; Deecke, L. Adult coeliac disease and brainstem encephalitis. J. Neurol. Neurosurg. Psychiatry 1988, 51, 456–457. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Keller, C.E.; Gamboa, E.T.; Hays, A.P.; Karlitz, J.; Lowe, G.; Green, P.H.; Bhagat, G. Fatal CNS vasculopathy in a patient with refractory celiac disease and lymph node cavitation. Virchows Arch. 2006, 448, 209–213. [Google Scholar] [CrossRef]
- Dimberg, E.L.; Crowe, S.E.; Trugman, J.M.; Swerdlow, R.H.; Lopes, M.B.; Bourne, T.D.; Burns, T.M. Fatal encephalitis in a patient with refractory celiac disease presenting with myorhythmia and carpal spasm. Mov. Disord. 2007, 22, 407–411. [Google Scholar] [CrossRef]
- Malamut, G.; Cellier, C. Refractory Celiac Disease. Gastroenterol. Clin. N. Am. 2019, 48, 137–144. [Google Scholar] [CrossRef]
- Hu, W.T.; Murray, J.A.; Greenaway, M.C.; Parisi, J.E.; Josephs, K.A. Cognitive impairment and celiac disease. Arch. Neurol. 2006, 63, 1440–1446. [Google Scholar] [CrossRef]
- La Mantia, L.; Pollo, B.; Savoiardo, M.; Costa, A.; Eoli, M.; Allegranza, A.; Boiardi, A.; Cestari, C. Meningo-cortical calcifying angiomatosis and celiac disease. Clin. Neurol. Neurosurg. 1998, 100, 209–215. [Google Scholar] [CrossRef]
- Shneker, B.F.; Fountain, N.B. Epilepsy. Dis. Mon. 2003, 49, 426–478. [Google Scholar] [CrossRef]
- Julian, T.; Hadjivassiliou, M.; Zis, P. Gluten sensitivity and epilepsy: A systematic review. J. Neurol. 2019, 266, 1557–1565. [Google Scholar] [CrossRef]
- Bouquet, F.; Ventura, A.; Gobbi, G.; Greco, L.; Lambertini, A.; Zaniboni, M.G.; Tassinari, C.A. Coeliac disease, epilepsy, and cerebral calcifications. The Italian Working Group on Coeliac Disease and Epilepsy. Lancet 1992, 340, 439–443. [Google Scholar] [CrossRef]
- Bye, A.M.; Andermann, F.; Robitaille, Y.; Oliver, M.; Bohane, T.; Andermann, E. Cortical vascular abnormalities in the syndrome of celiac disease, epilepsy, bilateral occipital calcifications, and folate deficiency. Ann. Neurol. 1993, 34, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Orstavik, K.H.; Stromme, P.; Ek, J.; Torvik, A.; Skjeldal, O.H. Macrocephaly, epilepsy, autism, dysmorphic features, and mental retardation in two sisters: A new autosomal recessive syndrome? J. Med. Genet. 1997, 34, 849–851. [Google Scholar] [CrossRef] [PubMed]
- Van Gils, T.; Rootsaert, B.; Bouma, G.; Mulder, C.J. Celiac Disease in The Netherlands: Demographic Data of Members of the Dutch Celiac Society. J. Gastrointest. Liver Dis. 2016, 25, 441–445. [Google Scholar]
- Rashtak, S.; Murray, J.A. Celiac disease in the elderly. Gastroenterol. Clin. N. Am. 2009, 38, 433–446. [Google Scholar] [CrossRef] [PubMed]
- Ventura, A.; Magazzu, G.; Greco, L. Duration of exposure to gluten and risk for autoimmune disorders in patients with celiac disease. SIGEP Study Group for Autoimmune Disorders in Celiac Disease. Gastroenterology 1999, 117, 297–303. [Google Scholar] [CrossRef]
- Sategna Guidetti, C.; Solerio, E.; Scaglione, N.; Aimo, G.; Mengozzi, G. Duration of gluten exposure in adult coeliac disease does not correlate with the risk for autoimmune disorders. Gut 2001, 49, 502–505. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Grünewald, R.A.; Sanders, D.S.; Zis, P.; Croall, I.; Shanmugarajah, P.D.; Sarrigiannis, P.G.; Trott, N.; Wild, G.; Hoggard, N. The Significance of Low Titre Antigliadin Antibodies in the Diagnosis of Gluten Ataxia. Nutrients 2018, 10, 1444. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Aeschlimann, P.; Strigun, A.; Sanders, D.S.; Woodroofe, N.; Aeschlimann, D. Autoantibodies in gluten ataxia recognize a novel neuronal transglutaminase. Ann. Neurol. 2008, 64, 332–343. [Google Scholar] [CrossRef]
- Aziz, I.; Peerally, M.F.; Barnes, J.H.; Kandasamy, V.; Whiteley, J.C.; Partridge, D.; Vergani, P.; Cross, S.S.; Green, P.H.; Sanders, D.S. The clinical and phenotypical assessment of seronegative villous atrophy; a prospective UK centre experience evaluating 200 adult cases over a 15-year period (2000–2015). Gut 2017, 66, 1563–1572. [Google Scholar] [CrossRef]
- Rodrigo, L.; Hernández-Lahoz, C.; Lauret, E.; Rodriguez-Peláez, M.; Soucek, M.; Ciccocioppo, R.; Kruzliak, P. Gluten ataxia is better classified as non-celiac gluten sensitivity than as celiac disease: A comparative clinical study. Immunol. Res. 2016, 64, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Bañares, F.; Carrasco, A.; Martín, A.; Esteve, M. Systematic Review and Meta-Analysis: Accuracy of Both Gamma Delta+ Intraepithelial Lymphocytes and Coeliac Lymphogram Evaluated by Flow Cytometry for Coeliac Disease Diagnosis. Nutrients 2019, 11, 1992. [Google Scholar] [CrossRef]
- Fernández-Bañares, F.; Carrasco, A.; García-Puig, R.; Rosinach, M.; González, C.; Alsina, M.; Loras, C.; Salas, A.; Viver, J.M.; Esteve, M. Intestinal intraepithelial lymphocyte cytometric pattern is more accurate than subepithelial deposits of anti-tissue transglutaminase IgA for the diagnosis of celiac disease in lymphocytic enteritis. PLoS ONE 2014, 9, e101249. [Google Scholar] [CrossRef] [PubMed]
- Herisson, F.; Frodermann, V.; Courties, G.; Rohde, D.; Sun, Y.; Vandoorne, K.; Wojtkiewicz, G.R.; Masson, G.S.; Vinegoni, C.; Kim, J.; et al. Direct vascular channels connect skull bone marrow and the brain surface enabling myeloid cell migration. Nat. Neurosci. 2018, 21, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Hadjivassiliou, M.; Boscolo, S.; Davies–Jones, G.A.; Grünewald, R.A.; Not, T.; Sanders, D.S.; Simpson, J.E.; Tongiorgi, E.; Williamson, C.A. The humoral response in the pathogenesis of gluten ataxia. Neurology 2002, 58, 1221–1226. [Google Scholar] [CrossRef]
- Boscolo, S.; Lorenzon, A.; Sblattero, D.; Florian, F.; Stebel, M.; Marzari, R.; Not, T.; Aeschlimann, D.; Ventura, A.; Hadjivassiliou, M.; et al. Anti transglutaminase antibodies cause ataxia in mice. PLoS ONE 2010, 5, e9698. [Google Scholar] [CrossRef]
- Stamnaes, J.; Dorum, S.; Fleckenstein, B.; Aeschlimann, D.; Sollid, L.M. Gluten T cell epitope targeting by TG3 and TG6; implications for dermatitis herpetiformis and gluten ataxia. Amino Acids 2010, 39, 1183–1191. [Google Scholar] [CrossRef]
- Thomas, H.; Beck, K.; Adamczyk, M.; Aeschlimann, P.; Langley, M.; Oita, R.C.; Thiebach, L.; Hils, M.; Aeschlimann, D. Transglutaminase 6: A protein associated with central nervous system development and motor function. Amino Acids 2013, 44, 161–177. [Google Scholar] [CrossRef]
- Hadjivassiliou, M.; Aeschlimann, P.; Sanders, D.S.; Mäki, M.; Kaukinen, K.; Grünewald, R.A.; Bandmann, O.; Woodroofe, N.; Haddock, G.; Aeschlimann, D.P. Transglutaminase 6 antibodies in the diagnosis of gluten ataxia. Neurology 2013, 80, 1740–1745. [Google Scholar] [CrossRef]
- Zis, P.; Rao, D.G.; Sarrigiannis, P.G.; Aeschlimann, P.; Aeschlimann, D.P.; Sanders, D.; Grünewald, R.A.; Hadjivassiliou, M. Transglutaminase 6 antibodies in gluten neuropathy. Dig. Liver Dis. 2017, 49, 1196–1200. [Google Scholar] [CrossRef]
- Addolorato, G.; Di Giuda, D.; De Rossi, G.; Valenza, V.; Domenicali, M.; Caputo, F.; Gasbarrini, A.; Capristo, E.; Gasbarrini, G. Regional cerebral hypoperfusion in patients with celiac disease. Am. J. Med. 2004, 116, 312–317. [Google Scholar] [CrossRef] [PubMed]
- Usai, P.; Serra, A.; Marini, B.; Mariotti, S.; Satta, L.; Boi, M.F.; Spanu, A.; Loi, G.; Piga, M. Frontal cortical perfusion abnormalities related to gluten intake and associated autoimmune disease in adult coeliac disease: 99mTc-ECD brain SPECT study. Dig. Liver Dis. 2004, 36, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Malamut, G.; Afchain, P.; Verkarre, V.; Lecomte, T.; Amiot, A.; Damotte, D.; Bouhnik, Y.; Colombel, J.F.; Delchier, J.C.; Allez, M.; et al. Presentation and long-term follow-up of refractory celiac disease: Comparison of type I with type II. Gastroenterology 2009, 136, 81–90. [Google Scholar] [CrossRef] [PubMed]

| Neurological Disorder Papers n First Author/Year/Country | Patients n | Tissue | Main Neuropathological Findings |
|---|---|---|---|
| Ataxia 9 | 18 | ||
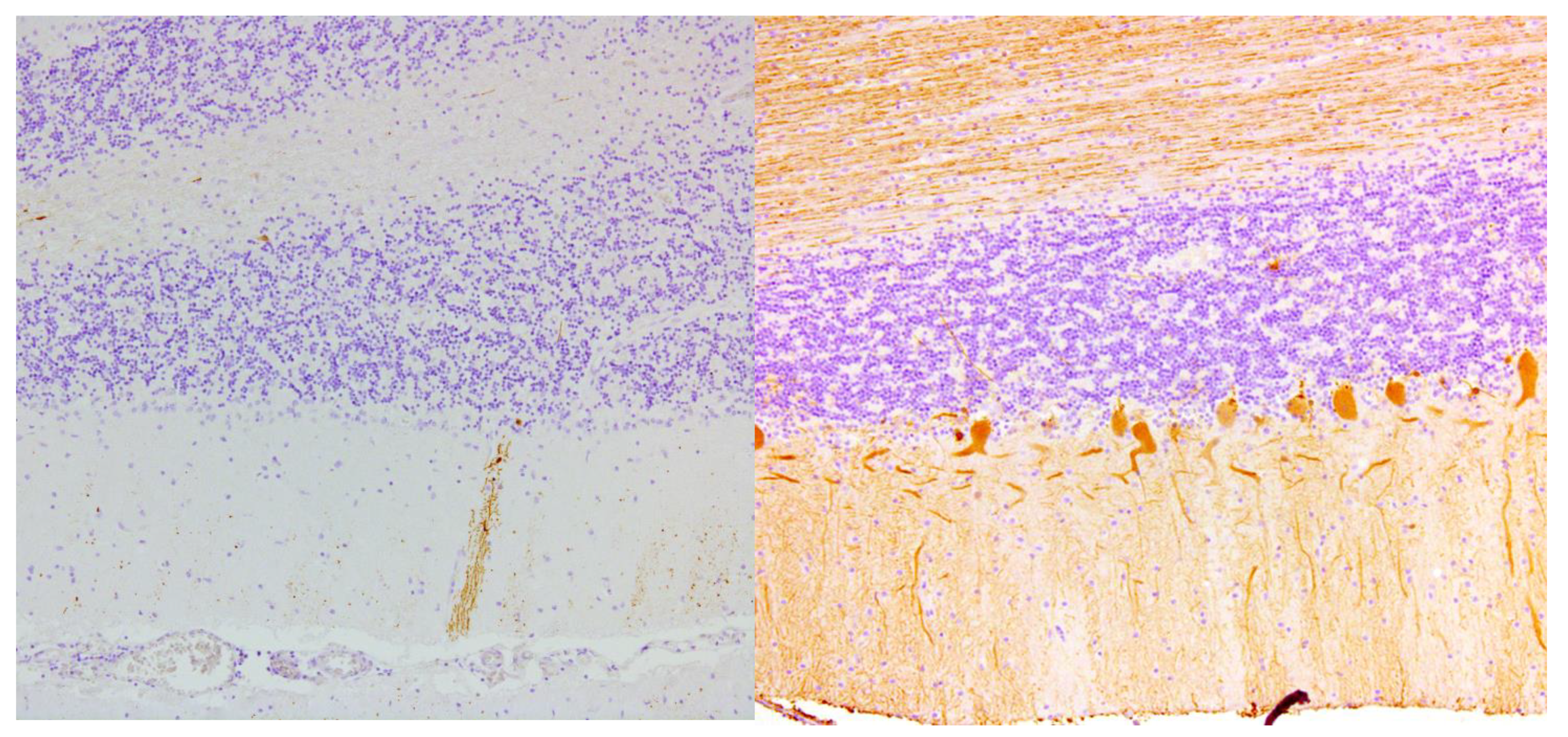
| Bhatia 1995, UK | 1 | Brain Spinal cord | Loss of Purkinje cells and cells in inferior olives; Cerebellar gliosis; Astrocytosis of the granular layer, dentate nucleus & inferior olives |
| Cooke 1966, UK (I) * | 9 | Brain Spinal cord | Loss of Purkinje cells; atrophy and gliosis of the dentate nucleus, cerebrum, inferior olives, thalamus and hypothalamus; demyelination of post. & ant.-lat. columns; focal perivascular lymphocytic cuffing, chromatolysis and sudanophil lipophages throughout the CNS. |
| Finelli 1980, USA | 1 | Brain Spinal cord | Loss of Purkinje and granular layer cells; Neuronal loss & gliosis basal ganglia, inferior olives, substantia nigra; Demyelination ant. & lat. corticospinal tracts |
| Hadjivassiliou 1998, UK | 2 | Brain Spinal cord | Loss of Purkinje cells; Cerebellar atrophy & astrocytic gliosis & vacuolation of neutrophils; Diffuse infiltration of lymphocytes & perivascular cuffing of T-lymphocytes in the cerebellum and the post. columns |
| Hadjivassiliou 2006, UK ** | 1 | Brain | Capillary changes in the white matter, hippocampus and olives marked by vascular and perivascular inflammatory cell infiltrates (CD68+ cells and a smaller CD45Ro+ cell population). Purkinje cell loss and Bergmann gliosis and loss of neurons in the inferior olives |
| Kinney 1982, USA | 1 | Brain | Loss of Purkinje cells; atrophy, gliosis of the dentate nucleus, cerebellar granular layer, thalamus, hypothalamus & periaqueductal grey; Senile plaques in the neocortex & hippocampi; Cerebral gliosis subcortical & white matter |
| Mittelbronn 2010, Germany | 1 | Brain | Loss of Purkinje cells & cerebellar granular layer cells; Cerebellar atrophy and astrocytic gliosis; Severe neuronal loss inferior olives & accumulation of corpora amylacea. Cerebral reactive astrogliosis and microglial activation Inflammation dominated by CD8+/granzyme B+ & CD20-/CD138- diffuse infiltrates & perivascular cuffing in the cerebellum and brainstem |
| Nanri 2011, Japan | 1 | Brain | Loss of Purkinje cells; Minimal cerebellar atrophy; Mild Bergmann gliosis. Empty basket cells, Edematous splitting of Purkinje cell layer, loss of granular cells. No lymphocytic infiltration (CD3-, CD4-, CD8-, CD20-, CD68-, CD79A-) |
| Tuzun 2001, Turkey | 1 | Skin | skin biopsy: Periodic Acid Schiff-positive inclusions, diastase resistant intracellular inclusion bodies in apocrine sweat gland cells |
| Neuropathy 10 | 37 | ||
| Brannagan 2005, USA | 8 | Skin | Reduced epidermal nerve density, distal > proximal |
| Cooke 1966, UK (I) * | 11 | Nerve | Axonal swelling, loss of myelinated fibers, focal proliferation of sarcolemmal nuclei and collateral reinnervation |
| Cooke 1966, UK (II) | |||
| Chin 2003, USA | 3 | Sural nerve | Mild to moderately severe chronic axonopathy with loss of myelinated fibers |
| De Sousa 2006, USA | 6 | Skin | Morphological changes & a reduced epidermal nerve density |
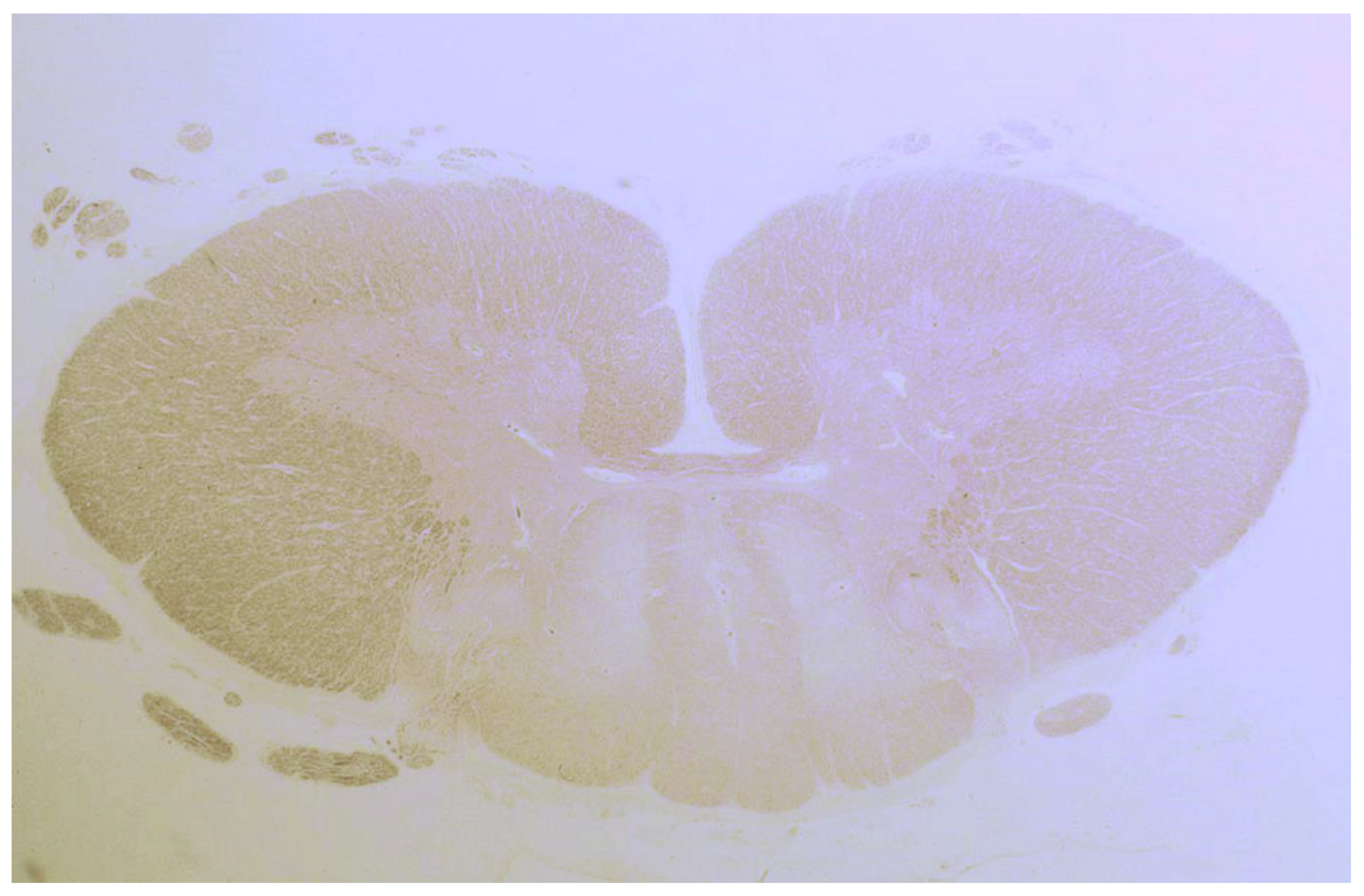
| Hadjivassiliou 2006, UK ** | 3 | Sural nerve | Focal inflammatory cell infiltrate in the epineurium & perivascular cuffing of lymphocytes; Patchy loss of myelinated fibers & occasional degeneration |
| Hadjivassiliou 2010, UK | 2 | Spinal cord | Degeneration of the dorsal columns; Preservation of the ant.-lat. white matter; Subtotal loss of myelin; Axonal loss; Lymphocytic infiltration |
| Simonati 1998, Italy | 1 | Sural nerve | Chronic axonal neuropathy; Significant loss of myelinated fibers & Schwann cell nuclei; Low density of unmyelinated fibers; No inflammatory cells objectified |
| Souayah 2008, USA | 2 | Skin | Low to normal epidermal nerve fiber density; Sparse nerve fibers; axonal swelling; increased branching; uneven distribution of epidermal fibers in calf and thigh both |
| Squintani 2009, Italy | 1 | Sural nerve | Loss of myelinated axons; Axonal degeneration with focal distribution in different fascicles; Mild perivascular mononuclear cell infiltration of epineural blood vessels; Thickened perineurium |
| Myopathy 7 | 36 | ||
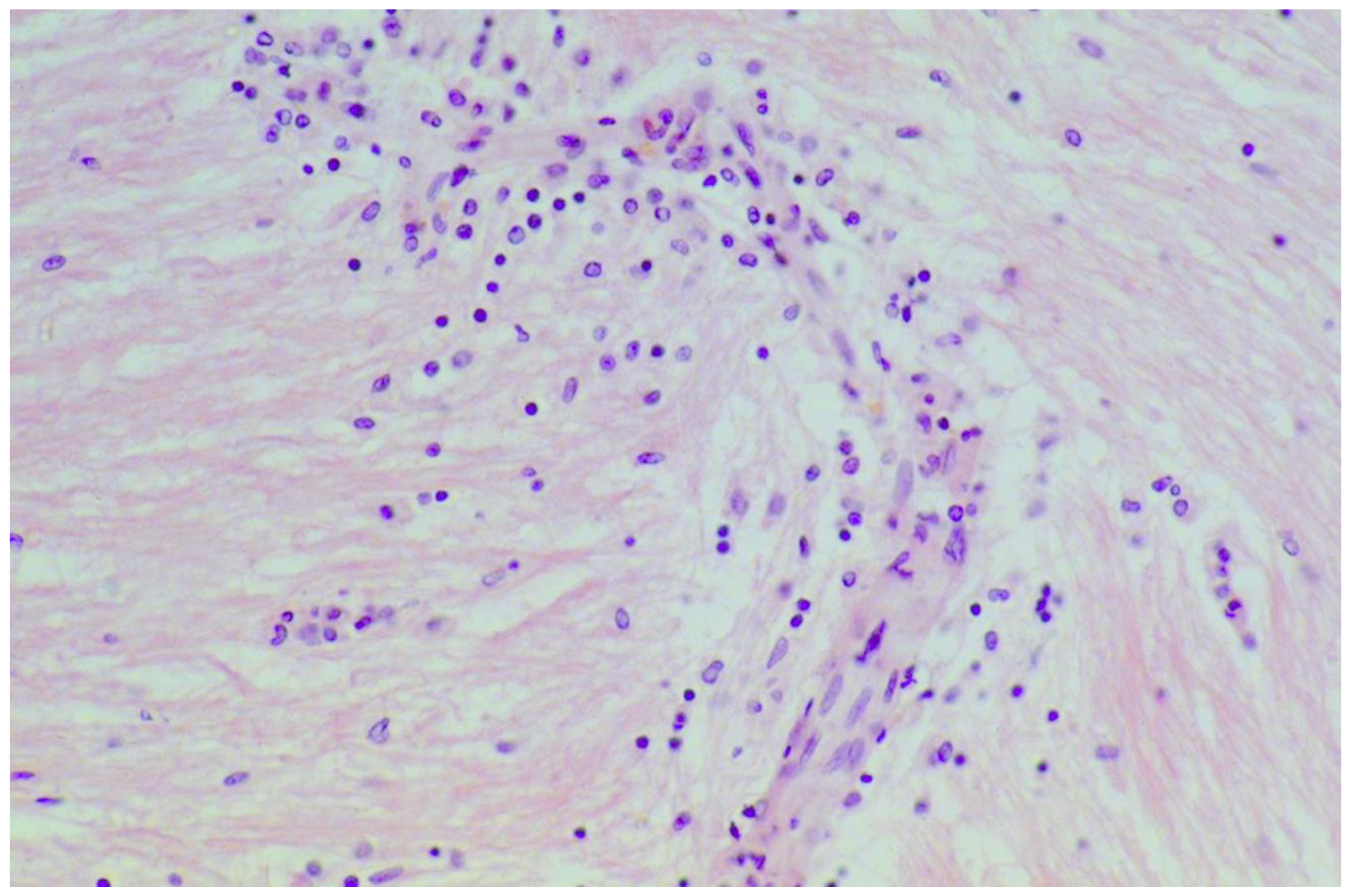
| Alawneh 2008, Jordan | 1 | Muscle | Muscle necrosis; Neutrophilic infiltration; Secondary leukocytoclastic vasculitis |
| Danielsson 2017, Sweden | 13 | Muscle | Inflammatory infiltrates & muscle fiber degeneration |
| Hadjivassiliou 1997, UK | 2 | Muscle | Inflammatory myopathy & Basophilic rimmed vacuoles |
| Hadjivassiliou 2007, UK | 13 | Muscle | Internal nuclei; Basophilic rimmed vacuoles; fiber splitting; Endomysial chronic inflammatory cell infiltrate (CD3+ cells), Fibrosis |
| Hendriksson 1982, Sweden | 5 | Muscle | Basophilic sarcoplasm; Vesicular nuclei, Muscle Fibre Atrophy, Splitting & Internally placed nuclei |
| Kleopa 2004, Cyprus | 1 | Muscle | Inflammatory cell infiltrates; Rimmed vacuoles; Sural nerve biopsy: Chronic active axonopathy; Loss of myelinated fibers; Regeneration & Necrotic fibers |
| Williams 2003, USA | 1 | Muscle | Basophilic rimmed vacuoles |
| Encephalopathy 5 | 9 | ||
| Brucke 1988, Austria | 1 | Brain | Brain edema; Periventricular lesions; Inflammatory necrosis; Demyelinated fibers; hypertrophy of the inferior olives; Vermal Bergmann gliosis; Lymphocyte infiltration of the pons and mesencephalon |
| Dimberg 2007, USA | 1 | Brain | Loss of Purkinje cells & granular layer cells; Astrocytic gliosis of the frontal, parietal, occipital cortices, globus pallidus, hippocampus, midbrain, pons, medulla; Widespread perivascular lymphocytosis in the cortex hippocampus and temporal gyrus, frontoparietal atrophy; |
| Hu 2006, USA | 5 | Brain | Non-specific gliosis & astrocytosis; Ubiquitin-positive inclusions in 1 patient |
| Keller 2006, USA | 1 | Brain Spinal cord | Loss of Purkinje cells; Neuronal loss of the dentate nucleus; Perivascular cuffing of lymphocytes; Arterial deformations of the pons, midbrain, thalamus and basal ganglia; Mild degeneration of the pyramidal tract & posterior columns; Loss of myelinated nerve fibers in the nerve roots |
| La Mantia, 1998, Italy | 1 | Brain | Brain edema; Calcifications, Increased vascularity, Mild neuronal loss, Reactive gliosis & Demyelination |
| Epilepsy 2 | 2 | ||
| Bye 1993, Australia | 1 | Brain | Pial angiomatosis consisting of groups of small veins entrapped by collagen; Severe sclerosis of veins; Lymphocytic perivascular cuffing in the cortical neuropil. Atrophy of the white matter and influx of macrophages & reactive gliosis (all occipital lobe) |
| Orstavik 1997, Norway | 1 | Brain | Megalencephaly |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rouvroye, M.D.; Zis, P.; Van Dam, A.-M.; Rozemuller, A.J.M.; Bouma, G.; Hadjivassiliou, M. The Neuropathology of Gluten-Related Neurological Disorders: A Systematic Review. Nutrients 2020, 12, 822. https://doi.org/10.3390/nu12030822
Rouvroye MD, Zis P, Van Dam A-M, Rozemuller AJM, Bouma G, Hadjivassiliou M. The Neuropathology of Gluten-Related Neurological Disorders: A Systematic Review. Nutrients. 2020; 12(3):822. https://doi.org/10.3390/nu12030822
Chicago/Turabian StyleRouvroye, Maxine D, Panagiotis Zis, Anne-Marie Van Dam, Annemieke J.M. Rozemuller, Gerd Bouma, and Marios Hadjivassiliou. 2020. "The Neuropathology of Gluten-Related Neurological Disorders: A Systematic Review" Nutrients 12, no. 3: 822. https://doi.org/10.3390/nu12030822
APA StyleRouvroye, M. D., Zis, P., Van Dam, A.-M., Rozemuller, A. J. M., Bouma, G., & Hadjivassiliou, M. (2020). The Neuropathology of Gluten-Related Neurological Disorders: A Systematic Review. Nutrients, 12(3), 822. https://doi.org/10.3390/nu12030822







