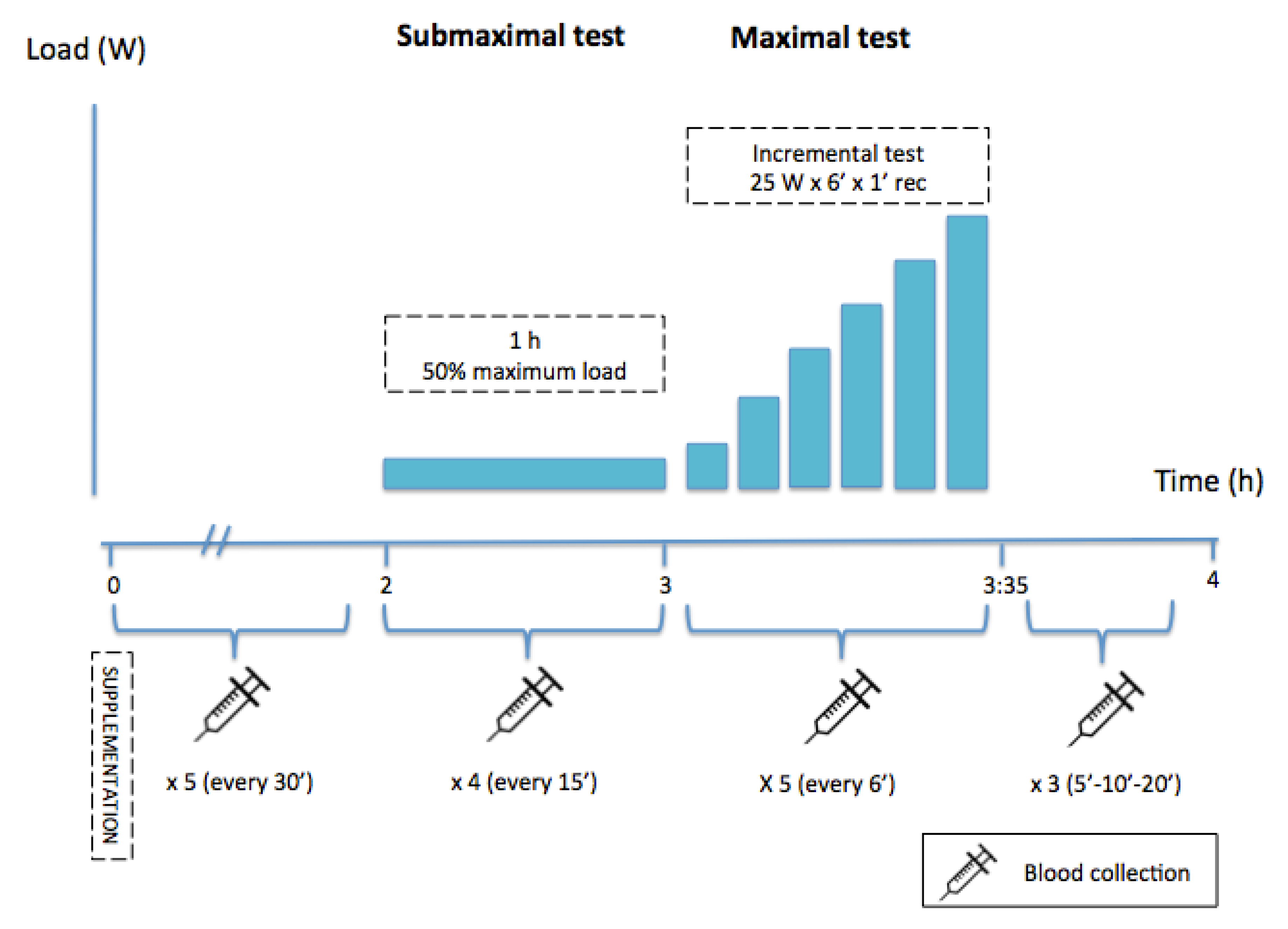
1. Introduction
In endurance sports, the pattern of energy substrate use changes over time, even when the exercise intensity remains constant. The longer the time spent on exercise, the greater the energy substrate contribution of fat [
1,
2]. In endurance exercise, the hormone-dependent release and oxidation of plasma fatty acids (FAs) increase in parallel to the gradual exhaustion of muscle glycogen reserves. This increased oxidation occurs in response to increased levels of circulating catecholamine (adrenaline and noradrenaline) and decreased levels of circulating insulin. Catecholamines and insulin play an important role in stimulating and inhibiting lipolytic activity, respectively [
3,
4].
Trained individuals make greater use of FAs as an energy source and perform better in endurance exercise due to their ability to increase glycogen stores and use them sparingly in submaximal efforts [
5,
6,
7,
8]. For this reason, acute FA supplementation has the effect of storing muscle glycogen during prolonged exercise because the oxidation rate of non-esterified fatty acids (NEFAs) partly depends on their blood plasma concentrations [
4,
8,
9].
Previous studies [
10,
11,
12,
13,
14,
15,
16,
17,
18], exploring the effects of acute FA supplementation through high fat intake before exercise, have not demonstrated improved performance. They have, in fact, shown the opposite, with intake causing gastrointestinal problems resulting from delayed gastric emptying induced by long-chain triglycerides (TGs). Those studies have shown that, during endurance exercises, while plasma NEFA availability increases, the pattern of substrate oxidation remains unchanged.
Almonds (
Prunus dulcis) are highly nutritional, mainly due to their high lipid content (25–66 g per 100 g
−1 (fresh weight)), which also makes them highly calorific (
Table 1). They are very rich in unsaturated FAs, especially oleic acid (monounsaturated) and linoleic acid (polyunsaturated), which account for around 90% of their total lipid content, although proportions vary widely depending on the almond variety. The amounts of saturated FAs they contain, such as myristic, palmitic, and stearic acids, are low (<10%), while the concentrations of carbohydrate, fibre, and protein per 100 g range between 1.8 and 7.4 g, 11 and 14 g, and 14 and 26 g, respectively [
19].
Almonds are a good source of α-tocopherol, riboflavin, magnesium, manganese, copper, and phosphorus. They are also rich in arginine, a substrate necessary for nitric oxide [
21,
22,
23]. The phenolic and polyphenolic compounds in almonds include mostly flavonoids, especially isorhamnetin-3-O-rutinoside and catechin [
24,
25] (
Table 2). This nutrient profile has been demonstrated to be important for humans, as the consumption of almonds is associated with improved oxidative stress biomarkers [
26,
27] and reduced inflammation [
28,
29] and is inversely related to cardiovascular diseases, diabetes, and certain cancers [
26,
30,
31].
Intense and prolonged physical effort increases reactive oxygen species (ROS) production due to, among other reasons, improved mitochondrial respiration chain oxidation flows; ROS are produced by an electron transfer that requires a high energy input with a very short lifetime (from milliseconds to nanoseconds) [
32]. Repeated and programmed exercise improves the ability to defend against ROS. However, ROS overproduction during exercise can overcome antioxidant defence capabilities, causing imbalances in the immune and endocrine systems, inducing fatigue, and impairing performance [
33,
34,
35]. As almonds are a good source of unsaturated FAs, antioxidants, and certain micronutrients, they can help maintain or improve exercise performance by modulating energy use and strengthening antioxidant defences. For example, quercetin [
36,
37,
38,
39] may help augment the training effectiveness on exercise performance by up-regulating mitochondrial biogenesis and oxygen sparing capacity and facilitating oxygen delivery to skeletal muscle, and arginine [
40,
41,
42,
43,
44,
45] may decrease ammonia liberation. Nonetheless, the impact of antioxidants and physiological markers on physical performance is not completely known [
46].
Based on the assumption that unsaturated FA-rich diets and endurance exercise both have positive (if different) effects on metabolic and cardiovascular health, and given that they both increase the oxidative capacity of fats, their combination is likely to be synergistic [
47]. While studies have demonstrated that unsaturated FA supplementation slightly increases fat oxidation after sports training compared to control supplementation, this change has not been reported to be reflected in maximum oxygen uptake (VO
2max) or other performance and physiological parameters [
47,
48,
49,
50].
In our experimental study, we evaluated the effect of acute FA supplementation (almonds), containing unsaturated FA and antioxidant micronutrients, on prolonged resistance training tests in laboratory conditions.
4. Discussion
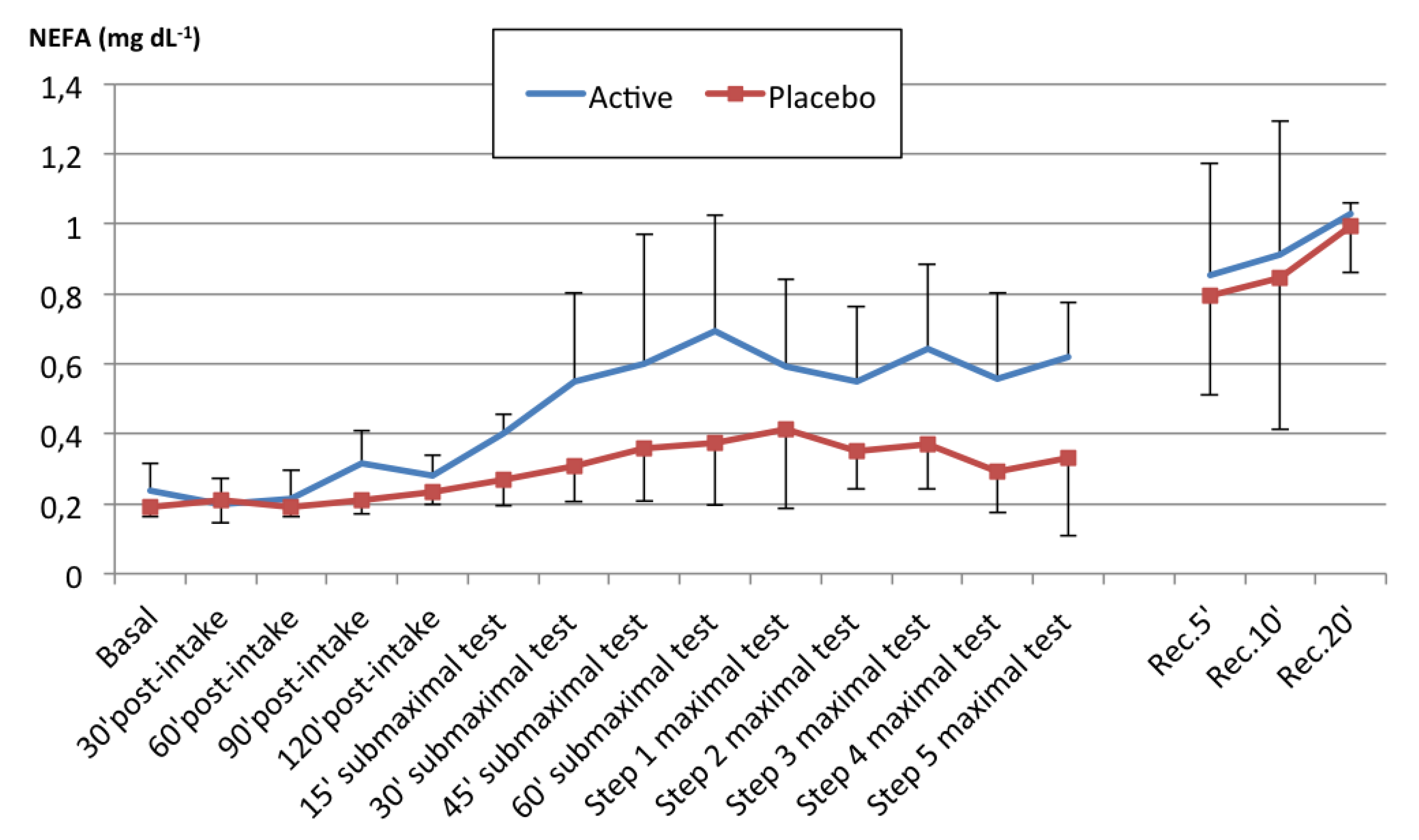
We evaluated the impact of acute FA supplementation in the form of almonds on plasma NEFA values and endurance performance, comparing it to a placebo. Our results indicate that acute almond supplementation modified the energy substrate availability pattern in plasma. In contrast to the little increase produced by the placebo supplementation, almond supplementation before exercise significantly increased plasma NEFA concentrations during the physical test by a mean value of 0.09 mg dL−1 (>30% vs. the placebo).
NEFAs, as an oxidisable fuel for physical exercise, can improve performance. According to previous studies [
14,
16,
51,
52,
53], NEFA availability may save on the use of muscle glycogen during exercise and, thus, delay fatigue. Those previous studies administered heparin to increase plasma NEFA levels; however, while this reduces the oxidation of muscle glycogen, it is not an acceptable pre-competition strategy. Our study demonstrated that NEFA values can be increased using exclusively nutritional strategies. Other studies using fat supplementation unaccompanied by heparin administration [
10,
11,
15] have reported increased NEFA values, but no improvement in performance. By contrast, we found that exercise performance improved by 20.6% following fat supplementation.
An important factor in fat supplementation during exercise is the type of FAs administered. It is important to bear in mind that fat digestion and absorption is a lengthy process, depending on the length of the FA chain, with long-chain TGs, for instance, being absorbed more slowly than short- or medium-chain TGs [
54]. Diet almond consumption (>42.5 g) may reduce the risk of CVD by improving blood lipids and by decreasing body weight and apolipoprotein B, but triglycerides, systolic blood pressure, apolipoprotein A1, high-sensitivity C-reactive protein, and lipoprotein (a) showed no difference [
55]. In a dose–response study, the results indicated that almond consumption increases oleic acid and monounsaturated fat content in serum triacylglycerol and non-esterified fatty acids fractions, which are inversely associated with CHD lipid risk factors and overall estimated 10-year CHD risk [
56]. However, after an acute intake of 60 g of almonds, triglycerides in plasma may be elevated because they are highly nutritional, mainly due to their high lipid content (25–66 g per 100 g
−1 (fresh weight)), and richness in unsaturated FAs, especially oleic acid (monounsaturated) and linoleic acid (polyunsaturated). In our data, triglycerides increased 14.7% with almond supplementation with respect to placebo after submaximal exercise, with similar dietetic and exercise conditions.
Several studies have evaluated the effects of supplements made with healthy foods rich in monounsaturated FAs, such as almonds, pistachios, and extra-virgin olive oil. In a study by Nieman et al. [
57], performance (measured as exercise time) worsened, possibly due to increases in plasma levels of compounds such as raffinose, sucrose, or myo-inositol, accompanied by an increase in leukotoxin (derived from linoleic acid), which may have had a negative impact on mitochondrial function. In studies by Boss et al. [
47], Capó et al. [
48,
49], and Esquius et al. [
50], performance (again measured as exercise time) was not affected by supplementation. However, Yi et al. [
58], in their evaluation involving 75 g of almonds administered as single pre-exercise supplements over 4 weeks, reported improved performance (measured as distance travelled). While the amount of almonds administered in that study (70 g) was similar to the 60 g administered in our study, the administration protocol differed.
The supplements used in our study were well received by the subjects and did not cause any gastrointestinal problems. The high fat content of the supplements had no negative influence on exercise performance in our study, unlike in other studies reporting impaired physical performance due to fat intake [
59]. Digestibility was good and plasma NEFA levels were observed to increase around 90 min after almond supplementation (
Figure 2). The increase in plasma NEFA levels occurred at an earlier stage in our study than in other studies [
60,
61].
The calorie difference between the active supplement (405 kcal) and the placebo (315 kcal) was 90 kcal, while the carbohydrate content was lower. The main difference was in the amount of fat. Further research is warranted to explore differences between different FAs used for acute supplementation in endurance exercise.
Studies in humans have shown that the consumption of almonds increases circulating levels of α-tocopherol in a dose-dependent manner [
62,
63] and reduces oxidative stress biomarker levels [
23,
26,
27]. The phenolic compounds in almonds have been shown to exert an antioxidant effect against free radicals [
27,
64] and to decrease inflammatory markers [
28,
29]. The phenolic and polyphenolic compounds in almonds may, therefore, contribute to improving the antioxidant capacity of athletes (not determined in this study).
No differences were observed for oxygen consumption or CO2 production during the submaximal effort, which could have been because there were no differences in the energy substrate used. During the maximal effort, there were also no differences in the maximum oxygen consumption reached, with more CO2 being produced. This could indicate that the positive effect on performance was due to the peripheral effect of the polyphenols rather than the energy savings from the extra lipid supplement, resulting in a reduced perception of fatigue that enabled the physical effort to last longer.
The main limitation of this study was the small number of participants and the fact that our sample was composed only of men. The complexity of the protocol, however, needed a homogeneous and trained sample to rule out any training effect. Studies with larger samples would be necessary to confirm our findings, explore individual differences in responses, and test differences arising from other factors such as gender. Since oxidative stress and inflammatory biomarkers could not be assessed in this research, we cannot evaluate the role of phenolic and polyphenolic compounds in almonds in improving the antioxidant capacity of the athletes. Therefore, further research is warranted analysing inflammatory and oxidation markers to confirm this hypothesis.
Our results, in summary, suggest that almonds can be included in pre-training or pre-competition supplements for endurance athletes.