Supplementation with a Mango Leaf Extract (Zynamite®) in Combination with Quercetin Attenuates Muscle Damage and Pain and Accelerates Recovery after Strenuous Damaging Exercise
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. General Overview
2.3. Pre-Tests and Familiarization
2.4. Main Experiments and Supplement administration
2.5. Assessment of Pain and Effectiveness of Concealment.
2.6. Vertical Jump Performance
2.7. Blood Sampling and Assessment of Biomarkers of Muscle Damage
2.8. Statistical Analysis
3. Results
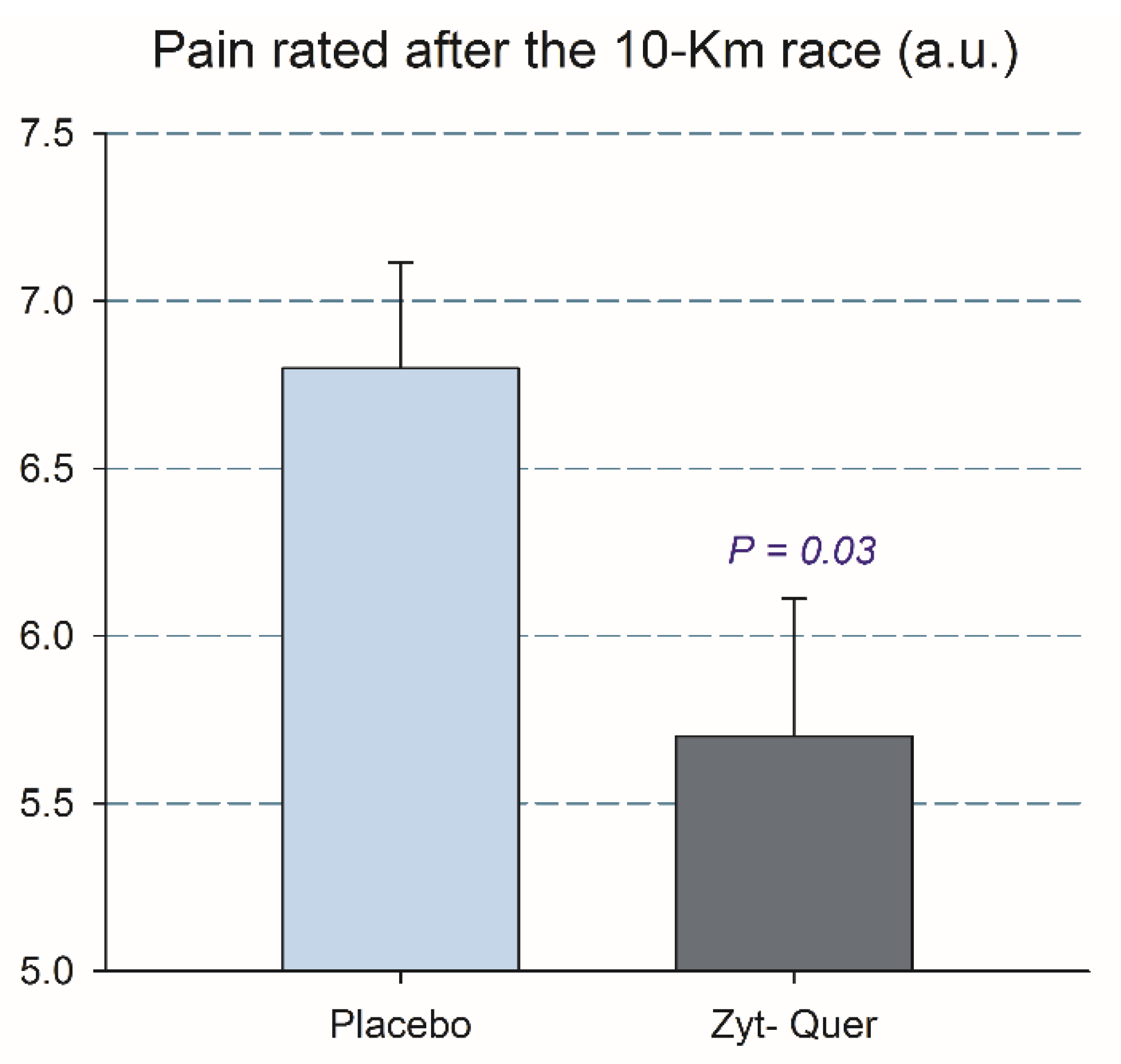
3.1. Effects on 10 km Running Time, Blood Lactate, RPE and Leg Pain
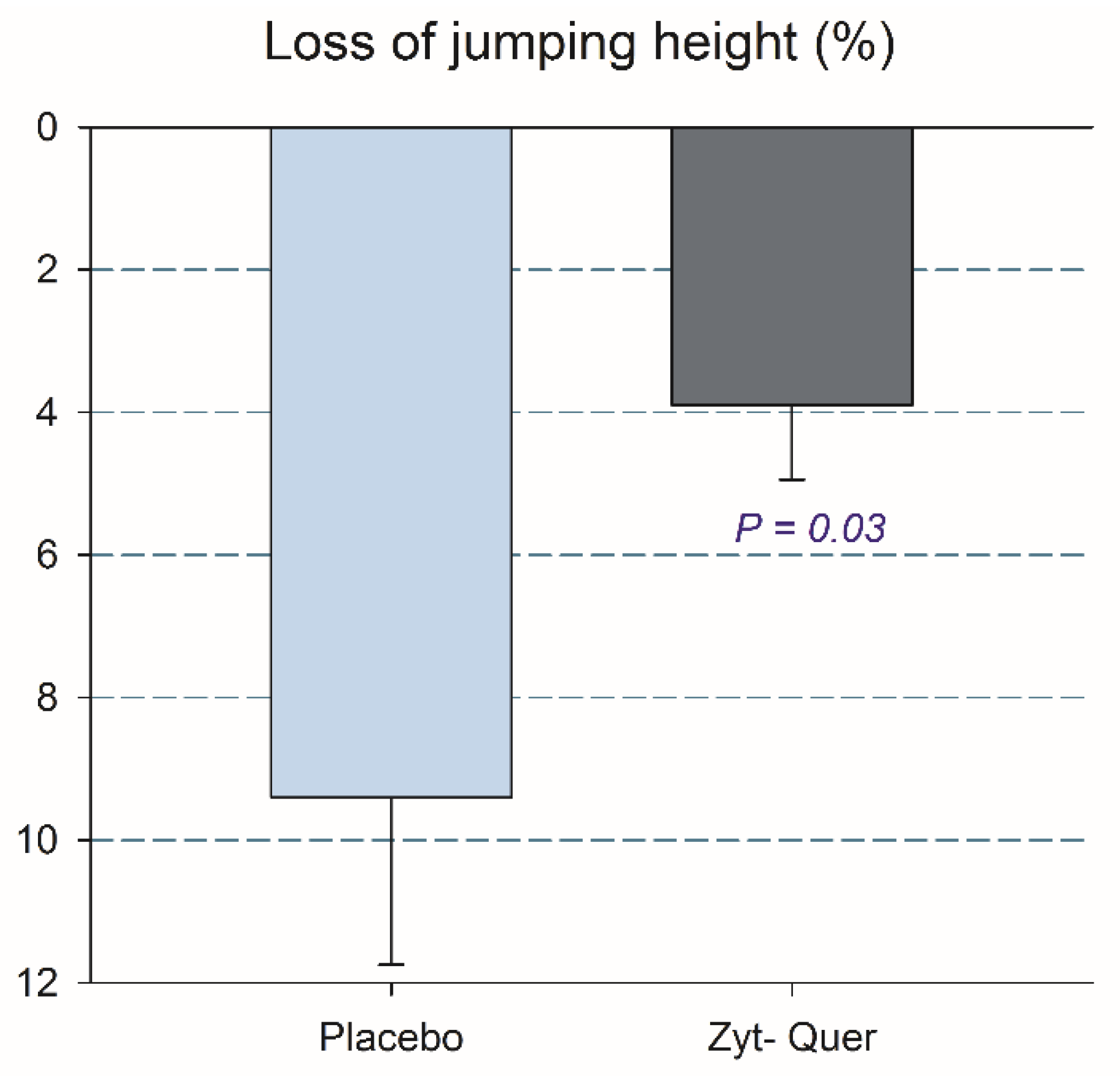
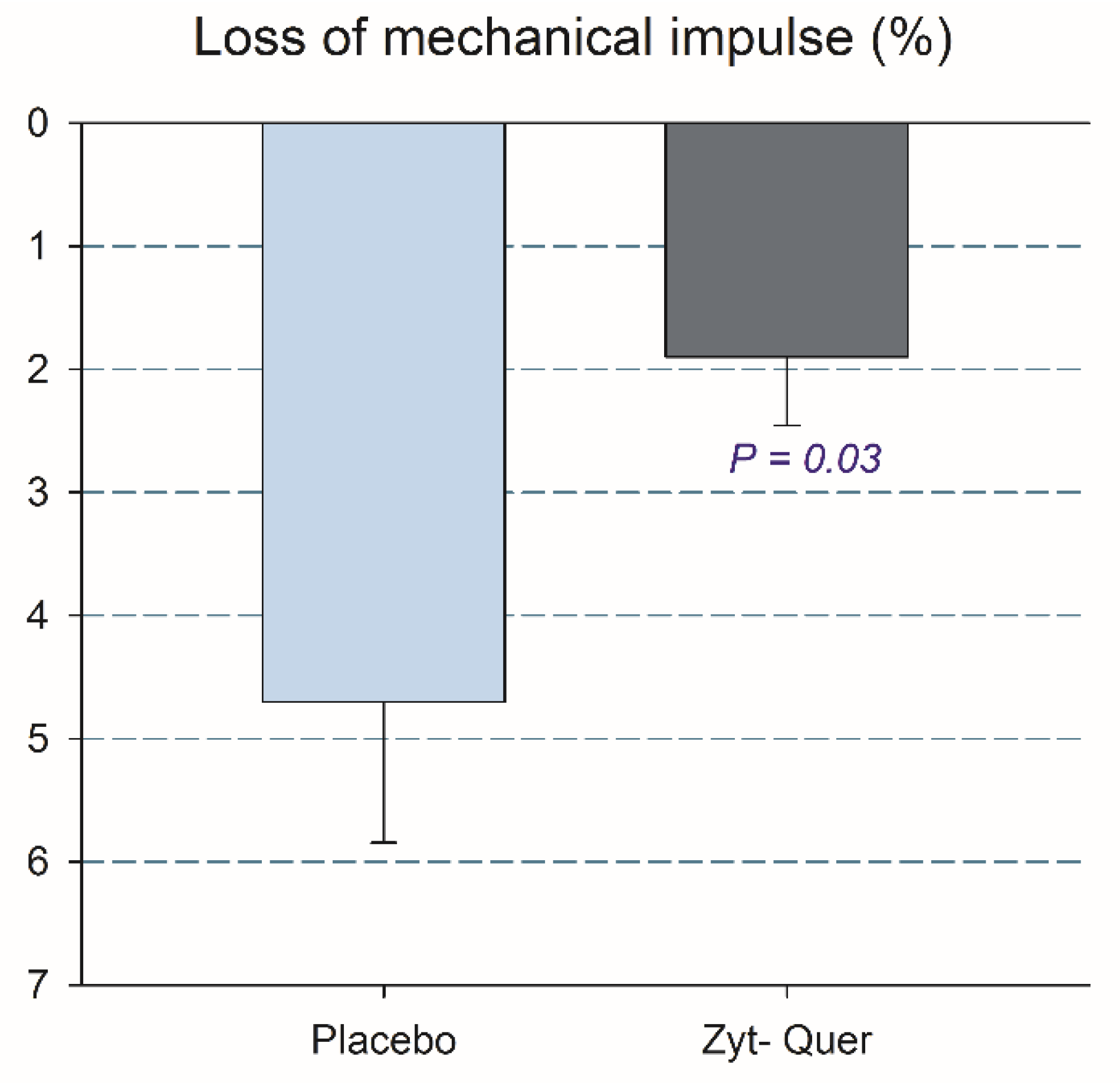
3.2. Effects on Vertical Jump Performance
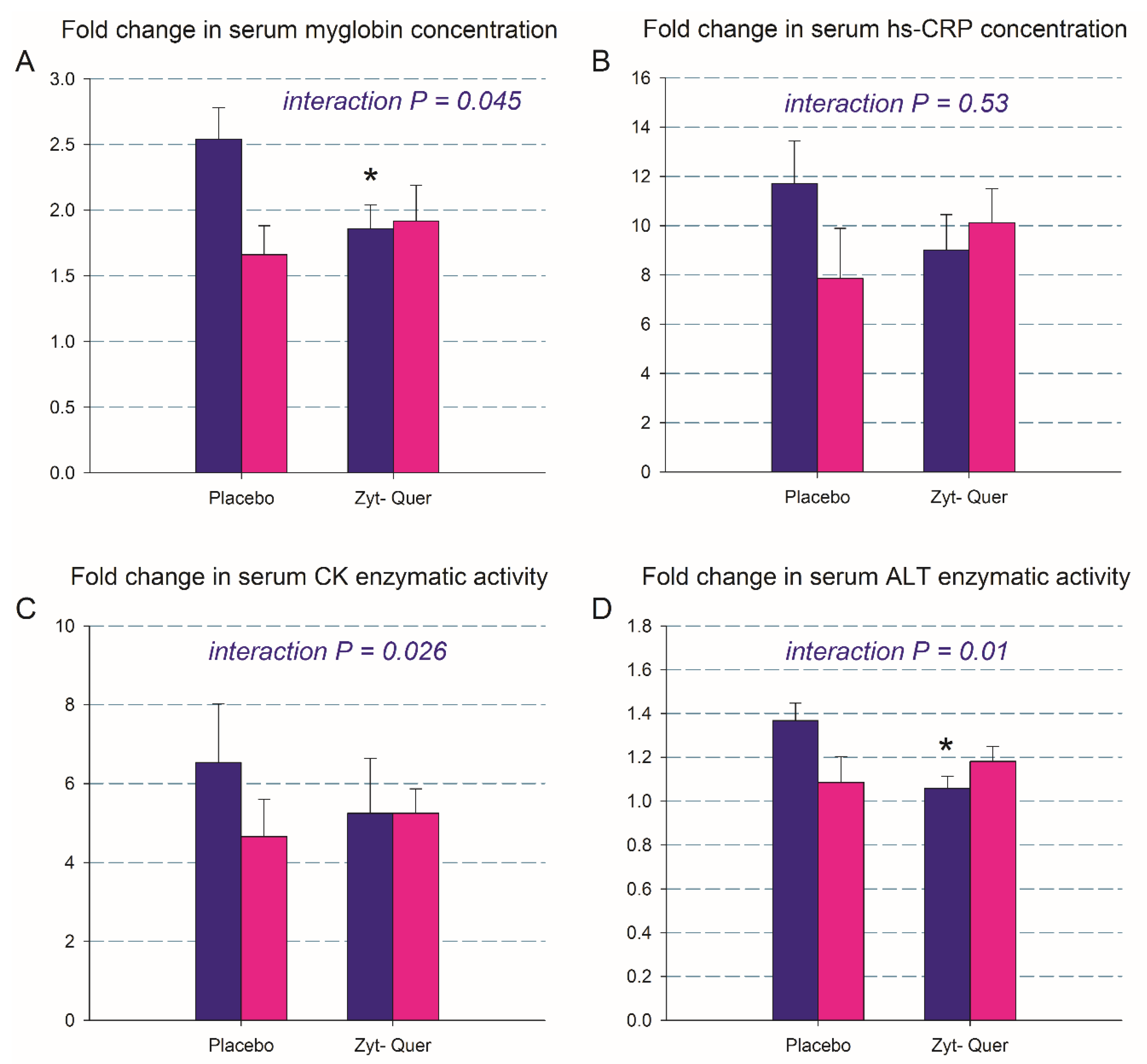
3.3. Biomarkers of Muscle Damage
3.4. Efficiency of Concealment
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Brancaccio, P.; Lippi, G.; Maffulli, N. Biochemical markers of muscular damage. Clin. Chem. Lab. Med. 2010, 48, 757–767. [Google Scholar] [CrossRef] [PubMed]
- Owens, D.J.; Twist, C.; Cobley, J.N.; Howatson, G.; Close, G.L. Exercise-induced muscle damage: What is it, what causes it and what are the nutritional solutions? Eur. J. Sport Sci. 2019, 19, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, J.C.; van Doornen, L.J.; Kemper, H.C. The role of antioxidant vitamins and enzymes in the prevention of exercise-induced muscle damage. Sports Med. 1996, 21, 213–238. [Google Scholar] [CrossRef] [PubMed]
- Peternelj, T.T.; Coombes, J.S. Antioxidant supplementation during exercise training: Beneficial or detrimental? Sports Med. 2011, 41, 1043–1069. [Google Scholar] [CrossRef] [PubMed]
- McGinley, C.; Shafat, A.; Donnelly, A.E. Does antioxidant vitamin supplementation protect against muscle damage? Sports Med. 2009, 39, 1011–1032. [Google Scholar] [CrossRef] [PubMed]
- Bowtell, J.; Kelly, V. Fruit-derived polyphenol supplementation for athlete recovery and performance. Sports Med. 2019, 49, 3–23. [Google Scholar] [CrossRef]
- Pal, R.; Chaudhary, M.J.; Tiwari, P.C.; Nath, R.; Pant, K.K. Pharmacological and biochemical studies on protective effects of mangiferin and its interaction with nitric oxide (no) modulators in adjuvant-induced changes in arthritic parameters, inflammatory, and oxidative biomarkers in rats. Inflammopharmacology 2019, 27, 291–299. [Google Scholar] [CrossRef]
- Li, M.; Wu, C.; Guo, H.; Chu, C.; Hu, M.; Zhou, C. Mangiferin improves hepatic damage-associated molecular patterns, lipid metabolic disorder and mitochondrial dysfunction in alcohol hepatitis rats. Food Funct. 2019, 10, 3514–3534. [Google Scholar] [CrossRef]
- Fu, Y.Y.; Zhang, F.; Zhang, L.; Liu, H.Z.; Zhao, Z.M.; Wen, X.R.; Wu, J.; Qi, D.S.; Sun, Y.; Du, Y.; et al. Mangiferin regulates interleukin-6 and cystathionine-b-synthase in lipopolysaccharide-induced brain injury. Cell. Mol. Neurobiol. 2014, 34, 651–657. [Google Scholar] [CrossRef]
- Mohammadi-Sartang, M.; Mazloom, Z.; Sherafatmanesh, S.; Ghorbani, M.; Firoozi, D. Effects of supplementation with quercetin on plasma c-reactive protein concentrations: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2017, 71, 1033–1039. [Google Scholar] [CrossRef]
- Bazzucchi, I.; Patrizio, F.; Ceci, R.; Duranti, G.; Sgro, P.; Sabatini, S.; Di Luigi, L.; Sacchetti, M.; Felici, F. The effects of quercetin supplementation on eccentric exercise-induced muscle damage. Nutrients 2019, 11, 205. [Google Scholar] [CrossRef] [PubMed]
- Casuso, R.A.; Martinez-Lopez, E.J.; Nordsborg, N.B.; Hita-Contreras, F.; Martinez-Romero, R.; Canuelo, A.; Martinez-Amat, A. Oral quercetin supplementation hampers skeletal muscle adaptations in response to exercise training. Scand. J. Med. Sci. Sports 2014, 24, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Byrne, C.; Twist, C.; Eston, R. Neuromuscular function after exercise-induced muscle damage: Theoretical and applied implications. Sports Med. 2004, 34, 49–69. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.M.; Suzuki, K.; Coombes, J.S. The influence of antioxidant supplementation on markers of inflammation and the relationship to oxidative stress after exercise. J. Nutr. Biochem. 2007, 18, 357–371. [Google Scholar] [CrossRef]
- Vassilakopoulos, T.; Karatza, M.H.; Katsaounou, P.; Kollintza, A.; Zakynthinos, S.; Roussos, C. Antioxidants attenuate the plasma cytokine response to exercise in humans. J. Appl. Physiol. 2003, 94, 1025–1032. [Google Scholar] [CrossRef]
- Michailidis, Y.; Karagounis, L.G.; Terzis, G.; Jamurtas, A.Z.; Spengos, K.; Tsoukas, D.; Chatzinikolaou, A.; Mandalidis, D.; Stefanetti, R.J.; Papassotiriou, I.; et al. Thiol-based antioxidant supplementation alters human skeletal muscle signaling and attenuates its inflammatory response and recovery after intense eccentric exercise. Am. J. Clin. Nutr. 2013, 98, 233–245. [Google Scholar] [CrossRef]
- Gelabert-Rebato, M.; Wiebe, J.C.; Martin-Rincon, M.; Gericke, N.; Perez-Valera, M.; Curtelin, D.; Galvan-Alvarez, V.; Lopez-Rios, L.; Morales-Alamo, D.; Calbet, J.A.L. Mangifera indica l. Leaf extract in combination with luteolin or quercetin enhances vo2peak and peak power output, and preserves skeletal muscle function during ischemia-reperfusion in humans. Front. Physiol. 2018, 9, 740. [Google Scholar] [CrossRef]
- Gelabert-Rebato, M.; Wiebe, J.C.; Martin-Rincon, M.; Galvan-Alvarez, V.; Curtelin, D.; Perez-Valera, M.; Habib, J.J.; Pérez-López, A.; Vega, T.; Morales-Alamo, D.; et al. Enhancement of exercise performance by 48 hours, and 15-day supplementation with mangiferin and luteolin in men. Nutrients 2019, 11, 344. [Google Scholar] [CrossRef]
- Gelabert-Rebato, M.; Martin-Rincon, M.; Galvan-Alvarez, V.; Gallego-Selles, A.; Martinez-Canton, M.; Vega-Morales, T.; Wiebe, J.C.; Fernandez-Del Castillo, C.; Castilla-Hernandez, E.; Diaz-Tiberio, O.; et al. A single dose of the mango leaf extract zynamite((r)) in combination with quercetin enhances peak power output during repeated sprint exercise in men and women. Nutrients 2019, 11, 2592. [Google Scholar] [CrossRef]
- Masibo, M.; He, Q. Major mango polyphenols and their potential significance to human health. Compr. Rev. Food Sci. Food Saf. 2008, 7, 309–319. [Google Scholar] [CrossRef]
- Das, J.; Ghosh, J.; Roy, A.; Sil, P.C. Mangiferin exerts hepatoprotective activity against d-galactosamine induced acute toxicity and oxidative/nitrosative stress via nrf2-nfkappab pathways. Toxicol. Appl. Pharmacol. 2012, 260, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Luczkiewicz, P.; Kokotkiewicz, A.; Dampc, A.; Luczkiewicz, M. Mangiferin: A promising therapeutic agent for rheumatoid arthritis treatment. Med. Hypotheses 2014, 83, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Suchal, K.; Malik, S.; Khan, S.I.; Malhotra, R.K.; Goyal, S.N.; Bhatia, J.; Kumari, S.; Ojha, S.; Arya, D.S. Protective effect of mangiferin on myocardial ischemia-reperfusion injury in streptozotocin-induced diabetic rats: Role of age-rage/mapk pathways. Sci. Rep. 2017, 7, 42027. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Liu, J.; Liu, H.Y.; Gao, L.H.; Feng, G.H.; Liu, X.; Li, L. Hypouricaemic action of mangiferin results from metabolite norathyriol via inhibiting xanthine oxidase activity. Pharm. Biol. 2016, 54, 1680–1686. [Google Scholar] [CrossRef] [PubMed]
- Braakhuis, A.J.; Hopkins, W.G. Impact of dietary antioxidants on sport performance: A review. Sports Med. 2015, 45, 939–955. [Google Scholar] [CrossRef] [PubMed]
- Ekinci Akdemir, F.N.; Gulcin, I.; Karagoz, B.; Soslu, R. Quercetin protects rat skeletal muscle from ischemia reperfusion injury. J. Enzyme Inhib. Med. Chem. 2016, 31, 162–166. [Google Scholar] [CrossRef]
- Ozyurek, M.; Bektasoglu, B.; Guclu, K.; Apak, R. Measurement of xanthine oxidase inhibition activity of phenolics and flavonoids with a modified cupric reducing antioxidant capacity (cuprac) method. Anal. Chim. Acta 2009, 636, 42–50. [Google Scholar] [CrossRef]
- Holland, J.A.; O’Donnell, R.W.; Chang, M.M.; Johnson, D.K.; Ziegler, L.M. Endothelial cell oxidant production: Effect of nadph oxidase inhibitors. Endothelium 2000, 7, 109–119. [Google Scholar] [CrossRef]
- Calbet, J.A.; Moysi, J.S.; Dorado, C.; Rodriguez, L.P. Bone mineral content and density in professional tennis players. Calcif. Tissue Int. 1998, 62, 491–496. [Google Scholar] [CrossRef]
- Poole, D.C.; Jones, A.M. Measurement of the maximum oxygen uptake vo2max: Vo2peak is no longer acceptable. J. Appl. Physiol. 2017, 122, 997–1002. [Google Scholar] [CrossRef]
- Borg, E.; Kaijser, L. A comparison between three rating scales for perceived exertion and two different work tests. Scand. J. Med. Sci. Sports 2006, 16, 57–69. [Google Scholar] [CrossRef] [PubMed]
- Perez-Gomez, J.; Olmedillas, H.; Delgado-Guerra, S.; Royo, I.A.; Vicente-Rodriguez, G.; Ortiz, R.A.; Chavarren, J.; Calbet, J.A. Effects of weight lifting training combined with plyometric exercises on physical fitness, body composition, and knee extension velocity during kicking in football. Appl. Physiol. Nutr. Metab. 2008, 33, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Arteaga, R.; Dorado, C.; Chavarren, J.; Calbet, J.A. Reliability of jumping performance in active men and women under different stretch loading conditions. J. Sports Med. Phys. Fitness 2000, 40, 26–34. [Google Scholar] [PubMed]
- Komi, P.V.; Bosco, C. Utilization of stored elastic energy in leg extensor muscles by men and women. Med. Sci. Sports 1978, 10, 261–265. [Google Scholar] [PubMed]
- Lippi, G.; Sanchis-Gomar, F.; Salvagno, G.L.; Aloe, R.; Schena, F.; Guidi, G.C. Variation of serum and urinary neutrophil gelatinase associated lipocalin (ngal) after strenuous physical exercise. Clin. Chem. Lab. Med. 2012, 50, 1585–1589. [Google Scholar] [CrossRef] [PubMed]
- Damas, F.; Nosaka, K.; Libardi, C.A.; Chen, T.C.; Ugrinowitsch, C. Susceptibility to exercise-induced muscle damage: A cluster analysis with a large sample. Int. J. Sports Med. 2016, 37, 633–640. [Google Scholar] [CrossRef]
- Newham, D.J.; McPhail, G.; Mills, K.R.; Edwards, R.H. Ultrastructural changes after concentric and eccentric contractions of human muscle. J. Neurol. Sci. 1983, 61, 109–122. [Google Scholar] [CrossRef]
- Lieber, R.L.; Woodburn, T.M.; Friden, J. Muscle damage induced by eccentric contractions of 25% strain. J. Appl. Physiol. 1991, 70, 2498–2507. [Google Scholar] [CrossRef]
- Peake, J.M.; Neubauer, O.; Della Gatta, P.A.; Nosaka, K. Muscle damage and inflammation during recovery from exercise. J. Appl. Physiol. 2017, 122, 559–570. [Google Scholar] [CrossRef]
- Hyldahl, R.D.; Hubal, M.J. Lengthening our perspective: Morphological, cellular, and molecular responses to eccentric exercise. Muscle Nerve 2014, 49, 155–170. [Google Scholar] [CrossRef]
- Proske, U.; Morgan, D.L. Muscle damage from eccentric exercise: Mechanism, mechanical signs, adaptation and clinical applications. J. Physiol. 2001, 537, 333–345. [Google Scholar] [CrossRef] [PubMed]
- Morgan, D.L.; Allen, D.G. Early events in stretch-induced muscle damage. J. Appl. Physiol. 1999, 87, 2007–2015. [Google Scholar] [CrossRef] [PubMed]
- McHugh, M.P.; Connolly, D.A.; Eston, R.G.; Gleim, G.W. Exercise-induced muscle damage and potential mechanisms for the repeated bout effect. Sports Med. 1999, 27, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Tebay, L.E.; Robertson, H.; Durant, S.T.; Vitale, S.R.; Penning, T.M.; Dinkova-Kostova, A.T.; Hayes, J.D. Mechanisms of activation of the transcription factor nrf2 by redox stressors, nutrient cues, and energy status and the pathways through which it attenuates degenerative disease. Free Radic. Biol. Med. 2015, 88, 108–146. [Google Scholar] [CrossRef]
- Duan, F.F.; Guo, Y.; Li, J.W.; Yuan, K. Antifatigue effect of luteolin-6-c-neohesperidoside on oxidative stress injury induced by forced swimming of rats through modulation of nrf2/are signaling pathways. Oxid. Med. Cell. Longev. 2017, 2017, 3159358. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, W.; Su, Z.Y.; Kong, A.N. The complexity of the nrf2 pathway: Beyond the antioxidant response. J. Nutr. Biochem. 2015, 26, 1401–1413. [Google Scholar] [CrossRef] [PubMed]
- Trevisan, G.; Hoffmeister, C.; Rossato, M.F.; Oliveira, S.M.; Silva, M.A.; Silva, C.R.; Fusi, C.; Tonello, R.; Minocci, D.; Guerra, G.P.; et al. Trpa1 receptor stimulation by hydrogen peroxide is critical to trigger hyperalgesia and inflammation in a model of acute gout. Free Radic. Biol. Med. 2014, 72, 200–209. [Google Scholar] [CrossRef]
- Schmidt, A.P.; Bohmer, A.E.; Antunes, C.; Schallenberger, C.; Porciuncula, L.O.; Elisabetsky, E.; Lara, D.R.; Souza, D.O. Anti-nociceptive properties of the xanthine oxidase inhibitor allopurinol in mice: Role of a1 adenosine receptors. Br. J. Pharmacol. 2009, 156, 163–172. [Google Scholar] [CrossRef]
- Chen, T.C.; Lin, K.Y.; Chen, H.L.; Lin, M.J.; Nosaka, K. Comparison in eccentric exercise-induced muscle damage among four limb muscles. Eur. J. Appl. Physiol. 2011, 111, 211–223. [Google Scholar] [CrossRef]
- Jamurtas, A.Z.; Theocharis, V.; Tofas, T.; Tsiokanos, A.; Yfanti, C.; Paschalis, V.; Koutedakis, Y.; Nosaka, K. Comparison between leg and arm eccentric exercises of the same relative intensity on indices of muscle damage. Eur. J. Appl. Physiol. 2005, 95, 179–185. [Google Scholar] [CrossRef]
- Stupka, N.; Lowther, S.; Chorneyko, K.; Bourgeois, J.M.; Hogben, C.; Tarnopolsky, M.A. Gender differences in muscle inflammation after eccentric exercise. J Appl Physiol (1985) 2000, 89, 2325–2332. [Google Scholar] [CrossRef] [PubMed]
- Danielsson, T.; Carlsson, J.; Schreyer, H.; Ahnesjo, J.; Ten Siethoff, L.; Ragnarsson, T.; Tugetam, A.; Bergman, P. Blood biomarkers in male and female participants after an ironman-distance triathlon. PLoS ONE 2017, 12, e0179324. [Google Scholar] [CrossRef] [PubMed]
- Tiidus, P.M. Estrogen and gender effects on muscle damage, inflammation, and oxidative stress. Can. J. Appl. Physiol. 2000, 25, 274–287. [Google Scholar] [CrossRef] [PubMed]
- Minahan, C.; Joyce, S.; Bulmer, A.C.; Cronin, N.; Sabapathy, S. The influence of estradiol on muscle damage and leg strength after intense eccentric exercise. Eur. J. Appl. Physiol. 2015, 115, 1493–1500. [Google Scholar] [CrossRef] [PubMed]
| Men | Women | p | |||||
|---|---|---|---|---|---|---|---|
| Age (years) | 23.1 | ± | 2.5 | 23.3 | ± | 3.4 | 0.75 |
| Height (cm) | 176.6 | ± | 5.8 | 165.7 | ± | 5.2 | 0.000 |
| Weight (kg) | 74.7 | ± | 6.8 | 61.0 | ± | 6.2 | 0.000 |
| Body fat (%) | 18.7 | ± | 4.1 | 26.9 | ± | 4.5 | 0.000 |
| Fat body mass (kg) | 14.1 | ± | 3.8 | 16.5 | ± | 4.2 | 0.04 |
| Lean body mass (kg) | 57.4 | ± | 5.3 | 41.9 | ± | 3.8 | 0.000 |
| Legs lean mass (kg) | 20.2 | ± | 2.1 | 14.8 | ± | 1.6 | 0.000 |
| VO2max (mL min−1) | 3246 | ± | 358 | 2360 | ± | 415 | 0.000 |
| VO2max (mL kg−1 min−1) | 43.6 | ± | 3.8 | 38.6 | ± | 4.5 | 0.000 |
| LLM VO2max (mL kg−1 min−1) | 161.3 | ± | 15.4 | 159.4 | ± | 18.1 | 0.71 |
| Placebo | Polyphenols | p | |||||
|---|---|---|---|---|---|---|---|
| Age (years) | 23.9 | ± | 3.2 | 22.5 | ± | 2.2 | 0.08 |
| Height (cm) | 172.3 | ± | 8.3 | 172.6 | ± | 7.2 | 0.90 |
| Weight (kg) | 69.2 | ± | 10.0 | 69.8 | ± | 9.4 | 0.85 |
| Body fat (%) | 21.0 | ± | 6.7 | 22.5 | ± | 4.8 | 0.37 |
| Fat body mass (kg) | 14.3 | ± | 4.5 | 15.6 | ± | 3.7 | 0.29 |
| Lean body mass (kg) | 52.0 | ± | 9.8 | 51.2 | ± | 8.2 | 0.77 |
| Legs lean mass (kg) | 18.2 | ± | 3.4 | 18.1 | ± | 3.2 | 0.91 |
| VO2max (mL min−1) | 2898 | ± | 621 | 2928 | ± | 539 | 0.86 |
| VO2max (mL kg−1 min−1) | 41.6 | ± | 4.6 | 41.9 | ± | 4.9 | 0.83 |
| LLM VO2max (mL kg−1 min−1) | 159 | ± | 11 | 163 | ± | 20 | 0.40 |
| 5-km run (s) | 1611 | ± | 212 | 1642 | ± | 201 | 0.60 |
| Placebo | Polyphenols | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Men (N = 14) | Women (N = 9) | p | Men (N = 16) | Women (N = 9) | p | |||||||||
| Age (years) | 23.9 | ± | 2.5 | 24.0 | ± | 4.3 | 0.91 | 22.4 | ± | 2.2 | 22.7 | ± | 2.4 | 0.76 |
| Height (cm) | 177.4 | ± | 4.8 | 164.4 | ± | 6.0 | 0.000 | 175.9 | ± | 6.5 | 166.9 | ± | 4.3 | 0.001 |
| Weight (kg) | 74.3 | ± | 8.0 | 61.3 | ± | 7.4 | 0.001 | 74.9 | ± | 6.6 | 60.7 | ± | 6.2 | 0.000 |
| Body fat (%) | 16.9 | ± | 4.0 | 27.3 | ± | 4.9 | 0.000 | 20.2 | ± | 3.5 * | 26.4 | ± | 4.2 | 0.001 |
| Fat body mass (kg) | 12.7 | ± | 3.8 | 16.9 | ± | 4.4 | 0.02 | 15.3 | ± | 3.6 | 16.2 | ± | 4.2 | 0.55 |
| Lean body mass (kg) | 58.4 | ± | 6.0 | 42.0 | ± | 4.5 | 0.000 | 56.5 | ± | 4.5 | 41.9 | ± | 3.3 | 0.000 |
| Legs lean mass (kg) | 20.3 | ± | 2.3 | 15.0 | ± | 1.8 | 0.000 | 20.1 | ± | 2.0 | 14.6 | ± | 1.3 | 0.000 |
| VO2max (mL min−1) | 3285 | ± | 416 | 2297 | ± | 334 | 0.000 | 3211 | ± | 310 | 2424 | ± | 495 | 0.000 |
| VO2max (mL kg−1 min−1) | 44.2 | ± | 3.5 | 37.4 | ± | 2.4 | 0.00 | 43.0 | ± | 4.1 | 39.8 | ± | 5.8 | 0.12 |
| LLM VO2max (mL kg−1 min−1) | 162 | ± | 11 | 153 | ± | 8 | 0.06 | 161 | ± | 19 | 166 | ± | 23 | 0.58 |
| 5-km run (s) | 1509 | ± | 144 | 1770 | ± | 209 | 0.002 | 1558 | ± | 159 | 1792 | ± | 186 | 0.003 |
| Placebo | Polyphenols | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Men (N = 14) | Women (N = 9) | p | Men (N = 16) | Women (N = 9) | p | |||||||||
| 10-km race (s) | 3128 | ± | 357 | 3802 | ± | 590 | 0.003 | 3209 | ± | 351 | 3763 | ± | 487 | 0.003 |
| Ratio 10km/5Km | 2.07 | ± | 0.12 | 2.14 | ± | 0.10 | 0.18 | 2.06 | ± | 0.10 | 2.10 | ± | 0.10 | 0.43 |
| Lactate (mM) | 5.9 | ± | 2.4 | 4.8 | ± | 1.2 | 0.21 | 6.2 | ± | 2.8 | 4.1 | ± | 1.6 | 0.05 |
| RPE Post 10 Km | 7.9 | ± | 1.2 | 8.7 | ± | 1.1 | 0.11 | 7.6 | ± | 1.5 | 8.6 | ± | 0.9 | 0.06 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martin-Rincon, M.; Gelabert-Rebato, M.; Galvan-Alvarez, V.; Gallego-Selles, A.; Martinez-Canton, M.; Lopez-Rios, L.; Wiebe, J.C.; Martin-Rodriguez, S.; Arteaga-Ortiz, R.; Dorado, C.; et al. Supplementation with a Mango Leaf Extract (Zynamite®) in Combination with Quercetin Attenuates Muscle Damage and Pain and Accelerates Recovery after Strenuous Damaging Exercise. Nutrients 2020, 12, 614. https://doi.org/10.3390/nu12030614
Martin-Rincon M, Gelabert-Rebato M, Galvan-Alvarez V, Gallego-Selles A, Martinez-Canton M, Lopez-Rios L, Wiebe JC, Martin-Rodriguez S, Arteaga-Ortiz R, Dorado C, et al. Supplementation with a Mango Leaf Extract (Zynamite®) in Combination with Quercetin Attenuates Muscle Damage and Pain and Accelerates Recovery after Strenuous Damaging Exercise. Nutrients. 2020; 12(3):614. https://doi.org/10.3390/nu12030614
Chicago/Turabian StyleMartin-Rincon, Marcos, Miriam Gelabert-Rebato, Victor Galvan-Alvarez, Angel Gallego-Selles, Miriam Martinez-Canton, Laura Lopez-Rios, Julia C. Wiebe, Saul Martin-Rodriguez, Rafael Arteaga-Ortiz, Cecilia Dorado, and et al. 2020. "Supplementation with a Mango Leaf Extract (Zynamite®) in Combination with Quercetin Attenuates Muscle Damage and Pain and Accelerates Recovery after Strenuous Damaging Exercise" Nutrients 12, no. 3: 614. https://doi.org/10.3390/nu12030614
APA StyleMartin-Rincon, M., Gelabert-Rebato, M., Galvan-Alvarez, V., Gallego-Selles, A., Martinez-Canton, M., Lopez-Rios, L., Wiebe, J. C., Martin-Rodriguez, S., Arteaga-Ortiz, R., Dorado, C., Perez-Regalado, S., Santana, A., Morales-Alamo, D., & Calbet, J. A. L. (2020). Supplementation with a Mango Leaf Extract (Zynamite®) in Combination with Quercetin Attenuates Muscle Damage and Pain and Accelerates Recovery after Strenuous Damaging Exercise. Nutrients, 12(3), 614. https://doi.org/10.3390/nu12030614








