Duality of Tocopherol Isoforms and Novel Associations with Vitamins Involved in One-Carbon Metabolism: Results from an Elderly Sample of the LifeLines Cohort Study
Abstract
1. Introduction
2. Methods
2.1. Study Population
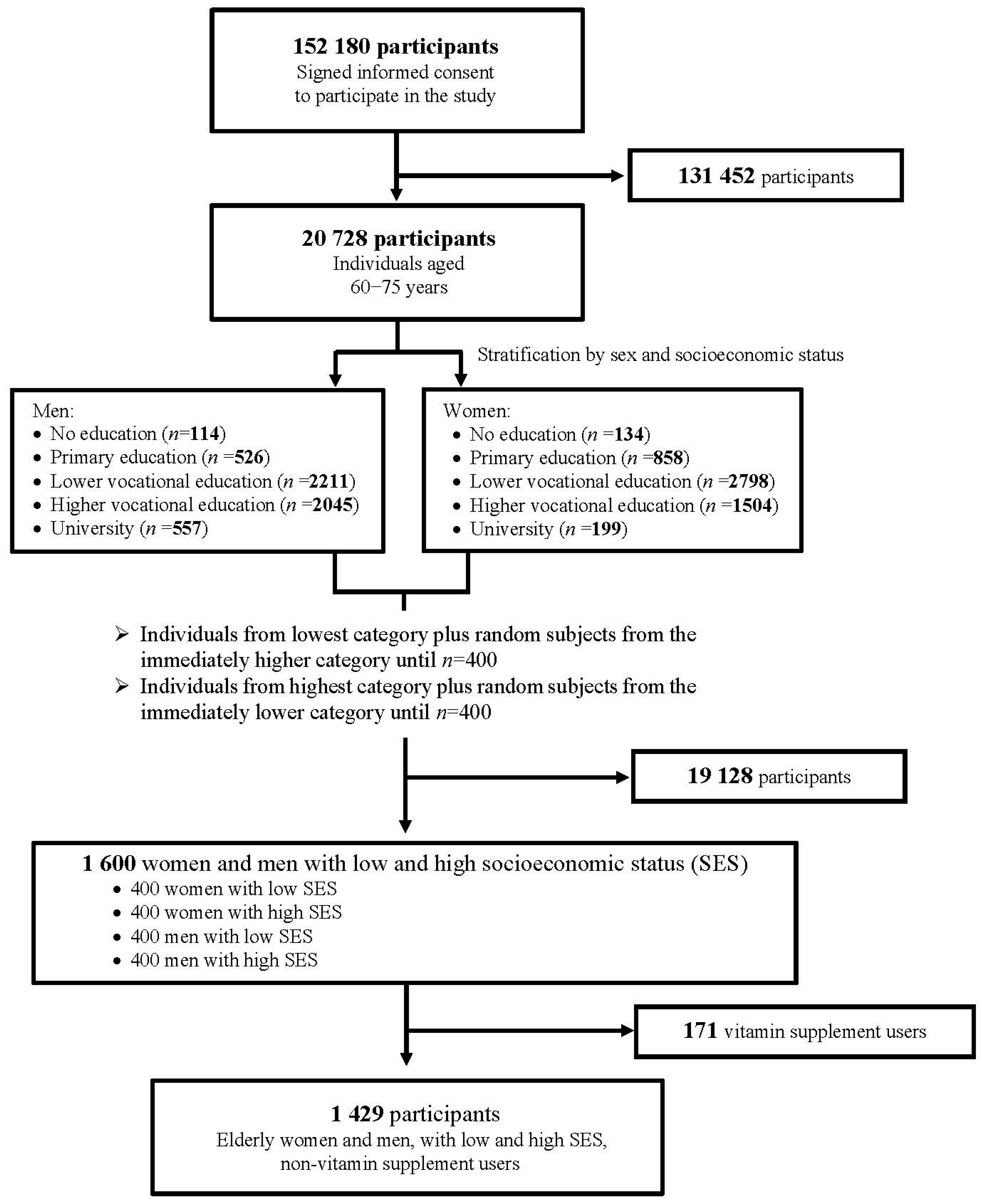
2.2. Selection of Study Population
2.3. Data Collection
2.4. Biochemical Measurements
2.5. Statistical Analyses
3. Results
3.1. Non-Indexed and Indexed α-Tocopherol and Biological Characteristics
3.2. Non-Indexed and Indexed γ-Tocopherol and Biological Characteristics
3.3. Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Catalgol, B.; Ozer, N.K. Protective effects of vitamin E against hypercholesterolemia-induced age-related diseases. Genes Nutr. 2012, 7, 91–98. [Google Scholar] [CrossRef]
- Karademir, B.; Ozer, N.K. Molecular function of tocopherols in age related diseases. Curr. Pharm. Des. 2014, 20, 3030–3035. [Google Scholar] [CrossRef]
- Nadiger, H.A.; Krishnan, R.; Radhaiah, G. Studies on interactions of vitamin E with thiamine, niacin and vitamin B12. Clin. Chim. Acta 1981, 116, 9–16. [Google Scholar] [CrossRef]
- Galli, F.; Azzi, A.; Birringer, M.; Cook-Mills, J.M.; Eggersdorfer, M.; Frank, J.; Cruciani, G.; Lorkowski, S.; Özer, N.K. Vitamin E: Emerging aspects and new directions. Free Radic. Biol. Med. 2017, 102, 16–36. [Google Scholar] [CrossRef]
- Thurnham, D.I.; Davies, J.A.; Crump, B.J.; Situnayake, R.D.; Davis, M. The use of different lipids to express serum tocopherol: Lipid ratios for the measurement of vitamin E status. Ann. Clin. Biochem. 1986, 23, 514–520. [Google Scholar] [CrossRef]
- Traber, M.G.; Mah, E.; Leonard, S.W.; Bobe, G.; Bruno, R.S. Metabolic syndrome increases dietary α-tocopherol requirements as assessed using urinary and plasma vitamin E catabolites: A double-blind, crossover clinical trial. Am. J. Clin. Nutr. 2017, 105, 571–579. [Google Scholar] [CrossRef]
- Bakker, S.J.; Gans, R.O.; ter Maaten, J.C.; Teerlink, T.; Westerhoff, H.V.; Heine, R.J. The potential role of adenosine in the pathophysiology of the insulin resistance syndrome. Atherosclerosis 2001, 155, 283–290. [Google Scholar] [CrossRef]
- Bakker, S.J.; IJzerman, R.G.; Teerlink, T.; Westerhoff, H.V.; Gans, R.O.; Heine, R.J. Cytosolic triglycerides and oxidative stress in central obesity: The missing link between excessive atherosclerosis, endotelial dysfunction, and beta-cell failure? Atherosclerosis 2000, 148, 17–21. [Google Scholar] [CrossRef]
- Hulsegge, G.; Herber-Gast, G.-C.M.; Spijkerman, A.M.W.; Susan, H.; Picavet, J.; van der Schouw, Y.T.; Bakker, S.J.L.; Gansevoort, R.T.; Dollé, M.E.T.; Smit, H.A.; et al. Obesity and Age-Related Changes in Markers of Oxidative Stress and Inflammation Across Four Generations. Obesity 2016, 24, 1389–1396. [Google Scholar] [CrossRef] [PubMed]
- Schindhelm, R.K.; Diamant, M.; Bakker, S.J.L.; van Dijk, R.A.J.M.; Scheffer, P.G.; Teerlink, T.; Kostense, P.J.; Heine, R.J. Liver alanine aminotransferase, insulin resistance and endothelial dysfunction in normotriglyceridaemic subjects with type 2 diabetes mellitus. Eur. J. Clin. Investig. 2005, 35, 369–374. [Google Scholar] [CrossRef] [PubMed]
- van der Harst, P.; Bakker, S.J.L.; de Boer, R.A.; Wolffenbuttel, B.H.R.; Johnson, T.; Caulfield, M.J.; Navis, G. Replication of the five novel loci for uric acid concentrations and potential mediating mechanisms. Hum. Mol. Genet. 2010, 19, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Selhub, J. Folate, vitamin B12 and vitamin B6 and one carbon metabolism. J. Nutr. Health Aging 2002, 6, 39–42. [Google Scholar] [PubMed]
- Zhang, J.; Hu, X.; Zhang, J. Associations between serum vitamin E concentration and bone mineral density in the US elderly population. Osteoporos. Int. 2017, 28, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H. Vitamin E administration may decrease the incidence of pneumonia in elderly males. Clin. Interv. Aging 2016, 11, 1379–1385. [Google Scholar] [CrossRef]
- Shi, W.; Liu, J.; Cao, Y.; Zhu, Y.; Guan, K.; Chen, Y. Association of dietary and serum vitamin E with bone mineral density in middle-aged and elderly Chinese adults: A cross-sectional study. Br. J. Nutr. 2016, 115, 113–120. [Google Scholar] [CrossRef]
- Michaëlsson, K.; Wolk, A.; Byberg, L.; Ärnlöv, J.; Melhus, H. Intake and serum concentrations of α-tocopherol in relation to fractures in elderly women and men: 2 cohort studies. Am. J. Clin. Nutr. 2014, 99, 107–114. [Google Scholar] [CrossRef]
- Shahar, S.; Lee, L.K.; Rajab, N.; Lim, C.L.; Harun, N.A.; Noh, M.F.N.M.; Mian-Then, S.; Jamal, R. Association between vitamin A, vitamin E and apolipoprotein E status with mild cognitive impairment among elderly people in low-cost residential areas. Nutr. Neurosci. 2013, 16, 6–12. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, A.; Li, Y.; Han, X.; Wang, Q.; Liang, H. Vitamin E supplementation protects erythrocyte membranes from oxidative stress in healthy Chinese middle-aged and elderly people. Nutr. Res. 2012, 32, 328–334. [Google Scholar] [CrossRef]
- Belisle, S.E.; Hamer, D.H.; Leka, L.S.; Dallal, G.E.; Delgado-Lista, J.; Fine, B.C.; Jacques, P.F.; Ordovas, J.M.; Meydani, S.N. IL-2 and IL-10 gene polymorphisms are associated with respiratory tract infection and may modulate the effect of vitamin E on lower respiratory tract infections in elderly nursing home residents. Am. J. Clin. Nutr. 2010, 92, 106–114. [Google Scholar] [CrossRef]
- Lee, G.Y.; Han, S.N. The Role of Vitamin E in Immunity. Nutrients 2018, 10, 1614. [Google Scholar] [CrossRef]
- Waniek, S.; di Giuseppe, R.; Esatbeyoglu, T.; Ratjen, I.; Enderle, J.; Jacobs, G.; Nöthlings, U.; Koch, M.; Schlesinger, S.; Rimbach, G.; et al. Association of Circulating Vitamin E (α- and γ-Tocopherol) Levels with Gallstone Disease. Nutrients 2018, 10, 133. [Google Scholar] [CrossRef] [PubMed]
- Stolk, R.P.; Rosmalen, J.G.M.; Postma, D.S.; de Boer, R.A.; Navis, G.; Slaets, J.P.J.; Ormel, J.; Wolffenbuttel, B.H.R. Universal risk factors for multifactorial diseases: LifeLines: A three-generation population-based study. Eur. J. Epidemiol. 2008, 23, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Korošec, T.; Tomažin, U.; Horvat, S.; Keber, R.; Salobir, J. The diverse effects of α- and γ-tocopherol on chicken liver transcriptome. Poult. Sci. 2017, 96, 667–680. [Google Scholar] [CrossRef] [PubMed]
- Scholtens, S.; Smidt, N.; Swertz, M.A.; Bakker, S.J.L.; Dotinga, A.; Vonk, J.M.; van Dijk, F.; van Zon, S.K.R.; Wijmenga, C.; Wolffenbuttel, B.H.R.; et al. Cohort Profile: LifeLines, a three-generation cohort study and biobank. Int. J. Epidemiol. 2015, 44, 1172–1780. [Google Scholar] [CrossRef] [PubMed]
- Vart, P.; Reijneveld, S.A.; Bültmann, U.; Gansevoort, R.T. Added value of screening for CKD among the elderly or persons with low socioeconomic status. Clin. J. Am. Soc. Nephrol. 2015, 10, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Riphagen, I.J.; van der Molen, J.C.; van Faassen, M.; Navis, G.; de Borst, M.H.; Muskiet, F.A.J.; de Jong, W.H.A.; Bakker, S.J.L.; Kema, I.P. Measurement of plasma vitamin K1 (phylloquinone) and K2 (menaquinones-4 and -7) using HPLC-tandem mass spectrometry. Clin. Chem. Lab. Med. 2016, 54, 1201–1210. [Google Scholar] [CrossRef]
- Ren, X.; Chen, Z.A.; Zheng, S.; Han, T.; Li, Y.; Liu, W.; Hu, Y. Association between Triglyceride to HDL-C Ratio (TG/HDL-C) and Insulin Resistance in Chinese Patients with Newly Diagnosed Type 2 Diabetes Mellitus. PLoS ONE 2016, 11, e0154345. [Google Scholar] [CrossRef]
- Boenzi, S.; Rizzo, C.; Di Ciommo, V.M.; Martinelli, D.; Goffredo, B.M.; la Marca, G.; Dionisi-Vici, C. Simultaneous determination of creatine and guanidinoacetate in plasma by liquid chromatography-tandem mass spectrometry (LC-MS/MS). J. Pharm. Biomed. Anal. 2011, 56, 792–798. [Google Scholar] [CrossRef]
- Talwar, D.; Quasim, T.; McMillan, D.C.; Kinsella, J.; Williamson, C.; O’Reilly, D.S.J. Optimisation and validation of a sensitive high-performance liquid chromatography assay for routine measurement of pyridoxal 5-phosphate in human plasma and red cells using pre-column semicarbazide derivatisation. J. Chromatogr. B 2003, 792, 333–343. [Google Scholar] [CrossRef]
- Blom, H.J.; van Rooij, A.; Hogeveen, M. A simple high-throughput method for the determination of plasma methylmalonic acid by liquid chromatography-tandem mass spectrometry. Clin. Chem. Lab. Med. 2007, 45, 645–650. [Google Scholar] [CrossRef]
- Casetta, B.; Jans, I.; Billen, J.; Vanderschueren, D.; Bouillon, R. Development of a method for the quantification of 1alpha,25(OH)2-vitamin D3 in serum by liquid chromatography tandem mass spectrometry without derivatization. Eur. J. Mass Spectrom. 2010, 16, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Moazzami, A.A.; Frank, S.; Gombert, A.; Sus, N.; Bayram, B.; Rimbach, G.; Frank, J. Non-targeted 1H-NMR-metabolomics suggest the induction of master regulators of energy metabolism in the liver of vitamin E-deficient rats. Food Funct. 2015, 6, 1090–1097. [Google Scholar] [CrossRef] [PubMed]
- Podszun, M.C.; Grebenstein, N.; Spruss, A.; Schlueter, T.; Kremoser, C.; Bergheim, I.; Frank, J. Dietary α-tocopherol and atorvastatin reduce high-fat-induced lipid accumulation and down-regulate CD36 protein in the liver of guinea pigs. J. Nutr. Biochem. 2014, 25, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Chai, W.; Novotny, R.; Maskarinec, G.; Le Marchand, L.; Franke, A.A.; Cooney, R.V. Serum Coenzyme Q10, α-Tocopherol, γ-Tocopherol, and C-Reactive Protein Levels and Body Mass Index in Adolescent and Premenopausal Females. J Am Coll Nutr. 2014, 33, 192–197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zou, Y.; Wang, D.-H.; Sakano, N.; Sato, Y.; Iwanaga, S.; Taketa, K.; Kubo, M.; Takemoto, K.; Masatomi, C.; Inoue, K.; et al. Associations of serum retinol, α-tocopherol, and γ-tocopherol with biomarkers among healthy Japanese men. Int. J. Environ. Res. Public Health 2014, 11, 1647–1660. [Google Scholar] [CrossRef] [PubMed]
- Sotomayor, C.G.; Rodrigo, R.; Gomes-Neto, A.W.; Gormaz, J.G.; Pol, R.A.; Minović, I.; Eggersdorfer, M.L.; Vos, M.; Riphagen, I.J.; de Borst, M.H.; et al. Plasma versus Erythrocyte Vitamin E in Renal. Transplant Recipients, and Duality of Tocopherol Species. Nutrients 2019, 11, 2821. [Google Scholar] [CrossRef]
- Azzi, A.; Gysin, R.; Kempná, P.; Munteanu, A.; Villacorta, L.; Visarius, T.; Zingg, J.M. Regulation of gene expression by α-tocopherol. Biol. Chem. 2004, 385, 585–591. [Google Scholar] [CrossRef]
- Azzi, A.; Gysin, R.; Kempná, P.; Munteanu, A.; Negis, Y.; Villacorta, L.; Visarius, T.; Zingg, J.M. Vitamin E mediates cell signaling and regulation of gene expression. Ann. N. Y. Acad. Sci. 2004, 1031, 86–95. [Google Scholar] [CrossRef]
- Fischer, A.; Rimbach, G. Gene Regulatory Activity of Vitamin E. In Vitamin E in Human Health; Springer International Publishing: Berlin, Germany, 2019; pp. 81–98. [Google Scholar]
- Cook-mills, J.M.; Abdala-valencia, H.; Hartert, T. Two Faces of Vitamin E in the Lung. Am. J. Respir. Crit. Care Med. 2013, 188, 279–1284. [Google Scholar] [CrossRef]
- Cooney, R.V.; Franke, A.A.; Wilkens, L.R.; Gill, J.; Kolonel, L.N. Elevated plasma gamma-tocopherol and decreased alpha-tocopherol in men are associated with inflammatory markers and decreased plasma 25-OH vitamin D. Nutr. Cancer 2008, 60, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wongsiriroj, N.; Blaner, W.S. The multifaceted nature of retinoid transport and metabolism. Hepatobiliary Surg. Nutr. 2014, 3, 126–139. [Google Scholar] [PubMed]
- Shamim, A.A.; Kabir, A.; Merrill, R.D.; Ali, H.; Rashid, M.; Schulze, K.; Labrique, A.; West, K.P.; Christian, P. Plasma zinc, vitamin B(12) and α-tocopherol are positively and plasma γ-tocopherol is negatively associated with Hb concentration in early pregnancy in north-west Bangladesh. Public Health Nutr. 2013, 16, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Gaedicke, S.; Zhang, X.; Schmelzer, C.; Lou, Y.; Doering, F.; Frank, J.; Rimbach, G. Vitamin E dependent microRNA regulation in rat liver. FEBS Lett. 2008, 582, 3542–3546. [Google Scholar] [CrossRef]
| Baseline Characteristics | n = 1429 | α-Tocopherol | γ-Tocopherol | ||
|---|---|---|---|---|---|
| Standardization | Standardization | ||||
| None | Lipids | None | Lipids | ||
| Demographics and Anthropometrics | |||||
| Age, years † | 66 ± 4 § | −0.04 | −0.01 | −0.08 ** | −0.07 * |
| Sex, male † | 727 (51) ¶ | 0.17 *** | 0.08 ** | 0.10 *** | −0.04 |
| Body mass index, kg/m2 | 26.4 (24.1–29.4) ¥ | −0.03 | −0.22 *** | 0.10 *** | 0.06 * |
| Waist circumference, cm | 101 ± 9 | −0.02 | −0.17 *** | 0.08 ** | 0.04 |
| Systolic blood pressure, mmHg | 133 (122–145) | 0.08 ** | −0.11 *** | 0.08 ** | 0.002 |
| Diastolic blood pressure, mmHg | 75 (69–81) | 0.08 ** | −0.06 * | 0.05 | −0.02 |
| Smoking status | |||||
| Never smoker | 484 (34) | — | — | — | — |
| Former smoker | 759 (53) | 0.02 | 0.02 | 0.07 * | 0.06 * |
| Current smoker | 173 (12) | −0.004 | −0.09 *** | 0.02 | −0.03 |
| Dietary intake | |||||
| Total protein, g/d | 69 (57–82) | −0.03 | 0.07 * | −0.05 | −0.01 |
| Animal protein, g/d | 41 (32–50) | −0.02 | 0.03 | −0.04 | −0.03 |
| Vegetable protein, g/d | 28 (22–34) | −0.02 | 0.10 ** | −0.04 | 0.02 |
| Total carbohydrates, g/d | 202 ± 74 | −0.04 | 0.05 | −0.09 ** | −0.05 |
| Total fat, g/d | 72 ± 31 | −0.03 | 0.06 * | 0.01 | 0.06 |
| Alcohol intake, g/d | 6.2 (0.8–16.4) | 0.06 * | 0.06 | 0.09 ** | 0.07 * |
| Energy intake, kCal/d | 1849 ± 643 | −0.03 | 0.07 * | −0.03 | 0.01 |
| Use of multivitamins | 0 (0) | — | — | — | |
| Vitamins | |||||
| Urinary creatine/creatinine ratio × 1000 | 14 (9–53) | 0.06 * | <0.001 | 0.05 | 0.03 |
| Pyridoxal phosphate, nmol/L | 51 (36–76) | 0.20 *** | 0.22 *** | −0.12 *** | −0.12 *** |
| Cobalamin, nmol/L | 285 (221–354) | 0.07 ** | 0.09 *** | −0.15 *** | −0.14 *** |
| Methylmalonic acid, nmol/L | 170 (138–217) | −0.03 | −0.01 | 0.04 | 0.05 |
| Folate, nmol/L | 15.8 (10.8–23.3) | 0.08 ** | 0.08 ** | −0.12 *** | −0.10 *** |
| Homocysteine, µmol/L | 12 (11–16) | −0.07 * | −0.12 *** | 0.08 ** | 0.05 |
| Vitamin D3, nmol/L | 62.0 (47.0–76.8) | 0.09 ** | 0.16 *** | −0.05 | −0.03 |
| Laboratory characteristics | |||||
| Lipids | |||||
| Total cholesterol, mg/dL | 209 ± 42 | 0.71 *** | −0.06 * | 0.30 *** | −0.13 *** |
| HDL cholesterol, mg/dL | 58 (46–70) | 0.03 | 0.40 *** | −0.04 | 0.10 *** |
| Non-HDL cholesterol, mg/dL | 150 ± 40 | 0.69 *** | −0.21 *** | 0.32 *** | −0.16 *** |
| LDL cholesterol, mg/dL | 135 (108–162) | 0.62 *** | −0.09 *** | 0.25 *** | −0.15 *** |
| Triglycerides, mg/dL | 98 (74–133) | 0.46 *** | −0.51 *** | 0.29 *** | −0.18 *** |
| Total lipid, mg/dL | 309 (269–360) | 0.73 *** | −0.38 *** | 0.38 *** | −0.19 *** |
| Glucose homeostasis | |||||
| Glucose, mmol/L | 5.2 (4.8–5.7) | −0.06 * | −0.20 *** | 0.09 *** | 0.07 ** |
| HbA1C, % | 5.8 (5.6–6.0) | −0.05 * | −0.14 *** | 0.09 *** | 0.10 *** |
| Thyroid function | |||||
| TSH, mU/L | 2.4 (1.6–3.4) | 0.08 | 0.11 * | 0.03 | 0.04 |
| Free T3, pmol/L | 5.0 ± 0.6 | −0.13 * | −0.13 * | −0.11 * | −0.09 |
| Free T4, pmol/L | 16.0 ± 2.4 | −0.13 * | −0.15 ** | −0.15 ** | −0.16 ** |
| Kidney function and inflammation | |||||
| Creatinine, µmol/L | 75 (66–85) | 0.05 | −0.06 * | 0.04 | −0.02 |
| eGFR, mL/min/1.73 m2 | 91 (79–104) | −0.10 | 0.11 ** | −0.01 | 0.05 |
| Urinary albumin, mg/24 hrs | 5.2 (3.0–9.5) | −0.02 | −0.06 | −0.01 | 0.02 |
| Uric acid, mmol/L | 0.32 ± 0.07 | 0.07 | −0.21 *** | 0.15 ** | 0.05 |
| hs-CRP, mg/L | 1.5 (0.7–2.9) | 0.09 * | 0.02 | 0.10 * | 0.08 |
| Liver characteristics | |||||
| ASAT, U/L | 24 (21–28) | 0.001 | −0.03 | 0.02 | −0.02 |
| ALAT, U/L | 21 (16–26) | −0.001 | −0.15 *** | −0.01 | −0.10 * |
| Alkaline phosphatase, U/L | 65 (56–76) | −0.001 | −0.11 * | 0.02 | −0.02 |
| γ-Glutamyltransferase, U/L | 23 (18–34) | 0.08 | −0.10 * | 0.04 | −0.05 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sotomayor, C.G.; Minović, I.; Eggersdorfer, M.L.; Riphagen, I.J.; de Borst, M.H.; Dekker, L.H.; Nolte, I.M.; Frank, J.; van Zon, S.K.R.; Reijneveld, S.A.; et al. Duality of Tocopherol Isoforms and Novel Associations with Vitamins Involved in One-Carbon Metabolism: Results from an Elderly Sample of the LifeLines Cohort Study. Nutrients 2020, 12, 580. https://doi.org/10.3390/nu12020580
Sotomayor CG, Minović I, Eggersdorfer ML, Riphagen IJ, de Borst MH, Dekker LH, Nolte IM, Frank J, van Zon SKR, Reijneveld SA, et al. Duality of Tocopherol Isoforms and Novel Associations with Vitamins Involved in One-Carbon Metabolism: Results from an Elderly Sample of the LifeLines Cohort Study. Nutrients. 2020; 12(2):580. https://doi.org/10.3390/nu12020580
Chicago/Turabian StyleSotomayor, Camilo G., Isidor Minović, Manfred L. Eggersdorfer, Ineke J. Riphagen, Martin H. de Borst, Louise H. Dekker, Ilja M. Nolte, Jan Frank, Sander K.R. van Zon, Sijmen A. Reijneveld, and et al. 2020. "Duality of Tocopherol Isoforms and Novel Associations with Vitamins Involved in One-Carbon Metabolism: Results from an Elderly Sample of the LifeLines Cohort Study" Nutrients 12, no. 2: 580. https://doi.org/10.3390/nu12020580
APA StyleSotomayor, C. G., Minović, I., Eggersdorfer, M. L., Riphagen, I. J., de Borst, M. H., Dekker, L. H., Nolte, I. M., Frank, J., van Zon, S. K. R., Reijneveld, S. A., van der Molen, J. C., Vos, M. J., Kootstra-Ros, J. E., Rodrigo, R., Kema, I. P., Navis, G. J., & Bakker, S. J. L. (2020). Duality of Tocopherol Isoforms and Novel Associations with Vitamins Involved in One-Carbon Metabolism: Results from an Elderly Sample of the LifeLines Cohort Study. Nutrients, 12(2), 580. https://doi.org/10.3390/nu12020580






