Gut–Joint Axis: The Role of Physical Exercise on Gut Microbiota Modulation in Older People with Osteoarthritis
Abstract
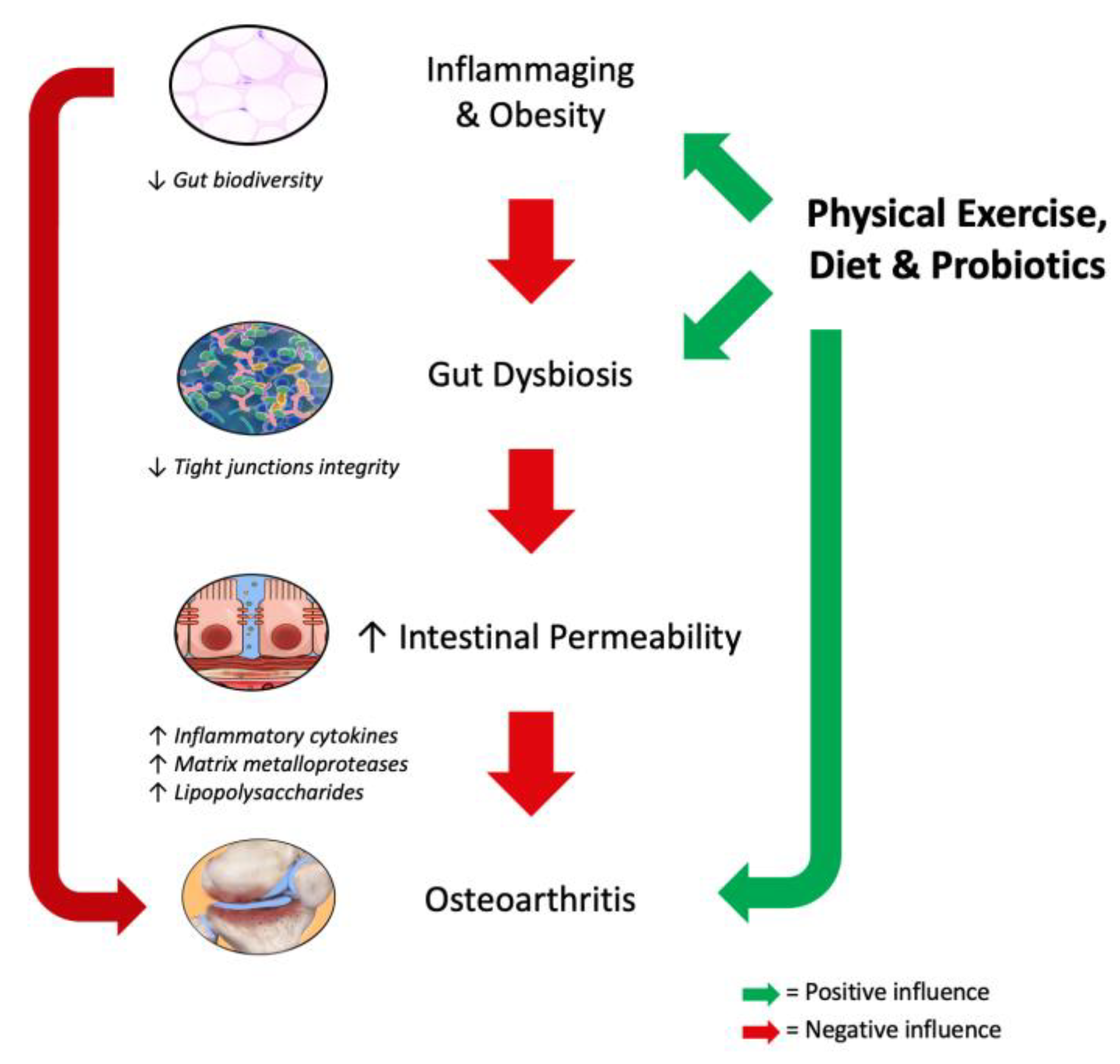
1. Introduction
2. Inflammaging
3. Obesity and Metabolic Syndrome
4. Gut Microbiota Modulation as Treatment of Osteoarthritis
5. Physical Exercise and Gut Microbiota
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef]
- McDonough, C.M.; Jette, A.M. The contribution of osteoarthritis to functional limitations and disability. Clin. Geriatr. Med. 2010, 26, 387–399. [Google Scholar] [CrossRef]
- McAlindon, T.E.; Cooper, C.; Kirwan, J.R.; Dieppe, P.A. Determinants of disability in osteoarthritis of the knee. Ann. Rheum. Dis. 1993, 52, 258–262. [Google Scholar] [CrossRef]
- Iolascon, G.; Gimigliano, F.; Moretti, A.; de Sire, A.; Migliore, A.; Brandi, M.L.; Piscitelli, P. Early osteoarthritis: How to define, diagnose, and manage. A systematic review. Eur. Geriatr. Med. 2017, 8, 383–396. [Google Scholar] [CrossRef]
- Malfait, A.M. Osteoarthritis year in review 2015: Biology. Osteoarthr. Cartil. 2016, 24, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xiao, W.; Luo, W.; Zeng, C.; Deng, Z.; Ren, W.; Wu, G.; Lei, G. Alterations of amino acid metabolism in osteoarthritis: Its implications for nutrition and health. Amino Acids 2016, 48, 907–914. [Google Scholar] [CrossRef] [PubMed]
- Biver, E.; Berenbaum, F.; Valdes, A.M.; de Carvalho, I.A.; Bindels, L.B.; Brandi, M.L.; Calder, P.C.; Castronovo, V.; Cavalier, E.; Cherubini, A.; et al. Gut microbiota and osteoarthritis management: An expert consensus of the European society for clinical and economic aspects of osteoporosis, osteoarthritis and musculoskeletal diseases (ESCEO). Ageing Res. Rev. 2019, 55, 100946. [Google Scholar] [CrossRef] [PubMed]
- Sommer, F.; Bäckhed, F. The gut microbiota-masters of host development and physiology. Nat. Rev. Microbiol. 2013, 11, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; Le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the human gut microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef]
- Kamada, N.; Seo, S.U.; Chen, G.Y.; Nunez, G. Role of the gut microbiota in immunity and inflammatory disease. Nat. Rev. Immunol. 2011, 13, 321–335. [Google Scholar] [CrossRef]
- De Sire, R.; Talocco, C.; Petito, V.; Lopetuso, L.R.; Graziani, C.; Gasbarrini, A.; Scaldaferri, F. Microbiota and inflammatory bowel disease: An update. Recenti Prog. Med. 2018, 109, 570–573. [Google Scholar] [PubMed]
- Li, Y.; Luo, W.; Deng, Z.; Lei, G. Diet-Intestinal Microbiota Axis in Osteoarthritis: A Possible Role. Mediat. Inflamm. 2016. [Google Scholar] [CrossRef] [PubMed]
- de Sire, R.; Rizzatti, G.; Ingravalle, F.; Pizzoferrato, M.; Petito, V.; Lopetuso, L.; Graziani, C.; de Sire, A.; Mentella, M.C.; Mele, M.C.; et al. Skeletal muscle-gut axis: Emerging mechanisms of sarcopenia for intestinal and extra intestinal diseases. Minerva Gastroenterol. Dietol. 2018, 64, 1351–1362. [Google Scholar] [CrossRef]
- Berthelot, J.-M.; Sellam, J.; Maugars, Y.; Berenbaum, F. Cartilage-gut-microbiome axis: A new paradigm for novel therapeutic opportunities in osteoarthritis. RMD Open 2019, 2, e001037. [Google Scholar] [CrossRef] [PubMed]
- Szychlinska, M.A.; Di Rosa, M.; Castorina, A.; Mobasheri, A.; Musumeci, G. A correlation between intestinal microbiota dysbiosis and osteoarthritis. Heliyon 2019, 1, e01134. [Google Scholar] [CrossRef] [PubMed]
- Favazzo, L.J.; Hendesi, H.; Villani, D.A.; Soniwala, S.; Dar, Q.A.; Schott, E.M.; Gill, S.R.; Zuscik, M.J. The gut microbiome-joint connection: Implications in osteoarthritis. Curr. Opin. Rheumatol. 2020, 1, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Monda, V.; Villano, I.; Messina, A.; Valenzano, A.; Esposito, T.; Moscatelli, F.; Viggiano, A.; Cibelli, G.; Chieffi, S.; Monda, M.; et al. Exercise modifies the gut microbiota with positive health effects. Oxid. Med. Cell. Longev. 2017. [Google Scholar] [CrossRef]
- Peters, H.P.; De Vries, W.R.; Vanberge-Henegouwen, G.P.; Akkermans, L.M. Potential benefits and hazards of physical activity and exercise on the gastrointestinal tract. Gut 2001, 48, 435–439. [Google Scholar] [CrossRef]
- Pizzoferrato, M.; de Sire, R.; Ingravalle, F.; Mentella, M.C.; Petito, V.; Martone, A.M.; Landi, F.; Miggiano, G.A.; Mele, M.C.; Lopetuso, L.R.; et al. Characterization of Sarcopenia in an IBD Population Attending an Italian Gastroenterology Tertiary Center. Nutrients 2019, 24, 2281. [Google Scholar] [CrossRef]
- Ticinesi, A.; Nouvenne, A.; Cerundolo, N.; Catania, P.; Prati, B.; Tana, C.; Meschi, T. Gut Microbiota, Muscle Mass and Function in Aging: A Focus on Physical Frailty and Sarcopenia. Nutrients 2019, 17, 1633. [Google Scholar] [CrossRef] [PubMed]
- Picca, A.; Ponziani, F.R.; Calvani, R.; Marini, F.; Biancolillo, A.; Coelho-Júnior, H.J.; Gervasoni, J.; Primiano, A.; Putignani, L.; Del Chierico, F.; et al. Gut Microbial, Inflammatory and Metabolic Signatures in Older People with Physical Frailty and Sarcopenia: Results from the BIOSPHERE Study. Nutrients 2019, 26, 65. [Google Scholar] [CrossRef] [PubMed]
- Attur, M.; Krasnokutsky, S.; Statnikov, A.; Samuels, J.; Li, Z.; Friese, O.; Le Graverand-Gastineau, M.P.H.; Rybak, L.; Kraus, V.B.; Jordan, J.M.; et al. Low-grade inflammation in symptomatic knee osteoarthritis: Prognostic value of inflammatory plasma lipids and peripheral blood leukocyte biomarkers. Arthritis Rheumatol. 2015, 67, 2905–2915. [Google Scholar] [CrossRef] [PubMed]
- Scanzello, C.R.; Loeser, R.F. Inflammatory activity in symptomatic knee osteoarthritis: Not all inflammation is local. Arthritis Rheumatol. 2015, 67, 2797–2800. [Google Scholar] [CrossRef] [PubMed]
- Greene, M.A.; Loeser, R.F. Aging-related inflammation in osteoarthritis. Osteoarthr. Cartil. 2015, 23, 1966–1971. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Bonafè, M.; Valensin, S.; Olivieri, F.; De Luca, M.; Ottaviani, E.; De Benedictis, G. Inflamm-aging: An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef]
- Davidson, E.B.; Van Caam, A.P.; Vitters, E.L.; Bennink, M.B.; Thijssen, E.; Van Den Berg, W.B.; Koenders, M.I.; Van Lent, P.L.; Van De Loo, F.A.; Van Der Kraan, P.M. TGF-â is a potent inducer of nerve growth factor in articular cartilage via the ALK5-Smad2/3 pathway. Potential role in OA related pain? Osteoarthr. Cartil. 2015, 23, 478–486. [Google Scholar] [CrossRef]
- Davidson, E.N.; Remst, D.F.; Vitters, E.L.; van Beuningen, H.M.; Blom, A.B.; Goumans, M.J.; van den Berg, W.B.; van der Kraan, P.M. Increase in ALK1/ALK5 ratio as a cause for elevated MMP-13 expression in osteoarthritis in humans and mice. J. Immunol. 2009, 182, 7937–7945. [Google Scholar] [CrossRef]
- Finnson, K.W.; Parker, W.L.; ten Dijke, P.; Thorikay, M.; Philip, A. ALK1 opposes ALK5/Smad3 signaling and expression of extracellular matrix components in human chondrocytes. J. Bone Miner. Res. 2008, 23, 896–906. [Google Scholar] [CrossRef]
- van der Kraan, P.; Matta, C.; Mobasheri, A. Age-Related Alterations in Signaling Pathways in Articular Chondrocytes: Implications for the Pathogenesis and Progression of Osteoarthritis—A Mini-Review. Gerontology 2017, 63, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Livshits, G.; Zhai, G.; Hart, D.J.; Kato, B.S.; Wang, H.; Williams, F.M.; Spector, T.D. Interleukin-6 is a significant predictor of radiographic knee osteoarthritis: The Chingford study. Arthritis Rheum. 2009, 60, 2037–2045. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.; Newman, A.B. Inflammatory markers in population studies of aging. Ageing Res. Rev. 2011, 10, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A.; Ostan, R.; Candela, M.; Biagi, E.; Brigidi, P.; Capri, M.; Franceschi, C. Gut microbiota changes in the extreme decades of human life: A focus on centenarians. Cell. Mol. Life Sci. 2018, 75, 129–148. [Google Scholar] [CrossRef]
- Biagi, E.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkïla, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through ageing, and beyond: Gut microbiota and inflammatory status in seniors and centenarians. PLoS ONE 2010, 5, e10667. [Google Scholar] [CrossRef]
- Kong, F.; Hua, Y.; Zeng, B.; Ning, R.; Li, Y.; Zhao, J. Gut microbiota signatures of longevity. Curr. Biol. 2016, 26, R832–R833. [Google Scholar] [CrossRef] [PubMed]
- Graziani, C.; Talocco, C.; De Sire, R.; Petito, V.; Lopetuso, L.R.; Gervasoni, J.; Persichilli, S.; Franceschi, F.; Ojetti, V.; Gasbarrini, A.; et al. Intestinal permeability in physiological and pathological conditions: Major determinants and assessment modalities. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 795–810. [Google Scholar] [PubMed]
- Collins, K.H.; Paul, H.A.; Reimer, R.A.; Seerattan, R.A.; Hart, D.A.; Herzog, W. Relationship between inflammation, the gut microbiota, and metabolic osteoarthritis development: Studies in a rat model. Osteoarthr. Cartil. 2015, 11, 1989–1998. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.Y.; Stabler, T.; Pei, F.X.; Kraus, V.B. Both systemic and local lipopolysaccharide (LPS) burden are associated with knee OA severity and inflammation. Osteoarthr. Cartil. 2016, 10, 1769–1775. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Garagnani, P.; Parini, P.; Giuliani, C.; Santoro, A. Inflammaging: A new immune-metabolic viewpoint for age-related diseases. Nat. Rev. Endocrinol. 2018, 14, 576–590. [Google Scholar] [CrossRef] [PubMed]
- Milani, C.; Ferrario, C.; Turroni, F.; Duranti, S.; Mangifesta, M.; van Sinderen, D.; Ventura, M. The human gut microbiota and its interactive connections to diet. J. Hum. Nutr. Diet. 2016, 29, 539–546. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Raoul, P.; Cintoni, M.; Franceschi, F.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms 2019, 1, 14. [Google Scholar] [CrossRef] [PubMed]
- Gentile, C.L.; Weir, T.L. The gut microbiota at the intersection of diet and human health. Science 2018, 16, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.A.; Velazquez, K.T.; Herbert, K.M. Influence of high-fat diet on gut microbiota: A driving force for chronic disease risk. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Lauretani, F.; Milani, C.; Nouvenne, A.; Tana, C.; Del Rio, D.; Maggio, M.; Ventura, M.; Meschi, T. Aging Gut Microbiota at the Cross-Road between Nutrition, Physical Frailty, and Sarcopenia: Is There a Gut-Muscle Axis? Nutrients 2017, 30, 1303. [Google Scholar] [CrossRef] [PubMed]
- Lucas, S.; Omata, Y.; Hofmann, J.; Böttcher, M.; Iljazovic, A.; Sarter, K.; Albrecht, O.; Schulz, O.; Krishnacoumar, B.; Krönke, G.; et al. Short-chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat. Commun. 2018, 4, 55. [Google Scholar] [CrossRef] [PubMed]
- Iolascon, G.; Gimigliano, R.; Bianco, M.; De Sire, A.; Moretti, A.; Giusti, A.; Malavolta, N.; Migliaccio, S.; Migliore, A.; Napoli, N.; et al. Are Dietary Supplements and Nutraceuticals Effective for Musculoskeletal Health and Cognitive Function? A Scoping Review. J. Nutr. Health Aging 2017, 21, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Calvani, R.; Tosato, M.; Martone, A.M.; Ortolani, E.; Savera, G.; D’Angelo, E.; Sisto, A.; Marzetti, E. Protein Intake and Muscle Health in Old Age: From Biological Plausibility to Clinical Evidence. Nutrients 2016, 14, 295. [Google Scholar] [CrossRef]
- Courtney-Martin, G.; Ball, R.O.; Pencharz, P.B.; Elango, R. Protein Requirements during Aging. Nutrients 2016, 11, 492. [Google Scholar] [CrossRef]
- Iolascon, G.; Moretti, A.; de Sire, A.; Calafiore, D.; Gimigliano, F. Effectiveness of Calcifediol in Improving Muscle Function in Post-Menopausal Women: A Prospective Cohort Study. Adv. Ther. 2017, 34, 744–752. [Google Scholar] [CrossRef]
- Tessier, A.J.; Chevalier, S. An Update on Protein, Leucine, Omega-3 Fatty Acids, and Vitamin D in the Prevention and Treatment of Sarcopenia and Functional Decline. Nutrients 2018, 16, 1099. [Google Scholar] [CrossRef]
- Iolascon, G.; Moretti, A.; de Sire, A.; Liguori, S.; Toro, G.; Gimigliano, F. Pharmacological therapy of sarcopenia: Past, present and future. Clin. Cases Miner. Bone Metab. 2018, 15, 411–419. [Google Scholar]
- de Sire, A.; Invernizzi, M.; Lippi, L.; Curci, C.; Cisari, C.; Iolascon, G. Nutritional supplementation in hip fracture sarcopenic patients: A narrative review. Clin. Cases Miner. Bone Metab. 2019, 16, 27–30. [Google Scholar]
- Invernizzi, M.; de Sire, A.; D’Andrea, F.; Carrera, D.; Renò, F.; Migliaccio, S.; Iolascon, G.; Cisari, C. Effects of essential amino acid supplementation and rehabilitation on functioning in hip fracture patients: A pilot randomized controlled trial. Aging Clin. Exp. Res. 2019, 10, 1517–1524. [Google Scholar] [CrossRef] [PubMed]
- de Sire, A.; Baricich, A.; Renò, F.; Cisari, C.; Fusco, N.; Invernizzi, M. Myostatin as a potential biomarker to monitor sarcopenia in hip fracture patients undergoing a multidisciplinary rehabilitation and nutritional treatment: A preliminary study. Aging Clin. Exp. Res. 2019, 14, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Veronese, N.; Koyanagi, A.; Stubbs, B.; Cooper, C.; Guglielmi, G.; Rizzoli, R.; Punzi, L.; Rogoli, D.; Caruso, M.G.; Rotolo, O.; et al. Mediterranean diet and knee osteoarthritis outcomes: A longitudinal cohort study. Clin. Nutr. 2019, 13, 2735–2739. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.; Zimmet, P.; Shaw, J. The metabolic syndrome—A new worldwide definition. Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef]
- Gao, X.; Zhang, M.; Xue, J.; Huang, J.; Zhuang, R.; Zhou, X.; Zhang, H.; Fu, Q.; Hao, Y. Body Mass Index Differences in the Gut Microbiota Are Gender Specific. Front. Microbiol. 2018, 9, 1250. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Hu, Y.; Bruner, D.W. Composition of gut microbiota and its association with body mass index and lifestyle factors in a cohort of 7-18 years old children from the American Gut Project. Pediatr. Obes. 2019, 4, e12480. [Google Scholar] [CrossRef]
- Guss, J.D.; Ziemian, S.N.; Luna, M.; Sandoval, T.N.; Holyoak, D.T.; Guisado, G.G.; Roubert, S.; Callahan, R.L.; Brito, I.L.; van der Meulen, M.C.; et al. The effects of metabolic syndrome, obesity, and the gut microbiome on load-induced osteoarthritis. Osteoarthr. Cartil. 2019, 1, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Ding, W.; Wang, H.L.; Dai, L.L.; Zong, W.H.; Wang, Y.Z.; Bi, J.; Han, W.; Dong, G.J. Gut microbiota and obesity-associated osteoarthritis. Osteoarthr. Cartil. 2019, 9, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Boulangé, C.L.; Neves, A.L.; Chilloux, J.; Nicholson, J.K.; Dumas, M.E. Impact of the gut microbiota on inflammation, obesity, and metabolic disease. Genome Med. 2016, 20, 42. [Google Scholar] [CrossRef]
- Visser, M.; Pahor, M.; Taaffe, D.R.; Goodpaster, B.H.; Simonsick, E.M.; Newman, A.B.; Nevitt, M.; Harris, T.B. Relationship of interleukin-6 and tumor necrosis factor-alpha with muscle mass and muscle strength in elderly men and women: The Health ABC Study. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 5, M326–M332. [Google Scholar] [CrossRef] [PubMed]
- Gimigliano, F.; Moretti, A.; de Sire, A.; Calafiore, D.; Iolascon, G. The combination of vitamin D deficiency and overweight affects muscle mass and function in older post-menopausal women. Aging Clin. Exp. Res. 2018, 6, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Satokari, R. Modulation of Gut Microbiota for Health by Current and Next-Generation Probiotics. Nutrients 2019, 8, 1921. [Google Scholar] [CrossRef] [PubMed]
- Korotkyi, O.H.; Vovk, A.A.; Dranitsina, A.S.; Falalyeyeva, T.M.; Dvorshchenko, K.O.; Fagoonee, S.; Ostapchenko, L.I. The influence of probiotic diet and chondroitin sulfate administration on Ptgs2, Tgfb1 and Col2a1 expression in rat knee cartilage during monoiodoacetate-induced osteoarthritis. Minerva Med. 2019, 110, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Rios, J.L. Protective effect of prebiotic and exercise intervention on knee health in a rat model of diet-induced obesity. Cartilage 2019, 9, 3893. [Google Scholar] [CrossRef] [PubMed]
- Mazor, M.; Best, T.M.; Cesaro, A.; Lespessailles, E.; Toumi, H. Osteoarthritis biomarker responses and cartilage adaptation to exercise: A review of animal and human models. Scand. J. Med. Sci. Sports 2019, 29, 1072–1082. [Google Scholar] [CrossRef]
- Richmond, J.; Hunter, D.; Irrgang, J.; Jones, M.H.; Levy, B.; Marx, R.; Snyder-Mackler, L.; Watters, W.C.; Haralson, R.H.; Turkelson, C.M.; et al. American Academy of Orthopaedic Surgeons. Treatment of osteoarthritis of the knee (nonarthroplasty). J. Am. Acad. Orthop. Surg. 2009, 17, 591–600. [Google Scholar] [CrossRef]
- Hochberg, M.C.; Altman, R.D.; April, K.T.; Benkhalti, M.; Guyatt, G.; McGowan, J.; Towheed, T.; Welch, V.; Wells, G.; Tugwell, P. American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res. 2012, 64, 465–474. [Google Scholar] [CrossRef] [PubMed]
- McAlindon, T.E.; Bannuru, R.; Sullivan, M.C.; Arden, N.K.; Berenbaum, F.; Bierma-Zeinstra, S.M.; Hawker, G.A.; Henrotin, Y.; Hunter, D.J.; Kawaguchi, H.; et al. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr. Cartil. 2014, 22, 363–388. [Google Scholar] [CrossRef] [PubMed]
- Cerda, B.; Perez, M.; Perez-Santiago, J.D.; Tornero-Aguilera, J.F.; Gonzalez-Soltero, R.; Larrosa, M. Gut Microbiota Modification: Another Piece in the Puzzle of the Benefits of Physical Exercise in Health? Front. Physiol. 2016, 7, 51. [Google Scholar] [CrossRef] [PubMed]
- Evans, C.C.; LePard, K.J.; Kwak, J.W.; Stancukas, M.C.; Laskowski, S.; Dougherty, J.; Moulton, L.; Glawe, A.; Wang, Y.; Leone, V.; et al. Exercise prevents weight gain and alters the gut microbiota in a mouse model of high fat diet-induced obesity. PLoS ONE 2014, 26, e92193. [Google Scholar] [CrossRef] [PubMed]
- Morita, E.; Yokoyama, H.; Imai, D.; Takeda, R.; Ota, A.; Kawai, E.; Hisada, T.; Emoto, M.; Suzuki, Y.; Okazaki, K. Aerobic Exercise Training with Brisk Walking Increases Intestinal Bacteroides in Healthy Elderly Women. Nutrients 2019, 4, 868. [Google Scholar] [CrossRef] [PubMed]
- Mach, N.; Fuster-Botella, D. Endurance exercise and gut microbiota: A review. J. Sport Health Sci. 2017, 6, 179–197. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.J.; Chiu, C.C.; Li, Y.P.; Huang, W.C.; Huan, Y.T.; Huang, C.C.; Chuang, H.L. Effect of intestinal microbiota on exercise performance in mice. J. Strength Cond. Res. 2015, 29, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.J.; Chiu, C.C.; Li, Y.P.; Huang, W.C.; Te Huang, Y.; Huang, C.C.; Chuang, H.L. Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc. Natl. Acad. Sci. USA 2008, 28, 16767–16772. [Google Scholar]
- Bermon, S.; Petriz, B.; Kajėnienė, A.; Prestes, J.; Castell, L.; Franco, O.L. The microbiota: An exercise immunology perspective. Exerc. Immunol. Rev. 2015, 21, 70–79. [Google Scholar] [PubMed]
- Campbell, S.C.; Wisniewski, P.J.; Noji, M.; McGuinness, L.R.; Häggblom, M.M.; Lightfoot, S.A.; Joseph, L.B.; Kerkhof, L.J. The Effect of Diet and Exercise on Intestinal Integrity and Microbial Diversity in Mice. PLoS ONE 2016, 8, e0150502. [Google Scholar] [CrossRef] [PubMed]
- Estaki, M.; Pither, J.; Baumeister, P.; Little, J.P.; Gill, S.K.; Ghosh, S.; Ahmadi-Vand, Z.; Marsden, K.R.; Gibson, D.L. Cardiorespiratory fitness as a predictor of intestinal microbial diversity and distinct metagenomic functions. Microbiome 2016, 8, 42. [Google Scholar] [CrossRef]
- Román, E.; Nieto, J.C.; Gely, C.; Vidal, S.; Pozuelo, M.; Poca, M.; Juárez, C.; Guarner, C.; Manichanh, C.; Soriano, G. Effect of a multistrain probiotic on cognitive function and risk of falls in patients with cirrhosis: A randomized trial. Hepatol. Commun. 2019, 3, 632–645. [Google Scholar] [CrossRef]
| Authors, Year | Study Design | Population | Intervention/Groups | Outcomes | Main Results | Conclusions |
|---|---|---|---|---|---|---|
| Bermon S, et al. 2015 [75] | Review | Animals/Humans | N/A | N/A | N/A | Exercise might modulate and help to restore the GM when altered by a high fat diet. |
| Campbell SC, et al. 2016 [76] | Randomized controlled trial | Thirty-six, 6-week old C57BL/6NTac male mice | The mice were divided into our groups: 1) lean sedentary (LS); 2) diet-induced obesity (DIO) sedentary (OS); 3) lean exercise (LX); 4) DIO exercise (OX) groups. | Duodenum/ileum tissues were fixed for immune-histo-chemistry for occludin, Ecadherin, and cyclooxygenase-2 (COX-2). Bacterial communities were assayed in fecal samples using terminal restriction fragment length polymorphism (TRFLP) analysis and pyrosequencing of 16S rRNA gene amplicons. | LS mice presented normal histologic villi while OS mice had similar villi height with more than twice the width of the LS animals. Both LX and OX mice duodenum and ileum were histologically normal. COX-2 expression was the greatest in the OS group, followed by LS, LX and OX. The TRFLP and pyrosequencing indicated that members of the Clostridiales order were predominant in all diet groups. Specific phylotypes were observed with exercise, including Faecalibacterium prausnitzi, Clostridium spp., and Allobaculum spp. | Exercise has a strong influence on gut integrity and host microbiome. |
| Cerda B, et al. 2016 [72] | Review | Animals/Humans | N/A | N/A | N/A | The release of hormones that occurs during exercise could modify the GM profile of subjects performing physical exercise at certain intensities or durations. |
| Estaki M, et al. 2016 [80] | Cross-sectional study | Healthy young adults aged 18–35 years | A continuous incremental ramp maximal exercise test on an electronically braked cycle ergometer to assess the Peak oxygen uptake (VO2 peak). According to the VO2 peak, participants were categorized into 3 groups: 1) low (LOW); 2) average (AVG); 3) high fitness (HI). | Peak oxygen uptake (VO2peak), as an indicator of physical fitness. Short-chain fatty acids (acetic, propionic, heptanoic, valeric, caproic, and butyric acid) were analyzed from the feces by gas chromatography (GC) as described previously Using high-throughput sequencing to analyze fecal microbiota | GM diversity in healthy humans is associated with aerobic fitness. Moreover, the GM profile of fit individuals appears to favor butyrate production (indicator of gut health) through increases in Clostridiales, Roseburia, Lachnospiraceae, and Erysipelotrichaceae genera. | |
| Evans CC, et al. 2014 [73] | Randomized controlled trial | Male C57BL/6 littermate mice (5 weeks) | Mice were divided into 4 groups: 1) low fat and sedentary (LF/Sed); 2) low fat and exercise (LF/Ex); 3) high fat and sedentary (HF/Sed); 4) high fat and exercise (HF/Ex). LF/Ex and HF/Ex cages were equipped with a wheel and odometer to record Ex. | Fecal samples were collected at baseline, 6 weeks and 12 weeks and used for bacterial DNA isolation. DNA was subjected both to quantitative PCR using primers specific to the 16S rRNA encoding genes for Bacteroidetes and Firmicutes and to sequencing for lower taxonomic identification using the Illumina MiSeq platform | HF diet resulted in significantly greater body weight and adiposity as well as decreased glucose tolerance that were prevented by voluntary exercise (p < 0.05). Sequencing demonstrated exercise-induced changes in the percentage of major bacterial phyla at 12 weeks. A correlation between total Ex distance and the ΔCt Bacteroidetes/ΔCt Firmicutes ratio from qPCR demonstrated a significant inverse correlation (r2 = 0.35, p = 0.043). | The exercise induces a unique shift in the GM that is different from dietary effects. The Bacteroidetes phylum increased while it decreased firmicutes in a manner that was proportional to the distance run in mice fed with high-fat diet. GM changes might play a role in exercise prevention of the high-fat diet-induced obesity. |
| Hsu YJ, et al. 2015 [76] | Prospective study | Male mice aged 12 weeks | The mice were divided into 3 groups: 1) specific pathogen-free (SPF); 2) germ-free (GF) 3) Bacteroides fragilis (BF) gnotobiotic mice Swimming was performed in plastic containers. | Endurance Swimming, weight of liver, muscle, brown adipose, and epididymal fat pads. | Endurance swimming time was longer for SPF and BF than GF mice, and the weight of liver, muscle, brown adipose, and epididymal fat pads was higher for SPF and BF than GF mice. | GM status could be crucial for exercise performance and its potential action linked with the antioxidant enzyme system in athletes. |
| Mach N & Fuster-Botella D. 2017 [75] | Review | Animals/Humans | N/A | N/A | N/A | Exercise might modulate and help to restore the GM when altered by a high fat diet. |
| Monda V, et al., 2017 [17] | Review | Animals/Humans | N/A | N/A | N/A | Exercise seems to be an environmental factor that might determine changes in the GM composition with possible benefits for the host. Physical exercise is able to enrich the microflora diversity and to improve the Bacteroidetes–Firmicutes ratio, that might contribute to reduce obesity-associated pathologies, and gastrointestinal disorders |
| Morita E, et al. 2019 [74] | Prospective study | Thirty-two sedentary healthy elderly women | The subjects were allocated into two groups receiving different exercise interventions, trunk muscle training or aerobic exercise training. | The GM composition in fecal samples was determined before and after the training period. Clinical outcome measures were: a) daily physical activity, using an accelerometer; b) trunk muscle strength, by the modified Kraus–Weber test; c) cardiorespiratory fitness, through 6-min walk test (6MWT). | Exercise intervention modified microbiota composition and improved 6MWT. | Aerobic exercise targeting an increase of the time spent in brisk walking could increase intestinal Bacteroides correlated with an improved cardiorespiratory fitness in a cohort of healthy elderly women. |
| Picca A, et al. 2020 [21] | Cross-sectional study | Thirty-five participants community-dwellers aged more than 70 years. | The presence of PF&S was established according to physical frailty, based on: a) Short Physical Performance Battery between 3 and 9; b) low appendicular muscle mass; c) absence of mobility disability. They were divided into 2 groups: 18 older adults with physical frailty and sarcopenia (PF & S), mean aged 75.5 ± 3.9 years, and 17 non-PF & S controls, mean aged 73.9 ± 3.2 years | A multi-marker immunoassay was used to measure circulating levels of a panel of inflammatory markers. Serum concentrations of 37 amino acids and derivatives were determined by ultraperformance liquid chromatography/ mass spectrometry. Total genome DNA was extracted from fecal samples using the QIAmp Fast DNA Stool mini kit (Qiagen, Germany). | PF&S participants showed higher serum concentrations of aspartic acid, lower circulating levels of concentrations of threonine and macrophage inflammatory protein 1, increased abundance of Oscillospira and Ruminococcus microbial taxa, and decreased abundance of Barnesiellaceae and Christensenellaceae. | The Authors showed a specific relationship among GM, systemic inflammatory mediators, and metabolic alterations in older adults with PF&S. |
| Ticinesi A, et al. 2019 [20] | Review | Animals/Humans | N/A | N/A | N/A | The presence of a gut-muscle axis involved in the pathophysiology of PF & S is biologically plausible and is supported by a limited number of animal and human studies. However, the Authors conclude affirming that the causal link was still uncertain. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Sire, A.; de Sire, R.; Petito, V.; Masi, L.; Cisari, C.; Gasbarrini, A.; Scaldaferri, F.; Invernizzi, M. Gut–Joint Axis: The Role of Physical Exercise on Gut Microbiota Modulation in Older People with Osteoarthritis. Nutrients 2020, 12, 574. https://doi.org/10.3390/nu12020574
de Sire A, de Sire R, Petito V, Masi L, Cisari C, Gasbarrini A, Scaldaferri F, Invernizzi M. Gut–Joint Axis: The Role of Physical Exercise on Gut Microbiota Modulation in Older People with Osteoarthritis. Nutrients. 2020; 12(2):574. https://doi.org/10.3390/nu12020574
Chicago/Turabian Stylede Sire, Alessandro, Roberto de Sire, Valentina Petito, Letizia Masi, Carlo Cisari, Antonio Gasbarrini, Franco Scaldaferri, and Marco Invernizzi. 2020. "Gut–Joint Axis: The Role of Physical Exercise on Gut Microbiota Modulation in Older People with Osteoarthritis" Nutrients 12, no. 2: 574. https://doi.org/10.3390/nu12020574
APA Stylede Sire, A., de Sire, R., Petito, V., Masi, L., Cisari, C., Gasbarrini, A., Scaldaferri, F., & Invernizzi, M. (2020). Gut–Joint Axis: The Role of Physical Exercise on Gut Microbiota Modulation in Older People with Osteoarthritis. Nutrients, 12(2), 574. https://doi.org/10.3390/nu12020574








