Narrative Review: Nutrient Deficiencies in Adults and Children with Treated and Untreated Celiac Disease
Abstract
1. Introduction
2. Materials and Methods
3. Nutritional Deficiencies in Celiac Disease at Diagnosis and on a Gluten-Free Diet
3.1. Nutritional Deficiencies at Moment of Diagnosis in Untreated Celiac Disease
3.2. Nutritional Deficiencies While on a Gluten-Free Diet in Treated Celiac Disease
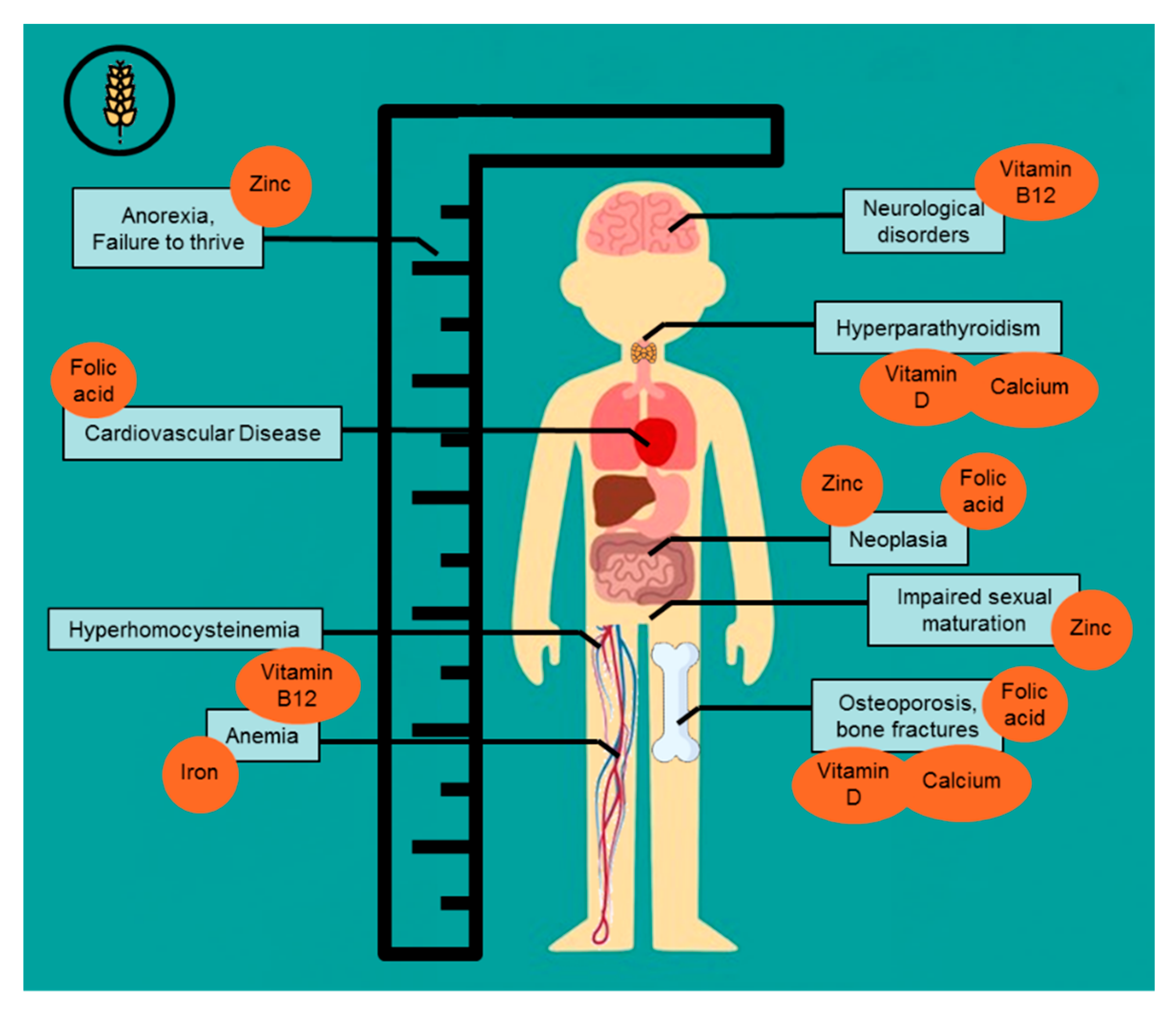
3.3. Comorbidities Potentially Related to Nutrient Deficiencies in Celiac Disease
3.4. Role of Nutrient Supplementation in Celiac Disease Management
4. Causes of Nutritional Deficiencies in Celiac Disease
4.1. Impaired Absorption DUE to Compromised Intestinal Epithelial Function
4.2. Histological Recovery on a Gluten-Free Diet
4.3. Nutrient Imbalance associated with a Gluten-Free Diet
Intake of Micronutrients
5. Discussion
6. Summary and Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Green, P.H.; Cellier, C. Celiac disease. N. Engl. J. Med. 2007, 357, 1731–1743. [Google Scholar] [CrossRef]
- Halfdanarson, T.R.; Litzow, M.R.; Murray, J.A. Hematologic manifestations of celiac disease. Blood 2007, 109, 412–421. [Google Scholar] [CrossRef]
- Corazza, G.R.; Di Stefano, M.; Maurino, E.; Bai, J.C. Bones in coeliac disease: Diagnosis and treatment. Best Pract. Res. Clin. Gastroenterol. 2005, 19, 453–465. [Google Scholar] [CrossRef]
- Bushara, K.O. Neurologic presentation of celiac disease. Gastroenterology 2005, 128, S92–S97. [Google Scholar] [CrossRef]
- Ciacci, C.; Cirillo, M.; Cavallaro, R.; Mazzacca, G. Long-term follow-up of celiac adults on gluten-free diet: Prevalence and correlates of intestinal damage. Digestion 2002, 66, 178–185. [Google Scholar] [CrossRef]
- Hutchinson, J.M.; West, N.P.; Robins, G.G.; Howdle, P.D. Long-term histological follow-up of people with coeliac disease in a UK teaching hospital. QJM Mon. J. Assoc. Physicians 2010, 103, 511–517. [Google Scholar] [CrossRef]
- Lee, S.K.; Lo, W.; Memeo, L.; Rotterdam, H.; Green, P.H. Duodenal histology in patients with celiac disease after treatment with a gluten-free diet. Gastrointest. Endosc. 2003, 57, 187–191. [Google Scholar] [CrossRef]
- Wahab, P.J.; Meijer, J.W.; Mulder, C.J. Histologic follow-up of people with celiac disease on a gluten-free diet: Slow and incomplete recovery. Am. J. Clin. Pathol. 2002, 118, 459–463. [Google Scholar] [CrossRef]
- Thompson, T. Folate, iron, and dietary fiber contents of the gluten-free diet. J. Am. Diet. Assoc. 2000, 100, 1389–1396. [Google Scholar] [CrossRef]
- Thompson, T. Thiamin, riboflavin, and niacin contents of the gluten-free diet: Is there cause for concern? J. Am. Diet. Assoc. 1999, 99, 858–862. [Google Scholar] [CrossRef]
- Miranda, J.; Lasa, A.; Bustamante, M.A.; Churruca, I.; Simon, E. Nutritional differences between a gluten-free diet and a diet containing equivalent products with gluten. Plant Foods Hum. Nutr. 2014, 69, 182–187. [Google Scholar] [CrossRef]
- Grant, M.J.; Booth, A. A typology of reviews: An analysis of 14 review types and associated methodologies. Health Inf. Libr. J. 2009, 26, 91–108. [Google Scholar] [CrossRef]
- OCEBM Levels of Evidence Working Group; Howick, J.; Chalmers, I.; Glasziou, P.; Greenhalgh, T.; Heneghan, C.; Liberati, A.; Moschetti, I.; Phillips, B.; Thornton, H.; et al. The Oxford 2011 Levels of Evidence; University of Oxford: Oxford, UK, 2011. [Google Scholar]
- Howick, J.; Phillips, B.; Ball, C.; Sackett, D.; Badenoch, D.; Straus, S.; Haynes, B.; Dawes, M. Oxford Centre for Evidence-based Medicine–Levels of Evidence (March 2009); University of Oxford: Oxford, UK, 2009. [Google Scholar]
- Tikkakoski, S.; Savilahti, E.; Kolho, K.L. Undiagnosed coeliac disease and nutritional deficiencies in adults screened in primary health care. Scand. J. Gastroenterol. 2007, 42, 60–65. [Google Scholar] [CrossRef]
- Harper, J.W.; Holleran, S.F.; Ramakrishnan, R.; Bhagat, G.; Green, P.H. Anemia in celiac disease is multifactorial in etiology. Am. J. Hematol. 2007, 82, 996–1000. [Google Scholar] [CrossRef]
- Vilppula, A.; Kaukinen, K.; Luostarinen, L.; Krekela, I.; Patrikainen, H.; Valve, R.; Luostarinen, M.; Laurila, K.; Maki, M.; Collin, P. Clinical benefit of gluten-free diet in screen-detected older celiac disease patients. BMC Gastroenterol. 2011, 11, 136. [Google Scholar] [CrossRef]
- Wierdsma, N.J.; van Bokhorst-de van der Schueren, M.A.; Berkenpas, M.; Mulder, C.J.; van Bodegraven, A.A. Vitamin and mineral deficiencies are highly prevalent in newly diagnosed celiac disease patients. Nutrients 2013, 5, 3975–3992. [Google Scholar] [CrossRef]
- Zanini, B.; Caselani, F.; Magni, A.; Turini, D.; Ferraresi, A.; Lanzarotto, F.; Villanacci, V.; Carabellese, N.; Ricci, C.; Lanzini, A. Celiac disease with mild enteropathy is not mild disease. Clin. Gastroenterol. Hepatol. 2013, 11, 253–258. [Google Scholar] [CrossRef]
- Schosler, L.; Christensen, L.A.; Hvas, C.L. Symptoms and findings in adult-onset celiac disease in a historical Danish patient cohort. Scand. J. Gastroenterol. 2016, 51, 288–294. [Google Scholar] [CrossRef]
- Sansotta, N.; Amirikian, K.; Guandalini, S.; Jericho, H. Celiac Disease Symptom Resolution: Effectiveness of the Gluten-free Diet. J. Pediatric Gastroenterol. Nutr. 2018, 66, 48–52. [Google Scholar] [CrossRef]
- Berry, N.; Basha, J.; Varma, N.; Varma, S.; Prasad, K.K.; Vaiphei, K.; Dhaka, N.; Sinha, S.K.; Kochhar, R. Anemia in celiac disease is multifactorial in etiology: A prospective study from India. JGH Open 2018, 2, 196–200. [Google Scholar] [CrossRef]
- Haapalahti, M.; Kulmala, P.; Karttunen, T.J.; Paajanen, L.; Laurila, K.; Maki, M.; Mykkanen, H.; Kokkonen, J. Nutritional status in adolescents and young adults with screen-detected celiac disease. J. Pediatric Gastroenterol. Nutr. 2005, 40, 566–570. [Google Scholar] [CrossRef]
- Kemppainen, T.A.; Kosma, V.M.; Janatuinen, E.K.; Julkunen, R.J.; Pikkarainen, P.H.; Uusitupa, M.I. Nutritional status of newly diagnosed celiac disease patients before and after the institution of a celiac disease diet--association with the grade of mucosal villous atrophy. Am. J. Clin. Nutr. 1998, 67, 482–487. [Google Scholar] [CrossRef]
- Lerner, A.; Shapira, Y.; Agmon-Levin, N.; Pacht, A.; Ben-Ami Shor, D.; Lopez, H.M.; Sanchez-Castanon, M.; Shoenfeld, Y. The clinical significance of 25OH-Vitamin D status in celiac disease. Clin. Rev. Allergy Immunol. 2012, 42, 322–330. [Google Scholar] [CrossRef]
- Chakravarthi, S.D.; Jain, K.; Kochhar, R.; Bhadada, S.K.; Khandelwal, N.; Bhansali, A.; Dutta, U.; Nain, C.K.; Singh, K. Prevalence and predictors of abnormal bone mineral metabolism in recently diagnosed adult celiac patients. Indian J. Gastroenterol. 2012, 31, 165–170. [Google Scholar] [CrossRef]
- Garcia-Manzanares, A.; Tenias, J.M.; Lucendo, A.J. Bone mineral density directly correlates with duodenal Marsh stage in newly diagnosed adult celiac patients. Scand. J. Gastroenterol. 2012, 47, 927–936. [Google Scholar] [CrossRef]
- Posthumus, L.; Al-Toma, A. Duodenal histopathology and laboratory deficiencies related to bone metabolism in coeliac disease. Eur. J. Gastroenterol. Hepatol. 2017, 29, 897–903. [Google Scholar] [CrossRef]
- Kemppainen, T.; Kroger, H.; Janatuinen, E.; Arnala, I.; Lamberg-Allardt, C.; Karkkainen, M.; Kosma, V.M.; Julkunen, R.; Jurvelin, J.; Alhava, E.; et al. Bone recovery after a gluten-free diet: A 5-year follow-up study. Bone 1999, 25, 355–360. [Google Scholar] [CrossRef]
- McFarlane, X.A.; Bhalla, A.K.; Reeves, D.E.; Morgan, L.M.; Robertson, D.A. Osteoporosis in treated adult coeliac disease. Gut 1995, 36, 710–714. [Google Scholar] [CrossRef]
- Fernandez, A.; Gonzalez, L.; de-la-Fuente, J. Coeliac disease: Clinical features in adult populations. Rev. Esp. Enferm. Dig. 2010, 102, 466–471. [Google Scholar] [CrossRef]
- Saibeni, S.; Lecchi, A.; Meucci, G.; Cattaneo, M.; Tagliabue, L.; Rondonotti, E.; Formenti, S.; De Franchis, R.; Vecchi, M. Prevalence of hyperhomocysteinemia in adult gluten-sensitive enteropathy at diagnosis: Role of B12, folate, and genetics. Clin. Gastroenterol. Hepatol. 2005, 3, 574–580. [Google Scholar] [CrossRef]
- Hallert, C.; Grant, C.; Grehn, S.; Granno, C.; Hulten, S.; Midhagen, G.; Strom, M.; Svensson, H.; Valdimarsson, T. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment. Pharmacol. Ther. 2002, 16, 1333–1339. [Google Scholar] [CrossRef]
- Crofton, R.W.; Glover, S.C.; Ewen, S.W.; Aggett, P.J.; Mowat, N.A.; Mills, C.F. Zinc absorption in celiac disease and dermatitis herpetiformis: A test of small intestinal function. Am. J. Clin. Nutr. 1983, 38, 706–712. [Google Scholar] [CrossRef]
- Boyd, S.; Collins, B.J.; Bell, P.M.; Love, A.H. Clinical presentation of coeliac disease in adult gastroenterological practice. Ulst. Med. J. 1985, 54, 140–147. [Google Scholar]
- Bode, S.; Gudmand-Hoyer, E. Symptoms and haematologic features in consecutive adult coeliac patients. Scand. J. Gastroenterol. 1996, 31, 54–60. [Google Scholar] [CrossRef]
- Kuloglu, Z.; Kirsaclioglu, C.T.; Kansu, A.; Ensari, A.; Girgin, N. Celiac disease: Presentation of 109 children. Yonsei Med. J. 2009, 50, 617–623. [Google Scholar] [CrossRef]
- Wessels, M.M.S.; van, V., II; Vriezinga, S.L.; Putter, H.; Rings, E.H.; Mearin, M.L. Complementary Serologic Investigations in Children with Celiac Disease Is Unnecessary during Follow-Up. J. Pediatrics 2016, 169, 55–60. [Google Scholar] [CrossRef]
- Deora, V.; Aylward, N.; Sokoro, A.; El-Matary, W. Serum Vitamins and Minerals at Diagnosis and Follow-up in Children with Celiac Disease. J. Pediatric Gastroenterol. Nutr. 2017, 65, 185–189. [Google Scholar] [CrossRef]
- Zanchi, C.; Di Leo, G.; Ronfani, L.; Martelossi, S.; Not, T.; Ventura, A. Bone metabolism in celiac disease. J. Pediatrics 2008, 153, 262–265. [Google Scholar] [CrossRef]
- Mager, D.R.; Qiao, J.; Turner, J. Vitamin D and K status influences bone mineral density and bone accrual in children and adolescents with celiac disease. Eur. J. Clin. Nutr. 2012, 66, 488–495. [Google Scholar] [CrossRef]
- Tokgoz, Y.; Terlemez, S.; Karul, A. Fat soluble vitamin levels in children with newly diagnosed celiac disease, a case control study. BMC Pediatr. 2018, 18, 130. [Google Scholar] [CrossRef]
- Rea, F.; Polito, C.; Marotta, A.; Di Toro, A.; Iovene, A.; Collini, R.; Rea, L.; Sessa, G. Restoration of body composition in celiac children after one year of gluten-free diet. J. Pediatric Gastroenterol. Nutr. 1996, 23, 408–412. [Google Scholar] [CrossRef]
- Kavak, U.S.; Yuce, A.; Kocak, N.; Demir, H.; Saltik, I.N.; Gurakan, F.; Ozen, H. Bone mineral density in children with untreated and treated celiac disease. J. Pediatric Gastroenterol. Nutr. 2003, 37, 434–436. [Google Scholar] [CrossRef]
- Tau, C.; Mautalen, C.; De Rosa, S.; Roca, A.; Valenzuela, X. Bone mineral density in children with celiac disease. Effect of a Gluten-free diet. Eur. J. Clin. Nutr. 2006, 60, 358–363. [Google Scholar] [CrossRef]
- Volkan, B.; Fettah, A.; Islek, A.; Kara, S.S.; Kurt, N.; Cayir, A. Bone mineral density and vitamin K status in children with celiac disease: Is there a relation? Turk. J. Gastroenterol. 2018, 29, 215–220. [Google Scholar] [CrossRef]
- Rawal, P.; Thapa, B.R.; Prasad, R.; Prasad, K.K.; Nain, C.K.; Singh, K. Zinc supplementation to patients with celiac disease—Is it required? J. Trop. Pediatrics 2010, 56, 391–397. [Google Scholar] [CrossRef]
- Rujner, J.; Socha, J.; Syczewska, M.; Wojtasik, A.; Kunachowicz, H.; Stolarczyk, A. Magnesium status in children and adolescents with coeliac disease without malabsorption symptoms. Clin. Nutr. 2004, 23, 1074–1079. [Google Scholar] [CrossRef]
- Dickey, W.; Ward, M.; Whittle, C.R.; Kelly, M.T.; Pentieva, K.; Horigan, G.; Patton, S.; McNulty, H. Homocysteine and related B-vitamin status in coeliac disease: Effects of gluten exclusion and histological recovery. Scand. J. Gastroenterol. 2008, 43, 682–688. [Google Scholar] [CrossRef]
- Altuntas, B.; Filik, B.; Ensari, A.; Zorlu, P.; Tezic, T. Can zinc deficiency be used as a marker for the diagnosis of celiac disease in Turkish children with short stature? Pediatrics Int. 2000, 42, 682–684. [Google Scholar] [CrossRef]
- Sategna-Guidetti, C.; Grosso, S.B.; Grosso, S.; Mengozzi, G.; Aimo, G.; Zaccaria, T.; Di Stefano, M.; Isaia, G.C. The effects of 1-year gluten withdrawal on bone mass, bone metabolism and nutritional status in newly-diagnosed adult coeliac disease patients. Aliment. Pharmacol. Ther. 2000, 14, 35–43. [Google Scholar] [CrossRef]
- Wild, D.; Robins, G.G.; Burley, V.J.; Howdle, P.D. Evidence of high sugar intake, and low fibre and mineral intake, in the gluten-free diet. Aliment. Pharmacol. Ther. 2010, 32, 573–581. [Google Scholar] [CrossRef]
- Keaveny, A.P.; Freaney, R.; McKenna, M.J.; Masterson, J.; O’Donoghue, D.P. Bone remodeling indices and secondary hyperparathyroidism in celiac disease. Am. J. Gastroenterol. 1996, 91, 1226–1231. [Google Scholar]
- Murray, J.A.; Rubio-Tapia, A.; Van Dyke, C.T.; Brogan, D.L.; Knipschield, M.A.; Lahr, B.; Rumalla, A.; Zinsmeister, A.R.; Gostout, C.J. Mucosal atrophy in celiac disease: Extent of involvement, correlation with clinical presentation, and response to treatment. Clin. Gastroenterol. Hepatol. 2008, 6, 186–193. [Google Scholar] [CrossRef]
- Ferrara, M.; Coppola, L.; Coppola, A.; Capozzi, L. Iron deficiency in childhood and adolescence: Retrospective review. Hematology 2006, 11, 183–186. [Google Scholar] [CrossRef]
- Capristo, E.; Addolorato, G.; Mingrone, G.; De Gaetano, A.; Greco, A.V.; Tataranni, P.A.; Gasbarrini, G. Changes in body composition, substrate oxidation, and resting metabolic rate in adult celiac disease patients after a 1-y gluten-free diet treatment. Am. J. Clin. Nutr. 2000, 72, 76–81. [Google Scholar] [CrossRef]
- Annibale, B.; Severi, C.; Chistolini, A.; Antonelli, G.; Lahner, E.; Marcheggiano, A.; Iannoni, C.; Monarca, B.; Delle Fave, G. Efficacy of gluten-free diet alone on recovery from iron deficiency anemia in adult celiac patients. Am. J. Gastroenterol. 2001, 96, 132–137. [Google Scholar] [CrossRef]
- Kapur, G.; Patwari, A.K.; Narayan, S.; Anand, V.K. Iron supplementation in children with celiac disease. Indian J. Pediatrics 2003, 70, 955–958. [Google Scholar] [CrossRef]
- Popov, J.; Baldawi, M.; Mbuagbaw, L.; Gould, M.; Mileski, H.; Brill, H.; Pai, N. Iron Status in Pediatric Celiac Disease: A Retrospective Chart Review. J. Pediatric Gastroenterol. Nutr. 2017. [Google Scholar] [CrossRef]
- Hogberg, L.; Danielsson, L.; Jarleman, S.; Sundqvist, T.; Stenhammar, L. Serum zinc in small children with coeliac disease. Acta Paediatr. 2009, 98, 343–345. [Google Scholar] [CrossRef]
- Hallert, C.; Svensson, M.; Tholstrup, J.; Hultberg, B. Clinical trial: B vitamins improve health in patients with coeliac disease living on a gluten-free diet. Aliment. Pharmacol. Ther. 2009, 29, 811–816. [Google Scholar] [CrossRef]
- Hadithi, M.; Mulder, C.J.; Stam, F.; Azizi, J.; Crusius, J.B.; Pena, A.S.; Stehouwer, C.D.; Smulders, Y.M. Effect of B vitamin supplementation on plasma homocysteine levels in celiac disease. World J. Gastroenterol. WJG 2009, 15, 955–960. [Google Scholar] [CrossRef]
- Saukkonen, J.; Kaukinen, K.; Koivisto, A.M.; Maki, M.; Laurila, K.; Sievanen, H.; Collin, P.; Kurppa, K. Clinical Characteristics and the Dietary Response in Celiac Disease Patients Presenting with or without Anemia. J. Clin. Gastroenterol. 2016. [Google Scholar] [CrossRef]
- Rajalahti, T.; Repo, M.; Kivela, L.; Huhtala, H.; Maki, M.; Kaukinen, K.; Lindfors, K.; Kurppa, K. Anemia in Pediatric Celiac Disease: Association with Clinical and Histological Features and Response to Gluten-free Diet. J. Pediatric Gastroenterol. Nutr. 2017, 64, e1–e6. [Google Scholar] [CrossRef]
- Singh, P.; Arora, S.; Makharia, G.K. Presence of anemia in patients with celiac disease suggests more severe disease. Indian J. Gastroenterol. 2014, 33, 161–164. [Google Scholar] [CrossRef]
- O’Leary, F.; Samman, S. Vitamin B12 in health and disease. Nutrients 2010, 2, 299–316. [Google Scholar] [CrossRef]
- Iyer, R.; Tomar, S.K. Folate: A functional food constituent. J. Food Sci. 2009, 74, R114–R122. [Google Scholar] [CrossRef]
- Ahmad, I.; Mirza, T.; Qadeer, K.; Nazim, U.; Vaid, F.H. Vitamin B6: Deficiency diseases and methods of analysis. Pak. J. Pharm. Sci. 2013, 26, 1057–1069. [Google Scholar]
- Wu, X.Y.; Lu, L. Vitamin B6 deficiency, genome instability and cancer. Asian Pac. J. Cancer Prev. APJCP 2012, 13, 5333–5338. [Google Scholar] [CrossRef]
- Blom, H.J.; Smulders, Y. Overview of homocysteine and folate metabolism. With special references to cardiovascular disease and neural tube defects. J. Inherit. Metab. Dis. 2011, 34, 75–81. [Google Scholar] [CrossRef]
- Peterson, J.C.; Spence, J.D. Vitamins and progression of atherosclerosis in hyper-homocyst(e)inaemia. Lancet 1998, 351, 263. [Google Scholar] [CrossRef]
- Eichinger, S. Are B vitamins a risk factor for venous thromboembolism? Yes. J. Thromb. Haemost. JTH 2006, 4, 307–308. [Google Scholar] [CrossRef]
- Bergen, N.E.; Jaddoe, V.W.; Timmermans, S.; Hofman, A.; Lindemans, J.; Russcher, H.; Raat, H.; Steegers-Theunissen, R.P.; Steegers, E.A. Homocysteine and folate concentrations in early pregnancy and the risk of adverse pregnancy outcomes: The Generation R Study. BJOG 2012, 119, 739–751. [Google Scholar] [CrossRef]
- Tyagi, N.; Vacek, T.P.; Fleming, J.T.; Vacek, J.C.; Tyagi, S.C. Hyperhomocysteinemia decreases bone blood flow. Vasc. Health Risk Manag. 2011, 7, 31–35. [Google Scholar] [CrossRef][Green Version]
- Charoenngam, N.; Shirvani, A.; Holick, M.F. Vitamin D for skeletal and non-skeletal health: What we should know. J. Clin. Orthop. Trauma 2019, 10, 1082–1093. [Google Scholar] [CrossRef]
- Kurppa, K.; Collin, P.; Sievanen, H.; Huhtala, H.; Maki, M.; Kaukinen, K. Gastrointestinal symptoms, quality of life and bone mineral density in mild enteropathic coeliac disease: A prospective clinical trial. Scand. J. Gastroenterol. 2010, 45, 305–314. [Google Scholar] [CrossRef]
- Barera, G.; Beccio, S.; Proverbio, M.C.; Mora, S. Longitudinal changes in bone metabolism and bone mineral content in children with celiac disease during consumption of a gluten-free diet. Am. J. Clin. Nutr. 2004, 79, 148–154. [Google Scholar] [CrossRef]
- Kalayci, A.G.; Kansu, A.; Girgin, N.; Kucuk, O.; Aras, G. Bone mineral density and importance of a gluten-free diet in patients with celiac disease in childhood. Pediatrics 2001, 108, E89. [Google Scholar] [CrossRef]
- Mora, S.; Barera, G.; Beccio, S.; Menni, L.; Proverbio, M.C.; Bianchi, C.; Chiumello, G. A prospective, longitudinal study of the long-term effect of treatment on bone density in children with celiac disease. J. Pediatrics 2001, 139, 516–521. [Google Scholar] [CrossRef]
- Mora, S.; Barera, G.; Ricotti, A.; Weber, G.; Bianchi, C.; Chiumello, G. Reversal of low bone density with a gluten-free diet in children and adolescents with celiac disease. Am. J. Clin. Nutr. 1998, 67, 477–481. [Google Scholar] [CrossRef]
- Barera, G.; Mora, S.; Brambilla, P.; Ricotti, A.; Menni, L.; Beccio, S.; Bianchi, C. Body composition in children with celiac disease and the effects of a gluten-free diet: A prospective case-control study. Am. J. Clin. Nutr. 2000, 72, 71–75. [Google Scholar] [CrossRef]
- Mora, S.; Weber, G.; Barera, G.; Bellini, A.; Pasolini, D.; Prinster, C.; Bianchi, C.; Chiumello, G. Effect of gluten-free diet on bone mineral content in growing patients with celiac disease. Am. J. Clin. Nutr. 1993, 57, 224–228. [Google Scholar] [CrossRef]
- Jatla, M.; Zemel, B.S.; Bierly, P.; Verma, R. Bone mineral content deficits of the spine and whole body in children at time of diagnosis with celiac disease. J. Pediatric Gastroenterol. Nutr. 2009, 48, 175–180. [Google Scholar] [CrossRef]
- Corazza, G.R.; Di Sario, A.; Cecchetti, L.; Jorizzo, R.A.; Di Stefano, M.; Minguzzi, L.; Brusco, G.; Bernardi, M.; Gasbarrini, G. Influence of pattern of clinical presentation and of gluten-free diet on bone mass and metabolism in adult coeliac disease. Bone 1996, 18, 525–530. [Google Scholar] [CrossRef]
- Kotze, L.M.; Skare, T.; Vinholi, A.; Jurkonis, L.; Nisihara, R. Impact of a gluten-free diet on bone mineral density in celiac patients. Rev. Esp. Enferm. Dig. 2016, 108, 84–88. [Google Scholar] [CrossRef]
- Pazianas, M.; Butcher, G.P.; Subhani, J.M.; Finch, P.J.; Ang, L.; Collins, C.; Heaney, R.P.; Zaidi, M.; Maxwell, J.D. Calcium absorption and bone mineral density in celiacs after long term treatment with gluten-free diet and adequate calcium intake. Osteoporos. Int. 2005, 16, 56–63. [Google Scholar] [CrossRef]
- Jafri, M.R.; Nordstrom, C.W.; Murray, J.A.; Van Dyke, C.T.; Dierkhising, R.A.; Zinsmeister, A.R.; Melton, L.J., III. Long-term fracture risk in patients with celiac disease: A population-based study in Olmsted County, Minnesota. Dig. Dis. Sci. 2008, 53, 964–971. [Google Scholar] [CrossRef]
- Heikkila, K.; Pearce, J.; Maki, M.; Kaukinen, K. Celiac disease and bone fractures: A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2015, 100, 25–34. [Google Scholar] [CrossRef]
- Crofton, R.W.; Aggett, P.J.; Gvozdanovic, S.; Gvozdanovic, D.; Mowat, N.A.; Brunt, P.W. Zinc metabolism in celiac disease. Am. J. Clin. Nutr. 1990, 52, 379–382. [Google Scholar] [CrossRef]
- Coleman, J.E. Zinc enzymes. Curr. Opin. Chem. Biol. 1998, 2, 222–234. [Google Scholar] [CrossRef]
- Salgueiro, M.J.; Zubillaga, M.; Lysionek, A.; Cremaschi, G.; Goldman, C.G.; Caro, R.; De Paoli, T.; Hager, A.; Weill, R.; Boccio, J. Zinc status and immune system relationship: A review. Biol. Trace Elem. Res. 2000, 76, 193–205. [Google Scholar] [CrossRef]
- Jeejeebhoy, K. Zinc: An essential trace element for parenteral nutrition. Gastroenterology 2009, 137, S7–S12. [Google Scholar] [CrossRef]
- Hambidge, K.M.; Hambidge, C.; Jacobs, M.; Baum, J.D. Low levels of zinc in hair, anorexia, poor growth, and hypogeusia in children. Pediatric Res. 1972, 6, 868–874. [Google Scholar] [CrossRef]
- Tran, C.D.; Katsikeros, R.; Manton, N.; Krebs, N.F.; Hambidge, K.M.; Butler, R.N.; Davidson, G.P. Zinc homeostasis and gut function in children with celiac disease. Am. J. Clin. Nutr. 2011, 94, 1026–1032. [Google Scholar] [CrossRef]
- Dahele, A.; Ghosh, S. Vitamin B12 deficiency in untreated celiac disease. Am. J. Gastroenterol. 2001, 96, 745–750. [Google Scholar] [CrossRef]
- Caraceni, M.P.; Molteni, N.; Bardella, M.T.; Ortolani, S.; Nogara, A.; Bianchi, P.A. Bone and mineral metabolism in adult celiac disease. Am. J. Gastroenterol. 1988, 83, 274–277. [Google Scholar]
- Mautalen, C.; Gonzalez, D.; Mazure, R.; Vazquez, H.; Lorenzetti, M.P.; Maurino, E.; Niveloni, S.; Pedreira, S.; Smecuol, E.; Boerr, L.A.; et al. Effect of treatment on bone mass, mineral metabolism, and body composition in untreated celiac disease patients. Am. J. Gastroenterol. 1997, 92, 313–318. [Google Scholar]
- Muzzo, S.; Burrows, R.; Burgueño, M.; Ríos, G.; Bergenfreid, C.; Chavez, E.; Leiva, L. Effect of calcium and vitamin D supplementation on bone mineral density of celiac children. Nutr. Rev. 2000, 20, 1241–1247. [Google Scholar] [CrossRef]
- Kinsey, L.; Burden, S.T.; Bannerman, E. A dietary survey to determine if patients with coeliac disease are meeting current healthy eating guidelines and how their diet compares to that of the British general population. Eur. J. Clin. Nutr. 2008, 62, 1333–1342. [Google Scholar] [CrossRef]
- Mariani, P.; Viti, M.G.; Montuori, M.; La Vecchia, A.; Cipolletta, E.; Calvani, L.; Bonamico, M. The gluten-free diet: A nutritional risk factor for adolescents with celiac disease? J. Pediatric Gastroenterol. Nutr. 1998, 27, 519–523. [Google Scholar] [CrossRef]
- Shepherd, S.J.; Gibson, P.R. Nutritional inadequacies of the gluten-free diet in both recently-diagnosed and long-term patients with coeliac disease. J. Hum. Nutr. Diet. 2013, 26, 349–358. [Google Scholar] [CrossRef]
- Fry, L.; Madden, A.M.; Fallaize, R. An investigation into the nutritional composition and cost of gluten-free versus regular food products in the UK. J. Hum. Nutr. Diet. 2018, 31, 108–120. [Google Scholar] [CrossRef]
- Newberry, C.; McKnight, L.; Sarav, M.; Pickett-Blakely, O. Going Gluten Free: The History and Nutritional Implications of Today’s Most Popular Diet. Curr. Gastroenterol. Rep. 2017, 19, 54. [Google Scholar] [CrossRef]
- Thompson, T.; Dennis, M.; Higgins, L.A.; Lee, A.R.; Sharrett, M.K. Gluten-free diet survey: Are Americans with coeliac disease consuming recommended amounts of fibre, iron, calcium and grain foods? J. Hum. Nutr. Diet. 2005, 18, 163–169. [Google Scholar] [CrossRef]
- Sue, A.; Dehlsen, K.; Ooi, C.Y. Paediatric Patients with Coeliac Disease on a Gluten-Free Diet: Nutritional Adequacy and Macro- and Micronutrient Imbalances. Curr. Gastroenterol. Rep. 2018, 20, 2. [Google Scholar] [CrossRef]
- Larretxi, I.; Simon, E.; Benjumea, L.; Miranda, J.; Bustamante, M.A.; Lasa, A.; Eizaguirre, F.J.; Churruca, I. Gluten-free-rendered products contribute to imbalanced diets in children and adolescents with celiac disease. Eur. J. Nutr. 2019, 58, 775–783. [Google Scholar] [CrossRef]
- Mijatov, M.A.; Micetic-Turk, D. Dietary Intake in Adult Female Coeliac Disease Patients in Slovenia: PREHRANSKI VNOS ODRASLIH BOLNIC S CELIAKIJO V SLOVENIJI. Zdr. Varst. 2016, 55, 86–93. [Google Scholar] [CrossRef][Green Version]
- Grehn, S.; Fridell, K.; Killiecreutz, M.; Hallert, C. Dietary habits of Swedish adult coeliac patients treated by a gluten-free diet for 10 years. Scand. J. Nutr. 2001, 45, 178–182. [Google Scholar] [CrossRef]
- Collins, B.J.; Bell, P.M.; Thomson, J.M.; Fee, D.B.; Wilson, E.A.; Love, A.H. Dietary history and nutritional state in treated coeliac patients. J. R. Soc. Med. 1986, 79, 206–209. [Google Scholar] [CrossRef][Green Version]
- Pham-Short, A.; Donaghue, K.C.; Ambler, G.; Garnett, S.; Craig, M.E. Greater postprandial glucose excursions and inadequate nutrient intake in youth with type 1 diabetes and celiac disease. Sci. Rep. 2017, 7, 45286. [Google Scholar] [CrossRef]
- Valente, F.X.; Campos Tdo, N.; Moraes, L.F.; Hermsdorff, H.H.; Cardoso Lde, M.; Pinheiro-Sant’Ana, H.M.; Gilberti, F.A.; Peluzio Mdo, C. B vitamins related to homocysteine metabolism in adults celiac disease patients: A cross-sectional study. Nutr. J. 2015, 14, 110. [Google Scholar] [CrossRef]
- Bottaro, G.; Cataldo, F.; Rotolo, N.; Spina, M.; Corazza, G.R. The clinical pattern of subclinical/silent celiac disease: An analysis on 1026 consecutive cases. Am. J. Gastroenterol. 1999, 94, 691–696. [Google Scholar] [CrossRef]
- Ohlund, K.; Olsson, C.; Hernell, O.; Ohlund, I. Dietary shortcomings in children on a gluten-free diet. J. Hum. Nutr. Diet. 2010, 23, 294–300. [Google Scholar] [CrossRef]
- Prasad, K.K.; Thapa, B.R.; Nain, C.K.; Sharma, A.K.; Singh, K. Brush border enzyme activities in relation to histological lesion in pediatric celiac disease. J. Gastroenterol. Hepatol. 2008, 23, e348–e352. [Google Scholar] [CrossRef]
- Jameson, S. Coeliac disease, insulin-like growth factor, bone mineral density, and zinc. Scand. J. Gastroenterol. 2000, 35, 894–896. [Google Scholar] [CrossRef]
- Arikan, C.; Zihni, C.; Cakir, M.; Alkanat, M.; Aydogdu, S. Morphometric analysis of small-bowel mucosa in Turkish children with celiac disease and relationship with the clinical presentation and laboratory findings. Dig. Dis. Sci. 2007, 52, 2133–2139. [Google Scholar] [CrossRef]
- Andrews, N.C.; Schmidt, P.J. Iron homeostasis. Annu. Rev. Physiol. 2007, 69, 69–85. [Google Scholar] [CrossRef]
- Wang, J.; Pantopoulos, K. Regulation of cellular iron metabolism. Biochem. J. 2011, 434, 365–381. [Google Scholar] [CrossRef]
- Logan, R.F. Men and postmenopausal women with iron deficiency had increased risk for gastrointestinal malignancy. ACP J. Club 2003, 138, 80. [Google Scholar] [CrossRef]
- Mant, M.J.; Bain, V.G.; Maguire, C.G.; Murland, K.; Yacyshyn, B.R. Prevalence of occult gastrointestinal bleeding in celiac disease. Clin. Gastroenterol. Hepatol. 2006, 4, 451–454. [Google Scholar] [CrossRef]
- Said, H.M. Intestinal absorption of water-soluble vitamins in health and disease. Biochem. J. 2011, 437, 357–372. [Google Scholar] [CrossRef]
- Molteni, N.; Bardella, M.T.; Vezzoli, G.; Pozzoli, E.; Bianchi, P. Intestinal calcium absorption as shown by stable strontium test in celiac disease before and after gluten-free diet. Am. J. Gastroenterol. 1995, 90, 2025–2028. [Google Scholar]
- Pistorius, L.R.; Sweidan, W.H.; Purdie, D.W.; Steel, S.A.; Howey, S.; Bennett, J.R.; Sutton, D.R. Coeliac disease and bone mineral density in adult female patients. Gut 1995, 37, 639–642. [Google Scholar] [CrossRef]
- Staun, M.; Jarnum, S. Measurement of the 10,000-molecular weight calcium-binding protein in small-intestinal biopsy specimens from patients with malabsorption syndromes. Scand. J. Gastroenterol. 1988, 23, 827–832. [Google Scholar] [CrossRef]
- Graham, L.A.; Caesar, J.J.; Burgen, A.S. Gastrointestinal absorption and excretion of Mg 28 in man. Metab. Clin. Exp. 1960, 9, 646–659. [Google Scholar]
- Musso, C.G. Magnesium metabolism in health and disease. Int. Urol. Nephrol. 2009, 41, 357–362. [Google Scholar] [CrossRef]
- Said, H.M. Recent advances in carrier-mediated intestinal absorption of water-soluble vitamins. Annu. Rev. Physiol. 2004, 66, 419–446. [Google Scholar] [CrossRef]
- Dickey, W. Low serum vitamin B12 is common in coeliac disease and is not due to autoimmune gastritis. Eur. J. Gastroenterol. Hepatol. 2002, 14, 425–427. [Google Scholar] [CrossRef]
- Haere, P.; Hoie, O.; Schulz, T.; Schonhardt, I.; Raki, M.; Lundin, K.E. Long-term mucosal recovery and healing in celiac disease is the rule-not the exception. Scand. J. Gastroenterol. 2016, 51, 1439–1446. [Google Scholar] [CrossRef]
- Biagi, F.; Vattiato, C.; Agazzi, S.; Balduzzi, D.; Schiepatti, A.; Gobbi, P.; Corazza, G.R. A second duodenal biopsy is necessary in the follow-up of adult coeliac patients. Ann. Med. 2014, 46, 430–433. [Google Scholar] [CrossRef]
- Laurikka, P.; Salmi, T.; Collin, P.; Huhtala, H.; Maki, M.; Kaukinen, K.; Kurppa, K. Gastrointestinal Symptoms in Celiac Disease Patients on a Long-Term Gluten-Free Diet. Nutrients 2016, 8, 429. [Google Scholar] [CrossRef]
- Rubio-Tapia, A.; Rahim, M.W.; See, J.A.; Lahr, B.D.; Wu, T.T.; Murray, J.A. Mucosal recovery and mortality in adults with celiac disease after treatment with a gluten-free diet. Am. J. Gastroenterol. 2010, 105, 1412–1420. [Google Scholar] [CrossRef]
- Bardella, M.T.; Velio, P.; Cesana, B.M.; Prampolini, L.; Casella, G.; Di Bella, C.; Lanzini, A.; Gambarotti, M.; Bassotti, G.; Villanacci, V. Coeliac disease: A histological follow-up study. Histopathology 2007, 50, 465–471. [Google Scholar] [CrossRef]
- Belei, O.; Dobrescu, A.; Heredea, R.; Iacob, E.R.; David, V.; Marginean, O. Histologic recovery among children with celiac disease on a gluten-free diet. A long-term follow-up single-center experience. Arch. Med. Sci. 2018, 14, 94–100. [Google Scholar] [CrossRef]
- Hopman, E.G.; le Cessie, S.; von Blomberg, B.M.; Mearin, M.L. Nutritional management of the gluten-free diet in young people with celiac disease in The Netherlands. J. Pediatric Gastroenterol. Nutr. 2006, 43, 102–108. [Google Scholar] [CrossRef]
- Alvarez-Jubete, L.; Arendt, E.K.; Gallagher, E. Nutritive value and chemical composition of pseudocereals as gluten-free ingredients. Int. J. Food Sci. Nutr. 2009, 60 (Suppl. 4), 240–257. [Google Scholar] [CrossRef]
- Lee, A.R.; Ng, D.L.; Dave, E.; Ciaccio, E.J.; Green, P.H. The effect of substituting alternative grains in the diet on the nutritional profile of the gluten-free diet. J. Hum. Nutr. Diet. 2009, 22, 359–363. [Google Scholar] [CrossRef]
- Zuccotti, G.; Fabiano, V.; Dilillo, D.; Picca, M.; Cravidi, C.; Brambilla, P. Intakes of nutrients in Italian children with celiac disease and the role of commercially available gluten-free products. J. Hum. Nutr. Diet. 2013, 26, 436–444. [Google Scholar] [CrossRef]
- Anderson, J.W.; Baird, P.; Davis, R.H., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205. [Google Scholar] [CrossRef]
- Tortora, R.; Capone, P.; De Stefano, G.; Imperatore, N.; Gerbino, N.; Donetto, S.; Monaco, V.; Caporaso, N.; Rispo, A. Metabolic syndrome in patients with coeliac disease on a gluten-free diet. Aliment. Pharmacol. Ther. 2015, 41, 352–359. [Google Scholar] [CrossRef]
- Kabbani, T.A.; Goldberg, A.; Kelly, C.P.; Pallav, K.; Tariq, S.; Peer, A.; Hansen, J.; Dennis, M.; Leffler, D.A. Body mass index and the risk of obesity in coeliac disease treated with the gluten-free diet. Aliment. Pharmacol. Ther. 2012, 35, 723–729. [Google Scholar] [CrossRef]
- Dyner, L.; Drago, S.R.; Pineiro, A.; Sanchez, H.; Gonzalez, R.; Villaamil, E.; Valencia, M.E. [Composition and potential contribution of iron, calcium and zinc of bread and pasta made with wheat and amaranth flours]. Arch. Latinoam. Nutr. 2007, 57, 69–77. [Google Scholar]
- Pourhoseingholi, M.A.; Rostami-Nejad, M.; Barzegar, F.; Rostami, K.; Volta, U.; Sadeghi, A.; Honarkar, Z.; Salehi, N.; Asadzadeh-Aghdaei, H.; Baghestani, A.R.; et al. Economic burden made celiac disease an expensive and challenging condition for Iranian patients. Gastroenterol. Hepatol. Bed Bench 2017, 10, 258–262. [Google Scholar]
- Mogul, D.; Nakamura, Y.; Seo, J.; Blauvelt, B.; Bridges, J.F. The unknown burden and cost of celiac disease in the U.S. Expert Rev. Pharm. Outcomes Res. 2017, 17, 181–188. [Google Scholar] [CrossRef]
- Ross, A.C. The 2011 report on dietary reference intakes for calcium and vitamin D. Public Health Nutr. 2011, 14, 938–939. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef]
- Bouillon, R.; Van Schoor, N.M.; Gielen, E.; Boonen, S.; Mathieu, C.; Vanderschueren, D.; Lips, P. Optimal vitamin D status: A critical analysis on the basis of evidence-based medicine. J. Clin. Endocrinol. Metab. 2013, 98, E1283–E1304. [Google Scholar] [CrossRef]
- Lowe, N.M.; Fekete, K.; Decsi, T. Methods of assessment of zinc status in humans: A systematic review. Am. J. Clin. Nutr. 2009, 89, 2040S–2051S. [Google Scholar] [CrossRef]
- Nielsen, F.H.; Johnson, L.A. Data from Controlled Metabolic Ward Studies Provide Guidance for the Determination of Status Indicators and Dietary Requirements for Magnesium. Biol. Trace Elem. Res. 2017, 177, 43–52. [Google Scholar] [CrossRef]
| Nutrient | Percentage of Untreated CD Patients with Circulating Levels below Reference Value (%) | Percentage of Treated CD Patients with Circulating Levels below Reference Value (%) Short Term Follow-up, <2 years on a GFD (Time on GFD) | Percentage of Treated CD Patients with Circulating Levels below Reference Value (%) Long Term Follow-up, >2 years on a GFD (Time on GFD) | Percentage of Individuals in the General Reference Population with Circulating Levels below Reference Value (%) |
|---|---|---|---|---|
| Adults | ||||
| Iron (iron/ferritin) | 6%–82% [15,16,17,18,19,20,21,22,23] | Serum iron: 44% Serum ferritin: 15% (1 year) [24] * | data not available | 17 % [23] |
| Vitamin D (25(OH)D) | 5%–88% [18,20,25,26,27,28] | 50% (1 year) [29] * | 7.6% (5 years) 0% (mean 4 years for men, 9 years for women) [29,30] * | 50% [25] |
| Calcium | 0%–26% [17,19,20,28,31] | 0% (1–2 years) [17] | data not available | data not available |
| Vitamin B12 | 5%–19% [16,17,18,20,22,32] | data not available | 0% (8–12 years) [33] | 7%–17% [18,23] |
| Vitamin B6 (vitamin B6/Plasma pyridoxal 5 phosphatase) | 15% [18] | data not available | 37% (8–12 years) [33] | 0% [18] |
| Folic acid (folic acid/folate) | 11%–75% [15,16,17,18,19,20,22,23,31,32] | data not available | 20% (8–40 years) [33] * | 4%–14% [18,23,32] |
| Zinc | 67% [18] | 30%; (1 year) [24] * | 20% (range 8 months–7 years) [34] * | data not available |
| Magnesium | 13%–17% [35,36] * | data not available | data not available | data not available |
| Children | ||||
| Iron (iron/ferritin) | 12%–82% [22,23,37,38,39] | Serum iron: 5%–10% Serum ferritin: 21%–27% (6 months–2 years) [38,39] | Serum iron: 4%–8% (3–5.5 years) [38] | 17% [23] |
| Vitamin D | 0%–70 % [25,38,39,40,41,42] | 0%–57% (6 months–2 years) [38,39,40,41] | 12%–25% (2–5.5 years) [38] | 4%–30% [25,40,42] |
| Calcium | 0%–41% [37,38,40,43,44,45,46] | 0% (6 months–2 years) [38,40] | 0% (3 years–5.5 years) [38] | 0% [40] |
| Vitamin B12 | 1%–14% [22,23,37,38] | 0%–1% (6 months–2 years) [38,39] | 0% (3–5.5 years) [38] | 7% [23] |
| Vitamin B6 | data not available | data not available | data not available | data not available |
| Folic acid | 14%–31% [22,23,37,38] | 0%–3% (1–2 years) [38] | 0% (3–5.5 years) [38] | 14% [23] |
| Zinc | 19%–72% [37,39,47] | 16%–18% (6–18 months) [39] | data not available | data not available |
| Magnesium | 7%–11% [40,48] | data not available | 4% (11 years; range 3–17) [48] | 0% [40] |
| Nutrient | Percentage of CD Patients with Nutrient Intake below Recommendations (%) | Percentage of Individuals in the General Reference Population with Nutrient Intake below Recommendations (%) |
|---|---|---|
| Adults | ||
| Iron | 46%–54% [52,99,101,104,107] | 14% [52,99] |
| Vitamin D | 53%–100% [99,107,108] | data not available |
| Calcium | 12%–78% [99,101,104,107,108,109,110] | 6%–29% [52,99] |
| Vitamin B12 | 10%–61% [99,107,111] | 1%–65% [99,111] |
| Vitamin B6 | 33% [111] | 17% [111] |
| Folic acid | 35%–100% [54,101,107,110,111,112] | 3%–100% [52,99,111] |
| Zinc | 11%–58% [52,101,107,108,110] | 30% [52] |
| Magnesium | 28%–50% [52,101] | 29% [52] |
| Children | ||
| Iron | 8% [113] | 43%–79% [100,110]* |
| Vitamin D | 68% [113] | data not available |
| Calcium | 8%–54% [48,113] | 86% [110]* |
| Vitamin B12 | 0% [113] | data not available |
| Vitamin B6 | 8% [113] | data not available |
| Folic acid | 80% [110] | 57% [110] * |
| Zinc | 40% [110] | 43% [110] * |
| Magnesium | 29%–76% [48,113] | data not available |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kreutz, J.M.; Adriaanse, M.P.M.; van der Ploeg, E.M.C.; Vreugdenhil, A.C.E. Narrative Review: Nutrient Deficiencies in Adults and Children with Treated and Untreated Celiac Disease. Nutrients 2020, 12, 500. https://doi.org/10.3390/nu12020500
Kreutz JM, Adriaanse MPM, van der Ploeg EMC, Vreugdenhil ACE. Narrative Review: Nutrient Deficiencies in Adults and Children with Treated and Untreated Celiac Disease. Nutrients. 2020; 12(2):500. https://doi.org/10.3390/nu12020500
Chicago/Turabian StyleKreutz, Johanna M., Marlou P. M. Adriaanse, Elisabeth M. C. van der Ploeg, and Anita C. E. Vreugdenhil. 2020. "Narrative Review: Nutrient Deficiencies in Adults and Children with Treated and Untreated Celiac Disease" Nutrients 12, no. 2: 500. https://doi.org/10.3390/nu12020500
APA StyleKreutz, J. M., Adriaanse, M. P. M., van der Ploeg, E. M. C., & Vreugdenhil, A. C. E. (2020). Narrative Review: Nutrient Deficiencies in Adults and Children with Treated and Untreated Celiac Disease. Nutrients, 12(2), 500. https://doi.org/10.3390/nu12020500




