Vitamin and Mineral Supplementation During Pregnancy on Maternal, Birth, Child Health and Development Outcomes in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
1.1. Background
1.2. Current Strategies and Interventions
1.3. Why it is Important to Do this Review
2. Materials and Methods
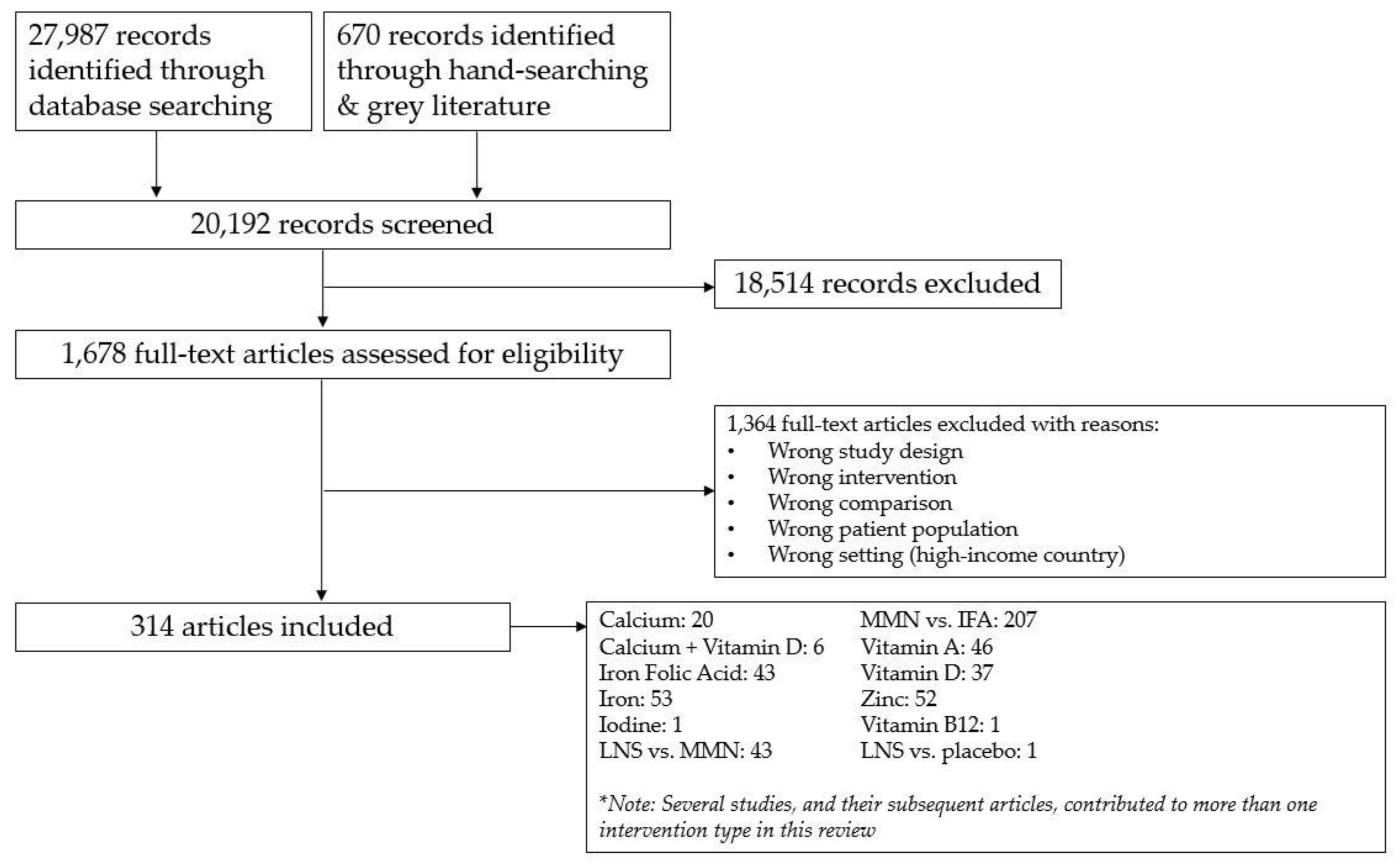
2.1. Literature Search
2.2. Study Selection and Data Abstraction
2.3. Quality Assessment
2.4. Data Synthesis
3. Results
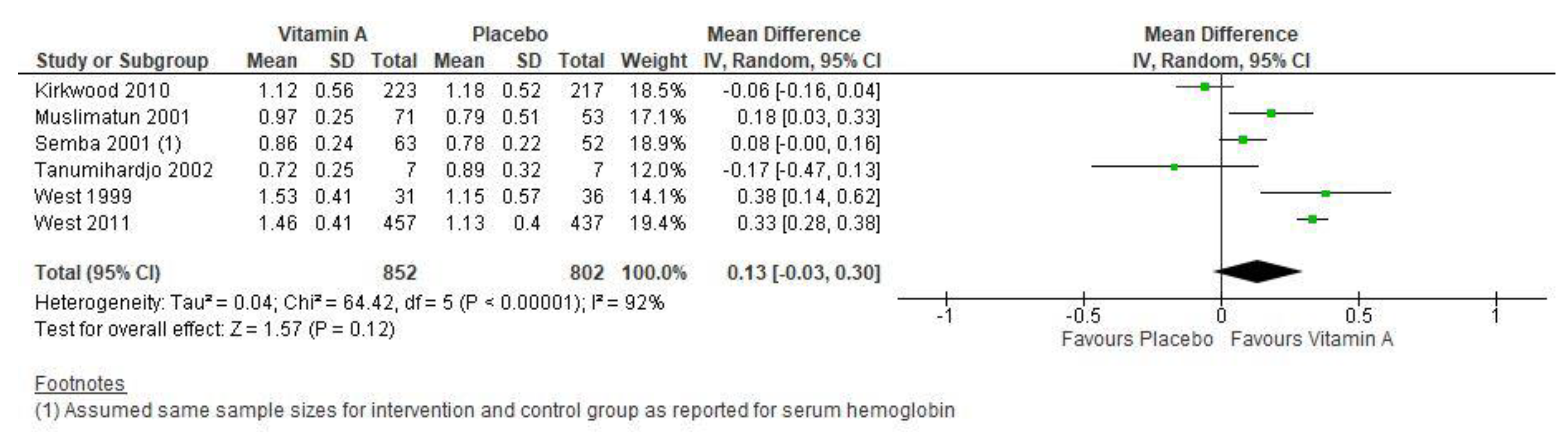
3.1. Vitamin A Supplementation Versus Placebo
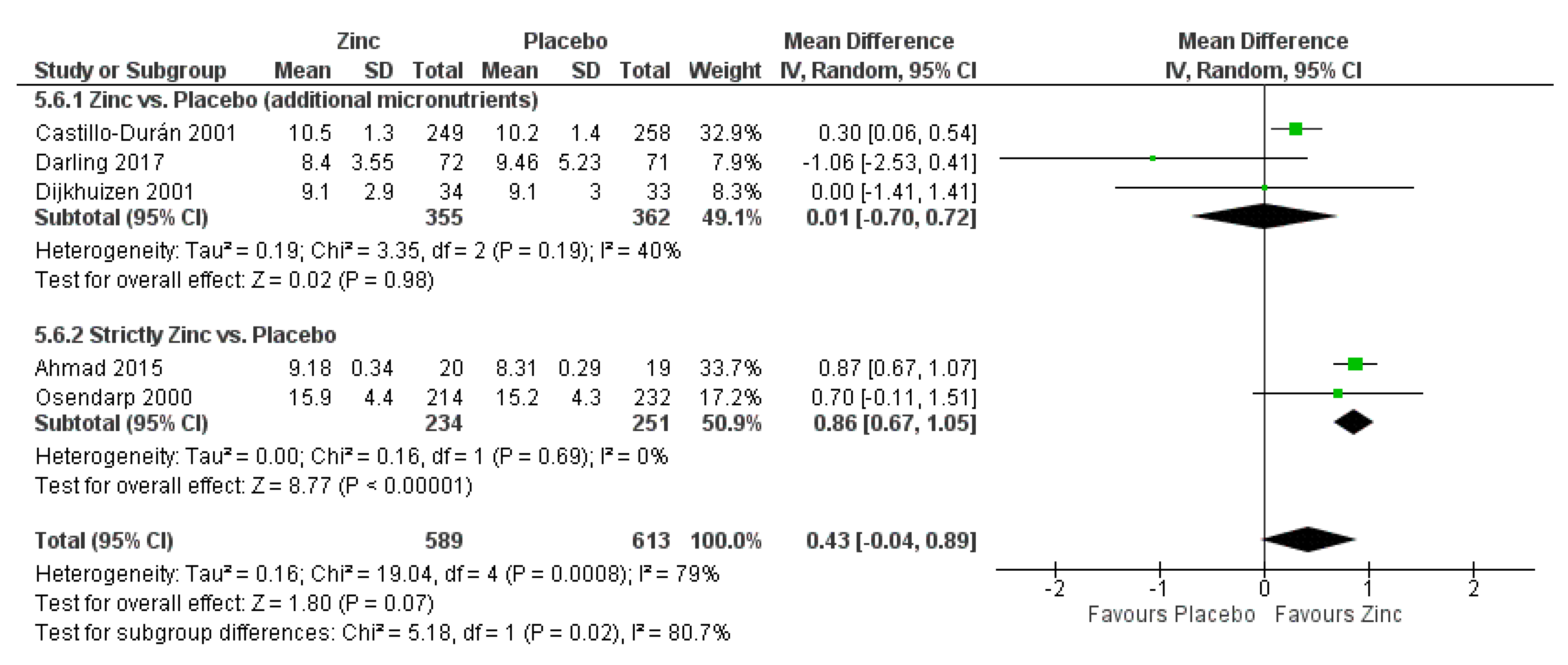
3.2. Zinc Supplementation Versus Placebo
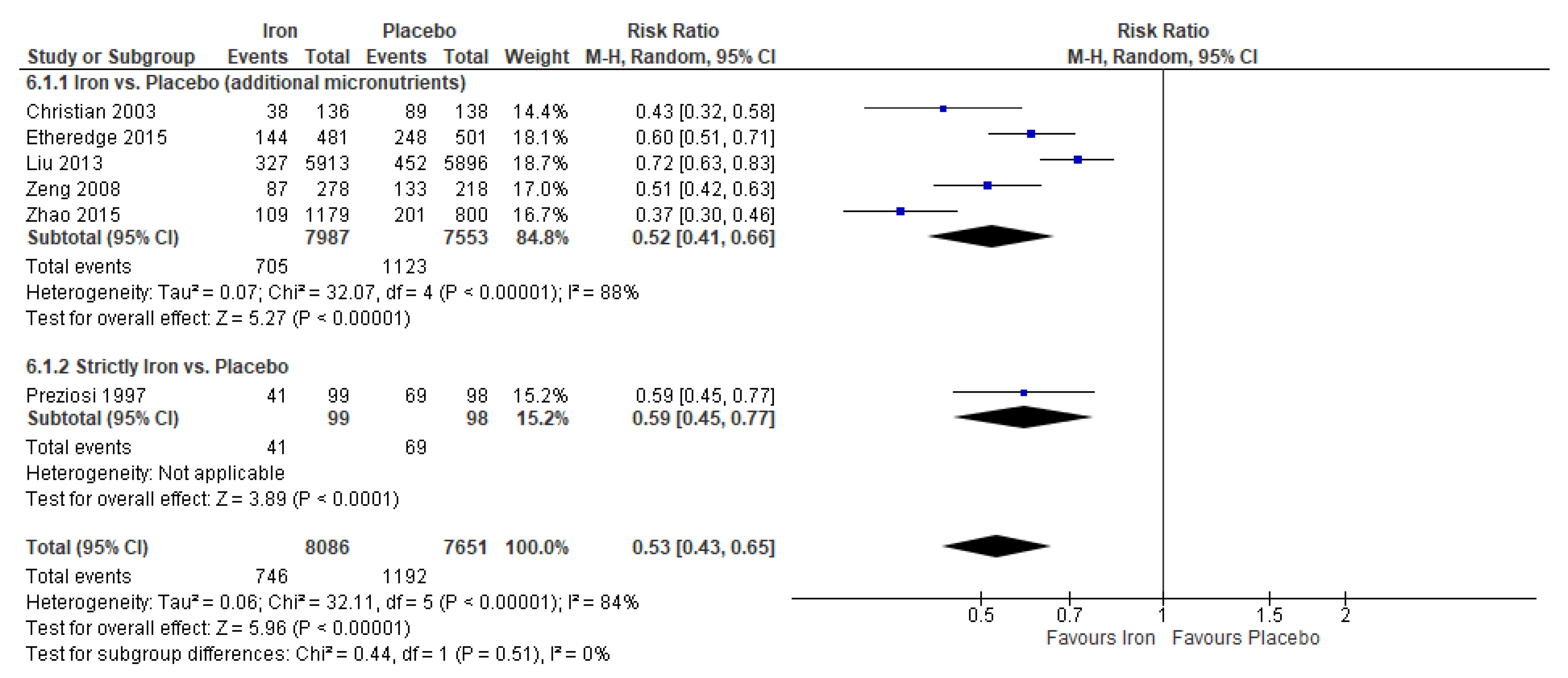
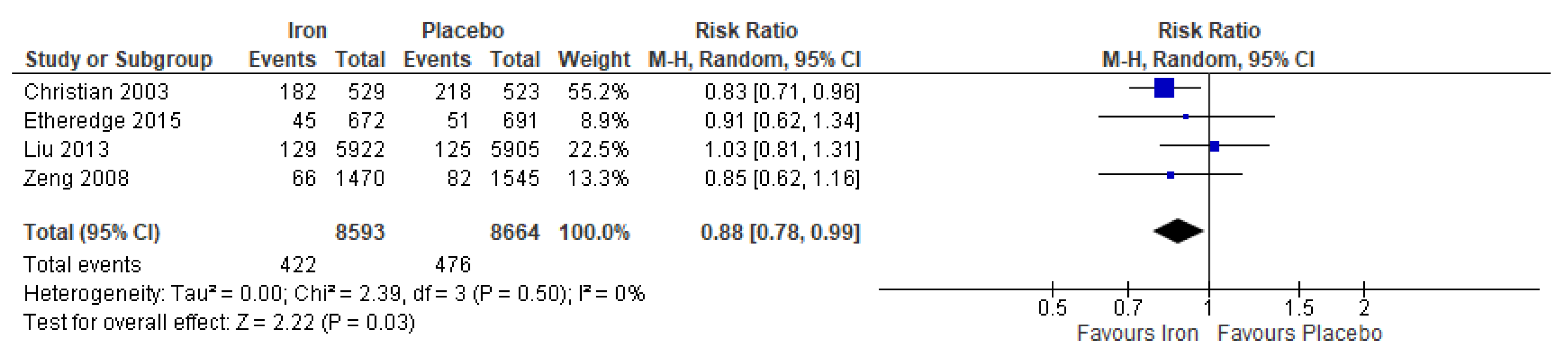
3.3. Iron Supplementation Versus Placebo
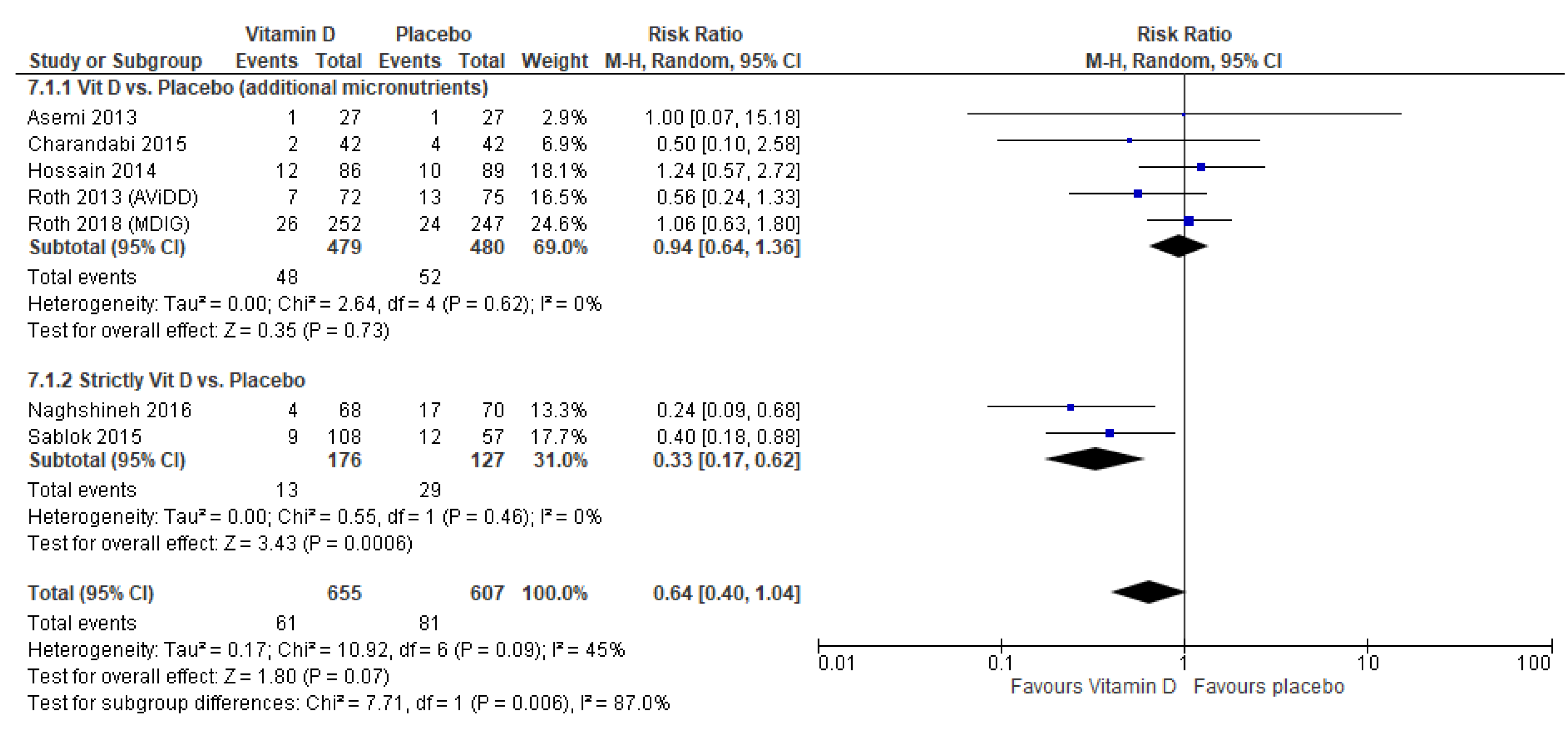
3.4. Vitamin D Supplementation Versus Placebo
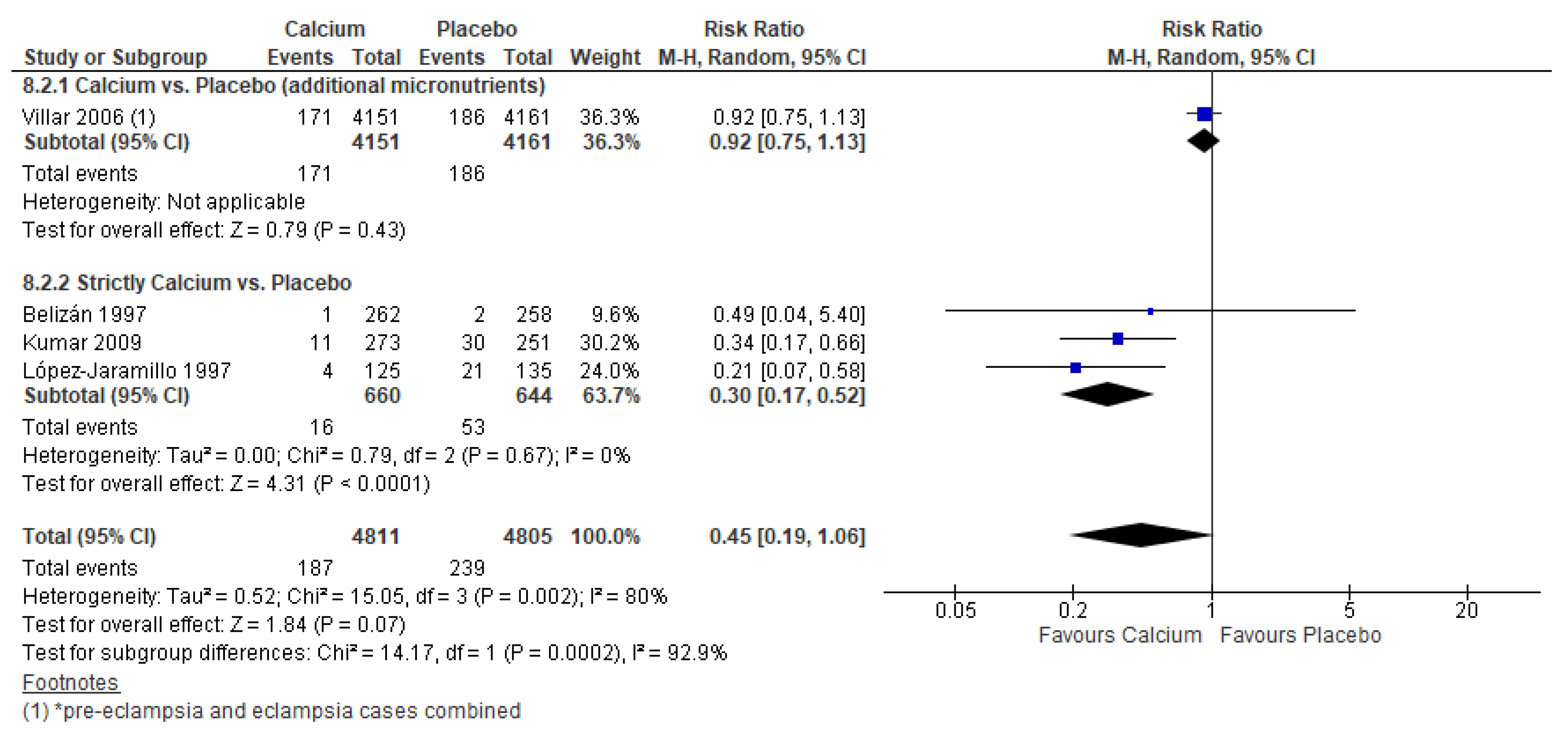
3.5. Calcium Supplementation Versus Placebo
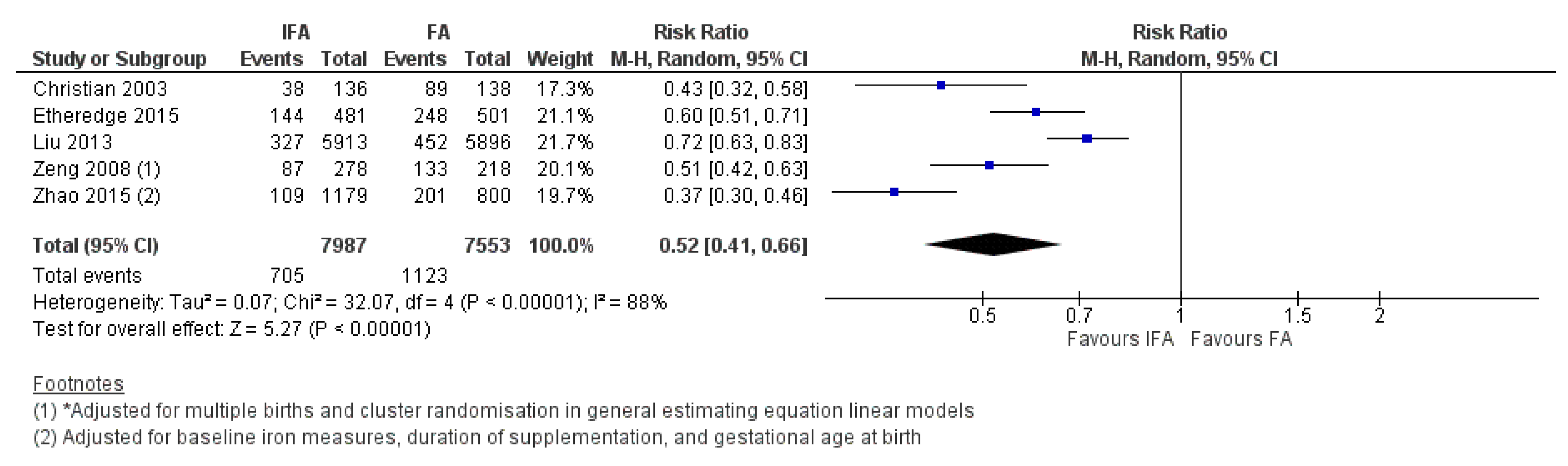
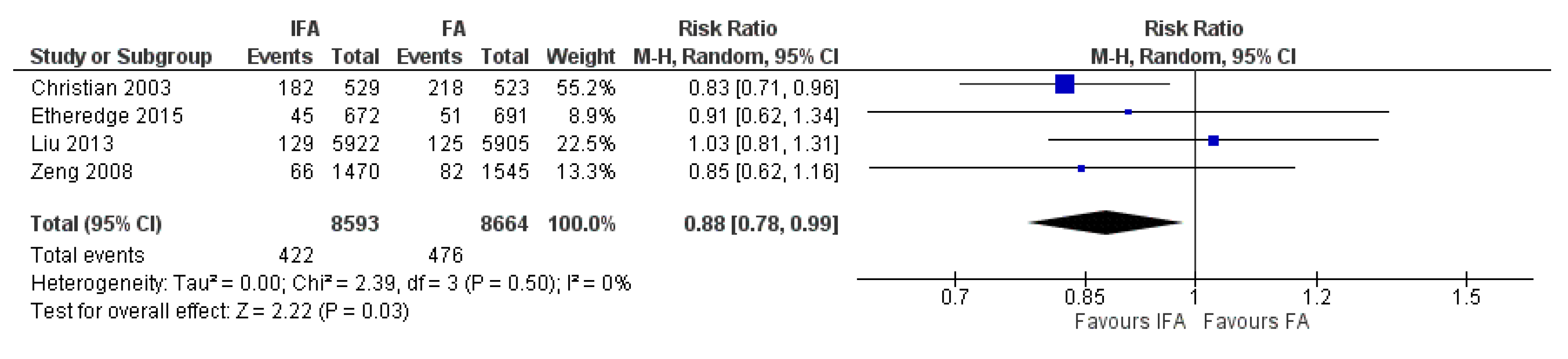
3.6. Iron and Folic Acid Versus Folic Acid Supplementation
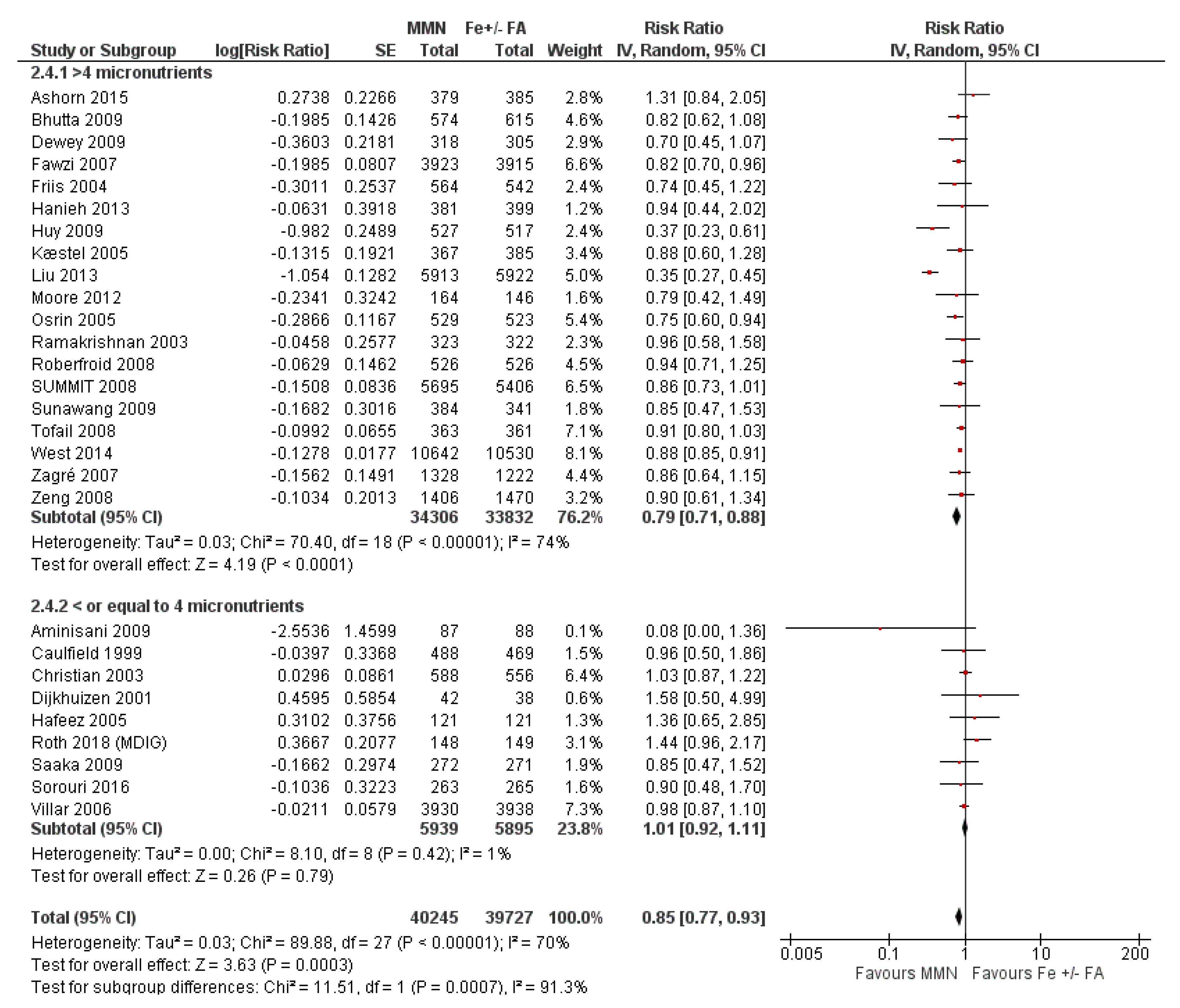
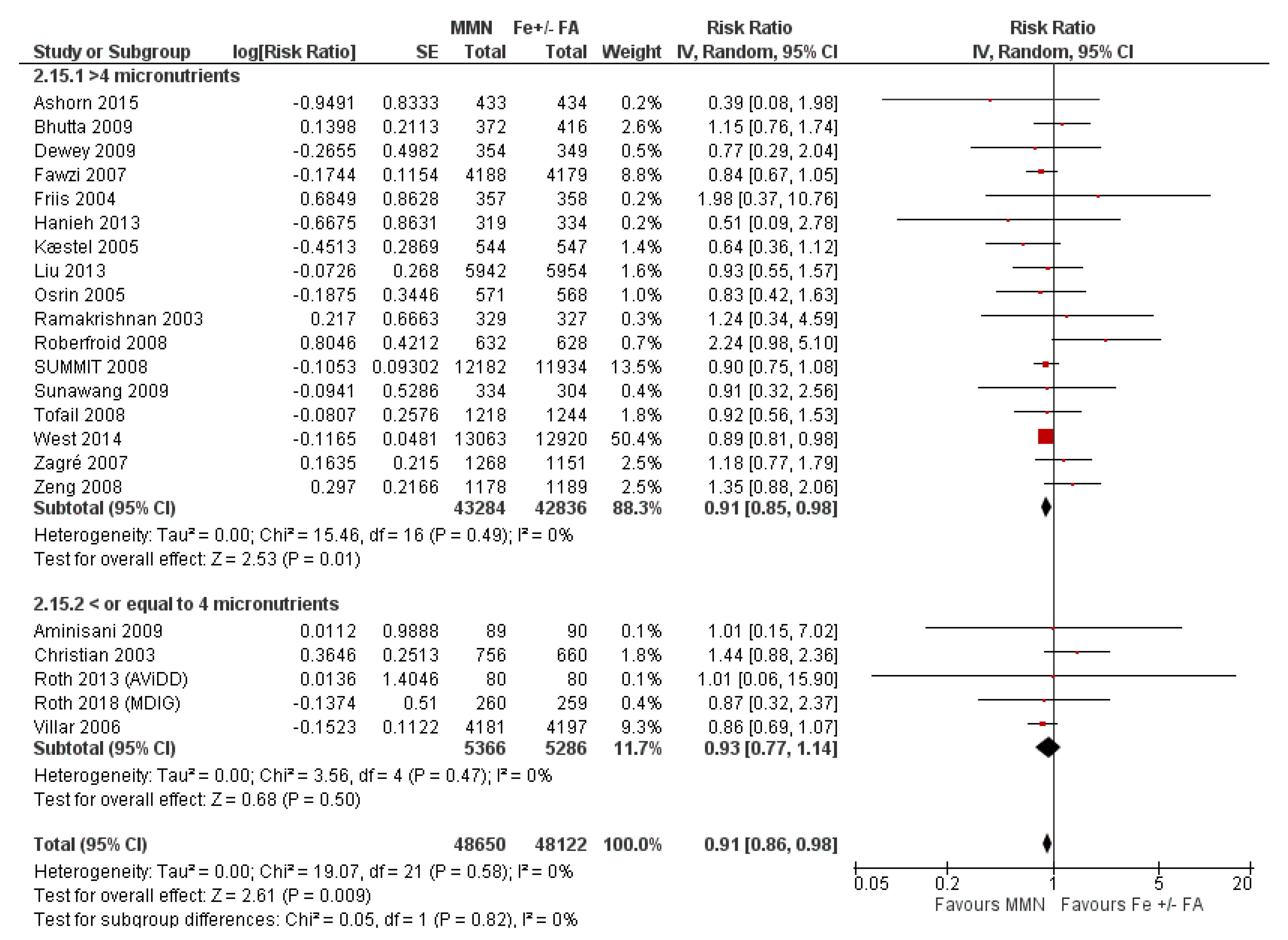
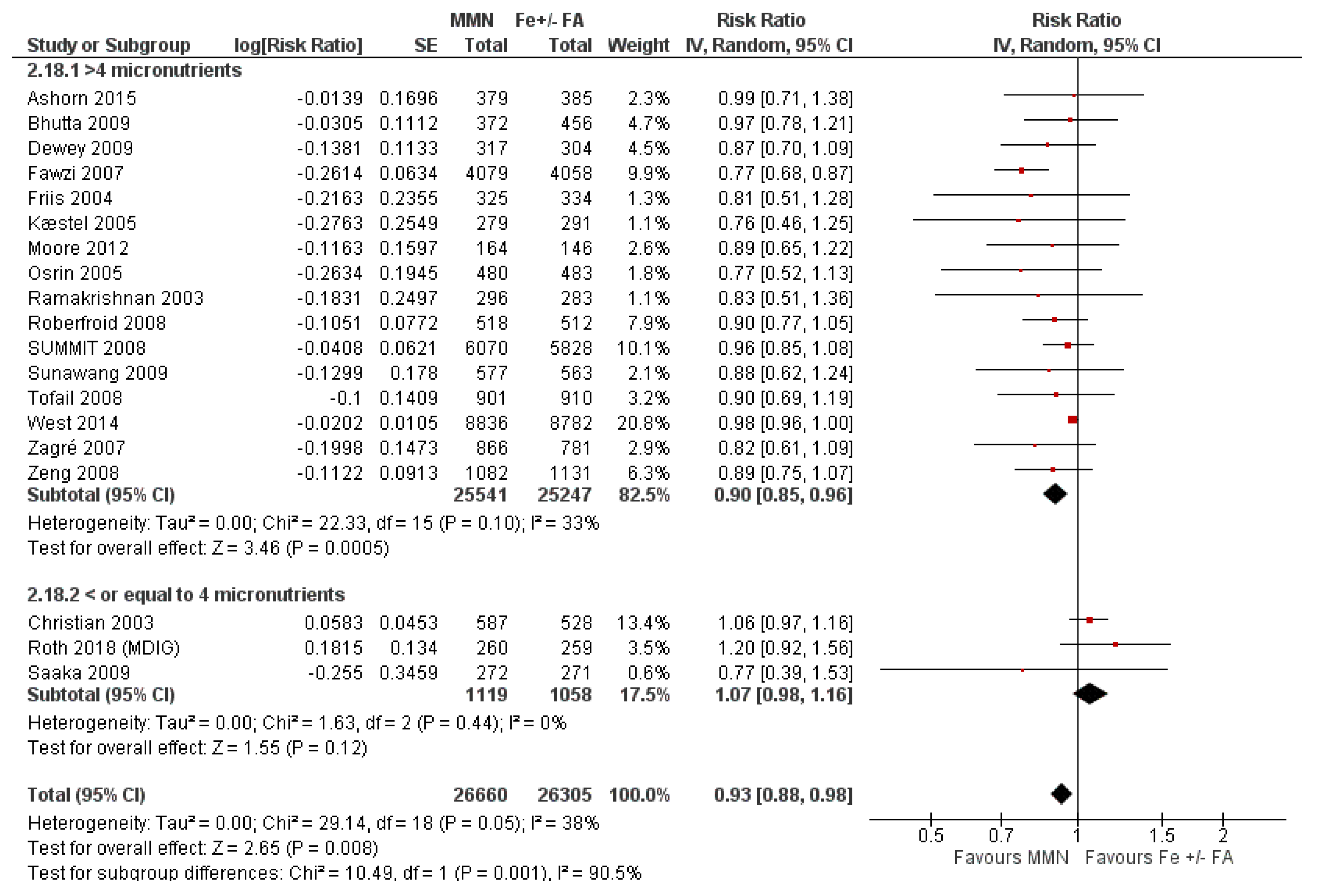
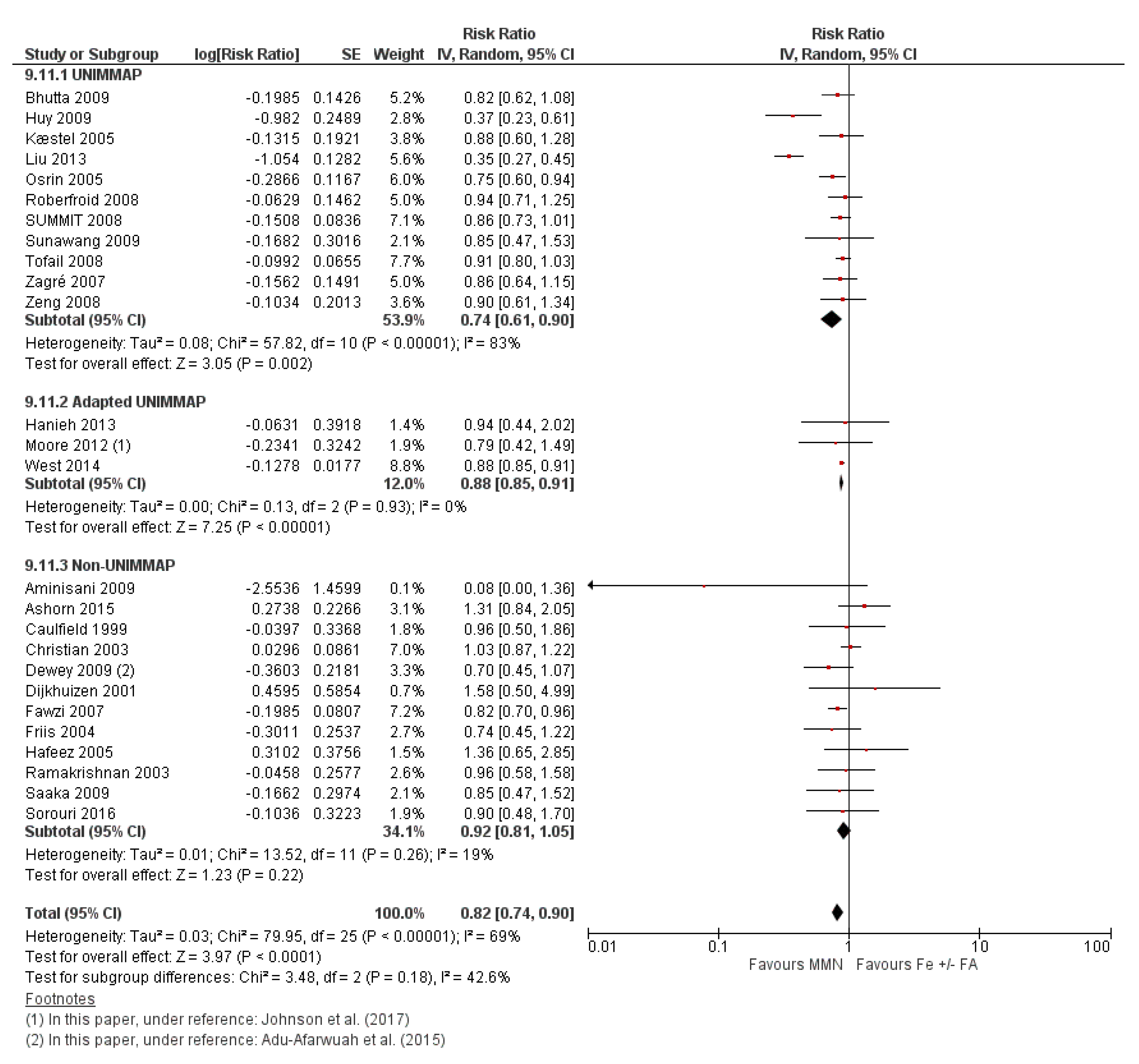
3.7. Multiple Micronutrient Supplementation Versus Iron Folic Acid Supplementation
3.7.1. Child Health and Developmental Outcomes
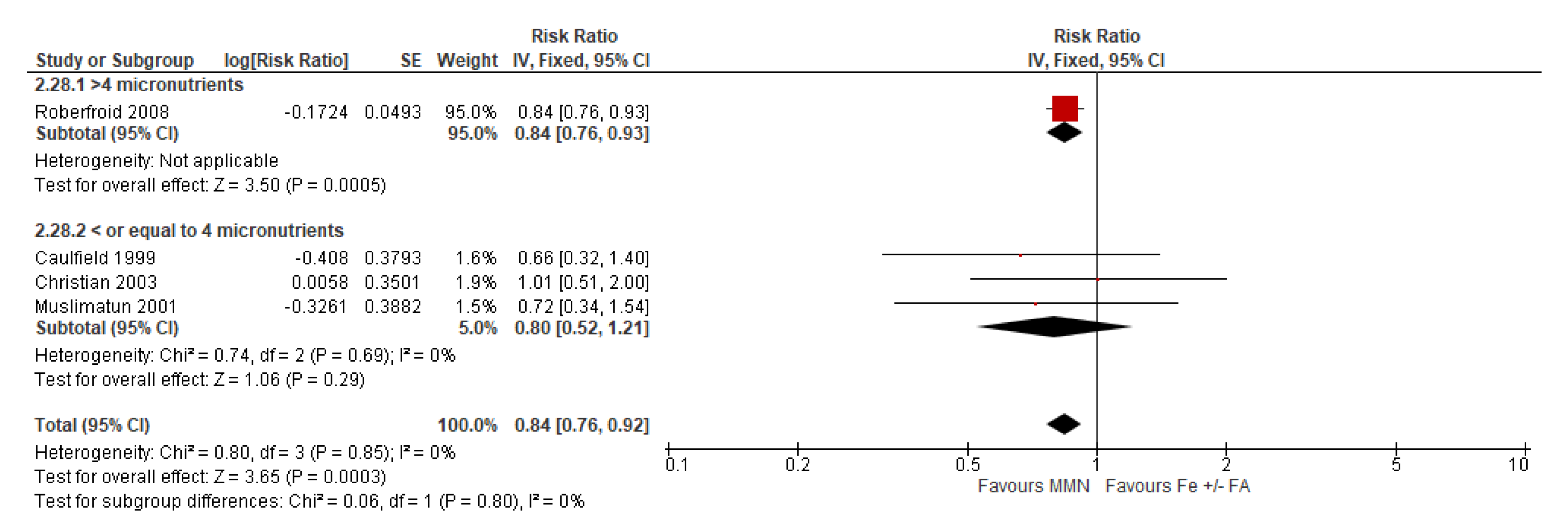
3.7.2. Subgroup Analyses for MMN vs. IFA Supplementation
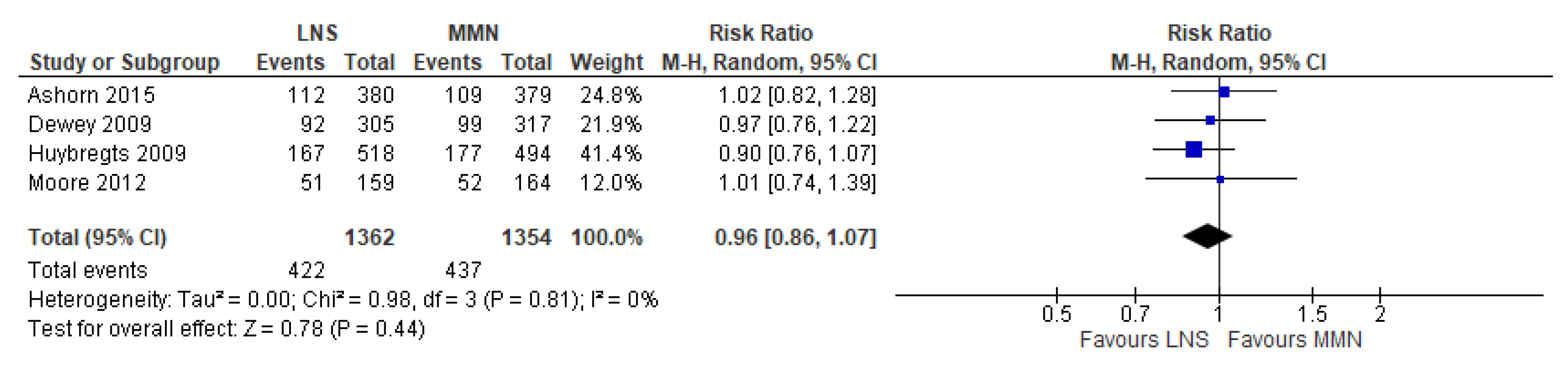
3.8. Lipid-Based Nutrient Supplementation (LNS) Versus MMN Supplementation
3.9. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; de Onis, M.; Ezzati, M.; Grantham-McGregor, S.; Katz, J.; Martorell, R.; et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Darnton-Hill, I.; Mkparu, U.C. Micronutrients in pregnancy in low- and middle-income countries. Nutrients 2015, 7, 1744–1768. [Google Scholar] [CrossRef] [PubMed]
- WHO. Vitamin and Mineral Requirements in Human Nutrition. Joint FAO/WHO Expert Consultation on Human Vitamin and Mineral Requirements; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Bailey, R.L.; West, K.P., Jr.; Black, R.E. The epidemiology of global micronutrient deficiencies. Ann. Nutr. Metab. 2015, 66 (Suppl. 2), 22–33. [Google Scholar] [CrossRef] [PubMed]
- FAO; IFAD; WHO; WFP; UNICEF. The State of Food Security and Nutrition in the World 2019. Safeguarding against Economic Slowdowns and Downturns; FAO: Rome, Italy, 2019. [Google Scholar]
- Fanzo, J.; Hawkes, C.; Udomkesmalee, E.; Afshin, A.; Allemandi, L.; Assery, O.; Baker, P.; Battersby, J.; Bhutta, Z.; Chen, K.; et al. 2018 Global Nutrition Report: Shining a light to spur action on nutrition. Dev. Initiat. 2018. [Google Scholar]
- UNICEF. The State of the World’s Children 2019. Children, Food and Nutrition: Growing Well in a Changing World; UNICEF: New York, NY, USA, 2019. [Google Scholar]
- Allen, L.H.; Peerson, J.M. Maternal Micronutrient Supplementation Study Group. Impact of multiple micronutrient versus iron-folic acid supplements on maternal anemia and micronutrient status in pregnancy. Food Nutr. Bull. 2009, 30, S527–S532. [Google Scholar] [CrossRef]
- Haider, B.A.; Olofin, I.; Wang, M.; Spiegelman, D.; Ezzati, M.; Fawzi, W.W.; Nutrition Impact Model Study Group. Anaemia, prenatal iron use, and risk of adverse pregnancy outcomes: Systematic review and meta-analysis. BMJ 2013, 346, f3443. [Google Scholar] [CrossRef]
- Allen, L.H. Biological Mechanisms That Might Underlie Iron’s Effects on Fetal Growth and Preterm Birth. J. Nutr. 2001, 131, 581S–589S. [Google Scholar] [CrossRef]
- Christian, P. Micronutrients, birth weight, and survival. Annu. Rev. Nutr. 2010, 30, 83–104. [Google Scholar] [CrossRef]
- De-Regil, L.M.; Pena-Rosas, J.P.; Fernandez-Gaxiola, A.C.; Rayco-Solon, P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Dunn, J.T. Iodine supplementation and the prevention of cretinism. Ann. N. Y. Acad. Sci. 1993, 678, 158–168. [Google Scholar] [CrossRef]
- Ortega, R.M.; Martinez, R.M.; Lopez-Sobaler, A.M.; Andres, P.; Quintas, M.E. Influence of calcium intake on gestational hypertension. Ann. Nutr. Metab. 1999, 43, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Bucher, H.C.; Cook, R.J.; Guyatt, G.H.; Lang, J.D.; Cook, D.J.; Hatala, R.; Hunt, D.L. Effects of dietary calcium supplementation on blood pressure. A meta-analysis of randomized controlled trials. JAMA 1996, 275, 1016–1022. [Google Scholar] [CrossRef] [PubMed]
- Dror, D.K. Vitamin D status during pregnancy: Maternal, fetal, and postnatal outcomes. Curr. Opin. Obs. Gynecol. 2011, 23, 422–426. [Google Scholar] [CrossRef]
- MacKay, A.P.; Berg, C.J.; Atrash, H.K. Pregnancy-related mortality from preeclampsia and eclampsia. Obs. Gynecol. 2001, 97, 533–538. [Google Scholar] [CrossRef]
- De-Regil, L.M.; Palacios, C.; Lombardo, L.K.; Pena-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ota, E.; Mori, R.; Middleton, P.; Tobe-Gai, R.; Mahomed, K.; Miyazaki, C.; Bhutta, Z.A. Zinc supplementation for improving pregnancy and infant outcome. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P., Jr.; Christian, P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention. Nat. Rev. Endocrinol 2016, 12, 274–289. [Google Scholar] [CrossRef]
- Victora, C.G.; Adair, L.; Fall, C.; Hallal, P.C.; Martorell, R.; Richter, L.; Sachdev, H.S.; Maternal; Child Undernutrition Study Group. Maternal and child undernutrition: Consequences for adult health and human capital. Lancet 2008, 371, 340–357. [Google Scholar] [CrossRef]
- Bhutta, Z.A.; Ahmed, T.; Black, R.E.; Cousens, S.; Dewey, K.; Giugliani, E.; Haider, B.A.; Kirkwood, B.; Morris, S.S.; Sachdev, H.P.S.; et al. What works? Interventions for maternal and child undernutrition and survival. Lancet 2008, 371, 417–440. [Google Scholar] [CrossRef]
- WHO. WHO Recommendations on Antenatal Care for a Positive Pregnancy Experience; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes tables and application. Available online: http://nationalacademies.org/hmd/Activities/Nutrition/SummaryDRIs/DRI-Tables.aspx (accessed on 20 December 2019).
- UNICEF; WHO; UNU. Composition of a Multi-Micronutrient Supplement to Be Used in Pilot Programmes among Pregnant Women in Developing Countries; Report of a United Nations Children’s Fund (UNICEF), World Health Organization (WHO), United Nations University (UNU) Workshop; UNICEF: New York, NY, USA, 1999. [Google Scholar]
- Arimond, M.; Zeilani, M.; Jungjohann, S.; Brown, K.H.; Ashorn, P.; Allen, L.H.; Dewey, K.G. Considerations in developing lipid-based nutrient supplements for prevention of undernutrition: Experience from the International Lipid-Based Nutrient Supplements (iLiNS) Project. Matern. Child Nutr. 2015, 11 (Suppl. 4), 31–61. [Google Scholar] [CrossRef]
- Lassi, Z.S.; Salam, R.A.; Haider, B.A.; Bhutta, Z.A. Folic acid supplementation during pregnancy for maternal health and pregnancy outcomes. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef] [PubMed]
- Rumbold, A.; Ota, E.; Nagata, C.; Shahrook, S.; Crowther, C.A. Vitamin C supplementation in pregnancy. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Bougma, K.; Aboud, F.E.; Harding, K.B.; Marquis, G.S. Iodine and mental development of children 5 years old and under: A systematic review and meta-analysis. Nutrients 2013, 5, 1384–1416. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.J.; Anderson, A.J.; Gibson, R.A.; Makrides, M. Effect of iodine supplementation in pregnancy on child development and other clinical outcomes: A systematic review of randomized controlled trials. Am. J. Clin. Nutr. 2013, 98, 1241–1254. [Google Scholar] [CrossRef] [PubMed]
- Pena-Rosas, J.P.; De-Regil, L.M.; Garcia-Casal, M.N.; Dowswell, T. Daily oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Imdad, A.; Ramakrishnan, U.; Martorell, R. Is it time to replace iron folate supplements in pregnancy with multiple micronutrients? Paediatr. Perinat. Epidemiol. 2012, 26 (Suppl. 1), 27–35. [Google Scholar] [CrossRef]
- Christian, P.; Osrin, D.; Manandhar, D.S.; Khatry, S.K.; Anthony, M.D.L.; West, K.P., Jr. Antenatal micronutrient supplements in Nepal. Lancet 2005, 366, 711–712. [Google Scholar] [CrossRef]
- Kawai, K.; Spiegelman, D.; Shankar, A.H.; Fawzi, W.W. Maternal multiple micronutrient supplementation and pregnancy outcomes in developing countries: Meta-analysis and meta-regression. Bull. World Health Organ. 2011, 89, 402–411. [Google Scholar] [CrossRef]
- Keats, E.C.; Haider, B.A.; Tam, E.; Bhutta, Z.A. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019, 3, CD004905. [Google Scholar] [CrossRef]
- Margetts, B.M.; Fall, C.H.; Ronsmans, C.; Allen, L.H.; Fisher, D.J.; Maternal Micronutrient Supplementation Study Group. Multiple micronutrient supplementation during pregnancy in low-income countries: Review of methods and characteristics of studies included in the meta-analyses. Food Nutr. Bull. 2009, 30, S517–S526. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Grant, F.K.; Goldenberg, T.; Bui, V.; Imdad, A.; Bhutta, Z.A. Effect of multiple micronutrient supplementation on pregnancy and infant outcomes: A systematic review. Paediatr. Perinat. Epidemiol. 2012, 26 (Suppl. 1), 153–167. [Google Scholar] [CrossRef] [PubMed]
- Das, J.K.; Hoodbhoy, Z.; Salam, R.A.; Bhutta, A.Z.; Valenzuela-Rubio, N.G.; Weise Prinzo, Z.; Bhutta, Z.A. Lipid-based nutrient supplements for maternal, birth, and infant developmental outcomes. Cochrane Database Syst. Rev. 2018, 8, CD012610. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; Kostiuk, L.K.; Pena-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019, 7, CD008873. [Google Scholar] [CrossRef] [PubMed]
- McCauley, M.E.; van den Broek, N.; Dou, L.; Othman, M. Vitamin A supplementation during pregnancy for maternal and newborn outcomes. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Hofmeyr, G.J.; Lawrie, T.A.; Atallah, A.N.; Torloni, M.R. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst. Rev. 2018, 10, CD001059. [Google Scholar] [CrossRef] [PubMed]
- Sudfeld, C.R.; Smith, E.R. New Evidence Should Inform WHO Guidelines on Multiple Micronutrient Supplementation in Pregnancy. J. Nutr. 2019, 149, 359–361. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.R.; Shankar, A.H.; Wu, L.S.; Aboud, S.; Adu-Afarwuah, S.; Ali, H.; Agustina, R.; Arifeen, S.; Ashorn, P.; Bhutta, Z.A.; et al. Modifiers of the effect of maternal multiple micronutrient supplementation on stillbirth, birth outcomes, and infant mortality: A meta-analysis of individual patient data from 17 randomised trials in low-income and middle-income countries. Lancet Glob. Health 2017, 5, e1090–e1100. [Google Scholar] [CrossRef]
- Mwangi, M.N.; Prentice, A.M.; Verhoef, H. Safety and benefits of antenatal oral iron supplementation in low-income countries: A review. Br. J. Haematol. 2017, 177, 884–895. [Google Scholar] [CrossRef]
- Paganini, D.; Uyoga, M.A.; Zimmermann, M.B. Iron Fortification of Foods for Infants and Children in Low-Income Countries: Effects on the Gut Microbiome, Gut Inflammation, and Diarrhea. Nutrients 2016, 8, 494. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (Updated July 2019); Cochrane, 2019; Available online: www.training.cochrane.org/handbook (accessed on 20 December 2019).
- Keats, E.C.; Imad, A.; Bhutta, Z. Protocol: Effects of vitamin and mineral supplementation during pregnancy on maternal, birth, child health and development outcomes in low- and middle-income countries: A systematic review. Campbell Syst. Rev. 2018, 14, 1–33. [Google Scholar] [CrossRef]
- Diogenes, M.E.; Bezerra, F.F.; Rezende, E.P.; Taveira, M.F.; Pinhal, I.; Donangelo, C.M. Effect of calcium plus vitamin D supplementation during pregnancy in Brazilian adolescent mothers: A randomized, placebo-controlled trial. Am. J. Clin. Nutr. 2013, 98, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Duggan, C.; Srinivasan, K.; Thomas, T.; Samuel, T.; Rajendran, R.; Muthayya, S.; Finkelstein, J.L.; Lukose, A.; Fawzi, W.; Allen, L.H.; et al. Vitamin B-12 supplementation during pregnancy and early lactation increases maternal, breast milk, and infant measures of vitamin B-12 status. J. Nutr. 2014, 144, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Gowachirapant, S.; Jaiswal, N.; Melse-Boonstra, A.; Galetti, V.; Stinca, S.; Mackenzie, I.; Thomas, S.; Thomas, T.; Winichagoon, P.; Srinivasan, K.; et al. Effect of iodine supplementation in pregnant women on child neurodevelopment: A randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2017, 5, 853–863. [Google Scholar] [CrossRef]
- Jarjou, L.M.; Prentice, A.; Sawo, Y.; Laskey, M.A.; Bennett, J.; Goldberg, G.R.; Cole, T.J. Randomized, placebo-controlled, calcium supplementation study in pregnant Gambian women: Effects on breast-milk calcium concentrations and infant birth weight, growth, and bone mineral accretion in the first year of life. Am. J. Clin. Nutr. 2006, 83, 657–666. [Google Scholar] [CrossRef]
- Korkmaz, V.; Ozkaya, E.; Seven, B.Y.; Duzguner, S.; Karsli, M.F.; Kucukozkan, T. Comparison of oxidative stress in pregnancies with and without first trimester iron supplement: A randomized double-blind controlled trial. J. Matern. Fetal Neonatal Med. 2014, 27, 1535–1538. [Google Scholar] [CrossRef]
- Prawirohartono, E.P.; Nystrom, L.; Ivarsson, A.; Stenlund, H.; Lind, T. The impact of prenatal vitamin A and zinc supplementation on growth of children up to 2 years of age in rural Java, Indonesia. Public Health Nutr. 2011, 14, 2197–2206. [Google Scholar] [CrossRef]
- Taherian, A.A.; Taherian, A.; Shirvani, A. Prevention of pre-eclampsia with low-dose aspirin or calcium supplementation. Arch. Iran. Med. 2002, 5, 151–156. [Google Scholar]
- Hambidge, K.M.; Westcott, J.E.; Garces, A.; Figueroa, L.; Goudar, S.S.; Dhaded, S.M.; Pasha, O.; Ali, S.A.; Tshefu, A.; Lokangaka, A.; et al. A multicountry randomized controlled trial of comprehensive maternal nutrition supplementation initiated before conception: The Women First trial. Am. J. Clin. Nutr. 2019, 109, 457–469. [Google Scholar] [CrossRef]
- Dijkhuizen, M.A.; Wieringa, F.T.; West, C.E.; Muherdiyantiningsih; Muhilal. Concurrent micronutrient deficiencies in lactating mothers and their infants in Indonesia. Am. J. Clin. Nutr. 2001, 73, 786–791. [Google Scholar]
- Hanieh, S.; Ha, T.T.; Simpson, J.A.; Casey, G.J.; Khuong, N.C.; Thoang, D.D.; Thuy, T.T.; Pasricha, S.R.; Tran, T.D.; Tuan, T.; et al. The effect of intermittent antenatal iron supplementation on maternal and infant outcomes in rural Viet Nam: A cluster randomised trial. PLoS Med. 2013, 10, e1001470. [Google Scholar] [CrossRef]
- Huy, N.D.; Le Hop, T.; Shrimpton, R.; Hoa, C.V. An effectiveness trial of multiple micronutrient supplementation during pregnancy in Vietnam: Impact on birthweight and on stunting in children at around 2 years of age. Food Nutr. Bull. 2009, 30, S506–S516. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.M.; Mei, Z.; Ye, R.; Serdula, M.K.; Ren, A.; Cogswell, M.E. Micronutrient supplementation and pregnancy outcomes: Double-blind randomized controlled trial in China. JAMA Intern. Med. 2013, 173, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Muslimatun, S.; Schmidt, M.K.; Schultink, W.; West, C.E.; Hautvast, J.A.; Gross, R.; Muhilal. Weekly supplementation with iron and vitamin A during pregnancy increases hemoglobin concentration but decreases serum ferritin concentration in Indonesian pregnant women. J. Nutr. 2001, 131, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Supplementation with Multiple Micronutrients Intervention Trial Study Group. Effect of maternal multiple micronutrient supplementation on fetal loss and infant death in Indonesia: A double-blind cluster-randomised trial. Lancet 2008, 371, 215–227. [Google Scholar] [CrossRef]
- Sunawang; Utomo, B.; Hidayat, A.; Kusharisupeni; Subarkah. Preventing low birthweight through maternal multiple micronutrient supplementation: A cluster-randomized, controlled trial in Indramayu, West Java. Food Nutr. Bull. 2009, 30, S488–S495. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.A. Vitamin A and iron status are improved by vitamin A and iron supplementation in pregnant Indonesian women. J. Nutr. 2002, 132, 1909–1912. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Dibley, M.J.; Cheng, Y.; Dang, S.; Chang, S.; Kong, L.; Yan, H. Impact of micronutrient supplementation during pregnancy on birth weight, duration of gestation, and perinatal mortality in rural western China: Double blind cluster randomised controlled trial. BMJ 2008, 337, a2001. [Google Scholar] [CrossRef]
- Zhao, G.; Xu, G.; Zhou, M.; Jiang, Y.; Richards, B.; Clark, K.M.; Kaciroti, N.; Georgieff, M.K.; Zhang, Z.; Tardif, T.; et al. Prenatal Iron Supplementation Reduces Maternal Anemia, Iron Deficiency, and Iron Deficiency Anemia in a Randomized Clinical Trial in Rural China, but Iron Deficiency Remains Widespread in Mothers and Neonates. J. Nutr. 2015, 145, 1916–1923. [Google Scholar] [CrossRef]
- Aminisani, N.; Ehdaivand, F.; Shamshirgaran, S.M.; Mohajery, M.; Pourfarzi, F.; Ahari, S. Zinc supplementation during pregnancy: A randomized controlled trial. Iran. J. Pharmacol. Therapeutics. 2009, 8, 67–71. [Google Scholar]
- Asemi, Z.; Samimi, M.; Tabassi, Z.; Shakeri, H.; Esmaillzadeh, A. Vitamin D supplementation affects serum high-sensitivity C-reactive protein, insulin resistance, and biomarkers of oxidative stress in pregnant women. J. Nutr. 2013, 143, 1432–1438. [Google Scholar] [CrossRef]
- Asemi, Z.; Samimi, M.; Siavashani, M.A.; Mazloomi, M.; Tabassi, Z.; Karamali, M.; Jamilian, M.; Esmaillzadeh, A. Calcium-Vitamin D Co-supplementation Affects Metabolic Profiles, but not Pregnancy Outcomes, in Healthy Pregnant Women. Int. J. Prev. Med. 2016, 7, 49. [Google Scholar] [CrossRef]
- Mohammad-Alizadeh-Charandabi, S.; Mirghafourvand, M.; Mansouri, A.; Najafi, M.; Khodabande, F. The Effect of Vitamin D and Calcium plus Vitamin D during Pregnancy on Pregnancy and Birth Outcomes: A Randomized Controlled Trial. J. Caring Sci. 2015, 4, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Falahi, E.; Akbari, S.; Ebrahimzade, F.; Gargari, B.P. Impact of prophylactic iron supplementation in healthy pregnant women on maternal iron status and birth outcome. Food Nutr. Bull. 2011, 32, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Naghshineh, E.; Sheikhaliyan, S. Effect of vitamin D supplementation in the reduce risk of preeclampsia in nulliparous women. Adv. Biomed. Res. 2016, 5, 7. [Google Scholar] [CrossRef]
- Ouladsahebmadarek, E.; Sayyah-Melli, M.; Taghavi, S.; Abbasalizadeh, S.; Seyedhejazie, M. The effect of supplemental iron elimination on pregnancy outcome. Pak. J. Med. Sci 2011, 27, 641–645. [Google Scholar]
- Sabet, Z.; Ghazi, A.A.; Tohidi, M.; Oladi, B. Vitamin D Supplementation in Pregnant Iranian Women: Effects on Maternal and Neonatal Vitamin D and Parathyroid Hormone Status. Acta Endocrinol.-Buch. 2012, 8, 59–66. [Google Scholar] [CrossRef]
- Zahiri Sorouri, Z.; Sadeghi, H.; Pourmarzi, D. The effect of zinc supplementation on pregnancy outcome: A randomized controlled trial. J. Matern. Fetal Neonatal Med. 2016, 29, 2194–2198. [Google Scholar] [CrossRef]
- Vaziri, F.; Nasiri, S.; Tavana, Z.; Dabbaghmanesh, M.H.; Sharif, F.; Jafari, P. A randomized controlled trial of vitamin D supplementation on perinatal depression: In Iranian pregnant mothers. BMC Pregnancy Childbirth 2016, 16, 239. [Google Scholar] [CrossRef]
- Ziaei, S.; Norrozi, M.; Faghihzadeh, S.; Jafarbegloo, E. A randomised placebo-controlled trial to determine the effect of iron supplementation on pregnancy outcome in pregnant women with haemoglobin > or = 13.2 g/dl. BJOG 2007, 114, 684–688. [Google Scholar] [CrossRef]
- Ziaei, S.; Mehrnia, M.; Faghihzadeh, S. Iron status markers in nonanemic pregnant women with and without iron supplementation. Int. J. Gynaecol. Obs. 2008, 100, 130–132. [Google Scholar] [CrossRef]
- Ashorn, P.; Alho, L.; Ashorn, U.; Cheung, Y.B.; Dewey, K.G.; Gondwe, A.; Harjunmaa, U.; Lartey, A.; Phiri, N.; Phiri, T.E.; et al. Supplementation of Maternal Diets during Pregnancy and for 6 Months Postpartum and Infant Diets Thereafter with Small-Quantity Lipid-Based Nutrient Supplements Does Not Promote Child Growth by 18 Months of Age in Rural Malawi: A Randomized Controlled Trial. J. Nutr. 2015, 145, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Cox, S.E.; Arthur, P.; Kirkwood, B.R.; Yeboah-Antwi, K.; Riley, E.M. Vitamin A supplementation increases ratios of proinflammatory to anti-inflammatory cytokine responses in pregnancy and lactation. Clin. Exp. Immunol. 2006, 144, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Adu-Afarwuah, S.; Lartey, A.; Okronipa, H.; Ashorn, P.; Zeilani, M.; Peerson, J.M.; Arimond, M.; Vosti, S.; Dewey, K.G. Lipid-based nutrient supplement increases the birth size of infants of primiparous women in Ghana. Am. J. Clin. Nutr. 2015, 101, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Etheredge, A.J.; Premji, Z.; Gunaratna, N.S.; Abioye, A.I.; Aboud, S.; Duggan, C.; Mongi, R.; Meloney, L.; Spiegelman, D.; Roberts, D.; et al. Iron Supplementation in Iron-Replete and Nonanemic Pregnant Women in Tanzania: A Randomized Clinical Trial. JAMA Pediatr. 2015, 169, 947–955. [Google Scholar] [CrossRef]
- Fawzi, W.W.; Msamanga, G.I.; Urassa, W.; Hertzmark, E.; Petraro, P.; Willett, W.C.; Spiegelman, D. Vitamins and perinatal outcomes among HIV-negative women in Tanzania. N. Engl. J. Med. 2007, 356, 1423–1431. [Google Scholar] [CrossRef]
- Friis, H.; Gomo, E.; Nyazema, N.; Ndhlovu, P.; Krarup, H.; Kaestel, P.; Michaelsen, K.F. Effect of multimicronutrient supplementation on gestational length and birth size: A randomized, placebo-controlled, double-blind effectiveness trial in Zimbabwe. Am. J. Clin. Nutr. 2004, 80, 178–184. [Google Scholar] [CrossRef]
- Huybregts, L.; Roberfroid, D.; Lanou, H.; Menten, J.; Meda, N.; Van Camp, J.; Kolsteren, P. Prenatal food supplementation fortified with multiple micronutrients increases birth length: A randomized controlled trial in rural Burkina Faso. Am. J. Clin. Nutr. 2009, 90, 1593–1600. [Google Scholar] [CrossRef]
- Kaestel, P.; Michaelsen, K.F.; Aaby, P.; Friis, H. Effects of prenatal multimicronutrient supplements on birth weight and perinatal mortality: A randomised, controlled trial in Guinea-Bissau. Eur. J. Clin. Nutr. 2005, 59, 1081–1089. [Google Scholar] [CrossRef]
- Kirkwood, B.R.; Hurt, L.; Amenga-Etego, S.; Tawiah, C.; Zandoh, C.; Danso, S.; Hurt, C.; Edmond, K.; Hill, Z.; Ten Asbroek, G.; et al. Effect of vitamin A supplementation in women of reproductive age on maternal survival in Ghana (ObaapaVitA): A cluster-randomised, placebo-controlled trial. Lancet 2010, 375, 1640–1649. [Google Scholar] [CrossRef]
- Menendez, C.; Todd, J.; Alonso, P.L.; Francis, N.; Lulat, S.; Ceesay, S.; M’Boge, B.; Greenwood, B.M. The effects of iron supplementation during pregnancy, given by traditional birth attendants, on the prevalence of anaemia and malaria. Trans. R. Soc. Trop. Med. Hyg. 1994, 88, 590–593. [Google Scholar] [CrossRef]
- Johnson, W.; Darboe, M.K.; Sosseh, F.; Nshe, P.; Prentice, A.M.; Moore, S.E. Association of prenatal lipid-based nutritional supplementation with fetal growth in rural Gambia. Matern. Child Nutr. 2017, 13, e12367. [Google Scholar] [CrossRef] [PubMed]
- Preziosi, P.; Prual, A.; Galan, P.; Daouda, H.; Boureima, H.; Hercberg, S. Effect of iron supplementation on the iron status of pregnant women: Consequences for newborns. Am. J. Clin. Nutr. 1997, 66, 1178–1182. [Google Scholar] [CrossRef] [PubMed]
- Roberfroid, D.; Huybregts, L.; Lanou, H.; Henry, M.C.; Meda, N.; Menten, J.; Kolsteren, P.; Group, M.S. Effects of maternal multiple micronutrient supplementation on fetal growth: A double-blind randomized controlled trial in rural Burkina Faso. Am. J. Clin. Nutr. 2008, 88, 1330–1340. [Google Scholar] [CrossRef] [PubMed]
- Saaka, M.; Oosthuizen, J.; Beatty, S. Effect of joint iron and zinc supplementation on malarial infection and anaemia. East. Afr. J. Public Health 2009, 6, 55–62. [Google Scholar] [CrossRef]
- Semba, R.D.; Kumwenda, N.; Taha, T.E.; Mtimavalye, L.; Broadhead, R.; Garrett, E.; Miotti, P.G.; Chiphangwi, J.D. Impact of vitamin A supplementation on anaemia and plasma erythropoietin concentrations in pregnant women: A controlled clinical trial. Eur. J. Haematol. 2001, 66, 389–395. [Google Scholar] [CrossRef]
- Zagre, N.M.; Desplats, G.; Adou, P.; Mamadoultaibou, A.; Aguayo, V.M. Prenatal multiple micronutrient supplementation has greater impact on birthweight than supplementation with iron and folic acid: A cluster-randomized, double-blind, controlled programmatic study in rural Niger. Food Nutr. Bull. 2007, 28, 317–327. [Google Scholar] [CrossRef]
- Darling, A.M.; Mugusi, F.M.; Etheredge, A.J.; Gunaratna, N.S.; Abioye, A.I.; Aboud, S.; Duggan, C.; Mongi, R.; Spiegelman, D.; Roberts, D.; et al. Vitamin A and Zinc Supplementation Among Pregnant Women to Prevent Placental Malaria: A Randomized, Double-Blind, Placebo-Controlled Trial in Tanzania. Am. J. Trop. Med. Hyg. 2017, 96, 826–834. [Google Scholar] [CrossRef]
- Ahmad, S.M.; Hossain, M.B.; Monirujjaman, M.; Islam, S.; Huda, M.N.; Kabir, Y.; Raqib, R.; Lonnerdal, B.L. Maternal zinc supplementation improves hepatitis B antibody responses in infants but decreases plasma zinc level. Eur. J. Nutr. 2016, 55, 1823–1829. [Google Scholar] [CrossRef]
- Bhutta, Z.A.; Rizvi, A.; Raza, F.; Hotwani, S.; Zaidi, S.; Moazzam Hossain, S.; Soofi, S.; Bhutta, S. A comparative evaluation of multiple micronutrient and iron-folic acid supplementation during pregnancy in Pakistan: Impact on pregnancy outcomes. Food Nutr. Bull. 2009, 30, S496–S505. [Google Scholar] [CrossRef]
- Choudhury, N.; Aimone, A.; Hyder, S.M.; Zlotkin, S.H. Relative efficacy of micronutrient powders versus iron-folic acid tablets in controlling anemia in women in the second trimester of pregnancy. Food Nutr. Bull. 2012, 33, 142–149. [Google Scholar] [CrossRef]
- Christian, P.; West, K.P.; Khatry, S.K.; Leclerq, S.C.; Pradhan, E.K.; Katz, J.; Shrestha, S.R.; Sommer, A. Effects of maternal micronutrient supplementation on fetal loss and infant mortality: A cluster-randomized trial in Nepal. Am. J. Clin. Nutr. 2003, 78, 1194–1202. [Google Scholar] [CrossRef] [PubMed]
- Hafeez, A.; Mehmood, G.; Mazhar, F. Oral zinc supplementation in pregnant women and its effect on birth weight: A randomised controlled trial. Arch. Dis. Child. Fetal Neonatal Ed. 2005, 90, F170–F171. [Google Scholar] [CrossRef] [PubMed]
- Hossain, N.; Kanani, F.; Khanani, R.; Ayaz, S.; Pal, L. O317 Effect of Maternal Supplementation with Vitamin D during Pregnancy on Neonatal Serum Vitamin D Levels and Anthropometric Measurements. Int. J. Gynecol. Obstet. 2012, 119, S372. [Google Scholar] [CrossRef]
- Khan, F.R.; Ahmad, T.; Hussain, R.; Bhutta, Z.A. A randomized controlled trial of oral vitamin D supplementation in pregnancy to improve maternal periodontal health and birth weight. J. Int. Oral Health 2016, 8, 657. [Google Scholar] [CrossRef]
- Kumar, A.; Devi, S.G.; Batra, S.; Singh, C.; Shukla, D.K. Calcium supplementation for the prevention of pre-eclampsia. Int. J. Gynaecol. Obs. 2009, 104, 32–36. [Google Scholar] [CrossRef]
- Osendarp, S.J.; van Raaij, J.M.; Arifeen, S.E.; Wahed, M.; Baqui, A.H.; Fuchs, G.J. A randomized, placebo-controlled trial of the effect of zinc supplementation during pregnancy on pregnancy outcome in Bangladeshi urban poor. Am. J. Clin. Nutr. 2000, 71, 114–119. [Google Scholar] [CrossRef]
- Osrin, D.; Vaidya, A.; Shrestha, Y.; Baniya, R.B.; Manandhar, D.S.; Adhikari, R.K.; Filteau, S.; Tomkins, A.; Costello, A.M. Effects of antenatal multiple micronutrient supplementation on birthweight and gestational duration in Nepal: Double-blind, randomised controlled trial. Lancet 2005, 365, 955–962. [Google Scholar] [CrossRef]
- Roth, D.E.; Perumal, N.; Al Mahmud, A.; Baqui, A.H. Maternal vitamin D3 supplementation during the third trimester of pregnancy: Effects on infant growth in a longitudinal follow-up study in Bangladesh. J. Pediatr. 2013, 163, 1605–1611. [Google Scholar] [CrossRef]
- Roth, D.E.; Morris, S.K.; Zlotkin, S.; Gernand, A.D.; Ahmed, T.; Shanta, S.S.; Papp, E.; Korsiak, J.; Shi, J.; Islam, M.M.; et al. Vitamin D Supplementation in Pregnancy and Lactation and Infant Growth. N. Engl. J. Med. 2018, 379, 535–546. [Google Scholar] [CrossRef]
- Sablok, A.; Batra, A.; Thariani, K.; Batra, A.; Bharti, R.; Aggarwal, A.R.; Kabi, B.C.; Chellani, H. Supplementation of vitamin D in pregnancy and its correlation with feto-maternal outcome. Clin. Endocrinol. (Oxf.) 2015, 83, 536–541. [Google Scholar] [CrossRef]
- Tofail, F.; Persson, L.A.; El Arifeen, S.; Hamadani, J.D.; Mehrin, F.; Ridout, D.; Ekstrom, E.C.; Huda, S.N.; Grantham-McGregor, S.M. Effects of prenatal food and micronutrient supplementation on infant development: A randomized trial from the Maternal and Infant Nutrition Interventions, Matlab (MINIMat) study. Am. J. Clin. Nutr. 2008, 87, 704–711. [Google Scholar] [CrossRef] [PubMed]
- West, K.P.; Shamim, A.A.; Mehra, S.; Labrique, A.B.; Ali, H.; Shaikh, S.; Klemm, R.D.W.; Wu, L.S.F.; Mitra, M.; Haque, R.; et al. Effect of Maternal Multiple Micronutrient vs Iron–Folic Acid Supplementation on Infant Mortality and Adverse Birth Outcomes in Rural Bangladesh. JAMA 2014, 312, 2649. [Google Scholar] [CrossRef] [PubMed]
- West, K.P., Jr.; Christian, P.; Labrique, A.B.; Rashid, M.; Shamim, A.A.; Klemm, R.D.; Massie, A.B.; Mehra, S.; Schulze, K.J.; Ali, H.; et al. Effects of vitamin A or beta carotene supplementation on pregnancy-related mortality and infant mortality in rural Bangladesh: A cluster randomized trial. JAMA 2011, 305, 1986–1995. [Google Scholar] [CrossRef]
- West, K.P., Jr.; Katz, J.; Khatry, S.K.; LeClerq, S.C.; Pradhan, E.K.; Shrestha, S.R.; Connor, P.B.; Dali, S.M.; Christian, P.; Pokhrel, R.P.; et al. Double blind, cluster randomised trial of low dose supplementation with vitamin A or beta-carotene on mortality related to pregnancy in Nepal: The nnips-2 study group. BMJ 1999, 318, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Sahu, M.; Das, V.; Aggarwal, A.; Rawat, V.; Saxena, P.; Bhatia, V. Vitamin D replacement in pregnant women in rural north India: A pilot study. Eur. J. Clin. Nutr. 2009, 63, 1157–1159. [Google Scholar] [CrossRef]
- Belizan, J.M.; Villar, J.; Bergel, E.; del Pino, A.; Di Fulvio, S.; Galliano, S.V.; Kattan, C. Long-term effect of calcium supplementation during pregnancy on the blood pressure of offspring: Follow up of a randomised controlled trial. BMJ 1997, 315, 281–285. [Google Scholar] [CrossRef]
- Castillo-Duran, C.; Marin, V.B.; Alcazar, L.S.; Iturralde, H.; Ruz, M.O. Controlled trial of zinc supplementation in Chilean pregnant adolescents. Nutr. Res. 2001, 21, 715–724. [Google Scholar] [CrossRef]
- Caulfield, L.E.; Zavaleta, N.; Figueroa, A. Adding zinc to prenatal iron and folate supplements improves maternal and neonatal zinc status in a Peruvian population. Am. J. Clin. Nutr. 1999, 69, 1257–1263. [Google Scholar] [CrossRef]
- Lopezjaramillo, P.; Delgado, F.; Jacome, P.; Teran, E.; Ruano, C.; Rivera, J. Calcium supplementation and the risk of preeclampsia in ecuadorian pregnant teenagers. Obstet. Gynecol. 1997, 90, 162–167. [Google Scholar] [CrossRef]
- Merialdi, M.; Caulfield, L.E.; Zavaleta, N.; Figueroa, A.; Dominici, F.; Dipietro, J.A. Randomized controlled trial of prenatal zinc supplementation and the development of fetal heart rate. Am. J. Obs. Gynecol. 2004, 190, 1106–1112. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Gonzalez-Cossio, T.; Neufeld, L.M.; Rivera, J.; Martorell, R. Multiple micronutrient supplementation during pregnancy does not lead to greater infant birth size than does iron-only supplementation: A randomized controlled trial in a semirural community in Mexico. Am. J. Clin. Nutr. 2003, 77, 720–725. [Google Scholar] [CrossRef] [PubMed]
- Villar, J.; Abdel-Aleem, H.; Merialdi, M.; Mathai, M.; Ali, M.M.; Zavaleta, N.; Purwar, M.; Hofmeyr, J.; Nguyen, T.N.; Campodonico, L.; et al. World Health Organization randomized trial of calcium supplementation among low calcium intake pregnant women. Am. J. Obs. Gynecol. 2006, 194, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Roth, D.E.; Leung, M.; Mesfin, E.; Qamar, H.; Watterworth, J.; Papp, E. Vitamin D supplementation during pregnancy: State of the evidence from a systematic review of randomised trials. BMJ 2017, 359, j5237. [Google Scholar] [CrossRef] [PubMed]
- Lassi, Z.S.; Dean, S.V.; Mallick, D.; Bhutta, Z.A. Preconception care: Delivery strategies and packages for care. Reprod. Health 2014, 11 (Suppl. 3), S7. [Google Scholar] [CrossRef]
- Dean, S.V.; Lassi, Z.S.; Imam, A.M.; Bhutta, Z.A. Preconception care: Nutritional risks and interventions. Reprod. Health 2014, 11 (Suppl. 3), S3. [Google Scholar] [CrossRef]
- Mason, E.; Chandra-Mouli, V.; Baltag, V.; Christiansen, C.; Lassi, Z.S.; Bhutta, Z.A. Preconception care: Advancing from ‘important to do and can be done’ to ‘is being done and is making a difference’. Reprod. Health 2014, 1, S3–S8. [Google Scholar] [CrossRef]
- Blum, R.W.; Gates, W.H. Girlhood Not Motherhood. Preventing Adolescent Pregnancy; United Nations Population Fund (UNFPA): New York, NY, USA, 2015. [Google Scholar]
- Zhao, D.; Zou, L.; Lei, X.; Zhang, Y. Gender Differences in Infant Mortality and Neonatal Morbidity in Mixed-Gender Twins. Sci. Rep. 2017, 7, 8736. [Google Scholar] [CrossRef]
- Stevenson, D.K.; Verter, J.; Fanaroff, A.A.; Oh, W.; Ehrenkranz, R.A.; Shankaran, S.; Donovan, E.F.; Wright, L.L.; Lemons, J.A.; Tyson, J.E.; et al. Sex differences in outcomes of very low birthweight infants: The newborn male disadvantage. Arch. Dis. Child. 2000, 83, F182–F185. [Google Scholar] [CrossRef]
- Lee, A.C.; Darmstadt, G.L.; Khatry, S.K.; LeClerq, S.C.; Shrestha, S.R.; Christian, P. Maternal-fetal disproportion and birth asphyxia in rural Sarlahi, Nepal. Arch. Pediatr. Adolesc. Med. 2009, 163, 616–623. [Google Scholar] [CrossRef]
- Hossain, M.; Choudhury, N.; Adib Binte Abdullah, K.; Mondal, P.; Jackson, A.A.; Walson, J.; Ahmed, T. Evidence-based approaches to childhood stunting in low and middle income countries: A systematic review. Arch. Dis. Child. 2017, 102, 903–909. [Google Scholar] [CrossRef]
| Vitamins, Micronutrients and Macronutrients | Unit | Institute of Medicine Recommended Dietary Allowance (RDA) for Pregnancy | ||
|---|---|---|---|---|
| 14–18 Years | 19–30 Years | 31–50 Years | ||
| Vitamin A (retinol) | µg | 750 | 770 | 770 |
| Vitamin B1 (thiamin) | mg | 1.4 | 1.4 | 1.4 |
| Vitamin B2 (riboflavin) | mg | 1.4 | 1.4 | 1.4 |
| Vitamin B3 (niacin) | mg | 18 | 18 | 18 |
| Vitamin B5 (pantothenic acid) | mg | 6 | 6 | 6 |
| Vitamin B6 (pyridoxine) | mg | 1.9 | 1.9 | 1.9 |
| Vitamin B7 (biotin) | µg | 30 | 30 | 30 |
| Vitamin B9 (folate) | µg | 600 | 600 | 600 |
| Vitamin B12 (cobalamine) | µg | 2.6 | 2.6 | 2.6 |
| Vitamin C (ascorbate) | mg | 80 | 85 | 85 |
| Vitamin D (cholecalciferol) | IU | 15 | 15 | 15 |
| Vitamin E (tocopherol acetate) | mg | 15 | 15 | 15 |
| Vitamin K (phytomenadione) | µg | 75 | 90 | 90 |
| Choline | mg | 450 | 450 | 450 |
| Calcium | mg | 1300 | 1000 | 1000 |
| Chromium | g | 29 | 30 | 30 |
| Copper | µg | 1000 | 1000 | 1000 |
| Fluoride | mg | 3 | 3 | 3 |
| Iodine | µg | 220 | 220 | 220 |
| Iron | mg | 27 | 27 | 27 |
| Magnesium | mg | 400 | 350 | 360 |
| Phosphorus | mg | 1250 | 700 | 700 |
| Selenium | µg | 60 | 60 | 60 |
| Zinc | mg | 12 | 11 | 11 |
| Potassium | mg | 2600 | 2900 | 2900 |
| Sodium | mg | 1500 | 1500 | 1500 |
| Chloride | g | 2.3 | 2.3 | 2.3 |
| Carbohydrate | g | 175 | 175 | 175 |
| Fat | g | Not determined | Not determined | Not determined |
| Linoleic Acid | g | 13 | 13 | 13 |
| α-Linoleic Acid | g | 1.4 | 1.4 | 1.4 |
| Protein | g | 71 | 71 | 71 |
| Inclusion Criteria |
|
|
|
|
|
| Exclusion Criteria |
|
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, C.; Keats, E.C.; Bhutta, Z.A. Vitamin and Mineral Supplementation During Pregnancy on Maternal, Birth, Child Health and Development Outcomes in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 491. https://doi.org/10.3390/nu12020491
Oh C, Keats EC, Bhutta ZA. Vitamin and Mineral Supplementation During Pregnancy on Maternal, Birth, Child Health and Development Outcomes in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis. Nutrients. 2020; 12(2):491. https://doi.org/10.3390/nu12020491
Chicago/Turabian StyleOh, Christina, Emily C. Keats, and Zulfiqar A. Bhutta. 2020. "Vitamin and Mineral Supplementation During Pregnancy on Maternal, Birth, Child Health and Development Outcomes in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis" Nutrients 12, no. 2: 491. https://doi.org/10.3390/nu12020491
APA StyleOh, C., Keats, E. C., & Bhutta, Z. A. (2020). Vitamin and Mineral Supplementation During Pregnancy on Maternal, Birth, Child Health and Development Outcomes in Low- and Middle-Income Countries: A Systematic Review and Meta-Analysis. Nutrients, 12(2), 491. https://doi.org/10.3390/nu12020491





