Beneficial Effects of Dark Chocolate for Episodic Memory in Healthy Young Adults: A Parallel-Groups Acute Intervention with a White Chocolate Control
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Participants
2.3. Outcome Measures
2.4. Procedures
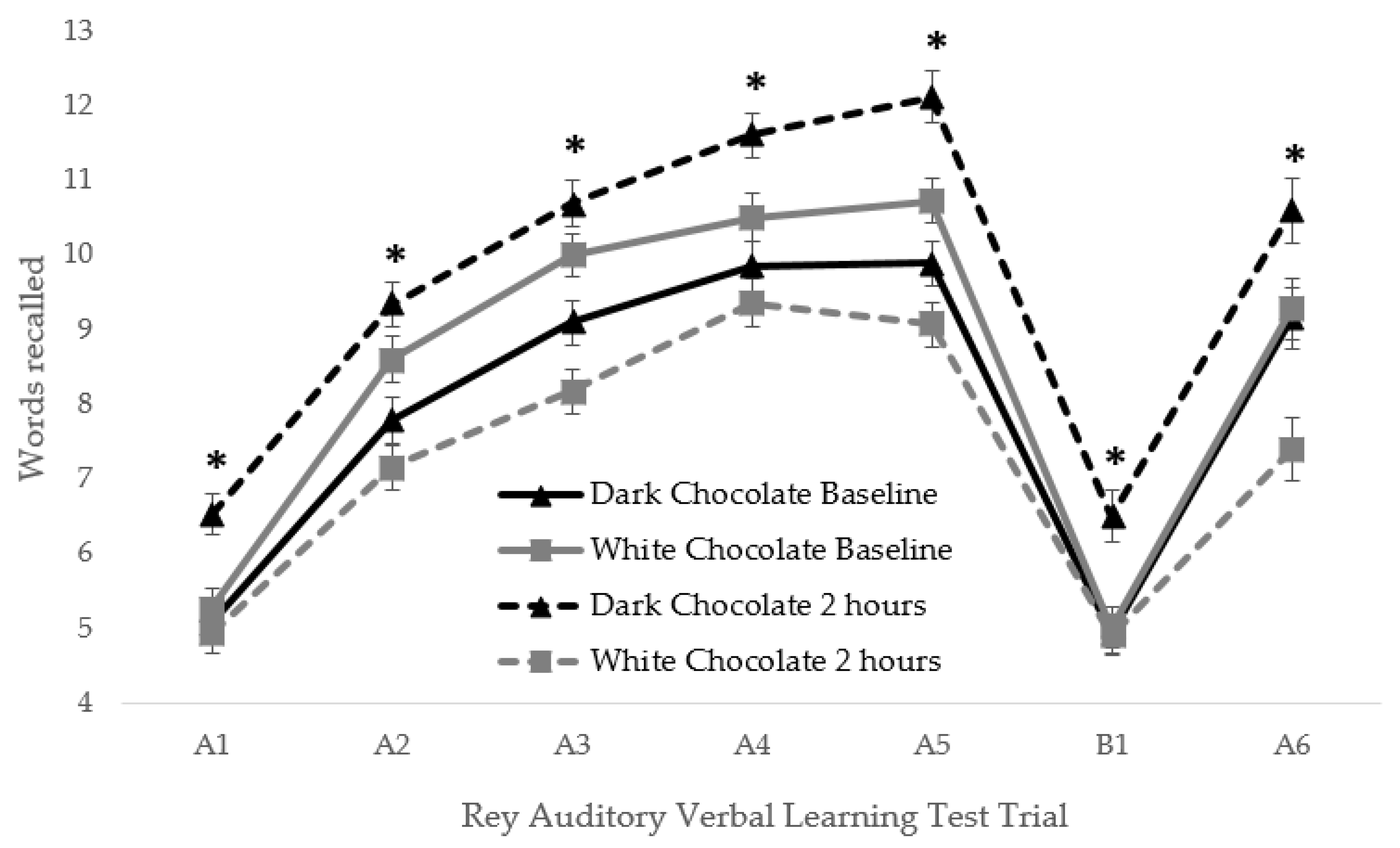
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rees, A.; Dodd, G.F.; Spencer, J.P.E. The effects of flavonoids on cardiovascular health: A review of human intervention trials and implications for cerebrovascular function. Nutrients 2018, 10, 1852. [Google Scholar] [CrossRef]
- Scholey, A.; Owen, L. Effects of chocolate on cognitive function and mood: A systematic review. Nutr. Rev. 2013, 71, 665–681. [Google Scholar] [CrossRef]
- Socci, V.; Tempesta, D.; Desideri, G.; De Gennaro, L.; Ferrara, M. Enhancing human cognition with cocoa flavonoids. Front. Nutr. 2017, 4, 19. [Google Scholar] [CrossRef] [PubMed]
- Brickman, A.M.; Khan, U.A.; Provenzano, F.A.; Yeung, L.K.; Suzuki, W.; Schroeter, H.; Wall, M.; Sloan, R.P.; Small, S.A. Enhancing dentate gyrus function with dietary flavanols improves cognition in older adults. Nat. Neurosci. 2014, 17, 1798–1803. [Google Scholar] [CrossRef] [PubMed]
- Mastroiacovo, D.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Raffaele, A.; Pistacchio, L.; Righetti, R.; Bocale, R.; Lechiara, M.C.; Marini, C.; et al. Cocoa flavanol consumption improves cognitive function, blood pressure control, and metabolic profile in elderly subjects: The cocoa, cognition, and aging (cocoa) study—A randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Desideri, G.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Ghiadoni, L.; Mastroiacovo, D.; Raffaele, A.; Ferri, L.; Bocale, R.; Lechiara, M.C.; et al. Benefits in cognitive function, blood pressure, and insulin resistance through cocoa flavanol consumption in elderly subjects with mild cognitive impairment: The cocoa, cognition, and aging (cocoa) study. Hypertension 2012, 60, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.T.; Head, K.; Morris, P.G.; Macdonald, I.A. The effect of flavanol-rich cocoa on the fmri response to a cognitive task in healthy young people. J. Cardiovasc. Pharmacol. 2006, 47 (Suppl. S2), S215–S220. [Google Scholar] [CrossRef]
- Haskell-Ramsay, C.F.; Schmitt, J.; Actis-Goretta, L. The impact of epicatechin on human cognition: The role of cerebral blood flow. Nutrients 2018, 10, 986. [Google Scholar] [CrossRef]
- Neshatdoust, S.; Saunders, C.; Castle, S.M.; Vauzour, D.; Williams, C.; Butler, L.; Lovegrove, J.A.; Spencer, J.P. High-flavonoid intake induces cognitive improvements linked to changes in serum brain-derived neurotrophic factor: Two randomised, controlled trials. Nutr. Healthy Aging 2016, 4, 81–93. [Google Scholar] [CrossRef]
- Lamport, D.J.; Pal, D.; Moutsiana, C.; Field, D.T.; Williams, C.M.; Spencer, J.P.E.; Butler, L.T. The effect of flavanol-rich cocoa on cerebral perfusion in healthy older adults during conscious resting state: A placebo controlled, crossover, acute trial. Psychopharmacology 2015, 232, 3227–3234. [Google Scholar] [CrossRef]
- Scholey, A.B.; French, S.J.; Morris, P.J.; Kennedy, D.O.; Milne, A.L.; Haskell, C.F. Consumption of cocoa flavanols results in acute improvements in mood and cognitive performance during sustained mental effort. J. Psychopharmacol. 2010, 24, 1505–1514. [Google Scholar] [CrossRef] [PubMed]
- Field, D.T.; Williams, C.M.; Butler, L.T. Consumption of cocoa flavanols results in an acute improvement in visual and cognitive functions. Physiol. Behav. 2011, 103, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Massee, L.A.; Ried, K.; Pase, M.; Travica, N.; Yoganathan, J.; Scholey, A.; Macpherson, H.; Kennedy, G.; Sali, A.; Pipingas, A. The acute and sub-chronic effects of cocoa flavanols on mood, cognitive and cardiovascular health in young healthy adults: A randomized, controlled trial. Front. Pharmacol. 2015, 6, 93. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Necozione, S.; di Giosia, P.; Barnabei, R.; Allegaert, L.; Bernaert, H.; Ferri, C. Cocoa consumption dose-dependently improves flow-mediated dilation and arterial stiffness decreasing blood pressure in healthy individuals. J. Hypertens. 2015, 33, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Decroix, L.; van Schuerbeek, P.; Tonoli, C.; van Cutsem, J.; Soares, D.D.; Heyman, E.; Vanderhasselt, T.; Verrelst, R.; Raeymaekers, H.; de Mey, J.; et al. The effect of acute cocoa flavanol intake on the BOLD response and cognitive function in type 1 diabetes: A randomized, placebo-controlled, double-blinded cross-over pilot study. Psychopharmacology 2019, 236, 3421–3428. [Google Scholar] [CrossRef] [PubMed]
- Shiina, Y.; Funabashi, N.; Lee, K.; Murayama, T.; Nakamura, K.; Wakatsuki, Y.; Daimon, M.; Komuro, I. Acute effect of oral flavonoid-rich dark chocolate intake on coronary circulation, as compared with non-flavonoid white chocolate, by transthoracic Doppler echocardiography in healthy adults. Int. J. Cardiol. 2009, 131, 424–429. [Google Scholar] [CrossRef]
- Rabin, J.C.; Karunathilake, N.; Patrizi, K. Effects of Milk vs Dark Chocolate Consumption on Visual Acuity and Contrast Sensitivity Within 2 Hours: A Randomized Clinical Trial. JAMA Ophthalmol. 2018, 136, 678–681. [Google Scholar] [CrossRef]
- Pase, M.P.; Scholey, A.B.; Pipingas, A.; Kras, M.; Nolidin, K.; Gibbs, A.; Wesnes, K.; Stough, C. Cocoa polyphenols enhance positive mood states but not cognitive performance: A randomized, placebo-controlled trial. J. Psychopharmacol. 2013, 27, 451–458. [Google Scholar] [CrossRef]
- Rey, A. L’examen Clinique en Psychologie; Presses Universitaires de France: Paris, France, 1964. [Google Scholar]
- Lezak, M.D. Neuropsychological Assessment; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Whyte, A.R.; Williams, C.M. Effects of a single dose of a flavonoid-rich blueberry drink on memory in 8 to 10 y old children. Nutrition 2015, 31, 531–553. [Google Scholar] [CrossRef]
- Barfoot, K.L.; May, G.; Lamport, D.J.; Ricketts, J.; Riddell, P.M.; Williams, C.M. The effects of acute wild blueberry supplementation on the cognition of 7–10-year-old schoolchildren. Eur. J. Nutr. 2018, 58, 2911–2920. [Google Scholar] [CrossRef]
- Crawford, J.R.; Henry, J.D. The Positive and Negative Affect Schedule (PANAS); Construct validity, measurement properties and normative data in a large non-clinical sample. Br. J. Clin. Psychol. 2004, 43, 245–265. [Google Scholar] [CrossRef] [PubMed]
- Bond, A.; Lader, M. The use of analogue scales in rating subjective feelings. Br. J. Med. Psychol. 1974, 47, 211–218. [Google Scholar] [CrossRef]
- Hein, S.; Whyte, A.R.; Wood, E.; Rodriguez-Mateos, A.; Williams, C.M. Systematic review of the effects of blueberry on cognitive performance as we age. J. Gerontol. Ser. A 2019, 74, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.; Williams, C.M. A pilot dose–response study of the acute effects of haskap berry extract (Lonicera caerulea L.) on cognition, mood, and blood pressure in older adults. Eur. J. Nutr. 2018, 58, 3325–3334. [Google Scholar] [CrossRef] [PubMed]
- Datla, K.P.; Christidou, M.; Widmer, W.W.; Rooprai, H.K.; Dexter, D.T. Tissue distribution and neuroprotective effects of citrus flavonoid tangeretin in a rat model of Parkinson’s disease. Neuroreport 2001, 12, 3871–3875. [Google Scholar] [CrossRef] [PubMed]
- Passamonti, S.; Vrhovsek, U.; Vanzo, A.; Mattivi, F. Fast access of some grape pigments to the brain. J. Agric. Food Chem. 2005, 53, 7029–7034. [Google Scholar] [CrossRef]
- Milbury, P.E.; Kalt, W. Xenobiotic metabolism and berry flavonoid transport across the blood-brain barrier. J. Agric. Food Chem. 2010, 58, 3950–3956. [Google Scholar] [CrossRef]
- Fisher, N.D.; Hollenberg, N.K. Aging and vascular responses to flavanol-rich cocoa. J. Hypertens. 2006, 24, 1575–1578. [Google Scholar] [CrossRef]
- Crichton, G.E.; Elias, M.F.; Davey, A.; Alkerwi, A. Cardiovascular health and cognitive function: The maine-syracuse longitudinal study. PLoS ONE 2014, 9, e89317. [Google Scholar] [CrossRef]
- Nurk, E.; Refsum, H.; Drevon, C.A.; Tell, G.S.; Nygaard, H.A.; Engedal, K.; Smith, A.D. Intake of flavonoid-rich wine, tea, and chocolate by elderly men and women is associated with better cognitive test performance. J. Nutr. 2008, 139, 120–127. [Google Scholar] [CrossRef]
- Moreira, A.; Diógenes, M.J.; de Mendonça, A.; Lunet, N.; Barros, H. Chocolate consumption is associated with a lower risk of cognitive decline. J. Alzheimer’s Dis. 2016, 53, 85–93. [Google Scholar] [CrossRef]
- Tuenter, E.; Foubert, K.; Pieters, L. Mood components in cocoa and chocolate: The mood pyramid. Planta Med. 2018, 84, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Langer, S.; Marshall, L.J.; Day, A.J.; Morgan, M.R. Flavanols and methylxanthines in commercially available dark chocolate: A study of the correlation with nonfat cocoa solids. J. Agric. Food Chem. 2011, 59, 8435–8441. [Google Scholar] [CrossRef] [PubMed]
- Cova, I.; Leta, V.; Mariani, C.; Pantoni, L.; Pomati, S. Exploring cocoa properties: Is theobromine a cognitive modulator? Psychopharmacology 2019, 236, 561–572. [Google Scholar] [CrossRef] [PubMed]
- Ruxton, C.H.S. The impact of caffeine on mood, cognitive function, performance and hydration: A review of benefits and risks. Nutr. Bull. 2008, 33, 15–25. [Google Scholar] [CrossRef]
| Dark Chocolate (n = 49) | White Chocolate (n = 49) | p-Value | |
|---|---|---|---|
| Age (years) | 20.7 (0.18) | 20.6 (0.19) | 0.59 |
| Height (m) | 1.71 (0.01) | 1.71 (0.02) | 0.99 |
| Weight (kg) | 64.6 (2.1) | 64.9 (2.6) | 0.95 |
| BMI (kg/m2) | 21.95 (0.57) | 21.86 (0.59) | 0.91 |
| Sex (male: female) | 22:27 | 19:30 | 0.54 ^ |
| Baseline Pre-Consumption | Two Hours Post-Consumption | Interaction p-Value | |||||
|---|---|---|---|---|---|---|---|
| Dark | White | p-Value * | Dark | White | p-Value * | ||
| Immediate word span | 5.08 (0.28) | 5.29 (0.25) | 0.59 | 6.53 (0.27) | 4.94 (0.27) | <0.00001 * | 0.00005 * |
| Words learnt | 4.82 (0.38) | 5.45 (0.27) | 0.18 | 5.59 (0.32) | 4.14 (0.35) | 0.003 | 0.0004 * |
| Final Acquisition | 9.9 (0.3) | 10.7 (0.29) | 0.051 | 12.1 (0.26) | 9.1 (2.91) | <0.00001 * | <0.00001 * |
| Total Acquisition | 41.7 (1.2) | 45.1 (1.2) | 0.048 | 50.3 (1.1) | 38.7 (1.3) | <0.00001 | <0.00001 * |
| Proactive Interference | 0.14 (0.33) | 0.27 (0.29) | 0.779 | 0.02 (0.3) | 0.02 (0.3) | 0.999 | 0.852 |
| Retroactive Interference | 0.73 (0.32) | 1.45 (0.35) | 0.134 | 1.53 (0.31) | 1.67 (0.37) | 0.769 | 0.374 |
| Delayed Recall | 9.16 (0.39) | 9.29 (0.44) | 0.835 | 10.6 (0.41) | 7.41 (0.47) | <0.00001 * | <0.00001 * |
| Total Recall | 55.8 (1.6) | 59.4 (1.7) | 0.124 | 67.4 (1.5) | 51.1 (1.9) | <0.00001 * | <0.00001 * |
| Positive Affect ^ | 33.71 (1) | 33.78 (1) | 0.96 | 34.86 (0.1) | 35.43 (0.1) | 0.69 | 0.633 |
| Negative Affect ^ | 18 (0.65) | 17.59 (0.7) | 0.66 | 15.43 (0.7) | 15.44 (0.7) | 0.99 | 0.582 |
| Alertness ~ | 68.8 (1.9) | 62.9 (1.9) | 0.03 | 73 (2.5) | 67.2 (2.5) | 0.11 | 0.99 |
| Anxiety ~ | 34.2 (3.1) | 30.3 (3.1) | 0.38 | 41.4 (3.3) | 37.9 (3.3) | 0.44 | 0.95 |
| Contentment ~ | 68.8 (2) | 68 (2) | 0.77 | 69.9 (2.1) | 66.8 (2.1) | 0.3 | 0.34 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lamport, D.J.; Christodoulou, E.; Achilleos, C. Beneficial Effects of Dark Chocolate for Episodic Memory in Healthy Young Adults: A Parallel-Groups Acute Intervention with a White Chocolate Control. Nutrients 2020, 12, 483. https://doi.org/10.3390/nu12020483
Lamport DJ, Christodoulou E, Achilleos C. Beneficial Effects of Dark Chocolate for Episodic Memory in Healthy Young Adults: A Parallel-Groups Acute Intervention with a White Chocolate Control. Nutrients. 2020; 12(2):483. https://doi.org/10.3390/nu12020483
Chicago/Turabian StyleLamport, Daniel J., Eleni Christodoulou, and Christina Achilleos. 2020. "Beneficial Effects of Dark Chocolate for Episodic Memory in Healthy Young Adults: A Parallel-Groups Acute Intervention with a White Chocolate Control" Nutrients 12, no. 2: 483. https://doi.org/10.3390/nu12020483
APA StyleLamport, D. J., Christodoulou, E., & Achilleos, C. (2020). Beneficial Effects of Dark Chocolate for Episodic Memory in Healthy Young Adults: A Parallel-Groups Acute Intervention with a White Chocolate Control. Nutrients, 12(2), 483. https://doi.org/10.3390/nu12020483






