The Effect of an 8 Week Prescribed Exercise and Low-Carbohydrate Diet on Cardiorespiratory Fitness, Body Composition and Cardiometabolic Risk Factors in Obese Individuals: A Randomised Controlled Trial
Abstract
1. Introduction
2. Methods
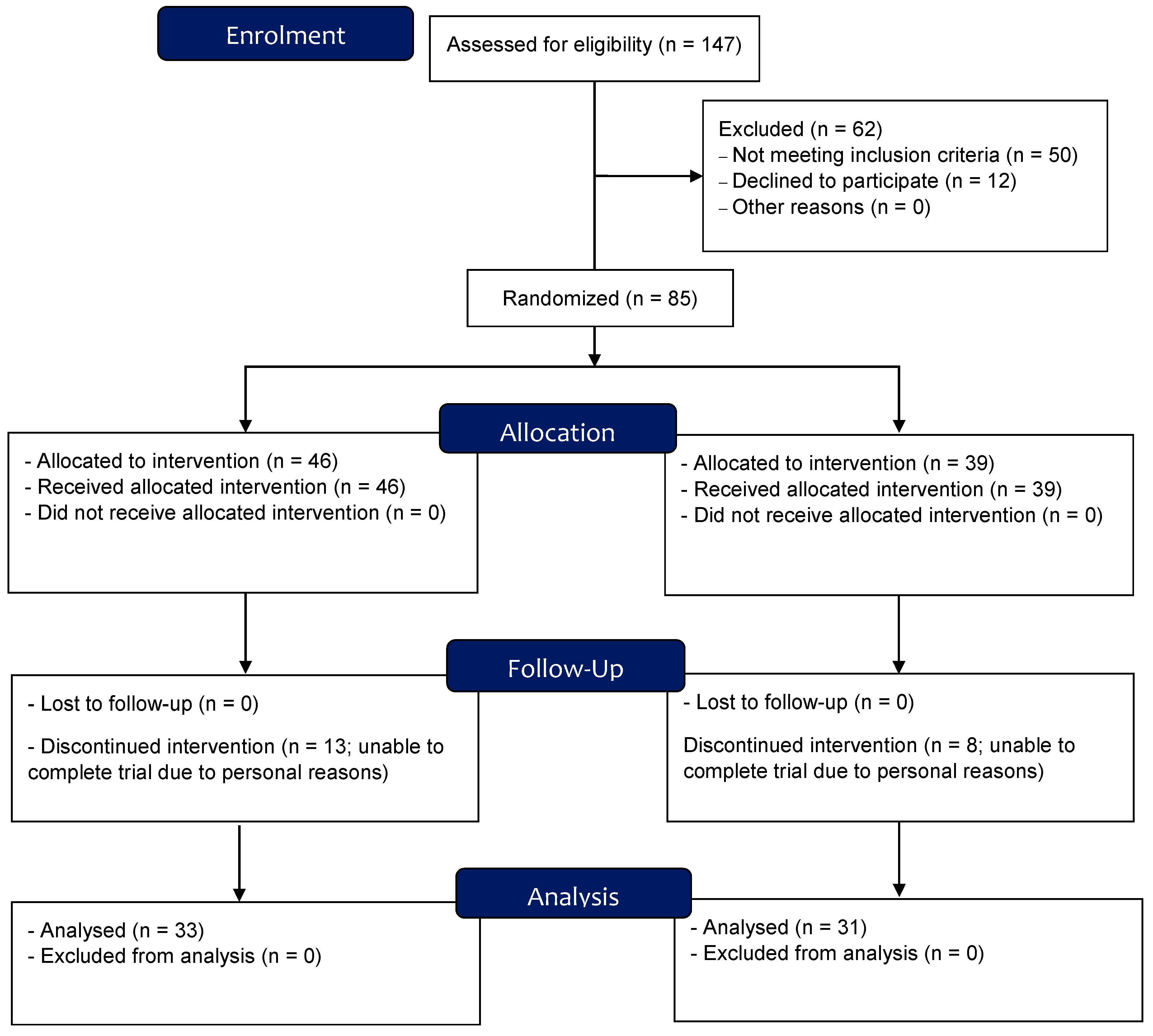
2.1. Participants
2.2. Study Design Overview
2.3. Dietary Intervention
2.4. Maximal Incremental Cycling Test
2.5. Body Composition Assessment
2.6. Blood Analysis
2.7. Resting Blood Pressure
3. Statistical Analysis
4. Results
4.1. Cardiorespiratory Fitness
4.2. Body Composition
4.3. Cardiometabolic Indices
4.4. Mixed Linear Regression Analysis
5. Discussion
5.1. Cardiorespiratory Fitness
5.2. Body Composition
5.3. Cardiometabolic Indices
5.4. Limitations and Issues of Concern
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Raynor, H.A.; Champagne, C.M. Position of the Academy of Nutrition and Dietetics: Interventions for the Treatment of Overweight and Obesity in Adults. J. Acad. Nutr. Diet. 2016, 116, 129–147. [Google Scholar] [CrossRef]
- Australian Bureau of Statistics: National Health Survey: First Results 2017-2018. Cat. No. 4364.0.55.001. ABS: Canberra. 2019. Available online: https://www.abs.gov.au/ (accessed on 15 December 2019).
- Ng, M.; Fleming, T.; Robinson, M.; Thomson, B.; Graetz, N.; Margono, C.; Mullany, E.C.; Biryukov, S.; Abbafati, C.; Abera, S.F.; et al. Regional, and national prevalence of overweight and obesity in children and adults during 1980–2013: A systematic analysis for the Global Burden of Disease Study 2013. Lancet 2014, 384, 766–781. [Google Scholar] [CrossRef]
- Walls, H.L.; Peeters, A.; Proietto, J.; McNeil, J.J. Public health campaigns and obesity—A critique. BMC Public Health 2011, 11, 136. [Google Scholar] [CrossRef] [PubMed]
- Dhatariya, K. Blood Ketones: Measurement, Interpretation, Limitations, and Utility in the Management of Diabetic Ketoacidosis. Rev. Diabet. Stud. RDS 2016, 13, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.S.; Noakes, M.; Keogh, J.B.; Clifton, P.M. Long-term effects of a low carbohydrate, low fat or high unsaturated fat diet compared to a no-intervention control. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Gjuladin-Hellon, T.; Davies, I.; Penson, P.; Amiri Baghbadorani, R. Effects of carbohydrate-restricted diets on low-density lipoprotein cholesterol levels in overweight and obese adults: A systematic review and meta-analysis. Nutr. Rev. 2019, 77, 161–180. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Wan, Y.; Yang, B.; Huggins, C.E.; Li, D. Effects of low-fat compared with high-fat diet on cardiometabolic indicators in people with overweight and obesity without overt metabolic disturbance: A systematic review and meta-analysis of randomised controlled trials. Br. J. Nutr. 2018, 119, 96–108. [Google Scholar] [CrossRef]
- Noakes, M.; Foster, P.R.; Keogh, J.B.; James, A.P.; Mamo, J.C.; Clifton, P.M. Comparison of isocaloric very low carbohydrate/high saturated fat and high carbohydrate/low saturated fat diets on body composition and cardiovascular risk. Nutr. Metab. 2006, 3, 7. [Google Scholar] [CrossRef]
- Tinsley, G.M.; Willoughby, D.S. Fat-Free Mass Changes During Ketogenic Diets and the Potential Role of Resistance Training. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 78–92. [Google Scholar] [CrossRef]
- Ding, J.; Xu, X.; Wu, X.; Huang, Z.; Kong, G.; Liu, J.; Huang, Z.; Liu, Q.; Li, R.; Yang, Z.; et al. Bone loss and biomechanical reduction of appendicular and axial bones under ketogenic diet in rats. Exp. Ther. Med. 2019, 17, 2503–2510. [Google Scholar] [CrossRef]
- Soeters, M.R.; Soeters, P.B.; Schooneman, M.G.; Houten, S.M.; Romijn, J.A. Adaptive reciprocity of lipid and glucose metabolism in human short-term starvation. Am. J. Physiol. Endocrinol. Metab. 2012, 303, E1397–E1407. [Google Scholar] [CrossRef] [PubMed]
- Crepaldi, G.; Romanato, G.; Tonin, P.; Maggi, S. Osteoporosis and body composition. J. Endocrinol. Invest. 2007, 30, 42–47. [Google Scholar] [PubMed]
- Janssen, I.; Heymsfield, S.B.; Ross, R. Low Relative Skeletal Muscle Mass (Sarcopenia) in Older Persons Is Associated with Functional Impairment and Physical Disability. J. Am. Geriatr. Soc. 2002, 50, 889–896. [Google Scholar] [CrossRef]
- Jabekk, P.T.; Moe, I.A.; Meen, H.D.; Tomten, S.E.; Høstmark, A.T. Resistance training in overweight women on a ketogenic diet conserved lean body mass while reducing body fat. Nutr. Metab. 2010, 7, 17. [Google Scholar] [CrossRef] [PubMed]
- Ramos, J.S.; Dalleck, L.S.; Borrani, F.; Beetham, K.S.; Wallen, M.P.; Clark, A.R.; Gomersall, S.; Keating, S.E.; Fassett, R.E.; Coombes, J.S. Low-Volume High-Intensity Interval Training Is Sufficient to Ameliorate the Severity of Metabolic Syndrome. Metab. Syndr. Relat. Disord. 2017, 15, 319–328. [Google Scholar] [CrossRef]
- Bishop, D.J.; Botella, J.; Genders, A.J.; Lee, M.J.; Saner, N.J.; Kuang, J.; Yan, X.; Granata, C. High-Intensity Exercise and Mitochondrial Biogenesis: Current Controversies and Future Research Directions. Physiology 2019, 34, 56–70. [Google Scholar] [CrossRef]
- You, T.; Arsenis, N.C.; Disanzo, B.L.; Lamonte, M.J. Effects of Exercise Training on Chronic Inflammation in Obesity. Sports Med. 2013, 43, 243–256. [Google Scholar] [CrossRef]
- Gomez-Ambrosi, J.; Catalán, V.; Rodríguez, A.; Andrada, P.; Ramírez, B.; Ibáñez, P.; Vila, N.; Romero, S.; Margall, M.A.; Gil, M.J.; et al. Increased cardiometabolic risk factors and inflammation in adipose tissue in obese subjects classified as metabolically healthy. Diabetes Care 2014, 37, 2813–2821. [Google Scholar] [CrossRef]
- Buemann, B.; Marckmann, P.; Christensen, N.J.; Astrup, A. The effect of ephedrine plus caffeine on plasma lipids and lipoproteins during a 4.2 MJ/day diet. Int. J. Obes. Relat. Metab. Disord. 1994, 18, 329–332. [Google Scholar]
- Lane, J.D.; Barkauskas, C.E.; Surwit, R.S.; Feinglos, M.N. Caffeine impairs glucose metabolism in type 2 diabetes. Diabetes Care 2004, 27, 2047–2048. [Google Scholar] [CrossRef][Green Version]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Eckert-Norton, M.; Kirk, S. Non-diabetic Hypoglycemia. J. Clin. Endocrinol. Metab. 2013, 98, 39A–40A. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.C.; Koceja, D.M.; Hamilton, E.J. A meta-analysis of the past 25 years of weight loss research using diet, exercise or diet plus exercise intervention. Int. J. Obes. Relat. Metab. Disord. 1997, 21, 941–947. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-Y. Statistical notes for clinical researchers: Assessing normal distribution (2) using skewness and kurtosis. Restor. Dent. Endod. 2013, 38, 52–54. [Google Scholar] [CrossRef] [PubMed]
- Rothman, K.J. No adjustments are needed for multiple comparisons. Epidemiology 1990, 1, 43–46. [Google Scholar] [CrossRef]
- Klement, R.J.; Frobel, T.; Albers, T.; Fikenzer, S.; Prinzhausen, J.; Kaemmerer, U. A pilot case study on the impact of a self-prescribed ketogenic diet on biochemical parameters and running performance in healthy and physically active individuals. Nutr. Med. 2013, 1, 10. [Google Scholar]
- Zajac, A.; Poprzecki, S.; Maszczyk, A.; Czuba, M.; Michalczyk, M.; Zydek, G. The effects of a ketogenic diet on exercise metabolism and physical performance in off-road cyclists. Nutrients 2014, 6, 2493–2508. [Google Scholar] [CrossRef]
- Brinkworth, G.D.; Noakes, M.; Clifton, P.M.; Buckley, J.D. Effects of a low carbohydrate weight loss diet on exercise capacity and tolerance in obese subjects. Obesity 2009, 17, 1916–1923. [Google Scholar] [CrossRef]
- Volek, J.S.; Noakes, T.; Phinney, S.D. Rethinking fat as a fuel for endurance exercise. Eur. J. Sport Sci. 2015, 15, 13–20. [Google Scholar] [CrossRef]
- Cox, P.J.; Kirk, T.; Ashmore, T.; Willerton, K.; Evans, R.; Smith, A.; Murray, A.J.; Stubbs, B.; West, J.; McLure, S.W.; et al. Nutritional Ketosis Alters Fuel Preference and Thereby Endurance Performance in Athletes. Cell Metab. 2016, 24, 256–268. [Google Scholar] [CrossRef]
- Cipryan, L.; Plews, D.; Ferretti, A.; Maffetone, P.; Laursen, P. Effects of a 4-week very low-carbohydrate diet on high-intensity interval training responses. J. Sports Sci. Med. 2018, 17, 259–268. [Google Scholar]
- Helge, J.W.; Watt, P.W.; Richter, E.A.; Rennie, M.J.; Kiens, B. Fat utilization during exercise: Adaptation to a fat-rich diet increases utilization of plasma fatty acids and very low density lipoprotein-triacylglycerol in humans. J. Physiol. 2001, 537, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Lakoski, S.G.; Barlow, C.E.; Farrell, S.W.; Berry, J.D.; Morrow, J.R., Jr.; Haskell, W.L. Impact of Body Mass Index, Physical Activity, and Other Clinical Factors on Cardiorespiratory Fitness (from the Cooper Center Longitudinal Study). Am. J. Cardiol. 2011, 108, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Edvardsen, E.; Hansen, B.H.; Holme, I.M.; Dyrstad, S.M.; Anderssen, S.A. Reference Values for Cardiorespiratory Response and Fitness on the Treadmill in a 20- to 85-Year-Old Population. Chest 2013, 144, 241–248. [Google Scholar] [CrossRef]
- Jin, C.-H.; Rhyu, H.-S.; Kim, J.Y. The effects of combined aerobic and resistance training on inflammatory markers in obese men. J. Exerc. Rehabil. 2018, 14, 660–665. [Google Scholar] [CrossRef]
- Lima, L.G.; Bonardi, J.T.M.; Campos, G.O.; Bertani, R.F.; Scher, L.M.L.; Moriguti, J.C.; Ferriolli, E.; Lima, N.K.C. Combined aerobic and resistance training: Are there additional benefits for older hypertensive adults? Clinics 2017, 72, 363–369. [Google Scholar] [CrossRef]
- Hashimoto, Y.; Fukuda, T.; Oyabu, C.; Tanaka, M.; Asano, M.; Yamazaki, M.; Fukui, M. Impact of low-carbohydrate diet on body composition: Meta-analysis of randomized controlled studies. Obes. Rev. 2016, 17, 499–509. [Google Scholar] [CrossRef]
- Masino, S.A.; Rho, J.M. Mechanisms of ketogenic diet action. Epilepsia 2010, 51, 85. [Google Scholar] [CrossRef]
- Fox, C.S.; Massaro, J.M.; Hoffmann, U.; Pou, K.M.; Maurovich-Horvat, P.; Liu, C.Y.; Vasan, R.S.; Murabito, J.M.; Meigs, J.B.; Cupples, L.A.; et al. Abdominal visceral and subcutaneous adipose tissue compartments: Association withmetabolic risk factors in the Framingham Heart Study. Circulation 2007, 116, 39–48. [Google Scholar] [CrossRef]
- Bank, A.J.; Wang, H.; Holte, J.E.; Mullen, K.; Shammas, R.; Kubo, S.H. Contribution of collagen, elastin, and smooth muscle to in vivo human brachial artery wall stress and elastic modulus. Circulation 1996, 94, 3263–3270. [Google Scholar] [CrossRef]
- Ritchie, S.A.; Connell, J.M. The link between abdominal obesity, metabolic syndrome and cardiovascular disease. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 319–326. [Google Scholar] [CrossRef]
- Hoenig, M.R.; Cowin, G.; Buckley, R.; McHenery, C.; Coulthard, A. Low density lipoprotein cholesterol is inversely correlated with abdominal visceral fat area: A magnetic resonance imaging study. Lipids Health Dis. 2011, 10, 12. [Google Scholar] [CrossRef]
- Macor, C.; Ruggeri, A.; Mazzonetto, P.; Federspil, G.; Cobelli, C.; Vettor, R. Visceral adipose tissue impairs insulin secretion and insulin sensitivity but not energy expenditure in obesity. Metabolism 1997, 46, 123–129. [Google Scholar] [CrossRef]
- Johnstone, A.M.; Horgan, G.W.; Murison, S.D.; Bremner, D.M.; Lobley, G.E. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am. J. Clin. Nutr. 2008, 87, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Cornier, M.A.; Draznin, B. Low-Carbohydrate Diets in the Treatment of The Metabolic Syndrome. In The Metabolic Syndrome: Pharmacology and Clinical Aspects; Springer: Vienna, Austria, 2013; pp. 87–98. [Google Scholar]
- Serra-Prat, M.; Lorenzo, I.; Palomera, E.; Yébenes, J.C.; Campins, L.; Cabré, M. Intracellular Water Content in Lean Mass is Associated with Muscle Strength, Functional Capacity, and Frailty in Community-Dwelling Elderly Individuals. A Cross-Sectional Study. Nutrients 2019, 11, 661. [Google Scholar] [CrossRef] [PubMed]
- Boison, D. New insights into the mechanisms of the ketogenic diet. Curr. Opin. Neurol. 2017, 30, 187–192. [Google Scholar] [CrossRef]
- Kinzig, K.P.; Honors, M.A.; Hargrave, S.L. Insulin sensitivity and glucose tolerance are altered by maintenance on a ketogenic diet. Endocrinology 2010, 151, 3105–3114. [Google Scholar] [CrossRef]
- Bouxsein, M.L. Determinants of skeletal fragility. Best Pract. Res. Clin. Rheumatol. 2005, 19, 897–911. [Google Scholar] [CrossRef]
- Wood, R.J.; Gregory, S.M.; Sawyer, J.; Milch, C.M.; Matthews, T.D.; Headley, S.A. Preservation of fat-free mass after two distinct weight loss diets with and without progressive resistance exercise. Metab. Syndr. Relat. Disord. 2012, 10, 167–174. [Google Scholar] [CrossRef]
- Murton, A.J.; Greenhaff, P.L. Resistance exercise and the mechanisms of muscle mass regulation in humans: Acute effects on muscle protein turnover and the gaps in our understanding of chronic resistance exercise training adaptation. Int. J. Biochem. Cell Biol. 2013, 45, 2209–2214. [Google Scholar] [CrossRef]
- Balducci, S.; Zanuso, S.; Nicolucci, A.; Fernando, F.; Cavallo, S.; Cardelli, P.; Fallucca, S.; Alessi, E.; Letizia, C.; Jimenez, A.; et al. Anti-inflammatory effect of exercise training in subjects with type 2 diabetes and the metabolic syndrome is dependent on exercise modalities and independent of weight loss. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 608–617. [Google Scholar] [CrossRef]
- Oh, E.G.; Bang, S.Y.; Hyun, S.S.; Kim, S.H.; Chu, S.H.; Jeon, J.Y.; Im, J.A.; Lee, M.K.; Lee, J.E. Effects of a 6-month lifestyle modification intervention on the cardiometabolic risk factors and health-related qualities of life in women with metabolic syndrome. Metabolism 2010, 59, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Gibala, M.J.; Little, J.P.; Macdonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 2012, 590, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Ramos, J.S.; Dalleck, L.C.; Tjonna, A.E.; Beetham, K.S.; Coombes, J.S. The impact of high-intensity interval training versus moderate-intensity continuous training on vascular function: A systematic review and meta-analysis. Sports Med. 2015, 45, 679–692. [Google Scholar] [CrossRef] [PubMed]
- Burgomaster, K.A.; Howarth, K.R.; Phillips, S.M.; Rakobowchuk, M.; Macdonald, M.J.; McGee, S.L.; Gibala, M.J. Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. J. Physiol. 2008, 586, 151–160. [Google Scholar] [CrossRef]
- Kaptoge, S.; Di Angelantonio, E.; Lowe, G.; Pepys, M.B.; Thompson, S.G.; Collins, R.; Danesh, J.; Emerging Risk Factors Collaboration. C-reactive protein concentration and risk of coronary heart disease, stroke, and mortality: An individual participant meta-analysis. Lancet 2010, 375, 132–140. [Google Scholar]
- Smidowicz, A.; Regula, J. Effect of nutritional status and dietary patterns on human serum C-reactive protein and interleukin-6 concentrations. Adv. Nutr. 2015, 6, 738–747. [Google Scholar] [CrossRef]
- Taggart, A.K.; Kero, J.; Gan, X.; Cai, T.Q.; Cheng, K.; Ippolito, M.; Ren, N.; Kaplan, R.; Wu, K.; Wu, T.J.; et al. (D)-beta-Hydroxybutyrate inhibits adipocyte lipolysis via the nicotinic acid receptor PUMA-G. J. Biol. Chem. 2005, 280, 26649–26652. [Google Scholar] [CrossRef]
- Rock, C.L.; Flatt, S.W.; Pakiz, B.; Barkai, H.S.; Heath, D.D.; Krumhar, K.C. Randomized clinical trial of portion-controlled prepackaged foods to promote weight loss. Obesity 2016, 24, 1230–1237. [Google Scholar] [CrossRef]
- Retterstol, K.; Svendsen, M.; Narverud, I.; Holven, K.B. Effect of low carbohydrate high fat diet on LDL cholesterol and gene expression in normal-weight, young adults: A randomized controlled study. Atherosclerosis 2018, 279, 52–61. [Google Scholar] [CrossRef]
- Noto, H.; Goto, A.; Tsujimoto, T.; Noda, M. Low-carbohydrate diets and all-cause mortality: A systematic review and meta-analysis of observational studies. PLoS ONE 2013, 8, e55030. [Google Scholar] [CrossRef]
- Seidelmann, S.B.; Claggett, B.; Cheng, S.; Henglin, M.; Shah, A.; Steffen, L.; Folsom, A.; Rimm, E.B.; Willett, W.; Solomon, S. Dietary carbohydrate intake and mortality: A prospective cohort study and meta-analysis. Lancet Public Health 2018, 3, e419–e428. [Google Scholar] [CrossRef]
- Dehghan, M.; Mente, A.; Zhang, X.; Swaminathan, S.; Li, W.; Mohan, V.; Iqbal, R.; Kumar, R.; Wentzel-Viljoen, E.; Rosengren, A.; et al. Prospective Urban Rural Epidemiology (PURE) study investigators. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): A prospective cohort study. Lancet 2017, 390, 2050–2062. [Google Scholar] [CrossRef]
| ALL | EX-CO | EX-LC | p Value | |
|---|---|---|---|---|
| (n =64) | (n = 33) | (n = 31) | ||
| Participant Characteristics | ||||
| Age (years) | 35 ± 9 | 34 ± 8 | 35 ± 6 | 0.45 |
| Height (m) | 1.74 ± 0.3 | 1.75 ± 0.5 | 1.77 ± 1.1 | 0.16 |
| Weight (kg) | 87 ± 17 | 84 ± 16 | 86 ± 15 | 0.20 |
| BMI (kg.m−2) | 30.3 ± 3 | 30.8 ± 4 | 31.2 ± 3 | 0.17 |
| Male Sex (%) | 43 | 38 | 41 | 0.25 |
| All | Time Effect | EX-CO | EX-LC | Condition Effect | ||||
|---|---|---|---|---|---|---|---|---|
| Baseline | Change | p | Baseline | Change | Baseline | Change | p | |
| Cardiorespiratory Fitness | ||||||||
| VO2peak (mL.kg−1.min−1) | 28.1 ± 6.3 | 2.4 ± 3.7 | 0.001 | 28.9 ± 6.3 | 1.7 ± 2.9 | 27.7 ± 6 | 3.0 ± 4.5 | 0.002 |
| Blood Biomarkers | ||||||||
| Total cholesterol (mmol/L) | 4.9 ± 0.8 | −0.3 ± 0.5 | 0.001 | 4.8 ± 0.8 | 0.0 ± 0.5 | 4.9 ± 1.0 | −0.3 ± 0.6 | 0.830 |
| HDL (mmol/L) | 1.3 ± 0.4 | 0.0 ± 0.4 | 0.230 | 1.2 ± 0.3 | 0.0 ± 0.1 | 1.4 ± 0.4 | 0.0 ± 0.2 | 0.260 |
| LDL (mmol/L) | 2.9 ± 0.9 | 0.2 ± 0.5 | 0.092 | 2.9 ± 0.8 | 0.0 ± 0.6 | 2.9 ± 0.9 | 0.0 ± 0.5 | 0.622 |
| Triglycerides (mmol/L) | 1.4 ± 0.7 | −0.3 ± 0.6 | 0.003 | 1.3 ± 0.5 | 0.0 ± 0.5 | 1.4 ± 0.9 | −0.4 ± 0.7 | 0.045 |
| Fasting glucose (mmol/L) | 5.5 ± 0.4 | −0.2 ± 0.3 | 0.005 | 5.4 ± 04 | 0.0 ± 0.4 | 5.6 ± 0.4 | −0.2 ± 0.4 | 0.027 |
| CRP (mmol/L) | 1.7 ± 2.3 | −0.6 ± 1.6 | 0.018 | 1.4 ± 2.2 | −0.2 ± 1.3 | 2.0 ± 2.5 | −0.9 ± 1.9 | 0.039 |
| Adiponectin (ug/L) | 23.0 ± 15 | −0.8 ± 14 | 0.911 | 20.6 ± 14 | 1.3 ± 18 | 25.9 ± 15 | −1.2 ± 9.5 | 0.417 |
| Blood Pressure | ||||||||
| Brachial SBP (mmHg) | 124 ± 10 | −3.6 ± 6 | 0.004 | 121 ± 4 | −3.3 ± 10 | 123 ± 7 | −3.7 ± 7 | 0.800 |
| Brachial DBP (mmHg) | 82 ± 8 | −3.5 ± 11 | 0.009 | 80 ± 7 | −1.0 ±8 | 83 ± 9 | −4.4 ± 7 | 0.092 |
| Body Composition | ||||||||
| Body weight (Kg) | 83.7 ± 18 | −3.8 ± 21 | 0.001 | 84.6 ± 13 | −1.8 ± 2.5 | 87.8 ± 16 | −4.4 ± 4 | 0.007 |
| Total body fat (%) | 40.5 ± 6.6 | −2.4 ± 5.7 | 0.001 | 40.0 ± 7.3 | −1.3 ± 1.4 | 40.9 ± 6.0 | −2.0 ± 2.0 | 0.090 |
| Visceral adipose tissue (cm3) | 114 ± 34 | −15 ± 24 | 0.001 | 116 ± 39 | −11 ± 15 | 116 ± 28 | −13 ± 14 | 0.764 |
| Lean muscle mass (cm3) | 47,549 ± 10,366 | −190 ± 2460 | 0.030 | 46,788 ± 9631 | 143 ± 976 | 48,106 ± 11,017 | −854 ± 1670 | 0.020 |
| Fat mass index (kg.m−2) | 12.7 ± 5.4 | −0.9 ± 0.9 | 0.001 | 13.1 ± 7.2 | −0.6 ± 0.7 | 12.3 ± 2.8 | −1.1 ± 0.9 | 0.013 |
| Total bone mineral density (g/cm2) | 1.106 ± 0.06 | 0.005 ± 0.00 | 0.025 | 1.116 ± 0.07 | 0.006 ± 0.01 | 1.102 ± 0.05 | 0.003 ± 0.00 | 0.659 |
| b | 95% CI | R | R2 | p | |
|---|---|---|---|---|---|
| Cardiorespiratory Fitness | |||||
| VO2peak mL.kg−1.min−1 | 2.54 | −1.78 to 6.87 | 0.252 | 0.064 | 0.026 |
| Blood Biomarkers | |||||
| Total cholesterol (mmol/L) | −0.33 | −0.73 to 0.44 | 0.068 | 0.015 | 0.618 |
| HDL (mmol/L) | −1.33 | −0.40 to 0.15 | 0.123 | 0.015 | 0.368 |
| LDL (mmol/L) | −0.10 | −0.47 to 0.67 | 0.048 | 0.002 | 0.724 |
| Triglycerides (mmol/L) | −0.21 | −0.86 to 0.50 | 0.190 | 0.036 | 0.161 |
| Fasting glucose (mmol/L) | −0.28 | −0.70 to 0.40 | 0.251 | 0.064 | 0.042 |
| CRP (mmol/L) | −0.97 | −0.56 to 1.58 | 0.245 | 0.060 | 0.041 |
| Adiponectin (ug/L) | −3.20 | −2.81 to 17.30 | 0.090 | 0.008 | 0.505 |
| Blood Pressure | |||||
| Brachial SBP (mmHg) | −0.94 | −6.96 to 5.07 | 0.040 | 0.002 | 0.755 |
| Brachial DBP (mmHg) | −1.03 | −6.40 to 4.33 | 0.050 | 0.002 | 0.701 |
| Body Composition | |||||
| Total body fat (%) | −1.52 | −0.35 to −2.68 | 0.317 | 0.101 | 0.011 |
| Visceral adipose tissue (cm3) | −23,779 | −3023 to −44,535 | 0.286 | 0.082 | 0.025 |
| Lean muscle mass (cm3) | −7081 | −250 to −13,913 | 0.261 | 0.068 | 0.042 |
| Fat mass index (kg.m−2) | −0.90 | −0.35 to −1.46 | 0.384 | 0.148 | 0.002 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perissiou, M.; Borkoles, E.; Kobayashi, K.; Polman, R. The Effect of an 8 Week Prescribed Exercise and Low-Carbohydrate Diet on Cardiorespiratory Fitness, Body Composition and Cardiometabolic Risk Factors in Obese Individuals: A Randomised Controlled Trial. Nutrients 2020, 12, 482. https://doi.org/10.3390/nu12020482
Perissiou M, Borkoles E, Kobayashi K, Polman R. The Effect of an 8 Week Prescribed Exercise and Low-Carbohydrate Diet on Cardiorespiratory Fitness, Body Composition and Cardiometabolic Risk Factors in Obese Individuals: A Randomised Controlled Trial. Nutrients. 2020; 12(2):482. https://doi.org/10.3390/nu12020482
Chicago/Turabian StylePerissiou, Maria, Erika Borkoles, Kent Kobayashi, and Remco Polman. 2020. "The Effect of an 8 Week Prescribed Exercise and Low-Carbohydrate Diet on Cardiorespiratory Fitness, Body Composition and Cardiometabolic Risk Factors in Obese Individuals: A Randomised Controlled Trial" Nutrients 12, no. 2: 482. https://doi.org/10.3390/nu12020482
APA StylePerissiou, M., Borkoles, E., Kobayashi, K., & Polman, R. (2020). The Effect of an 8 Week Prescribed Exercise and Low-Carbohydrate Diet on Cardiorespiratory Fitness, Body Composition and Cardiometabolic Risk Factors in Obese Individuals: A Randomised Controlled Trial. Nutrients, 12(2), 482. https://doi.org/10.3390/nu12020482





