A Fermented Milk Product with B. lactis CNCM I-2494 and Lactic Acid Bacteria Improves Gastrointestinal Comfort in Response to a Challenge Diet Rich in Fermentable Residues in Healthy Subjects
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
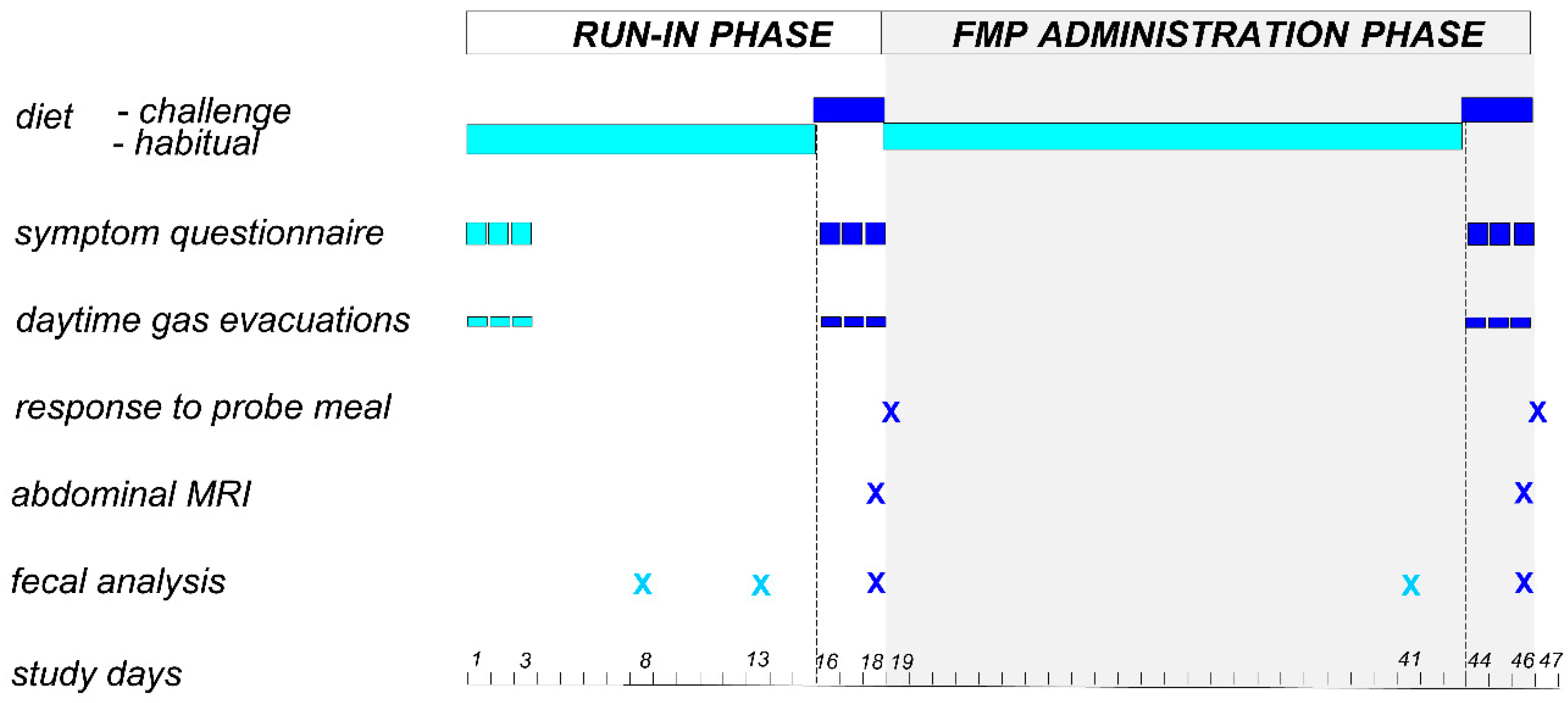
2.2. Study Design
2.3. Challenge Diet
2.4. Study Product
2.5. Main Outcomes
2.5.1. Daily Symptoms Questionnaire
2.5.2. Number of Anal Gas Evacuations
2.5.3. Response to a Probe Meal
2.6. Exploratory Outcomes
2.6.1. Colonic Gas Content
2.6.2. Fecal Microbiota Analysis
2.7. Statistical Analysis
2.7.1. Clinical Parameters
2.7.2. Gut Microbiota
3. Results
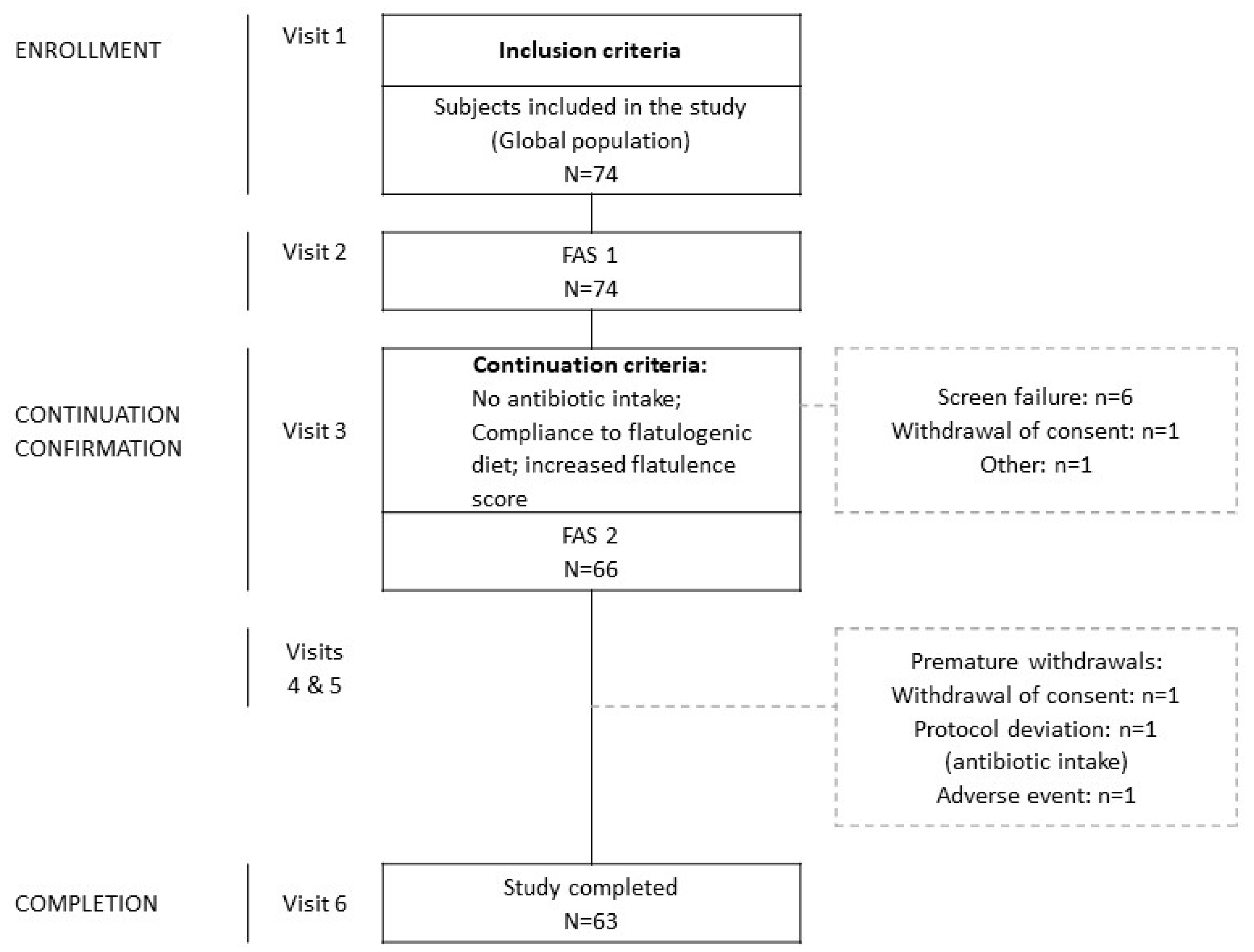
3.1. Demographics and Compliance to Study Procedures
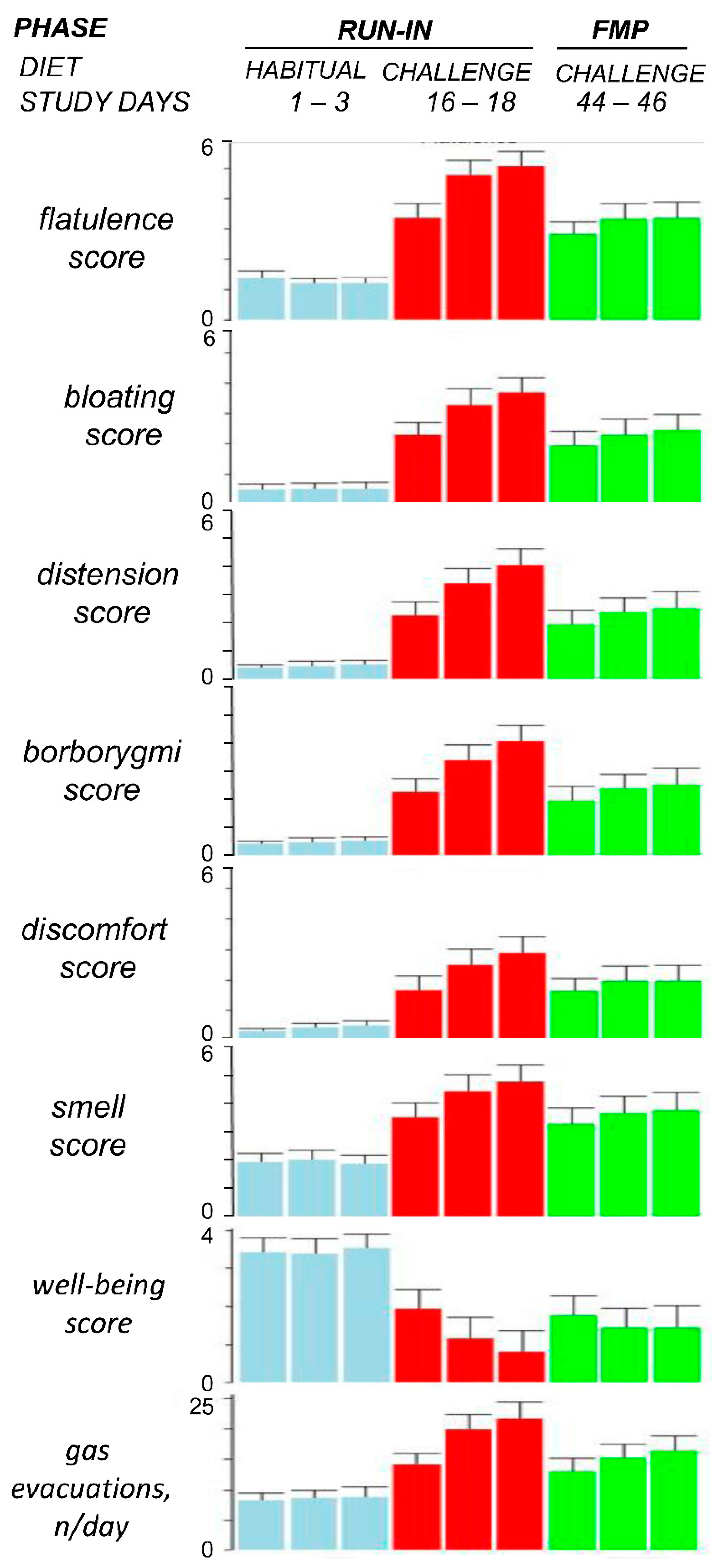
3.2. Effect of the Flatulogenic Diet during the Run-in Phase
3.3. Effect of the Fermented Milk Product Consumption on the Tolerance of the Flatulogenic Diet
3.4. Exploratory Outcomes
3.4.1. Colonic Gas Volume
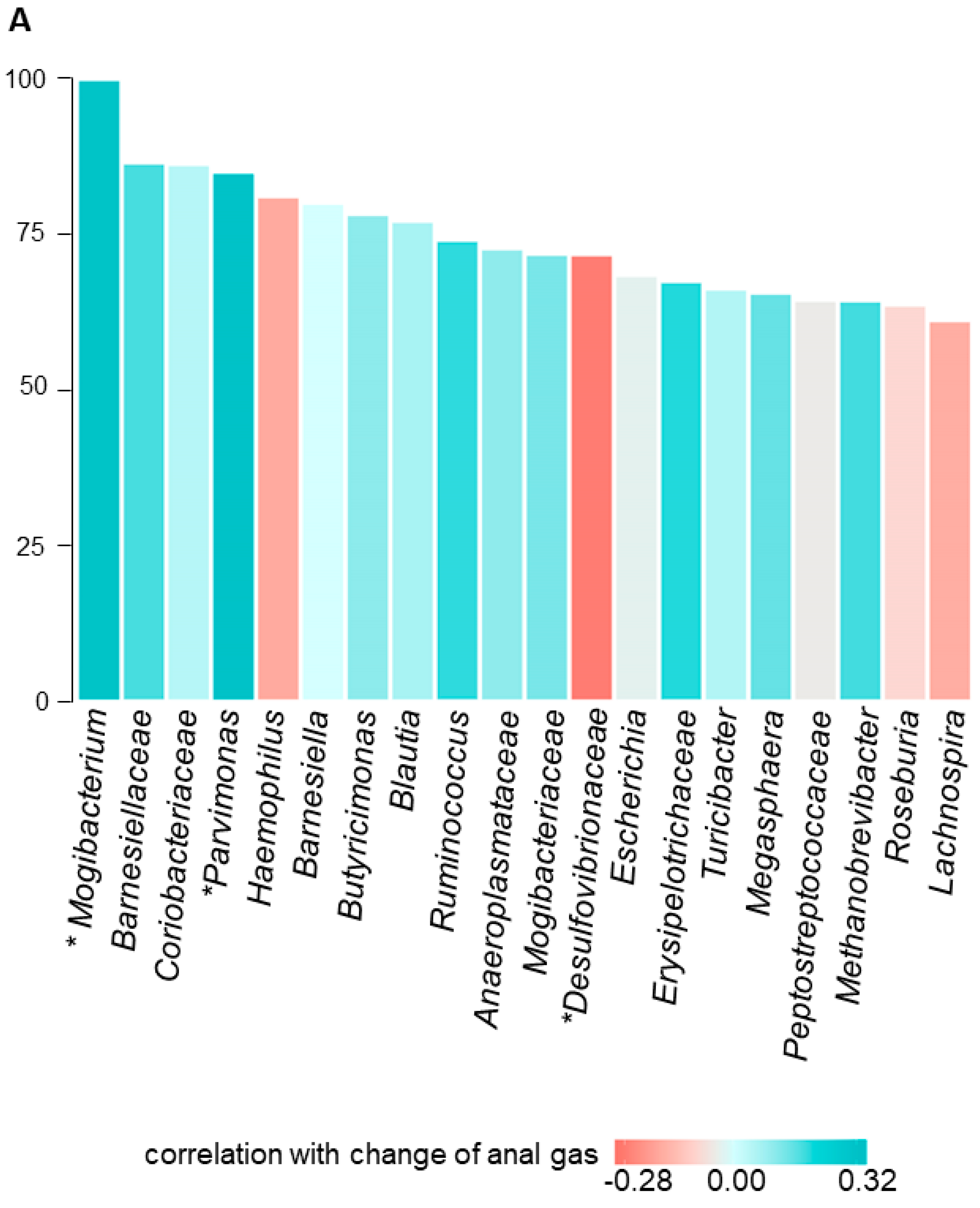
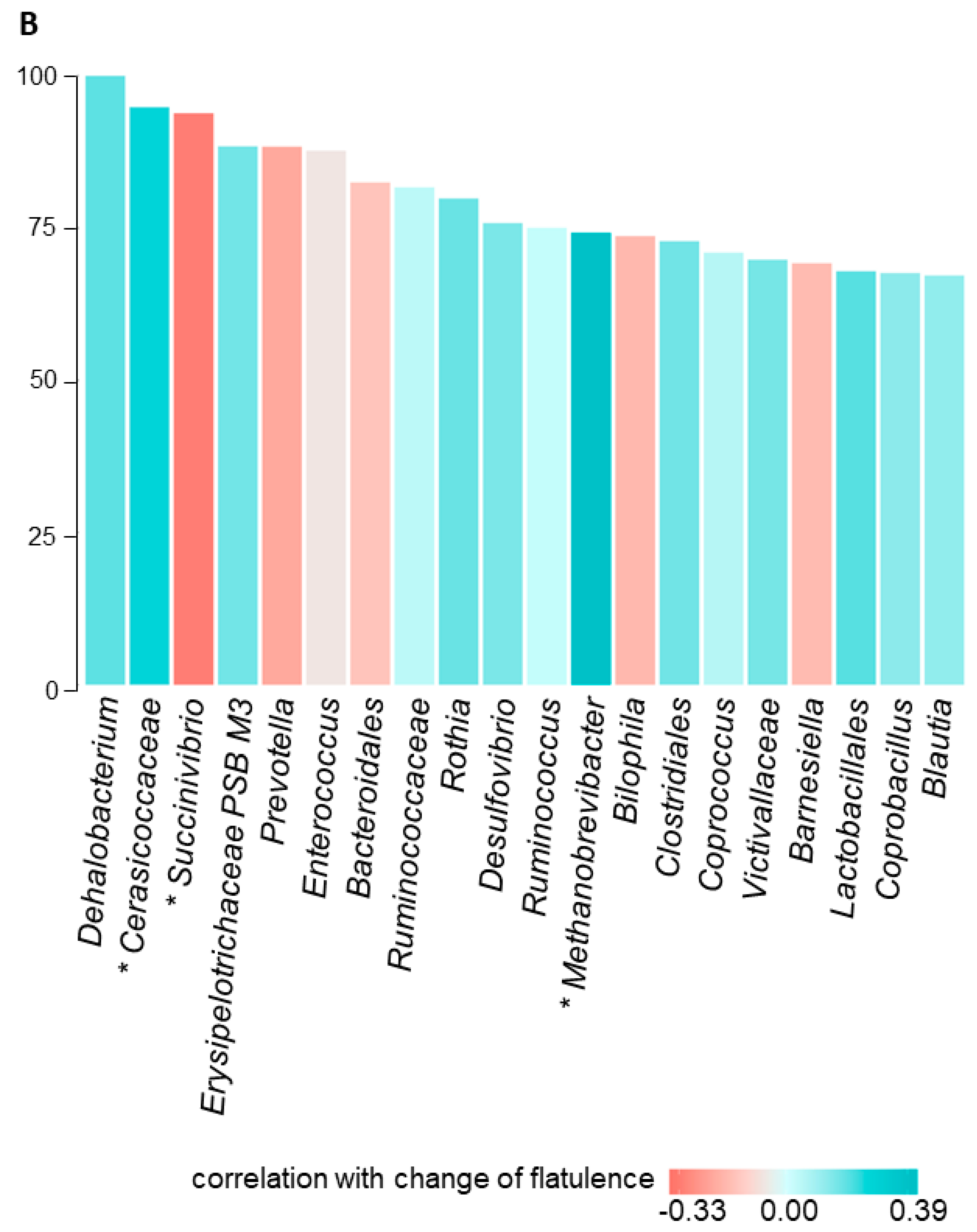
3.4.2. Fecal Microbiota
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jiang, X.; Locke, G.R.; Choung, R.S.; Zinsmeister, A.R.; Schleck, C.D.; Talley, N.J. Prevalence and risk factors for abdominal bloating and visible distention: A population-based study. Gut 2008, 57, 756–763. [Google Scholar] [CrossRef] [PubMed]
- Tielemans, M.M.; Jaspers Focks, J.; van Rossum, L.G.M.; Eikendal, T.; Jansen, J.B.M.J.; Laheij, R.J.F.; van Oijen, M.G.H. Gastrointestinal Symptoms are Still Prevalent and Negatively Impact Health-Related Quality of Life: A Large Cross-Sectional Population Based Study in The Netherlands. PLoS ONE 2013, 8, e69876. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Lee, O.Y.; Naliboff, B.; Schmulson, M.; Mayer, E.A. Sensation of bloating and visible abdominal distension in patients with irritable bowel syndrome. Am. J. Gastroenterol. 2001, 96, 3341–3347. [Google Scholar] [CrossRef] [PubMed]
- Malagelada, J.-R.; Accarino, A.; Azpiroz, F. Bloating and abdominal distension: Old misconceptions and current knowlege. Am. J. Gastroenterol. 2017, 112, 1221. [Google Scholar] [CrossRef] [PubMed]
- Azpiroz, F. Intestinal gas. In Pathophysiology, Diagnosis, Management; Feldman, M., Friedman, L.S., Brand, L.J., Eds.; Elsevier: Philadelphia, PA, USA, 2015; Volume 10, pp. 242–250. [Google Scholar]
- Grimble, G. Fibre, fermentation, flora, and flatus. Gut 1989, 30, 6–13. [Google Scholar] [CrossRef][Green Version]
- Steggerda, F.R. Gastrointestinal gas following food consumption. Ann. N. Y. Acad. Sci. 1968, 150, 57–66. [Google Scholar] [CrossRef]
- Wagner, J.R.; Carson, J.F.; Becker, R.; Gumbmann, M.R.; Danhof, I.E. Comparative Flatulence Activity of Beans and Bean Fractions for Man and the Rat. J. Nutr. 1977, 107, 680–689. [Google Scholar] [CrossRef]
- Manichanh, C.; Eck, A.; Varela, E.; Roca, J.; Clemente, J.C.; Gonzalez, A.; Knights, D.; Knight, R.; Estrella, S.; Hernandez, C.; et al. Anal gas evacuation and colonic microbiota in patients with flatulence: Effect of diet. Gut 2014, 63, 401–408. [Google Scholar] [CrossRef]
- Kaoutari, A.E.; Armougom, F.; Gordon, J.I.; Raoult, D.; Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 2013, 11, 497. [Google Scholar] [CrossRef]
- Frias, J.; Vidal-Valverde, C. Alpha-Galactosides: Antinutritional Factors or Functional Ingredients? Crit. Rev. Food Sci. Nutr. 2008, 48, 301–316. [Google Scholar] [CrossRef]
- Guyonnet, D.; Schlumberger, A.; Mhamdi, L.; Jakob, S.; Chassany, O. Fermented milk containing Bifidobacterium lactis DN-173 010 improves gastrointestinal well-being and digestive symptoms in women reporting minor digestive symptoms: A randomised, double-blind, parallel, controlled study. Br. J. Nutr. 2009, 102, 1654–1662. [Google Scholar] [CrossRef] [PubMed]
- Guyonnet, D.; Woodcock, A.; Stefani, B.; Trevisan, C.; Hall, C. Fermented milk containing Bifidobacterium lactis DN-173 010 improved self-reported digestive comfort amongst a general population of adults. A randomized, open-label, controlled, pilot study. J. Dig. Dis. 2009, 10, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Marteau, P.; Guyonnet, D.; Lafaye de Micheaux, P.; Gelu, S. A randomized, double-blind, controlled study and pooled analysis of two identical trials of fermented milk containing probiotic Bifidobacterium lactis CNCM I-2494 in healthy women reporting minor digestive symptoms. Neurogastroenterol. Motil. 2013, 25, 331–e252. [Google Scholar] [CrossRef] [PubMed]
- Marteau, P.; Le Neve, B.; Quinquis, L.; Pichon, C.; Whorwell, P.J.; Guyonnet, D. Consumption of a Fermented Milk Product Containing Bifidobacterium lactis CNCM I-2494 in Women Complaining of Minor Digestive Symptoms: Rapid Response Which Is Independent of Dietary Fibre Intake or Physical Activity. Nutrients 2019, 11, 92. [Google Scholar] [CrossRef]
- Agrawal, A.; Houghton, L.A.; Morris, J.; Reilly, B.; Guyonnet, D.; Goupil Feuillerat, N.; Schlumberger, A.; Jakob, S.; Whorwell, P.J. Clinical trial: The effects of a fermented milk product containing Bifidobacterium lactis DN-173 010 on abdominal distension and gastrointestinal transit in irritable bowel syndrome with constipation. Aliment. Pharmacol. Ther. 2009, 29, 104–114. [Google Scholar] [CrossRef]
- Guyonnet, D.; Chassany, O.; Ducrotte, P.; Picard, C.; Mouret, M.; Mercier, C.H.; Matuchansky, C. Effect of a fermented milk containing Bifidobacterium animalis DN-173 010 on the health-related quality of life and symptoms in irritable bowel syndrome in adults in primary care: A multicentre, randomized, double-blind, controlled trial. Aliment. Pharmacol. Ther. 2007, 26, 475–486. [Google Scholar] [CrossRef]
- Le Nevé, B.; Derrien, M.; Tap, J.; Brazeilles, R.; Cools Portier, S.; Guyonnet, D.; Ohman, L.; Störsrud, S.; Törnblom, H.; Simrén, M. Fasting breath H2 and gut microbiota metabolic potential are associated with the response to a fermented milk product in irritable bowel syndrome. PLoS ONE 2019, 14, e0214273. [Google Scholar] [CrossRef]
- Barba, E.; Burri, E.; Accarino, A.; Cisternas, D.; Quiroga, S.; Monclus, E.; Navazo, I.; Malagelada, J.R.; Azpiroz, F. Abdomino-thoracic mechanisms of functional abdominal distension and correction by biofeedback. Gastroenterology 2015, 148, 732–738. [Google Scholar] [CrossRef]
- Burri, E.; Barba, E.; Huaman, J.W.; Cisternas, D.; Accarino, A.; Soldevilla, A.; Malagelada, J.R.; Azpiroz, F. Mechanisms of postprandial abdominal bloating and distension in functional dyspepsia. Gut 2014, 63, 395–400. [Google Scholar] [CrossRef]
- Malagelada, C.; Drozdzal, M.; Segui, S.; Mendez, S.; Vitria, J.; Radeva, P.; Santos, J.; Accarino, A.; Malagelada, J.R.; Azpiroz, F. Classification of functional bowel disorders by objective physiological criteria based on endoluminal image analysis. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G413–G419. [Google Scholar] [CrossRef]
- Huaman, J.-W.; Mego, M.; Manichanh, C.; Cañellas, N.; Cañueto, D.; Segurola, H.; Jansana, M.; Malagelada, C.; Accarino, A.; Vulevic, J.; et al. Effects of Prebiotics vs a Diet Low in Fodmaps in Patients with Functional Gut Disorder. Gastroenterology 2018. [Google Scholar] [CrossRef] [PubMed]
- Mego, M.; Accarino, A.; Tzortzis, G.; Vulevic, J.; Gibson, G.; Guarner, F.; Azpiroz, F. Colonic gas homeostasis: Mechanisms of adaptation following HOST-G904 galactooligosaccharide use in humans. Neurogastroenterol. Mot. 2017, 29, e13080. [Google Scholar] [CrossRef] [PubMed]
- Pascal, V.; Pozuelo, M.; Borruel, N.; Casellas, F.; Campos, D.; Santiago, A.; Martinez, X.; Varela, E.; Sarrabayrouse, G.; Machiels, K.; et al. A microbial signature for Crohn’s disease. Gut 2017, 66, 813. [Google Scholar] [CrossRef] [PubMed]
- Serra, J.; Azpiroz, F.; Malagelada, J.R. Intestinal gas dynamics and tolerance in humans. Gastroenterology 1998, 115, 542–550. [Google Scholar] [CrossRef]
- Bendezu, R.A.; Mego, M.; Monclus, E.; Merino, X.; Accarino, A.; Malagelada, J.R.; Navazo, I.; Azpiroz, F. Colonic content: Effect of diet, meals, and defecation. Neurogastroenterol. Motil. 2017, 29, e12930. [Google Scholar] [CrossRef]
- Mego, M.; Manichanh, C.; Accarino, A.; Campos, D.; Pozuelo, M.; Varela, E.; Vulevic, J.; Tzortzis, G.; Gibson, G.; Guarner, F.; et al. Metabolic adaptation of colonic microbiota to galactooligosaccharides: A proof-of-concept-study. Aliment. Pharmacol. Ther. 2017, 45, 670–680. [Google Scholar] [CrossRef]
- Mego, M.; Bendezu, A.; Accarino, A.; Malagelada, J.R.; Azpiroz, F. Intestinal gas homeostasis: Disposal pathways. Neurogastroenterol. Motil. 2015, 27, 363–369. [Google Scholar] [CrossRef]
- Agostini, S.; Goubern, M.; Tondereau, V.; Salvador-Cartier, C.; Bezirard, V.; Lévèque, M.; Keränen, H.; Theodorou, V.; Bourdu-Naturel, S.; Goupil-Feuillerat, N.; et al. A marketed fermented dairy product containing Bifidobacterium lactis CNCM I-2494 suppresses gut hypersensitivity and colonic barrier disruption induced by acute stress in rats. Neurogastroenterol. Motil. 2012, 24, 376–e172. [Google Scholar] [CrossRef]
- Tillisch, K.; Labus, J.; Kilpatrick, L.; Jiang, Z.; Stains, J.; Ebrat, B.; Guyonnet, D.; Legrain–Raspaud, S.; Trotin, B.; Naliboff, B.; et al. Consumption of Fermented Milk Product With Probiotic Modulates Brain Activity. Gastroenterology 2013, 144, 1394–1401.e4. [Google Scholar] [CrossRef]
| Habitual Diet (n = 74) | Flatulogenic Diet (n = 74) | p Value | |
|---|---|---|---|
| Flatulence * | 1.2 [1.1; 1.2] | 4.9 [4.8; 5.1] | p < 0.0001 |
| Abdominal discomfort/pain * | 0.5 [0.4; 0.5] | 2.7 [2.5; 2.9] | p < 0.05 |
| Abdominal distension * | 0.4 [0.4; 0.5] | 3.4 [3.3; 3.6] | p < 0.0001 |
| Bloating * | 0.5 [0.5; 0.6] | 3.5 [3.3; 3.6] | p < 0.0001 |
| Borborygmi * | 0.8 [0.7; 0.8] | 2.8 [2.7; 3.0] | p < 0.0001 |
| Odor of flatus * | 1.8 [1.7; 1.9] | 4.3 [4.1; 4.5] | p < 0.0001 |
| Digestive well-being * | 3.4 [3.3; 3.6] | 1.0 [0.8; 1.2] | p < 0.05 |
| Anal gas evacuations * | 8.7 [8.2; 9.2] | 20.7 [19.9; 21.6] | p < 0.0001 |
| Bowel movements ** | 1.2 [1.2; 1.3] | 1.4 [1.4; 1.5] | p > 0.05 |
| Stool consistency ** | 3.7 [3.6; 3.7] | 3.9 [3.8; 3.9] | p > 0.05 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le Nevé, B.; Martinez-De la Torre, A.; Tap, J.; Derrien, M.; Cotillard, A.; Barba, E.; Mego, M.; Nieto Ruiz, A.; Hernandez-Palet, L.; Dornic, Q.; et al. A Fermented Milk Product with B. lactis CNCM I-2494 and Lactic Acid Bacteria Improves Gastrointestinal Comfort in Response to a Challenge Diet Rich in Fermentable Residues in Healthy Subjects. Nutrients 2020, 12, 320. https://doi.org/10.3390/nu12020320
Le Nevé B, Martinez-De la Torre A, Tap J, Derrien M, Cotillard A, Barba E, Mego M, Nieto Ruiz A, Hernandez-Palet L, Dornic Q, et al. A Fermented Milk Product with B. lactis CNCM I-2494 and Lactic Acid Bacteria Improves Gastrointestinal Comfort in Response to a Challenge Diet Rich in Fermentable Residues in Healthy Subjects. Nutrients. 2020; 12(2):320. https://doi.org/10.3390/nu12020320
Chicago/Turabian StyleLe Nevé, Boris, Adrian Martinez-De la Torre, Julien Tap, Muriel Derrien, Aurélie Cotillard, Elizabeth Barba, Marianela Mego, Adoración Nieto Ruiz, Laura Hernandez-Palet, Quentin Dornic, and et al. 2020. "A Fermented Milk Product with B. lactis CNCM I-2494 and Lactic Acid Bacteria Improves Gastrointestinal Comfort in Response to a Challenge Diet Rich in Fermentable Residues in Healthy Subjects" Nutrients 12, no. 2: 320. https://doi.org/10.3390/nu12020320
APA StyleLe Nevé, B., Martinez-De la Torre, A., Tap, J., Derrien, M., Cotillard, A., Barba, E., Mego, M., Nieto Ruiz, A., Hernandez-Palet, L., Dornic, Q., Faurie, J.-M., Butler, J., Merino, X., Lobo, B., Pinsach Batet, F., Accarino, A., Pozuelo, M., Manichanh, C., & Azpiroz, F. (2020). A Fermented Milk Product with B. lactis CNCM I-2494 and Lactic Acid Bacteria Improves Gastrointestinal Comfort in Response to a Challenge Diet Rich in Fermentable Residues in Healthy Subjects. Nutrients, 12(2), 320. https://doi.org/10.3390/nu12020320






