Addressing the Fortification Quality Gap: A Proposed Way Forward
Abstract
:1. Introduction
2. Background on Regulatory Monitoring and Compliance of Food Fortification Programs
- Internally at food production facilities—here food producers are expected to put in place internal quality assurance and quality control protocols that allow for regular checking and adjusting of the fortification production process, which should be built into already-existing food safety and quality measures, such as Hazard Analysis Critical Control Point (HACCP) and Good Manufacturing Practices (GMP) measures;
- Externally by government food control agencies who are expected to verify that food producers have appropriate quality assurance and quality control measures in place that are well documented via technical audits and, occasionally, qualitative or quantitative end product tests. (Technical audits compliment the internal monitoring procedures that food manufacturers regularly implement and track. During an audit, an inspector will verify the production procedures and ensure documentation of the procedures are in place by observing the fortification process, conducting critical location checks (e.g., inside the feeder and inside the premix storage area), confirming that quality assurance and quality control protocols are established and followed, and reviewing records that document internal monitoring practices [18]. Verification of procedures and documentation is generally a more effective way of ensuring the end product is of adequate quality than end product tests.);
- At the import level by government customs or food control agencies, who are expected to check to be sure fortified foods imported into the country meet national standards—this can include verifying certificates of analysis and, occasionally, conducting qualitative or quantitative tests; and
- At the market level by government food control agencies, who are expected to verify fortified products’ packaging and labelling; this may occasionally include qualitative or quantitative testing [19] to gauge fortification program performance.
2.1. Current Status of National Fortification Program Rates of Compliance
2.2. Documented Challenges Collecting and Acting Upon Regulatory Monitoring Data
2.3. Global Resources to Address Pressing Monitoring Challenges
2.3.1. Policy Guidance Document
- Implement a standardized, realistic systems-based approach to determine compliance, emphasizing the process of fortification over regular testing of fortified food samples.
- Develop a comprehensive audit checklist that covers food quality, food safety, and food fortification.
- Use the premix reconciliation calculation to determine whether the manufacturing (fortification) process is sufficiently adding micronutrients to foods. This equation compares whether the amount of premix used correlates appropriately to the amount of fortified food produced over a set time period. Premix reconciliation is one task conducted during an audit at a food production site.
- Within the country’s fortification standards, express each micronutrient specification as a target value encompassed by actionable limits.
- Analyze composite samples of fortified foods quantitatively only periodically and as a means to validate the findings of an audit.
- Implement a user-friendly, computerized management information system (MIS) to make the process of data collection, collation, analysis, interpretation, and results dissemination more efficient and effective.
- Establish incentives that appeal to the food industry in addition to meaningful and enforceable penalties that drive consistent compliance among food manufacturers.
- Involve non-traditional stakeholders in monitoring fortification programs at the commercial and household levels to extend resources and expand public engagement in the initiative.
2.3.2. FortifyMIS
2.3.3. PalmATrack
2.3.4. Micronutrient Fortification Index
3. Use of Premix Reconciliation to Infer Compliance
4. Discussion
- -
- Use premix reconciliation to infer compliance. Incorporate premix reconciliation into industry food safety and fortification audits. Follow the 2018 Global Regulatory Monitoring Policy Guidance Document recommendations regarding less frequent quantitative testing only as a means of verifying industry audits, potentially only once or twice a year.
- -
- Leverage producer associations or similar neutral bodies. If a country does not have functioning food safety processes into which the premix reconciliation calculation can be incorporated or does not have enough human or financial resources to deploy inspectors, seek an alternative means of collecting the two key data points: production data and premix usage over the same period of time from industry. This may be via producer associations, subcommittees, or already-existing databases within the Ministry of Finance, Ministry of Health, or Ministry of Trade and Customs that are trusted by industry.
- -
- Cultivate trust through open discussions with producers/processors. Discuss openly with producers and, importantly, producer associations where they exist, how these two data points could be obtained and shared in a way that both parties (government and producers) are comfortable with. Discuss what would cultivate a more trusting relationship between producers and government that would allow government to provide greater support to industry in their fortification efforts (e.g., what incentives would be most meaningful to them, what would timely audit and inspection feedback look like, etc.), and what would allow both parties to work towards generating a national picture of fortification compliance.
- -
- Food safety inspections take place and can effectively absorb fortification activities.
- -
- Regular and rigorous laboratory testing should be the primary means by which to determine compliance.
- -
- Industry will automatically be willing to share data with government inspectors without first building a relationship of trust.
- -
- Government inspectors can reliably communicate results back to producers and processors in time to correct production practices before a product reaches the market.
- -
- The only way to understand industry adherence to national fortification standards is through government inspectors.
- -
- Viewing fortification programming through the lens of the public sector is more effective than through the lens of the private sector.
- -
- Relying on government policing is more effective that private sector support.
- -
- Low- and middle-income countries can accommodate fortification quality protocols that are more stringent than what most developed countries practice.
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Global Panel of Agriculture and Food Systems for Nutrition. 2016. Available online: https://www.glopan.org (accessed on 15 November 2020).
- Horton, S. The economic impact of micronutrient deficiencies. In Micronutrient Deficiencies During the Weaning Period and the First Years of Life; Pettifor, J.M., Zlotkin, S., Eds.; Nestlé Nutrition Workshop Series Pediatric Program; Nestec Ltd.: Basel, Switzerland, 2004; Volume 54, pp. 187–202. [Google Scholar]
- Black, M.M. Micronutrient deficiencies and cognitive functioning. J. Nutr. 2003, 133, 3927S–3931S. [Google Scholar] [CrossRef] [Green Version]
- Hoddinott, J.; Rosegrant, M.; Torero, M. Hunger and malnutrition. In Global Problems, Smart Solutions: Costs and Benefits; Lomborg, B., Ed.; Cambridge University Press: Cambridge, UK; Copenhagen Consensus Center: Tewksbury, MA, USA, 2013; pp. 332–367. [Google Scholar]
- Horton, S. The economics of food fortification. J. Nutr. 2006, 136, 1068–1071. [Google Scholar] [CrossRef] [PubMed]
- Baltussen, R.; Knai, C.; Sharan, M. Iron fortification and iron supplementation are cost-effective interventions to reduce iron deficiency in four subregions of the world. J. Nutr. 2004, 134, 2678–2684. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beal, T.; Massiot, E.; Arsenault, J.E.; Smith, M.R.; Hijmans, R.J. Global trends in dietary micronutrient supplies and estimated prevalence of inadequate intakes. PLoS ONE 2017, 12, e0175554. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keats, E.C.; Neufeld, L.M.; Garrett, G.S.; Mbuya, M.N.N.; Bhutta, Z.A. Improved micronutrient status and health outcomes in low- and middle-income countries following large-scale fortification: Evidence from a systematic review and meta-analysis. Am. J. Clin. Nutr. 2019, 109, 1696–1708. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mkambula, P.; Mduduzi, M.N.N.; Rowe, L.A.; Sablah, M.; Friesen, V.M.; Chadha, M.; Osei, A.K.; Ringholz, C.; Vasta, F.C.; Gorstein, J. The unfinished agenda for food fortification in low and middle-income countries: Quantifying progress, gaps and potential solutions. Nutrients 2020, 12, 354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vijaya, K.; Chadha, M.; Rowe, L.A.; Thompson, A.; Jain, S.; Walters, D.; Martinez, H. Reducing the burden of anemia and neural tube defects in low- and middle-income countries: An analysis to identify countries with an immediate potential to benefit from large-scale mandatory fortification of wheat flour and rice. Nutrients 2020. Submitted. [Google Scholar]
- Dary, O.; Mora, J.O. Food fortification to reduce vitamin a deficiency: International Vitamin A consultative group recommendations. J. Nutr. 2002, 132, 2927S–2933S. [Google Scholar] [CrossRef]
- Horton, S.; Ross, J. The economics of iron deficiency. Food Policy 2003, 28, 51–75. [Google Scholar] [CrossRef]
- Luthringer, C.L.; Rowe, L.A.; Vossenaar, M.; Garrett, G.S. Regulatory monitoring of fortified foods: Identifying barriers and good practices. Glob. Health 2015, 3, 446–461. [Google Scholar] [CrossRef] [Green Version]
- Rowe, L.A.; Garrett, G.S.; Luthringer, C.L.; Pachón, H.; Verster, A. Summit recommendation 2: Regulatory monitoring. Sight and Life: 36–39 April 2016. Available online: http://www.sightandlife.org/fileadmin/data/Magazine/2016/Suppl_to_1_2016/FutureFortified.pdf (accessed on 15 November 2020).
- Fortification Monitoring “Challenge” Workshop. Smarter Futures and Lodestar Center of Excellence Workshop Proceedings. October 2020. Available online: https://static1.squarespace.com/static/5e1df234eef02705f5446453/t/5f3d65b4ea586222105e3f7b/1597859255601/2018_SADC_Workshop-Report_v2.pdf (accessed on 15 November 2020).
- Akhigbe, O.; Amyot, D.; Reichards, G.S. Monitoring and management of regulatory compliance: A literature review. Int. J. Inf. Process. Manag. 2016, 7, 20–35. Available online: https://www.researchgate.net/publication/307513105_Monitoring_and_Management_of_Regulatory_Compliance_A_Literature_Review (accessed on 15 November 2020).
- Bittisnich, D. Food Fortification Regulatory Monitoring Assessment; Iodine Global Network: Seattle, WA, USA, 2020. [Google Scholar]
- Global Alliance for Improved Nutrition (GAIN); Project Healthy Children (PHC). Regulatory Monitoring of National Food Fortification Programs: A Policy Guidance Document. Global Fortification Technical Advisory Group (GF-TAG). 2018. Available online: https://s3-us-west-2.amazonaws.com/gfdx-publishefiles/Regulatory+Monitoring+Policy+Guidance+April+2018.pdf (accessed on 15 November 2020).
- World Health Organization (WHO); Food and Agriculture Organization (FAO). Guidelines on Food Fortification with Micronutrients; Allen, L., de Benoist, B., Dary, O., Hurrel, R., Eds.; World Health Organization: Geneva, Switzerland, 2006; Available online: https://www.who.int/nutrition/publications/guide_food_fortification_micronutrients.pdf (accessed on 15 November 2020).
- Global Fortification Data Exchange (GFDx). Available online: http://www.fortificationdata.org (accessed on 28 October 2020).
- Mambu, S.; National Standards Laboratory, Monrovia, Liberia. Personal communication, 15 October 2020.
- Ndao, I.; Nutrition International, Dakar, Senegal. Personal communication, 16 November 2020.
- Osendarp, S.J.; Martinez, H.; Garrett, G.S.; Neufeld, L.M.; De-Regil, L.M.; Vossenaar, M. Large-scale food fortification and biofortification in low-and middle-income countries: A review of programs, trends, challenges, and evidence gaps. Food Nutr. Bull. 2018, 39, 315–331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van den Wijngaart, A.; Begin, F.; Codling, K.; Randall, P.; Johnson, Q.W. Regulatory monitoring systems of fortified salt and wheat flour in selected ASEAN countries. Food Nutr. Bull. 2013, 34, S102–S111. [Google Scholar] [CrossRef] [PubMed]
- Dijkhuizen, M.A.; Wieringa, F.T.; Soekarjo, D.; Van, K.T.; Laillou, A. Legal framework for food fortification: Examples from Vietnam and Indonesia. Food Nutr. Bull. 2013, 34, S112–S123. [Google Scholar] [CrossRef] [PubMed]
- Gayer, J.; Smith, G. Micronutrient fortification of food in Southeast Asia: Recommendations from an expert workshop. Nutrients 2015, 7, 646–658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The Arusha Statement on Food Fortification. 2015. Available online: https://www.fantaproject.org/sites/default/files/Final-Arusha-Statement-on-Food-Fortification-Sep2015.pdf (accessed on 15 November 2020).
- The Food Fortification Initiative. Available online: https://www.ffinetwork.org/tools#fortificationmonitoring (accessed on 10 October 2020).
- Dove, M.; Millhouse International, KwaZulu-Natal, South Africa. Personal communication, 18 November 2020.
- Durotoye, T.; SAPFF Technoserve, Lagos, Nigeria. Personal communication, 2 February 2020.
- Randal, P.; P Cubed, Johannesburg, South Africa. Personal communication, 16 November 2020.
- South Africa Grain Information Service. Available online: https://www.sagis.org.za/index.html (accessed on 16 October 2020).
- de Hoop, M.; National Department of Health, Pretoria, South Africa. Personal communication, 20 July 2020.
- Wirth, J.P.; Nichols, E.; Mas’d, H.; Barham, R.; Johnson, Q.W.; Serdula, M. External mill monitoring of wheat flour fortification programs. An approach for program managers using experiences in Jordan. Nutrients 2013, 5, 4741–4759. [Google Scholar] [CrossRef] [Green Version]
- Mildon, A.; Klaas, N.; OʹLeary, M.; Yiannakis, M. Can fortification be implemented in rural African communities where micronutrient deficiencies are greatest? Lessons from projects in Malawi, Tanzania, and Senegal. Food Nutr. Bull. 2015, 36, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Marsh, D.R.; Schroeder, D.G.; Dearden, K.A.; Sternin, J.; Sternin, M. The power of positive deviance. BMJ 2004, 329, 1177–1179. [Google Scholar] [CrossRef] [Green Version]
- Sternin, J.; Choo, R. The power of positive deviancy. An effort to reduce malnutrition in Vietnam offers an important lesson about managing change. Harv. Bus. Rev. 2000, 78, 14–15. [Google Scholar]
- Bradley, E.H.; Curry, L.A.; Ramanadhan, S.; Rowe, L.A.; Nembhard, I.M.; Krumholz, H.M. Research in action: Using positive deviance to improve quality of health care. Implement Sci. 2009, 4, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walker, L.O.; Sterling, B.S.; Hoke, M.M.; Dearden, K.A. Applying the concept of positive deviance to public health data: A tool for reducing health disparities. Public Health Nurs. 2007, 24, 571–576. [Google Scholar] [CrossRef] [PubMed]
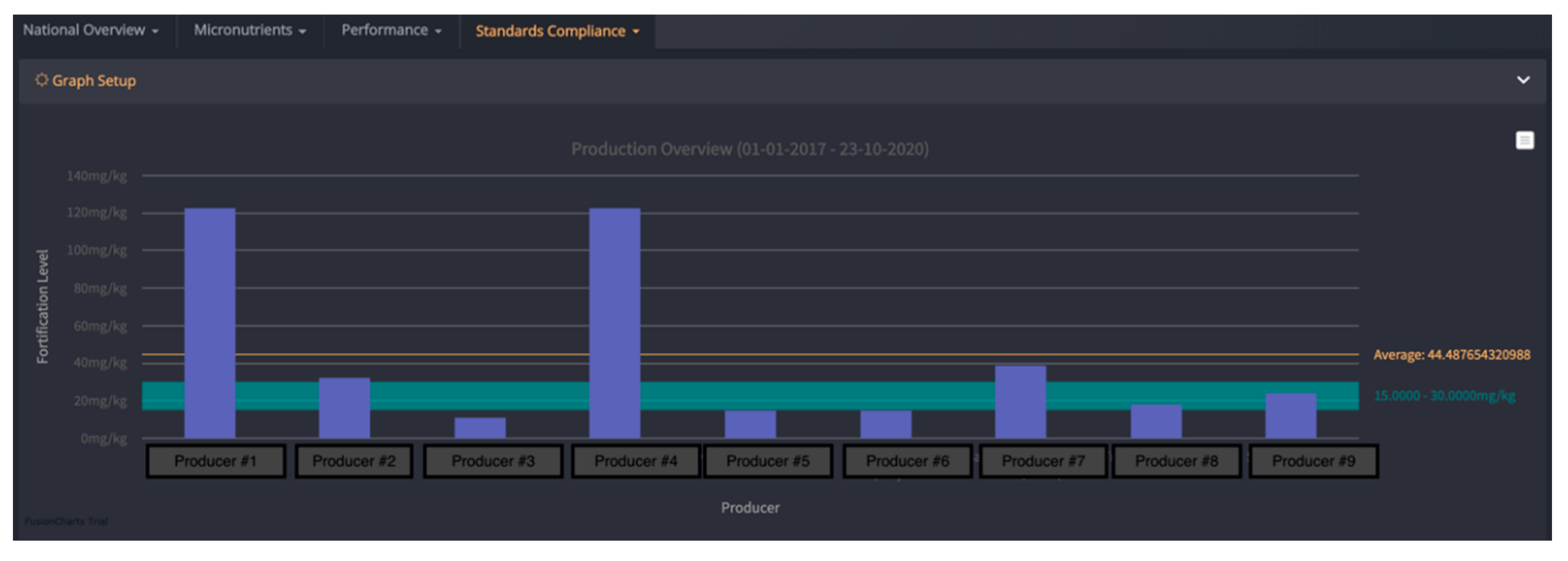
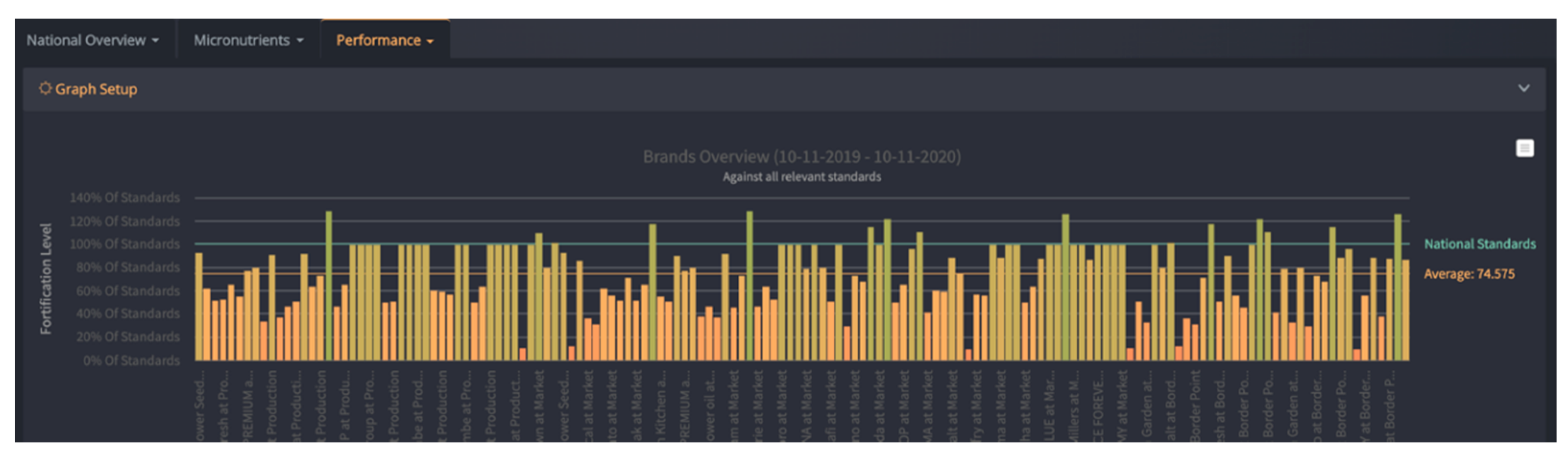
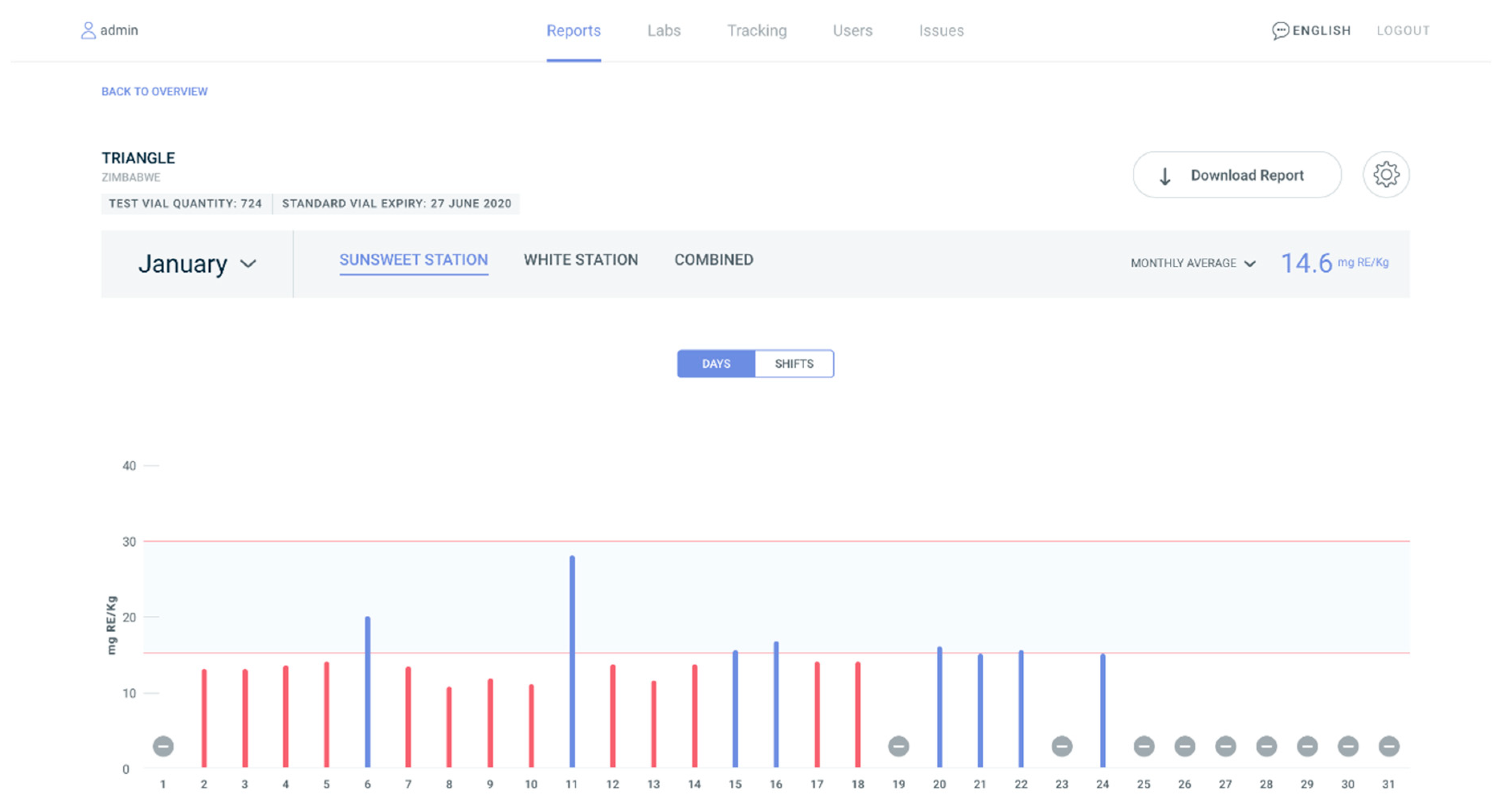
| Country | Timeframe | Staple Food/Condiment | Reported Compliance Range | Methods |
|---|---|---|---|---|
| Australia | 2010–2011 | Wheat flour | Unspecified | “Mills were audited by food control authorities to determine if they had quality assurance procedures in place and one wheat flour sample was taken per mill and analyzed in a lab for folic acid. All mills had quality assurance procedures in place and were deemed to be in compliance with the standard including five mills whose folic acid levels in samples were outside of the uncertainty range.” This was likely due to the challenges reliably detecting small quantities of this nutrient. |
| Brazil | 2006, 2007, 2010, 2011, 2012 | Wheat flour | 73–93% | “Wheat flour samples from mills were analyzed quantitatively for iron.” |
| 2006, 2008, 2010, 2011, 2012 | Maize flour | 16–100% | “Maize flour samples from mills were analyzed quantitatively for iron.” | |
| Chile | 2007–2014 (excluding 2013) | Wheat flour | 10–90% | “Flour samples were taken from national millers and imports four times a year. Samples were measured for thiamin, riboflavin, folic acid, and iron levels.” |
| Mexico | 2010–2017 | Wheat flour | 70–100% | “Government report of samples taken from mills. Samples measured for iron, folic acid, and zinc levels. Results reflect the lowest proportion of samples that met nutrient levels in the fortification standards.” |
| 2010–2017 | Maize flour | Not included | “Data obtained from a government report of samples taken from mills. The method is not detailed. Results reflect proportion of samples that met folic acid levels in the standard.” | |
| Peru | 2009–2017 | Wheat flour | 50–100% | “Government inspectors took flour samples from mills. Thiamine, riboflavin, niacin, folic acid, and iron levels were measured. Results reported use production volumes to generate the percent of flour compliant based on laboratory analyses. Premix reconciliation calculations were completed in a subset of mills.” |
| Liberia * | 2014–2020 | Wheat flour | 23–100% | Government inspectors take wheat flour samples from mills, import sites, and markets and test for iron using iCheck equipment. |
| 2014–2020 | Cooking oil | 6–83% | Government inspectors take cooking oil samples from import sites and markets and test for vitamin A using iCheck equipment. | |
| Salt | 21–100% | Government inspectors take salt samples from import sites and market and use the titration method to determine iodine content. | ||
| Senegal ** | 2018–2019 | Wheat flour | 90% | Wheat flour samples from mills are quantitatively analyzed at the control laboratory of the Ministry of Commerce. |
| Bangladesh | 2006–2018 | Salt | 79–97% | “Based on samples collected from production and tested for iodine.” |
| Colombia | 2014–2015 | Salt | 60–62% | “Based on samples collected at production level and tested by an authorized national lab for iodine and fluoride levels.” |
| Nigeria | 2011–2019 | Salt | 67–98% | “Based on quantitative tests of samples taken from production level. In 2018–2019, the USI/IDD Taskforce was set up in response to the decline in the compliance level of salt iodization at the household level in 2013. The Standards Organization of Nigeria (SON) authorized the release of the annual compliance data since the Taskforce started monitoring at the factory level. Data reported are the aggregate of onsite sampling assessment data collected by staff of SON during unscheduled monthly visits (January–December every year) to the two salt processing companies currently in operation.” |
| Peru | 2009–2010 | Salt | 41–59% | “Based on samples collected.” |
| Thailand | 2011–2018 | Salt | 13–92% | “Based on sample collection data from the Thai Food and Drug Administration (FDA).” |
| Uganda | 2018 | Salt | 79% | “Samples collected from production and tested for iodine content.” |
| Uzbekistan | 2004 and 2014 | Salt | 39–75% | “Data source unknown.” |
| Challenge Type | Government Regulatory Agency Challenges |
|---|---|
| Regulations/Government Structure | Need for clear regulations that identify the roles and responsibilities of stakeholders and how they should collaborate |
| Regulations related to monitoring, inspection, and enforcement are often fragmented and not appropriately embedded within legal frameworks, leading to a lack of (or weak) enforcement | |
| Need for improved regulatory agency structure | |
| Poor government coordination | |
| Need for better harmonization and integration of fortification efforts vertically into food safety mechanisms as a subset of food control within the appropriate national entities | |
| Lack of clear specifications and objective assignment of enforcement mechanisms and penalties in the legal framework | |
| Ongoing reorganization of regulatory agencies | |
| Fragmented system for collecting data/agency/inspector overlap | |
| Enforcement | Low priority and capacity for enforcement |
| Lack of willingness on the part of government inspectors to take on the “political risk” of enforcement (e.g., using penalties was seen as politically risky due to perceived or real resistance from interest groups or mill associations) and often lead to penalties that were not serve enough to encourage adequate fortification | |
| Laboratory/Testing/Equipment | Laboratory capacity constraints, both in terms of trained analysts and local availability/cost of equipment and reagents |
| Both regulators and industry have an over-reliance on end-product quantitative testing and lab results, which have a high margin of error and are time consuming to conduct | |
| Lack of reliance on industry audits in place of quantitative testing | |
| Need for food inspectors to consider laboratory analysis in combination with additional critical information, including information obtained from mill or factory inspections | |
| Lack of cost-effective and field-friendly tools to ensure quality to assist with effective enforcement of fortification legislation; cost and availability of reagents | |
| Quantitative testing of multiple micronutrients as opposed to one market nutrient and/or testing of micronutrients that are considerably difficult to measure because they are labile, added in minute quantities, or have a wide margin of error leading to high investment of lab resources and often unreliable results but often results that industry must respond to | |
| Budget | Need for improved regulatory agency financing |
| Limited national budget allocations for fortification | |
| Human Resources | Need for improved regulatory agency capacity |
| Lack of trained inspectors and analysts (for product sampling and laboratory testing) | |
| Limited personnel for legal action | |
| Lack of motivation at the implementing local government level | |
| Corruption among inspection personnel | |
| Delay in getting results to millers | |
| Data collection | Over-reliance on monitoring at the retail level in place of producer or import level |
| Lack of a centralized data collection mechanisms for collected data | |
| Need to simply the process of regulatory monitoring data collection for inspectors | |
| Fragmented system for collecting data/agency/inspector overlap | |
| Relationship between public and private sectors | Lack of trust between government and industry |
| Challenge Type | Industry (Food Producer) |
|---|---|
| Regulations | Need for clear regulations that identify the roles and responsibilities of stakeholders and how they should collaborate |
| Enforcement | Competition with non-compliant producers |
| Lack of realistic, meaningful, and consistent enforcement (incentives and penalties) | |
| Laboratory | Poor laboratory capacity |
| Lack of locally available reagents | |
| Equipment | Lack of fortification equipment that is locally available, accessible, and affordable |
| Premix | High price of premix |
| Poor quality premix | |
| Lack of duty-free premix | |
| Budget | Lack of internal budgets that include fortification |
| Human Resources | Lack of training |
| Need for training on internal monitoring with a special focus on process control | |
| Lack of awareness of standards | |
| Delay in results to millers | |
| Relationship between public and private sectors | Need for communication between sectors (e.g., industry and regulatory agency) |
| Lack of trust between government and industry | |
| Consumers | Lack of product market demand |
| Lack of accountability to consumers | |
| Other | Lack of an effective business model for fortification |
| Purposeful under-fortification |
| Item * | Unit | Where to Locate |
|---|---|---|
| MT ** | See facility records |
| MT | See facility records |
| MT | See facility records |
| MT | Calculate: A + B − C |
| MT | See facility records |
| g ***/MT | Calculate: D/E × 1000 |
| g/MT | Provided by premix producer |
| Result: Percent of target addition rate achieved | % | Calculate: F/G × 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rowe, L.A. Addressing the Fortification Quality Gap: A Proposed Way Forward. Nutrients 2020, 12, 3899. https://doi.org/10.3390/nu12123899
Rowe LA. Addressing the Fortification Quality Gap: A Proposed Way Forward. Nutrients. 2020; 12(12):3899. https://doi.org/10.3390/nu12123899
Chicago/Turabian StyleRowe, Laura A. 2020. "Addressing the Fortification Quality Gap: A Proposed Way Forward" Nutrients 12, no. 12: 3899. https://doi.org/10.3390/nu12123899




