Got Mylk? The Emerging Role of Australian Plant-Based Milk Alternatives as A Cow’s Milk Substitute
Abstract
1. Introduction
2. Materials and Methods
2.1. Modelling of Dietary Scenarios
2.2. Statistical Analysis
3. Results
3.1. Nutrient Composition
Comparisons with Cow’s Milk Under Australian Legislations
3.2. Projected Dietary Scenarios
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Ingredient | Number |
|---|---|
| F006081 | Nut, almond, with skin, raw, unsalted |
| F006088 | Nut, cashew, raw, unsalted |
| F006099 | Nut, macadamia, raw, unsalted |
| F006098 | Nut, hazelnut, raw, unsalted |
| F004002 | Flour, soya |
| F002982 | Coconut, cream, regular fat |
| F002991 | Coconut, milk, canned, regular fat |
| F003998 | Flour, rice |
| F006136 | Oats, hulled, uncooked |
| F007598 | Quinoa, uncooked |
| F008209 | Seed, chia, dried |
| F005638 | Milk, cow, fluid, skim (0.15% fat), added milk solids |
| F005637 | Milk, cow, fluid, skim (0.15% fat) |
| F005598 | Milk, cow, fluid, lactose-free, reduced-fat (1% fat) |
| F005614 | Milk, cow, fluid, reduced-fat (1% fat) |
| F005621 | Milk, cow, fluid, reduced-fat (1.5% fat), added omega 3 polyunsaturates |
| F005599 | Milk, cow, fluid, lactose-free, regular fat (3.5% fat) |
| F005634 | Milk, cow, fluid, regular fat (3.5% fat) |
| 12012 1 | Seeds, hemp seed, hulled |
References
- Evershed, R.P.; Payne, S.; Sherratt, A.G.; Copley, M.S.; Coolidge, J.; Urem-Kotsu, D.; Kotsakis, K.; Özdoğan, M.; Özdoğan, A.E.; Nieuwenhuyse, O.; et al. Earliest date for milk use in the Near East and southeastern Europe linked to cattle herding. Nature 2008, 455, 528–531. [Google Scholar] [CrossRef]
- Muehlhoff, E.; Bennett, A.; McMahon, D. Milk and Dairy Products in Human Nutrition; Food and Agriculture Organization of the United Nations (FAO): Rome, Italy, 2013. [Google Scholar]
- Mylan, J.; Morris, C.; Beech, E.; Geels, F.W. Rage against the regime: Niche-regime interactions in the societal embedding of plant-based milk. Environ. Innov. Soc. Transit. 2019, 31, 233–247. [Google Scholar] [CrossRef]
- IBISWorld. Soy and Almond Milk Production in Australia—Market Research Report; IBISWorld Pty Ltd.: Canberra, Australia, 2020. [Google Scholar]
- McCarthy, K.S.; Parker, M.; Ameerally, A.; Drake, S.L.; Drake, M.A. Drivers of choice for fluid milk versus plant-based alternatives: What are consumer perceptions of fluid milk? J. Dairy Sci. 2017, 100, 6125–6138. [Google Scholar] [CrossRef] [PubMed]
- Haas, R.; Schnepps, A.; Pichler, A.; Meixner, O. Cow Milk versus Plant-Based Milk Substitutes: A Comparison of Product Image and Motivational Structure of Consumption. Sustainability 2019, 11, 5046. [Google Scholar] [CrossRef]
- Abbring, S.; Hols, G.; Garssen, J.; van Esch, B.C.A.M. Raw cow’s milk consumption and allergic diseases—The potential role of bioactive whey proteins. Eur. J. Pharmacol. 2019, 843, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Hodges, J.K.; Cao, S.; Cladis, D.P.; Weaver, C.M. Lactose intolerance and bone health: The challenge of ensuring adequate calcium intake. Nutrients 2019, 11, 718. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.; Laganà, S.A.; Rapisarda, M.A.; La Ferrera, M.G.; Buscema, M.; Rossetti, P.; Nigro, A.; Muscia, V.; Valenti, G.; Sapia, F.; et al. Vitamin B12 among Vegetarians: Status, Assessment and Supplementation. Nutrients 2016, 8, 767. [Google Scholar] [CrossRef] [PubMed]
- Curtain, F.; Grafenauer, S. Plant-Based Meat Substitutes in the Flexitarian Age: An Audit of Products on Supermarket Shelves. Nutrients 2019, 11, 2603. [Google Scholar] [CrossRef]
- Tsakiridou, E.; Tsakiridou, H.; Mattas, K.; Arvaniti, E. Effects of animal welfare standards on consumers’ food choices. Acta Agric. Scand. Sect. C Food Econ. 2010, 7, 234–244. [Google Scholar] [CrossRef]
- Holden, L.A. Understanding the environmental impact of global dairy production. J. Anim. Sci. 2020, 98. [Google Scholar] [CrossRef]
- Ritchie, H.; Reay, D.S.; Higgins, P. The impact of global dietary guidelines on climate change. Glob. Environ. Chang. 2018, 49, 46–55. [Google Scholar] [CrossRef]
- National Dairy Council. Consumer Perceptions of Dairy milk and Plant-Based Milk Alternatives; Vermont General Assembly: Montpelier, Vermont, 2018. [Google Scholar]
- McClements, D.J.; Newman, E.; McClements, I.F. Plant-based Milks: A Review of the Science Underpinning Their Design, Fabrication, and Performance. Compr. Rev. Food Sci. Food Saf. 2019, 18, 2047–2067. [Google Scholar] [CrossRef]
- Poore, J.; Nemecek, T. Reducing food’s environmental impacts through producers and consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Grant, C.A.; Hicks, A.L. Comparative Life Cycle Assessment of Milk and Plant-Based Alternatives. Environ. Eng. Sci. 2018, 35, 1235–1247. [Google Scholar] [CrossRef]
- Wenzel, P.; Jungbluth, N. The Environmental Impact of Vegan Drinks Compared to Whole Milk; ESU-Services Ltd.: Schaffhausen, Switzerland, 2017. [Google Scholar]
- Gazan, R.; Brouzes, C.M.C.; Vieux, F.; Maillot, M.; Lluch, A.; Darmon, N. Mathematical Optimization to Explore Tomorrow’s Sustainable Diets: A Narrative Review. Adv. Nutr. 2018, 9, 602–616. [Google Scholar] [CrossRef]
- Perignon, M.; Sinfort, C.; El Ati, J.; Traissac, P.; Drogué, S.; Darmon, N.; Amiot, M.-J.; Amiot, M.J.; Achir, N.; Alouane, L.; et al. How to meet nutritional recommendations and reduce diet environmental impact in the Mediterranean region? An optimization study to identify more sustainable diets in Tunisia. Glob. Food Secur. 2019, 23, 227–235. [Google Scholar] [CrossRef]
- Shingfield, K.J.; Chilliard, Y.; Toivonen, V.; Kairenius, P.; Givens, D.I. Trans fatty acids and bioactive lipids in ruminant milk. In Bioactive Components of Milk; Springer: New York, NY, USA, 2008; pp. 3–65. [Google Scholar]
- Australian Bureau of Statistics. 4364.0.55.007-National Nutrition and Physical Activity Survey, 2011–2012. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/4364.0.55.012~2011-12~Main%20Features~Key%20Findings~1 (accessed on 5 January 2020).
- Dairy Australia. Consumption Summary. Available online: https://www.dairyaustralia.com.au/industry/production-and-sales/consumption-summary (accessed on 22 February 2020).
- Bureau of Statistics. 4364.0.55.008-Australian Health Survey: Usual Nutrient Intakes, 2011–2012. Available online: https://www.abs.gov.au/ausstats/abs@.nsf/mf/4364.0.55.008 (accessed on 6 November 2019).
- Auclair, O.; Han, Y.; Burgos, S.A. Consumption of Milk and Alternatives and Their Contribution to Nutrient Intakes among Canadian Adults: Evidence from the 2015 Canadian Community Health Survey—Nutrition. Nutrients 2019, 11, 1948. [Google Scholar] [CrossRef]
- Saito, A.; Okada, E.; Tarui, I.; Matsumoto, M.; Takimoto, H. The Association between Milk and Dairy Products Consumption and Nutrient Intake Adequacy among Japanese Adults: Analysis of the 2016 National Health and Nutrition Survey. Nutrients 2019, 11, 2361. [Google Scholar] [CrossRef]
- Murphy, M.M.; Douglass, J.S.; Johnson, R.K.; Spence, L.A. Drinking flavored or plain milk is positively associated with nutrient intake and is not associated with adverse effects on weight status in US children and adolescents. J. Am. Diet. Assoc. 2008, 108, 631–639. [Google Scholar] [CrossRef]
- Fayet, F.; Ridges, L.A.; Wright, J.K.; Petocz, P. Australian children who drink milk (plain or flavored) have higher milk and micronutrient intakes but similar body mass index to those who do not drink milk. Nutr. Res. 2013, 33, 95–102. [Google Scholar] [CrossRef]
- Cifelli, C.J.; Houchins, J.A.; Demmer, E.; Fulgoni, V.L. Increasing plant based foods or dairy foods differentially affects nutrient intakes: Dietary scenarios using NHANES 2007–2010. Nutrients 2016, 8, 422. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.E.; Vivian, W.J.; Oddy, W.H.; Beilin, L.J.; Mori, T.A.; O’Sullivan, T.A. Changes in dairy food and nutrient intakes in Australian adolescents. Nutrients 2012, 4, 1794–1811. [Google Scholar] [CrossRef] [PubMed]
- Rangan, A.M.; Flood, V.M.; Denyer, G.; Webb, K.; Marks, G.B.; Gill, T.P. Dairy consumption and diet quality in a sample of Australian children. J. Am. Coll. Nutr. 2012, 31, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.B.; Coveney, J.; Henderson, J.; Ward, P.R.; Taylor, A.W. Reconnecting Australian consumers and producers: Identifying problems of distrust. Food Policy 2012, 37, 634–640. [Google Scholar] [CrossRef]
- Jacobs, E.T.; Foote, J.A.; Kohler, L.N.; Skiba, M.B.; Thomson, C.A. Re-examination of dairy as a single commodity in US dietary guidance. Nutr. Rev. 2020, 78, 225–234. [Google Scholar] [CrossRef]
- Seo, H.-J.; Cho, Y.-E.; Kim, T.; Shin, H.-I.; Kwun, I.-S. Zinc may increase bone formation through stimulating cell proliferation, alkaline phosphatase activity and collagen synthesis in osteoblastic MC3T3-E1 cells. Nutr. Res. Pract. 2010, 4, 356–361. [Google Scholar] [CrossRef]
- Allgrove, J. Physiology of calcium, phosphate, magnesium and vitamin D. In Calcium and Bone Disorders in Children and Adolescents; Karger Publishers: Basel, Switzerland, 2015; Volume 28, pp. 7–32. [Google Scholar]
- Baroncelli, G.I.; Bertelloni, S.; Sodini, F.; Saggese, G. Osteoporosis in Children and Adolescents. Pediatric Drugs 2005, 7, 295–323. [Google Scholar] [CrossRef]
- Unni, J.; Garg, R.; Pawar, R. Bone mineral density in women above 40 years. J. Midlife Health 2010, 1, 19–22. [Google Scholar] [CrossRef]
- Demontiero, O.; Vidal, C.; Duque, G. Aging and bone loss: New insights for the clinician. Ther. Adv. Musculoskelet Dis. 2012, 4, 61–76. [Google Scholar] [CrossRef]
- Rizzoli, R.; Bischoff-Ferrari, H.; Dawson-Hughes, B.; Weaver, C. Nutrition and bone health in women after the menopause. Women’s Health 2014, 10, 599–608. [Google Scholar] [CrossRef]
- Ji, M.-X.; Yu, Q. Primary osteoporosis in postmenopausal women. Chronic Dis. Transl. Med. 2015, 1, 9–13. [Google Scholar] [CrossRef]
- Food Standards Australia New Zealand. Plant-based Milk Alternatives. Available online: https://www.foodstandards.gov.au/consumer/nutrition/milkaltern/Pages/default.aspx (accessed on 20 February 2020).
- Scholz-Ahrens, K.E.; Ahrens, F.; Barth, C.A. Nutritional and health attributes of milk and milk imitations. Eur. J. Nutr. 2019, 59, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chalupa-Krebzdak, S.; Long, C.J.; Bohrer, B.M. Nutrient density and nutritional value of milk and plant-based milk alternatives. Int. Dairy J. 2018, 87, 84–92. [Google Scholar] [CrossRef]
- Singhal, S.; Baker, R.D.; Baker, S.S. A Comparison of the Nutritional Value of Cow’s Milk and Nondairy Beverages. J. Pediatric Gastroenterol. Nutr. 2017, 64, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.A.; Kumar, S.; Kumar, V.; Sharma, R. Milk Analog: Plant based alternatives to conventional milk, production, potential and health concerns. Crit. Rev. Food Sci. Nutr. 2019, 16, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Sethi, S.; Tyagi, S.K.; Anurag, R.K. Plant-based milk alternatives an emerging segment of functional beverages: A review. J. Food Sci. Technol. 2016, 53, 3408–3423. [Google Scholar] [CrossRef]
- Jeske, S.; Zannini, E.; Arendt, E.K. Past, present and future: The strength of plant-based dairy substitutes based on gluten-free raw materials. Food Res. Int. 2018, 110, 42–51. [Google Scholar] [CrossRef]
- Food Standards Australia New Zealand. Voluntary Addition of Vitamins & Minerals to Nut-and Seed-Based Beverages. Available online: https://www.foodstandards.gov.au/code/applications/Pages/A1104-VitsMinsNutSeedBevs.aspx (accessed on 6 January 2020).
- Banwell, C.; Broom, D.; Davies, A.; Dixon, J. How Convenience Is Shaping Australian Diets: The Disappearing Dessert. In Weight of Modernity: An Intergenerational Study of the Rise of Obesity; Springer: Dordrecht, The Netherlands, 2012; pp. 41–58. [Google Scholar] [CrossRef]
- Shaw, M.; Nugent, A.P.; McNulty, B.A.; Walton, J.; McHugh, M.; Kane, A.; Heslin, A.M.; Morrissey, E.; Mullan, K.; Woodside, J.V. What is the availability of iodised salt in supermarkets on the Island of Ireland? Eur. J. Clin. Nutr. 2019, 73, 1636–1638. [Google Scholar] [CrossRef]
- National Health and Medical Research Council, New Zealand Ministry of Health. Nutrient Reference Values for Australia and New Zealand; National Health and Medical Research Council: Canberra, Australia, 2017. [Google Scholar]
- Curtain, F.; Grafenauer, S. Comprehensive nutrition review of grain-based muesli bars in Australia: An audit of supermarket products. Foods 2019, 8, 370. [Google Scholar] [CrossRef]
- Roy Morgan. Woolworths and Aldi Grow Grocery Market Share in 2018–2019. Available online: http://www.roymorgan.com/findings/7936-australian-grocery-market-december-2018-201904050426 (accessed on 2 February 2020).
- Food Standards Australia New Zealand. Application of Discretionary Foods Flag. Available online: https://www.foodstandards.gov.au/science/monitoringnutrients/australianhealthsurveyandaustraliandietaryguidelines/applicationofdiscretionary/Pages/default.aspx (accessed on 2 February 2020).
- Food Standards Australia New Zealand. Australian Food Composition Database. Available online: https://www.foodstandards.gov.au/science/monitoringnutrients/afcd/Pages/default.aspx (accessed on 2 February 2020).
- Food Standards Australia New Zealand. User Guide to Standard 1.2.8—Nutrition Information Requirements. Available online: https://www.foodstandards.gov.au/code/userguide/Documents/Userguide_Prescribed%20Nutrition%20Information%20Nov%2013%20Dec%202013.pdf (accessed on 2 February 2020).
- Baldi, A.; Pinotti, L. Lipophilic microconstituents of milk. In Bioactive Components of Milk; Springer: New York, NY, USA, 2008; pp. 109–125. [Google Scholar]
- Food Standards Australia New Zealand. Definitions used throughout the Code. In Standard 1.1.2, Food Standards Code Australia New Zealand. Available online: https://www.legislation.gov.au/Details/F2015L00385/Explanatory%20Statement/Text (accessed on 28 April 2020).
- Burton, N.W.; Brown, W.; Dobson, A. Accuracy of body mass index estimated from self-reported height and weight in mid-aged Australian women. Aust. N. Z. J. Public Health 2010, 34, 620–623. [Google Scholar] [CrossRef]
- Boehm, J.; Franklin, R.C.; Newitt, R.; McFarlane, K.; Grant, T.; Kurkowski, B. Barriers and motivators to exercise for older adults: A focus on those living in rural and remote areas of Australia. Aust. J. Rural Health 2013, 21, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.L.; Brown, W.J. Older Australians and physical activity levels: Do we know how many are meeting guidelines? Australas. J. Ageing 2012, 31, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Breda, J.; Jewell, J.; Keller, A. The importance of the World Health Organization sugar guidelines for dental health and obesity prevention. Caries Res. 2019, 53, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Bernat, N.; Cháfer, M.; Chiralt, A.; González-Martinez, C. Vegetable milks and their fermented derivative products. Int. J. Food Stud. 2014, 3, 93–124. [Google Scholar] [CrossRef]
- Sousa, A.; Kopf-Bolanz, K. Nutritional implications of an increasing consumption of non-dairy plant-based beverages instead of cow’s milk in Switzerland. J. Adv. Dairy Res. 2017, 5, 2. [Google Scholar] [CrossRef]
- Vanga, S.K.; Raghavan, V. How well do plant based alternatives fare nutritionally compared to cow’s milk? J. Food Sci. Technol. 2018, 55, 10–20. [Google Scholar] [CrossRef]
- Pistollato, F.; Sumalla Cano, S.; Elio, I.; Masias Vergara, M.; Giampieri, F.; Battino, M. Plant-based and plant-rich diet patterns during gestation: Beneficial effects and possible shortcomings. Adv. Nutr. 2015, 6, 581–591. [Google Scholar] [CrossRef]
- Slagman, A.; Harriss, L.; Campbell, S.; Muller, R.; McDermott, R. Folic acid deficiency declined substantially after introduction of the mandatory fortification programme in Queensland, Australia: A secondary health data analysis. Public Health Nutr. 2019, 22, 3426–3434. [Google Scholar] [CrossRef]
- Gallagher, C.M.; Black, L.J.; Oddy, W.H. Micronutrient intakes from food and supplements in Australian adolescents. Nutrients 2014, 6, 342–354. [Google Scholar] [CrossRef]
- Chiu, M.; Dillon, A.; Watson, S. Vitamin A deficiency and xerophthalmia in children of a developed country. J. Paediatr. Child Health 2016, 52, 699–703. [Google Scholar] [CrossRef]
- Gallegos, D.; Booth, S.; Kleve, S.; McKenchie, R.; Lindberg, R. Food insecurity in Australian households: From charity to entitlement. In A Sociology of Food and Nutrition: The Social Appetite; Oxford University Press: Oxford, UK, 2017; pp. 55–74. [Google Scholar]
- Gwynn, J.; Sim, K.; Searle, T.; Senior, A.; Lee, A.; Brimblecombe, J. Effect of nutrition interventions on diet-related and health outcomes of Aboriginal and Torres Strait Islander Australians: A systematic review. BMJ Open 2019, 9, e025291. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.R.; Davison, B.; Ma, G.Y.; Eastman, C.J.; Mackerras, D.E. Iodine status of Indigenous and non-Indigenous young adults in the Top End, before and after mandatory fortification. Med J. Aust. 2019, 210, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Charlton, K.; Probst, Y.; Kiene, G. Dietary Iodine Intake of the Australian Population after Introduction of a Mandatory Iodine Fortification Programme. Nutrients 2016, 8, 701. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.; Heath, A.-L. Population groups at risk of zinc deficiency in Australia and New Zealand. Nutr. Diet. 2011, 68, 97–108. [Google Scholar] [CrossRef]
- Rosado, J.; Caamano, M.; Montoya, Y.; de Lourdes Solano, M.; Santos, J.; Long, K. Interaction of zinc or vitamin A supplementation and specific parasite infections on Mexican infants’ growth: A randomized clinical trial. Eur. J. Clin. Nutr. 2009, 63, 1176–1184. [Google Scholar] [CrossRef]
- Grafenauer, S.; Curtain, F. An audit of Australian bread with a focus on loaf breads and whole grain. Nutrients 2018, 10, 1106. [Google Scholar] [CrossRef]
- Bouga, M.; Lean, M.E.; Combet, E. Iodine and pregnancy—A qualitative study focusing on dietary guidance and information. Nutrients 2018, 10, 408. [Google Scholar] [CrossRef]
- Bath, S.C.; Hill, S.; Infante, H.G.; Elghul, S.; Nezianya, C.J.; Rayman, M.P. Iodine concentration of milk-alternative drinks available in the UK in comparison with cows’ milk. Br. J. Nutr. 2017, 118, 525–532. [Google Scholar] [CrossRef]
- Crawford, B.A.; Cowell, C.T.; Emder, P.J.; Learoyd, D.L.; Chua, E.L.; Sinn, J.; Jack, M.M. Iodine toxicity from soy milk and seaweed ingestion is associated with serious thyroid dysfunction. Med. J. Aust. 2010, 193, 413–415. [Google Scholar] [CrossRef]
- Kalkwarf, H.J.; Khoury, J.C.; Lanphear, B.P. Milk intake during childhood and adolescence, adult bone density, and osteoporotic fractures in US women. Am. J. Clin. Nutr. 2003, 77, 257–265. [Google Scholar] [CrossRef]
- Bus, A.; Worsley, A. Consumers’ sensory and nutritional perceptions of three types of milk. Public Health Nutr. 2003, 6, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Burrow, K.; Young, W.; McConnell, M.; Carne, A.; Bekhit, A.E.-D. Do Dairy Minerals Have a Positive Effect on Bone Health? Compr. Rev. Food Sci. Food Saf. 2018, 17, 989–1005. [Google Scholar] [CrossRef]
- Dawson, A.; Dennison, E. Measuring the musculoskeletal aging phenotype. Maturitas 2016, 93, 13–17. [Google Scholar] [CrossRef]
- CSIRO. Protein Balance: New Concepts for Protein in Weight Management; Commonwealth Scientific and Industrial Research Organisation: Canberra, Australia, 2018. [Google Scholar]
- Berrazaga, I.; Micard, V.; Gueugneau, M.; Walrand, S. The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrients 2019, 11, 1825. [Google Scholar] [CrossRef]
- van Vliet, S.; Burd, N.A.; van Loon, L.J. The skeletal muscle anabolic response to plant-versus animal-based protein consumption. J. Nutr. 2015, 145, 1981–1991. [Google Scholar] [CrossRef]
- Nowson, C.; O’Connell, S. Protein requirements and recommendations for older people: A review. Nutrients 2015, 7, 6874–6899. [Google Scholar] [CrossRef] [PubMed]
- Nordin, B.C.; Need, A.G.; Morris, H.A.; O’Loughlin, P.D.; Horowitz, M. Effect of age on calcium absorption in postmenopausal women. Am. J. Clin. Nutr. 2004, 80, 998–1002. [Google Scholar] [CrossRef]
- Sepúlveda-Loyola, W.; Phu, S.; Bani Hassan, E.; Brennan-Olsen, S.L.; Zanker, J.; Vogrin, S.; Conzade, R.; Kirk, B.; Al Saedi, A.; Probst, V.; et al. The Joint Occurrence of Osteoporosis and Sarcopenia (Osteosarcopenia): Definitions and Characteristics. J. Am. Med Dir. Assoc. 2020, 21, 220–225. [Google Scholar] [CrossRef]
- Reginster, J.-Y.; Beaudart, C.; Buckinx, F.; Bruyère, O. Osteoporosis and sarcopenia: Two diseases or one? Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 31–36. [Google Scholar] [CrossRef]
- Abe, S.; Ezaki, O.; Suzuki, M. Medium-chain triglycerides (8: 0 and 10: 0) are promising nutrients for sarcopenia: A randomized controlled trial. Am. J. Clin. Nutr. 2019, 110, 652–665. [Google Scholar] [CrossRef]
- Abe, S.; Ezaki, O.; Suzuki, M. Medium-Chain Triglycerides in Combination with Leucine and Vitamin D Increase Muscle Strength and Function in Frail Elderly Adults in a Randomized Controlled Trial. The J. Nutr. 2016, 146, 1017–1026. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, C.J.; Hermans, W.J.H.; Holwerda, A.M.; Smeets, J.S.J.; Senden, J.M.; van Kranenburg, J.; Gijsen, A.P.; Wodzig, W.K.H.W.; Schierbeek, H.; Verdijk, L.B.; et al. Branched-chain amino acid and branched-chain ketoacid ingestion increases muscle protein synthesis rates in vivo in older adults: A double-blind, randomized trial. Am. J. Clin. Nutr. 2019, 110, 862–872. [Google Scholar] [CrossRef]
- Gobbi, L.; Ciano, S.; Rapa, M.; Ruggieri, R. Biogenic Amines Determination in “Plant Milks”. Beverages 2019, 5, 40. [Google Scholar] [CrossRef]
- Schuster, M.J.; Wang, X.; Hawkins, T.; Painter, J.E. Comparison of the Nutrient Content of Cow’s Milk and Nondairy Milk Alternatives: What’s the Difference? Nutr. Today 2018, 53, 153–159. [Google Scholar] [CrossRef]
- Ritz, E.; Hahn, K.; Ketteler, M.; Kuhlmann, M.K.; Mann, J. Phosphate additives in food—A health risk. Dtsch Arztebl. Int. 2012, 109, 49–55. [Google Scholar] [CrossRef]
- D’Alessandro, C.; Piccoli, G.B.; Cupisti, A. The “phosphorus pyramid”: A visual tool for dietary phosphate management in dialysis and CKD patients. BMC Nephrol. 2015, 16, 9. [Google Scholar] [CrossRef] [PubMed]
- Australian Institute of Health Welfare. Chronic Kidney Disease; AIHW: Canberra, Australia, 2019. [Google Scholar]
- Jeske, S.; Zannini, E.; Arendt, E.K. Evaluation of physicochemical and glycaemic properties of commercial plant-based milk substitutes. Plant Foods Hum. Nutr. 2017, 72, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Grieger, J.A.; Johnson, B.J.; Wycherley, T.P.; Golley, R.K. Comparing the nutritional impact of dietary strategies to reduce discretionary choice intake in the Australian adult population: A simulation modelling study. Nutrients 2017, 9, 442. [Google Scholar] [CrossRef]
- Rouf, A.S.; Sui, Z.; Rangan, A.; Grech, A.; Allman-Farinelli, M. Low calcium intakes among Australian adolescents and young adults are associated with higher consumption of discretionary foods and beverages. Nutrition 2018, 55, 146–153. [Google Scholar] [CrossRef]
- Fardet, A.; Dupont, D.; Rioux, L.-E.; Turgeon, S.L. Influence of food structure on dairy protein, lipid and calcium bioavailability: A narrative review of evidence. Crit. Rev. Food Sci. Nutr. 2019, 59, 1987–2010. [Google Scholar] [CrossRef]
- Platel, K.; Srinivasan, K. Bioavailability of Micronutrients from Plant Foods: An Update. Crit. Rev. Food Sci. Nutr. 2016, 56, 1608–1619. [Google Scholar] [CrossRef] [PubMed]
- Lenton, S.; Nylander, T.; Teixeira, S.C.M.; Holt, C. A review of the biology of calcium phosphate sequestration with special reference to milk. Dairy Sci. Technol. 2015, 95, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Mulet-Cabero, A.-I.; Torcello-Gómez, A.; Saha, S.; Mackie, A.R.; Wilde, P.J.; Brodkorb, A. Impact of caseins and whey proteins ratio and lipid content on in vitro digestion and ex vivo absorption. Food Chem. 2020. [Google Scholar] [CrossRef] [PubMed]
- Grazielle Siqueira Silva, J.; Paula Rebellato, A.; Tamirys dos Santos Caramês, E.; Greiner, R.; Azevedo Lima Pallone, J. In vitro digestion effect on mineral bioaccessibility and antioxidant bioactive compounds of plant-based beverages. Food Res. Int. 2020. [Google Scholar] [CrossRef]
- Theodoropoulos, V.C.T.; Turatti, M.A.; Greiner, R.; Macedo, G.A.; Pallone, J.A.L. Effect of enzymatic treatment on phytate content and mineral bioacessability in soy drink. Food Res. Int. 2018, 108, 68–73. [Google Scholar] [CrossRef]
- Lacerda Sanches, V.; Alves Peixoto, R.R.; Cadore, S. Phosphorus and zinc are less bioaccessible in soy-based beverages in comparison to bovine milk. J. Funct. Foods 2020, 65, 103728. [Google Scholar] [CrossRef]
- Zhao, Y.; Martin, B.R.; Weaver, C.M. Calcium Bioavailability of Calcium Carbonate Fortified Soymilk Is Equivalent to Cow’s Milk in Young Women. J. Nutr. 2005, 135, 2379–2382. [Google Scholar] [CrossRef]
- Cakebread, J.A.; Wallace, O.A.M.; Kruger, M.C.; Vickers, M.H.; Hodgkinson, A.J. Supplementation with Bovine Milk or Soy Beverages Recovers Bone Mineralization in Young Growing Rats Fed an Insufficient Diet, in Contrast to an Almond Beverage. Curr. Dev. Nutr. 2019, 3. [Google Scholar] [CrossRef]
- Rao, V.; Rao, L. Probiotics and Prebiotics in Human Nutrition and Health; IntechOpen: Rijeka, Croatia, 2016. [Google Scholar]
- Wallig, M.A.; Keenan, K.P. Chapter 36—Nutritional Toxicologic Pathology. In Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed.; Haschek, W.M., Rousseaux, C.G., Wallig, M.A., Eds.; Academic Press: Boston, MA, USA, 2013; pp. 1077–1121. [Google Scholar]
- van der Riet, W.B.; Wight, A.W.; Cilliers, J.J.L.; Datel, J.M. Food chemical analysis of tempeh prepared from South African-grown soybeans. Food Chem. 1987, 25, 197–206. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Laiño, J.E.; del Valle, M.J.; Vannini, V.; van Sinderen, D.; Taranto, M.P.; de Valdez, G.F.; de Giori, G.S.; Sesma, F. B-Group vitamin production by lactic acid bacteria-current knowledge and potential applications. J. Appl. Microbiol. 2011, 111, 1297–1309. [Google Scholar] [CrossRef]
- De Angelis, M.; Bottacini, F.; Fosso, B.; Kelleher, P.; Calasso, M.; Di Cagno, R.; Ventura, M.; Picardi, E.; van Sinderen, D.; Gobbetti, M. Lactobacillus rossiae, a vitamin B12 producer, represents a metabolically versatile species within the Genus Lactobacillus. PLoS ONE 2014, 9, e107232. [Google Scholar] [CrossRef] [PubMed]
- Öztürk, B. Nanoemulsions for food fortification with lipophilic vitamins: Production challenges, stability, and bioavailability. Eur. J. Lipid Sci. Technol. 2017, 119, 1500539. [Google Scholar] [CrossRef]
- Rovoli, M.; Pappas, I.; Lalas, S.; Gortzi, O.; Kontopidis, G. In vitro and in vivo assessment of vitamin A encapsulation in a liposome–protein delivery system. J. Liposome Res. 2019, 29, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Joye, I. Protein digestibility of cereal products. Foods 2019, 8, 199. [Google Scholar] [CrossRef] [PubMed]
- Bessada, S.M.; Barreira, J.C.; Oliveira, M.B.P. Pulses and food security: Dietary protein, digestibility, bioactive and functional properties. Trends Food Sci. Technol. 2019. [Google Scholar] [CrossRef]
- Sá, A.G.A.; Moreno, Y.M.F.; Carciofi, B.A.M. Food processing for the improvement of plant proteins digestibility. Crit. Rev. Food Sci. Nutr. 2019, 1–20, Epub ahead of print. [Google Scholar]
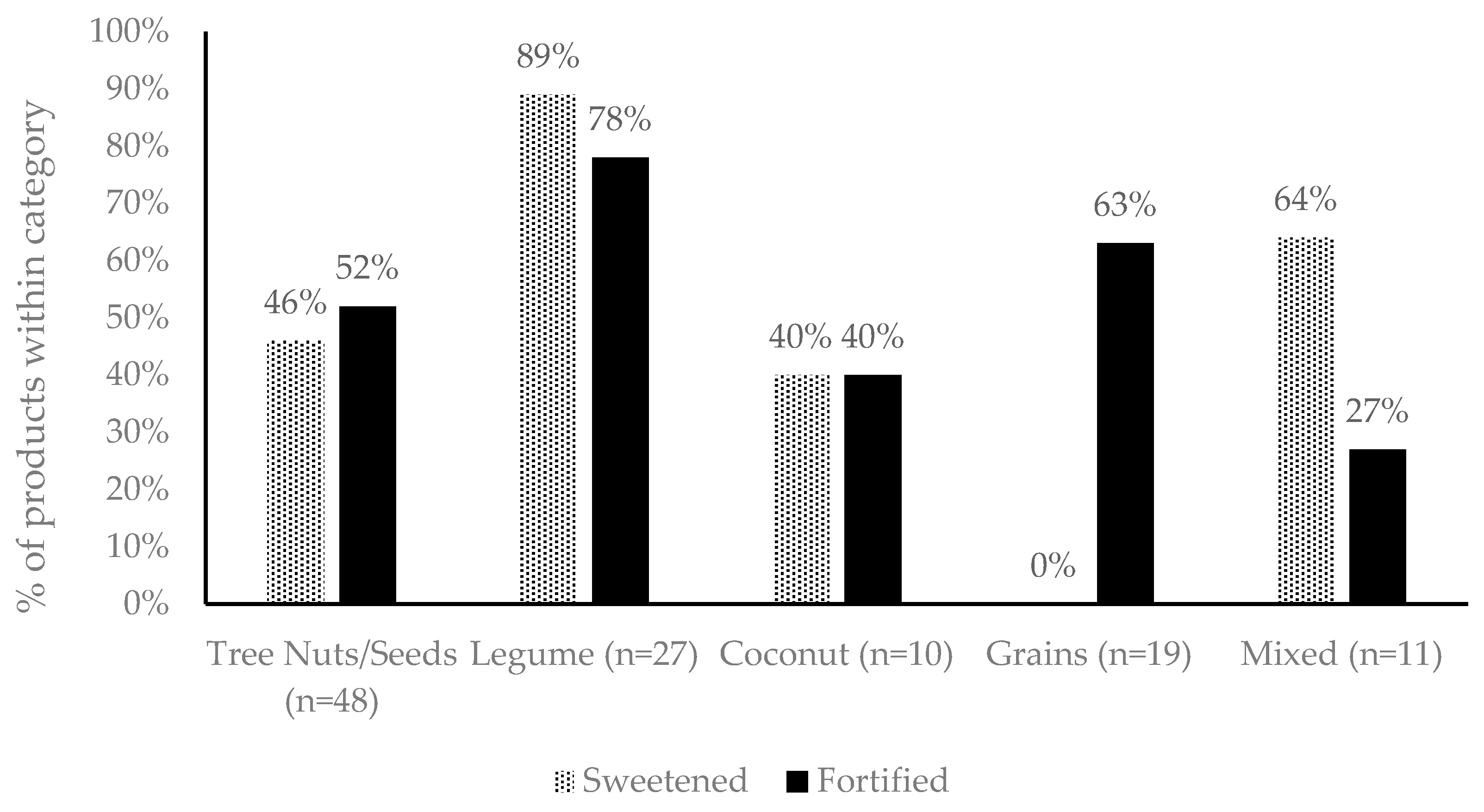
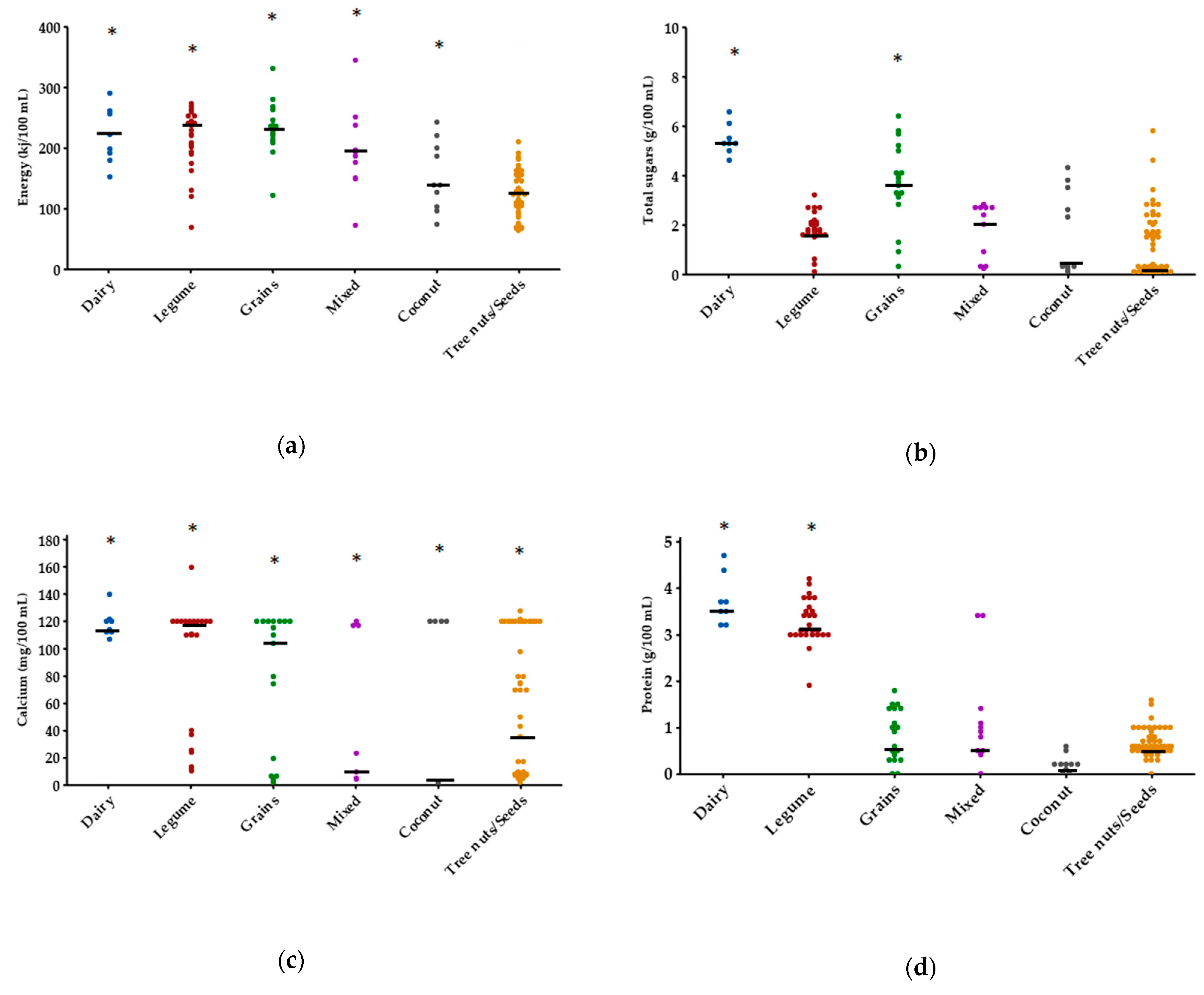
| Group | Ingredients Included |
|---|---|
| Tree nuts and seeds | Almond, Cashew, Macadamia, Hazelnut, Hemp |
| Legume | Soybean, pea |
| Coconut | Coconut Milk/Cream |
| Grains | Rice, Oat, Quinoa |
| Mixed 1 | Almond, Hazelnut, Coconut Cream, Oat, Cashew, Faba, Pea, Rice, Chia |
| Term | Definition |
|---|---|
| Sugars | Total sugars present in the product per 100 mL. Includes both free and intrinsic hexose monosaccharides and disaccharides. 1 |
| Sweetened | A product, containing one of the following ingredients: cane sugar, brown rice syrup, agave or tapioca syrup. 2 |
| Fortified | A product, containing micronutrients in addition to those intrinsically present in the original material, which are permitted for voluntary addition into plant-based beverages by FSANZ. Includes vitamin A, riboflavin (B2), cobalamin (B12), vitamin D, calcium, phosphorous and iodine. Added protein isolates were excluded from this definition. |
| Cow’s Milk | Tree Nuts/Seeds | Legumes | Coconut | Grains | Mixed Sources | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | Range | Median | Range | Median | Range | Median | Range | Median | Range | Median | Range | |
| Total fat (g) | 1.4 | 0–3.5 | 1.9 | 0.8–3.1 | 2.8 | 0–3.5 | 2.2 | 1.8–3.7 | 1.2 | 0–3.4 | 2.9 | 1.5–6.6 |
| Saturated fat (g) | 0.85 | 0–2.3 | 0.2 * | 0–0.4 | 0.3 | 0–0.8 | 2.1 | 1.4–3.4 | 0.17 | 0–0.5 | 0.5 | 0–4.8 |
| Dietary fibre (g) | 0 | 0–0.1 | 0 | 0–1.9 | 0 | 0–1.4 | 0 | 0–0.9 | 0 | 0–1.3 | 0 | 0–1 |
| Zinc (mg) | 0.39 | 0.12–0.48 | 0.11 * | 0.03–0.69 | 0.19 | 0.14–0.66 | 0.02 * | 0.02–0.06 | 0.22 | 0.07–0.37 | 0.25 | 0.09–0.26 |
| Sodium (mg) | 40.5 | 36–47 | 44 | 22–140 | 57 | 16–93 | 44.5 | 20–100 | 58 | 37–110 | 46 | 9–110 |
| Phosphorous (mg) | 94.5 | 85–119 | 17.05 * | 5–115.5 | 87.75 | 31.04–108 | 3 * | 2–8 | 30 * | 16–87 | 73 | 26.1–103 |
| Retinol equivalents (µg) | 18.5 | 0–54 | 0 * | 0–110 | 0.64 | 0–82 | 0 * | 0 | 0 * | 0 | 0 * | 0 |
| Riboflavin/Vitamin B2 (µg) | 0.21 | 0.16–0.23 | 0.0024 * | 0–0.17 | 0.04 | 0.004–0.42 | 0 * | 0 | 0.001 * | 0–0.002 | 0.007 | 0.003–0.17 |
| Cobalamin/Vitamin B12 (µg) | 0.6 | 0.5–0.8 | 0 * | 0–0.4 | 0 * | 0–0.9 | 0 * | 0 | 0 * | 0 | 0.4 | 0–0.4 |
| Iodine (µg) | 24.9 | 18.7–29.1 | 0 * | 0 | 0 * | 0–1.4 | 0.15 * | 0.1–0.4 | 5.2 | 0–11.8 | 0 * | 0–5.6 |
| Energy (kJ) | % EER 1 | Protein (g) | % EAR | Sugars (g) | % Energy | Calcium (mg) | % EAR | Zinc (mg) | % EAR | |
|---|---|---|---|---|---|---|---|---|---|---|
| Cow’s milk | 1838 | 13–17 | 32 | 64–90 | 46 | 0.3–7 | 1024 | 98 | 3.5 | 31–58 |
| Tree nuts/Seeds | 1076 | 8–10 | 5.3 | 11–15 | 9.6 | 0.1–2 | 407 | 39 | 1 | 9–16 |
| Legumes | 2004 | 14–18 | 27 | 55–78 | 16 | 0.1–2 | 1033 | 98 | 1.7 | 15–28 |
| Grains | 2021 | 14–19 | 7.9 | 16–23 | 32 | 0.2–5 | 910 | 87 | 1.9 | 18–32 |
| Coconut | 1216 | 9–11 | 1.8 | 4–5 | 11 | 0.1–2 | 5.3 | 0.5 | 0.2 | 2–3 |
| Mixed | 1689 | 12–15 | 7.9 | 16–23 | 21 | 0.2–3 | 82 | 8 | 2 | 20–36 |
| Energy (kJ) | % EER 1 | Protein (g) | % EAR | Sugars (g) | % Energy | Calcium (mg) | % EAR | Zinc (mg) | % EAR | |
|---|---|---|---|---|---|---|---|---|---|---|
| Cow’s milk | 2100 | 27 | 36 | 97 | 53 | 11 | 1170 | 106 | 3.95 | 61 |
| Tree nuts/Seeds | 1230 | 16 | 6 | 16 | 11 | 2 | 465 | 42 | 1.09 | 17 |
| Legumes | 2290 | 29 | 31 | 84 | 18 | 4 | 1180 | 107 | 1.9 | 29 |
| Grains | 2310 | 29 | 9 | 24 | 37 | 8 | 1040 | 95 | 2.2 | 34 |
| Coconut | 1390 | 18 | 2 | 5 | 13 | 3 | 6 | 0.6 | 0.2 | 3 |
| Mixed | 1930 | 24 | 9 | 24 | 24 | 5 | 94 | 9 | 2.5 | 38 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.Y.; Hughes, J.; Grafenauer, S. Got Mylk? The Emerging Role of Australian Plant-Based Milk Alternatives as A Cow’s Milk Substitute. Nutrients 2020, 12, 1254. https://doi.org/10.3390/nu12051254
Zhang YY, Hughes J, Grafenauer S. Got Mylk? The Emerging Role of Australian Plant-Based Milk Alternatives as A Cow’s Milk Substitute. Nutrients. 2020; 12(5):1254. https://doi.org/10.3390/nu12051254
Chicago/Turabian StyleZhang, Yianna Y., Jaimee Hughes, and Sara Grafenauer. 2020. "Got Mylk? The Emerging Role of Australian Plant-Based Milk Alternatives as A Cow’s Milk Substitute" Nutrients 12, no. 5: 1254. https://doi.org/10.3390/nu12051254
APA StyleZhang, Y. Y., Hughes, J., & Grafenauer, S. (2020). Got Mylk? The Emerging Role of Australian Plant-Based Milk Alternatives as A Cow’s Milk Substitute. Nutrients, 12(5), 1254. https://doi.org/10.3390/nu12051254






