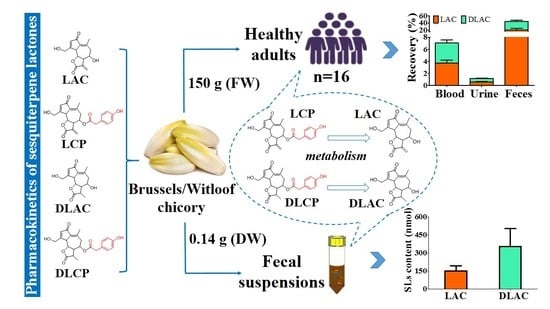
Low Oral Bioavailability and Partial Gut Microbiotic and Phase II Metabolism of Brussels/Witloof Chicory Sesquiterpene Lactones in Healthy Humans
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Clinical Pharmacokinetic Study
2.3. Sample Preparation
2.4. HPLC Method
2.5. Pharmacokinetic Analysis
2.6. Catabolism of SLs by Human Fecal Suspensions
2.7. Statistical Analysis
3. Results
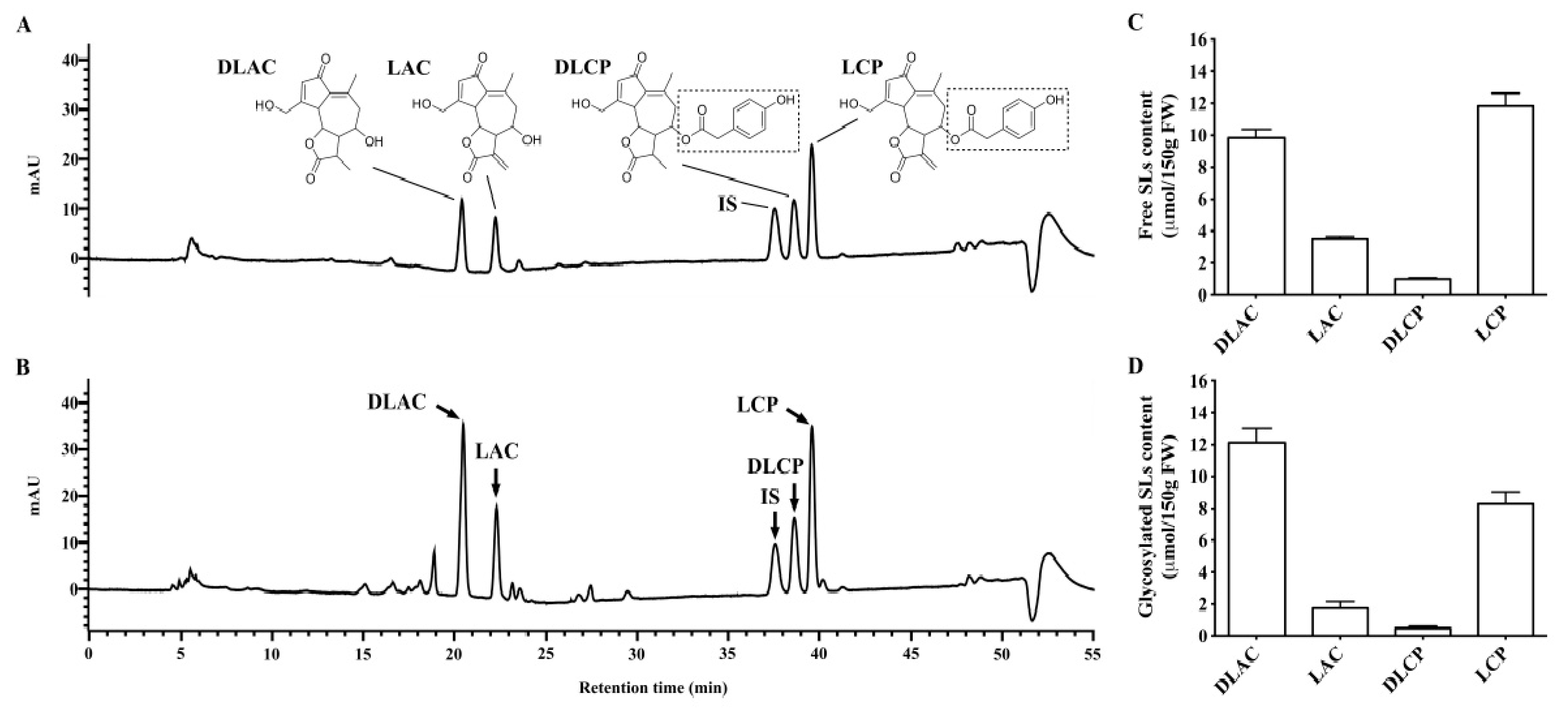
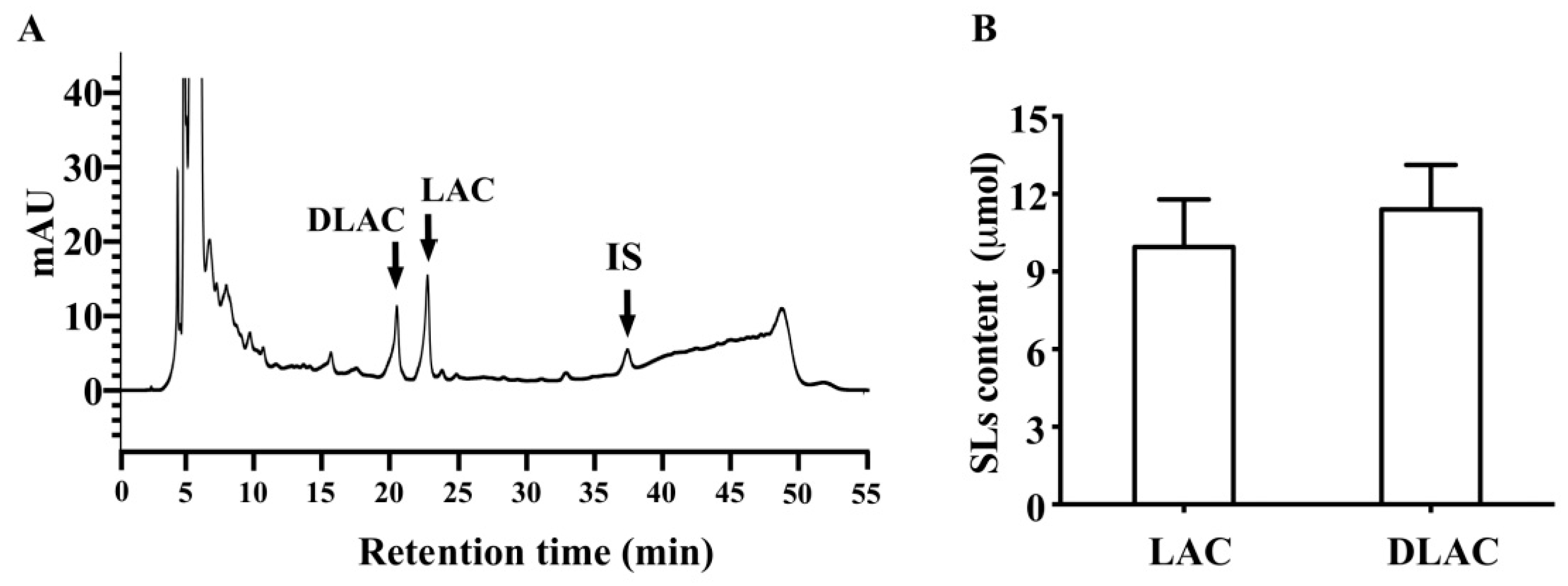
3.1. SLs in Brussels/Witloof Chicory
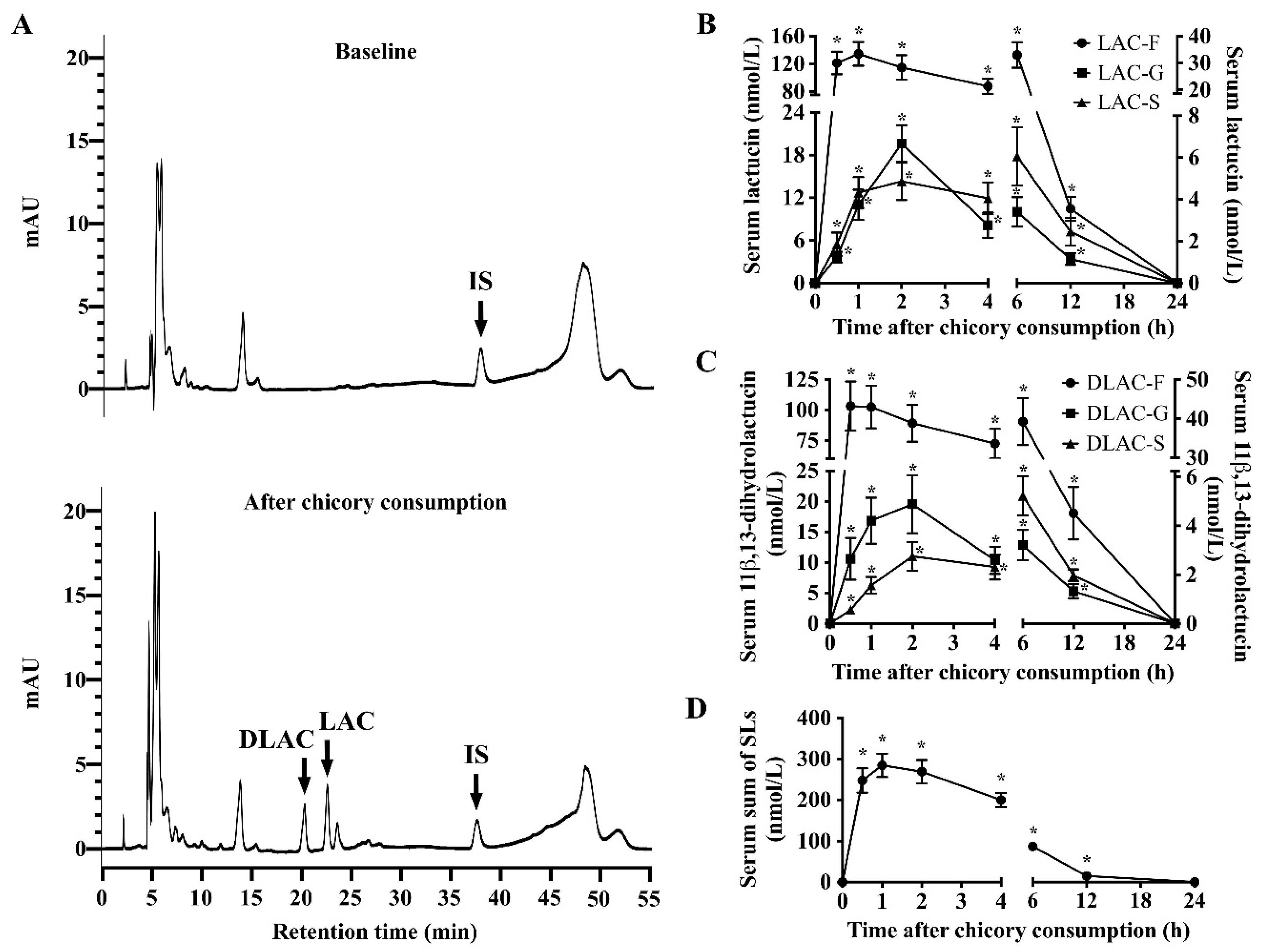
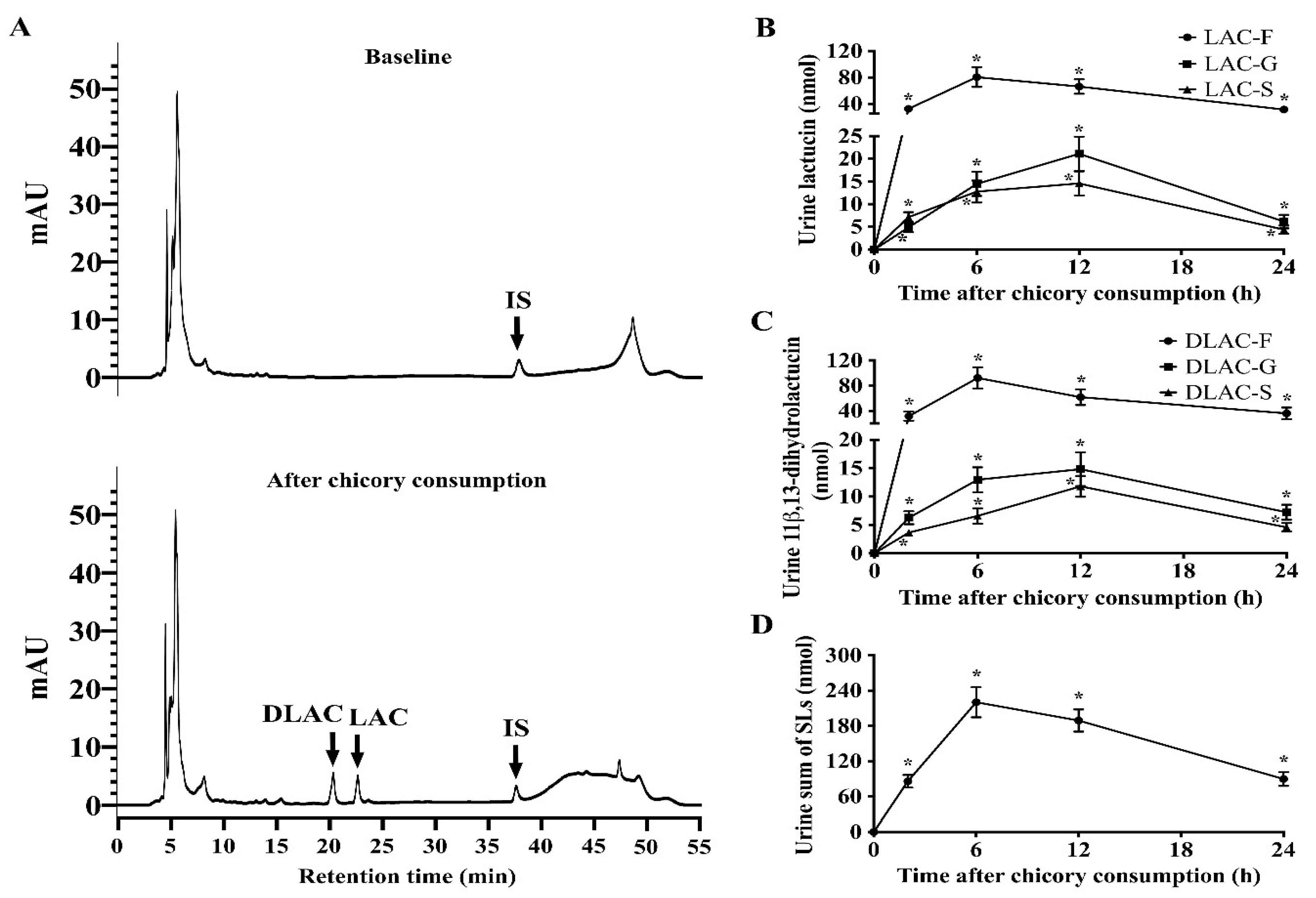
3.2. SLs and Metabolites in Serum, Urine and Feces
3.3. Recovery of SLs and Metabolites in Serum, Urine and Feces
3.4. Catabolism of SLs by Human Fecal Suspensions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ghantous, A.; Gali-Muhtasib, H.; Vuorela, H.; Saliba, N.A.; Darwiche, N. What made sesquiterpene lactones reach cancer clinical trials? Drug Discov. Today 2010, 15, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Amorim, M.H.; Gil, D.C.R.; Lopes, C.; Bastos, M.M. Sesquiterpene lactones: Adverse health effects and toxicity mechanisms. Crit. Rev. Toxicol. 2013, 43, 559–579. [Google Scholar] [CrossRef] [PubMed]
- Chadwick, M.; Trewin, H.; Gawthrop, F.; Wagstaff, C. Sesquiterpenoids lactones: Benefits to plants and people. Int. J. Mol. Sci. 2013, 14, 12780–12805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Price, K.R.; Dupont, M.S.; Shepherd, R.; Chan, H.W.S.; Fenwick, G.R. Relationship between the Chemical and Sensory Properties of Exotic Salad Crops -Coloured Lettuce (Lactuca sativa) and Chicory (Cichorium intybus. J. Sci. Food Agric. 1990, 53, 185–192. [Google Scholar] [CrossRef]
- Wulfkuehler, S.; Gras, C.; Carle, R. Influence of light exposure during storage on the content of sesquiterpene lactones and photosynthetic pigments in witloof chicory (Cichorium intybus L. var. foliosum Hegi). LWT Food Sci. Technol. 2014, 58, 417–426. [Google Scholar] [CrossRef]
- Wesolowska, A.; Nikiforuk, A.; Michalska, K.; Kisiel, W.; Chojnacka-Wojcik, E. Analgesic and sedative activities of lactucin and some lactucin-like guaianolides in mice. J. Ethnopharmacol. 2006, 107, 254–258. [Google Scholar] [CrossRef]
- Lin, W.; Liu, C.; Yang, H.; Wang, W.; Ling, W.; Wang, D. Chicory, a typical vegetable in Mediterranean diet, exerts a therapeutic role in established atherosclerosis in apolipoprotein E-deficient mice. Mol. Nutr. Food Res. 2015, 59, 1803–1813. [Google Scholar] [CrossRef]
- Liu, C.; Wang, W.; Lin, W.; Ling, W.; Wang, D. Established atherosclerosis might be a prerequisite for chicory and its constituent protocatechuic acid to promote endothelium-dependent vasodilation in mice. Mol. Nutr. Food Res. 2016, 60, 2141–2150. [Google Scholar] [CrossRef]
- Street, R.A.; Sidana, J.; Prinsloo, G. Cichorium intybus: Traditional Uses, Phytochemistry, Pharmacology, and Toxicology. Evid. Based Complement. Altern. 2013, 2013, 579319. [Google Scholar] [CrossRef] [Green Version]
- Nwafor, I.C.; Shale, K.; Achilonu, M.C. Chemical Composition and Nutritive Benefits of Chicory (Cichorium intybus) as an Ideal Complementary and/or Alternative Livestock Feed Supplement. Sci. World J. 2017, 2017, 7343928. [Google Scholar] [CrossRef] [Green Version]
- Bischoff, T.A.; Kelley, C.J.; Karchesy, Y.; Laurantos, M.; Nguyen-Dinh, P.; Arefi, A.G. Antimalarial activity of lactucin and lactucopicrin: Sesquiterpene lactones isolated from Cichorium intybus L. J. Ethnopharmacol. 2004, 95, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.L.; Zhou, Y.W.; Chen, X.Z.; Ye, Y.H. Discovery, structural determination and anticancer activities of lactucin-like guaianolides. Lett. Drug Des. Discov. 2005, 2, 444–450. [Google Scholar] [CrossRef]
- Zhang, F.H.; Yan, Y.L.; Wang, Y.; Liu, Z. Lactucin induces potent anti-cancer effects in HL-60 human leukemia cancer cells by inducing apoptosis and sub-G1 cell cycle arrest. Bangl. J. Pharmacol. 2016, 11, 478–484. [Google Scholar] [CrossRef] [Green Version]
- Venkatesan, R.; Subedi, L.; Yeo, E.J.; Kim, S.Y. Lactucopicrin ameliorates oxidative stress mediated by scopolamine-induced neurotoxicity through activation of the NRF2 pathway. Neurochem. Int. 2016, 99, 133–146. [Google Scholar] [CrossRef]
- Venkatesan, R.; Shim, W.S.; Yeo, E.J.; Kim, S.Y. Lactucopicrin potentiates neuritogenesis and neurotrophic effects by regulating Ca(2+)/CaMKII/ATF1 signaling pathway. J. Ethnopharmacol. 2017, 198, 174–183. [Google Scholar] [CrossRef]
- Garcia, C.J.; Beltran, D.; Tomas-Barberan, F.A. Human Gut Microbiota Metabolism of Dietary Sesquiterpene Lactones: Untargeted Metabolomics Study of Lactucopicrin and Lactucin Conversion In Vitro and In Vivo. Mol. Nutr. Food Res. 2020, 64, e2000619. [Google Scholar] [CrossRef]
- Zheng, J.; Xiong, H.; Li, Q.; He, L.; Weng, H.; Ling, W.; Wang, D. Protocatechuic acid from chicory is bioavailable and undergoes partial glucuronidation and sulfation in healthy humans. Food Sci. Nutr. 2019, 7, 3071–3080. [Google Scholar] [CrossRef] [Green Version]
- Ferioli, F.; D’Antuono, L.F. An update procedure for an effective and simultaneous extraction of sesquiterpene lactones and phenolics from chicory. Food Chem. 2012, 135, 243–250. [Google Scholar] [CrossRef]
- Xu, R.; Zhou, G.; Peng, Y.; Wang, M.; Li, X. Pharmacokinetics, tissue distribution and excretion of isoalantolactone and alantolactone in rats after oral administration of Radix Inulae extract. Molecules 2015, 20, 7719–7736. [Google Scholar] [CrossRef] [Green Version]
- Shelnutt, S.R.; Cimino, C.O.; Wiggins, P.A.; Ronis, M.J.; Badger, T.M. Pharmacokinetics of the glucuronide and sulfate conjugates of genistein and daidzein in men and women after consumption of a soy beverage. Am. J. Clin. Nutr. 2002, 76, 588–594. [Google Scholar] [CrossRef] [Green Version]
- Andreasen, M.F.; Kroon, P.A.; Williamson, G.; Garcia-Conesa, M.T. Esterase activity able to hydrolyze dietary antioxidant hydroxycinnamates is distributed along the intestine of mammals. J. Agric. Food Chem. 2001, 49, 5679–5684. [Google Scholar] [CrossRef] [PubMed]
- Gonthier, M.P.; Remesy, C.; Scalbert, A.; Cheynier, V.; Souquet, J.M.; Poutanen, K.; Aura, A.M. Microbial metabolism of caffeic acid and its esters chlorogenic and caftaric acids by human faecal microbiota in vitro. Biomed. Pharmacother. 2006, 60, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Broudiscou, L.; Cornu, A.; Rouzeau, A. In vitro degradation of 10 mono- and sesquiterpenes of plant origin by caprine rumen micro-organisms. J. Sci. Food Agric. 2007, 87, 1653–1658. [Google Scholar] [CrossRef]
- Malecky, M.; Albarello, H.; Broudiscou, L.P. Degradation of terpenes and terpenoids from Mediterranean rangelands by mixed rumen bacteria in vitro. Animal 2012, 6, 612–616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Compound | Cmax (nmol·L−1) | Tmax (h) | T1/2z (h) | AUC0–24 (nmol·h·L−1) |
|---|---|---|---|---|
| LAC-F | 142 ± 17.9 | 0.88 ± 0.12 | 2.13 ± 0.22 | 649 ± 84.3 |
| LAC-G | 19.8 ± 2.54 | 2.06 ± 0.14 | 2.80 ± 0.23 | 71.6 ± 10.8 |
| LAC-S | 16.3 ± 2.65 | 2.81 ± 0.31 | 4.14 ± 0.38 | 87.1 ± 14.5 |
| DLAC-F | 116 ± 19.5 | 0.78 ± 0.10 | 2.53 ± 0.22 | 563 ± 96.3 |
| DLAC-G | 22.1 ± 4.44 | 2.69 ± 0.30 | 3.17 ± 0.37 | 83.3 ± 15.2 |
| DLAC-S | 13.2 ± 2.17 | 2.38 ± 0.29 | 5.01 ± 0.55 | 67.1 ± 8.87 |
| Total Sesquiterpene Lactones | Recovery (of % Oral Consumption) | ||
|---|---|---|---|
| Serum | Urine | Feces | |
| Free | 5.51 ± 0.13 | 0.83 ± 0.03 | 43.75 ± 4.34 |
| Glucuronide | 0.75 ± 0.07 | 0.17 ± 0.02 | ND |
| Sulfate | 0.78 ± 0.11 | 0.13 ± 0.02 | ND |
| Compound (μmol) | Incubation Time (h) with Active Fecal Suspensions | Incubation Time (h) with Heat-Inactivated Fecal Suspensions | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.5 | 1 | 2 | 24 | 0 | 0.5 | 1 | 2 | 24 | |
| LCP | 0.84 ± 0.04 | 0.28 ± 0.07 * | 0.21 ± 0.10 * | ND | ND | 0.86 ± 0.03 | 0.91 ± 0.07 | 0.88 ± 0.08 | 0.86 ± 0.06 | 0.81 ± 0.12 |
| LACb | ND | 0.43 ± 0.16 * | 0.48 ± 0.13 * | 0.59 ± 0.17 * | 0.38 ± 0.14 * | ND | ND | ND | ND | ND |
| DLCP | 0.89 ± 0.05 | 0.34 ± 0.08 * | 0.16 ± 0.11 * | ND | ND | 0.91 ± 0.06 | 0.90 ± 0.05 | 0.84 ± 0.15 | 0.85 ± 0.11 | 0.83 ± 0.09 |
| DLACb | ND | 0.39 ± 0.15 * | 0.53 ± 0.12 * | 0.62 ± 0.14 * | 0.44 ± 0.16 * | ND | ND | ND | ND | ND |
| Compound (nmol) | Incubation Time (h) with Active Fecal Suspensions | Incubation Time (h) with Heat-Inactivated Fecal Suspensions | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 0.5 | 1 | 2 | 24 | 0 | 0.5 | 1 | 2 | 24 | |
| LAC | 50.4 ± 2.84 | 89.0 ± 14.6 * | 147 ± 32.7 * | 211 ± 64.1 * | 159 ± 43.0 * | 48.3 ± 2.07 | 42.0 ± 7.49 | 46.3 ± 6.82 | 42.2 ± 6.07 | 45.0 ± 10.3 |
| DLAC | 158 ± 7.43 | 274 ± 46.1 * | 435 ± 111 * | 514 ± 132 * | 384 ± 91.1 * | 154 ± 5.52 | 143 ± 24.4 | 157 ± 41.61 | 141 ± 13.6 | 135.47 ± 19.3 |
| LCP | 211 ± 13.3 | 183 ± 28.9 | 127 ± 20.6 * | ND | ND | 200 ± 10.2 | 209 ± 17.7 | 184 ± 29.4 | 199 ± 31.6 | 204 ± 27.8 |
| DLCP | 17.2 ± 1.14 | 9.76 ± 1.67 * | 4.37 ± 1.04 * | ND | ND | 18.1 ± 0.98 | 16.5 ±1.53 | 16.3 ± 1.28 | 16.6 ± 4.12 | 17.3 ± 3.61 |
| LAC-Gly | 46.1 ± 3.77 | 37.8 ± 4.57 | 13.2 ± 5.93 * | ND | ND | 48.2 ± 2.9 | 44.9 ± 9.47 | 42.6 ± 9.78 | 39.7 ± 8.58 | 42.0 ± 5.68 |
| DLAC-Gly | 249 ± 18.0 | 184 ± 26.0 * | 57.2 ± 17.8 * | ND | ND | 238 ± 15.78 | 226 ± 29.2 | 219 ± 25.5 | 209 ± 46 | 205 ± 43.2 |
| LCP-Gly | 153 ± 9.13 | 55.1 ± 14.1 * | 9.91 ± 2.1* | ND | ND | 149 ± 12.0 | 134 ± 25.6 | 124 ± 20.0 | 128 ± 15.3 | 127 ± 26.9 |
| DLCP-Gly | 9.04 ± 1.07 | 5.67 ± 1.19 * | 1.47 ± 0.75 * | ND | ND | 9.2 ± 1.13 | 8.46 ± 1.35 | 8.04 ± 1.21 | 8.19 ± 1.14 | 7.84 ± 1.36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weng, H.; He, L.; Zheng, J.; Li, Q.; Liu, X.; Wang, D. Low Oral Bioavailability and Partial Gut Microbiotic and Phase II Metabolism of Brussels/Witloof Chicory Sesquiterpene Lactones in Healthy Humans. Nutrients 2020, 12, 3675. https://doi.org/10.3390/nu12123675
Weng H, He L, Zheng J, Li Q, Liu X, Wang D. Low Oral Bioavailability and Partial Gut Microbiotic and Phase II Metabolism of Brussels/Witloof Chicory Sesquiterpene Lactones in Healthy Humans. Nutrients. 2020; 12(12):3675. https://doi.org/10.3390/nu12123675
Chicago/Turabian StyleWeng, Hui, Luanying He, Jiakun Zheng, Qing Li, Xiuping Liu, and Dongliang Wang. 2020. "Low Oral Bioavailability and Partial Gut Microbiotic and Phase II Metabolism of Brussels/Witloof Chicory Sesquiterpene Lactones in Healthy Humans" Nutrients 12, no. 12: 3675. https://doi.org/10.3390/nu12123675
APA StyleWeng, H., He, L., Zheng, J., Li, Q., Liu, X., & Wang, D. (2020). Low Oral Bioavailability and Partial Gut Microbiotic and Phase II Metabolism of Brussels/Witloof Chicory Sesquiterpene Lactones in Healthy Humans. Nutrients, 12(12), 3675. https://doi.org/10.3390/nu12123675