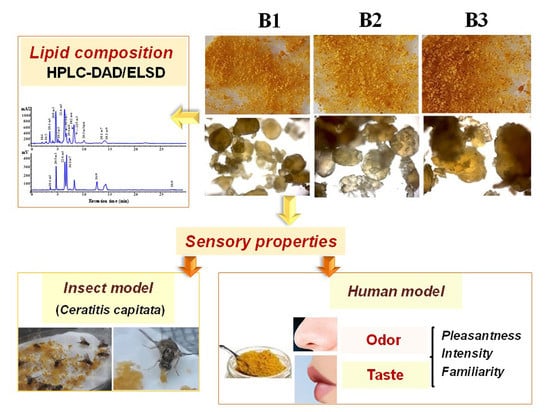
The Role of Lipid Composition in the Sensory Attributes and Acceptability of the Salted and Dried Mullet Roes (Bottarga): A Study in Human and Animal Models
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Bottarga Samples
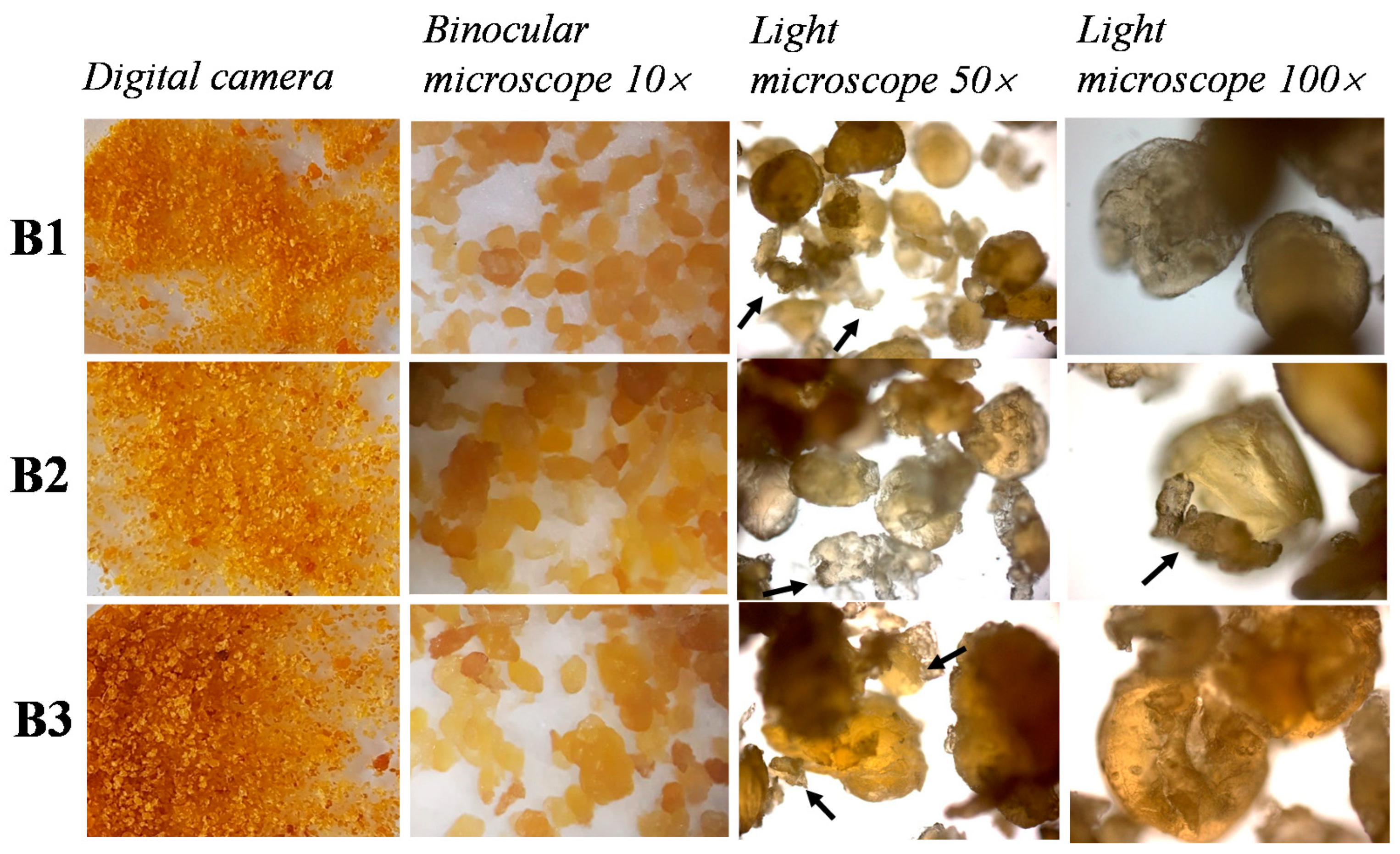
2.3. Microscopic Analysis of Bottarga Samples
2.4. Extraction and Saponification of Lipids
2.5. Analysis of Lipid Components
2.6. Insects
2.7. Behavioral Tests on Medfly
2.8. Participants
2.9. Procedures to Assess Pleasantness, Intensity and Familiarity in Odor and Taste for Bottarga Samples in a Human Model
2.10. Statistical Analyses
3. Results
3.1. Visual and Microscopic Analysis
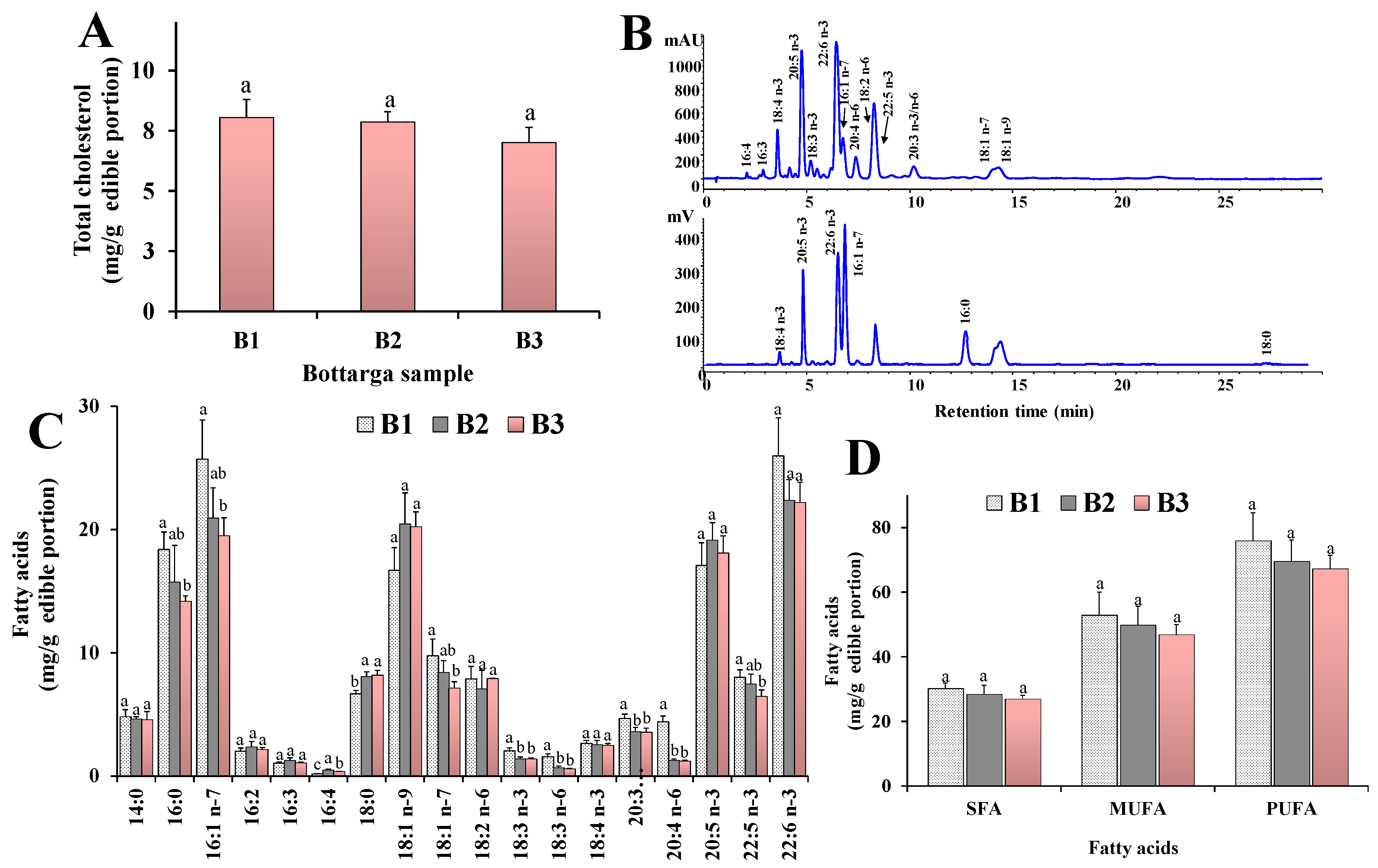
3.2. Main Lipid Components
3.3. Fatty Acid Profile
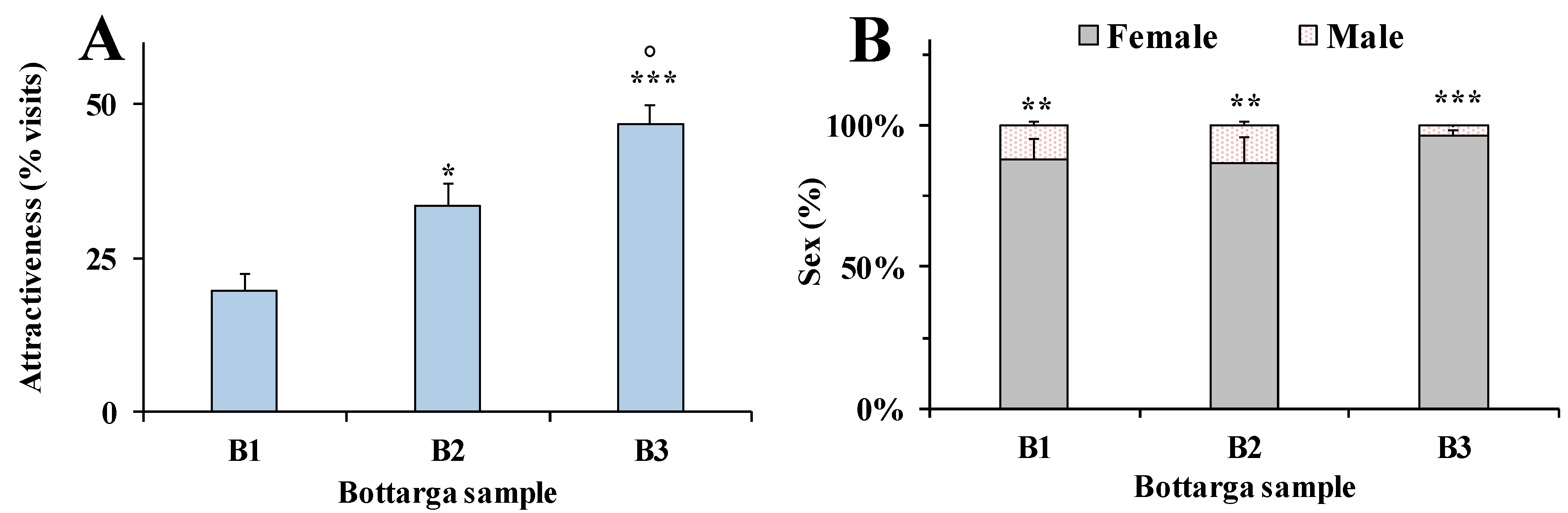
3.4. Behavioral Tests in Insects
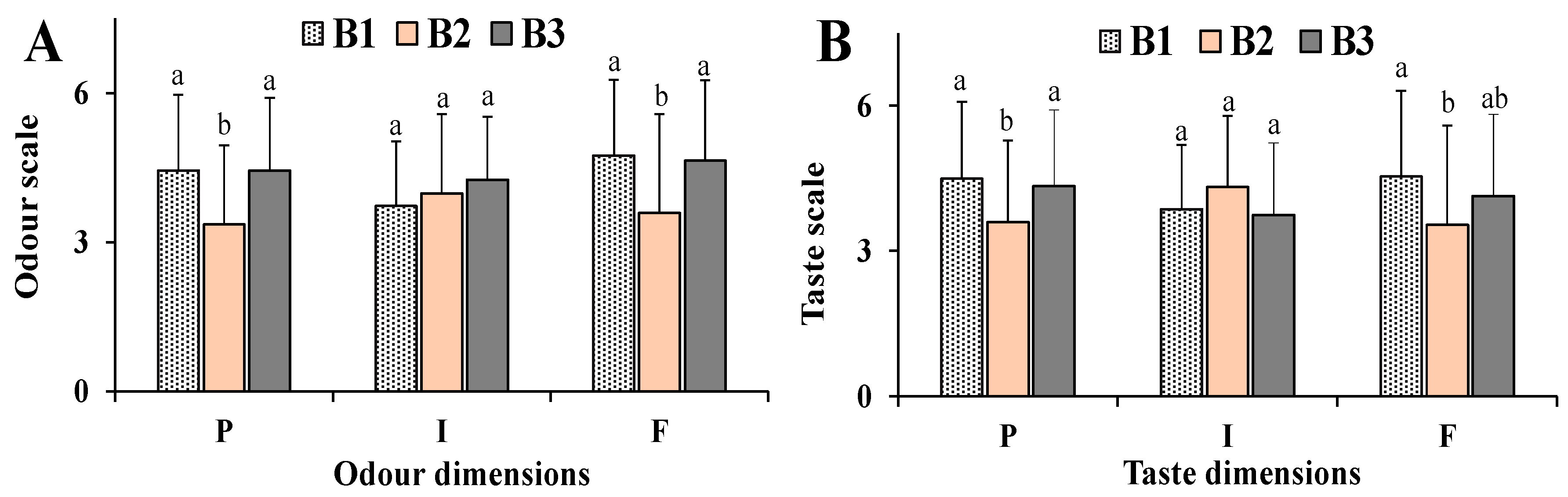
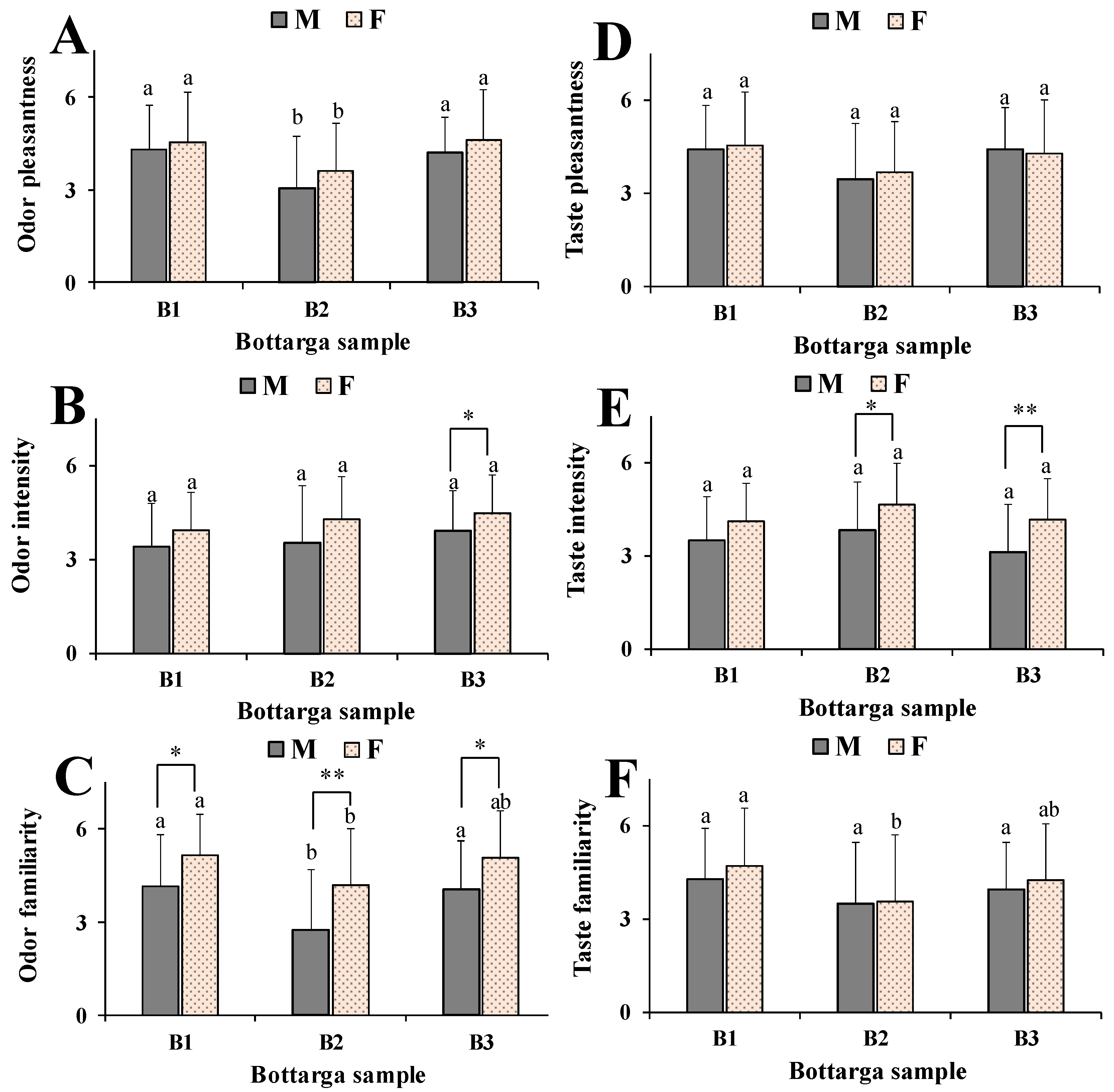
3.5. Human Model
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bledsoe, G.; Bledsoe, C.; Rasco, B.A. Caviars and Fish Roe Products. Crit. Rev. Food Sci. Nutr. 2003, 43, 317–356. [Google Scholar] [CrossRef] [PubMed]
- Kalogeropoulos, N.; Nomikos, T.; Chiou, A.; Fragopoulou, E.; Antonopoulou, S. Chemical Composition of Greek Avgotaracho Prepared from Mullet (Mugil cephalus): Nutritional and Health Benefits. J. Agric. Food Chem. 2008, 56, 5916–5925. [Google Scholar] [CrossRef] [PubMed]
- Cervera, M.R.; Suárez, M.; Guil-Guerrero, J. Fatty acid composition of selected roes from some marine species. Eur. J. Lipid Sci. Technol. 2009, 111, 920–925. [Google Scholar] [CrossRef]
- Bernasconi, R.; Bolzacchini, E.; Galliani, G.; Gugliersi, F.; Rindone, B.; Rindone, M.; Tacconi, M.T.; Terraneo, A. Determination of the content of wax esters in some sea foods and their molecular composition. A comparison with ω-3 enriched wax esters. LWT 2007, 40, 569–573. [Google Scholar] [CrossRef]
- Scano, P.; Rosa, A.; Marincola, F.C.; Locci, E.; Melis, M.; Dessì, M.; Lai, A. 13C NMR, GC and HPLC characterization of lipid components of the salted and dried mullet (Mugil cephalus) roe “bottarga”. Chem. Phys. Lipids 2008, 151, 69–76. [Google Scholar] [CrossRef]
- Caredda, M.; Addis, M.; Pes, M.; Fois, N.; Sanna, G.; Piredda, G.; Sanna, G. Physico-chemical, colorimetric, rheological parameters and chemometric discrimination of the origin of Mugil cephalus’ roes during the manufacturing process of Bottarga. Food Res. Int. 2018, 108, 128–135. [Google Scholar] [CrossRef]
- Piras, P.; Sardu, F.; Meloni, D.; Riina, M.V.; Beltramo, C.; Acutis, P.L. A case study on the labeling of bottarga produced in Sardinia from ovaries of grey mullets (Mugil cephalus and Mugil capurrii) caught in Eastern Central Atlantic coasts. Ital. J. Food Saf. 2018, 7, 6893. [Google Scholar] [CrossRef]
- Scano, P.; Rosa, A.; Mereu, S.; Piras, C.; Atzeri, A.; Dessi, M.A. Multivariate fatty acid and fatty alcohol profile of mullet bottarga. Eur. J. Lipid Sci. Technol. 2010, 112, 1369–1374. [Google Scholar] [CrossRef]
- Rosa, A.; Scano, P.; Melis, M.P.; Deiana, M.; Atzeri, A.; Dessi, M.A. Oxidative stability of lipid components of mullet (Mugil cephalus) roe and its product “bottarga”. Food Chem. 2009, 115, 891–896. [Google Scholar] [CrossRef]
- Rosa, A.; Scano, P.; Atzeri, A.; Deiana, M.; Mereu, S.; Dessi, M.A. Effect of Storage Conditions on Lipid Components and Color of Mugil cephalus Processed Roes. J. Food Sci. 2011, 77, C107–C114. [Google Scholar] [CrossRef]
- Rosa, A.; Atzeri, A.; Deiana, M.; Melis, M.P.; Loru, D.; Incani, A.; Cabboi, B.; Dessì, M.A. Effect of Aqueous and Lipophilic Mullet (Mugil cephalus) Bottarga Extracts on the Growth and Lipid Profile of Intestinal Caco-2 Cells. J. Agric. Food Chem. 2011, 59, 1658–1666. [Google Scholar] [CrossRef]
- Rosa, A.; Scano, P.; Atzeri, A.; Deiana, M.; Falchi, A.M. Potential anti-tumor effects of Mugil cephalus processed roe extracts on colon cancer cells. Food Chem. Toxicol. 2013, 60, 471–478. [Google Scholar] [CrossRef]
- Rosa, A.; Piras, A.; Nieddu, M.; Putzu, D.; Falchi, A.M.; Marincola, F.C. Mugil cephalus roe oil obtained by supercritical fluid extraction affects the lipid profile and viability in cancer HeLa and B16F10 cells. Food Funct. 2016, 7, 4092–4103. [Google Scholar] [CrossRef]
- Rosa, A.; Atzeri, A.; Putzu, D.; Scano, P. A diet enriched with Mugil cephalus processed roes modulates the tissue lipid profile in healthy rats: A biochemical and chemometric assessment. Food Funct. 2016, 7, 202–211. [Google Scholar] [CrossRef]
- Scano, P.; Rosa, A.; Locci, E.; Dessi, M.A.; Lai, A. NMR study of the lipid profile of mullet raw roe and bottarga. Eur. J. Lipid Sci. Technol. 2009, 111, 505–512. [Google Scholar] [CrossRef]
- Civille, G.V. Food quality: Consumer acceptance and sensory attributes. J. Food Qual. 1991, 14, 1–8. [Google Scholar] [CrossRef]
- Costell, E.; Tárrega, A.; Bayarri, S. Food Acceptance: The Role of Consumer Perception and Attitudes. Chemosens. Percept. 2009, 3, 42–50. [Google Scholar] [CrossRef]
- McCrickerd, K.; Forde, C.G. Sensory influences on food intake control: Moving beyond palatability. Obes. Rev. 2015, 17, 18–29. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, S.-Y.; Schmidt, S.J. Probing the Sensory Properties of Food Materials with Nuclear Magnetic Resonance Spectroscopy and Imaging. In Modern Magnetic Resonance; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2008; pp. 1889–1894. [Google Scholar]
- Spence, C. Multisensory Flavor Perception. Cell 2015, 161, 24–35. [Google Scholar] [CrossRef]
- Solari, P.; Sollai, G.; Masala, C.; Loy, F.; Palmas, F.; Sabatini, A.; Crnjar, R. Antennular Morphology and Contribution of Aesthetascs in the Detection of Food-related Compounds in the Shrimp Palaemon adspersus Rathke, 1837 (Decapoda: Palaemonidae). Biol. Bull. 2017, 232, 110–122. [Google Scholar] [CrossRef]
- Besnard, P.; Passilly-Degrace, P.; Khan, N.A. Taste of Fat: A Sixth Taste Modality? Physiol. Rev. 2016, 96, 151–176. [Google Scholar] [CrossRef] [PubMed]
- De Araujo, I.E.; Rolls, E.T. Representation in the Human Brain of Food Texture and Oral Fat. J. Neurosci. 2004, 24, 3086–3093. [Google Scholar] [CrossRef]
- Adachi, S.-I.; Eguchi, A.; Sakamoto, K.; Asano, H.; Manabe, Y.; Matsumura, S.; Tsuzuki, S.; Inoue, K.; Fushiki, T. Behavioral palatability of dietary fatty acids correlates with the intracellular calcium ion levels induced by the fatty acids in GPR120-expressing cells. Biomed. Res. 2014, 35, 357–367. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yang, Z.H.; Miyahara, H.; Doisaki, N.; Furihata, K.; Hatanaka, A. Different palatability of various fish oils to mice: Beta-endorphin may not be involved in the mechanism. Aust. J. Basic Appl. Sci. 2011, 5, 762–770. [Google Scholar]
- Chalé-Rush, A.; Burgess, J.R.; Mattes, R.D. Evidence for Human Orosensory (Taste?) Sensitivity to Free Fatty Acids. Chem. Senses 2007, 32, 423–431. [Google Scholar] [CrossRef]
- Yang, J.; Lee, J. Application of Sensory Descriptive Analysis and Consumer Studies to Investigate Traditional and Authentic Foods: A Review. Foods 2019, 8, 54. [Google Scholar] [CrossRef] [PubMed]
- Rosa, A.; Piras, A.; Carta, G.; Solari, P.; Crnjar, R.; Masala, C. Evaluation of the attractant effect and lipid profile modulation of natural fixed oils on the medfly Ceratitis capitata (Wiedemann). Arch. Insect Biochem. Physiol. 2018, 99, e21508. [Google Scholar] [CrossRef]
- Delplanque, S.; Grandjean, D.; Chrea, C.; Aymard, L.; Cayeux, I.; Le Calvé, B.; Velazco, M.I.; Scherer, K.R.; Sander, D. Emotional Processing of Odors: Evidence for a Nonlinear Relation between Pleasantness and Familiarity Evaluations. Chem. Senses 2008, 33, 469–479. [Google Scholar] [CrossRef]
- Oleszkiewicz, A.; Alizadeh, R.; Altundag, A.; Chen, B.; Corrai, A.; Fanari, R.; Farhadi, M.; Gupta, N.; Habel, R.; Hudson, R. Global study of variability in olfactory sensitivity. Behav. Neurosci. 2020, 134, 394–406. [Google Scholar] [CrossRef]
- Masala, C.; Käehling, C.; Fall, F.; Hummel, T. Correlation between olfactory function, trigeminal sensitivity, and nasal anatomy in healthy subjects. Eur. Arch. Oto-Rhino-Laryngology 2019, 276, 1649–1654. [Google Scholar] [CrossRef]
- Masala, C.; Saba, L.; Cecchini, M.P.; Solla, P.; Loy, F. Olfactory Function and Age: A Sniffin’ Sticks Extended Test Study Performed in Sardinia. Chemosens. Percept. 2017, 11, 19–26. [Google Scholar] [CrossRef]
- Doe, P.E.; Sikorski, Z.; Haard, N.; Olley, J.; Sun Pan, B. Basic principles. In Fish drying & smoking: Production and Quality; Doe, P.E., Ed.; Technomic Publishing Company: Lancaster, PA, USA, 1998; pp. 13–45. [Google Scholar]
- Fougeron, A.-S.; Farine, J.-P.; Flaven-Pouchon, J.; Everaerts, C.; Ferveur, J.-F. Fatty-Acid Preference Changes during Development in Drosophila melanogaster. PLoS ONE 2011, 6, e26899. [Google Scholar] [CrossRef]
- Loy, F.; Solari, P.; Isola, M.; Crnjar, R.; Masala, C. Morphological and electrophysiological analysis of tarsal sensilla in the medfly Ceratitis capitata (Wiedemann, 1824) (Diptera: Tephritidae). Ital. J. Zool. 2016, 83, 456–468. [Google Scholar] [CrossRef]
- Lopez, A.; Vasconi, M.; Bellagamba, F.; Mentasti, T.; Pazzaglia, T.M.M.; Moretti, V. Volatile Organic Compounds Profile in White Sturgeon (Acipenser transmontanus) Caviar at Different Stages of Ripening by Multiple Headspace Solid Phase Microextraction. Molecules 2020, 25, 1074. [Google Scholar] [CrossRef]
- Guiné, R. The Drying of Foods and Its Effect on the Physical-Chemical, Sensorial and Nutritional Properties. ETP Int. J. Food Eng. 2018, 4, 93–100. [Google Scholar] [CrossRef]
- Karam, M.C.; Petit, J.; Zimmer, D.; Djantou, E.B.; Scher, J. Effects of drying and grinding in production of fruit and vegetable powders: A review. J. Food Eng. 2016, 188, 32–49. [Google Scholar] [CrossRef]
- Yin, W.; Hewson, L.; Linforth, R.S.; Taylor, M.A.; Fisk, I.D. Effects of aroma and taste, independently or in combination, on appetite sensation and subsequent food intake. Appetite 2017, 114, 265–274. [Google Scholar] [CrossRef]
- Croy, I.; Negoias, S.; Novakova, L.; Landis, B.N.; Hummel, T. Learning about the Functions of the Olfactory System from People without a Sense of Smell. PLoS ONE 2012, 7, e33365. [Google Scholar] [CrossRef]
- Hartley, I.E.; Liem, D.G.; Keast, R. Umami as an ‘Alimentary’ Taste. A New Perspective on Taste Classification. Nutrients 2019, 11, 182. [Google Scholar] [CrossRef]
- Lim, J. Hedonic scaling: A review of methods and theory. Food Qual. Prefer. 2011, 22, 733–747. [Google Scholar] [CrossRef]
- Boesveldt, S.; Lundström, J.N. Detecting Fat Content of Food from a Distance: Olfactory-Based Fat Discrimination in Humans. PLoS ONE 2014, 9, e85977. [Google Scholar] [CrossRef]
- Kindleysides, S.; Beck, K.L.; Walsh, D.C.I.; Henderson, L.; Jayasinghe, S.N.; Golding, M.; Breier, B. Fat Sensation: Fatty Acid Taste and Olfaction Sensitivity and the Link with Disinhibited Eating Behaviour. Nutrients 2017, 9, 879. [Google Scholar] [CrossRef]
- Delompré, T.; Guichard, E.; Briand, L.; Salles, C. Taste Perception of Nutrients Found in Nutritional Supplements: A Review. Nutrients 2019, 11, 2050. [Google Scholar] [CrossRef]
- Kershaw, J.C.; Mattes, R.D. Nutrition and taste and smell dysfunction. World J. Otorhinolaryngol.-Head Neck Surg. 2018, 4, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Doty, R.L.; Cameron, E.L. Sex differences and reproductive hormone influences on human odor perception. Physiol. Behav. 2009, 97, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Sorokowski, P.; Karwowski, M.; Misiak, M.; Marczak, M.K.; Dziekan, M.; Hummel, T.; Sorokowska, A. Sex Differences in Human Olfaction: A Meta-Analysis. Front. Psychol. 2019, 10, 242. [Google Scholar] [CrossRef] [PubMed]


| Common Name | CA:DB 1 | B1 | B2 | B3 |
|---|---|---|---|---|
| Lauric acid | 12:0 | trace | trace | trace |
| Myristic acid | 14:0 | 3.02 ± 0.16 | 3.14 ± 0.21 | 3.22 ± 0.66 |
| Palmitic acid | 16:0 | 11.78 ± 0.64 | 10.60 ± 0.94 | 10.07 ± 0.43 |
| Palmitoleic acid | 16:1 n-7 | 16.51 ± 0.46 | 14.15 ± 0.26 | 13.81 ± 0.24 |
| Hexadecadienoic acid | 16:2 | 1.29 ± 0.06 | 1.59 ± 0.12 | 1.52 ± 0.10 |
| Hexadecatrienoic acid | 16:3 | 0.65 ± 0.01 | 0.84 ± 0.06 | 0.74 ± 0.01 |
| Hexadecatetranoic acid | 16:4 | 0.10 ± 0.00 | 0.32 ± 0.02 | 0.24 ± 0.01 |
| Stearic acid | 18:0 | 4.24 ± 0.58 | 5.50 ± 0.73 | 5.80 ± 0.08 |
| Oleic acid | 18:1 n-9 | 10.34 ± 0.46 | 13.82 ± 0.38 | 14.34 ± 0.09 |
| cis-Vaccenic acid | 18:1 n-7 | 6.38 ± 0.21 | 5.69 ± 0.24 | 5.05 ± 0.17 |
| Linoleic acid | 18:2 n-6 | 4.68 ± 0.20 | 4.77 ± 0.70 | 5.61 ± 0.34 |
| γ-Linolenic acid | 18:3 n-6 | 0.97 ± 0.07 | 0.44 ± 0.05 | 0.40 ± 0.03 |
| α -Linolenic acid | 18:3 n-3 | 1.29 ± 0.04 | 0.93 ± 0.02 | 0.97 ± 0.04 |
| Stearidonic acid | 18:4 n-3 | 1.66 ± 0.02 | 1.72 ± 0.07 | 1.75 ± 0.04 |
| Eicosatrienoic acid | 20:3 n-6 | 2.94 ± 0.19 | 2.42 ± 0.06 | 2.51 ± 0.11 |
| Arachidonic acid | 20:4 n-6 | 2.74 ± 0.07 | 0.85 ± 0.01 | 0.85 ± 0.03 |
| Eicosapentaenoic acid | 20:5 n-3 | 10.51 ± 0.43 | 12.98 ± 0.58 | 12.82 ± 0.41 |
| Docosapentaenoic acid | 22:5 n-3 | 5.06 ± 0.14 | 5.06 ± 0.25 | 4.57 ± 0.18 |
| Docosahexaenoic acid | 22:6 n-3 | 15.86 ± 0.77 | 15.18 ± 0.43 | 15.73 ± 0.40 |
| Saturated fatty acids | SFA | 19.03 ± 1.06 | 19.24 ± 0.31 | 19.09 ± 0.32 |
| Monounsaturated fatty acids | MUFA | 33.23 ± 0.90 | 33.66 ± 0.51 | 33.20 ± 0.45 |
| Polyunsaturated fatty acids | PUFA | 47.74 ± 0.96 | 47.09 ± 0.37 | 47.71 ± 0.30 |
| Data | FFA | IA | OP | OI | OF | TP | TI | TF |
|---|---|---|---|---|---|---|---|---|
| TFA | 0.9075 | −0.9909 | 0.1451 | −0.9866 | 0.2234 | 0.3002 | 0.0506 | 0.5328 |
| SFA | 0.8550 | −0.9996 | 0.0397 | −0.9983 | 0.1192 | 0.1977 | 0.1559 | 0.4404 |
| MUFA | 0.8466 | −1.0000 | 0.0178 | −0.9993 | 0.0974 | 0.1762 | 0.1775 | 0.4206 |
| PUFA | 0.9528 | −0.9663 | 0.2673 | −0.9585 | 0.3432 | 0.4168 | −0.0743 | 0.6342 |
| FFA | 1 | −0.8426 | 0.5472 | −0.8267 | 0.6122 | 0.6731 | −0.3736 | 0.8390 |
| IA | −0.8426 | 1 | −0.0104 | 0.9996 | −0.0900 | −0.1689 | −0.1848 | −0.4139 |
| OP | 0.5472 | −0.0104 | 1 | 0.0186 | 0.9968 | 0.9873 | −0.9808 | 0.9146 |
| OI | −0.8267 | −0.9996 | 0.0186 | 1 | 0.2234 | −0.1403 | −0.2132 | −0.3873 |
| OF | 0.6122 | −0.0900 | 0.9968 | 0.2234 | 1 | 0.9968 | −0.9622 | 0.9439 |
| TP | 0.6731 | −0.1689 | 0.9873 | −0.1403 | 0.9968 | 1 | −0.9375 | 0.9672 |
| TI | −0.3736 | −0.1848 | −0.9808 | −0.2132 | −0.9622 | −0.9375 | 1 | −0.8182 |
| TF | 0.8390 | −0.4139 | 0.9146 | −0.3873 | 0.9439 | 0.9672 | −0.8182 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosa, A.; Isola, R.; Nieddu, M.; Masala, C. The Role of Lipid Composition in the Sensory Attributes and Acceptability of the Salted and Dried Mullet Roes (Bottarga): A Study in Human and Animal Models. Nutrients 2020, 12, 3454. https://doi.org/10.3390/nu12113454
Rosa A, Isola R, Nieddu M, Masala C. The Role of Lipid Composition in the Sensory Attributes and Acceptability of the Salted and Dried Mullet Roes (Bottarga): A Study in Human and Animal Models. Nutrients. 2020; 12(11):3454. https://doi.org/10.3390/nu12113454
Chicago/Turabian StyleRosa, Antonella, Raffaella Isola, Mariella Nieddu, and Carla Masala. 2020. "The Role of Lipid Composition in the Sensory Attributes and Acceptability of the Salted and Dried Mullet Roes (Bottarga): A Study in Human and Animal Models" Nutrients 12, no. 11: 3454. https://doi.org/10.3390/nu12113454
APA StyleRosa, A., Isola, R., Nieddu, M., & Masala, C. (2020). The Role of Lipid Composition in the Sensory Attributes and Acceptability of the Salted and Dried Mullet Roes (Bottarga): A Study in Human and Animal Models. Nutrients, 12(11), 3454. https://doi.org/10.3390/nu12113454