Polyunsaturated Fatty Acids and Their Metabolites in Hyperemesis Gravidarum
Abstract
1. Introduction
2. Hyperemesis Gravidarum
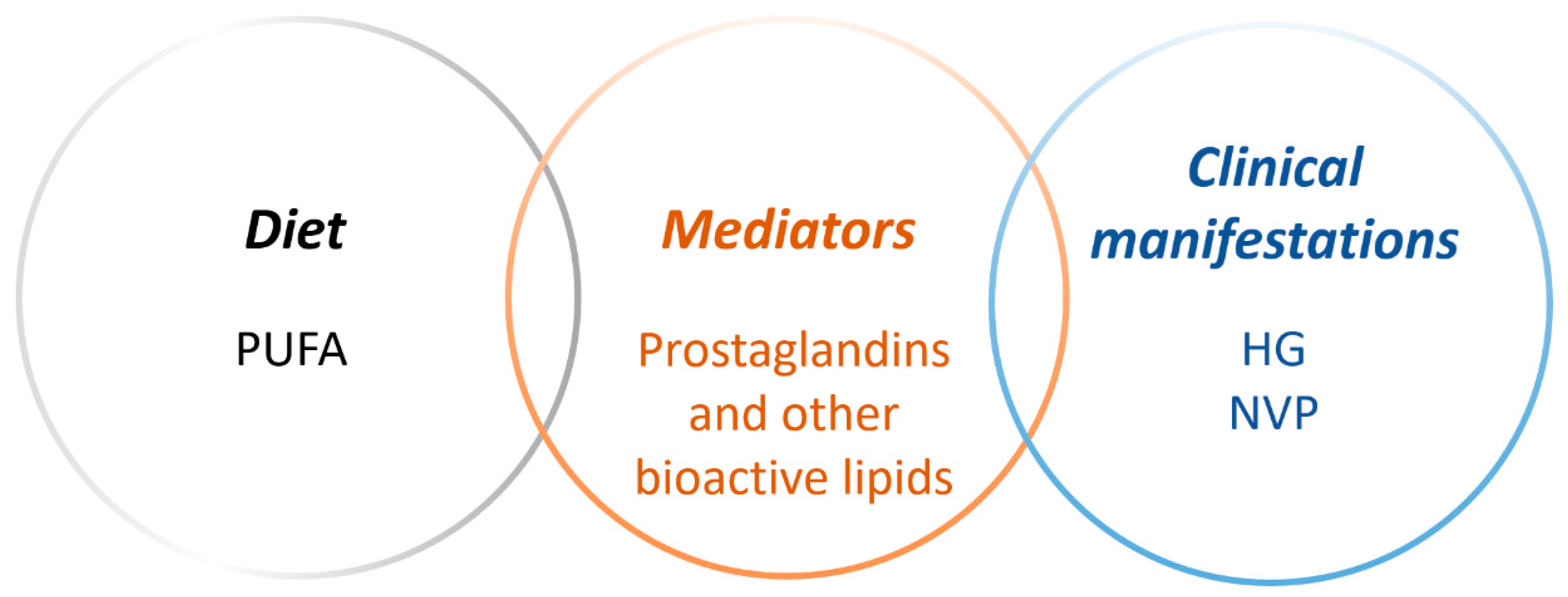
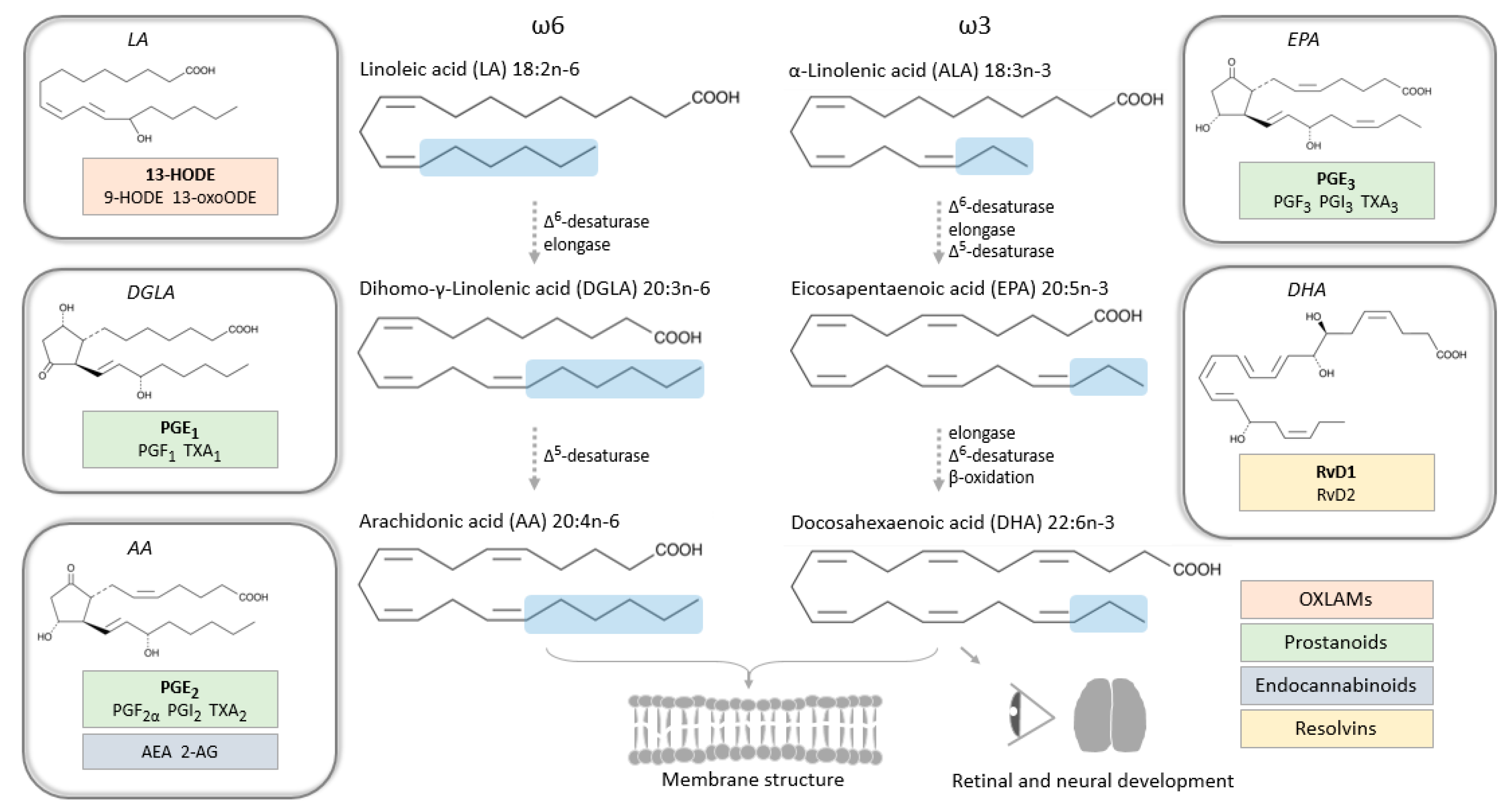
3. Polyunsaturated Fatty Acids and Bioactive Lipid Mediators in Pregnancy
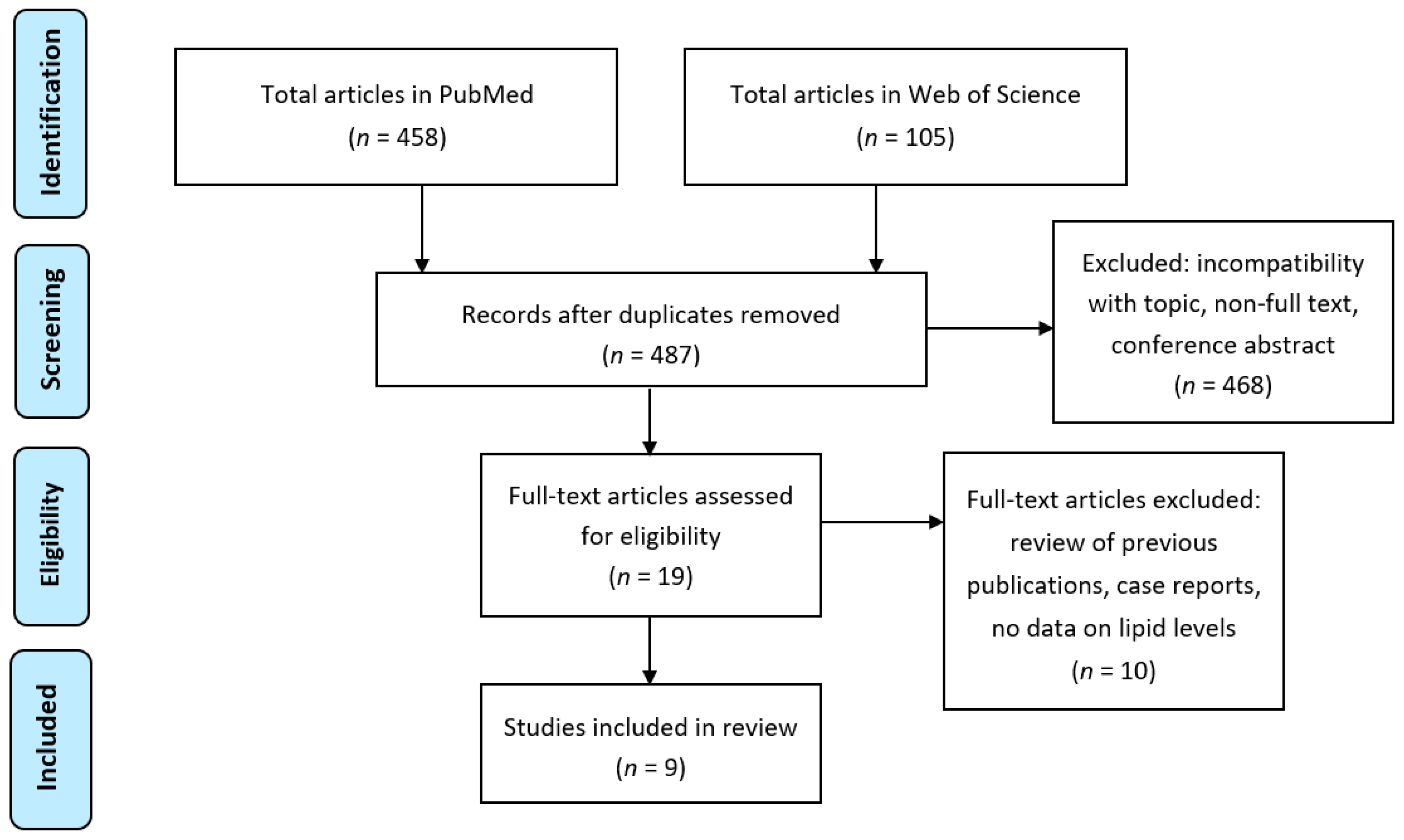
4. Results from Literature Search
5. Discussion
6. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Bloomfield, F.H.; Spiroski, A.M.; Harding, J.E. Fetal growth factors and fetal nutrition. Semin. Fetal Neonatal Med. 2013, 18, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Duttaroy, A.K.; Basak, S. Maternal dietary fatty acids and their roles in human placental development. Prostaglandins Leukot. Essent. Fatty Acids 2020, 155, 102080. [Google Scholar] [CrossRef] [PubMed]
- Haggarty, P. Fatty acid supply to the human fetus. Annu. Rev. Nutr. 2010, 30, 237–255. [Google Scholar] [CrossRef] [PubMed]
- Lager, S.; Powell, T.L. Regulation of nutrient transport across the placenta. J. Pregnancy 2012, 2012, 179827. [Google Scholar] [CrossRef] [PubMed]
- Novakovic, B.; Saffery, R. The ever growing complexity of placental epigenetics—Role in adverse pregnancy outcomes and fetal programming. Placenta 2012, 33, 959–970. [Google Scholar] [CrossRef] [PubMed]
- Gauster, M.; Hiden, U.; Blaschitz, A.; Frank, S.; Lang, U.; Alvino, G.; Cetin, I.; Desoye, G.; Wadsack, C. Dysregulation of placental endothelial lipase and lipoprotein lipase in intrauterine growth restricted pregnancies. J. Clin. Endocrinol. Metab. 2007, 92, 2256–2263. [Google Scholar] [CrossRef]
- Burton, G.J.; Jauniaux, E. Oxidative stress. Best. Pract. Res. Clin. Obstet. Gynaecol. 2011, 25, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Mackay, V.A.; Huda, S.S.; Stewart, F.M.; Tham, K.; McKenna, L.A.; Martin, I.; Jordan, F.; Brown, E.A.; Hodson, L.; Greer, I.A.; et al. Preeclampsia is associated with compromised maternal synthesis of long-chain polyunsaturated fatty acids, leading to offspring deficiency. Hypertension 2012, 60, 1078–1085. [Google Scholar] [CrossRef]
- Wadhwani, N.; Patil, V.; Joshi, S. Maternal long chain polyunsaturated fatty acid status and pregnancy complications. Prostaglandins Leukot. Essent. Fatty Acids 2018, 136, 143–152. [Google Scholar] [CrossRef]
- Fejzo, M.S.; Trovik, J.; Grooten, I.J.; Sridharan, K.; Roseboom, T.J.; Vikanes, Å.; Painter, R.C.; Mullin, P.M. Nausea and vomiting of pregnancy and hyperemesis gravidarum. Nat. Rev. Dis. Primers 2019, 5, 62. [Google Scholar] [CrossRef]
- Haugen, M.; Vikanes, A.; Brantsaeter, A.L.; Meltzer, H.M.; Grjibovski, A.M.; Magnus, P. Diet before pregnancy and the risk of hyperemesis gravidarum. Br. J. Nutr. 2011, 106, 596–602. [Google Scholar] [CrossRef]
- Fiaschi, L.; Nelson-Piercy, C.; Gibson, J.; Szatkowski, L.; Tata, L.J. Adverse maternal and birth outcomes in women admitted to hospital for hyperemesis gravidarum: A population- based cohort study. Paediatr. Perinat. Epidemiol. 2018, 32, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Dodds, L.; Fell, D.B.; Joseph, K.S.; Allen, V.M.; Butler, B. Outcomes of pregnancies complicated by hyperemesis gravidarum. Obstet. Gynecol. 2006, 107, 285–292. [Google Scholar] [CrossRef]
- Fejzo, M.; Kam, A.; Laguna, A.; MacGibbon, K.; Mullin, P. Analysis of neurodevelopmental delay in children exposed in utero to hyperemesis gravidarum reveals increased reporting of autism spectrum disorder. Reprod. Toxicol. 2019, 84, 59–64. [Google Scholar] [CrossRef]
- Getahun, D.; Fassett, M.J.; Jacobsen, S.J.; Xiang, A.H.; Takhar, H.S.; Wing, D.A.; Peltier, M.R. Autism spectrum disorders in children exposed in utero to hyperemesis gravidarum. Am. J. Perinatol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Rolls, E.T.; Du, X.; Du, J.; Yang, D.; Li, J.; Li, F.; Cheng, W.; Feng, J. Severe nausea and vomiting in pregnancy: Psychiatric and cognitive problems and brain structure in children. BMC Med. 2020, 18, 228. [Google Scholar] [CrossRef]
- Mullin, P.M.; Ching, C.; Schoenberg, F.; MacGibbon, K.; Romero, R.; Goodwin, T.M.; Fejzo, M.S. Risk factors, treatments, and outcomes associated with prolonged hyperemesis gravidarum. J. Matern. Fetal Neonatal Med. 2012, 25, 632–636. [Google Scholar] [CrossRef]
- Christodoulou-Smith, J.; Gold, J.I.; Romero, R.; Goodwin, T.M.; MacGibbon, K.W.; Mullin, P.M.; Fejzo, M.S. Posttraumatic stress symptoms following pregnancy complicated by hyperemesis gravidarum. J. Matern. Fetal Neonatal Med. 2011, 24, 1307–1311. [Google Scholar] [CrossRef]
- Fejzo, M.S.; MacGibbon, K.W.; Mullin, P.M. Why are women still dying from nausea and vomiting of pregnancy? Gynecol. Obstet. Case Rep. 2016, 2. [Google Scholar] [CrossRef]
- Gazmararian, J.A.; Petersen, R.; Jamieson, D.J.; Schild, L.; Adams, M.M.; Deshpande, A.D.; Franks, A.L. Hospitalizations during pregnancy among managed care enrollees. Obstet. Gynecol. 2002, 100, 94–100. [Google Scholar] [CrossRef]
- London, V.; Grube, S.; Sherer, D.M.; Abulafia, O. Hyperemesis gravidarum: A review of recent literature. Pharmacology 2017, 100, 161–171. [Google Scholar] [CrossRef]
- Källén, B. Hyperemesis during pregnancy and delivery outcome: A registry study. Eur. J. Obstet. Gynecol. Reprod. Biol. 1987, 26, 291–302. [Google Scholar] [CrossRef]
- Zhang, J.; Cai, W.W. Severe vomiting during pregnancy: Antenatal correlates and fetal outcomes. Epidemiology 1991, 2, 454–457. [Google Scholar] [CrossRef]
- Bustos, M.; Venkataramanan, R.; Caritis, S. Nausea and vomiting of pregnancy—What’s new? Auton. Neurosci. 2017, 202, 62–72. [Google Scholar] [CrossRef]
- Fejzo, M.S.; Sazonova, O.V.; Sathirapongsasuti, J.F.; Hallgrímsdóttir, I.B.; Vacic, V.; MacGibbon, K.W.; Schoenberg, F.P.; Mancuso, N.; Slamon, D.J.; Mullin, P.M. Placenta and appetite genes GDF15 and IGFBP7 are associated with hyperemesis gravidarum. Nat. Commun. 2018, 9, 1178. [Google Scholar] [CrossRef]
- Moore, A.G.; Brown, D.A.; Fairlie, W.D.; Bauskin, A.R.; Brown, P.K.; Munier, M.L.; Russell, P.K.; Salamonsen, L.A.; Wallace, E.M.; Breit, S.N. The transforming growth factor-ss superfamily cytokine macrophage inhibitory cytokine-1 is present in high concentrations in the serum of pregnant women. J. Clin. Endocrinol. Metab. 2000, 85, 4781–4788. [Google Scholar] [CrossRef]
- Fejzo, M.S.; Fasching, P.A.; Schneider, M.O.; Schwitulla, J.; Beckmann, M.W.; Schwenke, E.; MacGibbon, K.W.; Mullin, P.M. Analysis of GDF15 and IGFBP7 in hyperemesis gravidarum support causality. Geburtshilfe Frauenheilkd. 2019, 79, 382–388. [Google Scholar] [CrossRef]
- Petry, C.J.; Ong, K.K.; Burling, K.A.; Barker, P.; Goodburn, S.F.; Perry, J.R.B.; Acerini, C.L.; Hughes, I.A.; Painter, R.C.; Afink, G.B.; et al. Associations of vomiting and antiemetic use in pregnancy with levels of circulating GDF15 early in the second trimester: A nested case-control study. Wellcome Open Res. 2018, 21, 123. [Google Scholar] [CrossRef]
- Borner, T.; Shaulson, E.D.; Ghidewon, M.Y.; Barnett, A.B.; Horn, C.C.; Doyle, R.P.; Grill, H.J.; Hayes, M.R.; De Jonghe, B.C. GDF15 Induces Anorexia through Nausea and Emesis. Cell Metab. 2020, 31, 351–362.e5. [Google Scholar] [CrossRef]
- Chung, H.K.; Ryu, D.; Kim, K.S.; Chang, J.Y.; Kim, Y.K.; Yi, H.S.; Kang, S.G.; Choi, M.J.; Lee, S.E.; Jung, S.B.; et al. Growth differentiation factor 15 is a myomitokine governing systemic energy homeostasis. J. Cell Biol. 2017, 216, 149–165. [Google Scholar] [CrossRef] [PubMed]
- Chrysovergis, K.; Wang, X.; Kosak, J.; Lee, S.H.; Kim, J.S.; Foley, J.F.; Travlos, G.; Singh, S.; Baek, S.J.; Eling, T.E. NAG-1/GDF-15 prevents obesity by increasing thermogenesis, lipolysis and oxidative metabolism. Int. J. Obes. 2014, 38, 1555–1564. [Google Scholar] [CrossRef]
- Zhang, M.; Sun, W.; Qian, J.; Tang, Y. Fasting exacerbates hepatic growth differentiation factor 15 to promote fatty acid β-oxidation and ketogenesis via activating XBP1 signaling in liver. Redox Biol. 2018, 16, 87–96. [Google Scholar] [CrossRef]
- Ackermann, K.; Bonaterra, G.A.; Kinscherf, R.; Schwarz, A. Growth differentiation factor-15 regulates oxLDL-induced lipid homeostasis and autophagy in human macrophages. Atherosclerosis 2019, 281, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Alvarez-Guaita, A.; Melvin, A.; Rimmington, D.; Dattilo, A.; Miedzybrodzka, E.L.; Cimino, I.; Maurin, A.C.; Roberts, G.P.; Meek, C.L.; et al. GDF15 provides an endocrine signal of nutritional stress in mice and humans. Cell Metab. 2019, 29, 707–718.e8. [Google Scholar] [CrossRef]
- Kim, K.H.; Kim, S.H.; Jo, Y.S.; Lee, Y.H.; Lee, M.S. Growth differentiation factor 15 ameliorates nonalcoholic steatohepatitis and related metabolic disorders in mice. Sci. Rep. 2018, 8, 6789. [Google Scholar] [CrossRef]
- Dunlop, M.; Court, J.M. Lipogenesis in developing human adipose tissue. Early Hum. Dev. 1978, 2, 123–130. [Google Scholar] [CrossRef]
- Uauy, R.; Mena, P.; Rojas, C. Essential fatty acids in early life: Structural and functional role. Proc. Nutr. Soc. 2000, 59, 3–15. [Google Scholar] [CrossRef] [PubMed]
- Haggarty, P. Effect of placental function on fatty acid requirements during pregnancy. Eur. J. Clin. Nutr. 2004, 58, 1559–1570. [Google Scholar] [CrossRef]
- Brett, K.E.; Ferraro, Z.M.; Yockell-Lelievre, J.; Gruslin, A.; Adamo, K.B. Maternal-fetal nutrient transport in pregnancy pathologies: The role of the placenta. Int. J. Mol. Sci. 2014, 15, 16153–16185. [Google Scholar] [CrossRef] [PubMed]
- Chiurchiù, V.; Leuti, A.; Maccarrone, M. Bioactive lipids and chronic inflammation: Managing the fire within. Front. Immunol. 2018, 9, 38. [Google Scholar] [CrossRef]
- Dennis, E.A.; Norris, P.C. Eicosanoid storm in infection and inflammation. Nat. Rev. Immunol. 2015, 15, 511–523. [Google Scholar] [CrossRef]
- Morisseau, C.; Hammock, B.D. Impact of soluble epoxide hydrolase and epoxyeicosanoids on human health. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 37–58. [Google Scholar] [CrossRef]
- Gabbs, M.; Leng, S.; Devassy, J.G.; Monirujjaman, M.; Aukema, H.M. Advances in our understanding of oxylipins derived from dietary PUFAs. Adv. Nutr. 2015, 6, 513–540. [Google Scholar] [CrossRef]
- Hennebelle, M.; Morgan, R.K.; Sethi, S.; Zhang, Z.; Chen, H.; Grodzki, A.C.; Lein, P.J.; Taha, A.Y. Linoleic acid-derived metabolites constitute the majority of oxylipins in the rat pup brain and stimulate axonal growth in primary rat cortical neuron-glia co-cultures in a sex-dependent manner. J. Neurochem. 2020, 152, 195–207. [Google Scholar] [CrossRef]
- Taha, A.Y. Linoleic acid–good or bad for the brain? NPJ Sci. Food 2020, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, H.; Lee, E.; Kim, Y.; Ha, E.H.; Chang, N. Association between maternal intake of n-6 to n-3 fatty acid ratio during pregnancy and infant neurodevelopment at 6 months of age: Results of the MOCEH cohort study. Nutr. J. 2017, 16, 23. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Thomas, J.; Fairclough, A.; Kavanagh, J.; Kelly, A.J. Vaginal prostaglandin (PGE2 and PGF2a) for induction of labour at term. Cochrane Database Syst. Rev. 2014, 2014, CD003101. [Google Scholar] [CrossRef]
- Karim, S.M.M.; Filshie, G.M. Therapeutic abortion using prostaglandin F2a. Lancet 1970, 1, 157. [Google Scholar] [CrossRef]
- Karim, S.M.; Filshie, G.M. Use of prostaglandin E2 for therapeutic abortion. Br. Med. J. 1970, 3, 198–200. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Prata, N.; Weidert, K. Efficacy of misoprostol for the treatment of postpartum hemorrhage: Current knowledge and implications for health care planning. J. Women’s Health 2016, 29, 341–349. [Google Scholar] [CrossRef]
- Antonucci, R.; Zaffanello, M.; Puxeddu, E.; Porcella, A.; Cuzzolin, L.; Dolores Pilloni, M.; Fanos, V. Use of non-steroidal anti-inflammatory drugs in pregnancy: Impact on the fetus and newborn. Curr. Drug Metab. 2012, 13, 474–490. [Google Scholar] [CrossRef]
- Aung, M.T.; Yu, Y.; Ferguson, K.K.; Cantonwine, D.E.; Zeng, L.; McElrath, T.F.; Pennathur, S.; Mukherjee, B.; Meeker, J.D. Prediction and associations of preterm birth and its subtypes with eicosanoid enzymatic pathways and inflammatory markers. Sci. Rep. 2019, 9, 17049. [Google Scholar] [CrossRef] [PubMed]
- Gouveia-Figueira, S.; Martens, D.S.; Nawrot, T.S.; Nording, M.L. Cord blood eicosanoid signatures and newborn gestational age. Prostaglandins Other Lipid Mediat. 2017, 133, 123–127. [Google Scholar] [CrossRef]
- Welch, B.M.; Keil, A.P.; van‘t Erve, T.J.; Deterding, L.J.; Williams, J.G.; Lih, F.B.; Cantonwine, D.E.; McElrath, T.F.; Ferguson, K.K. Longitudinal profiles of plasma eicosanoids during pregnancy and size for gestational age at delivery: A nested case-control study. PLoS Med. 2020, 17, e1003271. [Google Scholar] [CrossRef]
- Basil, M.C.; Levy, B.D. Specialized pro-resolving mediators: Endogenous regulators of infection and inflammation. Nat. Rev. Immunol. 2016, 16, 51–67. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Krishnamoorthy, S.; Recchiuti, A.; Chiang, N. Novel anti-inflammatory–pro-resolving mediators and their receptors. Curr. Top. Med. Chem. 2011, 11, 629–647. [Google Scholar] [CrossRef]
- Ulu, A.; Sahoo, P.K.; Yuil-Valdes, A.G.; Mukherjee, M.; Van Ormer, M.; Muthuraj, P.G.; Thompson, M.; Anderson Berry, A.; Hanson, C.K.; Natarajan, S.K.; et al. Omega-3 fatty acid-derived resolvin D2 regulates human placental vascular smooth muscle and extravillous trophoblast activities. Int. J. Mol. Sci. 2019, 20, 4402. [Google Scholar] [CrossRef]
- Nordgren, T.M.; Anderson Berry, A.; Van Ormer, M.; Zoucha, S.; Elliott, E.; Johnson, R.; McGinn, E.; Cave, C.; Rilett, K.; Weishaar, K.; et al. Omega-3 fatty acid supplementation, pro-resolving mediators, and clinical outcomes in maternal-infant pairs. Nutrients 2019, 11, 98. [Google Scholar] [CrossRef]
- Maccarrone, M.; Valensise, H.; Bari, M.; Lazzarin, N.; Romanini, C.; Finazzi-Agrò, A. Relation between decreased anandamide hydrolase concentrations in human lymphocytes and miscarriage. Lancet 2000, 355, 1326–1329. [Google Scholar] [CrossRef]
- Maccarrone, M.; Bisogno, T.; Valensise, H.; Lazzarin, N.; Fezza, F.; Manna, C.; Di Marzo, V.; Finazzi-Agro, A. Low fatty acid amide hydrolase and high anandamide levels are associated with failure to achieve an ongoing pregnancy after IVF and embryo transfer. Mol. Hum. Reprod. 2002, 8, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Habayeb, O.M.; Taylor, A.H.; Evans, M.D.; Cooke, M.S.; Taylor, D.J.; Bell, S.C.; Konje, J.C. Plasma levels of the endocannabinoid anandamide in women—A potential role in pregnancy maintenance and labor? J. Clin. Endocrinol. Metab. 2004, 89, 5482–5487. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA group (2009). Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Aksoy, H.; Aksoy, A.N.; Ozkan, A.; Polat, H. Serum lipid profile, oxidative status, and paraoxonase 1 activity in hyperemesis gravidarum. J. Clin. Lab. Anal. 2009, 23, 105–109. [Google Scholar] [CrossRef]
- Asakura, H.; Watanabe, S.; Sekiguchi, A.; Power, G.G.; Araki, T. Severity of hyperemesis gravidarum correlates with serum levels of reverse T3. Arch. Gynecol. Obstet. 2000, 264, 57–62. [Google Scholar] [CrossRef]
- Asakura, H.; Nakai, A.; Araki, T. Correlations between interscapular deep temperature and plasma free fatty acid levels in pregnant women with hyperemesis gravidarum. Arch. Gynecol. Obstet. 2003, 268, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Gadsby, R.; Barnie-Adshead, A.; Grammatoppoulos, D.; Gadsby, P. Nausea and vomiting in pregnancy: An association between symptoms and maternal prostaglandin E2. Gynecol. Obstet. Investig. 2000, 50, 149–152. [Google Scholar] [CrossRef]
- Gebeh, A.K.; Willets, J.M.; Marczylo, T.H.; Konje, J.C. Plasma anandamide and related n-acylethanolamide levels are not elevated in pregnancies complicated by hyperemesis gravidarum. J. Matern. Fetal Neonatal Med. 2014, 27, 954–959. [Google Scholar] [CrossRef] [PubMed]
- Järnfelt-Samsioe, A.; Eriksson, B.; Mattsson, L.A.; Samsioe, G. Serum lipids and lipoproteins in pregnancies associated with emesis gravidarum. Gynecol. Endocrinol. 1987, 1, 51–60. [Google Scholar] [CrossRef]
- Ulubay, M.; Ozturk, M.; Ozturk, O.; Keskin, U.; Fidan, U.; Sertoglu, E.; Aydin, H.; Yilmaz, A.; Yenen, M.C. Plasma free fatty acids in hyperemesis gravidarum pregnancy. Open Med. 2017, 12, 70–75. [Google Scholar] [CrossRef]
- Ustün, Y.; Engin-Ustün, Y.; Dökmeci, F.; Söylemez, F. Serum concentrations of lipids and apolipoproteins in normal and hyperemetic pregnancies. J. Matern. Fetal Neonatal Med. 2004, 15, 287–290. [Google Scholar] [CrossRef]
- Watanabe, S.; Asakura, H.; Power, G.G.; Araki, T. Alterations of thermoregulation in women with hyperemesis gravidarum. Arch. Gynecol. Obstet. 2003, 267, 221–226. [Google Scholar] [CrossRef]
- Grennon, J.A.; Brech, W.J.; Gordon, E.S. Effect of a short period of cold exposure on plasma FFA level in lean and obese humans. Metabolism 1967, 16, 503–506. [Google Scholar] [CrossRef]
- Astrup, A.; Bulow, J.; Christiansen, N.J.; Madsen, J. Epinephrine-induced thermogenesis in man: No role for interscapular adipose tissue. Clin. Sci. 1984, 6, 179–186. [Google Scholar] [CrossRef]
- Gadsby, R.; Barnie-Adshead, A.M.; Jagger, C.A. prospective study of nausea and vomiting during pregnancy. Br. J. Gen. Pract. 1993, 43, 245–248. [Google Scholar]
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega-3 fatty acid addition during pregnancy. Cochrane Database Syst. Rev. 2018, 11, CD003402. [Google Scholar] [CrossRef] [PubMed]
- Signorello, L.; Harlow, B.; Wang, S.; Erick, M. Saturated fat intake and the risk of severe morning sickness. Epidemiology 1998, 9, 636–640. [Google Scholar] [CrossRef]
- Nording, M.L.; Yang, J.; Georgi, K.; Karbowski, C.H.; German, J.B.; Weiss, R.H.; Hogg, R.J.; Trygg, J.; Hammock, B.D.; Zivkovic, A.M. Individual variation in lipidomic profiles of healthy subjects in response to omega-3 Fatty acids. PLoS ONE 2013, 8, e76575. [Google Scholar] [CrossRef]
- Ramsden, C.E.; Makrides, M.; Yuan, Z.X.; Horowitz, M.S.; Zamora, D.; Yelland, L.N.; Best, K.; Jensen, J.; Taha, A.Y.; Gibson, R.A. Plasma oxylipins and unesterified precursor fatty acids are altered by DHA supplementation in pregnancy: Can they help predict risk of preterm birth? Prostaglandins Leukot. Essent. Fatty Acids 2020, 153, 102041. [Google Scholar] [CrossRef] [PubMed]
- Keelan, J.A.; Mas, E.; D’Vaz, N.; Dunstan, J.A.; Li, S.; Barden, A.E.; Mark, P.J.; Waddell, B.J.; Prescott, S.L.; Mori, T.A. Effects of maternal n-3 fatty acid supplementation on placental cytokines, pro-resolving lipid mediators and their precursors. Reproduction 2015, 149, 171–178. [Google Scholar] [CrossRef]
- Mozurkewich, E.L.; Greenwood, M.; Clinton, C.; Berman, D.; Romero, V.; Djuric, Z.; Qualls, C.; Gronert, K. Pathway markers for pro-resolving lipid mediators in maternal and umbilical cord blood: A secondary analysis of the mothers, omega-3, and mental health study. Front. Pharmacol. 2016, 7, 274. [Google Scholar] [CrossRef]
- Baek, S.J.; Kim, K.S.; Nixon, J.B.; Wilson, L.C.; Eling, T.E. Cyclooxygenase inhibitors regulate the expression of a TGF-beta superfamily member that has proapoptotic and antitumorigenic activities. Mol. Pharmacol. 2001, 59, 901–908. [Google Scholar] [CrossRef]
- Baek, S.J.; Wilson, L.C.; Lee, C.H.; Eling, T.E. Dual function of nonsteroidal anti-inflammatory drugs (NSAIDs): Inhibition of cyclooxygenase and induction of NSAID-activated gene. J. Pharmacol. Exp. Ther. 2002, 301, 1126–1131. [Google Scholar] [CrossRef]
- Iguchi, G.; Chrysovergis, K.; Lee, S.H.; Baek, S.J.; Langenbach, R.; Eling, T.E. A reciprocal relationship exists between non-steroidal anti-inflammatory drug-activated gene-1 (NAG-1) and cyclooxygenase-2. Cancer Lett. 2009, 282, 152–158. [Google Scholar] [CrossRef]
- Koren, G.; Madjunkova, S.; Maltepe, C. The protective effects of nausea and vomiting of pregnancy against adverse fetal outcome—A systematic review. Reprod. Toxicol. 2014, 47, 77–80. [Google Scholar] [CrossRef]
- Tran, T.; Yang, J.; Gardner, J.; Xiong, Y. GDF15 deficiency promotes high fat diet-induced obesity in mice. PLoS ONE 2018, 13, e0201584. [Google Scholar] [CrossRef]
- Zivkovic, A.M.; Wiest, M.M.; Nguyen, U.T.; Davis, R.; Watkins, S.M.; German, J.B. Effects of sample handling and storage on quantitative lipid analysis in human serum. Metabolomics 2009, 5, 507–516. [Google Scholar] [CrossRef]
- Dettmer, K.; Aronov, P.A.; Hammock, B.D. Mass spectrometry-based metabolomics. Mass Spectrom. Rev. 2007, 26, 51–78. [Google Scholar] [CrossRef]
- Martens, D.S.; Gouveia, S.; Madhloum, N.; Janssen, B.G.; Plusquin, M.; Vanpoucke, C.; Lefebvre, W.; Forsberg, B.; Nording, M.; Nawrot, T.S. Neonatal cord blood oxylipins and exposure to particulate matter in the early-life environment: An Environage Birth Cohort Study. Environ. Health Perspect. 2017, 125, 691–698. [Google Scholar] [CrossRef]
| Author, Year, Country | Study Participants | Analyte, Type of Lipid, PUFA or PUFA Metabolite | Analytical Method | Outcome | |
|---|---|---|---|---|---|
| Cases | Controls | ||||
| Aksoy et al., 2009, Turkey [64] | HG 1 patients (n = 36) | Healthy pregnant women (n = 36) | Serum TG, cholesterol, HDL, LDL, apo- A1 and -B | Enzymatic | TG, LDL-cholesterol, and apo B levels were not different; HDL-cholesterol and apo A1 were lower in HG vs. healthy pregnant controls. |
| Asakura et al., 2000, Japan [65] | HG patients (n = 80) and NVP (n = 30) | Healthy pregnant women (n = 30) | Plasma or serum free fatty acids | No info | Elevated free fatty acids in HG patients vs. healthy pregnant controls. |
| Asakura et al., 2003, Japan [66] | HG patients (n = 24) | Healthy pregnant women (n = 30) | Serum free fatty acids | Enzymatic | Elevated free fatty acids in HG patients vs. healthy pregnant controls and decreased free fatty acid levels in correlation with symptomatic improvement. |
| Gadsby et al., 2000, U.K. [67] | NVP symptomatic (n = 18) | NVP symptom-free (n = 18) | Serum PGE2 | Radio-immunoassay | Increased maternal serum levels of PGE2 during episodes of nausea. |
| Gebeh et al., 2014, U.K. [68] | HG patients (n = 15) | Healthy pregnant women (n = 30) | Plasma AEA, OEA, PEA | LC-MS/MS | No differences in levels between the groups and no correlation with dehydration markers. |
| Järnfelt-Samsioe et al., 1987, Sweden [69] | NVP in early (n = 60) and late (n = 53) pregnancy | Healthy pregnant women in early (n = 38) and late (n = 34) pregnancy, non-pregnant controls (n = 22) | Serum TG, cholesterol, phospholipids, HDL, LDL, VLDL | Enzymatic | Early pregnancy: serum levels of TC, VLDL, and LDL were higher in NVP vs. non-pregnant controls; late pregnancy: higher serum levels of TC and TG in previously NVP patients vs. pregnant controls; HDL lipid composition was differential in both early and late pregnancy vs. controls. |
| Ulubay et al., 2017, Turkey [70] | HG patients (n = 26) | Healthy pregnant women (n = 26) | Plasma free fatty acids | GC | No difference in levels of DHA, EPA and AA. Higher behenic acid and lower arachidic acid in the HG vs. control group. |
| Üstün et al., 2004, Turkey [71] | Women with HG (n = 35) | Healthy pregnant women (n = 39) | Serum TG, cholesterol, HDL, LDL, apo-A and -B | Enzymatic | Lower levels of HDL cholesterol, LDL cholesterol and total cholesterol, apo-A and apo-B in the HG vs. control group. |
| Watanabe et al., 2003, Japan [72] | HG patients (n = 13) before and after challenge, fasting and non-fasting | Pregnant women with other obstetrical disease (n = 13) | Serum free fatty acids | Enzymatic | Free fatty acids increased in response to cold stimulus in fasting HG subjects, but not in the other groups. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lindberg, R.; Lindqvist, M.; Trupp, M.; Vinnars, M.-T.; Nording, M.L. Polyunsaturated Fatty Acids and Their Metabolites in Hyperemesis Gravidarum. Nutrients 2020, 12, 3384. https://doi.org/10.3390/nu12113384
Lindberg R, Lindqvist M, Trupp M, Vinnars M-T, Nording ML. Polyunsaturated Fatty Acids and Their Metabolites in Hyperemesis Gravidarum. Nutrients. 2020; 12(11):3384. https://doi.org/10.3390/nu12113384
Chicago/Turabian StyleLindberg, Richard, Maria Lindqvist, Miles Trupp, Marie-Therese Vinnars, and Malin L. Nording. 2020. "Polyunsaturated Fatty Acids and Their Metabolites in Hyperemesis Gravidarum" Nutrients 12, no. 11: 3384. https://doi.org/10.3390/nu12113384
APA StyleLindberg, R., Lindqvist, M., Trupp, M., Vinnars, M.-T., & Nording, M. L. (2020). Polyunsaturated Fatty Acids and Their Metabolites in Hyperemesis Gravidarum. Nutrients, 12(11), 3384. https://doi.org/10.3390/nu12113384





